Candida Infection Associated with Salivary Gland—A Narrative Review
Abstract
1. Introduction
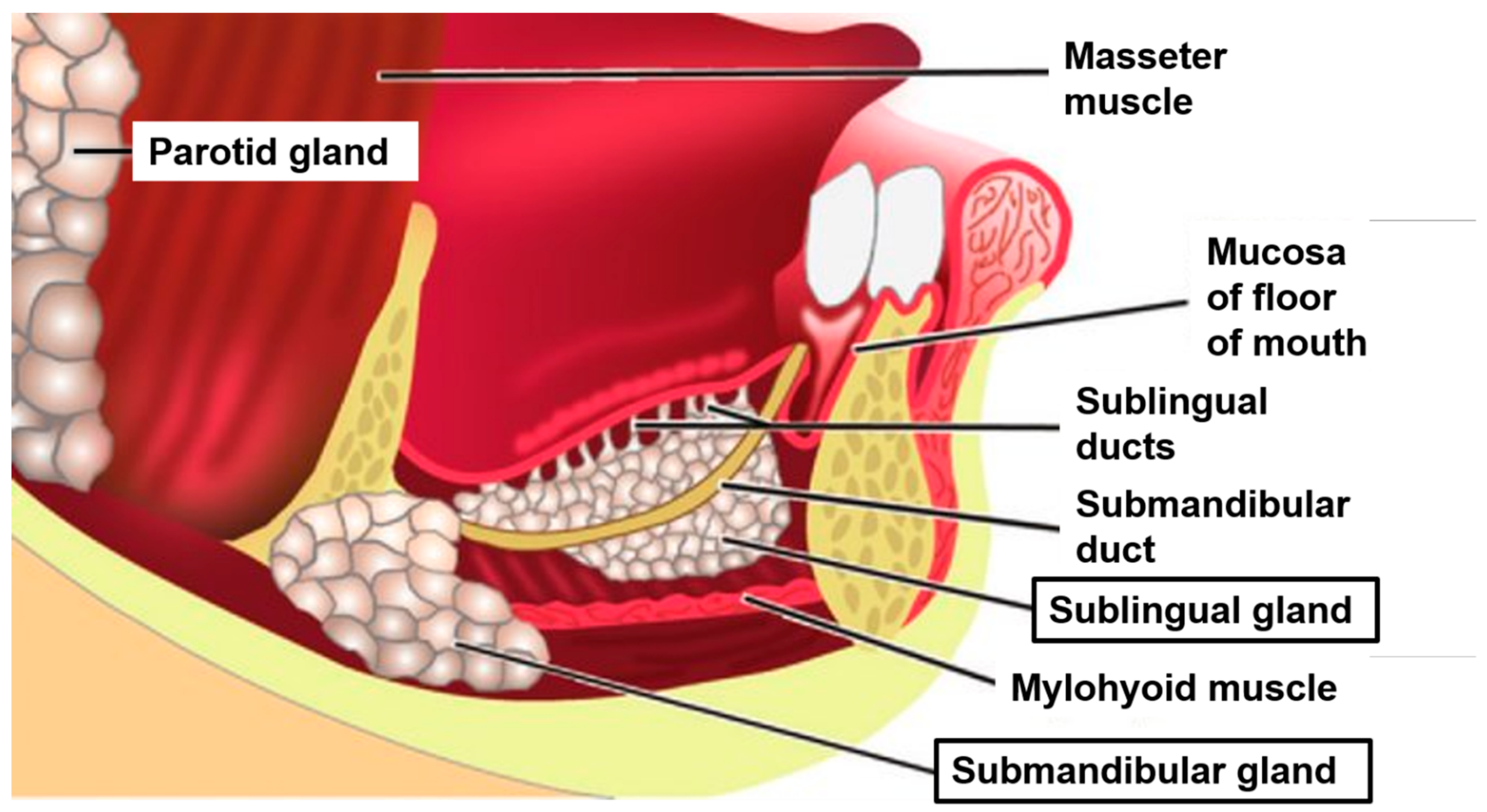
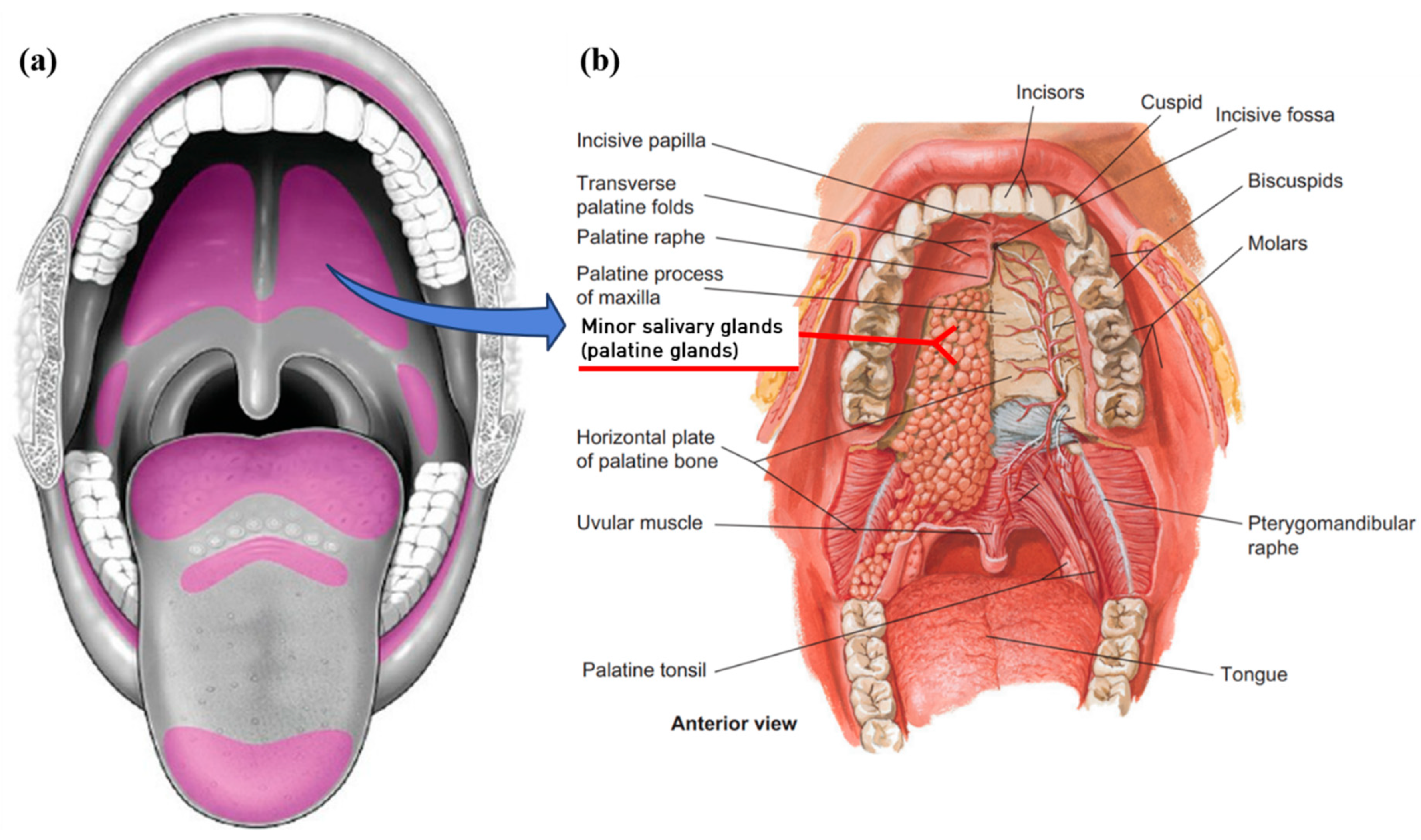
2. Anatomy and Function of the Salivary Glands
3. Pathology of Salivary Glands
3.1. Infectious Diseases Involving Virus, Bacteria, and Fungi
3.1.1. Viral and Bacterial Infectious Diseases
3.1.2. Infectious Diseases with Candida
3.2. Non-Infectious Inflammatory Disease
3.3. Secondary Candida Infection with Salivary Gland Tumors
4. Candida Infection and Salivary Gland Function
4.1. Candida Infection Affecting Salivation
4.2. Candida Infection and Host Immune Response
5. Diagnosis of Oral Candidiasis
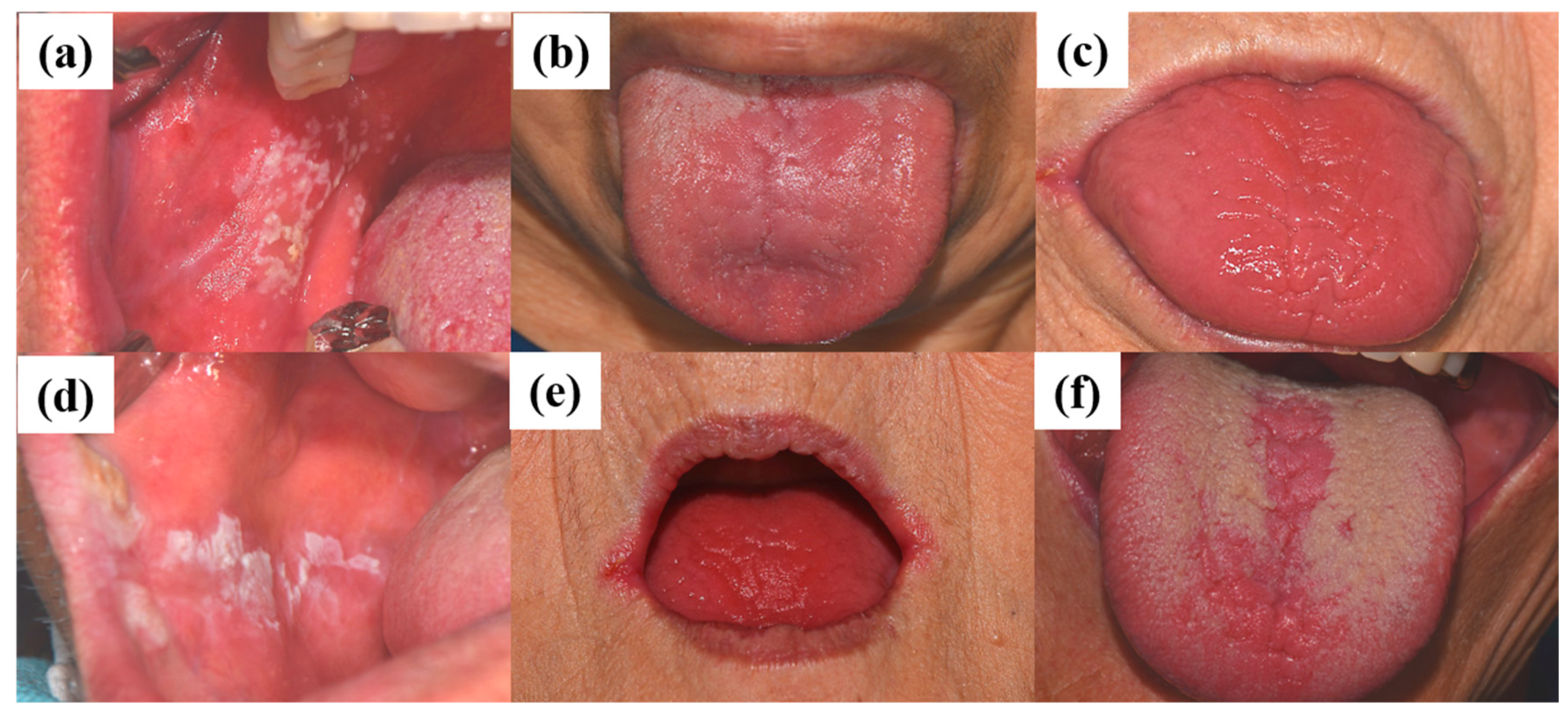
5.1. Tentative Diagnosis Using Clinical Features and Characteristics
5.2. Definite Diagnosis Using Cytology and Culture
6. Prevention and Treatments of Oral Candidiasis
6.1. Prevention
6.2. Treatments of Candida Infection
6.3. Treatments of Salivary Gland Dysfunction
6.3.1. Symptomatic Management
6.3.2. Gene Therapy and Cellular Stimulation
6.3.3. Stem Cell Therapy
6.3.4. Tissue Engineering
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Hellquist, H.; Skalova, A. Histopathology of the Salivary Glands; Springer: Berlin/Heidelberg, Germany, 2014. [Google Scholar]
- Aframian, D.J.; Keshet, N.; Nadler, C.; Zadik, Y.; Vered, M. Minor salivary glands: Clinical, histological and immunohistochemical features of common and less common pathologies. Acta Histochem. 2019, 121, 151451. [Google Scholar] [CrossRef] [PubMed]
- Amano, O.; Mizobe, K.; Bando, Y.; Sakiyama, K. Anatomy and histology of rodent and human major salivary glands: -Overview of the Japan salivary gland society-sponsored workshop-. Acta Histochem. Cytochem. 2012, 45, 241–250. [Google Scholar] [CrossRef] [PubMed]
- Hand, A.R.; Pathmanathan, D.; Field, R.B. Morphological features of the minor salivary glands. Arch. Oral Biol. 1999, 44 (Suppl. S1), S3–S10. [Google Scholar] [CrossRef]
- Won, S.; Kho, H.; Kim, Y.; Chung, S.; Lee, S. Analysis of residual saliva and minor salivary gland secretions. Arch. Oral Biol. 2001, 46, 619–624. [Google Scholar] [CrossRef]
- Rayment, S.A.; Liu, B.; Offner, G.D.; Oppenheim, F.G.; Troxler, R.F. Immunoquantification of human salivary mucins MG1 and MG2 in stimulated whole saliva: Factors influencing mucin levels. J. Dent. Res. 2000, 79, 1765–1772. [Google Scholar]
- Smith, D.J.; Joshipura, K.; Kent, R.; Taubman, M.A. Effect of age on immunoglobulin content and volume of human labial gland saliva. J. Dent. Res. 1992, 71, 1891–1894. [Google Scholar] [CrossRef]
- Rudney, J.D.; Krig, M.A.; Neuvar, E.K.; Soberay, A.H.; Iverson, L. Antimicrobial proteins in human unstimulated whole saliva in relation to each other, and to measures of health status, dental plaque accumulation and composition. Arch. Oral Biol. 1991, 36, 497–506. [Google Scholar] [CrossRef]
- Skinner, C.E.; Fletcher, D.W. A review of the genus candida. Bacteriol. Rev. 1960, 24, 397–416. [Google Scholar] [CrossRef]
- Enache-Angoulvant, A.; Torti, F.; Tassart, M.; Poirot, J.-L.; Jafari, A.; Roux, P.; Hennequin, C. Candidal abscess of the parotid gland due to Candida glabrata: Report of a case and literature review. Med. Mycol. 2010, 48, 402–405. [Google Scholar] [CrossRef]
- Edgerton, M.; Koshlukova, S.E. Salivary histatin 5 and its similarities to the other antimicrobial proteins in human saliva. Adv. Dent Res. 2000, 14, 16–21. [Google Scholar] [CrossRef]
- Tsui, C.; Kong, E.F.; Jabra-Rizk, M.A. Pathogenesis of Candida albicans biofilm. Pathog. Dis. 2016, 74, ftw018. [Google Scholar] [CrossRef] [PubMed]
- Reichart, P.A.; Samaranayake, L.P.; Philipsen, H.P. Pathology and clinical correlates in oral candidiasis and its variants: A review. Oral Dis. 2000, 6, 85–91. [Google Scholar] [CrossRef] [PubMed]
- Muadcheingka, T.; Tantivitayakul, P. Distribution of Candida albicans and non-albicans Candida species in oral candidiasis patients: Correlation between cell surface hydrophobicity and biofilm forming activities. Arch. Oral Biol. 2015, 60, 894–901. [Google Scholar] [CrossRef] [PubMed]
- Akpan, A.; Morgan, R. Oral candidiasis. Postgrad. Med. J. 2002, 78, 455–459. [Google Scholar] [CrossRef] [PubMed]
- Barnett, J.A. A history of research on yeasts 12: Medical yeasts part 1, Candida albicans. Yeast 2008, 25, 385–417. [Google Scholar] [CrossRef] [PubMed]
- Scully, C. Oral and Maxillofacial Medicine: The Basis of Diagnosis and Treatment, 2nd ed.; Elsevier: Philadelphia, PA, USA, 2008; pp. 191–200. [Google Scholar]
- Lalla, R.V.; Patton, L.L.; Dongari-Bagtzoglou, A. Oral candidiasis: Pathogenesis, clinical presentation, diagnosis and treatment strategies. J. Calif. Dent. Assoc. 2013, 41, 263–268. [Google Scholar]
- Vila, T.; Sultan, A.S.; Montelongo-Jauregui, D.; Jabra-Rizk, M.A. Oral candidiasis: A disease of opportunity. J. Fungi 2020, 6, 15. [Google Scholar] [CrossRef]
- Ogle, O.E. Salivary Gland Diseases. Dent. Clin. N. Am. 2020, 64, 87–104. [Google Scholar] [CrossRef]
- Ochiai, A.; Harada, K.; Hashimoto, K.; Shibata, K.; Ishiyama, Y.; Mitsui, T.; Tanaka, T.; Taniguchi, M. α-Amylase is a potential growth inhibitor of Porphyromonas gingivalis, a periodontal pathogenic bacterium. J. Periodontal Res. 2014, 49, 62–68. [Google Scholar] [CrossRef]
- Kondo, Y.; Nakamoto, T.; Jaramillo, Y.; Choi, S.; Catalan, M.A.; Melvin, J.E. Functional differences in the acinar cells of the murine major salivary glands. J. Dent. Res. 2015, 94, 715–721. [Google Scholar] [CrossRef]
- La’porte, S.J.; Juttla, J.K.; Lingam, R.K. Imaging the floor of the mouth and the sublingual space. Radiographics 2011, 31, 1215–1230. [Google Scholar] [CrossRef] [PubMed]
- Iorgulescu, G. Saliva between normal and pathological. Important factors in determining systemic and oral health. J. Med. Life 2009, 2, 303–307. [Google Scholar]
- Eliasson, L.; Birkhed, D.; Carlén, A. Feeling of dry mouth in relation to whole and minor gland saliva secretion rate. Arch. Oral Biol. 2009, 54, 263–267. [Google Scholar] [CrossRef] [PubMed]
- Bretz, W.A.; Loesche, W.J.; Chen, Y.M.; Schork, M.A.; Dominguez, B.L.; Grossman, N. Minor salivary gland secretion in the elderly. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endod. 2000, 89, 696–701. [Google Scholar] [CrossRef] [PubMed]
- Dijkema, T.; Raaijmakers, C.P.J.; Braam, P.M.; Roesink, J.M.; Monninkhof, E.M.; Terhaard, C.H.J. Xerostomia: A day and night difference. Radiother. Oncol. 2012, 104, 219–223. [Google Scholar] [CrossRef] [PubMed]
- Eliasson, L.; Carlén, A. An update on minor salivary gland secretions. Eur. J. Oral Sci. 2010, 118, 435–442. [Google Scholar] [CrossRef] [PubMed]
- Shern, R.J.; Fox, P.C.; Cain, J.L.; Li, S.H. A method for measuring the flow of saliva from the minor salivary glands. J. Dent. Res. 1990, 69, 1146–1149. [Google Scholar] [CrossRef] [PubMed]
- Marcotte, H.; Lavoie, M.C. Oral microbial ecology and the role of salivary immunoglobulin A. Microbiol. Mol. Biol. Rev. 1998, 62, 71–109. [Google Scholar] [CrossRef]
- Kessler, A.T.; Bhatt, A.A. Review of the major and minor salivary glands, part 1: Anatomy, infectious, and inflammatory processes. J. Clin. Imaging Sci. 2018, 8, 47. [Google Scholar] [CrossRef]
- Treuting, P.M.; Morton, T.H. Oral cavity and teeth. In Comparative Anatomy and Histology; Elsevier: Amsterdam, The Netherlands, 2012; pp. 95–110. [Google Scholar]
- Matsuo, R. Role of saliva in the maintenance of taste sensitivity. Crit. Rev. Oral Biol. Med. 2000, 11, 216–229. [Google Scholar] [CrossRef]
- Yoshikawa, H.; Furuta, K.; Ueno, M.; Egawa, M.; Yoshino, A.; Kondo, S.; Nariai, Y.; Ishibashi, H.; Kinoshita, Y.; Sekine, J. Oral symptoms including dental erosion in gastroesophageal reflux disease are associated with decreased salivary flow volume and swallowing function. J. Gastroenterol. 2012, 47, 412–420. [Google Scholar] [CrossRef] [PubMed]
- Hviid, A.; Rubin, S.; Mühlemann, K. Mumps. Lancet 2008, 371, 932–944. [Google Scholar] [CrossRef]
- Gupta, M.; Shorman, M. Cytomegalovirus. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2020. [Google Scholar]
- Xu, J.; Li, Y.; Gan, F.; Du, Y.; Yao, Y. Salivary Glands: Potential Reservoirs for COVID-19 Asymptomatic Infection. J. Dent. Res. 2020, 99, 989. [Google Scholar] [CrossRef]
- Pascolo, L.; Zupin, L.; Melato, M.; Tricarico, P.M.; Crovella, S. TMPRSS2 and ACE2 Coexpression in SARS-CoV-2 Salivary Glands Infection. J. Dent. Res. 2020, 99, 1120–1121. [Google Scholar] [CrossRef]
- Fisher, J.; Monette, D.L.; Patel, K.R.; Kelley, B.P.; Kennedy, M. COVID-19 associated parotitis: A case report. Am. J. Emerg. Med. 2020. [Google Scholar] [CrossRef]
- Chern, A.; Famuyide, A.O.; Moonis, G.; Lalwani, A.K. Sialadenitis: A Possible Early Manifestation of COVID-19. Laryngoscope 2020, 130, 2595–2597. [Google Scholar] [CrossRef]
- Lechien, J.R.; Chetrit, A.; Chekkoury-Idrissi, Y.; Distinguin, L.; Circiu, M.; Saussez, S.; Berradja, N.; Edjlali, M.; Hans, S.; Carlier, R. Parotitis-Like Symptoms Associated with COVID-19, France, March-April 2020. Emerg. Infect. Dis. 2020, 26, 2270. [Google Scholar] [CrossRef]
- Mariette, X.; Gozlan, J.; Clerc, D.; Bisson, M.; Morinet, F. Detection of Epstein-Barr virus DNA by in situ hybridization and polymerase chain reaction in salivary gland biopsy specimens from patients with Sjögren’s syndrome. Am. J. Med. 1991, 90, 286–294. [Google Scholar] [CrossRef]
- Rivera, H.; Nikitakis, N.G.; Castillo, S.; Siavash, H.; Papadimitriou, J.C.; Sauk, J.J. Histopathological analysis and demonstration of EBV and HIV p-24 antigen but not CMV expression in labial minor salivary glands of HIV patients affected by diffuse infiltrative lymphocytosis syndrome. J. Oral Pathol. Med. 2003, 32, 431–437. [Google Scholar] [CrossRef]
- Mariette, X.; Agbalika, F.; Zucker-Franklin, D.; Clerc, D.; Janin, A.; Cherot, P.; Brouet, J.C. Detection of the tax gene of HTLV-I in labial salivary glands from patients with Sjögren’s syndrome and other diseases of the oral cavity. Clin. Exp. Rheumatol. 2000, 18, 341–347. [Google Scholar]
- Ugga, L.; Ravanelli, M.; Pallottino, A.A.; Farina, D.; Maroldi, R. Diagnostic work-up in obstructive and inflammatory salivary gland disorders. Acta Otorhinolaryngol. Ital. 2017, 37, 83–93. [Google Scholar] [PubMed]
- Wray, W.; Scully, C.; Rennie, J.; Mason, D.K.; Love, W.C. Major and minor salivary gland swelling in Mycoplasma pneumoniae infection. Br. Med. J. 1980, 280, 1421. [Google Scholar] [CrossRef] [PubMed]
- De Gomes, P.S.; Juodzbalys, G.; Fernandes, M.H.; Guobis, Z. Diagnostic Approaches to Sjögren’s syndrome: A Literature Review and Own Clinical Experience. J. Oral Maxillofac. Res. 2012, 3, e3. [Google Scholar] [CrossRef] [PubMed]
- Even-Tov, E.; Niv, A.; Kraus, M.; Nash, M. Candida parotitis with abscess formation. Acta Otolaryngol. 2006, 126, 334–336. [Google Scholar] [CrossRef]
- Kantarcioğlu, A.S.; Gulenc, M.; Yücel, A.; Uzun, N.; Taskin, T.; Sakiz, D.; Altas, K. Cryptococcal parotid involvement: An uncommon localization of Cryptococcus neoformans. Med. Mycol. 2006, 44, 279–283. [Google Scholar] [CrossRef][Green Version]
- Marioni, G.; Rinaldi, R.; de Filippis, C.; Gaio, E.; Staffieri, A. Candidal abscess of the parotid gland associated with facial nerve paralysis. Acta Otolaryngol. 2003, 123, 661–663. [Google Scholar] [CrossRef]
- Namiq, A.L.; Tollefson, T.; Fan, F. Cryptococcal parotitis presenting as a cystic parotid mass: Report of a case diagnosed by fine-needle aspiration cytology. Diagn. Cytopathol. 2005, 33, 36–38. [Google Scholar] [CrossRef]
- Stefanopoulos, P.K.; Karakassis, D.T.; Triantafyllidou, A. Stensen’s duct obstruction by foreign body and subsequent candidal infection of the parotid gland. J. Laryngol. Otol. 2003, 117, 662–665. [Google Scholar] [CrossRef]
- Vargas, P.A.; Mauad, T.; Böhm, G.M.; Saldiva, P.H.N.; Almeida, O.P. Parotid gland involvement in advanced AIDS. Oral Dis. 2003, 9, 55–61. [Google Scholar] [CrossRef]
- Souza Filho, F.J.; Lopes, M.; Almeida, O.P.; Scully, C. Mucocutaneous histoplasmosis in AIDS. Br. J. Dermatol. 1995, 133, 472–474. [Google Scholar] [CrossRef]
- De Repentigny, L.; Lewandowski, D.; Jolicoeur, P. Immunopathogenesis of oropharyngeal candidiasis in human immunodeficiency virus infection. Clin. Microbiol. Rev. 2004, 17, 729–759. [Google Scholar] [CrossRef]
- Liang, Y.; Yang, Z.; Qin, B.; Zhong, R. Primary Sjogren’s syndrome and malignancy risk: A systematic review and meta-analysis. Ann. Rheum. Dis. 2014, 73, 1151–1156. [Google Scholar] [CrossRef]
- Wei, T.-W.; Lien, C.-F.; Hsu, T.-Y.; He, H.-L. Chronic sclerosing sialadenitis of the submandibular gland: An entity of IgG4-related sclerosing disease. Int. J. Clin. Exp. Pathol. 2015, 8, 8628–8631. [Google Scholar]
- Geyer, J.T.; Ferry, J.A.; Harris, N.L.; Stone, J.H.; Zukerberg, L.R.; Lauwers, G.Y.; Pilch, B.Z.; Deshpande, V. Chronic sclerosing sialadenitis (Küttner tumor) is an IgG4-associated disease. Am. J. Surg. Pathol. 2010, 34, 202–210. [Google Scholar] [CrossRef]
- Speight, P.M.; Barrett, A.W. Salivary gland tumours. Oral Dis. 2002, 8, 229–240. [Google Scholar] [CrossRef]
- Venkateswaran, L.; Gan, Y.J.; Sixbey, J.W.; Santana, V.M. Epstein-Barr virus infection in salivary gland tumors in children and young adults. Cancer 2000, 89, 463–466. [Google Scholar] [CrossRef]
- Mozaffari, H.R.; Ramezani, M.; Janbakhsh, A.; Sadeghi, M. Malignant Salivary Gland Tumors and Epstein-Barr Virus (EBV) Infection: A Systematic Review and Meta-Analysis. Asian Pac. J. Cancer Prev. 2017, 18, 1201–1206. [Google Scholar]
- Raab-Traub, N.; Rajadurai, P.; Flynn, K.; Lanier, A.P. Epstein-Barr virus infection in carcinoma of the salivary gland. J. Virol. 1991, 65, 7032–7036. [Google Scholar] [CrossRef]
- Sujatha, D.; Babitha, K.; Prasad, R.S.; Pai, A. Parotid lymphoepithelial cysts in human immunodeficiency virus: A review. J. Laryngol. Otol. 2013, 127, 1046–1049. [Google Scholar] [CrossRef]
- Nagao, Y.; Hashimoto, K.; Sata, M. Candidiasis and other oral mucosal lesions during and after interferon therapy for HCV-related chronic liver diseases. BMC Gastroenterol. 2012, 12, 155. [Google Scholar] [CrossRef]
- Sankari, S.L.; Gayathri, K.; Balachander, N.; Malathi, L. Candida in potentially malignant oral disorders. J. Pharm. Bioallied Sci. 2015, 7, S162–S164. [Google Scholar]
- Archambault, L.S.; Trzilova, D.; Gonia, S.; Gale, C.; Wheeler, R.T. Intravital Imaging Reveals Divergent Cytokine and Cellular Immune Responses to Candida albicans and Candida parapsilosis. MBio 2019, 10. [Google Scholar] [CrossRef]
- Ohta, H.; Tanimoto, T.; Taniai, M.; Taniguchi, M.; Ariyasu, T.; Arai, S.; Ohta, T.; Fukuda, S. Regulation of Candida albicans morphogenesis by tumor necrosis factor-alpha and potential for treatment of oral candidiasis. In Vivo 2007, 21, 25–32. [Google Scholar]
- Okamura, M.; Moto, M.; Kashida, Y.; Machida, N.; Mitsumori, K. Carcinogenic susceptibility to N-bis(2-hydroxypropyl)nitrosamine (DHPN) in rasH2 mice. Toxicol. Pathol. 2004, 32, 474–481. [Google Scholar] [CrossRef]
- Kim, D.; Hwang, Y.-I.; Choi, S.; Park, C.; Lee, N.; Kim, E.-A. A case of tracheal adenoid cystic carcinoma in a worker exposed to rubber fumes. Ann. Occup. Environ. Med. 2013, 25, 22. [Google Scholar] [CrossRef]
- Murtaugh, L.C.; Keefe, M.D. Regeneration and repair of the exocrine pancreas. Annu. Rev. Physiol. 2015, 77, 229–249. [Google Scholar] [CrossRef]
- Mahalakshmi, S.; Kandula, S.; Shilpa, P.; Kokila, G. Chronic Recurrent Non-specific Parotitis: A Case Report and Review. Ethiop. J. Health Sci. 2017, 27, 95–100. [Google Scholar] [CrossRef]
- Tanida, T.; Okamoto, T.; Okamoto, A.; Wang, H.; Hamada, T.; Ueta, E.; Osaki, T. Decreased excretion of antimicrobial proteins and peptides in saliva of patients with oral candidiasis. J. Oral Pathol. Med. 2003, 32, 586–594. [Google Scholar] [CrossRef]
- Lynge Pedersen, A.M.; Belstrøm, D. The role of natural salivary defences in maintaining a healthy oral microbiota. J. Dent. 2019, 80 (Suppl. S1), S3–S12. [Google Scholar] [CrossRef]
- Ciociola, T.; Giovati, L.; Conti, S.; Magliani, W.; Santinoli, C.; Polonelli, L. Natural and synthetic peptides with antifungal activity. Future Med. Chem. 2016, 8, 1413–1433. [Google Scholar] [CrossRef]
- Edgerton, M.; Koshlukova, S.E.; Araujo, M.W.; Patel, R.C.; Dong, J.; Bruenn, J.A. Salivary histatin 5 and human neutrophil defensin 1 kill Candida albicans via shared pathways. Antimicrob. Agents Chemother. 2000, 44, 3310–3316. [Google Scholar] [CrossRef]
- Sahasrabudhe, K.S.; Kimball, J.R.; Morton, T.H.; Weinberg, A.; Dale, B.A. Expression of the antimicrobial peptide, human beta-defensin 1, in duct cells of minor salivary glands and detection in saliva. J. Dent. Res. 2000, 79, 1669–1674. [Google Scholar] [CrossRef]
- Nadig, S.D.; Ashwathappa, D.T.; Manjunath, M.; Krishna, S.; Annaji, A.G.; Shivaprakash, P.K. A relationship between salivary flow rates and Candida counts in patients with xerostomia. J. Oral Maxillofac. Pathol. 2017, 21, 316. [Google Scholar] [CrossRef]
- Karbach, J.; Walter, C.; Al-Nawas, B. Evaluation of saliva flow rates, Candida colonization and susceptibility of Candida strains after head and neck radiation. Clin. Oral Investig. 2012, 16, 1305–1312. [Google Scholar] [CrossRef]
- Ohga, N.; Yamazaki, Y.; Sato, J.; Asaka, T.; Morimoto, M.; Hata, H.; Satoh, C.; Kitagawa, Y. Elimination of oral candidiasis may increase stimulated whole salivary flow rate. Arch. Oral Biol. 2016, 71, 129–133. [Google Scholar] [CrossRef]
- Porcheri, C.; Mitsiadis, T.A. Physiology, pathology and regeneration of salivary glands. Cells 2019, 8, 976. [Google Scholar] [CrossRef]
- Deshmukh, U.S.; Nandula, S.R.; Thimmalapura, P.-R.; Scindia, Y.M.; Bagavant, H. Activation of innate immune responses through Toll-like receptor 3 causes a rapid loss of salivary gland function. J. Oral Pathol. Med. 2009, 38, 42–47. [Google Scholar] [CrossRef]
- Ferretti, S.; Bonneau, O.; Dubois, G.R.; Jones, C.E.; Trifilieff, A. IL-17, produced by lymphocytes and neutrophils, is necessary for lipopolysaccharide-induced airway neutrophilia: IL-15 as a possible trigger. J. Immunol. 2003, 170, 2106–2112. [Google Scholar] [CrossRef]
- Kany, S.; Vollrath, J.T.; Relja, B. Cytokines in inflammatory disease. Int. J. Mol. Sci. 2019, 20, 6008. [Google Scholar] [CrossRef]
- Festa, A.; D’Agostino, R.; Howard, G.; Mykkänen, L.; Tracy, R.P.; Haffner, S.M. Chronic subclinical inflammation as part of the insulin resistance syndrome: The Insulin Resistance Atherosclerosis Study (IRAS). Circulation 2000, 102, 42–47. [Google Scholar] [CrossRef]
- Ohyama, Y.; Carroll, V.A.; Deshmukh, U.; Gaskin, F.; Brown, M.G.; Fu, S.M. Severe focal sialadenitis and dacryoadenitis in NZM2328 mice induced by MCMV: A novel model for human Sjögren’s syndrome. J. Immunol. 2006, 177, 7391–7397. [Google Scholar] [CrossRef] [PubMed]
- King, L.S.; Yasui, M. Aquaporins and disease: Lessons from mice to humans. Trends Endocrinol. Metab. 2002, 13, 355–360. [Google Scholar] [CrossRef]
- Yamamura, Y.; Motegi, K.; Kani, K.; Takano, H.; Momota, Y.; Aota, K.; Yamanoi, T.; Azuma, M. TNF-α inhibits aquaporin 5 expression in human salivary gland acinar cells via suppression of histone H4 acetylation. J. Cell Mol. Med. 2012, 16, 1766–1775. [Google Scholar] [CrossRef] [PubMed]
- Williams, D.W.; Lewis, M.A.O. Oral Microbiology: Isolation and identification of candida from the oral cavity. Oral Dis. 2008, 6, 3–11. [Google Scholar] [CrossRef]
- Hellstein, J.W.; Marek, C.L. Candidiasis: Red and white manifestations in the oral cavity. Head Neck Pathol. 2019, 13, 25–32. [Google Scholar] [CrossRef]
- Williams, D.; Lewis, M. Pathogenesis and treatment of oral candidosis. J. Oral Microbiol. 2011, 3, 3. [Google Scholar] [CrossRef]
- Reamy, B.V.; Derby, R.; Bunt, C.W. Common tongue conditions in primary care. Am. Fam. Physician 2010, 81, 627–634. [Google Scholar]
- Khan, T.S.; Muddebihal, F.; Koshy, A. Chronic atrophic candidiasis: A case report and review of literature. Univ. Res. J. Dent. 2015, 5, 123. [Google Scholar] [CrossRef]
- Aoun, G.; Berberi, A. Prevalence of Chronic Erythematous Candidiasis in Lebanese Denture Wearers: A Clinico-microbiological Study. Mater. Sociomed. 2017, 29, 26–29. [Google Scholar] [CrossRef]
- Sitheeque, M.A.M.; Samaranayake, L.P. Chronic hyperplastic candidosis/candidiasis (candidal leukoplakia). Crit. Rev. Oral Biol. Med. 2003, 14, 253–267. [Google Scholar] [CrossRef]
- Sharon, V.; Fazel, N. Oral candidiasis and angular cheilitis. Dermatol. Ther. 2010, 23, 230–242. [Google Scholar] [CrossRef] [PubMed]
- Zhou, P.R.; Hua, H.; Liu, X.S. Quantity of candida colonies in saliva: A diagnostic evaluation for oral candidiasis. Chin. J. Dent. Res. 2017, 20, 27–32. [Google Scholar] [PubMed]
- Coronado-Castellote, L.; Jiménez-Soriano, Y. Clinical and microbiological diagnosis of oral candidiasis. J. Clin. Exp. Dent. 2013, 5, e279–e286. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Cuesta, C.; Sarrion-Pérez, M.-G.; Bagán, J.V. Current treatment of oral candidiasis: A literature review. J. Clin. Exp. Dent. 2014, 6, e576–e582. [Google Scholar] [CrossRef]
- Vigneswaran, N.; Muller, S. Pharmacologic management of oral mucosal inflammatory and ulcerative diseases. In Contemporary Dental Pharmacology: Evidence-Based Considerations; Jeske, A.H., Ed.; Springer International Publishing: Cham, Switzerland, 2019; pp. 91–108. [Google Scholar]
- Aguirre Urizar, J.M. Oral candidiasis. Rev. Iberoam Micol. 2002, 19, 17–21. [Google Scholar]
- Martínez-Beneyto, Y.; López-Jornet, P.; Velandrino-Nicolás, A.; Jornet-García, V. Use of antifungal agents for oral candidiasis: Results of a national survey. Int. J. Dent. Hyg. 2010, 8, 47–52. [Google Scholar] [CrossRef]
- Odds, F.C.; Brown, A.J.P.; Gow, N.A.R. Antifungal agents: Mechanisms of action. Trends Microbiol. 2003, 11, 272–279. [Google Scholar] [CrossRef]
- Lombardi, A.; Ouanounou, A. Fungal infections in dentistry: Clinical presentations, diagnosis, and treatment alternatives. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. 2020, 130, 533–546. [Google Scholar] [CrossRef]
- Parente-Rocha, J.A.; Bailão, A.M.; Amaral, A.C.; Taborda, C.P.; Paccez, J.D.; Borges, C.L.; Pereira, M. Antifungal Resistance, Metabolic Routes as Drug Targets, and New Antifungal Agents: An Overview about Endemic Dimorphic Fungi. Mediators Inflamm. 2017, 2017, 9870679. [Google Scholar] [CrossRef]
- Cowen, L.E.; Sanglard, D.; Howard, S.J.; Rogers, P.D.; Perlin, D.S. Mechanisms of Antifungal Drug Resistance. Cold Spring Harb. Perspect. Med. 2014, 5, a019752. [Google Scholar] [CrossRef]
- Whaley, S.G.; Berkow, E.L.; Rybak, J.M.; Nishimoto, A.T.; Barker, K.S.; Rogers, P.D. Azole Antifungal Resistance in Candida albicans and Emerging Non-albicans Candida Species. Front. Microbiol. 2016, 7, 2173. [Google Scholar] [CrossRef] [PubMed]
- Chang, Y.-L.; Yu, S.-J.; Heitman, J.; Wellington, M.; Chen, Y.-L. New facets of antifungal therapy. Virulence 2017, 8, 222–236. [Google Scholar] [CrossRef] [PubMed]
- Pianalto, K.; Alspaugh, J. New horizons in antifungal therapy. J. Fungi 2016, 2, 26. [Google Scholar] [CrossRef] [PubMed]
- Ghannoum, M.A.; Rice, L.B. Antifungal agents: Mode of action, mechanisms of resistance, and correlation of these mechanisms with bacterial resistance. Clin. Microbiol. Rev. 1999, 12, 501–517. [Google Scholar] [CrossRef]
- Kumamoto, C.A. Candida biofilms. Curr. Opin. Microbiol. 2002, 5, 608–611. [Google Scholar] [CrossRef]
- Vissink, A.; Mitchell, J.B.; Baum, B.J.; Limesand, K.H.; Jensen, S.B.; Fox, P.C.; Elting, L.S.; Langendijk, J.A.; Coppes, R.P.; Reyland, M.E. Clinical management of salivary gland hypofunction and xerostomia in head-and-neck cancer patients: Successes and barriers. Int. J. Radiat. Oncol. Biol. Phys. 2010, 78, 983–991. [Google Scholar] [CrossRef]
- Silvestre, F.J.; Minguez, M.P.; Suñe-Negre, J.M. Clinical evaluation of a new artificial saliva in spray form for patients with dry mouth. Med. Oral Patol. Oral Cir. Bucal 2009, 14, E8–E11. [Google Scholar]
- Jansma, J.; Vissink, A.; Spijkervet, F.K.; Roodenburg, J.L.; Panders, A.K.; Vermey, A.; Szabó, B.G.; Gravenmade, E.J. Protocol for the prevention and treatment of oral sequelae resulting from head and neck radiation therapy. Cancer 1992, 70, 2171–2180. [Google Scholar] [CrossRef]
- Jellema, A.P.; Slotman, B.J.; Doornaert, P.; Leemans, C.R.; Langendijk, J.A. Impact of radiation-induced xerostomia on quality of life after primary radiotherapy among patients with head and neck cancer. Int. J. Radiat. Oncol. Biol. Phys. 2007, 69, 751–760. [Google Scholar] [CrossRef]
- Gibson, J.; Halliday, J.A.; Ewert, K.; Robertson, S. A controlled release pilocarpine buccal insert in the treatment of Sjögren’s syndrome. Br. Dent. J. 2007, 202, E17. [Google Scholar] [CrossRef]
- Tabata, Y. Tissue regeneration based on growth factor release. Tissue Eng. 2003, 9 (Suppl. S1), S5–S15. [Google Scholar] [CrossRef]
- Richards Grayson, A.C.; Choi, I.S.; Tyler, B.M.; Wang, P.P.; Brem, H.; Cima, M.J.; Langer, R. Multi-pulse drug delivery from a resorbable polymeric microchip device. Nat. Mater. 2003, 2, 767–772. [Google Scholar] [CrossRef] [PubMed]
- Hirsch, L.R.; Gobin, A.M.; Lowery, A.R.; Tam, F.; Drezek, R.A.; Halas, N.J.; West, J.L. Metal nanoshells. Ann. Biomed. Eng. 2006, 34, 15–22. [Google Scholar] [CrossRef] [PubMed]
- Sershen, S.R.; Mensing, G.A.; Ng, M.; Halas, N.J.; Beebe, D.J.; West, J.L. Independent Optical Control of Microfluidic Valves Formed from Optomechanically Responsive Nanocomposite Hydrogels. Adv. Mater. Weinheim 2005, 17, 1366–1368. [Google Scholar] [CrossRef]
- Delporte, C.; O’Connell, B.C.; He, X.; Lancaster, H.E.; O’Connell, A.C.; Agre, P.; Baum, B.J. Increased fluid secretion after adenoviral-mediated transfer of the aquaporin-1 cDNA to irradiated rat salivary glands. Proc. Natl. Acad. Sci. USA 1997, 94, 3268–3273. [Google Scholar] [CrossRef] [PubMed]
- Alevizos, I.; Zheng, C.; Cotrim, A.P.; Liu, S.; McCullagh, L.; Billings, M.E.; Goldsmith, C.M.; Tandon, M.; Helmerhorst, E.J.; Catalán, M.A.; et al. Late responses to adenoviral-mediated transfer of the aquaporin-1 gene for radiation-induced salivary hypofunction. Gene Ther. 2017, 24, 176–186. [Google Scholar] [CrossRef]
- Samuni, Y.; Baum, B.J. Gene delivery in salivary glands: From the bench to the clinic. Biochim. Biophys. Acta 2011, 1812, 1515–1521. [Google Scholar] [CrossRef]
- Varghese, J.J.; Schmale, I.L.; Wang, Y.; Hansen, M.E.; Newlands, S.D.; Ovitt, C.E.; Benoit, D.S.W. Retroductal nanoparticle injection to the murine submandibular gland. J. Vis. Exp. 2018, 135, e57521. [Google Scholar] [CrossRef]
- Shan, Z.; Li, J.; Zheng, C.; Liu, X.; Fan, Z.; Zhang, C.; Goldsmith, C.M.; Wellner, R.B.; Baum, B.J.; Wang, S. Increased fluid secretion after adenoviral-mediated transfer of the human aquaporin-1 cDNA to irradiated miniature pig parotid glands. Mol. Ther. 2005, 11, 444–451. [Google Scholar] [CrossRef]
- Redman, R.S.; Ball, W.D.; Mezey, E.; Key, S. Dispersed donor salivary gland cells are widely distributed in the recipient gland when infused up the ductal tree. Biotech. Histochem. 2009, 84, 253–260. [Google Scholar] [CrossRef]
- Grundmann, O.; Fillinger, J.L.; Victory, K.R.; Burd, R.; Limesand, K.H. Restoration of radiation therapy-induced salivary gland dysfunction in mice by post therapy IGF-1 administration. BMC Cancer 2010, 10, 417. [Google Scholar] [CrossRef] [PubMed]
- Okazaki, Y.; Kagami, H.; Hattori, T.; Hishida, S.; Shigetomi, T.; Ueda, M. Acceleration of rat salivary gland tissue repair by basic fibroblast growth factor. Arch. Oral Biol. 2000, 45, 911–919. [Google Scholar] [CrossRef]
- Zheng, C.; Cotrim, A.P.; Rowzee, A.; Swaim, W.; Sowers, A.; Mitchell, J.B.; Baum, B.J. Prevention of radiation-induced salivary hypofunction following hKGF gene delivery to murine submandibular glands. Clin. Cancer Res. 2011, 17, 2842–2851. [Google Scholar] [CrossRef] [PubMed]
- Marmary, Y.; Adar, R.; Gaska, S.; Wygoda, A.; Maly, A.; Cohen, J.; Eliashar, R.; Mizrachi, L.; Orfaig-Geva, C.; Baum, B.J.; et al. Radiation-Induced Loss of Salivary Gland Function Is Driven by Cellular Senescence and Prevented by IL6 Modulation. Cancer Res. 2016, 76, 1170–1180. [Google Scholar] [CrossRef]
- Lombaert, I.M.A.; Wierenga, P.K.; Kok, T.; Kampinga, H.H.; de Haan, G.; Coppes, R.P. Mobilization of bone marrow stem cells by granulocyte colony-stimulating factor ameliorates radiation-induced damage to salivary glands. Clin. Cancer Res. 2006, 12, 1804–1812. [Google Scholar] [CrossRef] [PubMed]
- Lombaert, I.M.A.; Brunsting, J.F.; Wierenga, P.K.; Kampinga, H.H.; de Haan, G.; Coppes, R.P. Cytokine treatment improves parenchymal and vascular damage of salivary glands after irradiation. Clin. Cancer Res. 2008, 14, 7741–7750. [Google Scholar] [CrossRef]
- Rocchi, C.; Emmerson, E. Mouth-Watering Results: Clinical Need, Current Approaches, and Future Directions for Salivary Gland Regeneration. Trends Mol. Med. 2020, 26, 649–669. [Google Scholar] [CrossRef]
- Jensen, D.H.; Oliveri, R.S.; Trojahn Kølle, S.-F.; Fischer-Nielsen, A.; Specht, L.; Bardow, A.; Buchwald, C. Mesenchymal stem cell therapy for salivary gland dysfunction and xerostomia: A systematic review of preclinical studies. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. 2014, 117, 335–342.e1. [Google Scholar] [CrossRef]
- Lim, J.-Y.; Yi, T.; Choi, J.-S.; Jang, Y.H.; Lee, S.; Kim, H.J.; Song, S.U.; Kim, Y.-M. Intraglandular transplantation of bone marrow-derived clonal mesenchymal stem cells for amelioration of post-irradiation salivary gland damage. Oral Oncol. 2013, 49, 136–143. [Google Scholar] [CrossRef] [PubMed]
- Kojima, T.; Kanemaru, S.-I.; Hirano, S.; Tateya, I.; Ohno, S.; Nakamura, T.; Ito, J. Regeneration of radiation damaged salivary glands with adipose-derived stromal cells. Laryngoscope 2011, 121, 1864–1869. [Google Scholar] [CrossRef] [PubMed]
- Grønhøj, C.; Jensen, D.H.; Glovinski, P.V.; Jensen, S.B.; Bardow, A.; Oliveri, R.S.; Specht, L.; Thomsen, C.; Darkner, S.; Kiss, K.; et al. First-in-man mesenchymal stem cells for radiation-induced xerostomia (MESRIX): Study protocol for a randomized controlled trial. Trials 2017, 18, 108. [Google Scholar] [CrossRef] [PubMed]
- Grønhøj, C.; Jensen, D.H.; Vester-Glowinski, P.; Jensen, S.B.; Bardow, A.; Oliveri, R.S.; Fog, L.M.; Specht, L.; Thomsen, C.; Darkner, S.; et al. Safety and Efficacy of Mesenchymal Stem Cells for Radiation-Induced Xerostomia: A Randomized, Placebo-Controlled Phase 1/2 Trial (MESRIX). Int. J. Radiat. Oncol. Biol. Phys. 2018, 101, 581–592. [Google Scholar] [CrossRef] [PubMed]
- Barkholt, L.; Flory, E.; Jekerle, V.; Lucas-Samuel, S.; Ahnert, P.; Bisset, L.; Büscher, D.; Fibbe, W.; Foussat, A.; Kwa, M.; et al. Risk of tumorigenicity in mesenchymal stromal cell-based therapies--bridging scientific observations and regulatory viewpoints. Cytotherapy 2013, 15, 753–759. [Google Scholar] [CrossRef]
- Van Luijk, P.; Pringle, S.; Deasy, J.O.; Moiseenko, V.V.; Faber, H.; Hovan, A.; Baanstra, M.; van der Laan, H.P.; Kierkels, R.G.J.; van der Schaaf, A.; et al. Sparing the region of the salivary gland containing stem cells preserves saliva production after radiotherapy for head and neck cancer. Sci. Transl. Med. 2015, 7, 305ra147. [Google Scholar] [CrossRef]
- Pringle, S.; Maimets, M.; van der Zwaag, M.; Stokman, M.A.; van Gosliga, D.; Zwart, E.; Witjes, M.J.H.; de Haan, G.; van Os, R.; Coppes, R.P. Human salivary gland stem cells functionally restore radiation damaged salivary glands. Stem Cells 2016, 34, 640–652. [Google Scholar] [CrossRef]
- Ihrler, S.; Blasenbreu-Vogt, S.; Sendelhofert, A.; Rössle, M.; Harrison, J.D.; Löhrs, U. Regeneration in chronic sialadenitis: An analysis of proliferation and apoptosis based on double immunohistochemical labelling. Virchows Arch. 2004, 444, 356–361. [Google Scholar] [CrossRef]
- Aure, M.H.; Konieczny, S.F.; Ovitt, C.E. Salivary gland homeostasis is maintained through acinar cell self-duplication. Dev. Cell 2015, 33, 231–237. [Google Scholar] [CrossRef]
- Boshell, J.L.; Pennington, C. Histological observations on the effects of isoproterenol on regenerating submandibular glands of the rat. Cell Tissue Res. 1980, 213, 411–416. [Google Scholar] [CrossRef]
- Maimets, M.; Bron, R.; de Haan, G.; van Os, R.; Coppes, R.P. Similar ex vivo expansion and post-irradiation regenerative potential of juvenile and aged salivary gland stem cells. Radiother. Oncol. 2015, 116, 443–448. [Google Scholar] [CrossRef]
- Feng, J.; van der Zwaag, M.; Stokman, M.A.; van Os, R.; Coppes, R.P. Isolation and characterization of human salivary gland cells for stem cell transplantation to reduce radiation-induced hyposalivation. Radiother. Oncol. 2009, 92, 466–471. [Google Scholar] [CrossRef]
- Patel, V.N.; Lombaert, I.M.A.; Cowherd, S.N.; Shworak, N.W.; Xu, Y.; Liu, J.; Hoffman, M.P. Hs3st3-modified heparan sulfate controls KIT+ progenitor expansion by regulating 3-O-sulfotransferases. Dev. Cell 2014, 29, 662–673. [Google Scholar] [CrossRef] [PubMed]
- Banh, A.; Xiao, N.; Cao, H.; Chen, C.-H.; Kuo, P.; Krakow, T.; Bavan, B.; Khong, B.; Yao, M.; Ha, C.; et al. A novel aldehyde dehydrogenase-3 activator leads to adult salivary stem cell enrichment in vivo. Clin. Cancer Res. 2011, 17, 7265–7272. [Google Scholar] [CrossRef] [PubMed]
- Nakao, K.; Morita, R.; Saji, Y.; Ishida, K.; Tomita, Y.; Ogawa, M.; Saitoh, M.; Tomooka, Y.; Tsuji, T. The development of a bioengineered organ germ method. Nat. Methods 2007, 4, 227–230. [Google Scholar] [CrossRef] [PubMed]
- Ogawa, M.; Oshima, M.; Imamura, A.; Sekine, Y.; Ishida, K.; Yamashita, K.; Nakajima, K.; Hirayama, M.; Tachikawa, T.; Tsuji, T. Functional salivary gland regeneration by transplantation of a bioengineered organ germ. Nat. Commun. 2013, 4, 2498. [Google Scholar] [CrossRef]
- Hirayama, M.; Oshima, M.; Tsuji, T. Development and prospects of organ replacement regenerative therapy. Cornea 2013, 32 (Suppl. S1), S13–S21. [Google Scholar] [CrossRef]
| Drug | Formulation | Dose | Side Effect |
|---|---|---|---|
| Amphotericin B | Infusion 50 mg | 100–200 mg/6 h | Renal, cardiovascular, spinal, and neurological |
| Nystatin | Suspension 60 mL | 4–6 mL/6 h | Well tolerated |
| Ointment 30 g | 2–4 times/day | ||
| Tablets | 2 every 8 h | Uncommon nausea, vomiting, and gastrointestinal effects | |
| Clotrimazole | Gel 1% | 3 times/day | Occasionally skin irritation and burning sensation. |
| Tablets 10 mg | 5 times/day | ||
| Miconazole | Gel | 100 mg/6 h | Uncommon burning, irritation, nausea, and diarrhea. |
| Ketoconazole | Gel 2% | 3 times/day | Nausea, vomiting |
| Tablets | 200 mg, 1–2/day | abdominal pain. | |
| Suspension 30 mL | |||
| Fluconazole | Tablets | 50–100 mg/day | Nausea, vomiting, diarrhea, and abdominal pain. |
| Suspension | 10 mg/mL | ||
| Itraconazole | Capsule | 100–200 mg/day | Nausea, vomiting, diarrhea, and abdominal pain. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ok, S.-M.; Ho, D.; Lynd, T.; Ahn, Y.-W.; Ju, H.-M.; Jeong, S.-H.; Cheon, K. Candida Infection Associated with Salivary Gland—A Narrative Review. J. Clin. Med. 2021, 10, 97. https://doi.org/10.3390/jcm10010097
Ok S-M, Ho D, Lynd T, Ahn Y-W, Ju H-M, Jeong S-H, Cheon K. Candida Infection Associated with Salivary Gland—A Narrative Review. Journal of Clinical Medicine. 2021; 10(1):97. https://doi.org/10.3390/jcm10010097
Chicago/Turabian StyleOk, Soo-Min, Donald Ho, Tyler Lynd, Yong-Woo Ahn, Hye-Min Ju, Sung-Hee Jeong, and Kyounga Cheon. 2021. "Candida Infection Associated with Salivary Gland—A Narrative Review" Journal of Clinical Medicine 10, no. 1: 97. https://doi.org/10.3390/jcm10010097
APA StyleOk, S.-M., Ho, D., Lynd, T., Ahn, Y.-W., Ju, H.-M., Jeong, S.-H., & Cheon, K. (2021). Candida Infection Associated with Salivary Gland—A Narrative Review. Journal of Clinical Medicine, 10(1), 97. https://doi.org/10.3390/jcm10010097





