Surgical Management of Gastric Cancer: A Systematic Review
Abstract
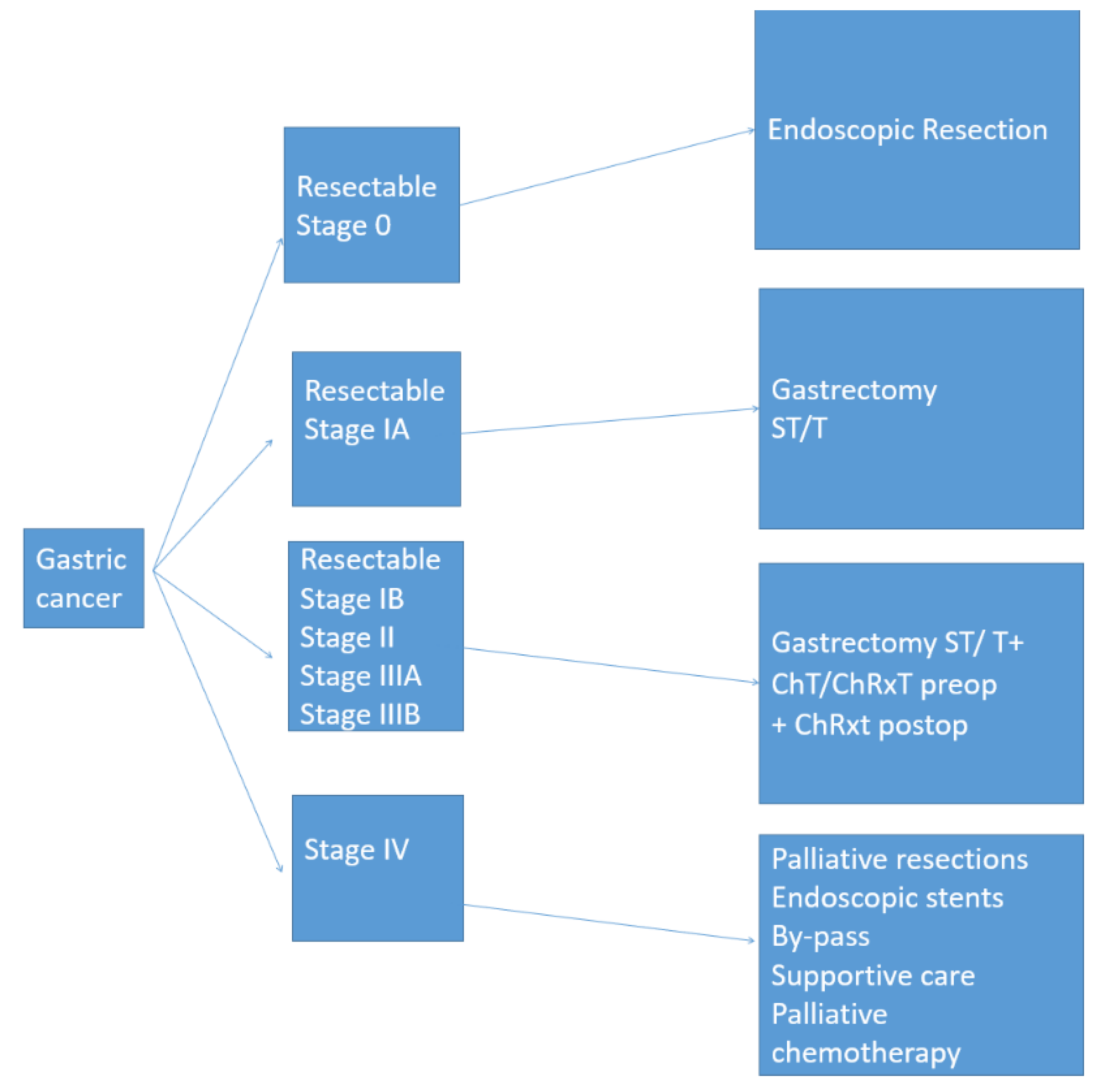
1. Introduction
2. Results and Discussion
2.1. Extent of Gastric Resection: Total Gastrectomy (TG), Subtotal Gastrectomy (SG), and Proximal Gastrectomy (PG)
2.2. Reconstruction Following Resection
2.3. Extending Resection to Adjacent Organs
2.4. Extent of Lymphadenectomy
2.5. Influence of Positive Resection Margins and Re-Resection
2.6. The Importance of High-Volume Centers
2.7. Laparoscopic Gastrectomy (LG)
3. Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Suh, Y.; Lee, J.; Woo, H.; Shin, D.; Kong, S.; Lee, H.; Shin, A.; Yang, H. National Cancer Screening Program for Gastric Cancer in Korea: Nationwide Treatment Benefit and Cost. Cancer 2020, 126, 1929–1939. [Google Scholar] [CrossRef]
- Petrillo, A.; Smyth, E.C. Multimodality Treatment for Localized Gastric Cancer: State of the Art and New Insights. Curr. Opin. Oncol. 2020, 32, 347–355. [Google Scholar] [CrossRef]
- Japanese Gastric Cancer Association. Japanese Gastric Cancer Treatment Guidelines 2018. Gastric Cancer 2021, 24, 1–21. [Google Scholar] [CrossRef] [PubMed]
- Song, Z.; Wu, Y.; Yang, J.; Yang, D.; Fang, X. Progress in the Treatment of Advanced Gastric Cancer. Tumor Biol. 2017, 39, 1010428317714626. [Google Scholar] [CrossRef] [PubMed]
- Tokunaga, M.; Sato, Y.; Nakagawa, M.; Aburatani, T.; Matsuyama, T.; Nakajima, Y.; Kinugasa, Y. Perioperative Chemotherapy for Locally Advanced Gastric Cancer in Japan: Current and Future Perspectives. Surg. Today 2020, 50, 30–37. [Google Scholar] [CrossRef] [PubMed]
- Zheng, L.; Wang, L.; Ajani, J.; Xie, K. Molecular Basis of Gastric Cancer Development and Progression. Gastric Cancer 2004, 7, 61–77. [Google Scholar] [CrossRef] [PubMed]
- Muir, J.; Aronson, M.; Esplen, M.; Pollett, A.; Swallow, C.J. Prophylactic Total Gastrectomy: A Prospective Cohort Study of Long-Term Impact on Quality of Life. J. Gastrointest. Surg. 2016, 20, 1950–1958. [Google Scholar] [CrossRef]
- Ilson, D.H. Advances in the Treatment of Gastric Cancer. Curr. Opin. Gastroenterol. 2018, 34, 465–468. [Google Scholar] [CrossRef]
- Bernards, N.; Creemers, G.; Nieuwenhuijzen, G.; Bosscha, K.; Pruijt, J.; Lemmens, V. No Improvement in Median Survival for Patients with Metastatic Gastric Cancer Despite Increased use of Chemotherapy. Ann. Oncol. 2013, 24, 3056–3060. [Google Scholar] [CrossRef]
- Nelen, S.; Heuthorst, L.; Verhoeven, R.; Polat, F.; Kruyt, P.M.; Reijnders, K.; Ferenschild, F.; Bonenkamp, J.; Rutter, J.; de Wilt, J. Impact of Centralizing Gastric Cancer Surgery on Treatment, Morbidity, and Mortality. J. Gastrointest. Surg. 2017, 21, 2000–2008. [Google Scholar] [CrossRef]
- Kumazu, Y.; Hayashi, T.; Yoshikawa, T.; Yamada, T.; Hara, K.; Shimoda, Y.; Nakazono, M.; Nagasawa, S.; Shiozawa, M.; Morinaga, S. Risk Factors Analysis and Stratification for Microscopically Positive Resection Margin in Gastric Cancer Patients. BMC Surg. 2020, 20, 95. [Google Scholar] [CrossRef]
- Kim, A.; Kim, B.S.; Yook, J.H.; Kim, B.S. Optimal Proximal Resection Margin Distance for Gastrectomy in Advanced Gastric Cancer. World J. Gastroenterol. 2020, 26, 2232–2246. [Google Scholar] [CrossRef]
- Qi, J.; Zhang, P.; Wang, Y.; Chen, H.; Li, Y. Does Total Gastrectomy Provide Better Outcomes than Distal Subtotal Gastrectomy for Distal Gastric Cancer? A Systematic Review and Meta-Analysis. PLoS ONE 2016, 11, e0165179. [Google Scholar] [CrossRef] [PubMed]
- Sugoor, P.; Shah, S.; Dusane, R.; Desouza, A.; Goel, M.; Shrikhande, S.V. Proximal Gastrectomy Versus Total Gastrectomy for Proximal Third Gastric Cancer: Total Gastrectomy is Not always Necessary. Langenbeck’s Arch. Surg. 2016, 401, 687–697. [Google Scholar] [CrossRef] [PubMed]
- Harrison, L.E.; Karpeh, M.S.; Brennan, M.F. Total Gastrectomy is Not Necessary for Proximal Gastric Cancer. Surgery 1998, 123, 127–130. [Google Scholar] [CrossRef]
- Hsu, C.; Chen, C.; Hsieh, Y.; Hsia, J.; Shai, S.; Kao, C. Esophageal Reflux After Total Or Proximal Gastrectomy in Patients with Adenocarcinoma of the Gastric Cardia. Am. J. Gastroenterol. 1997, 92, 1347–1350. [Google Scholar] [PubMed]
- Park, S.; Chung, H.Y.; Lee, S.S.; Kwon, O.; Yu, W. Serial Comparisons of Quality of Life After Distal Subtotal Or Total Gastrectomy: What are the Rational Approaches for Quality of Life Management? J. Gastric Cancer 2014, 14, 32–38. [Google Scholar] [CrossRef]
- Gouzi, J.L.; Huguier, M.; Fagniez, P.L.; Launois, B.; Flamant, Y.; Lacaine, F.; Paquet, J.C.; Hay, J.M. Total Versus Subtotal Gastrectomy for Adenocarcinoma of the Gastric Antrum. A French Prospective Controlled Study. Ann. Surg. 1989, 209, 162–166. [Google Scholar] [CrossRef]
- Robertson, C.S.; Chung, S.C.; Woods, S.D.; Griffin, S.M.; Raimes, S.A.; Lau, J.T.; Li, A.K. A Prospective Randomized Trial Comparing R1 Subtotal Gastrectomy with R3 Total Gastrectomy for Antral Cancer. Ann. Surg. 1994, 220, 176–182. [Google Scholar] [CrossRef]
- Bozzetti, F.; Marubini, E.; Bonfanti, G.; Miceli, R.; Piano, C.; Crose, N.; Gennari, L. Total Versus Subtotal Gastrectomy: Surgical Morbidity and Mortality Rates in a Multicenter Italian Randomized Trial. the Italian Gastrointestinal Tumor Study Group. Ann. Surg. 1997, 226, 613–620. [Google Scholar] [CrossRef]
- De Manzoni, G.; Verlato, G.; Roviello, F.; Di Leo, A.; Marrelli, D.; Morgagni, P.; Pasini, F.; Saragoni, L.; Tomezzoli, A. Subtotal Versus Total Gastrectomy for T3 Adenocarcinoma of the Antrum. Gastric Cancer 2003, 6, 237–242. [Google Scholar] [CrossRef]
- Diaz De Liano, A.; Oteiza Martinez, F.; Ciga, M.; Aizcorbe, M.; Cobo, F.; Trujillo, R. Impact of Surgical Procedure for Gastric Cancer on Quality of Life. Br. J. Surg. 2003, 90, 91–94. [Google Scholar] [CrossRef]
- Brennan, M.F. Current Status of Surgery for Gastric Cancer: A Review. Gastric Cancer 2005, 8, 64–70. [Google Scholar] [CrossRef] [PubMed]
- Ito, H.; Clancy, T.E.; Osteen, R.T.; Swanson, R.S.; Bueno, R.; Sugarbaker, D.J.; Ashley, S.W.; Zinner, M.J.; Whang, E.E. Adenocarcinoma of the Gastric Cardia: What is the Optimal Surgical Approach? J. Am. Coll. Surg. 2004, 199, 880–886. [Google Scholar] [CrossRef] [PubMed]
- Barbour, A.P.; Rizk, N.P.; Gonen, M.; Tang, L.; Bains, M.S.; Rusch, V.W.; Coit, D.G.; Brennan, M.F. Adenocarcinoma of the Gastroesophageal Junction: Influence of Esophageal Resection Margin and Operative Approach on Outcome. Ann. Surg. 2007, 246, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Xiao, H.; Ma, M.; Xiao, Y.; Ouyang, Y.; Tang, M.; Zhou, K.; Hong, Y.; Tang, B.; Zuo, C. Incomplete Resection and Linitis Plastica are Factors for Poor Survival After Extended Multiorgan Resection in Gastric Cancer Patients. Sci. Rep. 2017, 7, 15800. [Google Scholar] [CrossRef]
- Mao, X.; Xu, X.; Zhu, H.; Ji, C.; Lu, X.; Wang, B. A Comparison between Pylorus-Preserving and Distal Gastrectomy in Surgical Safety and Functional Benefit with Gastric Cancer: A Systematic Review and Meta-Analysis. World J. Surg. Oncol. 2020, 18, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Bueno, J.A.D.; Park, Y.; Ahn, S.; Park, D.J.; Kim, H. Function-Preserving Surgery in Gastric Cancer. J. Minim. Invasive Surg. 2018, 21, 141–147. [Google Scholar] [CrossRef]
- Hu, Y.; Zaydfudim, V.M. Quality of Life After Curative Resection for Gastric Cancer: Survey Metrics and Implications of Surgical Technique. J. Surg. Res. 2020, 251, 168–179. [Google Scholar] [CrossRef] [PubMed]
- Hirao, M.; Takiguchi, S.; Imamura, H.; Yamamoto, K.; Kurokawa, Y.; Fujita, J.; Kobayashi, K.; Kimura, Y.; Mori, M.; Doki, Y. Comparison of Billroth I and Roux-En-Y Reconstruction After Distal Gastrectomy for Gastric Cancer: One-Year Postoperative Effects Assessed by a Multi-Institutional RCT. Ann. Surg. Oncol. 2013, 20, 1591–1597. [Google Scholar] [CrossRef]
- Kimura, Y.; Mikami, J.; Yamasaki, M.; Hirao, M.; Imamura, H.; Fujita, J.; Takeno, A.; Matsuyama, J.; Kishi, K.; Hirao, T. Comparison of 5-year Postoperative Outcomes After Billroth I and Roux-en-Y Reconstruction Following Distal Gastrectomy for Gastric Cancer: Results from a multi-institutional Randomized Controlled Trial. Ann. Gastroenterol. Surg. 2021, 5, 93–101. [Google Scholar] [CrossRef]
- Angrisani, L.; Santonicola, A.; Iovino, P.; Formisano, G.; Buchwald, H.; Scopinaro, N. Bariatric Surgery Worldwide 2013. Obes. Surg. 2015, 25, 1822–1832. [Google Scholar] [CrossRef]
- Shen, J.; Ma, X.; Yang, J.; Zhang, J. Digestive Tract Reconstruction Options After Laparoscopic Gastrectomy for Gastric Cancer. World J. Gastrointest. Oncol. 2020, 12, 21. [Google Scholar] [CrossRef] [PubMed]
- Kano, Y.; Ohashi, M.; Ida, S.; Kumagai, K.; Makuuchi, R.; Sano, T.; Hiki, N.; Nunobe, S. Therapeutic Value of Splenectomy to Dissect Splenic Hilar Lymph Nodes for Type 4 Gastric Cancer Involving the Greater Curvature, Compared with Other Types. Gastric Cancer 2020, 23, 927–936. [Google Scholar] [CrossRef] [PubMed]
- Maruyama, K.; Okabayashi, K.; Kinoshita, T. Progress in Gastric Cancer Surgery in Japan and its Limits of Radicality. World J. Surg. 1987, 11, 418–425. [Google Scholar] [CrossRef]
- Lo, S.; Wu, C.; Shen, K.; Hsieh, M.; Lui, W. Higher Morbidity and Mortality After Combined Total Gastrectomy and Pancreaticosplenectomy for Gastric Cancer. World J. Surg. 2002, 26, 678–682. [Google Scholar] [CrossRef]
- Otsuji, E.; Yamaguchi, T.; Sawai, K.; Okamoto, K.; Takahashi, T. Total Gastrectomy with Simultaneous Pancreaticosplenectomy Or Splenectomy in Patients with Advanced Gastric Carcinoma. Br. J. Cancer 1999, 79, 1789–1793. [Google Scholar] [CrossRef]
- Shchepotin, I.B.; Chorny, V.A.; Nauta, R.J.; Shabahang, M.; Buras, R.R.; Evans, S.R. Extended Surgical Resection in T4 Gastric Cancer. Am. J. Surg. 1998, 175, 123–126. [Google Scholar] [CrossRef]
- Bonenkamp, J.; Hermans, J.; Sasako, M.; Welvaart, K.; Songun, I.; Meyer, S.; Plukker, J.; Van Elk, P.; Obertop, H.; Gouma, D. Extended Lymph-Node Dissection for Gastric Cancer. N. Engl. J. Med. 1999, 340, 908–914. [Google Scholar] [CrossRef]
- Weeden, S.; Cuschieri, A.; Fielding, J.; Bancewicz, J.; Craven, J.; Joypaul, V.; Sydes, M.; Fayers, P. Patient Survival After D1 and D2 Resections for Gastric Cancer: Long-Term Results of the UK Medical Research Council (MRC) Randomised Surgical Trial. Br. J. Cancer. 1999, 79, 1522–1530. [Google Scholar]
- Bonenkamp, J.; Songun, I.; Welvaart, K.; van de Velde, C.; Hermans, J.; Sasako, M.; Plukker, J.; van Elk, P.; Obertop, H.; Gouma, D. Randomised Comparison of Morbidity After D1 and D2 Dissection for Gastric Cancer in 996 Dutch Patients. Lancet 1995, 345, 745–748. [Google Scholar] [CrossRef]
- Seevaratnam, R.; Bocicariu, A.; Cardoso, R.; Mahar, A.; Kiss, A.; Helyer, L.; Law, C.; Coburn, N. A Meta-Analysis of D1 Versus D2 Lymph Node Dissection. Gastric Cancer 2012, 15, 60–69. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, M.; Baba, H.; Kakeji, Y.; Endo, K.; Ikeda, Y.; Toh, Y.; Kohnoe, S.; Okamura, T.; Maehara, Y. Postoperative morbidity/mortality and Survival Rates After Total Gastrectomy, with splenectomy/pancreaticosplenectomy for Patients with Advanced Gastric Cancer. Hepatogastroenterology 2004, 51, 298–302. [Google Scholar] [PubMed]
- Kasakura, Y.; Fujii, M.; Mochizuki, F.; Kochi, M.; Kaiga, T. Is there a Benefit of Pancreaticosplenectomy with Gastrectomy for Advanced Gastric Cancer? Am. J. Surg. 2000, 179, 237–242. [Google Scholar] [CrossRef]
- Elmessiry, M.M.; El-Fayoumi, T.A.; Fayed, H.M.; Gebaly, A.A.; Mohamed, E.A. Operative and Oncological Outcomes After D2 Versus D1 Gastrectomy of Operable Gastric Cancer: An Observational Study. J. Gastrointest. Cancer 2020. [Google Scholar] [CrossRef] [PubMed]
- Degiuli, M.; Reddavid, R.; Tomatis, M.; Ponti, A.; Morino, M.; Sasako, M.; Rebecchi, F.; Garino, M.; Vigano, L.; Scaglione, D. D2 Dissection Improves Disease-Specific Survival in Advanced Gastric Cancer Patients: 15-Year Follow-Up Results of the Italian Gastric Cancer Study Group D1 Versus D2 Randomised Controlled Trial. Eur. J. Cancer 2021, 150, 10–22. [Google Scholar] [CrossRef] [PubMed]
- Kung, C.; Tsai, J.; Lundell, L.; Johansson, J.; Nilsson, M.; Lindblad, M. Nationwide Study of the Impact of D2 Lymphadenectomy on Survival After Gastric Cancer Surgery. BJS Open 2020, 4, 424–431. [Google Scholar] [CrossRef]
- Oh, S.E.; Seo, J.E.; An, J.Y.; Choi, M.; Sohn, T.S.; Bae, J.M.; Kim, S.; Lee, J.H. Compliance with D2 Lymph Node Dissection in Reduced-Port Totally Laparoscopic Distal Gastrectomy in Patients with Gastric Cancer. Sci. Rep. 2021, 11, 3658. [Google Scholar] [CrossRef]
- Murakami, T. The General Rules for the Gastric Cancer Study in Surgery. Jpn. J. Surg. 1973, 3, 61–71. [Google Scholar]
- Faiz, Z.; Hayashi, T.; Yoshikawa, T. Lymph Node Dissection for Gastric Cancer: Establishment of D2 and the Current Position of Splenectomy in Europe and Japan. Eur. J. Surg. Oncol. 2021. [Google Scholar] [CrossRef]
- Sasako, M. Principles of Surgical Treatment for Curable Gastric Cancer. J. Clin. Oncol. 2003, 21, 274–275. [Google Scholar] [CrossRef] [PubMed]
- Hartgrink, H.; Van de Velde, C.; Putter, H.; Bonenkamp, J.; Meershoek-Klein Kranenbarg, E.; Songun, I.; Welvaart, K.; Van Krieken, J.; Meijer, S.; Plukker, J. Extended Lymph Node Dissection for Gastric Cancer: Who may Benefit? Final Results of the Randomized Dutch Gastric Cancer Group Trial. J. Clin. Oncol. 2004, 22, 2069–2077. [Google Scholar] [CrossRef] [PubMed]
- Songun, I.; Putter, H.; Kranenbarg, E.M.; Sasako, M.; van de Velde, C.J.H. Surgical Treatment of Gastric Cancer: 15-Year Follow-Up Results of the Randomised Nationwide Dutch D1D2 Trial. Lancet Oncol. 2010, 11, 439–449. [Google Scholar] [CrossRef]
- Cuschieri, A.; Weeden, S.; Fielding, J.; Bancewicz, J.; Craven, J.; Joypaul, V.; Sydes, M. Patient Survival After D 1 and D 2 Resections for Gastric Cancer: Long-Term Results of the MRC Randomized Surgical Trial. Br. J. Cancer 1999, 79, 1522–1530. [Google Scholar] [CrossRef] [PubMed]
- Degiuli, M.; Sasako, M.; Calgaro, M.; Garino, M.; Rebecchi, F.; Mineccia, M.; Scaglione, D.; Andreone, D.; Ponti, A.; Calvo, F. Morbidity and Mortality After D1 and D2 Gastrectomy for Cancer: Interim Analysis of the Italian Gastric Cancer Study Group (IGCSG) Randomised Surgical Trial. Eur. J. Surg. Oncol. (EJSO) 2004, 30, 303–308. [Google Scholar] [CrossRef] [PubMed]
- Yonemura, Y.; Wu, C.; Fukushima, N.; Honda, I.; Bandou, E.; Kawamura, T.; Kamata, T.; Kim, B.; Matsuki, N.; Sawa, T. Randomized Clinical Trial of D2 and Extended Paraaortic Lymphadenectomy in Patients with Gastric Cancer. Int. J. Clin. Oncol. 2008, 13, 132–137. [Google Scholar] [CrossRef] [PubMed]
- Mocellin, S.; McCulloch, P.; Kazi, H.; Gama-Rodrigues, J.J.; Yuan, Y.; Nitti, D. Extent of Lymph Node Dissection for Adenocarcinoma of the Stomach. Cochrane Database Syst. Rev. 2015. [Google Scholar] [CrossRef] [PubMed]
- Fujimura, T.; Nakamura, K.; Oyama, K.; Funaki, H.; Fujita, H.; Kinami, S.; Ninomiya, I.; Fushida, S.; Nishimura, G.; Kayahara, M. Selective Lymphadenectomy of Para-Aortic Lymph Nodes for Advanced Gastric Cancer. Oncol. Rep. 2009, 22, 509–514. [Google Scholar] [CrossRef] [PubMed]
- Schwarz, R.E.; Smith, D.D. Clinical Impact of Lymphadenectomy Extent in Resectable Gastric Cancer of Advanced Stage. Ann. Surg. Oncol. 2007, 14, 317–328. [Google Scholar] [CrossRef]
- Ye, J.; Ren, Y.; Dai, W.; Chen, J.; Cai, S.; Tan, M.; He, Y.; Yuan, Y. Does Lymphadenectomy with at Least 15 Perigastric Lymph Nodes Retrieval Promise an Improved Survival for Gastric Cancer: A Retrospective Cohort Study in Southern China. J. Cancer 2019, 10, 1444–1452. [Google Scholar] [CrossRef]
- Degiuli, M.; Sasako, M.; Ponti, A. Morbidity and Mortality in the Italian Gastric Cancer Study Group Randomized Clinical Trial of D1 Versus D2 Resection for Gastric Cancer. Br. J. Surg. 2010, 97, 643–649. [Google Scholar] [CrossRef]
- Saka, M.; Morita, S.; Fukagawa, T.; Katai, H. Present and Future Status of Gastric Cancer Surgery. Jpn. J. Clin. Oncol. 2011, 41, 307–313. [Google Scholar] [CrossRef]
- Jiang, Z.; Cai, Z.; Yin, Y.; Shen, C.; Huang, J.; Yin, Y.; Zhang, B. Impact of Surgical Margin Status on the Survival Outcome After Surgical Resection of Gastric Cancer: A Protocol for Systematic Review and Meta-Analysis. BMJ Open 2020, 10, e040282-2020-040282. [Google Scholar] [CrossRef] [PubMed]
- Sun, Z.; Li, D.; Wang, Z.; Huang, B.; Xu, Y.; Li, K.; Xu, H. Prognostic Significance of Microscopic Positive Margins for Gastric Cancer Patients with Potentially Curative Resection. Ann. Surg. Oncol. 2009, 16, 3028–3037. [Google Scholar] [CrossRef] [PubMed]
- Cho, B.C.; Jeung, H.C.; Choi, H.J.; Rha, S.Y.; Hyung, W.J.; Cheong, J.H.; Noh, S.H.; Chung, H.C. Prognostic Impact of Resection Margin Involvement After Extended (D2/D3) Gastrectomy for Advanced Gastric Cancer: A 15-year Experience at a Single Institute. J. Surg. Oncol. 2007, 95, 461–468. [Google Scholar] [CrossRef] [PubMed]
- Bissolati, M.; Desio, M.; Rosa, F.; Rausei, S.; Marrelli, D.; Baiocchi, G.L.; De Manzoni, G.; Chiari, D.; Guarneri, G.; Pacelli, F. Risk Factor Analysis for Involvement of Resection Margins in Gastric and Esophagogastric Junction Cancer: An Italian Multicenter Study. Gastric Cancer 2017, 20, 70–82. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.; Squires, M.H.; Poultsides, G.A.; Fields, R.C.; Weber, S.M.; Votanopoulos, K.I.; Kooby, D.A.; Worhunsky, D.J.; Jin, L.X.; Hawkins, W.G. Impact of Lymph Node Ratio in Selecting Patients with Resected Gastric Cancer for Adjuvant Therapy. Surgery 2017, 162, 285–294. [Google Scholar] [CrossRef] [PubMed]
- Schoenfeld, J.D.; Wo, J.Y.; Mamon, H.J.; Kwak, E.L.; Mullen, J.T.; Enzinger, P.Z.; Blaskowsky, L.S.; Ryan, D.P.; Hong, T.S. The Impact of Positive Margins on Outcome among Patients with Gastric Cancer Treated with Radiation. Am. J. Clin. Oncol. 2016, 39, 243–247. [Google Scholar] [CrossRef]
- Raziee, H.R.; Cardoso, R.; Seevaratnam, R.; Mahar, A.; Helyer, L.; Law, C.; Coburn, N. Systematic Review of the Predictors of Positive Margins in Gastric Cancer Surgery and the Effect on Survival. Gastric Cancer 2012, 15, 116–124. [Google Scholar] [CrossRef]
- Bickenbach, K.; Gonen, M.; Strong, V.; Brennan, M.; Coit, D. Association of Positive Transection Margins with Gastric Cancer Survival and Local Recurrence. Ann. Surg. Oncol. 2013, 20, 2663–2668. [Google Scholar] [CrossRef]
- Hasbahceci, M. Comment on “Dealing with the Gray Zones in the Management of Gastric Cancer: The Consensus Statement of the Istanbul Group”. Turk. J. Gastroenterol. 2020, 31, 838–839. [Google Scholar] [CrossRef] [PubMed]
- Ramos, M.F.K.P.; Pereira, M.A.; Yagi, O.K.; Dias, A.R.; Charruf, A.Z.; Oliveira, R.J.d.; Zaidan, E.P.; Zilberstein, B.; Ribeiro-Júnior, U.; Cecconello, I. Surgical Treatment of Gastric Cancer: A 10-Year Experience in a High-Volume University Hospital. Clinics 2018, 73. [Google Scholar] [CrossRef] [PubMed]
- Hansson, L.E.; Sparen, P.; Nyren, O. Survival in Stomach Cancer is Improving: Results of a Nationwide Population-Based Swedish Study. Ann. Surg. 1999, 230, 162–169. [Google Scholar] [CrossRef] [PubMed]
- Finlayson, E.V.; Goodney, P.P.; Birkmeyer, J.D. Hospital Volume and Operative Mortality in Cancer Surgery: A National Study. Arch. Surg. 2003, 138, 721–725. [Google Scholar] [CrossRef]
- Wainess, R.M.; Dimick, J.B.; Upchurch, G.R.; Cowan, J.A.; Mulholland, M.W. Epidemiology of Surgically Treated Gastric Cancer in the United States, 1988–2000. J. Gastrointest. Surg. 2003, 7, 879–883. [Google Scholar]
- Hundahl, S.A.; Macdonald, J.S.; Benedetti, J.; Fitzsimmons, T. Surgical Treatment Variation in a Prospective, Randomized Trial of Chemoradiotherapy in Gastric Cancer: The Effect of Undertreatment. Ann. Surg. Oncol. 2002, 9, 278–286. [Google Scholar] [CrossRef] [PubMed]
- Baxter, N.N.; Tuttle, T.M. Inadequacy of Lymph Node Staging in Gastric Cancer Patients: A Population-Based Study. Ann. Surg. Oncol. 2005, 12, 981–987. [Google Scholar] [CrossRef] [PubMed]
- Bilimoria, K.Y.; Talamonti, M.S.; Wayne, J.D.; Tomlinson, J.S.; Stewart, A.K.; Winchester, D.P.; Ko, C.Y.; Bentrem, D.J. Effect of Hospital Type and Volume on Lymph Node Evaluation for Gastric and Pancreatic Cancer. Arch. Surg. 2008, 143, 671–678. [Google Scholar] [CrossRef]
- Kampschöer, G.; Maruyama, K.; Van de Velde, C.; Sasako, M.; Kinoshita, T.; Okabayashi, K. Computer Analysis in Making Preoperative Decisions: A Rational Approach to Lymph Node Dissection in Gastric Cancer Patients. J. Br. Surg. 1989, 76, 905–908. [Google Scholar] [CrossRef]
- Bollschweiler, E.; Boettcher, K.; Hoelscher, A.; Sasako, M.; Kinoshita, T.; Maruyama, K.; Siewert, J. Preoperative Assessment of Lymph Node Metastases in Patients with Gastric Cancer: Evaluation of the Maruyama Computer Program. J. Br. Surg. 1992, 79, 156–160. [Google Scholar] [CrossRef]
- Mahar, A.L.; McLeod, R.S.; Kiss, A.; Paszat, L.; Coburn, N.G. A Systematic Review of the Effect of Institution and Surgeon Factors on Surgical Outcomes for Gastric Cancer. J. Am. Coll. Surg. 2012, 214, 860–868.e12. [Google Scholar] [CrossRef] [PubMed]
- Wanebo, H.J.; Kennedy, B.J.; Chmiel, J.; Steele, G., Jr.; Winchester, D.; Osteen, R. Cancer of the Stomach. A Patient Care Study by the American College of Surgeons. Ann. Surg. 1993, 218, 583–592. [Google Scholar] [CrossRef]
- Hoshino, N.; Murakami, K.; Hida, K.; Hisamori, S.; Tsunoda, S.; Obama, K.; Sakai, Y. Robotic Versus Laparoscopic Surgery for Gastric Cancer: An Overview of Systematic Reviews with Quality Assessment of Current Evidence. Updates Surg. 2020, 72, 573–582. [Google Scholar] [CrossRef]
- Son, S.Y.; Kim, H.H. Minimally Invasive Surgery in Gastric Cancer. World J. Gastroenterol. 2014, 20, 14132–14141. [Google Scholar] [CrossRef]
- Hyung, W.J.; Yang, H.; Park, Y.; Lee, H.; An, J.Y.; Kim, W.; Kim, H.; Kim, H.; Ryu, S.W.; Hur, H. Long-Term Outcomes of Laparoscopic Distal Gastrectomy for Locally Advanced Gastric Cancer: The KLASS-02-RCT Randomized Clinical Trial. J. Clin. Oncol. 2020, 38, 3304–3313. [Google Scholar] [CrossRef]
- Kim, H.; Hyung, W.J.; Cho, G.S.; Kim, M.C.; Han, S.; Kim, W.; Ryu, S.; Lee, H.; Song, K.Y. Morbidity and Mortality of Laparoscopic Gastrectomy Versus Open Gastrectomy for Gastric Cancer: An Interim report—A Phase III Multicenter, Prospective, Randomized Trial (KLASS Trial). Ann. Surg. 2010, 251, 417–420. [Google Scholar] [CrossRef]
- Katai, H.; Sasako, M.; Fukuda, H.; Nakamura, K.; Hiki, N.; Saka, M.; Yamaue, H.; Yoshikawa, T.; Kojima, K. Safety and Feasibility of Laparoscopy-Assisted Distal Gastrectomy with Suprapancreatic Nodal Dissection for Clinical Stage I Gastric Cancer: A Multicenter Phase II Trial (JCOG 0703). Gastric Cancer 2010, 13, 238–244. [Google Scholar] [CrossRef]
- Honda, M.; Hiki, N.; Kinoshita, T.; Yabusaki, H.; Abe, T.; Nunobe, S.; Terada, M.; Matsuki, A.; Sunagawa, H.; Aizawa, M.; et al. Long-Term Outcomes of Laparoscopic Versus Open Surgery for Clinical Stage I Gastric Cancer: The LOC-1 Study. Ann. Surg. 2016, 264, 214–222. [Google Scholar] [CrossRef]
- Kim, H.; Han, S.; Kim, M.; Kim, W.; Lee, H.; Ryu, S.W.; Cho, G.S.; Kim, C.Y.; Yang, H.; Park, D.J. Effect of Laparoscopic Distal Gastrectomy Vs Open Distal Gastrectomy on Long-Term Survival among Patients with Stage I Gastric Cancer: The KLASS-01 Randomized Clinical Trial. JAMA Oncol. 2019, 5, 506–513. [Google Scholar] [CrossRef] [PubMed]
- Uyama, I.; Suda, K.; Satoh, S. Laparoscopic Surgery for Advanced Gastric Cancer: Current Status and Future Perspectives. J. Gastric Cancer 2013, 13, 19–25. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.; Hu, Y.; Chen, T.; Mou, T.; Cheng, X.; Li, G. Laparoscopic Distal Gastrectomy with D2 Dissection for Advanced Gastric Cancer. Chin. J. Cancer Res. 2013, 25, 474–476. [Google Scholar]
- Caruso, S.; Patriti, A.; Roviello, F.; De Franco, L.; Franceschini, F.; Coratti, A.; Ceccarelli, G. Laparoscopic and Robot-Assisted Gastrectomy for Gastric Cancer: Current Considerations. World J. Gastroenterol. 2016, 22, 5694–5717. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mocan, L. Surgical Management of Gastric Cancer: A Systematic Review. J. Clin. Med. 2021, 10, 2557. https://doi.org/10.3390/jcm10122557
Mocan L. Surgical Management of Gastric Cancer: A Systematic Review. Journal of Clinical Medicine. 2021; 10(12):2557. https://doi.org/10.3390/jcm10122557
Chicago/Turabian StyleMocan, Lucian. 2021. "Surgical Management of Gastric Cancer: A Systematic Review" Journal of Clinical Medicine 10, no. 12: 2557. https://doi.org/10.3390/jcm10122557
APA StyleMocan, L. (2021). Surgical Management of Gastric Cancer: A Systematic Review. Journal of Clinical Medicine, 10(12), 2557. https://doi.org/10.3390/jcm10122557




