Sex Disparities in Long-Term Mortality after Paclitaxel Exposure in Patients with Peripheral Artery Disease: A Nationwide Claims-Based Cohort Study
Abstract
:1. Introduction
2. Materials and Methods
2.1. BARMER Cohort
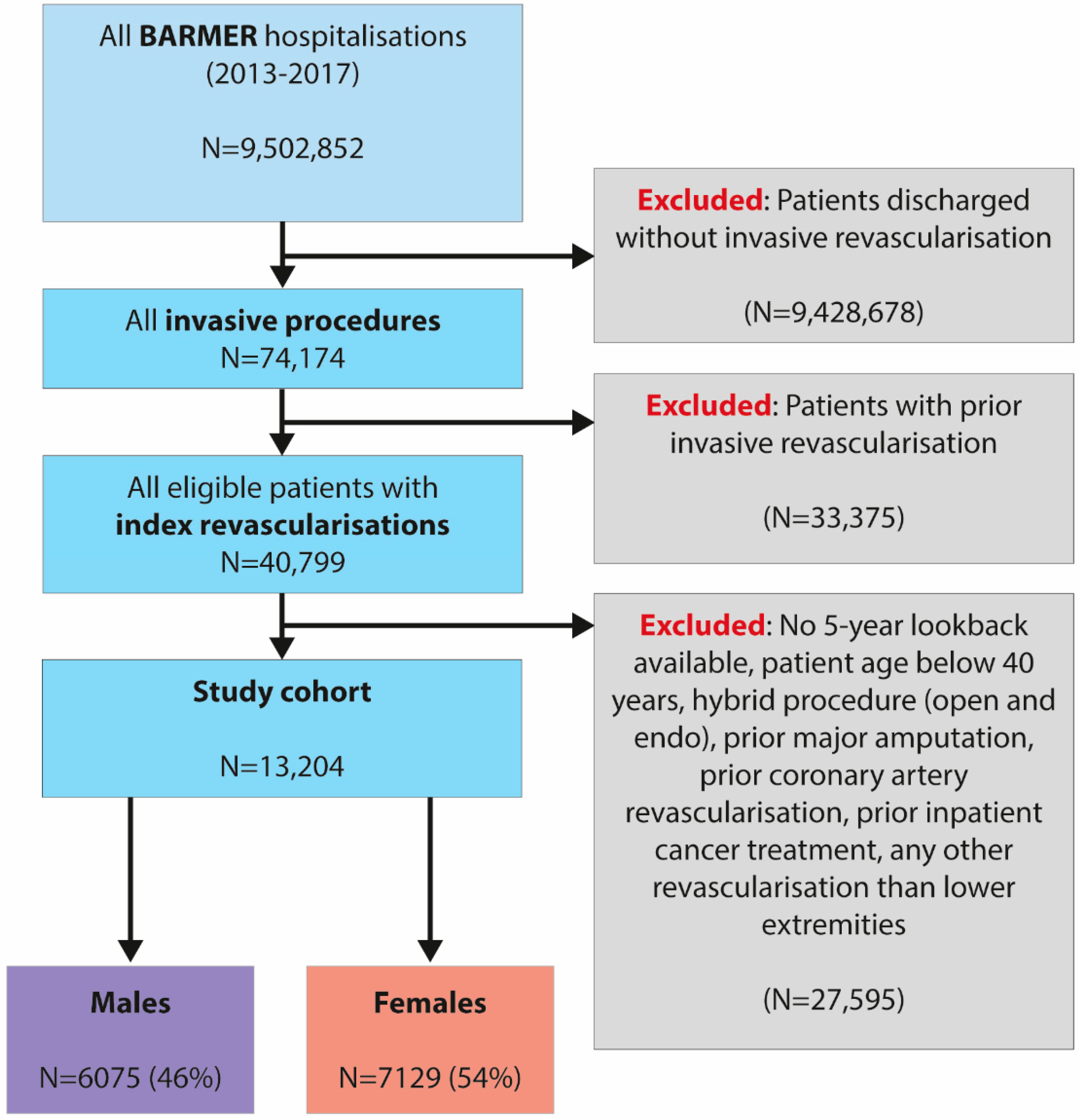
2.2. Study Population
2.3. Baseline Characteristics
2.4. Statistical Analysis
3. Results
3.1. Baseline Characteristics by Sex
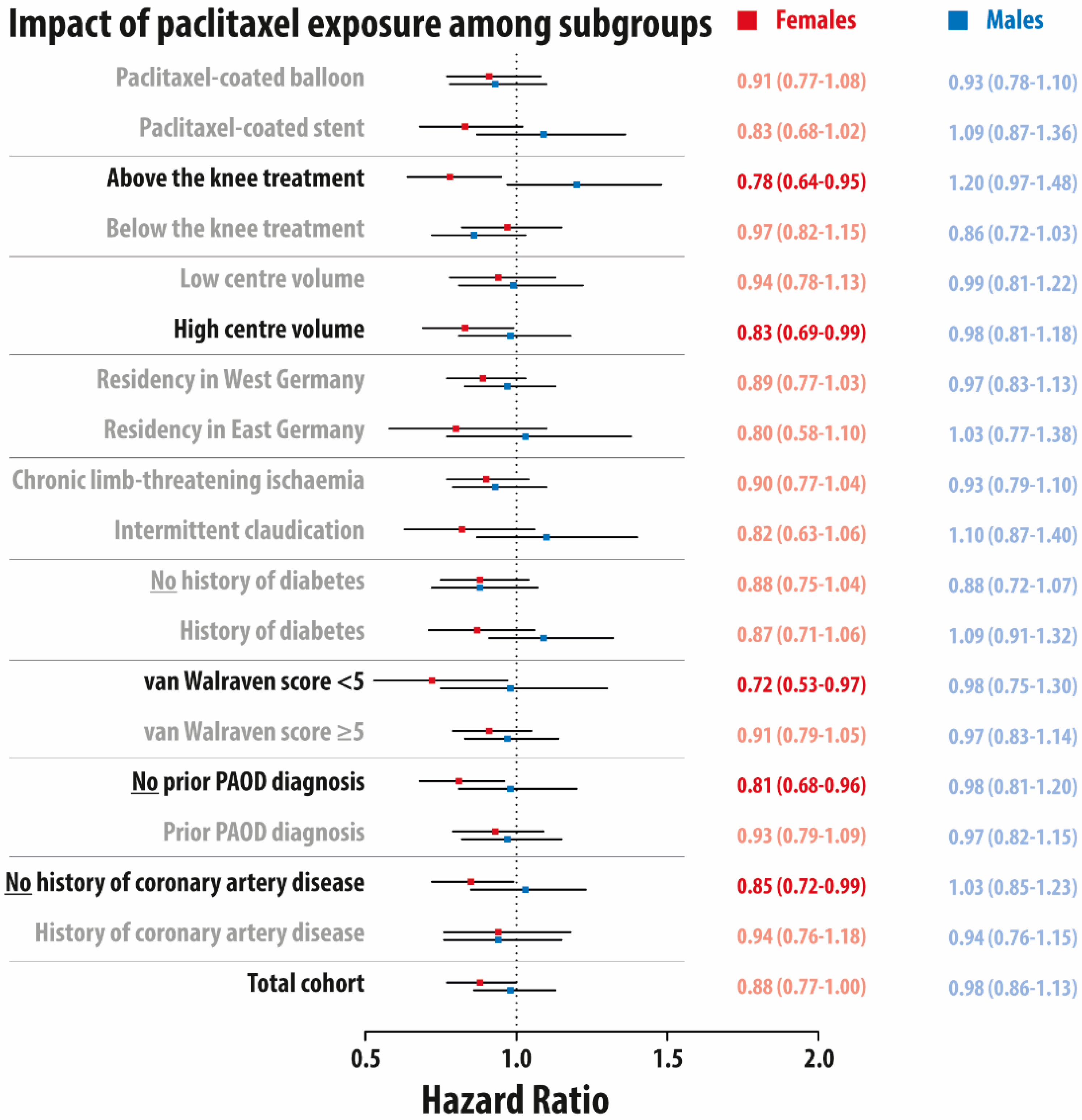
3.2. Impact of Paclitaxel Exposure on 5-Year Mortality among Subgroups
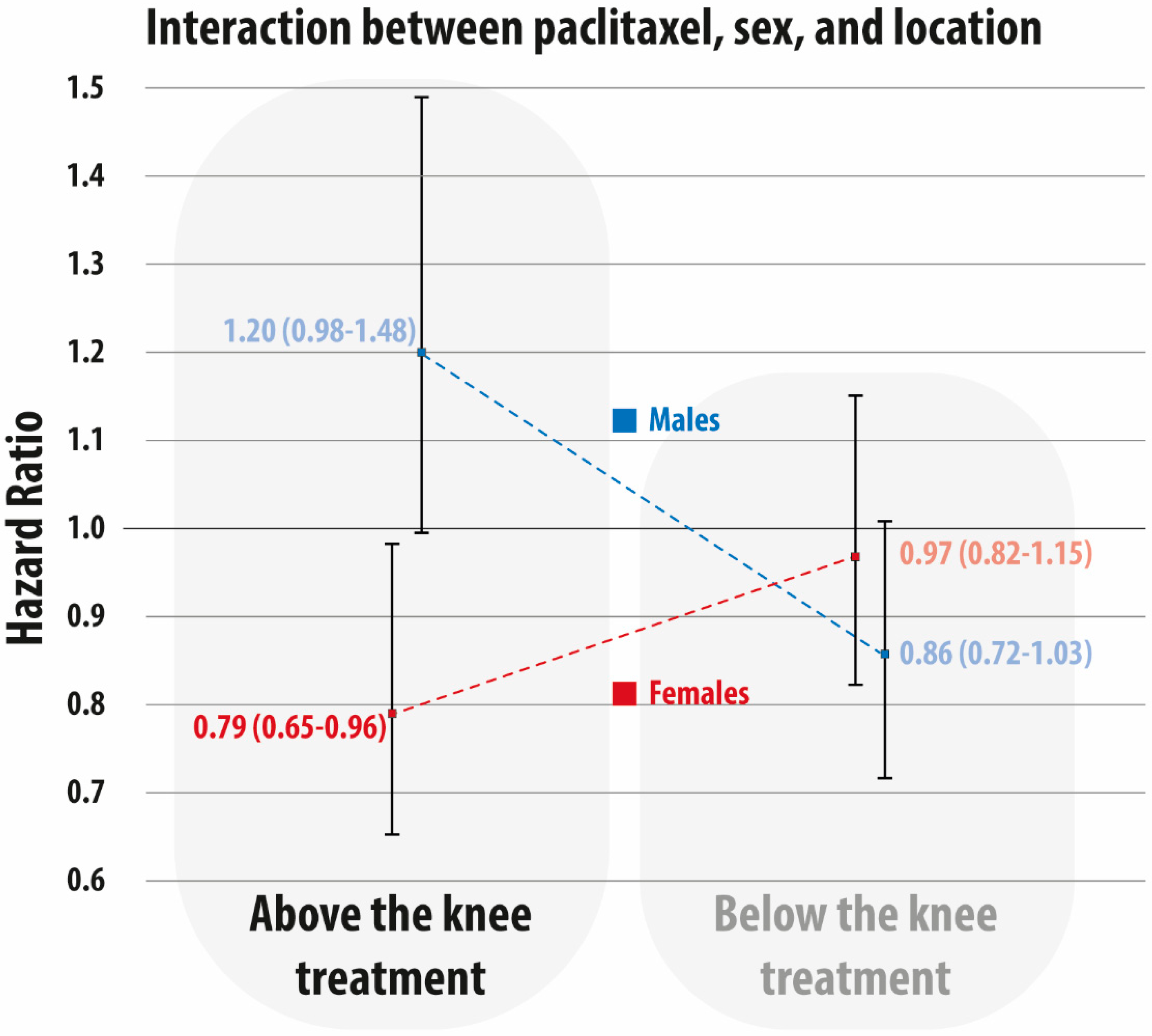
3.3. Interaction of Treatment Level, Sex, and Paclitaxel Exposure on 5-Year Mortality
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Katsanos, K.; Spiliopoulos, S.; Kitrou, P.; Krokidis, M.; Karnabatidis, D. Risk of Death Following Application of Paclitaxel-Coated Balloons and Stents in the Femoropopliteal Artery of the Leg: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. J. Am. Heart Assoc. 2018, 7, e011245. [Google Scholar] [CrossRef] [Green Version]
- Katsanos, K.; Spiliopoulos, S.; Kitrou, P.; Krokidis, M.; Paraskevopoulos, I.; Karnabatidis, D. Risk of Death and Amputation with Use of Paclitaxel-Coated Balloons in the Infrapopliteal Arteries for Treatment of Critical Limb Ischemia: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. J. Vasc. Interv. Radiol. 2020, 31, 202–212. [Google Scholar] [CrossRef] [PubMed]
- Rocha-Singh, K.J.; Duval, S.; Jaff, M.R.; Schneider, P.A.; Ansel, G.M.; Lyden, S.P.; Mullin, C.M.; Ioannidis, J.P.A.; Misra, S.; Tzafriri, A.R.; et al. Mortality and Paclitaxel-Coated Devices. Circulation 2020, 141, 1859–1869. [Google Scholar] [CrossRef] [PubMed]
- Secemsky, E.A.; Kundi, H.; Weinberg, I.; Jaff, M.R.; Krawisz, A.; Parikh, S.A.; Beckman, J.A.; Mustapha, J.; Rosenfield, K.; Yeh, R.W. Association of Survival with Femoropopliteal Artery Revascularization with Drug-Coated Devices. JAMA Cardiol. 2019, 4, 332–340. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Behrendt, C.A.; Sedrakyan, A.; Peters, F.; Kreutzburg, T.; Schermerhorn, M.; Bertges, D.J.; Larena-Avellaneda, A.; L’Hoest, H.; Kolbel, T.; Debus, E.S. Editor’s Choice—Long Term Survival after Femoropopliteal Artery Revascularisation with Paclitaxel Coated Devices: A Propensity Score Matched Cohort Analysis. Eur. J. Vasc. Endovasc. Surg. 2020, 59, 587–596. [Google Scholar] [CrossRef]
- Heidemann, F.; Peters, F.; Kuchenbecker, J.; Kreutzburg, T.; Sedrakyan, A.; Marschall, U.; L’Hoest, H.; Debus, E.S.; Behrendt, C.-A. Long Term Outcomes After Revascularisations Below the Knee with Paclitaxel Coated Devices: A Propensity Score Matched Cohort Analysis. Eur. J. Vasc. Endovasc. Surg. 2020, 60, 549–558. [Google Scholar] [CrossRef]
- Bertges, D.J.; Sedrakyan, A.; Sun, T.; Eslami, M.H.; Schermerhorn, M.; Goodney, P.P.; Beck, A.W.; Cronenwett, J.L.; Eldrup-Jorgensen, J. Mortality After Paclitaxel Coated Balloon Angioplasty and Stenting of Superficial Femoral and Popliteal Artery in the Vascular Quality Initiative. Circ. Cardiovasc. Interv. 2020, 13, e008528. [Google Scholar] [CrossRef] [PubMed]
- Böhme, T.; Noory, E.; Beschorner, U.; Bürgerlin, K.; Macharzina, R.; Kuhn, L.; Nührenberg, T.; Neumann, F.J.; Zeller, T. Evaluation of mortality following paclitaxel drug-coated balloon angioplasty of femoropopliteal lesions in patients with ulcerations and gangrene—A single center experience. VASA 2021, 50, 132–138. [Google Scholar] [CrossRef]
- Saratzis, A.; Lea, T.; Yap, T.; Batchelder, A.; Thomson, B.; Saha, P.; Diamantopoulos, A.; Saratzis, N.; Davies, R.; Zayed, H. Paclitaxel and Mortality Following Peripheral Angioplasty: An Adjusted and Case Matched Multicentre Analysis. Eur. J. Vasc. Endovasc. Surg. 2020, 60, 220–229. [Google Scholar] [CrossRef]
- Nordanstig, J.; James, S.; Andersson, M.; Andersson, M.; Danielsson, P.; Gillgren, P.; Delle, M.; Engström, J.; Fransson, T.; Hamoud, M.; et al. Mortality with Paclitaxel-Coated Devices in Peripheral Artery Disease. NEJM 2020, 383, 2538–2546. [Google Scholar] [CrossRef] [PubMed]
- Behrendt, C.A.; Sigvant, B.; Kuchenbecker, J.; Grima, M.J.; Schermerhorn, M.; Thomson, I.; Altreuther, M.; Setacci, C.; Svetlikov, A.; Laxdal, E.; et al. Editor’s Choice—International Variations and Gender Disparities in the Treatment of Peripheral Arterial Occlusive Disease—A report from VASCUNET and the International Consortium of Vascular Registries. Eur. J. Vasc. Endovasc. Surg. 2020, 60, 873–880. [Google Scholar] [CrossRef]
- Peters, F.; Kreutzburg, T.; Riess, H.C.; Heidemann, F.; Marschall, U.; L’Hoest, H.; Debus, E.S.; Sedrakyan, A.; Behrendt, C.A. Editor’s Choice—Optimal Pharmacological Treatment of Symptomatic Peripheral Arterial Occlusive Disease and Evidence of Female Patient Disadvantage: An Analysis of Health Insurance Claims Data. Eur. J. Vasc. Endovasc. Surg. 2020, 60, 421–429. [Google Scholar] [CrossRef] [PubMed]
- Ramkumar, N.; Suckow, B.D.; Brown, J.R.; Sedrakyan, A.; MacKenzie, T.; Stone, D.H.; Cronenwett, J.L.; Goodney, P.P. Role of Sex in Determining Treatment Type for Patients Undergoing Endovascular Lower Extremity Revascularization. J. Am. Heart Assoc. 2019, 8, e013088. [Google Scholar] [CrossRef] [PubMed]
- Ramkumar, N.; Suckow, B.D.; Brown, J.R.; Sedrakyan, A.; Cronenwett, J.L.; Goodney, P.P. Sex-Based Assessment of Patient Presentation, Lesion Characteristics, and Treatment Modalities in Patients Undergoing Peripheral Vascular Intervention. Circ. Cardiovasc. Interv. 2018, 11, e005749. [Google Scholar] [CrossRef]
- Parvar, S.L.; Thiyagarajah, A.; Nerlekar, N.; King, P.; Nicholls, S.J. A Systematic Review and Meta-analysis of Gender Differences in Long-term Mortality and Cardiovascular Events in Peripheral Artery Disease. J. Vasc. Surg. 2021, 73, 1456–1465.e7. [Google Scholar] [CrossRef] [PubMed]
- Peters, F.; Kreutzburg, T.; Kuchenbecker, J.; Marschall, U.; Remmel, M.; Dankhoff, M.; Trute, H.H.; Repgen, T.; Debus, E.S.; Behrendt, C.A. Quality of care in surgical/interventional vascular medicine: What can routinely collected data from the insurance companies achieve? Gefässchirurgie 2020, 25, 19–28. [Google Scholar]
- Czwikla, J.; Jobski, K.; Schink, T. The impact of the lookback period and definition of confirmatory events on the identification of incident cancer cases in administrative data. BMC Med. Res. Methodol. 2017, 17, 122. [Google Scholar] [CrossRef] [Green Version]
- Hoffmann, F.; Andersohn, F.; Giersiepen, K.; Scharnetzky, E.; Garbe, E. Validation of secondary data. Strengths and limitations. Bundesgesundheitsblatt 2008, 51, 1118–1126. [Google Scholar] [CrossRef]
- Ohlmeier, C.; Langner, I.; Hillebrand, K.; Schmedt, N.; Mikolajczyk, R.; Riedel, O.; Garbe, E. Mortality in the German Pharmacoepidemiological Research Database (GePaRD) compared to national data in Germany: Results from a validation study. BMC Public Health 2015, 15, 570. [Google Scholar] [CrossRef] [Green Version]
- Langner, I.; Ohlmeier, C.; Zeeb, H.; Haug, U.; Riedel, O. Individual mortality information in the German Pharmacoepidemiological Research Database (GePaRD): A validation study using a record linkage with a large cancer registry. BMJ Open 2019, 9, e028223. [Google Scholar] [CrossRef]
- Peters, F.; Kreutzburg, T.; Kuchenbecker, J.; Debus, E.; Marschall, U.; L’Hoest, H.; Behrendt, C.-A. A retrospective cohort study on the provision and outcomes of pharmacological therapy after revascularization for peripheral arterial occlusive disease: A study protocol. BMJ Surg. Interv. Health Technol. 2020, 2, e000020. [Google Scholar] [CrossRef] [Green Version]
- Quan, H.; Sundararajan, V.; Halfon, P.; Fong, A.; Burnand, B.; Luthi, J.C.; Saunders, L.D.; Beck, C.A.; Feasby, T.E.; Ghali, W.A. Coding algorithms for defining comorbidities in ICD-9-CM and ICD-10 administrative data. Med. Care 2005, 43, 1130–1139. [Google Scholar] [CrossRef]
- Elixhauser, A.; Steiner, C.; Harris, D.R.; Coffey, R.M. Comorbidity measures for use with administrative data. Med. Care 1998, 36, 8–27. [Google Scholar] [CrossRef]
- Kreutzburg, T.; Peters, F.; Riess, H.C.; Hischke, S.; Marschall, U.; Kriston, L.; L’Hoest, H.; Sedrakyan, A.; Debus, E.S.; Behrendt, C.A. Editor’s Choice—Comorbidity Patterns Among Patients with Peripheral Arterial Occlusive Disease in Germany: A Trend Analysis of Health Insurance Claims Data. Eur. J. Vasc. Endovasc. Surg. 2020, 59, 59–66. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Aboyans, V.; Ricco, J.B.; Bartelink, M.E.L.; Bjorck, M.; Brodmann, M.; Cohnert, T.; Collet, J.P.; Czerny, M.; De Carlo, M.; Debus, S.; et al. Editor’s Choice—2017 ESC Guidelines on the Diagnosis and Treatment of Peripheral Arterial Diseases, in collaboration with the European Society for Vascular Surgery (ESVS). Eur. J. Vasc. Endovasc. Surg. 2018, 55, 305–368. [Google Scholar] [CrossRef] [Green Version]
- Breiman, L. Random Forests. Mach. Learn. 2001, 45, 5–32. [Google Scholar] [CrossRef] [Green Version]
- Grambsch, P.M.; Therneau, T.M. Proportional Hazards Tests and Diagnostics Based on Weighted Residuals. Biometrika 1994, 81, 515–526. [Google Scholar] [CrossRef]
- Benchimol, E.I.; Smeeth, L.; Guttmann, A.; Harron, K.; Moher, D.; Petersen, I.; Sorensen, H.T.; von Elm, E.; Langan, S.M.; Committee, R.W. The REporting of studies Conducted using Observational Routinely-collected health Data (RECORD) statement. PLoS Med. 2015, 12, e1001885. [Google Scholar] [CrossRef] [PubMed]
- von Elm, E.; Altman, D.G.; Egger, M.; Pocock, S.J.; Gotzsche, P.C.; Vandenbroucke, J.P.; Initiative, S. The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement: Guidelines for reporting observational studies. J. Clin. Epidemiol. 2008, 61, 344–349. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schneider, P.A.; Laird, J.R.; Doros, G.; Gao, Q.; Ansel, G.; Brodmann, M.; Micari, A.; Shishehbor, M.H.; Tepe, G.; Zeller, T. Mortality Not Correlated with Paclitaxel Exposure: An Independent Patient-Level Meta-Analysis of a Drug-Coated Balloon. J. Am. Heart. Assoc. 2019, 73, 2550–2563. [Google Scholar] [CrossRef] [Green Version]
- Jelani, Q.U.; Petrov, M.; Martinez, S.C.; Holmvang, L.; Al-Shaibi, K.; Alasnag, M. Peripheral Arterial Disease in Women: An Overview of Risk Factor Profile, Clinical Features, and Outcomes. Curr. Atheroscler. Rep. 2018, 20, 40. [Google Scholar] [CrossRef] [Green Version]
- Khan, S.U.; Khan, M.Z.; Raghu Subramanian, C.; Riaz, H.; Khan, M.U.; Lone, A.N.; Khan, M.S.; Benson, E.M.; Alkhouli, M.; Blaha, M.J. Participation of Women and Older Participants in Randomized Clinical Trials of Lipid-Lowering Therapies: A Systematic Review. JAMA Netw Open 2020, 3, e205202. [Google Scholar] [CrossRef]
- Jin, X.; Chandramouli, C.; Allocco, B.; Gong, E.; Lam, C.S.P.; Yan, L.L. Women’s Participation in Cardiovascular Clinical Trials From 2010 to 2017. Circulation 2020, 141, 540–548. [Google Scholar] [CrossRef] [PubMed]
- Mayor, J.; Preventza, O.; Mills, J.L., Sr.; Montero-Baker, M.; Gilani, R.; Pallister, Z.; Chung, J. Persistent Underrepresentation of Female Patients in US Trials of Common Vascular Diseases Since 2008. J. Vasc. Surg. 2021, 73, e23. [Google Scholar] [CrossRef]
- Vyas, M.V.; Mrkobrada, M.; Donner, A.; Hackam, D.G. Underrepresentation of peripheral artery disease in modern cardiovascular trials: Systematic review and meta-analysis. Int. J. Cardiol. 2013, 168, 4875–4876. [Google Scholar] [CrossRef]
- Bonaca, M.P.; Bauersachs, R.M.; Anand, S.S.; Debus, E.S.; Nehler, M.R.; Patel, M.R.; Fanelli, F.; Capell, W.H.; Diao, L.; Jaeger, N. Rivaroxaban in Peripheral Artery Disease after Revascularization. NEJM 2020, 382, 1994–2004. [Google Scholar] [CrossRef]
- Conte, M.S.; Bradbury, A.W.; Kolh, P.; White, J.V.; Dick, F.; Fitridge, R.; Mills, J.L.; Ricco, J.B.; Suresh, K.R.; Murad, M.H. Global vascular guidelines on the management of chronic limb-threatening ischemia. J. Vasc. Surg. 2019, 69, 3S–125S.e40. [Google Scholar] [CrossRef] [Green Version]
- Behrendt, C.-A.; Peters, F.; Mani, K. The swinging pendulum of evidence—Is there a reality behind results from randomized trials and real-world data? Lessons learned from the Paclitaxel debate. Eur. J. Vasc. Endovasc. Surg. 2020, 59, 510–511. [Google Scholar] [CrossRef] [PubMed]
- De Martino, R.R.; Eldrup-Jorgensen, J.; Nolan, B.W.; Stone, D.H.; Adams, J.; Bertges, D.J.; Cronenwett, J.L.; Goodney, P.P. Perioperative management with antiplatelet and statin medication is associated with reduced mortality following vascular surgery. J. Vasc. Surg. 2014, 59, 1615–1621.e1. [Google Scholar] [CrossRef] [Green Version]
- Kokkinidis, D.G.; Arfaras-Melainis, A.; Giannopoulos, S.; Katsaros, I.; Jawaid, O.; Jonnalagadda, A.K.; Parikh, S.A.; Secemsky, E.A.; Giri, J.; et al. Statin therapy for reduction of cardiovascular and limb-related events in critical limb ischemia: A systematic review and meta-analysis. Vasc. Med. 2020, 25, 106–117. [Google Scholar] [CrossRef]
- Saratzis, A.; Paraskevopoulos, I.; Patel, S.; Donati, T.; Biasi, L.; Diamantopoulos, A.; Zayed, H.; Katsanos, K. Supervised Exercise Therapy and Revascularization for Intermittent Claudication: Network Meta-Analysis of Randomized Controlled Trials. JACC 2019, 12, 1125–1136. [Google Scholar]
- Secemsky, E.A.; Raja, A.; Shen, C.; Valsdottir, L.R.; Schermerhorn, M.; Yeh, R.W. Rationale and Design of the SAFE-PAD Study. Circ. Cardiovasc. Qual. Outcomes 2021, 14, e007040. [Google Scholar] [CrossRef] [PubMed]
- Björkman, P.; Weselius, E.-M.; Venermo, M. No Difference in Mid-term and Long-Term Mortality after Vascular Paclitaxel Exposure. Ann. Vasc. Surg. 2020, 72, 253–260. [Google Scholar] [CrossRef] [PubMed]
| N | % Females | N | % Males | SMD | |
|---|---|---|---|---|---|
| No of patients | 7129 | 100 | 6075 | 100 | |
| Paclitaxel exposure at index | 1611 | 22.6 | 1324 | 21.8 | 0.017 |
| Stent at index | 3030 | 42.5 | 2643 | 43.5 | 0.020 |
| Crural arteries involved | 2509 | 35.2 | 2254 | 37.1 | 0.038 |
| Intermittent claudication | 3949 | 55.4 | 3639 | 59.9 | 0.091 |
| Discharge year >2014 | 4434 | 62.2 | 3791 | 62.4 | 0.005 |
| High hospital volume | 3736 | 52.4 | 3177 | 52.3 | 0.001 |
| Patient residence East Germany | 1355 | 19.0 | 1361 | 22.4 | 0.086 |
| Prior outpatient PAOD visit | 3907 | 54.8 | 3663 | 60.3 | 0.112 # |
| Van Walraven score >5 | 3593 | 50.4 | 2855 | 47.0 | 0.069 |
| Coronary artery disease | 1611 | 22.6 | 2023 | 33.3 | 0.241 # |
| Dyslipidemia | 3151 | 44.2 | 3019 | 49.7 | 0.110 # |
| History of myocardial infarction | 364 | 5.1 | 377 | 6.2 | 0.046 |
| History of stroke or TIA | 549 | 7.7 | 504 | 8.3 | 0.021 |
| Congestive heart failure | 1576 | 22.1 | 1318 | 21.7 | 0.011 |
| Cardiac arrhythmias | 1739 | 24.4 | 1567 | 25.8 | 0.034 |
| Hypertension | 6003 | 84.2 | 4878 | 80.3 | 0.102 # |
| Neurodegenerative disorders | 428 | 6.0 | 413 | 6.8 | 0.033 |
| Chronic pulmonary disease | 984 | 13.8 | 778 | 12.8 | 0.030 |
| Diabetes, uncomplicated | 1668 | 23.4 | 1895 | 31.2 | 0.177 # |
| Diabetes, complicated | 1112 | 15.6 | 1458 | 24.0 | 0.214 # |
| Diabetes, total | 2082 | 29.2 | 2509 | 41.3 | 0.255 # |
| Hypothyroidism | 1547 | 21.7 | 486 | 8.0 | 0.391 # |
| Obesity | 763 | 10.7 | 796 | 13.1 | 0.074 |
| Weight loss | 349 | 4.9 | 164 | 2.7 | 0.115 # |
| Depression | 713 | 10.0 | 377 | 6.2 | 0.142 # |
| Smoking | 741 | 10.4 | 936 | 15.4 | 0.149 # |
| Optimal pharmacological therapy during the prior year | 1355 | 19.0 | 1640 | 27.0 | 0.190 # |
| Oral anticoagulation during the prior year | 1119 | 15.7 | 1027 | 16.9 | 0.030 |
| Age, mean (SD) | N/A | 77.01 (10.15) | N/A | 71.34 (10.51) | 0.549 # |
| Prior hospital visits, mean (SD) | N/A | 0.76 (1.21) | N/A | 0.76 (1.27) | <0.001 |
| No of different prescriptions during the prior year, mean (SD) | N/A | 10.03 (5.75) | N/A | 9.19 (5.85) | 0.144 # |
| Number of surgeries at index, mean (SD) | N/A | 1.76 (1.41) | N/A | 1.78 (1.69) | 0.007 |
| Hospital length of stay, mean (SD) | N/A | 5.85 (8.48) | N/A | 5.54 (9.01) | 0.036 |
| Follow-up time, median [Q1, Q3] | N/A | 1274 [846.0, 1798.0] | N/A | 1302 [874.0, 1816.5] | 0.047 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Behrendt, C.-A.; Sedrakyan, A.; Katsanos, K.; Nordanstig, J.; Kuchenbecker, J.; Kreutzburg, T.; Secemsky, E.A.; Debus, E.S.; Marschall, U.; Peters, F. Sex Disparities in Long-Term Mortality after Paclitaxel Exposure in Patients with Peripheral Artery Disease: A Nationwide Claims-Based Cohort Study. J. Clin. Med. 2021, 10, 2978. https://doi.org/10.3390/jcm10132978
Behrendt C-A, Sedrakyan A, Katsanos K, Nordanstig J, Kuchenbecker J, Kreutzburg T, Secemsky EA, Debus ES, Marschall U, Peters F. Sex Disparities in Long-Term Mortality after Paclitaxel Exposure in Patients with Peripheral Artery Disease: A Nationwide Claims-Based Cohort Study. Journal of Clinical Medicine. 2021; 10(13):2978. https://doi.org/10.3390/jcm10132978
Chicago/Turabian StyleBehrendt, Christian-Alexander, Art Sedrakyan, Konstantinos Katsanos, Joakim Nordanstig, Jenny Kuchenbecker, Thea Kreutzburg, Eric A. Secemsky, Eike Sebastian Debus, Ursula Marschall, and Frederik Peters. 2021. "Sex Disparities in Long-Term Mortality after Paclitaxel Exposure in Patients with Peripheral Artery Disease: A Nationwide Claims-Based Cohort Study" Journal of Clinical Medicine 10, no. 13: 2978. https://doi.org/10.3390/jcm10132978
APA StyleBehrendt, C.-A., Sedrakyan, A., Katsanos, K., Nordanstig, J., Kuchenbecker, J., Kreutzburg, T., Secemsky, E. A., Debus, E. S., Marschall, U., & Peters, F. (2021). Sex Disparities in Long-Term Mortality after Paclitaxel Exposure in Patients with Peripheral Artery Disease: A Nationwide Claims-Based Cohort Study. Journal of Clinical Medicine, 10(13), 2978. https://doi.org/10.3390/jcm10132978








