Advances in Intestinal Barrier Preservation and Restoration in the Allogeneic Hematopoietic Cell Transplantation Setting
Abstract
1. Introduction
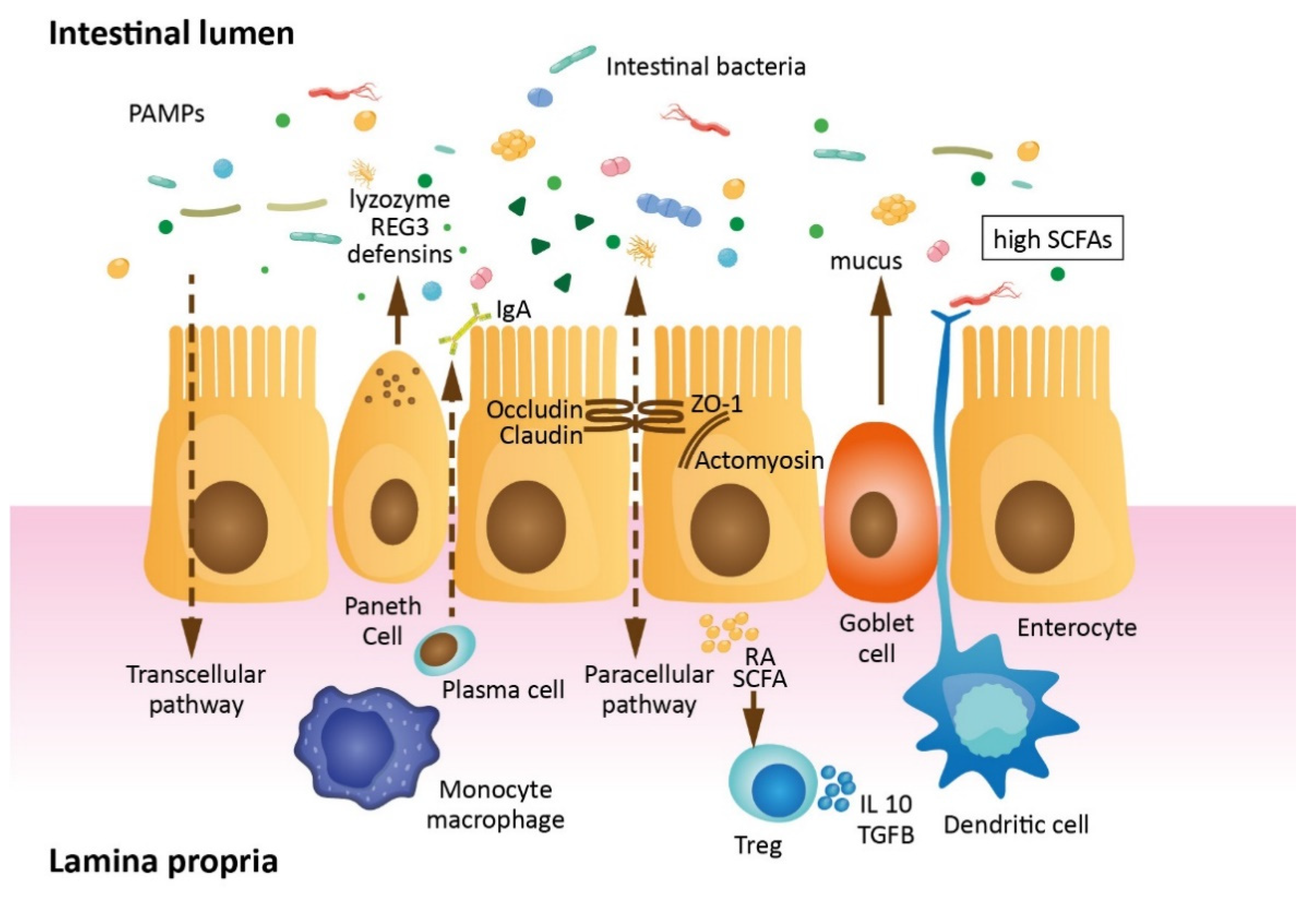
1.1. Intestinal Barrier Anatomy and Physiology
1.2. Gut Barrier Permeability
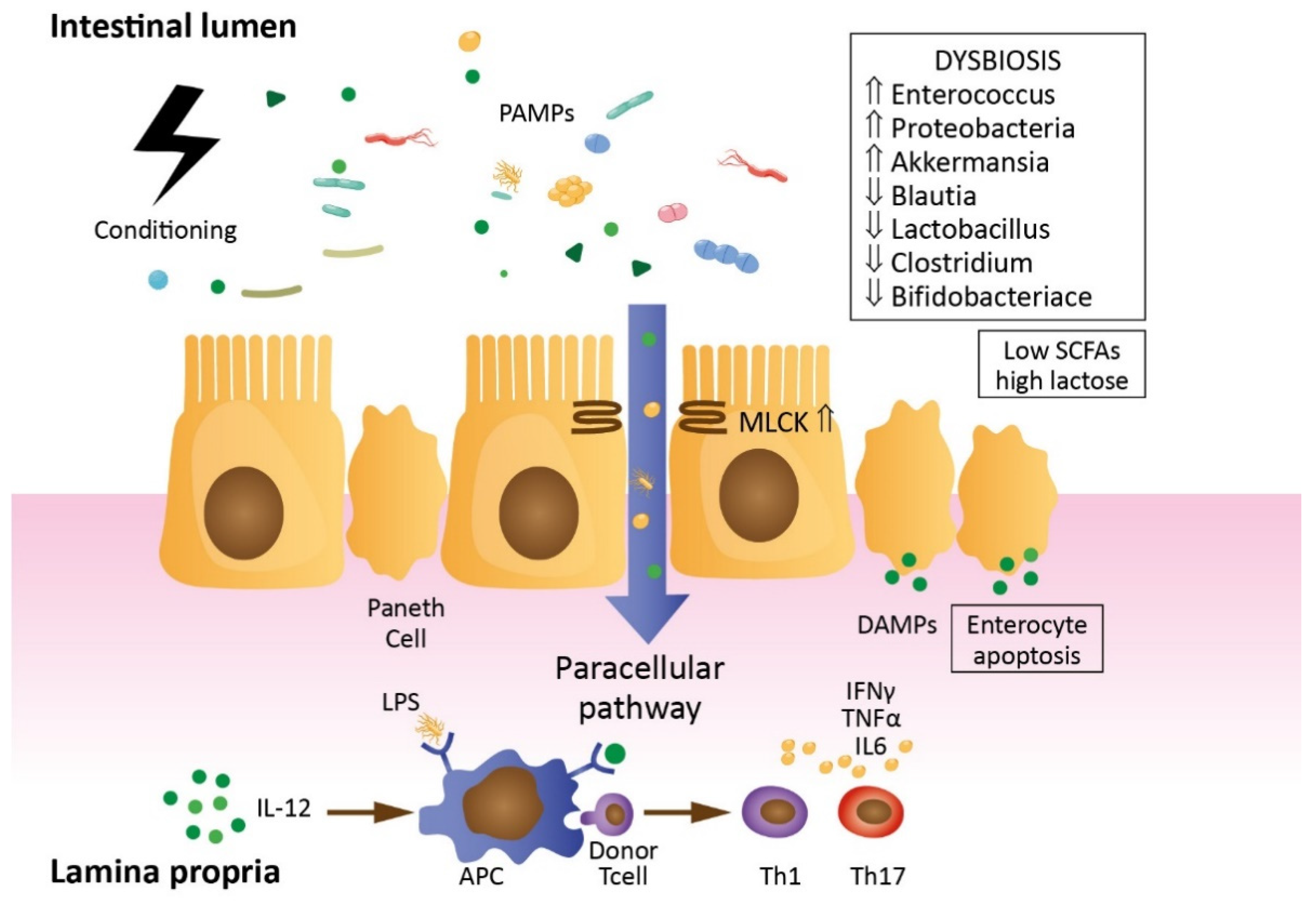
2. Impact of allo-HCT Procedure on Gut Barrier Permeability (Mechanisms and Summary of Studies)
3. Gut Barrier Preservation and Restoration in allo-HCT
3.1. Vitamin A
3.2. MLCK Inhibition
3.3. Indole Derivatives
3.4. Alloantigen Presentation Inhibition in the Gut via IL-12 Neutralization
3.5. Intestinal REG3
3.6. Butyrate
3.7. Prebiotic Use
3.8. Lactose-Free Diet
3.9. Fecal Microbiota Transplantation
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Cooke, K.R.; Gerbitz, A.; Crawford, J.M.; Teshima, T.; Hill, G.R.; Tesolin, A.; Rossignol, D.P.; Ferrara, J.L. LPS antagonism reduces graft-versus-host disease and preserves graft-versus-leukemia activity after experimental bone marrow transplantation. J. Clin. Investig. 2001, 107, 1581–1589. [Google Scholar] [CrossRef] [PubMed]
- Peled, J.U.; Gomes, A.L.; Devlin, S.M.; Littmann, E.R.; Taur, Y.; Sung, A.D.; Weber, D.; Hashimoto, D.; Slingerland, A.E.; Slingerland, J.B.; et al. Microbiota as Predictor of Mortality in Allogeneic Hematopoietic-Cell Transplantation. N. Engl. J. Med. 2020, 382, 822–834. [Google Scholar] [CrossRef]
- Scaldaferri, F.; Pizzoferrato, M.; Gerardi, V.; Lopetuso, L.; Gasbarrini, A. The gut barrier: New acquisitions and therapeutic approaches. J. Clin. Gastroenterol. 2012, 46, S12–S17. [Google Scholar] [CrossRef] [PubMed]
- Vancamelbeke, M.; Vermeire, S. The intestinal barrier: A fundamental role in health and disease. Expert Rev. Gastroenterol. Hepatol. 2017, 11, 821–834. [Google Scholar] [CrossRef]
- Hartsock, A.; Nelson, W.J. Adherens and tight junctions: Structure, function and connections to the actin cytoskeleton. Biochim. Biophys. Acta 2008, 1778, 660–669. [Google Scholar] [CrossRef] [PubMed]
- Ungewiß, H.; Vielmuth, F.; Suzuki, S.T.; Maiser, A.; Harz, H.; Leonhardt, H.; Kugelmann, D.; Schlegel, N.; Waschke, J. Desmoglein 2 regulates the intestinal epithelial barrier via p38 mitogen-activated protein kinase. Sci. Rep. 2017, 7, 6329. [Google Scholar] [CrossRef] [PubMed]
- Romero, E.S.; Cotoner, C.A.; Camacho, C.P.; Bedmar, M.C.; Vicario, M. The intestinal barrier function and its involvement in digestive disease. Rev. Esp. Enferm. Dig. 2015, 107, 686–696. [Google Scholar]
- Poyet, M.; Groussin, M.; Gibbons, S.M.; Avila-Pacheco, J.; Jiang, X.; Kearney, S.M.; Perrotta, A.R.; Berdy, B.; Zhao, S.; Lieberman, T.D.; et al. A library of human gut bacterial isolates paired with longitudinal multiomics data enables mechanistic microbiome research. Nat. Med. 2019, 25, 1442–1452. [Google Scholar] [CrossRef]
- Muniz, L.R.; Knosp, C.; Yeretssian, G. Intestinal antimicrobial peptides during homeostasis, infection, and disease. Front. Immunol. 2012, 3, 310. [Google Scholar] [CrossRef]
- Sorbara, M.T.; Pamer, E.G. Interbacterial mechanisms of colonization resistance and the strategies pathogens use to overcome them. Mucosal Immunol. 2019, 12, 1–9. [Google Scholar] [CrossRef]
- Bischoff, S.C.; Barbara, G.; Buurman, W.; Ockhuizen, T.; Schulzke, J.D.; Serino, M.; Tilg, H.; Watson, A.; Wells, J.M. Intestinal permeability—A new target for disease prevention and therapy. BMC Gastroenterol. 2014, 14, 189. [Google Scholar] [CrossRef]
- Furusawa, Y.; Obata, Y.; Fukuda, S.; Endo, T.A.; Nakato, G.; Takahashi, D.; Nakanishi, Y.; Uetake, C.; Kato, K.; Kato, T.; et al. Commensal microbe-derived butyrate induces the differentiation of colonic regulatory T cells. Nature 2013, 504, 446–450. [Google Scholar] [CrossRef] [PubMed]
- Belkaid, Y.; Harrison, O.J. Homeostatic Immunity and the Microbiota. Immunity 2017, 46, 562–576, ISSN 1074-7613. [Google Scholar] [CrossRef]
- Gordon, J.; M’Leod, J.W. Inhibition of bacterial growth by some amino acids and its bearing on the use of tryptic digests as culture media. J. Pathol. Bacteriol. 1926, 29, 13–25. [Google Scholar] [CrossRef]
- Zelante, T.; Iannitti, R.G.; Cunha, C.; De Luca, A.; Giovannini, G.; Pieraccini, G.; Zecchi, R.; D’Angelo, C.; Massi-Benedetti, C.; Fallarino, F.; et al. Tryptophan catabolites from microbiota engage aryl hydrocarbon receptor and balance mucosal reactivity via interleukin-22. Immunity 2013, 39, 372–385. [Google Scholar] [CrossRef]
- Bansal, T.; Alaniz, R.C.; Wood, T.K.; Jayaraman, A. The bacterial signal indole increases epithelial-cell tight-junction resistance and attenuates indicators of inflammation. Proc. Natl. Acad. Sci. USA 2010, 107, 228–233. [Google Scholar] [CrossRef]
- Menard, S.; Cerf-Bensussan, N.; Heyman, M. Multiple facets of intestinal permeability and epithelial handling of dietary antigens. Mucosal Immunol. 2010, 3, 247–259. [Google Scholar] [CrossRef] [PubMed]
- Rapoport, A.P.; Watelet, L.F.M.; Linder, T.; Rberly, S.; Raubertas, R.F.; Lipp, J.; Duerst, R.; Abboud, C.N.; Constine, L.; Andrews, J.; et al. Analysis of factors that correlate with mucositis in recipients of autologous and allogeneic stem-cell transplants. J. Clin. Oncol. 1999, 17, 2446–2453. [Google Scholar] [CrossRef]
- Johansson, J.E.; Ekman, T. Gastro-intestinal toxicity related to bone marrow transplantation: Disruption of the intestinal barrier precedes clinical findings. Bone Marrow Transplant. 1997, 19, 921–925. [Google Scholar] [CrossRef]
- Johansson, J.E.; Brune, M.; Ekman, T. The gut mucosa barrier is preserved during allogeneic, haemopoietic stem cell transplantation with reduced intensity conditioning. Bone Marrow Transplant. 2001, 28, 737–742. [Google Scholar] [CrossRef] [PubMed]
- Blijlevens, N.M.A.; Donnelly, J.P.; M’Rabet, L.; De Pauw, B.E.; Land, B.V. Measuring mucosal damage induced by cytotoxic therapy. Support. Care Cancer 2004, 12, 227–233. [Google Scholar] [CrossRef] [PubMed]
- Blijlevens, N.; Donnelly, J.; de Pauw, B. Prospective evaluation of gut mucosal barrier injury following various myeloablative regimens for haematopoietic stem cell transplant. Bone Marrow Transplant. 2005, 35, 707–711. [Google Scholar] [CrossRef]
- Johansson, J.E.; Ekman, T. Gut toxicity during hemopoietic stem cell transplantation may predict acute graft-versus-host disease severity in patients. Dig. Dis. Sci. 2007, 52, 2340–2345. [Google Scholar] [CrossRef] [PubMed]
- Bates, D.W.; Parsonnet, J.; Ketchum, P.A.; Miller, E.B.; Novitsky, T.J.; Sands, K.; Hibberd, P.L.; Graman, P.S.; Lanken, P.N.; Schwartz, J.S.; et al. Limulus amebocyte lysate assay for detection of endotoxin in patients with sepsis syndrome: AMCC sepsis project working group. Clin. Infect. Dis. 1998, 27, 582–591. [Google Scholar] [CrossRef]
- Strutz, F.; Heller, G.; Krasemann, K.; Krone, B.; Müller, G.A. Relationship of antibodies to endotoxin core to mortality in medical patients with sepsis syndrome. Intensive Care Med. 1999, 25, 435–444. [Google Scholar] [CrossRef]
- Blijlevens, N.M.; Lutgens, L.C.H.W.; Schattenberg, A.V.M.B.; Donnelly, J.P. Citrulline: A potentially simple quantitative marker of intestinal epithelial damage following myeloablative therapy. Bone Marrow Transplant. 2004, 34, 193–196. [Google Scholar] [CrossRef] [PubMed]
- Crenn, P.; Coudray-Lucas, C.; Thuillier, F.; Cynober, L.; Messing, B. Postabsorptive plasma citrulline concentration is a marker of absorptive enterocyte mass and intestinal failure in humans. Gastroenterology 2000, 119, 1496–1505. [Google Scholar] [CrossRef]
- Vreugdenhil, A.C.; Wolters, V.M.; Adriaanse, M.P.; van den Neucker, A.M.; van Bijnen, A.A.; Houwen, R.; Buurman, W.A. Additional value of serum I-FABP levels for evaluating celiac disease activity in children. Scand. J. Gastroenterol. 2011, 46, 1435–1441. [Google Scholar] [CrossRef]
- Prasad, S.; Mingrino, R.; Kaukinen, K.; Hayes, K.; Powell, R.; MacDonald, T.; Collins, J. Inflammatory processes have differential effects on claudins 2, 3 and 4 in colonic epithelial cells. Lab. Investig. 2005, 85, 1139–1162. [Google Scholar] [CrossRef]
- Blijlevens, N.M.; Donnelly, J.P.; DePauw, B.E. Inflammatory response to mucosal barrier injury after myeloablative therapy in allogeneic stem cell transplant recipients. Bone Marrow Transplant. 2005, 36, 703–707. [Google Scholar] [CrossRef][Green Version]
- Hill, G.R.; Crawford, J.M.; Cooke, K.R.; Brinson, Y.S.; Pan, L.; Ferrara, J.L. Total body irradiation and acute graft-versus-host disease: The role of gastrointestinal damage and inflammatory cytokines. Blood 1997, 90, 3204–3213. [Google Scholar] [CrossRef]
- Hill, G.R.; Ferrara, J.L. The primacy of the gastrointestinal tract as a target organ of acute graft-versus-host disease: Rationale for the use of cytokine shields in allogeneic bone marrow transplantation. Blood 2000, 95, 2754–2759. [Google Scholar] [CrossRef] [PubMed]
- Nalle, S.C.; Turner, J.R. Endothelial and epithelial barriers in graft-versus-host disease. Adv. Exp. Med. Biol. 2012, 763, 105–131. [Google Scholar] [PubMed]
- Noth, R.; Lange-Grumfeld, J.; Stüber, E.; Kruse, M.-L.; Ellrichmann, M.; Häsler, R.; Hampe, J.; Bewig, B.; Rosenstiel, P.; Schreiber, S.; et al. Increased intestinal permeability and tight junction disruption by altered expression and localization of occludin in a murine graft versus host disease model. BMC Gastroenterol. 2011, 11, 109. [Google Scholar] [CrossRef] [PubMed]
- Nalle, S.C.; Kwak, H.A.; Edelblum, K.L.; Joseph, N.E.; Singh, G.; Khramtsova, G.F.; Mortenson, E.D.; Savage, P.A.; Turner, J.R. Recipient NK cell inactivation and intestinal barrier loss are required for MHC-matched graft-versus-host disease. Sci. Transl. Med. 2014, 6, 243ra87. [Google Scholar] [CrossRef]
- Penack, O.; Holler, E.; van den Brink, M.R. Graft-versus-host disease: Regulation by microbe-associated molecules and innate immune receptors. Blood 2010, 115, 1865–1872. [Google Scholar] [CrossRef]
- Lounder, D.T.; Khandelwal, P.; Dandoy, C.; Jodele, S.; Grimley, M.S.; Wallace, G.; Lane, A.; Taggart, C.; Teusink-Cross, A.C.; Lake, K.E.; et al. Lower levels of vitamin A are associated with increased gastrointestinal graft-versus-host disease in children. Blood 2017, 129, 2801–2807. [Google Scholar] [CrossRef] [PubMed]
- Qiang, Y.; Xu, J.; Yan, C.; Jin, H.; Xiao, T.; Yan, N.; Zhou, L.; An, H.; Zhou, X.; Shao, Q.; et al. Butyrate and retinoic acid imprint mucosal-like dendritic cell development synergistically from bone marrow cells. Clin. Exp. Immunol. 2017, 189, 290–297. [Google Scholar] [CrossRef]
- Martin, J.C.; Bériou, G.; Heslan, M.; Bossard, C.; Jarry, A.; Abidi, A.; Hulin, P.; Ménoret, S.; Thinard, R.; Anegon, I.; et al. IL-22BP is produced by eosinophils in human gut and blocks IL-22 protective actions during colitis. Mucosal Immunol. 2016, 9, 539–549. [Google Scholar] [CrossRef] [PubMed]
- Rossi, J.L.; Velentza, A.V.; Steinhorn, D.M.; Watterson, D.M.; Wainwright, M.S. MLCK210 gene knockout or kinase inhibition preserves lung function following endotoxin-induced lung injury in mice. Am. J. Physiol. Lung Cell Mol. Physiol. 2007, 292, L1327–L1334. [Google Scholar] [CrossRef]
- Parker, J. Inhibitors of myosin light chain kinase and phosphodiesterase reduce ventilator-induced lung injury. J. Appl. Physiol. 2000, 89, 2241–2248. [Google Scholar] [CrossRef]
- Nalle, S.C.; Zuo, L.; Ong, M.L.D.M.; Singh, G.; Worthylake, A.M.; Choi, W.; Manresa, M.C.; Southworth, A.P.; Edelblum, K.L.; Baker, G.J.; et al. Graft-versus-host disease propagation depends on increased intestinal epithelial tight junction permeability. J. Clin. Investig. 2019, 129, 902–914. [Google Scholar] [CrossRef] [PubMed]
- Swimm, A.; Giver, C.R.; DeFilipp, Z.; Rangaraju, S.; Sharma, A.; Antonova, A.U.; Sonowal, R.; Capaldo, C.; Powell, D.; Qayed, M.; et al. Indoles derived from intestinal microbiota act via type I interferon signaling to limit graft-versus-host disease. Blood 2018, 132, 2506–2519. [Google Scholar] [CrossRef]
- Londei, M.; Lamb, J.R.; Bottazzo, G.F.; Feldmann, M. Epithelial cells expressing aberrant MHC class II determinants can present antigen to cloned human T cells. Nature 1984, 312, 639–641. [Google Scholar] [CrossRef] [PubMed]
- McDonald, G.B.; Jewell, D.P. Class II antigen (HLA-DR) expression by intestinal epithelial cells in inflammatory diseases of colon. J. Clin. Pathol. 1987, 40, 312–317. [Google Scholar] [CrossRef] [PubMed]
- Koyama, M.; Mukhopadhyay, P.; Schuster, I.S.; Henden, A.S.; Hülsdünker, J.; Varelias, A.; Vetizou, M.; Kuns, R.D.; Robb, R.J.; Zhang, P.; et al. MHC Class II Antigen Presentation by the Intestinal Epithelium Initiates Graft-versus-Host Disease and Is Influenced by the Microbiota. Immun. 2019, 51, 885–898.e7. [Google Scholar] [CrossRef] [PubMed]
- Pidala, J.; Beato, F.; Kim, J.; Betts, B.; Jim, H.; Sagatys, E.; Levine, J.E.; Ferrara, J.L.; Ozbek, U.; Ayala, E.; et al. In vivo IL-12/IL-23p40 neutralization blocks Th1/Th17 response after allogeneic hematopoietic cell transplantation. Haematol. 2017, 103, 531–539. [Google Scholar] [CrossRef]
- Zhao, D.; Kim, Y.H.; Jeong, S.; Greenson, J.K.; Chaudhry, M.S.; Hoepting, M.; Anderson, E.R.; van den Brink, M.R.; Peled, J.U.; Gomes, A.L.; et al. Survival signal REG3α prevents crypt apoptosis to control acute gastrointestinal graft-versus-host disease. J. Clin. Investig. 2018, 128, 4970–4979. [Google Scholar] [CrossRef] [PubMed]
- Hanash, A.M.; Dudakov, J.A.; Hua, G.; O’Connor, M.H.; Young, L.F.; Singer, N.V.; West, M.L.; Jenq, R.R.; Holland, A.M.; Kappel, L.W.; et al. Interleukin-22 protects intestinal stem cells from immune-mediated tissue damage and regulates sensitivity to graft versus host disease. Immunity 2012, 37, 339–350. [Google Scholar] [CrossRef]
- Bajic, D.; Niemann, A.; Hillmer, A.K.; Mejias-Luque, R.; Bluemel, S.; Docampo, M.; Funk, M.C.; Tonin, E.; Boutros, M.; Schnabl, B.; et al. Gut microbiota-derived propionate regulates the expression of reg3 mucosal lectins and ameliorates experimental colitis in mice. J. Crohns Colitis 2020, 14, 1462–1472. [Google Scholar] [CrossRef]
- Ferrara, J.L.M.; Harris, A.C.; Greenson, J.K.; Braun, T.M.; Holler, E.; Teshima, T.; Levine, J.E.; Choi, S.W.J.; Huber, E.; Landfried, K.; et al. Regenerating islet-derived 3-alpha is a biomarker of gastrointestinal graft-versus-host disease. Blood 2011, 118, 6702–6708. [Google Scholar] [CrossRef] [PubMed]
- Levine, J.E.; Huber, E.; Hammer, S.T.; Harris, A.C.; Greenson, J.K.; Braun, T.M.; Ferrara, J.L.; Holler, E. Low Paneth cell numbers at onset of gastrointestinal graft-versus-host disease identify patients at high risk for nonrelapse mortality. Blood 2013, 122, 1505–1509. [Google Scholar] [CrossRef]
- Brandl, K.; Plitas, G.; Mihu, C.N.; Ubeda, C.; Jia, T.; Fleisher, M.; Schnabl, B.; DeMatteo, R.P.; Pamer, E.G. Vancomycin-resistant enterococci exploit antibiotic-induced innate immune deficits. Nature 2008, 455, 804–807. [Google Scholar] [CrossRef]
- Mathewson, N.D.; Jenq, R.; Mathew, A.; Koenigsknecht, M.; Hanash, A.; Toubai, T.; Oravecz-Wilson, K.; Wu, S.-R.; Sun, Y.; Rossi, C.; et al. Gut microbiome–derived metabolites modulate intestinal epithelial cell damage and mitigate graft-versus-host disease. Nat. Immunol. 2016, 17, 505–513. [Google Scholar] [CrossRef]
- Markey, K.A.; Schluter, J.; Gomes, A.L.C.; Littmann, E.R.; Pickard, A.J.; Taylor, B.P.; Giardina, P.A.; Weber, D.; Dai, A.; Docampo, M.D.; et al. The microbe-derived short-chain fatty acids butyrate and propionate are associated with protection from chronic GVHD. Blood 2020, 136, 130–136. [Google Scholar] [CrossRef] [PubMed]
- Haak, B.W.; Littmann, E.R.; Chaubard, J.-L.; Pickard, A.; Fontana, E.; Adhi, F.; Gyaltshen, Y.; Ling, L.; Morjaria, S.M.; Peled, J.U.; et al. Impact of gut colonization with butyrate producing microbiota on respiratory viral infection following allo-HCT. Blood 2018, 131, 2978–2986. [Google Scholar] [CrossRef]
- Galloway-Peña, J.R.; Peterson, C.B.; Malik, F.; Sahasrabhojane, P.V.; Shah, D.P.; E Brumlow, C.; Carlin, L.G.; Chemaly, R.F.; Im, J.S.; Rondon, G.; et al. Fecal Microbiome, Metabolites, and Stem Cell Transplant Outcomes: A Single-Center Pilot Study. Open Forum Infect. Dis. 2019, 6, 173. [Google Scholar] [CrossRef]
- Yoshifuji, K.; Inamoto, K.; Kiridoshi, Y.; Takeshita, K.; Sasajima, S.; Shiraishi, Y.; Yamashita, Y.; Nisaka, Y.; Ogura, Y.; Takeuchi, R.; et al. Prebiotics protect against acute graft-versus-host disease and preserve the gut microbiota in stem cell transplantation. Blood Adv. 2020, 4, 4607–4617. [Google Scholar] [CrossRef]
- Holler, E.; Butzhammer, P.; Schmid, K.; Hundsrucker, C.; Koestler, J.; Peter, K.; Zhu, W.; Sporrer, D.; Hehlgans, T.; Kreutz, M.; et al. Metagenomic analysis of the stool microbiome in patients receiving allogeneic stem cell transplantation: Loss of diversity is associated with use of systemic antibiotics and more pronounced in gastrointestinal graft-versus-host disease. Biol. Blood Marrow Transplant. 2014, 20, 640–645. [Google Scholar] [CrossRef] [PubMed]
- Taur, Y.; Jenq, R.; Perales, M.-A.; Littmann, E.R.; Morjaria, S.; Ling, L.; No, D.; Gobourne, A.; Viale, A.; Dahi, P.; et al. The effects of intestinal tract bacterial diversity on mortality following allogeneic hematopoietic stem cell transplantation. Blood 2014, 124, 1174–1182. [Google Scholar] [CrossRef]
- Montassier, E.; Batard, E.; Massart, S.; Gastinne, T.; Carton, T.; Caillon, J.; Le Fresne, S.; Caroff, N.; Hardouin, J.B.; Moreau, P.; et al. 16S rRNA Gene Pyrosequencing Reveals Shift in Patient Faecal Microbiota During High-Dose Chemotherapy as Conditioning Regimen for Bone Marrow Transplantation. Microb. Ecol. 2014, 67, 690–699. [Google Scholar] [CrossRef] [PubMed]
- Weber, D.; Oefner, P.J.; Hiergeist, A.; Koestler, J.; Gessner, A.; Weber, M.; Hahn, J.; Wolff, D.; Stämmler, F.; Spang, R.; et al. Low urinary indoxyl sulfate levels early after transplantation reflect a disrupted microbiome and are associated with poor outcome. Blood 2015, 126, 1723–1728. [Google Scholar] [CrossRef] [PubMed]
- Stein-Thoeringer, C.K.; Nichols, K.B.; Lazrak, A.; Docampo, M.D.; Slingerland, A.E.; Slingerland, J.B.; Clurman, A.G.; Armijo, G.; Gomes, A.L.C.; Shono, Y.; et al. Lactose drives Enterococcus expansion to promote graft-versus-host disease. Science 2019, 366, 1143–1149. [Google Scholar] [CrossRef]
- Bilinski, J.; Grzesiowski, P.; Sorensen, N.; Madry, K.; Muszynski, J.; Robak, K.; Wroblewska, M.; Dzieciatkowski, T.; Dulny, G.; Dwilewicz-Trojaczek, J.; et al. Fecal microbiota transplantation in patients with blood disorders inhibits gut colonization with antibiotic-resistant bacteria: Results of a prospective, single-center study. Clin. Infect. Dis. 2017, 65, 364–370. [Google Scholar] [CrossRef] [PubMed]
- Battipaglia, G.; Malard, F.; Rubio, M.T.; Ruggeri, A.; Mamez, A.C.; Brissot, E.; Giannotti, F.; Dulery, R.; Joly, A.C.; Baylatry, M.T.; et al. Fecal microbiota transplantation before or after allogeneic hematopoietic transplantation in patients with hematologic malignancies carrying multidrug-resistance bacteria. Haematologica 2019, 104, 1682–1688. [Google Scholar] [CrossRef] [PubMed]
- Mohty, M.; Malard, F.; Vekhoff, A.; Lapusan, S.; Isnard, F.; D’Incan, E.; Rey, J.; Saillard, C.; Thomas, X.; Ducastelle-Lepretre, S.; et al. The Odyssee study: Prevention of dysbiosis complications with autologous Fecal Microbiota Transfer (FMT) in Acute Myeloid Leukemia (AML) patients undergoing intensive treatment: Results of a prospective multicenter trial. Blood 2018, 132 (Suppl. S1), 1444. [Google Scholar] [CrossRef]
- Webb, B.J.; Brunner, A.; Ford, C.D.; Gazdik, M.A.; Petersen, F.B.; Hoda, D. Fecal microbiota transplantation for recurrent Clostridium difficile infection in hematopoietic stem cell transplant recipients. Transpl. Infect. Dis. 2016, 18, 628–633. [Google Scholar] [CrossRef]
- Moss, E.L.; Falconer, S.B.; Tkachenko, E.; Wang, M.; Systrom, H.; Mahabamunuge, J.; Relman, D.A.; Hohmann, E.L.; Bhatt, A.S. Long-term taxonomic and functional divergence from donor bacterial strains following fecal microbiota transplantation in immunocompromised patients. PLoS ONE 2017, 12, e0182585. [Google Scholar] [CrossRef]
- Mao, D.; Jiang, Q.; Sun, Y.; Mao, Y.; Guo, L.; Zhang, Y.; Man, M.; Ouyang, G.; Sheng, L. Treatment of intestinal graft-versus-host disease with unrelated donor fecal microbiota transplantation capsules: A case report. Medicine 2020, 99, e22129. [Google Scholar] [CrossRef]
- Kakihana, K.; Fujioka, Y.; Suda, W.; Najima, Y.; Kuwata, G.; Sasajima, S.; Mimura, I.; Morita, H.; Sugiyama, D.; Nishikawa, H.; et al. Fecal microbiota transplantation for patients with steroid-resistant acute graft-versus-host disease of the gut. Blood 2016, 128, 2083–2088. [Google Scholar] [CrossRef] [PubMed]
- Spindelboeck, W.; Schulz, E.; Uhl, B.; Kashofer, K.; Aigelsreiter, A.; Zinke-Cerwenka, W.; Mulabecirovic, A.; Kump, P.K.; Halwachs, B.; Gorkiewicz, G.; et al. Repeated fecal microbiota transplantations attenuate diarrhea and lead to sustained changes in the fecal microbiota in acute, refractory gastrointestinal graft-versus-host-disease. Haematologica 2017, 102, e210–e213. [Google Scholar] [CrossRef] [PubMed]
- Qi, X.; Li, X.; Zhao, Y.; Wu, X.; Chen, F.; Ma, X.; Zhang, F.; Wu, D. Treating steroid refractory intestinal acute graft-vs.-host disease with fecal microbiota transplantation: A pilot study. Front Immunol. 2018, 9, 2195. [Google Scholar] [CrossRef]
- Van Lier, Y.F.; Davids, M.; Haverkate, N.J.E.; de Groot, P.F.; Donker, M.L.; Meijer, E.; Heubel-Moenen, F.C.J.I.; Nur, E.; Zeerleder, S.S.; Nieuwdorp, M.; et al. Donor fecal microbiota transplantation ameliorates intestinal graft-versus-host disease in allogeneic hematopoietic cell transplant recipients. Sci. Transl. Med. 2020, 12, eaaz8926. [Google Scholar] [CrossRef] [PubMed]
- Bilinski, J.; Lis, K.; Tomaszewska, A.; Grzesiowski, P.; Dzieciatkowski, T.; Tyszka, M.; Karakulska-Prystupiuk, E.; Boguradzki, P.; Tormanowska, M.; Halaburda, K.; et al. Fecal microbiota transplantation in patients with acute and chronic graft-versus-host disease—Spectrum of responses and safety profile. Results from a prospective, multicenter study. Am. J. Hematol. 2021, 96, E88–E91. [Google Scholar] [CrossRef]
- DeFilipp, Z.; Peled, J.U.; Li, S.; Mahabamunuge, J.; Dagher, Z.; Slingerland, A.E.; del Rio, C.; Valles, B.; Kempner, M.E.; Smith, M.; et al. Third-party fecal microbiota transplantation following allo-HCT reconstitutes microbiome diversity. Blood Adv. 2018, 2, 745–753. [Google Scholar] [CrossRef]
- Taur, Y.; Coyte, K.; Schluter, J.; Robilotti, E.; Figueroa, C.; Gjonbalaj, M.; Littmann, E.R.; Ling, L.; Miller, L.; Gyaltshen, Y.; et al. Reconstitution of the gut microbiota of antibiotic-treated patients by autologous fecal microbiota transplant. Sci. Transl. Med. 2018, 10, eaap9489. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tyszka, M.; Biliński, J.; Basak, G.W. Advances in Intestinal Barrier Preservation and Restoration in the Allogeneic Hematopoietic Cell Transplantation Setting. J. Clin. Med. 2021, 10, 2508. https://doi.org/10.3390/jcm10112508
Tyszka M, Biliński J, Basak GW. Advances in Intestinal Barrier Preservation and Restoration in the Allogeneic Hematopoietic Cell Transplantation Setting. Journal of Clinical Medicine. 2021; 10(11):2508. https://doi.org/10.3390/jcm10112508
Chicago/Turabian StyleTyszka, Martyna, Jarosław Biliński, and Grzegorz Władysław Basak. 2021. "Advances in Intestinal Barrier Preservation and Restoration in the Allogeneic Hematopoietic Cell Transplantation Setting" Journal of Clinical Medicine 10, no. 11: 2508. https://doi.org/10.3390/jcm10112508
APA StyleTyszka, M., Biliński, J., & Basak, G. W. (2021). Advances in Intestinal Barrier Preservation and Restoration in the Allogeneic Hematopoietic Cell Transplantation Setting. Journal of Clinical Medicine, 10(11), 2508. https://doi.org/10.3390/jcm10112508





