The Influence of Air Pollution on the Development of Allergic Inflammation in the Airways in Krakow’s Atopic and Non-Atopic Residents
Abstract
1. Introduction
2. Materials and Methods
2.1. Patients
- symptomatic (with atopy and an allergy to birch, no allergy to all-year allergens, non-desensitized);
- control group (with symptoms, without atopy).
2.2. PM Preparation
2.3. Basophil Activation Test
2.4. 1-OHP Assessment
2.5. Statistical Analysis
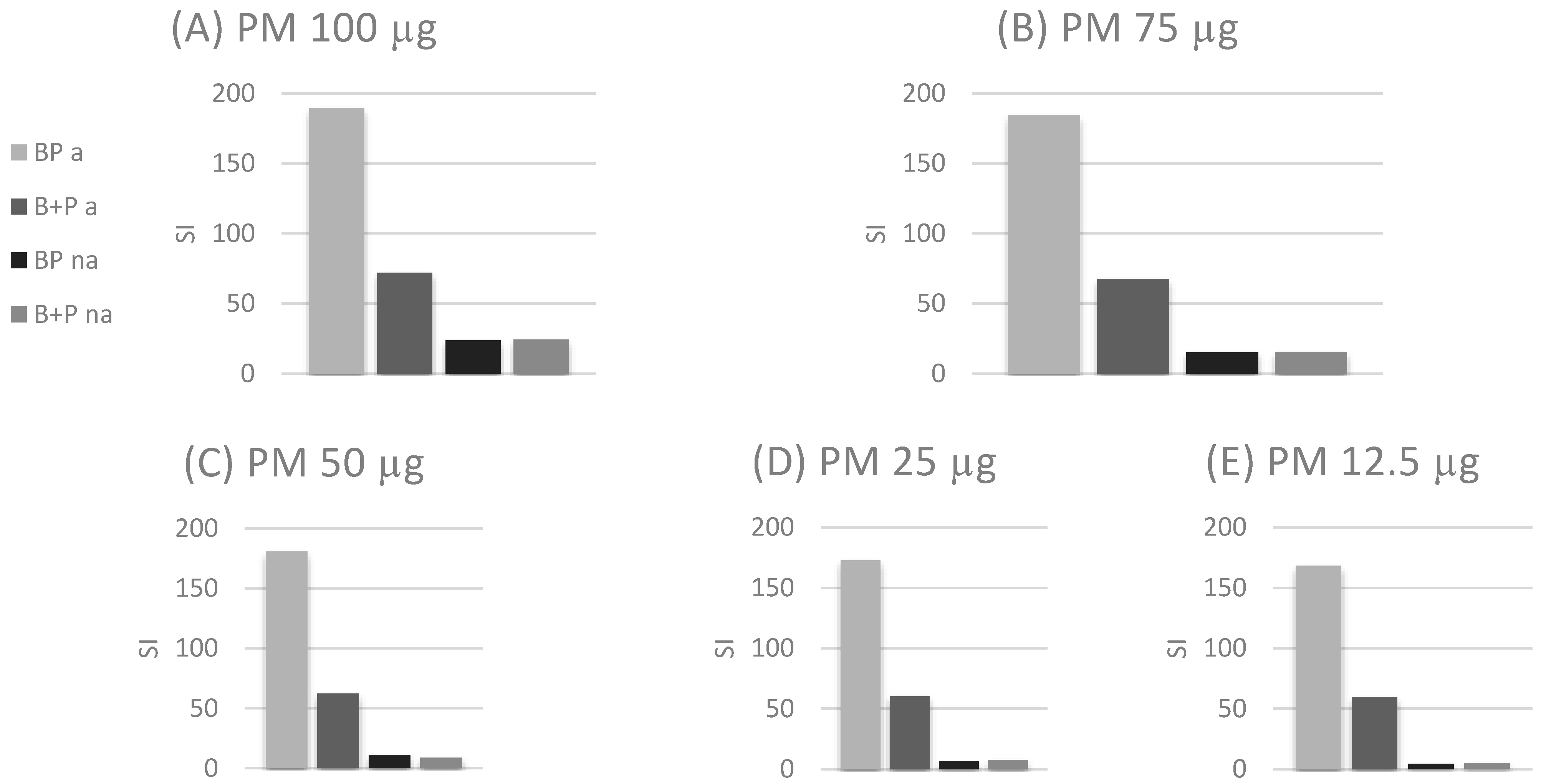
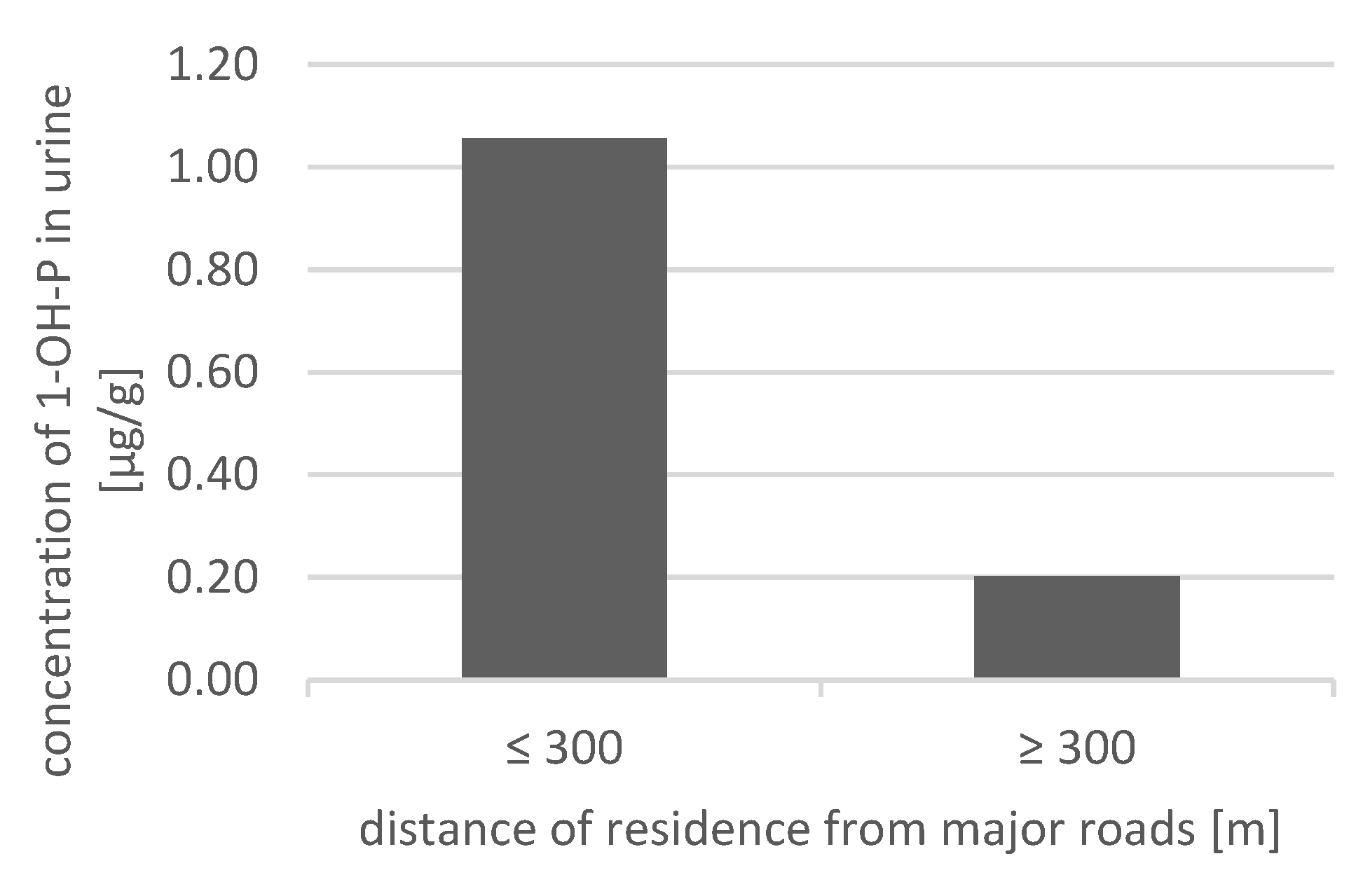
3. Results
4. Discussion
5. Conclusions
- 1.
- Particulate matter (PM) 2.5 in atopic and non-atopic people activates basophils of peripheral blood and causes the expression of CD63 receptors on their surface. Dust alone activates basophils in 83% of determinations in atopic people and 75% of determinations in non-atopic people in Kraków.
- 2.
- Exposure to birch pollen and PM2.5 has a synergistic effect in sensitized individuals.
- 3.
- The higher the exposure to pollutants, the higher the synergistic basophil response to the allergen and PM in atopic patients.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Pawankar, R. Allergic diseases and asthma: A global public health concern and a call to action. World Allergy Organ. J. 2014, 7, 1–12. [Google Scholar] [CrossRef]
- Bråbäck, L.; Forsberg, B. Does traffic exhaust contribute to the development of asthma and allergic sensitization in children: Findings from recent cohort studies. Environ. Heal. 2009, 8, 17. [Google Scholar] [CrossRef]
- Raport o stanie środowiska w województwie małopolskim w 2017 roku. Available online: http://www.krakow.pios.gov.pl/Press/publikacje/raporty/raport17/raport2017.pdf (accessed on 1 February 2021).
- Guarnieri, M.; Balmes, J.R. Outdoor air pollution and asthma. Lancet 2014, 383, 1581–1592. [Google Scholar] [CrossRef]
- Naclerio, R.; Ansotegui, I.J.; Bousquet, J.; Canonica, G.W.; D’Amato, G.; Rosario, N.; Pawankar, R.; Peden, D.; Bergmann, K.-C.; Bielory, L.; et al. International expert consensus on the management of allergic rhinitis (AR) aggravated by air pollutants. World Allergy Organ. J. 2020, 13, 100106. [Google Scholar] [CrossRef] [PubMed]
- Zając, J.; Gomółka, E.; Szot, W. Urinary 1-hydroxypyrene in occupationally-exposed and non-exposed individuals in Silesia, Poland. Ann. Agric. Environ. Med. 2018, 25, 625–629. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.; Bo, L.; Gong, C.; Cheng, P.; Kan, H.; Xie, Y.; Song, W. Preliminary study to explore gene-PM2.5 interactive effects on respiratory system in traffic policemen. Int. J. Occup. Med. Environ. Heal. 2015, 28, 971–983. [Google Scholar] [CrossRef]
- Chu, J.-H.; Hart, J.E.; Chhabra, D.; Garshick, E.; Raby, B.A.; Laden, F. Gene expression network analyses in response to air pollution exposures in the trucking industry. Environ. Heal. 2016, 15, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Schiavoni, G.; D’Amato, G.; Afferni, C. The dangerous liaison between pollens and pollution in respiratory allergy. Ann. Allergy, Asthma Immunol. 2017, 118, 269–275. [Google Scholar] [CrossRef]
- Ouyang, Y.; Xu, Z.; Fan, E.; Li, Y.; Miyake, K.; Xu, X.; Zhang, L. Changes in gene expression in chronic allergy mouse model exposed to natural environmental PM2.5-rich ambient air pollution. Sci. Rep. 2018, 8, 6326. [Google Scholar] [CrossRef]
- Santos, A.F.; Alpan, O.; Hoffmann, H. Basophil activation test: Mechanisms and considerations for use in clinical trials and clinical practice. Allergy 2021. [Google Scholar] [CrossRef]
- Jongeneelen, F.J. A guidance value of 1-hydroxypyrene in urine in view of acceptable occupational exposure to polycyclic aromatic hydrocarbons. Toxicol. Lett. 2014, 231, 239–248. [Google Scholar] [CrossRef] [PubMed]
- Mazur, M.; Czarnobilska, E. Wpływ zanieczyszczenia powietrza na rozwój chorób alergicznych. Przegl Lek. 2016, 73, 823. [Google Scholar] [PubMed]
- Liu, L.; Poon, R.; Chen, L.; Frescura, A.-M.; Montuschi, P.; Ciabattoni, G.; Wheeler, A.; Dales, R. Acute Effects of Air Pollution on Pulmonary Function, Airway Inflammation, and Oxidative Stress in Asthmatic Children. Environ. Heal. Perspect. 2009, 117, 668–674. [Google Scholar] [CrossRef]
- Schober, W.; Belloni, B.; Lubitz, S.; Eberlein-König, B.; Bohn, P.; Saritas, Y.; Lintelmann, J.; Matuschek, G.; Behrendt, H.; Buters, J. Organic Extracts of Urban Aerosol (≤PM2.5) Enhance rBet v 1-Induced Upregulation of CD63 in Basophils from Birch Pollen–Allergic Individuals. Toxicol. Sci. 2006, 90, 377–384. [Google Scholar] [CrossRef]
- Rössner, P.; Mrhalkova, A.; Uhlířová, K.; Spatova, M.; Rossnerova, A.; Libalova, H.; Schmuczerova, J.; Milcova, A.; Topinka, J.; Šrám, R.J. Nucleotide Excision Repair Is Not Induced in Human Embryonic Lung Fibroblasts Treated with Environmental Pollutants. PLOS ONE 2013, 8, e69197. [Google Scholar] [CrossRef] [PubMed]
- Gualtieri, M.; Longhin, E.; Mattioli, M.; Mantecca, P.; Tinaglia, V.; Mangano, E.; Proverbio, M.C.; Bestetti, G.; Camatini, M.; Battaglia, C. Gene expression profiling of A549 cells exposed to Milan PM2.5. Toxicol. Lett. 2012, 209, 136–145. [Google Scholar] [CrossRef]
- Ramanathan, M., Jr.; London, N.R., Jr.; Tharakan, A.; Surya, N.; Sussan, T.E.; Rao, X.; Lin, S.Y.; Toskala, E.; Rajagopalan, S.; Biswal, S. Airborne Particulate Matter Induces Nonallergic Eosinophilic Sinonasal Inflammation in Mice. Am. J. Respir. Cell Mol. Biol. 2017, 57, 59–65. [Google Scholar] [CrossRef]
- Canonica, G.W.; Compalati, E. Minimal persistent inflammation in allergic rhinitis: Implications for current treatment strategies. Clin. Exp. Immunol. 2009, 158, 260–271. [Google Scholar] [CrossRef]
- Diaz-Sanchez, D.; Garcia, M.P.; Wang, M.; Jyrala, M.; Saxon, A. Nasal challenge with diesel exhaust particles can induce sensitization to a neoallergen in the human mucosa. J. Allergy Clin. Immunol. 1999, 104, 1183–1188. [Google Scholar] [CrossRef]
- Kuroda, E.; Ozasa, K.; Temizoz, B.; Ohata, K.; Koo, C.X.; Kanuma, T.; Kusakabe, T.; Kobari, S.; Horie, M.; Morimoto, Y.; et al. Inhaled Fine Particles Induce Alveolar Macrophage Death and Interleukin-1α Release to Promote Inducible Bronchus-Associated Lymphoid Tissue Formation. Immun. 2016, 45, 1299–1310. [Google Scholar] [CrossRef]
- Ziemianin, M.; Myszkowska, D.; Piotrowicz, K.; Czarnobilska, E. Ekspozycja na pyłek olchy, leszczyny i brzozy, a stężenie pyłu zawieszonego PM10 w Krakowie w latach 2010–2015. Przegl Lek. 2016, 73, 802–808. [Google Scholar] [PubMed]
- Ifegwu, C.; Osunjaye, K.; Fashogbon, F.; Oke, K.; Adeniyi, A.; Anyakora, C. Urinary 1-Hydroxypyrene as a Biomarker to Carcinogenic Polycyclic Aromatic Hydrocarbon Exposure. Biomarkers Cancer 2012, 4, 7–17. Available online: https://ruj.uj.edu.pl/xmlui/handle/item/35642 (accessed on 24 May 2021). [CrossRef] [PubMed]
- Porebski, G.; Woźniak, M.; Czarnobilska, E. Residential proximity to major roadways is associated with increased prevalence of allergic respiratory symptoms in children. Ann. Agric. Environ. Med. 2014, 21, 760–766. [Google Scholar] [CrossRef] [PubMed]
- Health Effects Institute. Traffic-Related Air Pollution: A Critical Review of the Literature on Emissions, Exposure, and Health Effects; Panel on the Health Effects of Traffic-Related Air Pollution; Health Effects Institute: Boston, MA, USA, 2010. [Google Scholar]

| Age | Sex | Symptoms | BAT with Birch [%] | 1-OHP in Urine [μg/g] | |
|---|---|---|---|---|---|
| 1 | 19 | F | AR | 83.17 | 0.47 |
| 2 | 48 | F | AR + Asthma | 40.2 | 1.14 |
| 3 | 68 | F | AR | 80.21 | 3.50 |
| 4 | 49 | M | AR | 55.79 | 1.28 |
| 5 | 47 | F | AR | 42.62 | 0.92 |
| 6 | 65 | F | AR | 83.00 | 1.45 |
| 7 | 59 | M | AR + Asthma | 33.78 | 1.65 |
| 8 | 56 | F | AR + Asthma | 54.73 | 0.42 |
| 9 | 27 | M | AR | 29.09 | 0.06 |
| 10 | 47 | F | AR | 76.73 | 0.37 |
| 11 | 20 | F | AR + Asthma | 14.82 | 0.27 |
| 12 | 67 | M | AR | 81.30 | 0.19 |
| 13 | 70 | F | AR | 16.00 | 0.48 |
| 14 | 40 | F | AR | 84.40 | 0.93 |
| 15 | 47 | M | AR + Asthma | 76.08 | 0.54 |
| Age | Sex | Symptoms | BAT with Birch | 1-OHP in Urine [μg/g] | |
|---|---|---|---|---|---|
| 1 | 51 | M | AR + Asthma | 1.86 | 0.12 |
| 2 | 46 | F | AR + Asthma | 0.65 | 1.12 |
| 3 | 41 | F | AR | 2.18 | 0.01 |
| 4 | 18 | F | AR | 2.66 | 0.52 |
| 5 | 18 | M | AR | 1.31 | 0.33 |
| 6 | 60 | F | AR + Asthma | 1.69 | 0.32 |
| 7 | 56 | F | AR | 0.20 | 0.90 |
| 8 | 35 | M | AR + Asthma | 0.40 | 0.59 |
| 9 | 42 | F | AR | 0.90 | 0.40 |
| 10 | 38 | F | AR | 1.22 | 0.17 |
| 11 | 39 | F | AR | 0.50 | 0.03 |
| 12 | 55 | F | AR | 0.53 | 0.01 |
| 13 | 44 | M | AR | 1.49 | 0.01 |
| 14 | 29 | F | AR | 0.42 | 0.12 |
| 15 | 62 | F | AR | 0.57 | 0.56 |
| Average [%] | Minimum [%] | Median [%] | Maximum [%] | |
|---|---|---|---|---|
| PM 100 μg | 8.40 | 1.16 | 6.91 | 19.07 |
| PM 75 μg | 6.96 | 0.64 | 5.25 | 17.81 |
| PM 50 μg | 3.85 | 0.59 | 2.91 | 10.39 |
| PM 25 μg | 2.54 | 0.36 | 1.93 | 7.19 |
| PM 12.5 μg | 1.94 | 0.37 | 1.20 | 4.74 |
| BP 100 μg | 72.41 | 33.65 | 84.09 | 94.60 |
| BP 75 μg | 69.66 | 29.80 | 77.59 | 93.50 |
| BP 50 μg | 68.12 | 27.30 | 70.66 | 94.20 |
| BP 25 μg | 64.45 | 22.10 | 68.41 | 92.80 |
| BP 12.5 μg | 61.79 | 21.00 | 60.64 | 88.50 |
| negative control | 0.91 | 0.10 | 0.70 | 2.77 |
| Average [%] | Minimum [%] | Median [%] | Maximum [%] | |
|---|---|---|---|---|
| PM 100 μg | 8.84 | 1.10 | 8.16 | 24.54 |
| PM 75 μg | 6.27 | 0.77 | 5.52 | 17.60 |
| PM 50 μg | 4.38 | 0.54 | 2.60 | 11.80 |
| PM 25 μg | 3.47 | 0.24 | 1.85 | 9.70 |
| PM 12.5 μg | 2.03 | 0.32 | 1.47 | 6.03 |
| BP 100 μg | 8.38 | 1.17 | 7.37 | 22.75 |
| BP 75 μg | 6.96 | 0.77 | 5.08 | 19.10 |
| BP 50 μg | 4.62 | 0.62 | 4.07 | 10.80 |
| BP 25 μg | 3.12 | 0.56 | 2.14 | 8.35 |
| BP 12.5 μg | 2.13 | 0.46 | 1.83 | 5.14 |
| negative control | 0.87 | 0.08 | 0.54 | 2.51 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Czarnobilska, E.; Bulanda, M.; Bulanda, D.; Mazur, M. The Influence of Air Pollution on the Development of Allergic Inflammation in the Airways in Krakow’s Atopic and Non-Atopic Residents. J. Clin. Med. 2021, 10, 2383. https://doi.org/10.3390/jcm10112383
Czarnobilska E, Bulanda M, Bulanda D, Mazur M. The Influence of Air Pollution on the Development of Allergic Inflammation in the Airways in Krakow’s Atopic and Non-Atopic Residents. Journal of Clinical Medicine. 2021; 10(11):2383. https://doi.org/10.3390/jcm10112383
Chicago/Turabian StyleCzarnobilska, Ewa, Małgorzata Bulanda, Daniel Bulanda, and Marcel Mazur. 2021. "The Influence of Air Pollution on the Development of Allergic Inflammation in the Airways in Krakow’s Atopic and Non-Atopic Residents" Journal of Clinical Medicine 10, no. 11: 2383. https://doi.org/10.3390/jcm10112383
APA StyleCzarnobilska, E., Bulanda, M., Bulanda, D., & Mazur, M. (2021). The Influence of Air Pollution on the Development of Allergic Inflammation in the Airways in Krakow’s Atopic and Non-Atopic Residents. Journal of Clinical Medicine, 10(11), 2383. https://doi.org/10.3390/jcm10112383






