Outcomes of Universal Newborn Screening Programs: Systematic Review
Abstract
1. Introduction
2. Materials and Methods
2.1. PCHL Definition
2.2. Search Strategy
2.3. Inclusion and Exclusion Criteria
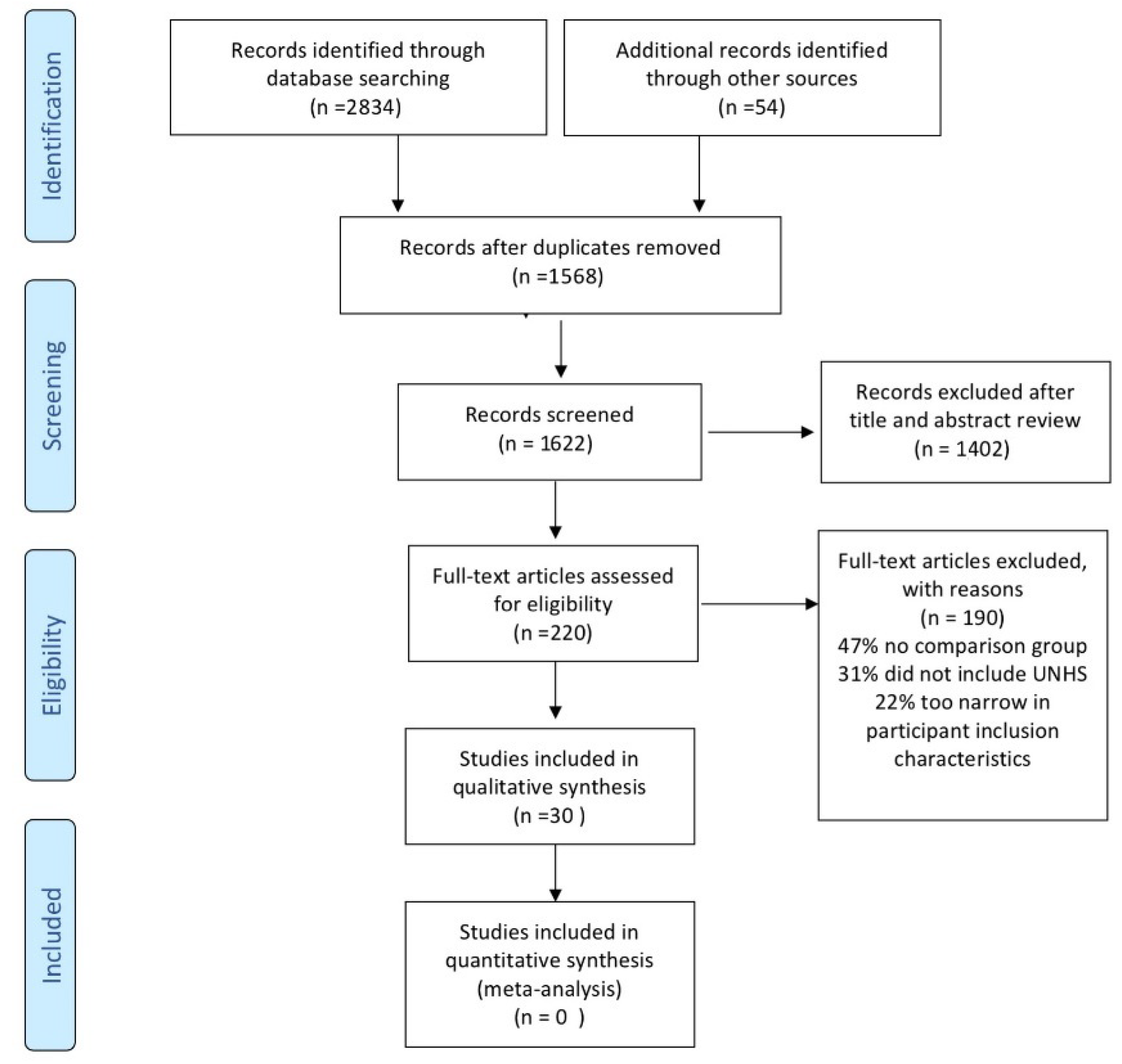
2.4. Article Selection and Data Extraction
2.5. Synthesis of Included Studies
2.6. Quality Analysis and Level of Evidence
3. Results
3.1. Study Characteristics
3.1.1. Question 1: Early identification and Intervention
Does UNHS Lower the Age of Identification and Increase the Number of Children Identified Early?
Does UNHS Lower the Age of Amplification Fit?
Does UNHS Lower the Age at Which Early Intervention Services Are Initiated?
3.1.2. Question 2: Developmental Outcomes
Does UNHS Improve Receptive and Expressive Language?
Does UNHS Improve Speech Perception and Speech Production?
Does UNHS Improve Literacy?
Does UNHS Improve Social Development?
Does UNHS Reduce Behavior Problems?
Does UNHS Improve Quality of Life?
3.1.3. Question 3: Cost Effectiveness
Is UNHS Cost Effective and Is There a Cost Benefit?
3.1.4. Question 4: Adverse Effects
Does UNHS Cause Social and/or Emotional Harm (e.g., Worry, Stress, Anxiety) to Parents (Mothers)?
3.2. Quality Analysis and Level of Evidence
3.2.1. Quality Analysis of Included Studies
3.2.2. Determination of Level of Cumulative Evidence
4. Discussion
4.1. Comparison of UNHS Outcomes of Other Newborn Screening Programs
4.2. Limitations of Existing Evidence and the Review Process
Supplementary Materials
Author Contributions
Funding
Institutional Reviewer Board Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| CASP | Critical Appraisal Skills Programme |
| EID | Early-identified |
| GRADE | Grading of Recommendations Assessment, Development and Evaluation |
| GDP | Guideline Development Group |
| HVDT | Health Visitor Distraction Testing |
| LID | Late-identified |
| LMIC | Low Middle Income Countries |
| NICU | Newborn intensive care unit |
| NS | No Screen |
| PCHL | Permanent childhood hearing loss |
| PICOTS | Participants: Intervention, Comparators, Outcomes, Timings and Study |
| PRISMA | Preferred Reporting Items for Systematic reviews and Meta-analyses |
| RFS | Risk Factor Screen |
| SD | Standard deviation |
| UNHS | Universal newborn hearing screening |
| UK | United Kingdom |
| US | United States |
| VHD | Very highly developed |
| WHO | World Hearing Organization |
| CYI | (Christine Yoshinaga-Itano) |
| CH | (Cynthia Hunnicutt) |
| VM | (Vinaya Manchaiah) |
References
- Butcher, E.; Dezateux, C.; Cortina-Borja, M.; Knowles, R.L. Prevalence of permanent childhood hearing loss detected at the universal newborn hearing screen: Systematic review and meta-analysis. PLoS ONE 2019, 14, e0219600. [Google Scholar] [CrossRef] [PubMed]
- Bussé, A.M.L.; Hoeve, H.L.J.; Nasserinejad, K.; Mackey, A.R.; Simonsz, H.J.; Goedegebure, A. Prevalence of permanent neonatal hearing impairment: Systematic review and Bayesian meta-analysis. Int. J. Audiol. 2020, 59, 475–485. [Google Scholar] [CrossRef]
- Qi, S.; Mitchell, R.E. Large-scale academic achievement testing of deaf and hard-of-hearing students: Past, present, and future. J. Deaf. Stud. Deaf. Educ. 2011, 17, 1–18. [Google Scholar] [CrossRef]
- Wauters, L.N.; Van Bon, W.H.J.; Tellings, A.E.J.M. Reading comprehension of Dutch deaf children. Read. Writ. 2006, 19, 49–76. [Google Scholar] [CrossRef]
- Nelson, H.D.; Bougatsos, C.; Nygren, P. Universal newborn hearing screening: Systematic review to update the 2001 US preventive services task force recommendation. Pediatrics 2008, 122, e266–e276. [Google Scholar] [CrossRef]
- Davis, A.; Bamford, J.; Wilson, I.; Ramkalawan, T.; Forshaw, M.; Wright, S. A critical review of the role of neonatal hearing screening in the detection of congenital hearing impairment. Health Technol. Assess. 1997, 10, 1–176. [Google Scholar] [CrossRef] [PubMed]
- The use of automated auditory brainstem response and otoacoustic emissions tests for newborn hearing screening. In IHE Report: Screening Newborns for Hearing; IHE: Edmonton, AB, Canada, 2007; p. 102.
- Merlin, T.; Hedayati, H.; Sullivan, T.; Buckley, E.; Newton, S.; Hodgkinson, B.; Bywood, P.; Jenner, F.; Moss, J. Universal Neonatal Hearing Screening; MSAC Reference 17 Assessment Report; MSAC: Canberra, Australia, 2007. [Google Scholar]
- Joint Committee on Infant Hearing. Year 2007 position statement: Principles and guidelines for early hearing detection and intervention programs. Pediatrics 2007, 120, 898–921. [Google Scholar] [CrossRef]
- Joint Committee on Infant Hearing. Year 2019 Position Statement: Principles and Guidelines for Early Hearing Detection and Intervention Programs. J. Early Hear. Detect. Interv. 2019, 4, 1–44. [Google Scholar]
- Yoshinaga-Itano, C.; Hunnicutt, C.; Manchaiah, V. A Systematic Review of the Evidence for the Effectiveness of Universal Newborn Hearing Screening. PROSPERO 2020, CRD42020175451. [Google Scholar]
- Schardt, C.; Adams, M.B.; Owens, T.; Keitz, S.; Fontelo, P. Utilization of the PICO Framework to Improve Searching PubMed for Clinical Questions. BMC Med. Inform. Decis. Mak. 2007, 7, 16. [Google Scholar] [CrossRef]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. BMJ 2009, 339. [Google Scholar] [CrossRef]
- Akers, J.; University of New York, Centre for Reviews and Dissemination. Systematic Reviews: CRD’s Guidance for Undertaking Reviews in Health Care; CRD University of York: York, UK, 2009. [Google Scholar]
- Campbell, M.; McKenzie, J.E.; Sowden, A.; Katikireddi, S.V.; Brennan, S.E.; Ellis, S.; Hartmann-Boyce, J.; Ryan, R.; Shepperd, S.; Thomas, J.; et al. Synthesis without meta-analysis (SWiM) in systematic reviews: Reporting guideline. BMJ 2020, 368. [Google Scholar] [CrossRef] [PubMed]
- Popay, J.; Roberts, H.; Sowden, A.; Petticrew, M.; Arai, L.; Rodgers, M.; Britten, N.; Roen, K.; Duffy, S.; Guidance on the Conduct of Narrative Synthesis in Systematic Reviews. A Product from the ESRC Methods Programme. Version 1. Available online: http://citeseerx.ist.psu.edu/viewdoc/download?doi=10.1.1.178.3100&rep=rep1&type=pdf (accessed on 15 May 2021).
- Taylor, R.S.; Reeves, B.C.; Ewings, P.E.; Taylor, R.J. Critical appraisal skills training for health care professionals: A randomized controlled trial [ISRCTN46272378]. BMC Med. Educ. 2004, 4. [Google Scholar] [CrossRef]
- Balshem, H.; Helfand, M.; Schünemann, H.J.; Oxman, A.D.; Kunz, R.; Brozek, J.; Vist, G.E.; Falck-Ytter, Y.; Meerpohl, J.; Norris, S. GRADE guidelines: 3. Rating the quality of evidence. J. Clin. Epidemiol. 2011, 64, 401–406. [Google Scholar] [CrossRef] [PubMed]
- Kennedy, C.R.; McCann, D.C.; Campbell, M.J.; Law, C.M.; Mullee, M.; Petrou, S.; Watkin, P.; Worsfold, S.; Yuen, H.M.; Stevenson, J. Language ability after early detection of permanent childhood hearing impairment. N. Engl. J. Med. 2006, 354, 2131–2141. [Google Scholar] [CrossRef] [PubMed]
- Uus, K.; Bamford, J. Effectiveness of population-based newborn hearing screening in England: Ages of interventions and profile of cases. Pediatrics 2006, 117, e887–e893. [Google Scholar] [CrossRef]
- Wood, S.A.; Sutton, G.J.; Davis, A.C. Performance and characteristics of the newborn hearing screening programme in England: The first seven years. Int. J. Audiol. 2015, 54, 353–358. [Google Scholar] [CrossRef]
- Wake, M.; Ching, T.Y.C.; Wirth, K.; Poulakis, Z.; Mensah, F.K.; Gold, L.; King, A.; Bryson, H.E.; Reilly, S.; Rickards, F. Population outcomes of three approaches to detection of congenital hearing loss. Pediatrics 2016, 137, e20151722. [Google Scholar] [CrossRef]
- Sininger, Y.S.; Martinez, A.; Eisenberg, L.; Christensen, E.; Grimes, A.; Hu, J. Newborn hearing screening speeds diagnosis and access to intervention by 20–25 months. J. Am. Acad. Audiol. 2009, 20, 049–057. [Google Scholar] [CrossRef]
- Dalzell, L.; Orlando, M.; MacDonald, M.; Berg, A.; Bradley, M.; Cacace, A.; Campbell, D.; DeCristofaro, J.; Gravel, J.; Greenberg, E.; et al. The New York State universal newborn hearing screening demonstration project: Ages of hearing loss identification, hearing aid fitting, and enrollment in early intervention. Ear Hear. 2000, 21, 118–130. [Google Scholar] [CrossRef]
- Yoshinaga-Itano, C.; Coulter, D.; Thomson, V. Developmental outcomes of children with hearing loss born in Colorado hospitals with and without universal newborn hearing screening programs. Semin. Neonatol. 2001, 6, 521–529. [Google Scholar] [CrossRef] [PubMed]
- McCann, D.C.; Worsfold, S.; Law, C.M.; Mullee, M.; Petrou, S.; Stevenson, J.; Yuen, H.M.; Kennedy, C.R. Reading and communication skills after universal newborn screening for permanent childhood hearing impairment. Arch. Dis. Child. 2008, 94, 293–297. [Google Scholar] [CrossRef]
- Worsfold, S.; Mahon, M.; Yuen, H.M.; Kennedy, C. Narrative skills following early confirmation of permanent childhood hearing impairment. Dev. Med. Child. Neurol. 2010, 52, 922–928. [Google Scholar] [CrossRef]
- Stevenson, J.; McCann, D.C.; Law, C.M.; Mullee, M.; Petrou, S.; Worsfold, S.; Yuen, H.M.; Kennedy, C.R. The effect of early confirmation of hearing loss on the behaviour in middle childhood of children with bilateral hearing impairment. Dev. Med. Child. Neurol. 2010, 53, 269–274. [Google Scholar] [CrossRef] [PubMed]
- Stevenson, J.; Pimperton, H.; Kreppner, J.; Worsfold, S.; Terlektsi, E.; Mahon, M.; Kennedy, C. Language and reading comprehension in middle childhood predicts emotional and behaviour difficulties in adolescence for those with permanent childhood hearing loss. J. Child. Psychol. Psychiatry 2018, 59, 180–190. [Google Scholar] [CrossRef]
- Pimperton, H.; Blythe, H.; Kreppner, J.; Mahon, M.; Peacock, J.L.; Stevenson, J.; Terlektsi, E.; Worsfold, S.; Yuen, H.M.; Kennedy, C.R. The impact of universal newborn hearing screening on long-term literacy outcomes: A prospective cohort study. Arch. Dis. Child. 2016, 101, 9–15. [Google Scholar] [CrossRef]
- Korver, A.M.H.; Konings, S.; Dekker, F.W.; Beers, M.; Wever, C.C.; Frijns, J.H.M.; Oudesluys-Murphy, A.M.; DECIBEL Collaborative Study Group. Newborn hearing screening vs later hearing screening and developmental outcomes in children with permanent childhood hearing impairment. JAMA 2010, 304, 1701. [Google Scholar] [CrossRef]
- Sininger, Y.S.; Grimes, A.; Christensen, E. Auditory development in early amplified children: Factors influencing auditory-based communication outcomes in children with hearing loss. Ear Hear. 2010, 31, 166–185. [Google Scholar] [CrossRef] [PubMed]
- Yoshinaga-Itano, C.; Coulter, D.; Thomson, V. The Colorado Newborn Hearing Screening Project: Effects on speech and language development for children with hearing loss. J. Perinatol. 2000, 20, S132–S137. [Google Scholar] [CrossRef]
- Schroeder, L.; Petrou, S.; Kennedy, C.; McCann, D.; Law, C.; Watkin, P.M.; Worsfold, S.; Yuen, H.M. The Economic Costs of Congenital Bilateral Permanent Childhood Hearing Impairment. Pediatrics 2006, 117, 1101–1112. [Google Scholar] [CrossRef]
- Chorozoglou, M.; Mahon, M.; Pimperton, H.; Worsfold, S.; Kennedy, C.R. Societal costs of permanent childhood hearing loss at teen age: A cross-sectional cohort follow-up study of universal newborn hearing screening. BMJ Paediatr. Open 2018, 2, e000228. [Google Scholar] [CrossRef]
- Mehl, A.L.; Thomson, V. The Colorado Newborn Hearing Screening Project, 1992-1999: On the threshold of effective population-based universal newborn hearing screening. Pediatrics 2002, 109, e7. [Google Scholar] [CrossRef]
- Keren, R.; Helfand, M.; Homer, C.; McPhillips, H.; Lieu, T.A. Projected cost-effectiveness of statewide universal newborn hearing screening. Pediatrics 2002, 110, 855–864. [Google Scholar] [CrossRef] [PubMed]
- Kennedy, C.R.; Kimm, L.; Cafarelli Dees, D.; Campbell, M.J.; Thornton, A.R.D.; Bamber, J.; Innes, V.; Lloyd-Hughes, S.; Parish, R.; Woodhead, C.; et al. Controlled trial of universal neonatal screening for early identification of permanent childhood hearing impairment. Lancet 1998, 352, 1957–1964. [Google Scholar] [CrossRef]
- Weichbold, V.; Welzl-Mueller, K. Maternal concern about positive test results in universal newborn hearing screening. Pediatrics 2001, 108, 1111–1116. [Google Scholar] [CrossRef]
- Tueller, S.J.; White, K.R. Maternal anxiety associated with newborn hearing screening. J. Early Hear. Detect. Interv. 2016, 1, 87–92. [Google Scholar]
- Watkin, P.M.; Baldwin, M.; Dixon, R.; Beckman, A. Maternal anxiety and attitudes to universal neonatal hearing screening. Br. J. Audiol. 1998, 32, 27–37. [Google Scholar] [CrossRef]
- Crockett, R.; Wright, A.J.; Uus, K.; Bamford, J.; Marteau, T.M. Maternal anxiety following newborn hearing screening: The moderating role of knowledge. J. Med. Screen. 2006, 13, 20–25. [Google Scholar] [CrossRef]
- Crockett, R.; Baker, H.; Uus, K.; Bamford, J.; Marteau, T.M. Maternal anxiety and satisfaction following infant hearing screening: A comparison of the health visitor distraction test and newborn hearing screening. J. Med. Screen. 2005, 12, 78–82. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Vohr, B.R.; Letourneau, K.S.; McDermott, C. Maternal worry about neonatal hearing screening. J. Perinatol. 2001, 21, 15–20. [Google Scholar] [CrossRef]
- Kolski, C.; Le Driant, B.; Lorenzo, P.; Vandromme, L.; Strunski, V. Early hearing screening: What is the best strategy? Int. J. Pediatr. Otorhinolaryngol 2007, 71, 1055–1060. [Google Scholar] [CrossRef]
- Khairi, M.D.M.; Rafidah, K.N.; Affizal, A.; Normastura, A.R.; Suzana, M.; Normani, Z.M. Anxiety of the Mothers with Referred Baby during Universal Newborn Hearing Screening. Int. J. Pediatric Otorhinolaryngol. 2011, 75, 513–517. [Google Scholar] [CrossRef]
- Stuart, A.; Moretz, M.; Yang, E.Y. An investigation of maternal stress after neonatal hearing screening. Am. J. Audiol. 2000, 9, 135–141. [Google Scholar] [CrossRef]
- Tueller, S.J. Maternal Worry about Infant Health, Maternal Anxiety, and Maternal Perceptions of Child Vulnerability Associated with Newborn Hearing Screening Results. Unpublished. Master’s Thesis, Utah State University, Logan, UT, USA, 2006. [Google Scholar]
- Chiong, C.; Ostrea, E.J.; Reyes, A.; Llanes, E.G.; Uy, M.E.; Chan, A. Correlation of hearing screening with developmental outcomes in infants over a 2-year period. Acta Oto-Laryngologica 2007, 127, 384–388. [Google Scholar] [CrossRef]
- Bevilacqua, M.C.; Alvarenga, K.d.F.; Costa, O.A.; Moret, A.L.M. The universal newborn hearing screening in Brazil: From identification to intervention. Int. J. Pediatr. Otorhinolaryngol. 2010, 74, 510–515. [Google Scholar] [CrossRef]
- Poonual, W.; Navacharoen, N.; Kangsanarak, J.; Namwongprom, S. Outcome of early identification and intervention on infants with hearing loss under universal hearing screening program. J. Med. Assoc. Thai. 2017, 100, 197. [Google Scholar] [PubMed]
- Sahli, A.S. Developments of children with hearing loss according to the age of diagnosis, amplification, and training in the early childhood period. Eur. Arch. Oto-Rhino-Laryngol. 2019, 276, 2457–2463. [Google Scholar] [CrossRef] [PubMed]
- Olusanya, B.O.; Somefun, A.O. Place of Birth and Characteristics of Infants with Congenital and Early-Onset Hearing Loss in a Developing Country. Int. J. Ptry. Int. J. Pediatr. Otorhinolaryngol. 2009, 73, 1263–1269. [Google Scholar] [CrossRef] [PubMed]
- EQUATOR Network Guidelines. 2021. Available online: https://www.equator-network.org/reporting-guidelines/ (accessed on 1 May 2021).
- Yoshinaga-Itano, C.; Sedey, A.L.; Coulter, D.K.; Mehl, A.L. Language of Early- and Later-identified Children With Hearing Loss. Pediatrics 1998, 102, 1161–1171. [Google Scholar] [CrossRef]
- Bishop, D.V.M. Test for Reception of Grammar; Age and Cognitive Performance Research Centre, University of Manchester: Manchester, UK, 1983. [Google Scholar]
- Dunn, L.M.; Whetton, C.; Burley, J. British Picture Vocabulary Scale, 2nd ed.; NFER-Nelson: Windsor, UK, 1997. [Google Scholar]
- Sparrow, S.S.; Balla, D.A.; Cicchetti, D.V. Vineland: Adaptive Behavior: Scales, Interview Edition: Survey form Manual; American Guidance Service: Circle Pines, MN, USA, 1984. [Google Scholar]
- Wechsler, D. Wechsler Objective Reading Dimensions; The Psychological Corporation: London, UK, 2003. [Google Scholar]
- Stothard, S.E.; Hulme, C.; Clarke, P.; Bowyer-Crane, C.; Harrington, A.; Truelove, E.; Nation, K. YARC York Assessment of Reading for Comprehension Secondary; GL Assessment: London, UK, 2010. [Google Scholar]
- Ireton, H.; Thwing, E. The Minnesota Child Development Inventory in the psychiatric-developmental evaluation of the preschool-age child. Child. Psychiatry Hum. Dev. 1972, 3, 102–114. [Google Scholar] [CrossRef]
- Zimmerman, I.L.; Steiner, V.G.; Pond, R.E. Preschool Language Scale, 4th ed.; Harcourt Assessment: London, UK, 2002. [Google Scholar]
- Dunn, L.M.; Dunn, D.M. The Peabody Picture Vocabulary Test, 4th ed.; Pearson: Minneapolis, MN, USA, 2007. [Google Scholar]
- Renfrew, C. Renfrew Bus Story Manual: A Test of Narrative Speech, 3rd ed.; Winslow Press: Oxford, UK, 1995. [Google Scholar]
- Bishop, D.V. Development of the Children’s Communication Checklist (CCC): A method for assessing qualitative aspects of communicative impairment in children. J. Child. Psychol. Psychiatry 1998, 39, 879–891. [Google Scholar] [CrossRef] [PubMed]
- Crystal, D.; Fletcher, P.; Garman, M. The Grammatical Analysis of Language Disability—A Procedure for Assessment and Remediation, 1st ed.; Edward Arnold: London, UK, 1976. [Google Scholar]
- Raven, C.; Raven, J.C.; Court, J.H. Manual for Raven’s Progressive Matrices and Vocabulary Scales; Oxford Psychologists Press: Oxford, UK, 1998. [Google Scholar]
- Goodman, R. The Strengths and Difficulties Questionnaire: A Research Note. J. Child. Psychol. Psychiatry 1997, 38, 581–586. [Google Scholar] [CrossRef]
- Varni, J.W.; Burwinkle, T.M.; Seid, M.; Skarr, D. The PedsQL 4.0 as a pediatric population health measure: Feasibility, reliability, and validity. Ambul. Pediatr. 2003, 3, 329–341. [Google Scholar] [CrossRef]
- Fenson, L.; Marchman, V.A.; Thal, D.; Dale, P.S.; Bates, E.; Reznick, J.S. The MacArthur-Bates Communicative Development Inventories: User’s Guide and Technical Manual, 2nd ed.; Paul, H. Brookes: Baltimore, MD, USA, 2007. [Google Scholar]
- Sumner, G.; Spietz, A. NCAST Caregiver/Parent-Child Interaction Teaching Manual; NCAST/Caregiver: Seattle, WA, USA, 1994. [Google Scholar]
- Jerger, S.; Lewis, S.; Hawkins, J.; Jerger, J. Pediatric speech intelligibility test. I. Generation of test materials. Int. J. Pediatr. Otorhinolaryngol. 1980, 2, 217–230. [Google Scholar] [CrossRef]
- Boothroyd, A.; Eisenberg, L.S.; Martinez, A.S. OLIMSPAC Version 3.1d; House Ear Institute: Los Angeles, CA, USA, 2005. [Google Scholar]
- Fudala, J. Arizona Test of Articulation-3; PRO-Ed, Inc.: Austin, TX, USA, 2000. [Google Scholar]
- Edwards, S.; Fletcher, P.; Garman, M.; Hughes, A.; Letts, C.; Sinda, I. Reynell Developmental Language Scales; NFER-Nelson Publishing: Windsor, UK, 1997. [Google Scholar]
- Marteau, T.M.; Bekker, H. The development of a 6-item short-form of the state scale of the Spielberger State Trait Anxiety In-ventory (STAI). Br. J. Clin. Psychol. 1992, 31, 301–306. [Google Scholar] [CrossRef]
- Montgomery, S.A.; Asberg, M. A new depression scale designed to be sensitive to change. Br. J. Psychiat. 1979, 134, 382–389. [Google Scholar] [CrossRef]
- Cox, J.L.; Holden, J.M.; Sagovsky, R. Detection of postnatal depression: Development of the 10 items Edinburgh Postnatal Depression Scale. Br. J. Psychiat. 1987, 150, 782–786. [Google Scholar] [CrossRef] [PubMed]
- Beck, A.T.; Epstein, N.; Brown, G.; Steer, R.A. An inventory for measuring clinical anxiety: Psychometric properties. J. Consult. Clin. Psychol. 1988, 56, 893–897. [Google Scholar] [CrossRef]
- Abidin, R.R. The Parenting Stress Index: Manual, 3rd ed.; Psychological Assessment Resources: Odessa, FL, USA, 1995. [Google Scholar]
| Concept 1 | AND | Concept 2 | AND | Concept 3 |
| (newborn hearing screening) OR (universal newborn hearing screening) | (hearing loss) OR (hearing impairment) OR (childhood hearing impairment) OR (permanent childhood hearing loss) | (outcome) OR (speech outcome) OR (language outcome) OR (literacy outcome) OR (maternal anxiety) OR (maternal stress) |
| Inclusion | Exclusion | |
|---|---|---|
| Participants | Newborns (gestational age ≥37 weeks at birth without complications) and children within the first year of life undergoing Newborn Hearing Screening (NHS)/ Universal Newborn Hearing Screening (UNHS) | Only reporting screening of high-risk babies |
| Interventions | Universal newborn hearing screening | Target (or selective) screening with the UNHS with no comparison to UNHS |
| Comparators | No screening or selected/targeted screen | |
| Outcomes |
| Not reporting the key outcomes listed |
| Study designs |
|
|
| Timings | No restrictions | No restrictions |
| Other criteria | English language articles | Non-English articles |
| Study | Early Identification and Intervention | Developmental Outcomes | Cost Analysis | Adverse Effects | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Age of Identification | Age at Amplify | Intervention Start | Receptive Language | Expressive Language | Speech Perception | Speech Production | Literacy | Social dev | Behavioral Problems | Quality of Life | |||
| Question 1: Early identification and intervention (7 studies) | |||||||||||||
| Kennedy et al. (2006) [19] | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | |||||||
| Uus and Bamford (2006) [20] | ✓ | ✓ | ✓ | ||||||||||
| Wood et al. (2015) [21] | ✓ | ✓ | ✓ | ||||||||||
| Wake et al. (2016) [22] | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | |||||||
| Sininger et al. (2009) [23] | ✓ | ✓ | ✓ | ||||||||||
| Dalzell et al. (2000) [24] | ✓ | ✓ | ✓ | ||||||||||
| Yoshinaga-Itano et al. (2001) [25] | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | |||||||
| Question 2: Developemental outcomes (11 studies, Kennedy et al., 2006 and Wake et al., 2016 listed earlier) | |||||||||||||
| McCann et al. (2008) [26] | ✓ | ✓ | ✓ | ||||||||||
| Worsfold et al. (2010) [27] | ✓ | ✓ | |||||||||||
| Stevenson et al. (2010) [28] | ✓ | ✓ | ✓ | ✓ | |||||||||
| Stevenson et al. (2018) [29] | ✓ | ✓ | ✓ | ✓ | ✓ | ||||||||
| Pimperton et al. (2016) [30] | ✓ | ✓ | ✓ | ||||||||||
| Korver et al. (2010) [31] | ✓ | ✓ | ✓ | ✓ | |||||||||
| Sininger et al. (2010) [32] | ✓ | ✓ | ✓ | ✓ | |||||||||
| Yoshinaga-Itano et al. (2000) [33] | ✓ | ✓ | ✓ | ||||||||||
| Yoshinaga-Itano et al. (2020) [11] | ✓ | ✓ | |||||||||||
| Question 3: Cost effectiveness (4 studies) | |||||||||||||
| Schroeder et al. (2006) [34] | ✓ | ||||||||||||
| Chorozoglou et al. (2018) [35] | ✓ | ||||||||||||
| Mehl and Thomson (2002) [36] | ✓ | ||||||||||||
| Keren et al. (2002) [37] | ✓ | ||||||||||||
| Question 4: Adverse effects (10 studies) | |||||||||||||
| Kennedy et al. (1998) [38] | ✓ | ||||||||||||
| Weichbold and Welzl-Mueller (2001) [39] | ✓ | ||||||||||||
| Tueller and White (2016) [40] | ✓ | ||||||||||||
| Watkin et al. (1998) [41] | ✓ | ||||||||||||
| Crockett et al. (2006) [42] | ✓ | ||||||||||||
| Crockett et al. (2005) [43] | ✓ | ||||||||||||
| Vohr et al. (2001) [44] | ✓ | ||||||||||||
| Kolski et al. (2007) [45] | ✓ | ||||||||||||
| Khairi et al. (2011) [46] | ✓ | ||||||||||||
| Stuart et al. (2000) [47] | ✓ | ||||||||||||
| a. CASP ratings for cohort studies (Y = Yes, C = Can’t tell, N = No, NA = Not applicable) | |||||||||||||||||||||||||
| Article | 1. Did the study address a clearly focused issue? | 2. Was the cohort recruited in an acceptable way? | 3. Was the exposure accurately measured to minimize bias? | 4. Was the outcome accurately measured to minimize bias? | 5. (a) Have the authors identified all important confounding factors? | 5. (b) Have they taken account of the confounding factors in the design and/or analysis? | 6. (a) Was the follow up of subjects complete enough? | 6. (b) Was the follow up of subjects long enough? | 7. What are the results of this study? | 8. How precise are the results? | 9. Do you believe the results? | 10. Can the results be applied to the local population? | 11. Do the results of this study fit with other available evidence? | 12. What are the implications of this study for practice? | |||||||||||
| Kennedy et al. (2006) [19] | Y | Y | Y | Y | C | C | Y | Y | Y | C | Y | Y | Y | Y | |||||||||||
| Uus and Bamford (2006) [20] | Y | Y | Y | Y | Y | Y | Y | Y | Y | Y | Y | Y | Y | Y | |||||||||||
| Wood et al. (2015) [21] | Y | Y | Y | Y | Y | Y | Y | Y | Y | Y | Y | Y | Y | Y | |||||||||||
| Wake et al. (2016) [22] | Y | Y | Y | Y | Y | Y | Y | C | Y | C | Y | Y | Y | Y | |||||||||||
| Sininger et al. (2010) [32] | Y | C | C | Y | C | C | C | Y | C | Y | Y | Y | Y | Y | |||||||||||
| Dalzell et al. (2000) [24] | Y | Y | Y | Y | Y | C | C | C | Y | Y | Y | Y | Y | Y | |||||||||||
| McCann et al. (2008) [26] | Y | Y | Y | Y | C | C | Y | Y | Y | Y | Y | Y | Y | Y | |||||||||||
| Worsfold et al. (2010) [27] | Y | Y | Y | Y | C | C | Y | Y | Y | C | Y | Y | Y | Y | |||||||||||
| Stevenson et al. (2010) [28] | Y | Y | Y | Y | C | C | Y | Y | C | C | Y | Y | Y | Y | |||||||||||
| Stevenson et al. (2018) [29] | Y | Y | Y | Y | C | C | Y | Y | Y | Y | Y | Y | Y | Y | |||||||||||
| Pimperton et al. (2016) [30] | Y | Y | Y | Y | C | C | Y | Y | Y | Y | Y | Y | Y | Y | |||||||||||
| Korver et al. (2010) [31] | Y | Y | C | C | C | C | Y | Y | C | C | Y | C | C | C | |||||||||||
| Sininger et al. (2009) [23] | Y | C | C | C | C | C | C | C | Y | C | Y | Y | Y | Y | |||||||||||
| Yoshinaga-Itano et al. (2020) [11] | Y | Y | Y | Y | C | C | Y | Y | Y | Y | Y | Y | Y | Y | |||||||||||
| Kennedy (1998) [38] | Y | Y | Y | Y | C | Y | Y | Y | Y | C | Y | Y | Y | Y | |||||||||||
| Weichbold and Welzl-Mueller (2001) [39] | Y | Y | Y | C | C | C | C | Y | C | C | Y | Y | Y | Y | |||||||||||
| Tueller and White (2016) [40] and Tueller (2006) [48] | Y | Y | Y | Y | Y | Y | Y | Y | Y | Y | Y | Y | Y | Y | |||||||||||
| Watkin et al. (1998) [41] | Y | Y | Y | Y | C | Y | Y | Y | Y | Y | Y | Y | Y | Y | |||||||||||
| Crockett et al. (2006) [42] | Y | Y | Y | C | Y | Y | C | C | Y | Y | Y | Y | Y | Y | |||||||||||
| Crockett et al. (2005) [43] | Y | C | Y | Y | C | C | Y | Y | Y | C | Y | Y | Y | Y | |||||||||||
| Vohr et al. (2001) [44] | Y | Y | C | C | C | C | C | C | Y | Y | Y | Y | Y | Y | |||||||||||
| Kolski et al. (2007) [45] | Y | Y | Y | Y | C | C | Y | C | Y | Y | Y | Y | Y | Y | |||||||||||
| Khairi et al. (2011) [46] | Y | Y | Y | C | C | C | C | C | Y | C | Y | Y | Y | Y | |||||||||||
| Stuart at al. (2000) [47] | Y | Y | Y | C | C | C | Y | C | Y | Y | Y | Y | Y | Y | |||||||||||
| b. CASP ratings case-control studies (Y = Yes, C = Can’t tell, N = No, NA = Not applicable) | |||||||||||||||||||||||||
| Article | 1. Did the study address a clearly focused issue? | 2. Did the authors use an appropriate method to answer their question? | 3. Were the cases recruited in an acceptable way? | 4. Were the controls selected in an acceptable way? | 5. Was the exposure accurately measured to minimize bias? | 6. (a) Aside from the experimental intervention, were the groups treated equally? | 6. (b) Have the authors taken account of the potential confounding factors in the design and/or in their analysis? | 7. How large was the treatment effect? | 8. How precise was the estimate of the treatment effect? | 9. Do you believe the results? | 10. Can the results be applied to the local population? | 11. Do the results of this study fit with other available evidence? | |||||||||||||
| Yoshinaga-Itano et al. (2001) [25] | Y | Y | Y | Y | Y | C | Y | Y | Y | Y | Y | Y | |||||||||||||
| Yoshinaga-Itano et al. (2000) [33] | Y | Y | C | C | Y | Y | C | Y | Y | Y | Y | Y | |||||||||||||
| c. CASP ratings economic evaluation studies (Y = Yes, C = Can’t tell, N = No, NA = Not applicable) | |||||||||||||||||||||||||
| Article | 1. Was a well-defined question posed? | 2. Was a comprehensive description of the competing alternatives given? | 3. Does the paper provide evidence that the programme would be effective? (i.e., would the programme do more good than harm?) | 4. Were the effects of the intervention identified, measured and valued appropriately? | 5. Were all important and relevant resources required, and health outcome costs for each alternative identified, measured in appropriate units and valued credibly? | 6. Were costs and consequences adjusted for different times at which they occurred (discounting)? | 7. What were the results of the evaluation?their analysis? | 8. Was an incremental analysis of the consequences and cost of alternatives performed? | 9. Was an adequate sensitivity analysis performed? | 10. Is the programme likely to be equally effective in your context or setting? | 11. Are the costs translatable to your setting? | 12. Is it worth doing in your setting? | |||||||||||||
| Schroeder et al. (2006) [34] | Y | Y | Y | Y | Y | C | Y | C | NA | Y | Y | Y | |||||||||||||
| Chorozoglou et al. (2018) [35] | Y | Y | Y | Y | Y | C | Y | C | NA | Y | Y | Y | |||||||||||||
| Mehl and Thomson (2002) [36] | Y | Y | Y | C | C | C | Y | C | N | Y | Y | Y | |||||||||||||
| Keren et al. (2002) [37] | Y | Y | Y | Y | C | N | Y | Y | NA | Y | C | Y | |||||||||||||
| Certainty Assessment | № of Patients | Effect | Certainty | Importance | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| № of Studies | Study Design | Risk of Bias | Inconsistency | Indirectness | Imprecision | Other Considerations | UNHS | No Screen and/or Targeted Screen | Relative (95% CI) | Absolute (95% CI) | ||
| Early identification and intervention (follow up: median 3 years) | ||||||||||||
| 7 | observational studies | not serious | serious | not serious | not serious | strong association all outcomes indicate lower age ID, age amp, and age Rx | 5,882,275 | 596,874 | not estimable | NA | ⨁⨁◯◯ LOW | CRITICAL |
| Developmental outcomes (follow up: median 3 years) | ||||||||||||
| 11 | observational studies | serious | serious | not serious | serious | strong association all plausible residual confounding would reduce the demonstrated effect | 725,037 | 1,556,737 | not estimable | NA | ⨁◯◯◯ VERY LOW | CRITICAL |
| Cost-benefit (follow up: median 5 years) | ||||||||||||
| 4 | observational studies | serious | serious | not serious | not serious | none | 68,714 | 88,019 | not estimable | NA | ⨁◯◯◯ VERY LOW | CRITICAL |
| Negative side effects (follow up: median 3 months) | ||||||||||||
| 10 | observational studies | serious | not serious | not serious | not serious | none | 717,826 | 88,019 | not estimable | NA | ⨁◯◯◯ VERY LOW | IMPORTANT |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yoshinaga-Itano, C.; Manchaiah, V.; Hunnicutt, C. Outcomes of Universal Newborn Screening Programs: Systematic Review. J. Clin. Med. 2021, 10, 2784. https://doi.org/10.3390/jcm10132784
Yoshinaga-Itano C, Manchaiah V, Hunnicutt C. Outcomes of Universal Newborn Screening Programs: Systematic Review. Journal of Clinical Medicine. 2021; 10(13):2784. https://doi.org/10.3390/jcm10132784
Chicago/Turabian StyleYoshinaga-Itano, Christine, Vinaya Manchaiah, and Cynthia Hunnicutt. 2021. "Outcomes of Universal Newborn Screening Programs: Systematic Review" Journal of Clinical Medicine 10, no. 13: 2784. https://doi.org/10.3390/jcm10132784
APA StyleYoshinaga-Itano, C., Manchaiah, V., & Hunnicutt, C. (2021). Outcomes of Universal Newborn Screening Programs: Systematic Review. Journal of Clinical Medicine, 10(13), 2784. https://doi.org/10.3390/jcm10132784







