Visual Outcome after Intravitreal Anti-VEGF Therapy for Macular Neovascularisation Secondary to Sorsby’s Fundus Dystrophy: A Systematic Review
Abstract
1. Introduction
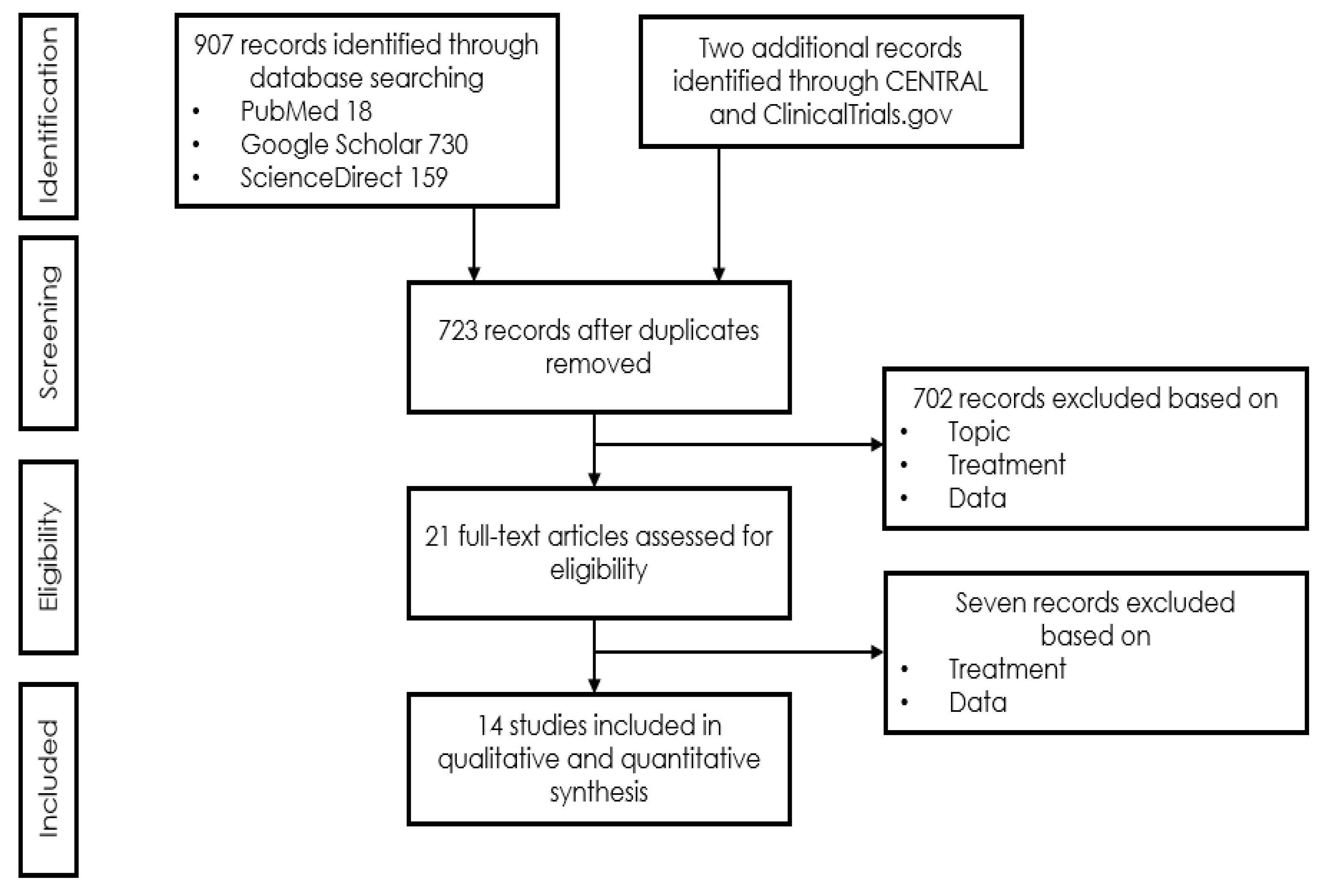
2. Materials and Methods
2.1. Eligibility Criteria
- Report of single or multiple patient case or cohort study including patients diagnosed with Sorsby’s Fundus Dystrophy published or treated until February 2021;
- Additional or pre-treatment with corticosteroids or photodynamic therapy was accepted;
- Reporting of evolution of visual function.
2.2. Information Retrieved from the Included Publications
2.3. Assessment of Risk of Bias
3. Results
Meta-Analysis
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Sorsby, A.; Mason, M.E.J.; Gardener, N. A Fundus Dystrophy with Unusual Features (Late onset and dominant inheritance of a central retinal lesion showing oedema, haemorrhage and exudates developing into generalised choroidal atrophy with massive pigment proliferation). Br. J. Ophthalmol. 1949, 33, 67–97. [Google Scholar] [CrossRef]
- Capon, M.R.C.; Polkinghorne, P.J.; Fitzke, F.W.; Bird, A.C. Sorsby’s pseudoinflammatory macula dystrophy—Sorsby’s fundus dystrophies. Eye 1988, 2, 114–122. [Google Scholar] [CrossRef] [PubMed]
- Jacobson, S.G.; Cideciyan, A.V.; Regunath, G.; Rodriguez, F.J.; VanDenburgh, K.; Sheffield, V.C.; Stone, E.M. Night blindness in Sorsby’s fundus dystrophy reversed by vitamin A. Nat. Genet. 1995, 11, 27–32. [Google Scholar] [CrossRef]
- Handsley, M.M.; Edwards, D.R. Metalloproteinases and their inhibitors in tumor angiogenesis. Int. J. Cancer 2005, 115, 849–860. [Google Scholar] [CrossRef] [PubMed]
- Qi, J.H.; Ebrahem, Q.; Ali, M.; Cutler, A.; Bell, B.; Prayson, N.; Sears, J.; Knauper, V.; Murphy, G.; Anand-Apte, B. Tissue Inhibitor of Metalloproteinases-3 Peptides Inhibit Angiogenesis and Choroidal Neovascularization in Mice. PLoS ONE 2013, 8, e55667. [Google Scholar] [CrossRef]
- Qi, J.H.; Ebrahem, Q.; Moore, N.; Murphy, G.; Claesson-Welsh, L.; Bond, M.; Baker, A.; Anand-Apte, B. A novel function for tissue inhibitor of metalloproteinases-3 (TIMP3): Inhibition of angiogenesis by blockage of VEGF binding to VEGF receptor-2. Nat. Med. 2003, 9, 407–415. [Google Scholar] [CrossRef] [PubMed]
- Fariss, R.N.; Apte, S.S.; Olsen, B.R.; Iwata, K.; Milam, A.H. Tissue inhibitor of metalloproteinases-3 is a compo-nent of Bruch’s membrane of the eye. Am. J. Pathol. 1997, 150, 323–328. [Google Scholar]
- Langton, K.P.; McKie, N.; Smith, B.M.; Brown, N.J.; Barker, M.D. Sorsby’s fundus dystrophy mutations impair turnover of TIMP-3 by retinal pigment epithelial cells. Hum. Mol. Genet. 2005, 14, 3579–3586. [Google Scholar] [CrossRef]
- Christensen, D.R.; Brown, F.E.; Cree, A.J.; Ratnayaka, J.A.; Lotery, A.J. Sorsby fundus dystrophy—A review of pathology and disease mechanisms. Exp. Eye Res. 2017, 165, 35–46. [Google Scholar] [CrossRef] [PubMed]
- Anand-Apte, B.; Chao, J.R.; Singh, R.; Stöhr, H. Sorsby fundus dystrophy:Insights from the past and looking to the future. J. Neurosci. Res. 2018, 97, 88–97. [Google Scholar] [CrossRef]
- Sivaprasad, S.; Webster, A.R.; Egan, C.; Bird, A.C.; Tufail, A. Clinical Course and Treatment Outcomes of Sorsby Fundus Dystrophy. Am. J. Ophthalmol. 2008, 146, 228–234.e2. [Google Scholar] [CrossRef]
- Daniel, E.; Toth, C.A.; Grunwald, J.E.; Jaffe, G.J.; Martin, D.F.; Fine, S.L.; Huang, J.; Ying, G.-S.; Hagstrom, S.A.; Winter, K.; et al. Risk of Scar in the Comparison of Age-related Macular Degeneration Treatments Trials. Ophthalmology 2014, 121, 656–666. [Google Scholar] [CrossRef]
- Hamilton, W.K.; Ewing, C.C.; Ives, E.J.; Carruthers, J.D. Sorsby’s Fundus Dystrophy. Ophthalmology 1989, 96, 1755–1762. [Google Scholar] [CrossRef]
- Polkinghorne, P.J.; Capon, M.R.; Berninger, T.; Lyness, A.L.; Sehmi, K.; Bird, A.C. Sorsby’s Fundus Dystrophy: A clinical study. Ophthalmology 1989, 96, 1763–1768. [Google Scholar] [CrossRef]
- Gliem, M.; Müller, P.L.; Mangold, E.; Bolz, H.J.; Stöhr, H.; Weber, B.H.; Holz, F.G.; Issa, P.C. Reticular Pseudodrusen in Sorsby Fundus Dystrophy. Ophthalmology 2015, 122, 1555–1562. [Google Scholar] [CrossRef]
- Menassa, N.; Burgula, S.; Empeslidis, T.; Tsaousis, K.T. Bilateral choroidal neovascular membrane in a young patient with Sorsby fundus dystrophy: The value of prompt treatment. BMJ Case Rep. 2017, 2017, 2017220488. [Google Scholar] [CrossRef] [PubMed]
- Gliem, M.; Müller, P.L.; Mangold, E.; Holz, F.G.; Bolz, H.J.; Stöhr, H.; Weber, B.H.F.; Issa, P.C. Sorsby Fundus Dystrophy: Novel Mutations, Novel Phenotypic Characteristics, and Treatment Outcomes. Investig. Opthalmol. Vis. Sci. 2015, 56, 2664–2676. [Google Scholar] [CrossRef] [PubMed]
- Holz, F.G.; Haimovici, R.; Wagner, D.G.; Bird, A.C. Recurrent Choroidal Neovascularization after Laser Photocoagulation in Sorsbyʼs Fundus Dystrophy. Retina 1994, 14, 329–334. [Google Scholar] [CrossRef] [PubMed]
- Keller, J.; Giralt, J.; Alforja, S.; Casaroli-Marano, R.P. Altering the Clinical Course of Sorsby Fundus Dystrophy with the Use of Anti-Vascular Endothelial Growth Factor Intraocular Therapy. Retin. Cases Brief Rep. 2015, 9, 104–105. [Google Scholar] [CrossRef]
- Spaide, R.F. Long-Term Visual Acuity Preservation in Sorsby Fundus Dystrophy with Corticosteroid Treatment. Retin. Cases Brief Rep. 2019. [Google Scholar] [CrossRef]
- Fung, A.T.; Stöhr, H.; Weber, B.H.F.; Holz, F.G.; Yannuzzi, L.A. Atypical Sorsby Fundus Dystrophy with a Novel Tyr159cys Timp-3 Mutation. Retin. Cases Brief Rep. 2013, 7, 71–74. [Google Scholar] [CrossRef] [PubMed]
- Kapoor, K.G.; Bakri, S.J. Intravitreal Anti-Vascular Endothelial Growth Factor Therapy for Choroidal Neovascularization Due to Sorsby Macular Dystrophy. J. Ocul. Pharmacol. Ther. 2013, 29, 444–447. [Google Scholar] [CrossRef]
- Peiretti, E.; Klancnik, J.M.; Spaide, R.F.; Yannuzzi, L. Choroidal Neovascularization in Sorsby Fundus Dystrophy Treated with Photodynamic Therapy and Intravitreal Triamcinolone Acetonide. Retina 2005, 25, 377–379. [Google Scholar] [CrossRef]
- Wong, S.C.; Fong, K.C.S.; Lee, N.; Gregory-Evans, K.; Gregory-Evans, C.Y. Successful photodynamic therapy for subretinal neovascularisation due to Sorsby’s fundus dystrophy: 1 year follow up. Br. J. Ophthalmol. 2003, 87, 796–797. [Google Scholar] [CrossRef][Green Version]
- Cohen, J. A Coefficient of Agreement for Nominal Scales. Educ. Psychol. Meas. 1960, 20, 37–46. [Google Scholar] [CrossRef]
- Sanz, G.F.; Alonso-Gonzalez, R.; Keane, P.; Carreno, E.; Liew, G.; Sim, D.; Patel, P.; Webster, A.; Egan, C.; Tufail, A. Treatment with Intravitreal An-ti-VEGF for Choroidal Neovascular Membrane secondary to Sorsby’s Fundus Dystrophy: A 24-Month Analysis. Investig. Ophthalmol. Vis. Sci. 2013, 54, 3863. [Google Scholar]
- Kaye, R.; Lotery, A. Long-term Outcome of Bevacizumab Therapy in Sorsby Fundus Dystrophy, A Case Series. Investig. Ophthalmol. Vis. Sci. 2017, 58, 229. [Google Scholar]
- Mohla, A.; Khan, K.; Kasilian, M.; Michaelides, M. OCT angiography in the management of choroidal neovascular membrane secondary to Sorsby fundus dystrophy. BMJ Case Rep. 2016, 2016, 2016216453. [Google Scholar] [CrossRef] [PubMed]
- Tsokolas, G.; Almuhtaseb, H.; Lotery, A. Evaluation of Pro-re-Nata (PRN) and Treat and Extend Bevacizumab treatment protocols in Sorsby Fundus Dystrophy. Eur. J. Ophthalmol. 2018, 30, 26–33. [Google Scholar] [CrossRef]
- Gemenetzi, M.K.; Luff, A.J.; Lotery, A.J. Successful Treatment of Choroidal Neovascularization Secondary to Sorsby Fundus Dystrophy with Intravitreal Bevacizumab. Retin. Cases Brief Rep. 2011, 5, 132–135. [Google Scholar] [CrossRef] [PubMed]
- Gray, T.L.; Wong, H.-C.; Raymond, G.L. Choroidal Neovascularization Secondary to Sorsby Fundus Dystrophy Treated with Intravitreal Bevacizumab. Retin. Cases Brief Rep. 2012, 6, 193–196. [Google Scholar] [CrossRef] [PubMed]
- Balaskas, K.; Hovan, M.; Mahmood, S.; Bishop, P. Ranibizumab for the management of Sorsby fundus dystrophy. Eye 2012, 27, 101–102. [Google Scholar] [CrossRef] [PubMed]
- Copete-Piqueras, S.; Cava-Valenciano, C.; Flores-Moreno, I.; Moreno-Valladares, A.; Ruescas, V.B. Tratamiento antiangiogénico en fondo de distrofia de Sorsby sin mutación en gen de TIMP-3. Arch. Soc. Española Oftalmol. 2013, 88, 240–243. [Google Scholar] [CrossRef] [PubMed]
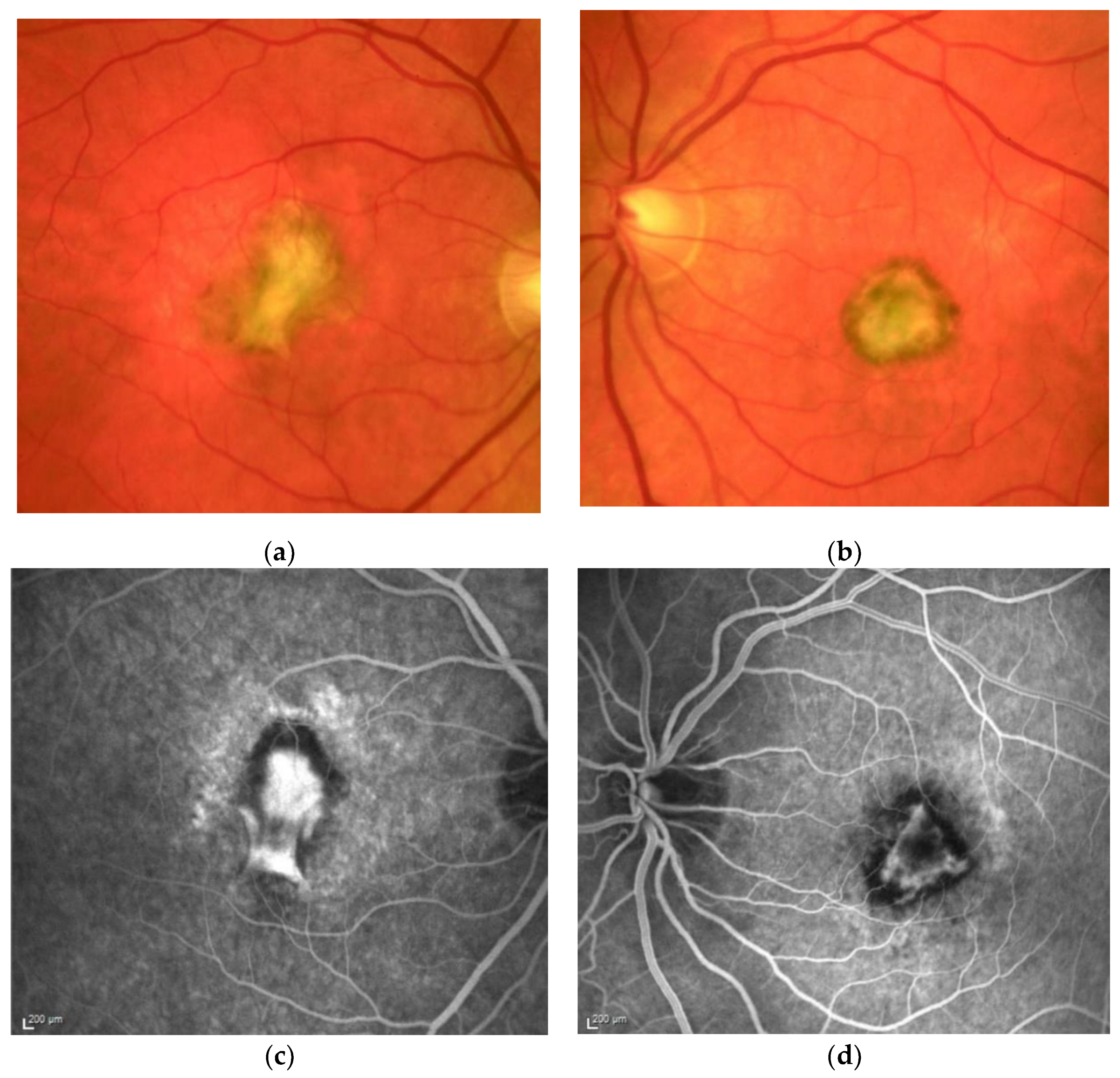
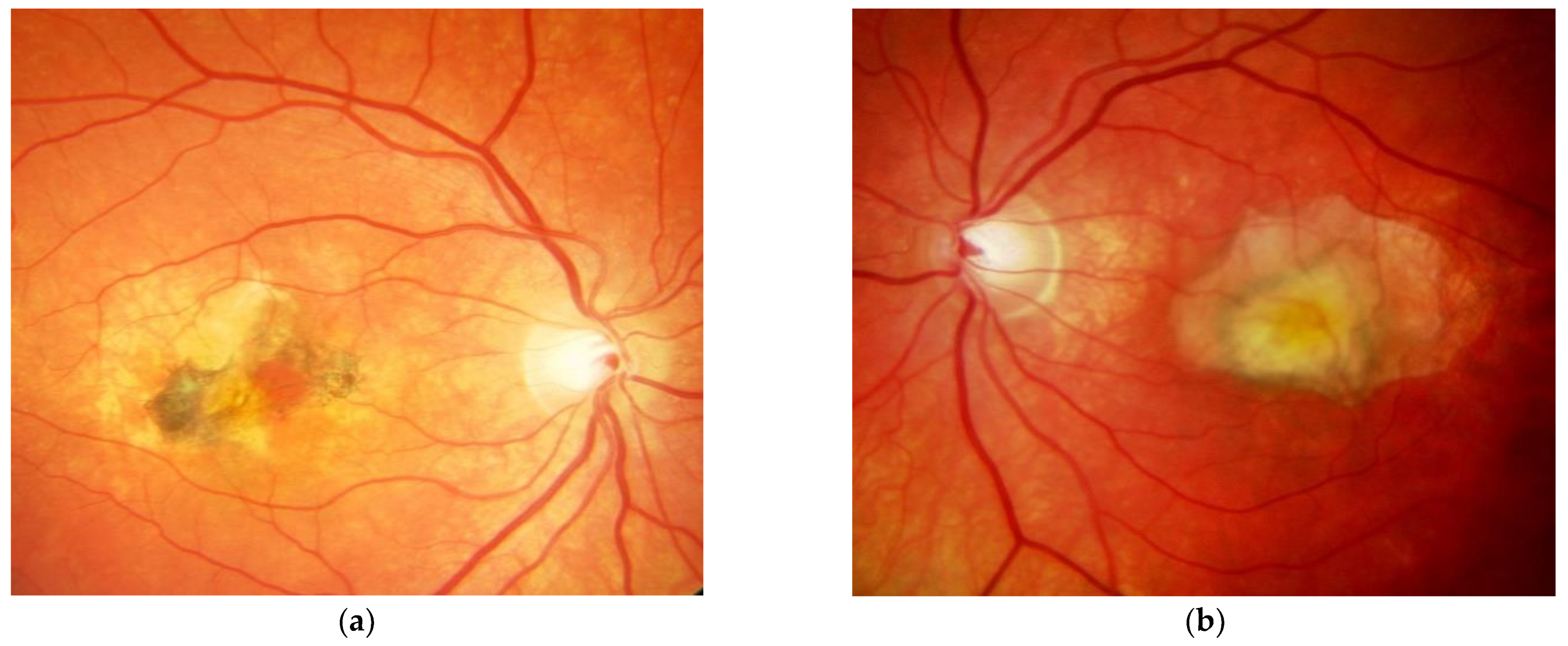
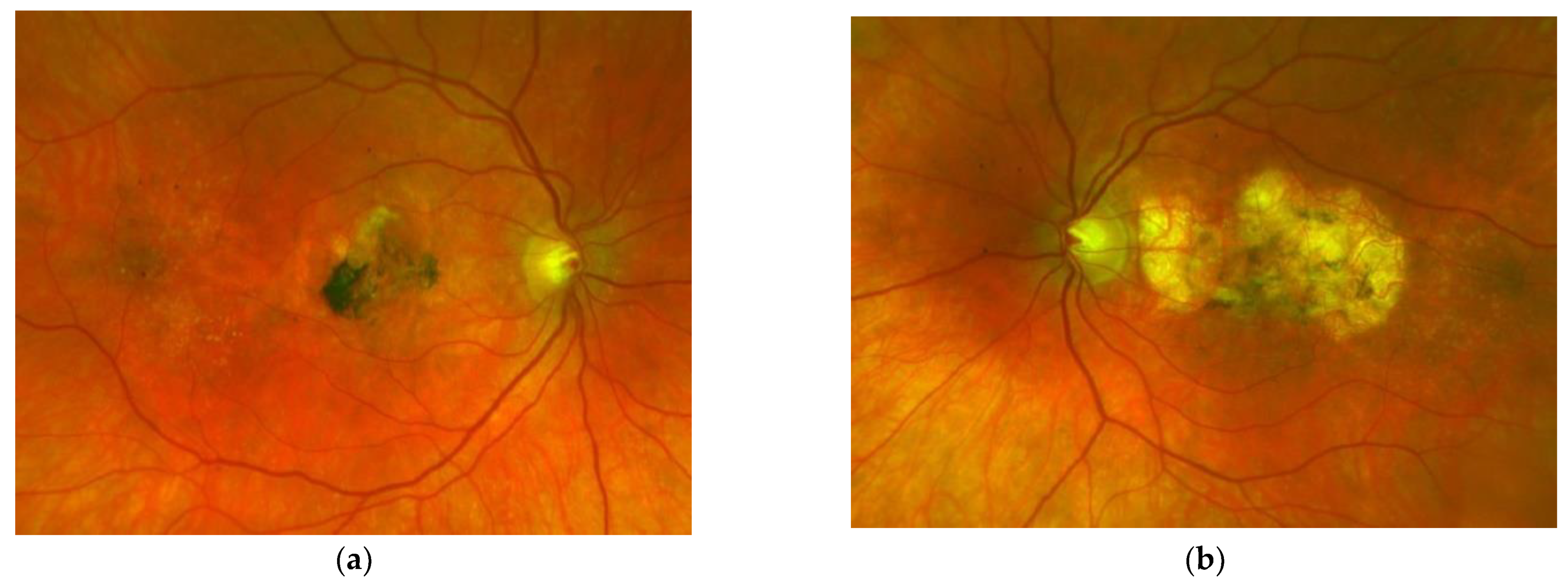
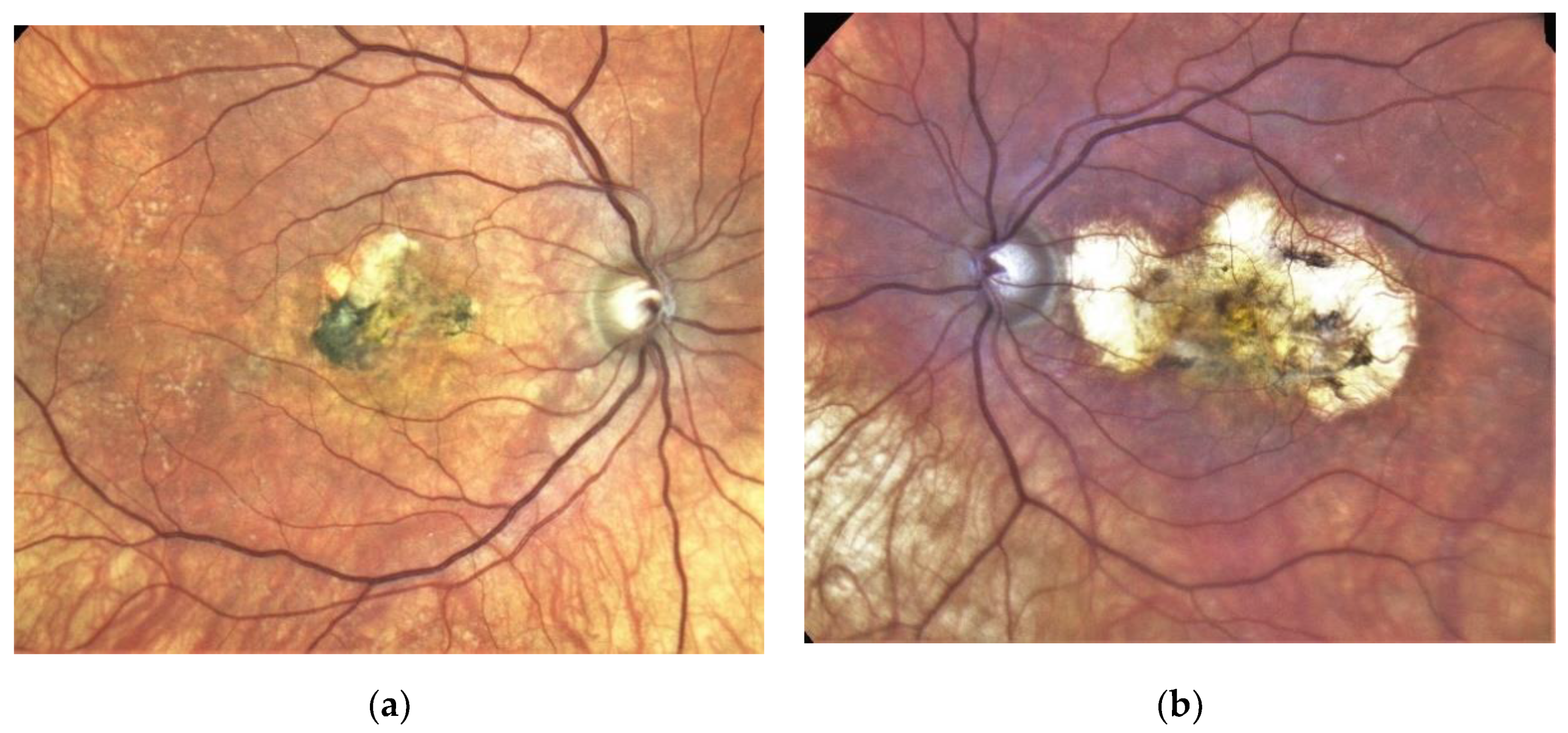
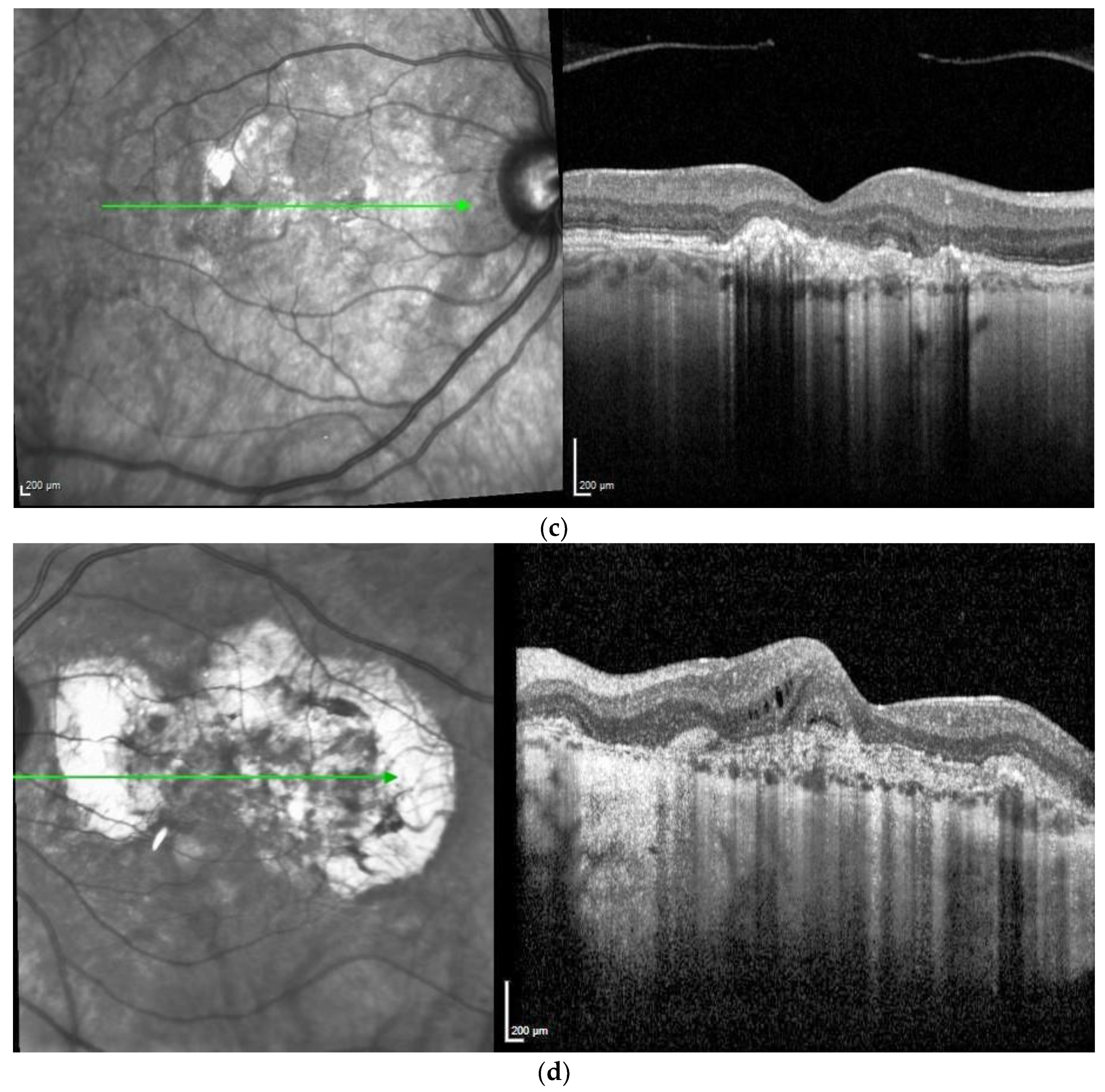
| Patient | First Author | Year of Publication | Gender | NR of Eyes | Age at Onset | Family History Positive | Prior Treatment | VA before Onset | Eye | ||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Sivaprasad | 2008 | m | 1 | nr | yes | PDT | nr | nr | ||||
| 2 | Gemenetzi | 2011 | f | 2 | 34 | yes | no | nr | r | ||||
| 2 | Gemenetzi | 2011 | f | 37 | yes | PDT | 1.00 | l | |||||
| 3 | Gemenetzi | 2011 | f | 1 | 44 | yes | no | 1.00 | r | ||||
| 4 | Gray | 2012 | f | 1 | 38 | yes | no | nr | l | ||||
| 5 | Balaskas | 2013 | m | 1 | 41 | nr | no | 1.25 | r | ||||
| 6 | Copete-Piqueras | 2013 | m | 2 | 32 | nr | no | nr | r | ||||
| 6 | Copete-Piqueras | 2013 | m | 32 | nr | no | nr | l | |||||
| 7 | Fung | 2013 | m | 1 | 44 | yes | no | 1.00 | r | ||||
| 8 | Kapoor | 2013 | m | 2 | 57 | yes | no | 1.25 | r | ||||
| 8 | Kapoor | 2013 | m | 57 | yes | no | 1.00 | l | |||||
| 9 | Gliem | 2015 | nr | 1 | 54 | yes | no | 1.00 | l | ||||
| 10 | Gliem | 2015 | nr | 1 | 56 | yes | no | 1.00 | r | ||||
| 11 | Gliem | 2015 | m | 1 | 45 | yes | no | 1.00 | r | ||||
| 12 | Keller | 2015 | m | 2 | 32 | yes | nr | nr | r | ||||
| 12 | Keller | 2015 | m | 32 | yes | PDT | nr | l | |||||
| 13 | Keller | 2015 | m | 2 | 28 | yes | no | nr | r | ||||
| 13 | Keller | 2015 | m | 28 | yes | no | nr | l | |||||
| 14 | Mohla | 2016 | f | 1 | 52 | nr | no | 0.63 | r | ||||
| 15 | Menassa | 2017 | m | 2 | 44 | yes | no | 1.60 | r | ||||
| 15 | Menassa | 2017 | m | 38 | yes | no | nr | l | |||||
| 16 | Tsokolas | 2020 | f | 2 | 34 | yes | no | nr | r | ||||
| 16 | Tsokolas | 2020 | f | 37 | yes | PDT | nr | l | |||||
| 17 | Tsokolas | 2020 | f | 2 | 36 | yes | no | 1.25 | r | ||||
| 17 | Tsokolas | 2020 | f | 38 | yes | no | 1.00 | l | |||||
| 18 | Own patient | 2004 | m | 2 | 33 | yes | no | nr | r | ||||
| 18 | Own patient | 2004 | m | 33 | yes | no | nr | l | |||||
| 19–23 * | Kaye | 2017 | nr | 5 | nr | nr | nr | nr | nr | ||||
| 24–31 * | Sanz | 2013 | 62.5% m | 9 | 45.3 (6.9) | nr | nr | nr | nr | ||||
| Mean | 62.5% m | 41 | 41.2 | 48.4% | 9.8% | 1.08 | |||||||
| Patient | First Author | VA at Onset | Treatment Delay (Months) | Last VA | Follow-Up after Onset of Anti-VEGF Treatment (Months) | Total Number of Intravitreal Injections | Drug | Mutation | |||||
| 1 | Sivaprasad | 0.50 | 2 | 0.50 | 6 | 2 | 2 Bev | Ser181Cys | |||||
| 2 | Gemenetzi | 0.10 | 0 | 0.16 | 33 | 6 | 6 Bev | p.S204C | |||||
| 2 | Gemenetzi | 1.60 | 0.75 | 1.25 | 5 | 3 | 3 Bev | p.S204C | |||||
| 3 | Gemenetzi | 0.10 | 0 | 1.00 | 3 | 1 | 1 Bev | p.S204C | |||||
| 4 | Gray | 1.00 | 1.5 | 1.00 | 13 | 3 | 3 Bev | Ser181Cys | |||||
| 5 | Balaskas | 0.16 | nr | 0.40 | 27 | 14 | 14 Ran | c.610A4T (p.Ser204Cys) | |||||
| 6 | Copete-Piqueras | 0.63 | 0 | 1.00 | 6 | 1 | 1 Ran | mutations in Exon 5 of gene 22.12.3 | |||||
| 6 | Copete-Piqueras | 0.80 | 0 | 1.00 | 6 | 1 | 1 Ran | mutations in Exon 5 of gene 22.12.3 | |||||
| 7 | Fung | 0.63 | 0 | 0.80 | 48 | 6 | 6 Bev, PDT | Tyr159Cys | |||||
| 8 | Kapoor | 0.50 | 0 | 0.10 | 55 | 8 | 8 Bev, several Bev-Dex | normal coding sequence (codons 124–188 of the mature protein) | |||||
| 8 | Kapoor | 0.63 | 0 | 0.40 | 77 | 31 | 8 Bev, min. 18 Bev-Dex, 5 Ran, PDT | normal coding sequence (codons 124–188 of the mature protein) | |||||
| 9 | Gliem | 0.80 | 0 | 1.00 | 12 | 1 | 1 Bev | c.530A > G (p.Tyr200Cys) | |||||
| 10 | Gliem | 0.63 | 0 | 1.00 | 8 | nr | multiple Bev | c.530A > G (p.Tyr200Cys) | |||||
| 11 | Gliem | nr | 0 | 1.00 | nr | 35 | 35 Bev | c.545A > G(p.Tyr182Cys) | |||||
| 12 | Keller | nr | nr | 0.70 | 60 | nr | several Ran and Bev | nr | |||||
| 12 | Keller | nr | nr | 0.03 | 60 | 3 | PDT, 3 Ran | nr | |||||
| 13 | Keller | nr | nr | 0.10 | 48 | nr | Multiple Ran | nr | |||||
| 13 | Keller | 1.00 | nr | 0.20 | 48 | nr | Multiple Ran | nr | |||||
| 14 | Mohla | 0.10 | 0 | 0.32 | 7 | 2 | 2 Bev | p.Arg204Cys | |||||
| 15 | Menassa | 1.25 | 0.3 | 0.80 | 6 | 5 | 5 Ran | c.610A > T | |||||
| 15 | Menassa | nr | nr | 0.10 | nr | 6 | 6 Ran | c.610A > T | |||||
| 16 | Tsokolas | 0.10 | 0 | 0.08 | 144 | 5 | 5 Bev | Ser204Cys | |||||
| 16 | Tsokolas | 1.25 | 1 | 0.16 | 108 | 79 | 79 Bev | Ser204Cys | |||||
| 17 | Tsokolas | nr | 4 | 0.06 | 72 | 24 | 24 Bev | Ser204Cys | |||||
| 17 | Tsokolas | nr | 0 | 0.50 | 60 | 42 | 42 Bev | Ser204Cys | |||||
| 18 | Own patient | 0 | 1.0 | 192 | 24 | 3 PDT, Tri, 24 Ran | mutation in the TIMP3 gene | ||||||
| 18 | Own patient | 0 | 0.16 | 192 | 9 | 4 PDT, multiple Tri, 9 Ran | mutation in the TIMP3 gene | ||||||
| 19–23 * | Kaye | 0.8 (0.8) | nr | 0.2 (0.4) | Min. 60 | 16 | Bev | mutation in tissue inhibitor of metalloproteinases-3 (TIMP3) | |||||
| 24–31 * | Sanz | 0.25 (0.2) | nr | nr | nr | 9.11 (6.01) | Bev, Ran | p.Ser204Cys | |||||
| Mean | 0.56 | 0.45 | 0.49 | 54 | 12.78 | ||||||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Baston, A.; Gerhardt, C.; Zandi, S.; Garweg, J.G. Visual Outcome after Intravitreal Anti-VEGF Therapy for Macular Neovascularisation Secondary to Sorsby’s Fundus Dystrophy: A Systematic Review. J. Clin. Med. 2021, 10, 2433. https://doi.org/10.3390/jcm10112433
Baston A, Gerhardt C, Zandi S, Garweg JG. Visual Outcome after Intravitreal Anti-VEGF Therapy for Macular Neovascularisation Secondary to Sorsby’s Fundus Dystrophy: A Systematic Review. Journal of Clinical Medicine. 2021; 10(11):2433. https://doi.org/10.3390/jcm10112433
Chicago/Turabian StyleBaston, Arthur, Christin Gerhardt, Souska Zandi, and Justus G. Garweg. 2021. "Visual Outcome after Intravitreal Anti-VEGF Therapy for Macular Neovascularisation Secondary to Sorsby’s Fundus Dystrophy: A Systematic Review" Journal of Clinical Medicine 10, no. 11: 2433. https://doi.org/10.3390/jcm10112433
APA StyleBaston, A., Gerhardt, C., Zandi, S., & Garweg, J. G. (2021). Visual Outcome after Intravitreal Anti-VEGF Therapy for Macular Neovascularisation Secondary to Sorsby’s Fundus Dystrophy: A Systematic Review. Journal of Clinical Medicine, 10(11), 2433. https://doi.org/10.3390/jcm10112433






