The Influence of Talc Addition on the Performance of Polypropylene Membranes Formed by TIPS Method
Abstract
1. Introduction
- high liquid entry pressure (LEP), which is the minimum transmembrane hydrostatic pressure that is applied on the membrane before a liquid solution penetrates into the pores;
- good thermal stability—up to the boiling temperature of water;
- high chemical resistance to separated solutions;
- high permeability;
- low thermal conductivity;
- narrow pore size distribution.
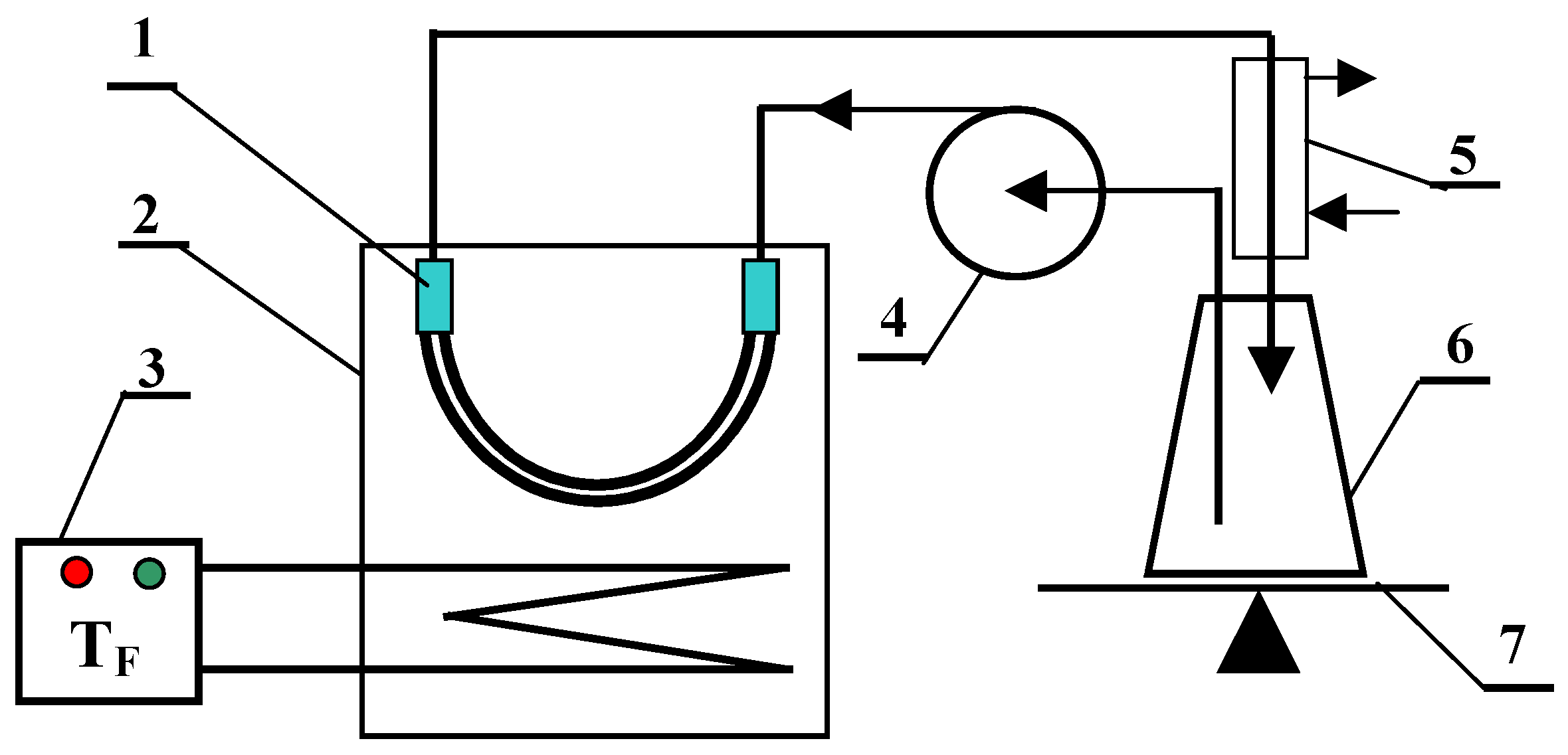
2. Materials and Methods
3. Results and Discussion
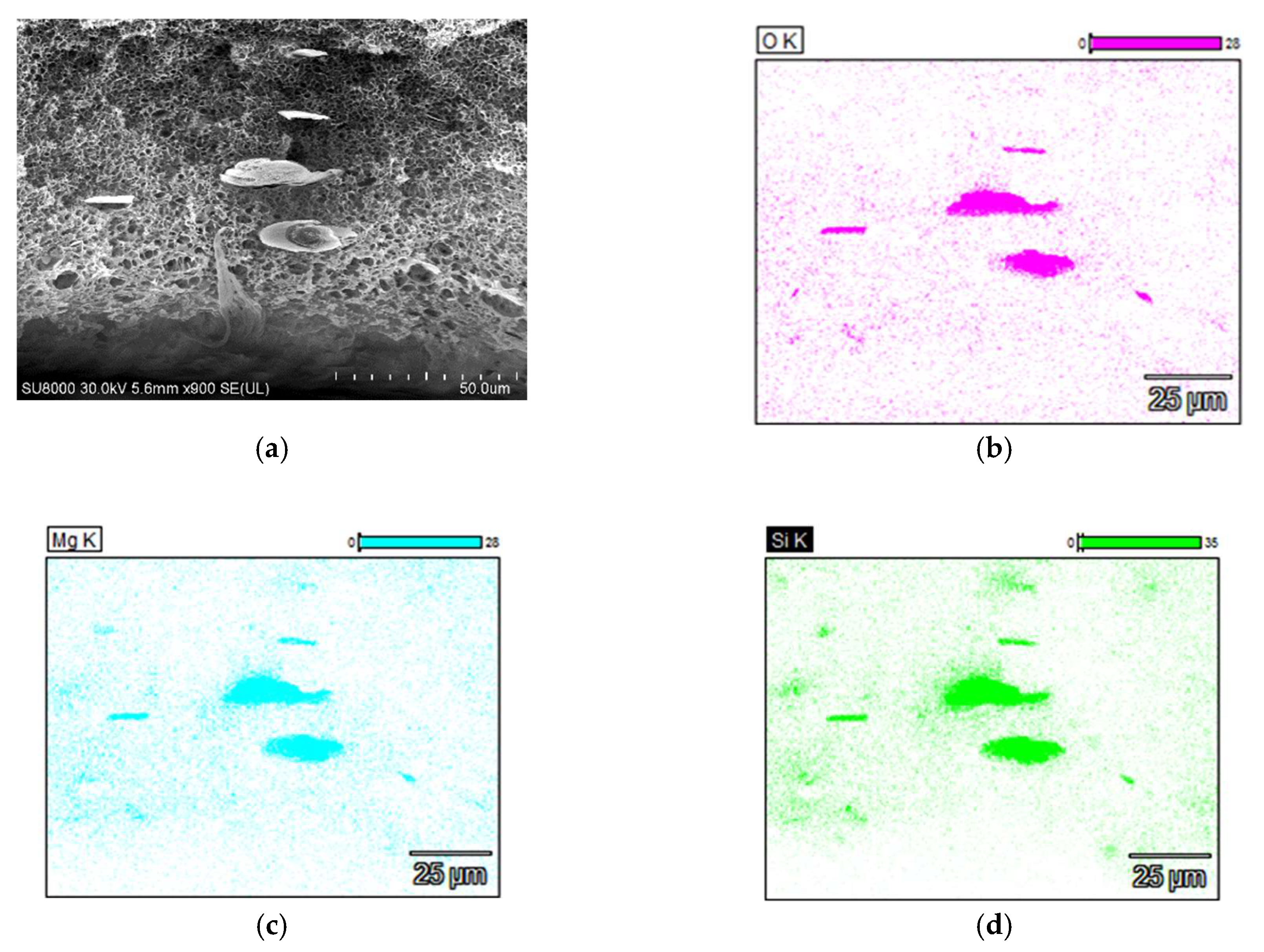
3.1. SEM Examinations of Membranes
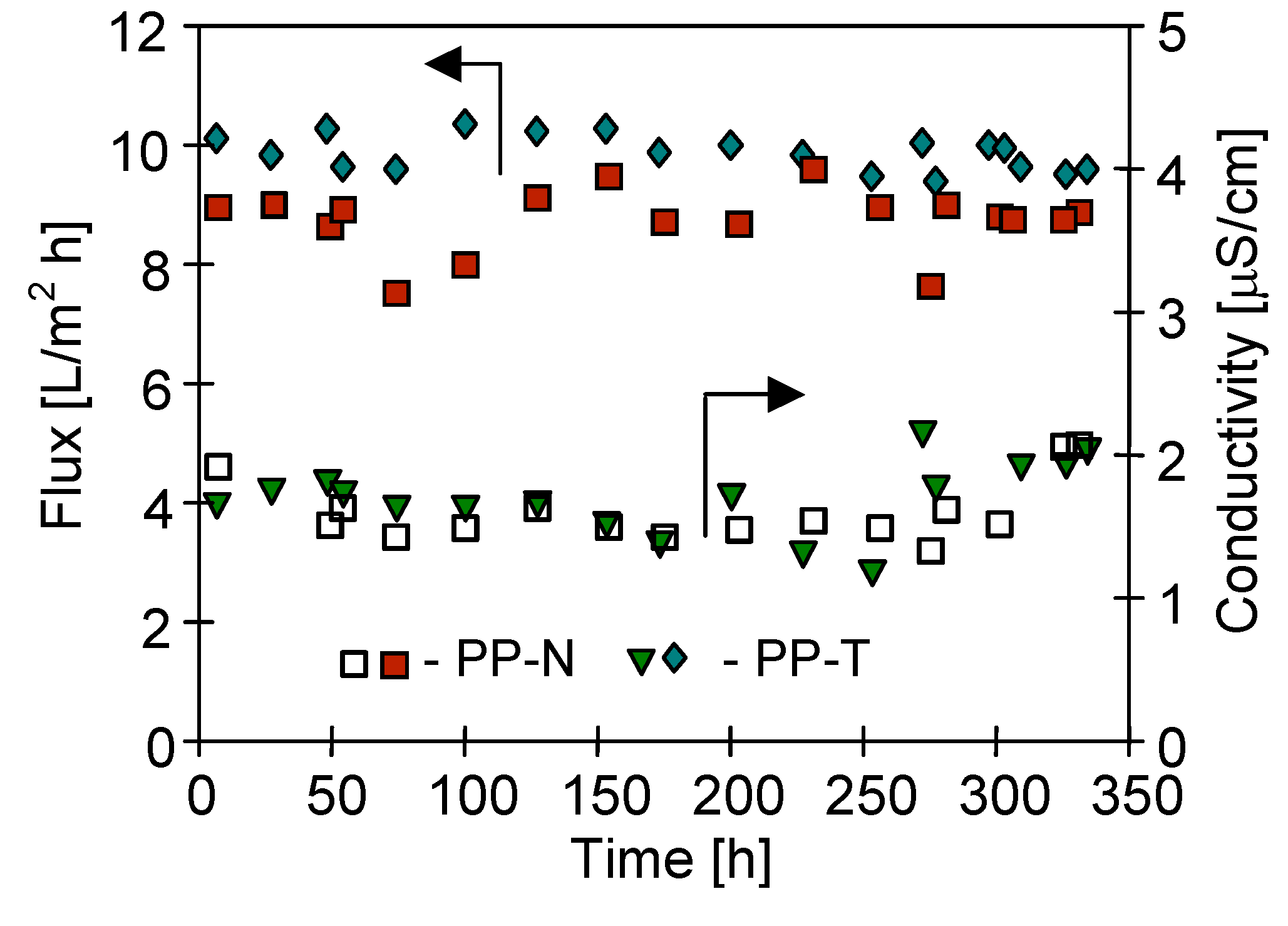
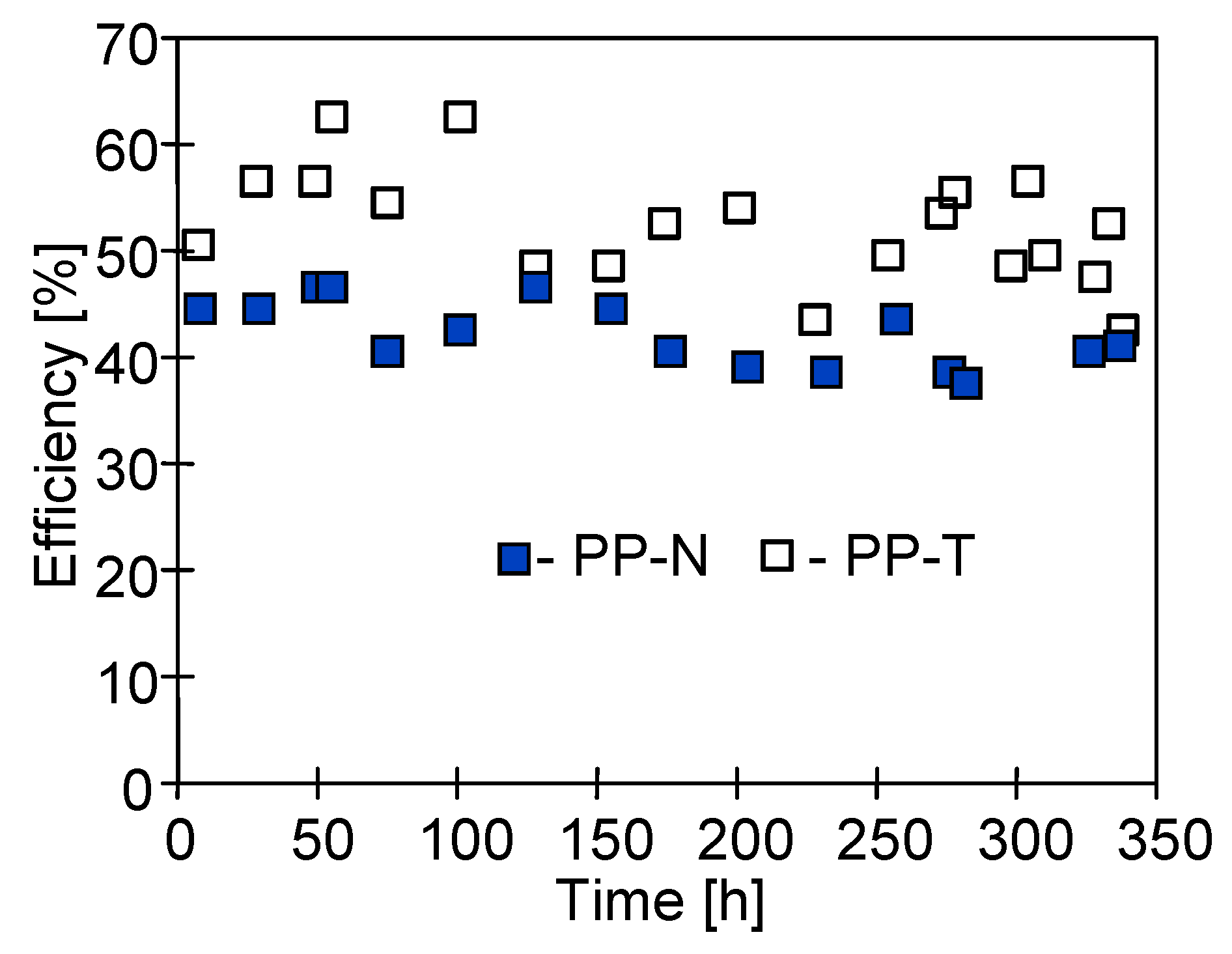
3.2. Long-Term MD Studies
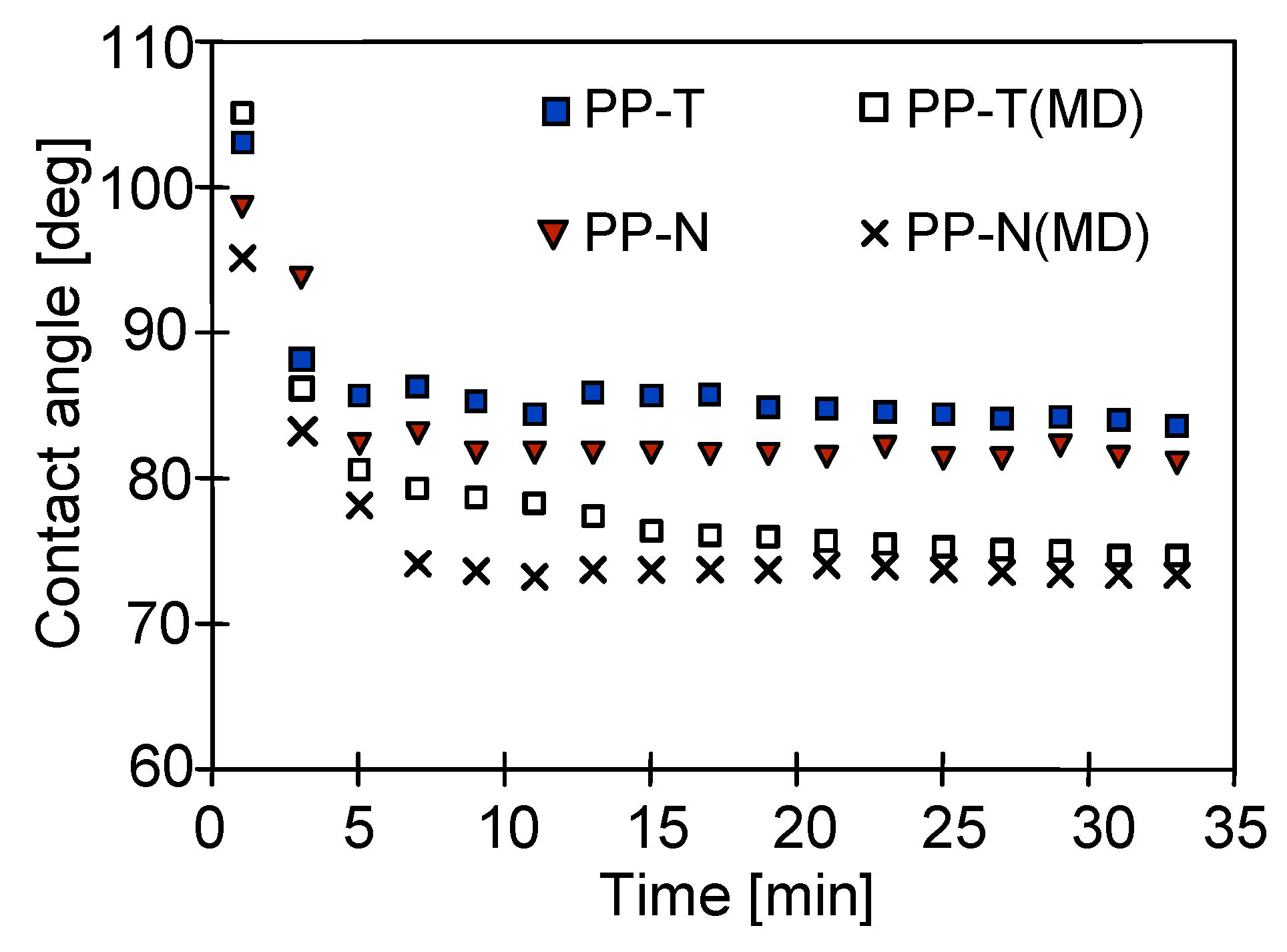
3.3. Membrane Performance
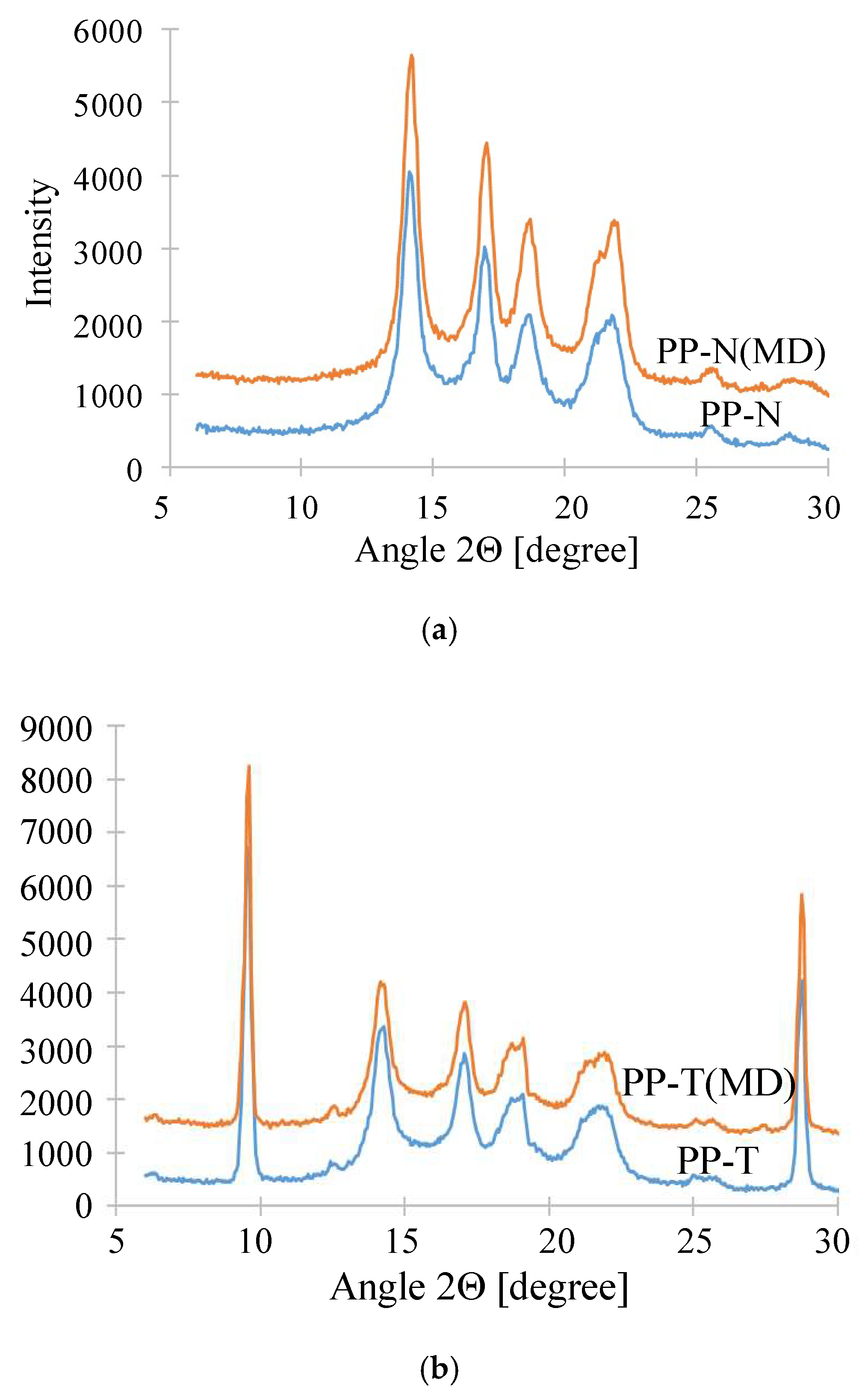
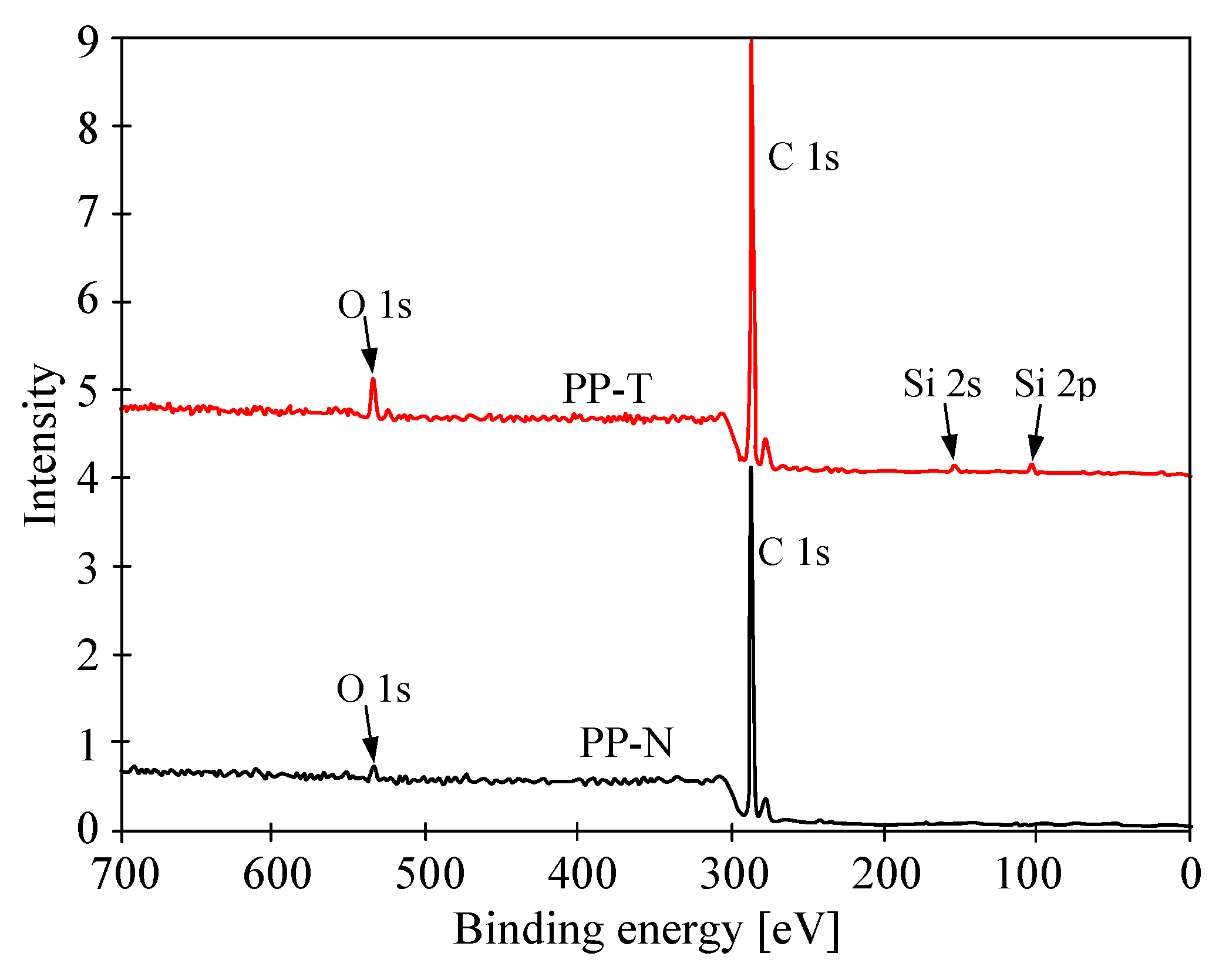
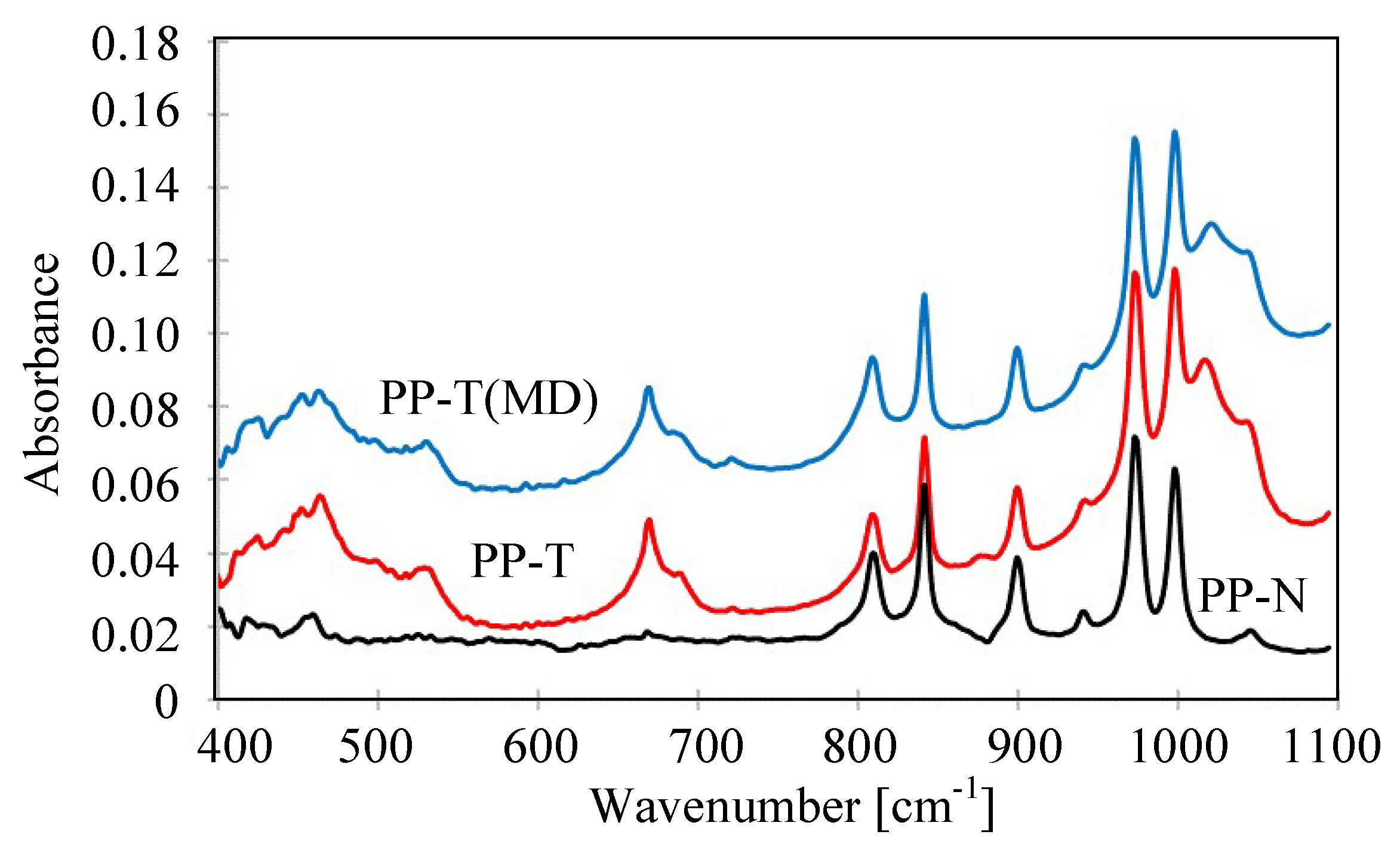
3.4. Stability of Membrane Matrix
4. Conclusions
Funding
Conflicts of Interest
References
- Bodell, B.R. Silicone Rubber Vapor Diffusion in Saline Water Desalination. U.S. Patent 285032, 3 June 1963. [Google Scholar]
- Lizeth, D.; Mendez, M.; Castel, C.; Lemaitre, C.; Favre, E. Membrane distillation (MD) processes for water desalination applications. Can dense selfstanding membranes compete with microporous hydrophobic materials? Chem. Eng. Sci. 2018, 188, 84–96. [Google Scholar]
- Winter, D.; Koschikowski, J.; Wieghaus, M. Desalination using membrane distillation: Experimental studies on full scale spiral wound modules. J. Membr. Sci. 2011, 375, 104–112. [Google Scholar] [CrossRef]
- Guillén-Burrieza, E.; Blanco, J.; Zaragoza, G.; Alarcón, D.-C.; Palenzuela, P.; Ibarra, M.; Gernjak, W. Experimental analysis of an air gap membrane distillation solar desalination pilot system. J. Membr. Sci. 2011, 379, 386–396. [Google Scholar] [CrossRef]
- Duong, H.C.; Chivas, A.R.; Nelemans, B.; Duke, M.; Gray, S.; Cath, T.Y.; Nghiem, L.D. Treatment of RO brine from CSG produced water by spiral-wound air gap membrane distillation—A pilot study. Desalination 2015, 366, 121–129. [Google Scholar] [CrossRef]
- Schwantes, R.; Cipollina, A.; Gross, F.; Koschikowski, J.; Pfeifle, D.; Rolletschek, M.; Subiela, V. Membrane distillation: Solar and waste heat driven demonstration plants for desalination. Desalination 2013, 323, 93–106. [Google Scholar] [CrossRef]
- Qtaishat, M.R.; Banat, F. Desalination by solar powered membrane distillation systems. Desalination 2013, 308, 186–197. [Google Scholar] [CrossRef]
- Eykens, L.; De Sitter, K.; Dotremont, C.; Pinoy, L.; Van der Bruggen, B. Membrane synthesis for membrane distillation: A review. Sep. Purif. Technol. 2017, 182, 36–51. [Google Scholar] [CrossRef]
- Rezaeia, M.; Warsinger, D.M.; Lienhard V, J.H.; Samhaber, W.M. Wetting prevention in membrane distillation through superhydrophobicity and recharging an air layer on the membrane surface. J. Membr. Sci. 2017, 530, 42–52. [Google Scholar] [CrossRef]
- Gryta, M. The application of polypropylene membranes for production of fresh water from brines by membrane distillation. Chem. Pap. 2017, 71, 775–784. [Google Scholar] [CrossRef]
- Adnan, S.; Hoang, M.; Wang, H.T.; Xie, Z.L. Commercial PTFE membranes for membrane distillation application: Effect of microstructure and support material. Desalination 2012, 284, 297–308. [Google Scholar] [CrossRef]
- Eykens, L.; De Sitter, K.; Dotremont, C.; Pinoy, L.; Van der Bruggen, B. Characterization and performance evaluation of commercially available hydrophobic membranes for direct contact membrane distillation. Desalination 2016, 392, 63–73. [Google Scholar] [CrossRef]
- Wang, P.; Chung, T.S. Recent advances in membrane distillation processes: Membrane development, configuration design and application exploring. J. Membr. Sci. 2015, 474, 39–56. [Google Scholar] [CrossRef]
- González, D.; Amigo, J.; Suárez, F. Membrane distillation: Perspectives for sustainable and improved desalination. Renew. Sustain. Energy Rev. 2017, 80, 238–259. [Google Scholar] [CrossRef]
- Khalifa, A.; Ahmad, H.; Antar, M.; Laoui, T.; Khayet, M. Experimental and theoretical investigations on water desalination using direct contact membrane distillation. Desalination 2017, 404, 22–34. [Google Scholar] [CrossRef]
- Rezaei, M.; Warsinger, D.M.; Lienhard V, J.H.; Duke, M.C.; Matsuura, T.; Samhaber, W.M. Wetting phenomena in membrane distillation: Mechanisms, reversal, and prevention. Water Res. 2018, 139, 329–352. [Google Scholar] [CrossRef]
- Chamani, H.; Matsuura, T.; Rana, D.; Lan, C.Q. Modeling of pore wetting in vacuum membrane distillation. J. Membr. Sci. 2019, 572, 332–342. [Google Scholar] [CrossRef]
- Thomas, N.; Mavukkandy, M.O.; Loutatidou, S.; Arafat, H.A. Membrane distillation research & implementation: Lessons from the past five decades. Sep. Purif. Technol. 2017, 189, 108–127. [Google Scholar]
- Lee, E.-J.; An, A.K.; He, T.; Woo, Y.C.; Shon, H.K. Electrospun nanofiber membranes incorporating fluorosilane-coated TiO2 nanocomposite for direct contact membrane distillation. J. Membr. Sci. 2016, 520, 145–154. [Google Scholar] [CrossRef]
- Ali, A.; Tsai, J.-H.; Tung, K.-L.; Drioli, E.; Macedonio, F. Designing and optimization of continuous direct contact membrane distillation process. Desalination 2018, 426, 97–107. [Google Scholar] [CrossRef]
- Eykens, L.; Hitsov, I.; DeSitter, K.; Dotremont, C.; Pinoy, L.; Nopens, I.; Van der Bruggen, B. Influence of membrane thickness and process conditions on direct contact membrane distillation at different salinities. J. Membr. Sci. 2016, 498, 353–364. [Google Scholar] [CrossRef]
- Guillen-Burrieza, E.; Ruiz-Aguirre, A.; Zaragoza, G.; Arafat, H.A. Membrane fouling and cleaning in long term plant-scale membrane distillation operations. J. Membr. Sci. 2014, 68, 360–372. [Google Scholar] [CrossRef]
- Himma, N.F.; Anisah, S.; Prasetya, N.; Wenten, I.G. Advances in preparation, modification, and application of polypropylene membrane. Polym. Eng. 2016, 36, 329–362. [Google Scholar] [CrossRef]
- Eykens, L.; De Sitter, K.; Dotremont, C.; Pinoy, L.; Van der Bruggen, B. Coating techniques for membrane distillation: An experimental assessment. Sep. Purif. Technol. 2018, 193, 38–48. [Google Scholar] [CrossRef]
- Cui, Z.; Zhang, Y.; Li, X.; Wang, X.; Drioli, E.; Wang, Z.; Zhao, S. Optimization of novel composite membranes for water and mineral recovery by vacuum membrane distillation. Desalination 2018, 440, 39–47. [Google Scholar] [CrossRef]
- Ali, A.; Criscuoli, A.; Macedonio, F.; Drioli, E. A comparative analysis of flat sheet and capillary membranes for membrane distillation applications. Desalination 2019, 456, 1–12. [Google Scholar] [CrossRef]
- Silva, T.L.S.; Morales-Torres, S.; Figueiredo, J.L.; Silva, A.M.T. Multi-walled carbon nanotube/PVDF blended membranes with sponge- and finger-like pores for direct contact membrane distillation. Desalination 2015, 357, 233–245. [Google Scholar] [CrossRef]
- Fahmey, M.S.; El-Aassar, A.-H.M.; Abo-Elfadel, M.M.; Orabi, A.S.; Das, R. Comparative performance evaluations of nanomaterials mixed polysulfone: A scale-up approach through vacuum enhanced direct contact membrane distillation for water desalination. Desalination 2019, 451, 111–116. [Google Scholar] [CrossRef]
- Baghbanzadeh, M.; Rana, D.; Lan, C.Q.; Matsuura, T. Effects of hydrophilic silica nanoparticles and backing material in improving the structure and performance of VMD PVDF membranes. Sep. Purif. Technol. 2016, 157, 60–71. [Google Scholar] [CrossRef]
- Makhlouf, A.; Satha, H.; Frihi, D.; Gherib, S.; Seguela, R. Optimization of the crystallinity of polypropylene/submicronic-talc composites: The role of filler ratio and cooling rate. eXPRESS Polym. Lett. 2016, 10, 237–247. [Google Scholar] [CrossRef]
- Efome, J.E.; Baghbanzadeh, M.; Rana, D.; Matsuura, T.; Lan, C.Q. Effects of superhydrophobic SiO2 nanoparticles on the performance of PVDF flat sheet membranes for vacuum membrane distillation. Desalination 2015, 373, 47–57. [Google Scholar] [CrossRef]
- Santoro, S.; Vidorreta, I.; Coelhoso, I.; Lima, J.C.; Desiderio, G.; Lombardo, G.; Drioli, E.; Mallada, R.; Crespo, J.; Criscuoli, A.; et al. Experimental Evaluation of the Thermal Polarization in Direct Contact Membrane Distillation Using Electrospun Nanofiber Membranes Doped with Molecular Probes. Molecules 2019, 24, 638. [Google Scholar] [CrossRef]
- Kujawa, J.; Al-Gharabli, S.; Kujawski, W.; Knozowska, K. Molecular Grafting of Fluorinated and Nonfluorinated Alkylsiloxanes on Various Ceramic Membrane Surfaces for the Removal of Volatile Organic Compounds Applying Vacuum Membrane Distillation. ACS Appl. Mater. Interfaces 2017, 9, 6571–6590. [Google Scholar] [CrossRef]
- Hubadillah, S.K.; Tai, Z.S.; Othman, M.H.D.; Harun, Z.; Jamalludin, M.R.; Rahmana, M.A.; Jaafar, J.; Ismail, A.F. Hydrophobic ceramic membrane for membrane distillation: A mini review on preparation, characterization, and applications. Sep. Purif. Technol. 2019, 217, 71–84. [Google Scholar] [CrossRef]
- Ashoor, B.B.; Mansour, S.; Giwa, A.; Dufour, V.; Hasan, S.W. Principles and applications of direct contact membrane distillation (DCMD): A comprehensive review. Desalination 2016, 398, 222–246. [Google Scholar] [CrossRef]
- Gryta, M.; Grzechulska-Damszel, J.; Markowska, A.; Karakulski, K. The influence of polypropylene degradation on the membrane wettability during membrane distillation. J. Membr. Sci. 2009, 326, 493–502. [Google Scholar] [CrossRef]
- Wang, Y.-J.; Zhao, Z.-P.; Xi, Z.-Y.; Yan, S.-Y. Microporous polypropylene membrane prepared via TIPS using environment-friendly binary diluents and its VMD performance. J. Membr. Sci. 2018, 548, 332–344. [Google Scholar] [CrossRef]
- Tang, N.; Han, H.; Yuan, L.; Zhang, L.; Wang, X.; Cheng, P. Preparation of a hydrophobically enhanced antifouling isotactic polypropylene/silicone dioxide flat-sheet membrane via thermally induced phase separation for vacuum membrane distillation. J. Appl. Polym. Sci. 2015, 132, 1–11. [Google Scholar] [CrossRef]
- Xu, K.; Cai, Y.; Hassankiadeh, N.T.; Cheng, Y.; Li, X.; Wang, X.; Wang, Z.; Drioli, E.; Cui, Z. ECTFE membrane fabrication via TIPS method using ATBC diluent for vacuum membrane distillation. Desalination 2019, 456, 13–22. [Google Scholar] [CrossRef]
- Zhang, X.; Zhang, D.; Liu, T. Influence of Nucleating Agent on Properties of Isotactic Polypropylene. Energy Procedia 2012, 17, 1829–1835. [Google Scholar] [CrossRef]
- Tang, N.; Jia, Q.; Zhang, H.; Li, J.; Cao, S. Preparation and morphological characterization of narrow pore size distributed polypropylene hydrophobic membranes for vacuum membrane distillation via thermally induced phase separation. Desalination 2010, 256, 27–36. [Google Scholar] [CrossRef]
- Lin, Y.K.; Chen, G.; Yang, J.; Wang, X.L. Formation of isotactic polypropylene membranes with bicontinuous structure and good strength via thermally induced phase separation method. Desalination 2009, 236, 8–15. [Google Scholar] [CrossRef]
- Tang, N.; Feng, C.; Han, H.; Hua, X.; Zhang, L.; Xiang, J.; Cheng, P.; Du, W.; Wang, X. High permeation flux polypropylene/ethylene vinyl acetate co-blending membranes via thermally induced phase separation for vacuum membrane distillation desalination. Desalination 2016, 394, 44–55. [Google Scholar] [CrossRef]
- Zhao, J.; Shi, L.; Loh, C.H.; Wang, R. Preparation of PVDF/PTFE hollow fiber membranes for direct contact membrane distillation via thermally induced phase separation method. Desalination 2018, 430, 86–97. [Google Scholar] [CrossRef]
- Matsuyama, M.; Okafuji, H.; Maki, T.; Teramoto, M.; Kubota, N. Preparation of polyethylene hollow fiber membrane via thermally induced phase separation. J. Membr. Sci. 2003, 223, 119–126. [Google Scholar] [CrossRef]
- Li, M.; Qi, Y.; Zhao, Z.; Xiang, Z.; Liao, X.; Niu, Y.; Kong, M. Morphology evolution and crystalline structure of controlled-rheology polypropylene in micro-injection molding. Polym. Adv. Technol. 2016, 27, 494–503. [Google Scholar] [CrossRef]
- Ammar, O.; Bouaziz, Y.; Haddar, N.; Mnif, N. Talc as Reinforcing Filler in Polypropylene Compounds: Effect on Morphology and Mechanical Properties. Polym Sci. 2017, 3, 1–7. [Google Scholar]
- Alvarez, V.A.; Pérez, C.J. Effect of different inorganic filler over isothermal and non-isothermal crystallization of polypropylene homopolymer. J. Therm. Anal. Calorim. 2012, 107, 633–643. [Google Scholar] [CrossRef]
- Luo, B.; Zhang, J.; Wang, X.; Zhou, Y.; Wen, J. Effects of nucleating agents and extractants on the structure of polypropylene microporous membranes via thermally induced phase separation. Desalination 2006, 192, 142–150. [Google Scholar] [CrossRef]
- Gryta, M. Long-term performance of membrane distillation process. J. Membr. Sci. 2005, 265, 153–159. [Google Scholar] [CrossRef]
- Ursino, C.; Di Nicolò, E.; Gabriele, B.; Criscuoli, A.; Figoli, A. Development of a novel perfluoropolyether (PFPE) hydrophobic/hydrophilic coated membranes for water treatment. J. Membr. Sci. 2019, 581, 58–71. [Google Scholar] [CrossRef]
- Kalčíková, G.; Marolt, G.; Kokalj, A.J.; Gotvajn, A.Ž. The use of multiwell culture plates in the duckweed toxicity test—A case study on Zn nanoparticles. New Biotechnol. 2018, 47, 67–72. [Google Scholar] [CrossRef]
- Rekulapally, R.; Murthy Chavali, L.N.; Idris, M.M.; Singh, S. Toxicity of TiO2, SiO2, ZnO, CuO, Au and Ag engineered nanoparticles on hatching and early nauplii of Artemia sp. PeerJ 2019, 6, e6138. [Google Scholar] [CrossRef]
- Boffetta, P.; Mundt, K.A.; Thompson, W.J. The epidemiologic evidence for elongate mineral particle (EMP)-related human cancer risk. Toxicol. Appl. Pharmacol. 2018, 361, 100–106. [Google Scholar] [CrossRef]
- Drechsel, D.A.; Barlow, C.A.; Bare, J.L.; Jacobs, N.F.; Henshaw, J.L. Historical evolution of regulatory standards for occupational and consumer exposures to industrial talc. Regul. Toxicol. Pharmacol. 2018, 92, 251–267. [Google Scholar] [CrossRef]
- Chang, C.-J.; Yang, Y.-H.; Chen, P.-C.; Peng, H.-Y.; Lu, Y.-C.; Song, S.-R.; Yang, H.-Y. Stomach Cancer and Exposure to Talc Powder without Asbestos via Chinese Herbal Medicine: A Population-Based Cohort Study. Int. J. Environ. Res. Public Health 2019, 16, 717. [Google Scholar] [CrossRef] [PubMed]
- Moulik, S.; Kumar, F.D.; Archana, K.; Sridhar, S. Enrichment of hydrazine from aqueous solutions by vacuum membrane distillation through microporous polystyrene membranes of enhanced hydrophobicity. Sep. Purif. Technol. 2018, 203, 159–167. [Google Scholar] [CrossRef]
- Li, L.; Li, W.; Geng, L.; Chen, B.; Mi, H.; Hong, K.; Peng, X.; Kuang, T. Formation of stretched fibrils and nanohybrid shish-kebabs in isotactic polypropylene-based nanocomposites by application of a dynamic oscillatory shear. Chem. Eng. J. 2018, 348, 546–556. [Google Scholar] [CrossRef]
- Gryta, M. Influence of polypropylene membrane surface porosity on the performance of membrane distillation process. J. Membr. Sci. 2007, 287, 67–78. [Google Scholar] [CrossRef]
- Li, Z.; Rana, D.; Wang, Z.; Matsuura, T.; Lan, C.Q. Synergic effects of hydrophilic and hydrophobic nanoparticles on performance of nanocomposite distillation membranes: An experimental and numerical study. Sep. Purif. Technol. 2018, 202, 45–58. [Google Scholar] [CrossRef]
- Sadeghi, F.; Ajji, A.; Carreau, P.J. Analysis of microporous membranes obtained from polypropylene films by stretching. J. Membr. Sci. 2007, 292, 62–71. [Google Scholar] [CrossRef]
- Lv, Y.; Huang, Y.; Kong, M.; Li, G. Improved thermal oxidation stability of polypropylene films in the presence of β-nucleating agent. Polym. Test. 2013, 32, 179–186. [Google Scholar] [CrossRef]
- Fontanella, S.; Bonhomme, S.; Brusson, J.-M.; Pitteri, S.; Samuel, G.; Pichon, G.; Lacoste, J.; Fromageot, D.; Lemaire, J.; Delort, A.-M. Comparison of biodegradability of various polypropylene films containing pro-oxidant additives based on Mn, Mn/Fe or Co. Polym. Degrad. Stabil. 2013, 98, 875–884. [Google Scholar] [CrossRef]
- Echeverrigaray, S.G.; Cruz, R.C.D.; Oliveira, R.V.B. Reactive processing of a non-additivated isotactic polypropylene: Mechanical and morphological properties on molten and solid states. Polym. Bull. 2013, 70, 1237–1250. [Google Scholar] [CrossRef]
- He, P.; Xiao, Y.; Zhang, P.; Xing, C.; Zhu, N.; Zhu, X.; Yan, D. Thermal degradation of syndiotactic polypropylene and the influence of stereoregularity on the thermal degradation behaviour by in situ FTIR spectroscopy. Polym. Degrad. Stab. 2005, 88, 473–479. [Google Scholar] [CrossRef]
- Qian, S.; Igarashi, T.; Nitta, K. Thermal degradation behavior of polypropylene in the melt state: Molecular weight distribution changes and chain scission mechanism. Polym. Bull. 2011, 67, 1661–1670. [Google Scholar] [CrossRef]
- Zhang, H.; Li, B.; Sun, D.; Miao, X.; Gu, Y. SiO2-PDMS-PVDF hollow fiber membrane with high flux for vacuum membrane distillation. Desalination 2018, 429, 33–43. [Google Scholar] [CrossRef]





| Membrane | Internal Diameter [mm] | Wall [mm] | Porosity [%] | Pore Diameter [μm] |
|---|---|---|---|---|
| PP-N | 1.8 | 0.4 | 81 | 0.2 |
| PP-T | 1.8 | 0.4 | 84 | 0.2 |
| Membrane | Young’s Modulus [MPa] | Elongation at Break [%] | Tensile Strength [MPa] |
|---|---|---|---|
| PP-N | 106.1 +/− 18.3 | 154.5 +/− 26.8 | 1.98 +/− 0.13 |
| PP-T | 135.5 +/− 12.8 | 172.2 +/− 19.7 | 2.55 +/− 0.14 |
| Membrane | Tm [°C] | ΔHm [J/g] | TC [°C] | ΔHC [J/g] | X [%] |
|---|---|---|---|---|---|
| PP-N | 164.6 | 91.2 | 116.1 | 82.3 | 44 |
| PP-N (MD) | 157.2 | 84.9 | 117.1 | 68.4 | 41 |
| PP-T | 163.7 | 117.1 | 119.1 | 93.2 | 56 |
| PP-T (MD) | 162.7 | 109.8 | 114.6 | 91.7 | 53 |
| Membrane | Peak 14.1 [°] | Peak 16.9 [°] |
|---|---|---|
| PP-N | 0.89 +/− 0.01 | 1.0 +/− 0.02 |
| PP-N (MD) | 0.72 +/− 0.01 | 0.74 +/− 0.02 |
| PP-T | 0.67 +/− 0.01 | 0.71 +/− 0.02 |
| PP-T (MD) | 0.69 +/− 0.01 | 0.68 +/− 0.01 |
| Membrane | C [%] | O [%] | H [%] | O/C [%] |
|---|---|---|---|---|
| PP-N | 85.613 +/− 0.811 | 0.976 +/− 0.009 | 14.423 +/− 0.126 | 1.14 |
| PP-N (MD) | 82.668 +/− 0.116 | 0.804 +/− 0.116 | 13.961 +/− 0.116 | 0.97 |
| PP-T | 79.788 +/− 0.116 | 3.783 +/− 0.281 | 13.312 +/− 0.015 | 4.74 |
| PP-T (MD) | 76.333 +/− 0.116 | 3.224 +/− 0.116 | 12.856 +/− 0.116 | 4.22 |
| Membrane | C [%] | O [%] | Si [%] | Mg [%] |
|---|---|---|---|---|
| PP-N (EDS) | 98.3 | 0.9 | - | - |
| PP-N (XPS) | 98.1 | 1.9 | - | - |
| PP-T (EDS) | 93.1 | 2.7 | 2.2 | 1.8 |
| PP-T (XPS) | 91.8 | 4.2 | 2.5 | 1.4 |
© 2019 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gryta, M. The Influence of Talc Addition on the Performance of Polypropylene Membranes Formed by TIPS Method. Membranes 2019, 9, 63. https://doi.org/10.3390/membranes9050063
Gryta M. The Influence of Talc Addition on the Performance of Polypropylene Membranes Formed by TIPS Method. Membranes. 2019; 9(5):63. https://doi.org/10.3390/membranes9050063
Chicago/Turabian StyleGryta, Marek. 2019. "The Influence of Talc Addition on the Performance of Polypropylene Membranes Formed by TIPS Method" Membranes 9, no. 5: 63. https://doi.org/10.3390/membranes9050063
APA StyleGryta, M. (2019). The Influence of Talc Addition on the Performance of Polypropylene Membranes Formed by TIPS Method. Membranes, 9(5), 63. https://doi.org/10.3390/membranes9050063





