A Study of Ralex Membrane Morphology by SEM
Abstract
1. Introduction
2. Experimental
2.1. Membranes
2.2. Membrane Characterization
2.3. Scanning Electron Microscopy
2.4. Software
3. Results and Discussion
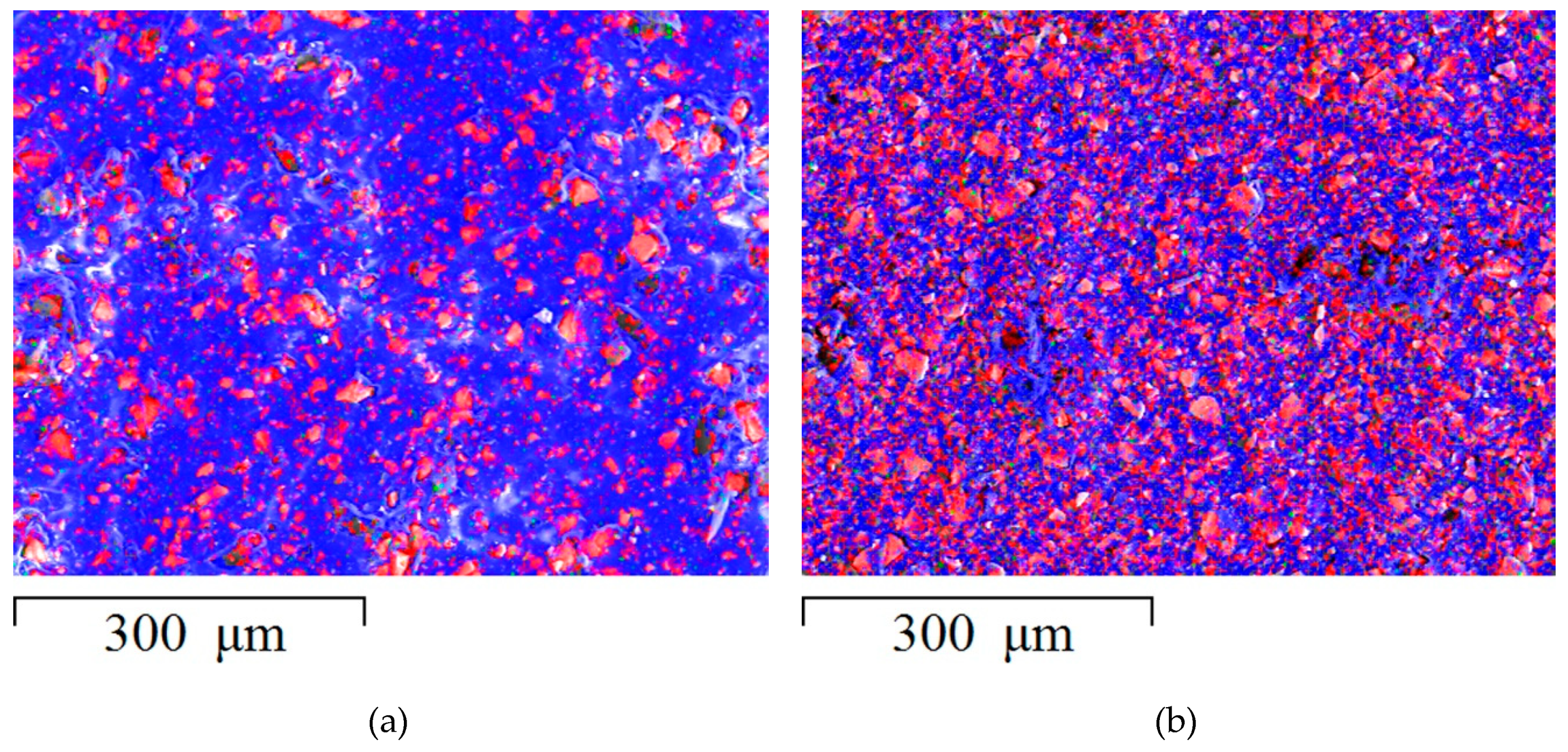
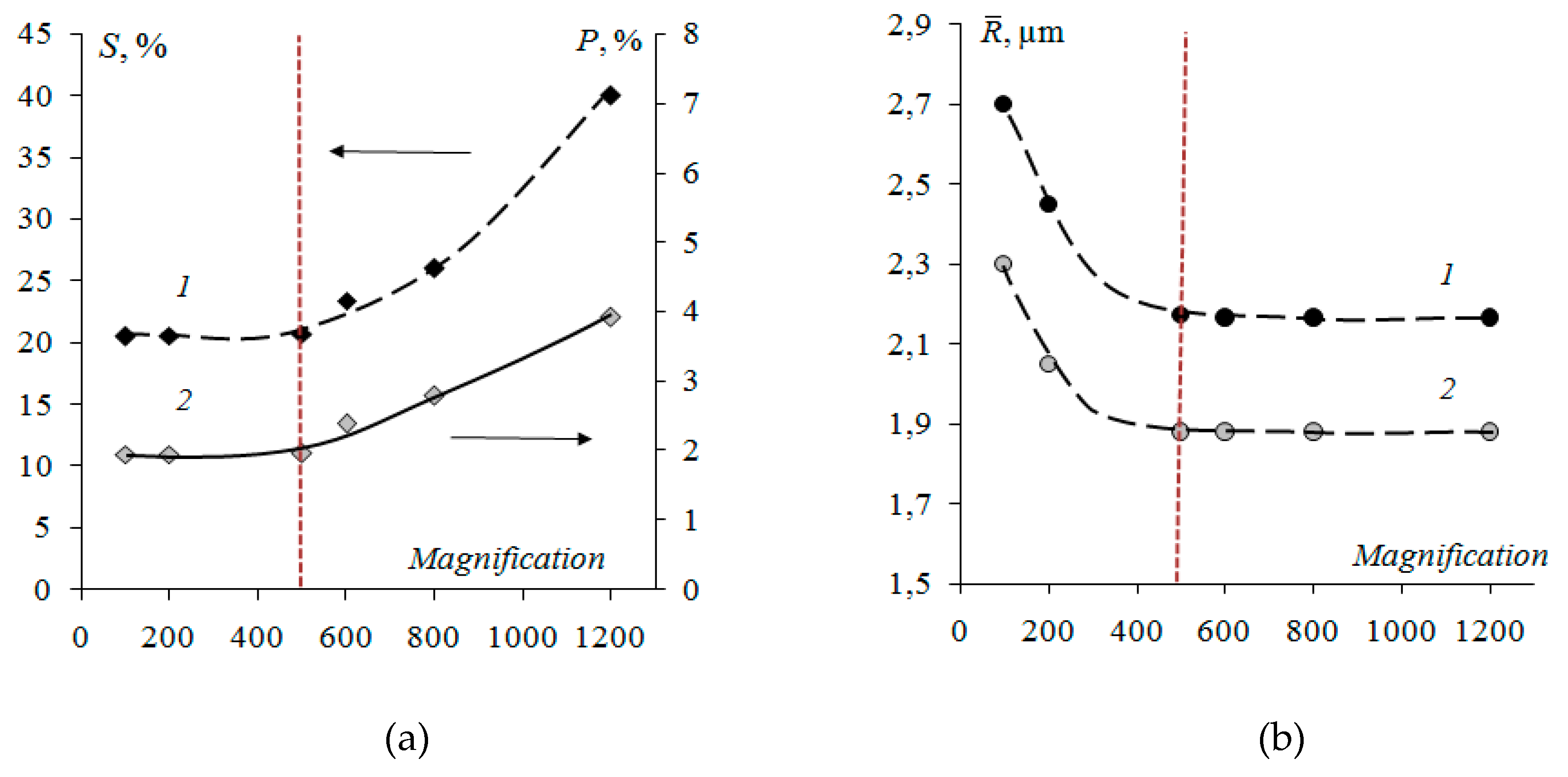
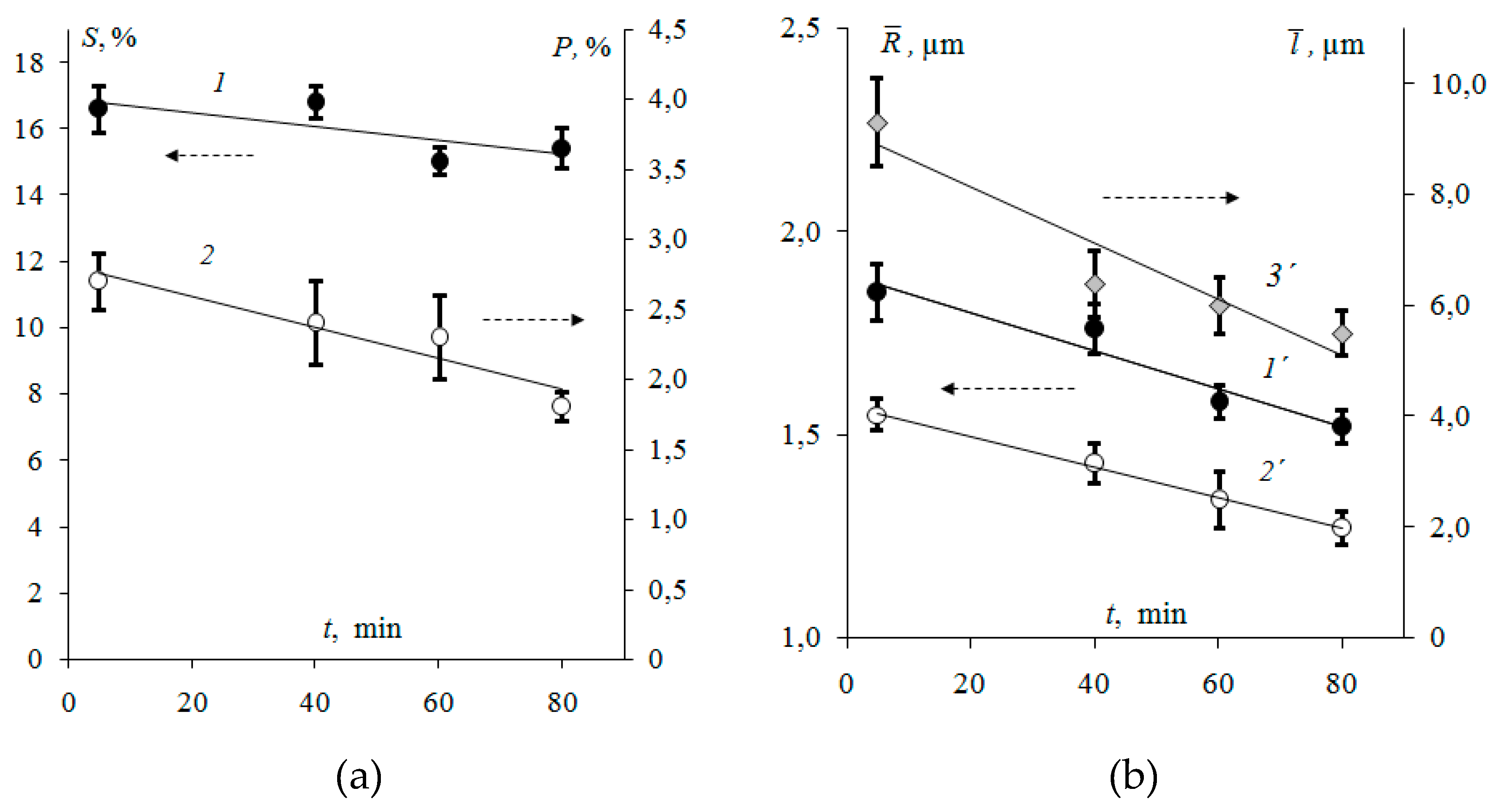
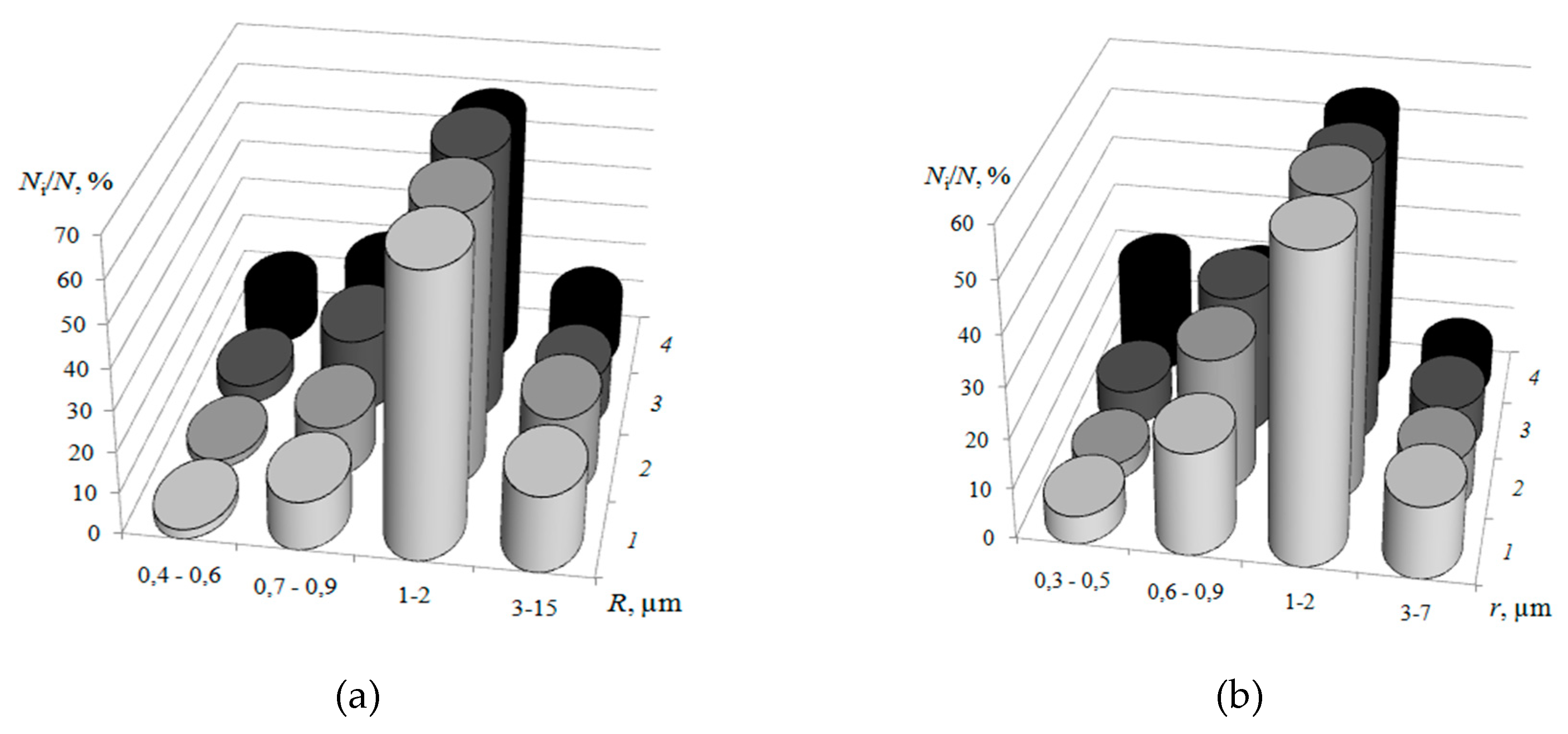
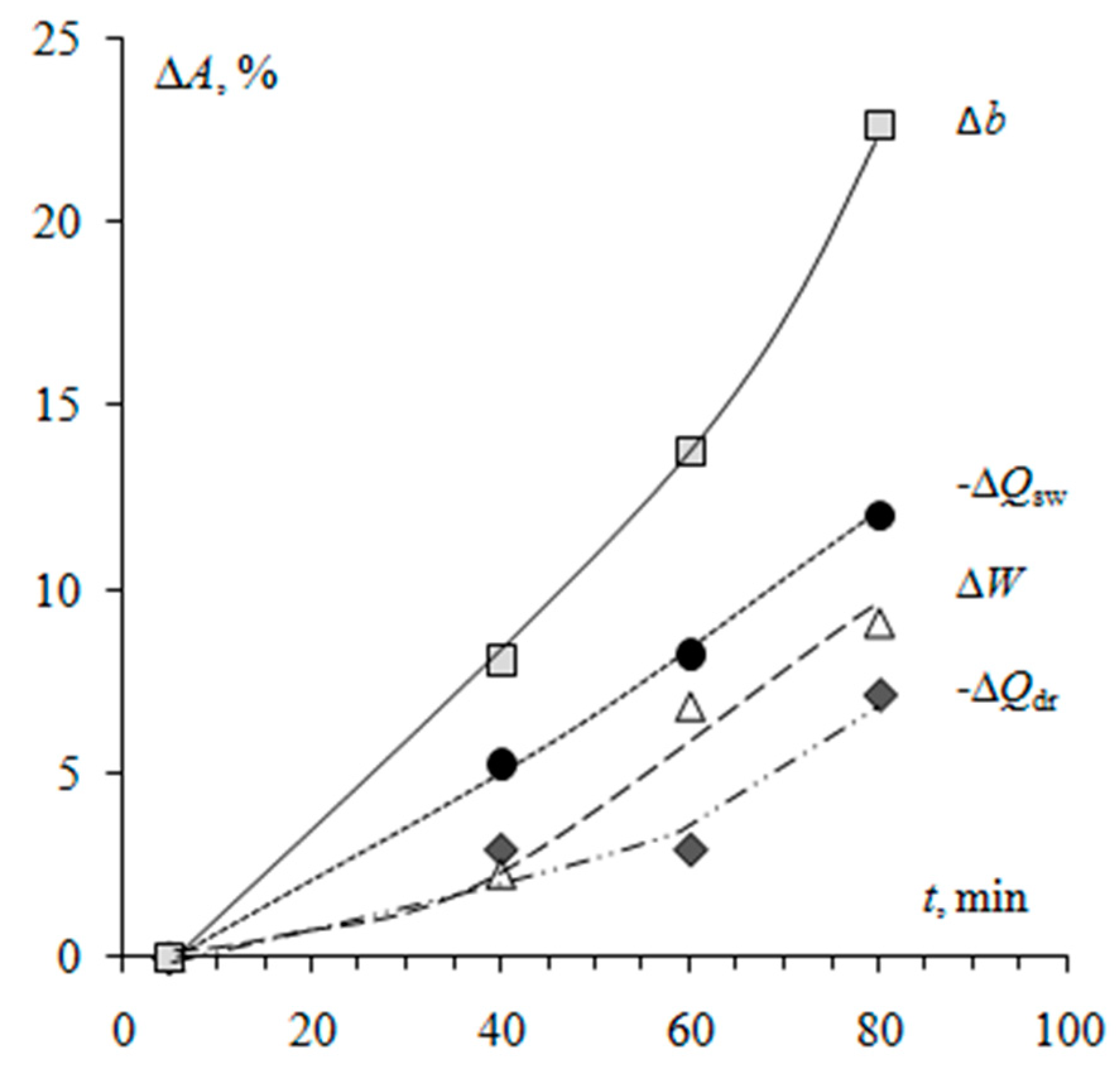
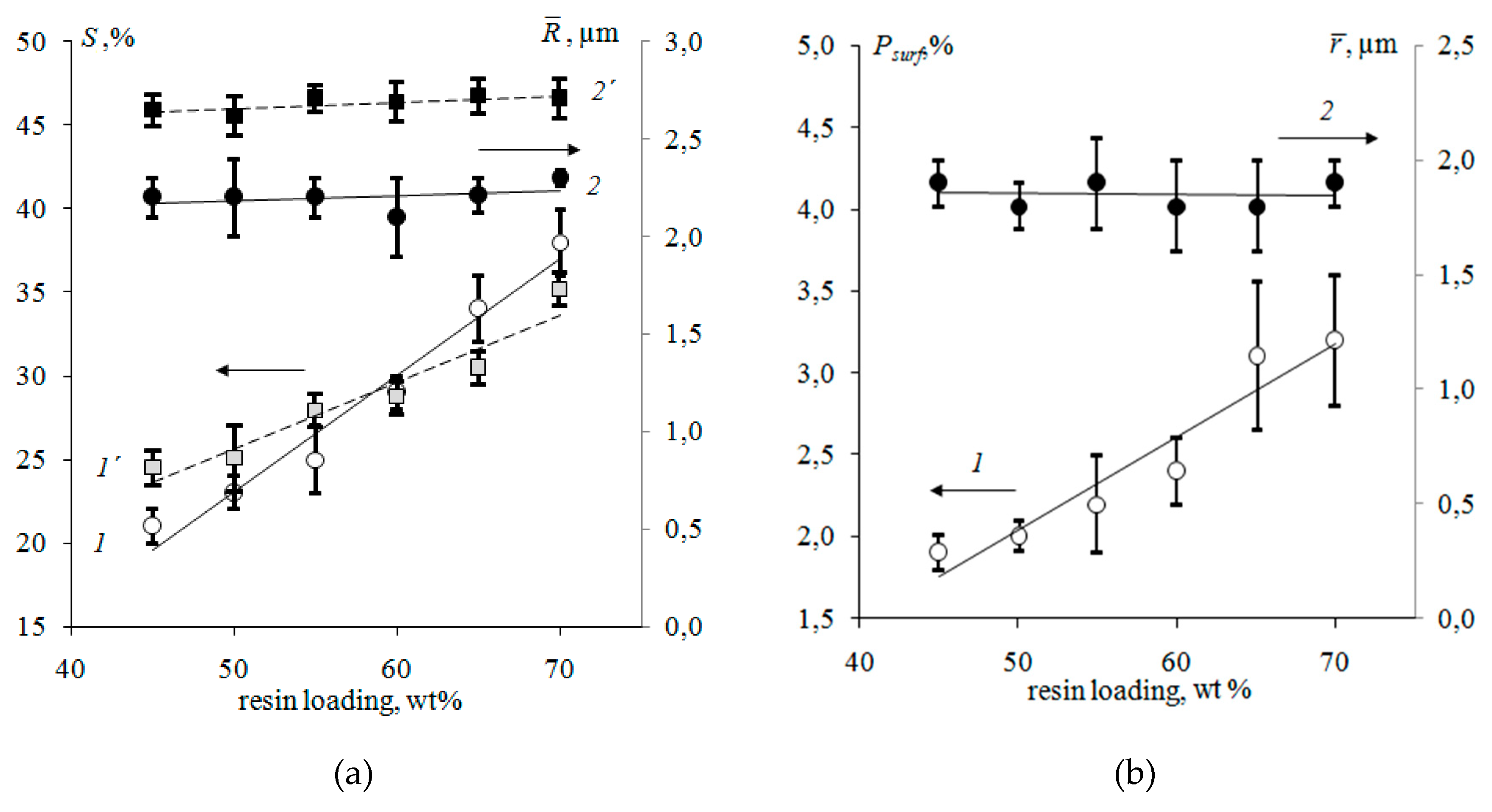
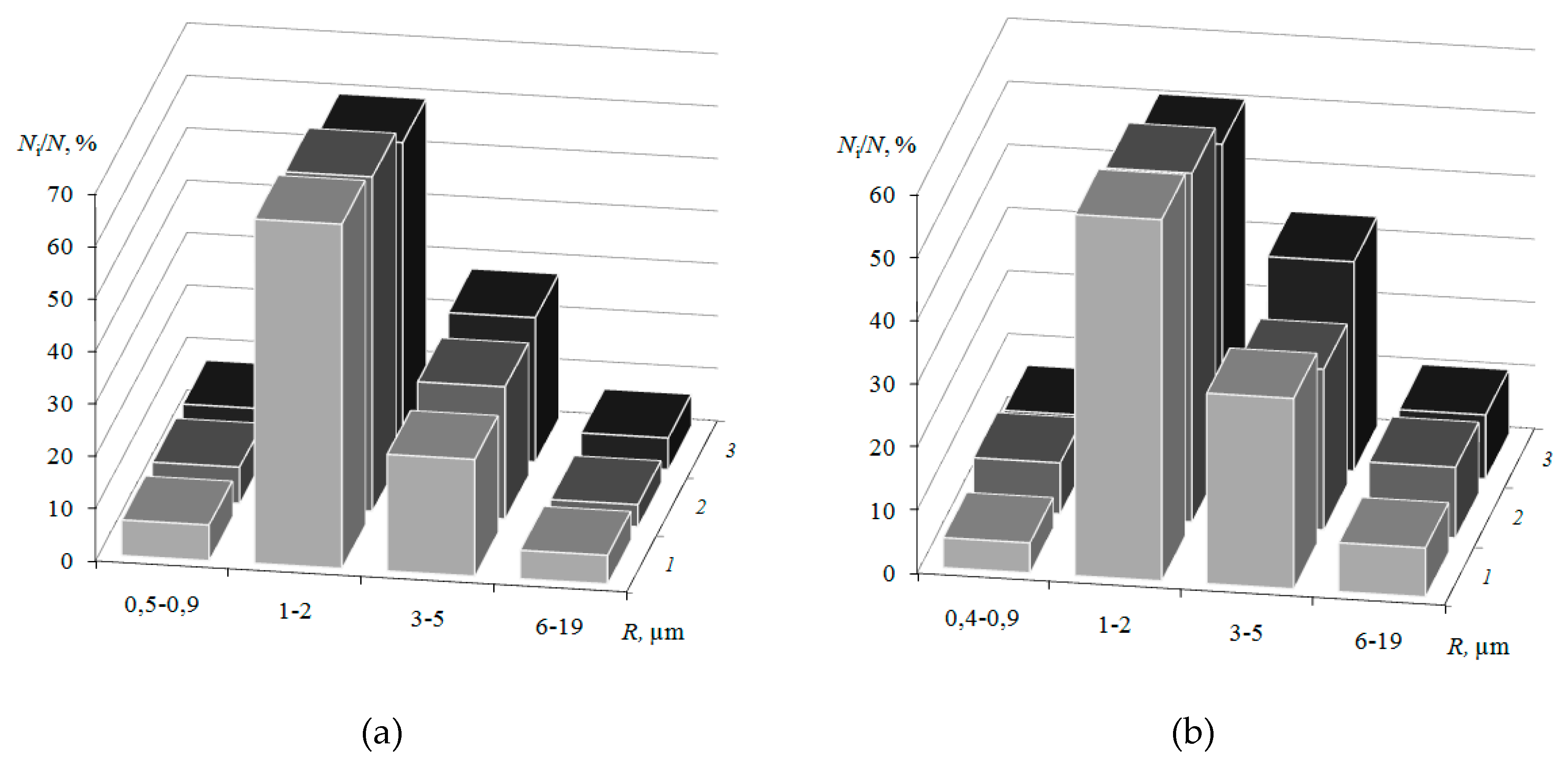
3.1. Surface Morphology of Heterogeneous Membranes with Different Resin Particle Sizes
3.2. Surface Morphology of Heterogeneous Membranes with Different Resin/Inert Binder Ratios
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Balster, J.; Yildirim, M.H.; Stamatialis, D.F.; Ibanez, R.; Lammertink, R.G.H.; Jordan, V.; Wessling, M. Morphology and microtopology of cation-exchange polymers and the origin of the overlimiting current. J. Phys. Chem. B 2007, 111, 2152–2165. [Google Scholar] [CrossRef]
- Nikonenko, V.V.; Pismenskaya, N.D.; Belova, E.I.; Sistat, P.; Huguet, P.; Pourcelly, G.; Larchet, C. Intensive current transfer in membrane systems: Modelling, mechanisms and application in electrodialysis. Adv. Colloid Interface Sci. 2010, 160, 101–123. [Google Scholar] [CrossRef]
- De Valenca, J.C.; Wagterveld, R.M.; Lammertink, R.G.H.; Tsai, P.A. Dynamics of microvortices induced by ion concentration polarization. Phys. Rev. E 2015, 92, 031003. [Google Scholar] [CrossRef]
- Wessling, M.; Morcillo, L.G.; Abdu, S. Nanometer-thick lateral polyelectrolyte micropatterns induce macrosopic electro-osmotic chaotic fluid instabilities. Sci. Rep. 2014, 4, 4294. [Google Scholar] [CrossRef]
- Tadimeti, J.G.D.; Kurian, V.; Chandra, A.; Chattopadhyay, S. Corrugated membrane surfaces for effective ion transport in electrodialysis. J. Membr. Sci. 2016, 499, 418–428. [Google Scholar] [CrossRef]
- Zabolotsky, V.I.; Nikonenko, V.V.; Urtenov, M.H.; Lebedev, K.A.; Bugakov, V.V. Electroconvection in systems with heterogeneous ion-exchange membranes. Russ. J. Electrochem. 2012, 48, 692–703. [Google Scholar] [CrossRef]
- Zabolotsky, V.I.; Novak, L.; Kovalenko, A.V.; Nikonenko, V.V.; Urtenov, M.H.; Lebedev, K.A.; But, A.Y. Electroconvection in systems with heterogeneous ion-exchange membranes. Pet. Chem. 2017, 57, 779–789. [Google Scholar] [CrossRef]
- Nikonenko, V.V.; Mareev, S.A.; Pis’menskaya, N.D.; Uzdenova, A.M.; Kovalenko, A.V.; Urtenov, M.K.; Pourcelly, G. Effect of electroconvection and its use in intensifying the mass transfer in electrodialysis (Review). Russ. J. Electrochem. 2017, 53, 1122–1144. [Google Scholar] [CrossRef]
- Vasil’eva, V.I.; Akberova, E.M.; Zabolotskii, V.I. Electroconvection in systems with heterogeneous ion-exchange membranes after thermal modification. Russ. J. Electrochem. 2017, 53, 398–410. [Google Scholar] [CrossRef]
- Akberova, E.M.; Vasil’eva, V.I.; Zabolotsky, V.I.; Novak, L. Effect of the sulfocation-exchanger dispersity on the surface morphology, microrelief of heterogeneous membranes and development of electroconvection in intense current modes. J. Membr. Sci. 2018, 566, 317–328. [Google Scholar] [CrossRef]
- Kamusewitz, H.; Schossig-Tiedemann, M.; Keller, M.; Paul, D. Characterization of polymeric membranes by means of scanning force microscopy (SFM) in comparison to results of scanning electron microscopy (SEM). Surf. Sci. 1997, 377–379, 1076–1081. [Google Scholar] [CrossRef]
- Sun, W.; Chen, T.; Chen, C.; Li, J. A study on membrane morphology by digital image processing. J. Membr. Sci. 2007, 305, 93–102. [Google Scholar] [CrossRef]
- Bouzek, K.; Moravcova, S.; Schauer, J.; Brozova, L.; Pientka, Z. Heterogeneous ion-selective membranes: The influence of the inert matrix polymer on the membrane properties. J. Appl. Electrochem. 2010, 40, 1005–1018. [Google Scholar] [CrossRef]
- Ghalloussi, R.; Garcia-Vasquez, W.; Chaabane, L.; Dammak, L.; Larchet, C.; Deabate, S.V.; Nevakshenova, E.; Nikonenko, V.; Grande, D. Ageing of ion-exchange membranes in electrodialysis: A structural and physicochemical investigation. J. Membr. Sci. 2013, 436, 68–78. [Google Scholar] [CrossRef]
- Vasil’eva, V.I.; Kranina, N.A.; Malykhin, M.D.; Akberova, E.M.; Zhiltsova, A.V. The surface inhomogeneity of ion-exchange membranes by SEM and AFM data. J. Surf. Investig. X-ray Synchrotron Neutron Tech. 2013, 7, 144–153. [Google Scholar]
- Vasil’eva, V.I.; Akberova, E.M.; Zhiltsova, A.V.; Chernykh, E.I.; Sirota, E.A.; Agapov, B.L. SEM diagnostics of the surface of MK-40 and MA-40 heterogeneous ion-exchange membranes in the swollen state after thermal treatment. J. Surf. Investig. X-ray Synchrotron Neutron Tech. 2013, 7, 833–840. [Google Scholar]
- Martí-Calatayud, M.C.; Buzzi, D.C.; García-Gabaldón, M.; Bernardes, A.M.; Tenório, J.A.S.; Pérez-Herranz, V. Ion transport through homogeneous and heterogeneous ion-exchange membranes in single salt and multicomponent electrolyte solutions. J. Membr. Sci. 2014, 466, 45–57. [Google Scholar] [CrossRef]
- Asraf-Snir, M.; Gilron, J.; Oren, Y. Gypsum scaling of anion exchange membranes in electrodialysis. J. Membr. Sci. 2016, 520, 176–186. [Google Scholar] [CrossRef]
- Lehmani, A.; Durand-Vidal, S.; Turq, P. Surface morphology of Nafion 117 membrane by tapping mode atomic force microscope. J. Appl. Polym. Sci. 1998, 68, 503–508. [Google Scholar] [CrossRef]
- Araya-Farias, M.; Bazinet, L. Effect of calcium and carbonate concentrations on anionic membrane fouling during electrodialysis. J. Colloid Interface Sci. 2006, 296, 242–247. [Google Scholar] [CrossRef]
- Pismenskaya, N.D.; Pokhidnia, E.V.; Pourcelly, G.; Nikonenko, V.V. Can the electrochemical performance of heterogeneous ion-exchange membranes be better than that of homogeneous membranes? J. Membr. Sci. 2018, 566, 54–68. [Google Scholar] [CrossRef]
- Casademont, C.; Pourcelly, G.; Bazinet, L. Effect of magnesium/calcium ratios in solutions treated by electrodialysis: Morphological characterization and identification of anion-exchange membrane fouling. J. Colloid Interface Sci. 2008, 322, 215–223. [Google Scholar] [CrossRef] [PubMed]
- Vasil’eva, V.I.; Akberova, E.M.; Goleva, E.A.; Yatsev, A.M.; Tzkhai, A.A. Changes in the microstructure and operational characteristics of the MK-40 sulfocation-exchange membrane during the electrodialysis of natural waters. J. Surf. Investig. X-ray Synchrotron Neutron Tech. 2017, 11, 429–436. [Google Scholar]
- Zhao, Z.; Shia, S.; Cao, H.; Shan, B.; Sheng, Y. Property characterization and mechanism analysis on organic fouling of structurally different anion exchange membranes in electrodialysis. Desalination 2018, 428, 199–206. [Google Scholar] [CrossRef]
- Belashova, E.; Mikhaylin, S.; Pismenskaya, N.; Nikonenko, V.; Bazinet, L. Impact of cation-exchange membrane scaling nature on the electrochemical characteristics of membrane system. Sep. Purif. Technol. 2017, 189, 441–448. [Google Scholar] [CrossRef]
- Butylskii, D.Y.; Mareev, S.A.; Nikonenko, V.V.; Pismenskaya, N.D.; Larchet, C.; Dammak, L.; Grande, D.; Apel, P.Y. In situ investigation of electrical inhomogeneity of ion exchange membrane surface using scanning electrochemical microscopy. Pet. Chem. 2016, 56, 1006–1013. [Google Scholar] [CrossRef]
- Volodina, E.; Pismenskaya, N.D.; Nikonenko, V.V.; Larchet, C.; Pourcelly, G. Ion transfer across ion-exchange membranes with homogeneous and heterogeneous surfaces. J. Colloid Interface Sci. 2005, 285, 247–258. [Google Scholar] [CrossRef]
- Vasil’eva, V.I.; Pismenskaya, N.D.; Akberova, E.M.; Nebavskaya, K.A. Effect of thermochemical treatment on the surface morphology and hydrophobicity of heterogeneous ion-exchange membranes. Russ. J. Phys. Chem. A 2014, 88, 1293–1299. [Google Scholar] [CrossRef]
- Vasil’eva, V.I.; Akberova, E.M.; Shaposhnik, V.A.; Malykhin, M.D. Electrochemical properties and structure of ion-exchange membranes upon thermochemical treatment. Russ. J. Electrochem. 2014, 50, 789–797. [Google Scholar] [CrossRef]
- Urtenov, M.K.; Uzdenova, A.M.; Kovalenko, A.V.; Vasil’eva, V.V.; Sistat, P.; Pourcelly, G. Basic mathematical model of overlimiting transfer enhanced by electroconvection in flow-through electrodialysis membrane cells. J. Membr. Sci. 2013, 447, 190–202. [Google Scholar] [CrossRef]
- Kovalenko, A.V.; Zabolotsky, V.I.; Nikonenko, V.V.; Urtenov, M.K. Mathematical modeling of the influence of surface morphology of heterogeneous ion-exchange membrane to the electroconvection. Sci. J. KubSAU 2014, 104, 630–643. [Google Scholar]
- Zabolotsky, V.I.; Lebedev, K.A.; Vasilenko, P.A.; Kuzyakina, M.V. Mathematical modeling of vortex structures during electroconvection in the channel of an electrodialyzer cell on model membranes with two conducting sections. Ecol. Bull. Res. Cent. Black Sea Econ. Coop. 2019, 1, 73–82. [Google Scholar] [CrossRef]
- Tanaka, Y. Ion Exchange Membrane: Fundamentals and Applications, 2nd ed.; Elsevier Science: Amsterdam, The Netherlands, 2015. [Google Scholar]
- MEGA, a.s. Products. Available online: https://www.mega.cz/membranes/ (accessed on 31 October 2019).
- Berezina, N.; Gnusin, N.; Dyomina, O.; Tymofeyev, S. Water electrotransport in membrane systems. Experiment and model description. J. Membr. Sci. 1994, 86, 207–229. [Google Scholar] [CrossRef]
- Reed, S.J.B. Electron Microprobe Analysis and Scanning Electron Microscopy in Geology; Cambridge University Press: Cambridge, UK, 2010. [Google Scholar]
- Uglov, V.V.; Koval’, N.N.; Kuleshov, A.K.; Ivanov, Y.F.; Teresov, A.D.; Soldatenko, E.A. Structure-phase transformation in surface layers of hard alloy as a result of action of high-current electron beams. J. Surf. Investig. X-ray Synchrotron Neutron Tech. 2011, 5, 350–357. [Google Scholar] [CrossRef]
- Sirota, E.A.; Kranina, N.A.; Vasil’eva, V.I.; Malykhin, M.D.; Selemenev, V.F. Development and experimental approbation of program complex for definition of a share ion-spending surfaces of heterogeneous membranes according to raster electronic microscopy. Proc. Voronezh State Uni. Series: Chem. Biol. Pharm. 2011, 2, 53–59. [Google Scholar]
- Taylor, J.R. An Introduction to Error Analysis: The Study of Uncertainties in Physical Measurements; University Science Books: Sausalito, CA, USA, 1982. [Google Scholar]
- Vasil’eva, V.I.; Zhil’tsova, A.V.; Akberova, E.M.; Fataeva, A.I. Influence of surface inhomogeneity on current-voltage characteristics of heterogeneous ion exchange membranes. Condens. Matter Interphases 2014, 16, 257–261. [Google Scholar]
- Melnikov, S.; Loza, S.; Sharafan, M.; Zabolotskiy, V. Electrodialysis treatment of secondary steam condensate obtained during production of ammonium nitrate. Technical and economic analysis. Sep. Purif. Technol. 2015, 157, 179–191. [Google Scholar] [CrossRef]
- Svoboda, M.; Slouka, Z.; Schrott, W.; Šnita, D. Cation exchange membrane integrated into a microfluidic device. Microelectron. Eng. 2009, 86, 1371–1374. [Google Scholar] [CrossRef]
- Zabolotskii, V.I.; Loza, S.A.; Sharafan, M.V. Physicochemical properties of profiled heterogeneous ion-exchange membranes. Russ. J. Electrochem. 2005, 41, 1053–1060. [Google Scholar] [CrossRef]
- Pismenskaya, N.; Melnik, N.; Nevakshenova, E.; Nebavskaya, K.; Nikonenko, V. Enhancing ion transfer in overlimiting electrodialysis of dilute solutions by modifying the surface of heterogeneous ion-exchange membranes. Int. J. Chem. Eng. 2012, 528290. [Google Scholar] [CrossRef]
- Berezina, N.P.; Kononenko, N.A.; Dyomina, O.A.; Gnusin, N.P. Characterization of ion-exchange membrane materials: Properties vs. structure. Adv. Colloid Interface Sci 2008, 139, 3–28. [Google Scholar] [CrossRef]
- Berezina, N.P.; Vol’fkovich, Y.M.; Kononenko, N.A.; Blinov, I.A. Investigation by standard porosimetry method of water distribution in heterogeneous ion-exchange membranes. Elektrokhimiya 1987, 23, 912–916. [Google Scholar]



| Resin Content, wt % | Qsw, mmol/g | Qdr, mmol/g | b, μm | |
|---|---|---|---|---|
| 45 | 1.88 ± 0.07 | 2.66 ± 0.09 | 0.29 | 540 ± 20 |
| 50 | 1.90 ± 0.02 | 2.83 ± 0.02 | 0.33 | 550 ± 10 |
| 55 | 1.93 ± 0.05 | 3.00 ± 0.07 | 0.36 | 585 ± 5 |
| 60 | 2.07 ± 0.04 | 3.38 ± 0.07 | 0.39 | 610 ± 5 |
| 65 | 2.16 ± 0.06 | 3.76 ± 0.09 | 0.42 | 655 ± 5 |
| 70 | 2.34 ± 0.05 | 4.23 ± 0.09 | 0.45 | 715 ± 15 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Akberova, E.M.; Vasil’eva, V.I.; Zabolotsky, V.I.; Novak, L. A Study of Ralex Membrane Morphology by SEM. Membranes 2019, 9, 169. https://doi.org/10.3390/membranes9120169
Akberova EM, Vasil’eva VI, Zabolotsky VI, Novak L. A Study of Ralex Membrane Morphology by SEM. Membranes. 2019; 9(12):169. https://doi.org/10.3390/membranes9120169
Chicago/Turabian StyleAkberova, Elmara M., Vera I. Vasil’eva, Victor I. Zabolotsky, and Lubos Novak. 2019. "A Study of Ralex Membrane Morphology by SEM" Membranes 9, no. 12: 169. https://doi.org/10.3390/membranes9120169
APA StyleAkberova, E. M., Vasil’eva, V. I., Zabolotsky, V. I., & Novak, L. (2019). A Study of Ralex Membrane Morphology by SEM. Membranes, 9(12), 169. https://doi.org/10.3390/membranes9120169






