Progress and Perspectives on Ceramic Membranes for Solvent Recovery
Abstract
1. Introduction
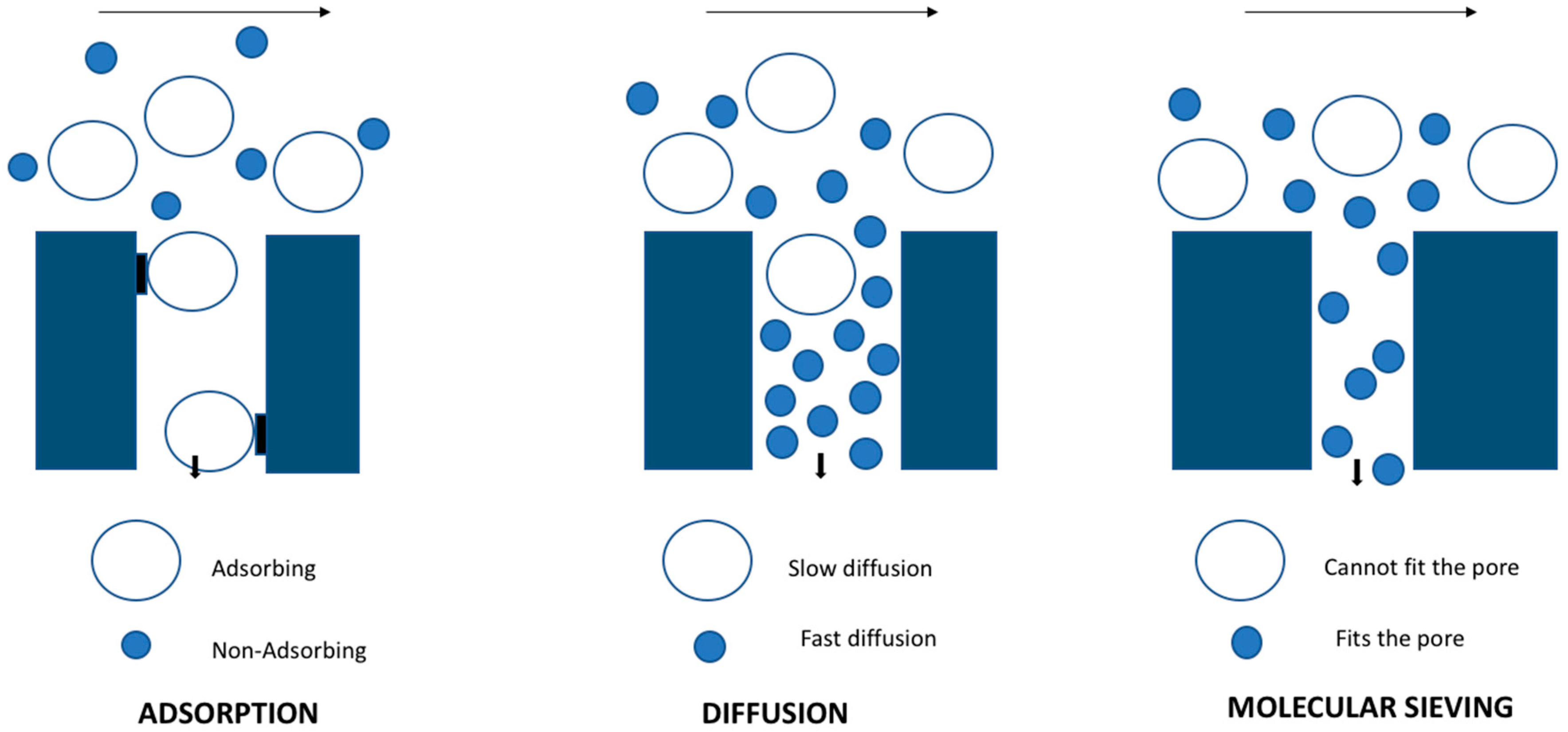
2. Inorganic Membranes
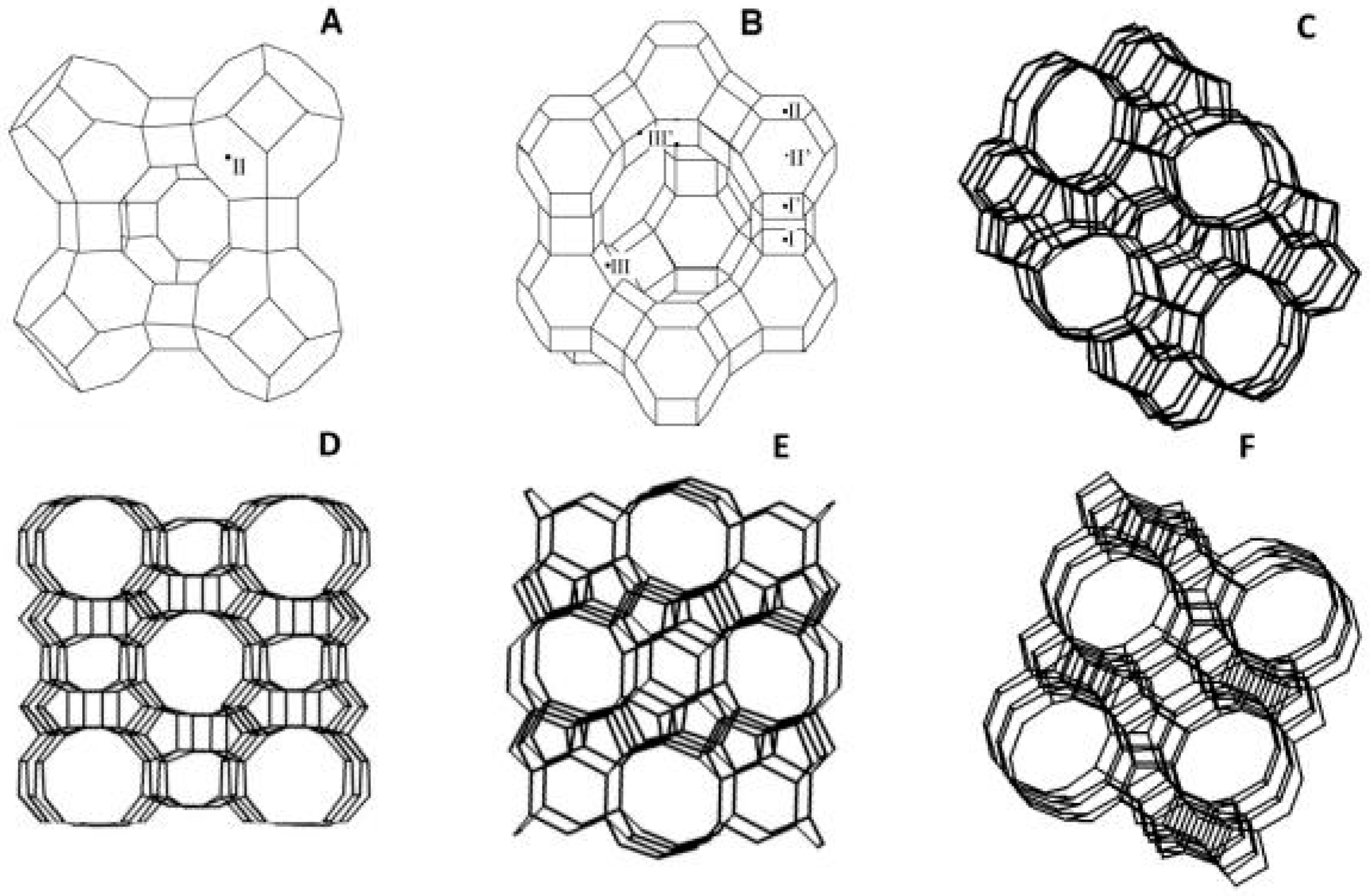
2.1. Zeolite-Based Membranes
2.2. Silica-Based Membranes
2.3. Mixed Matrix Membrane
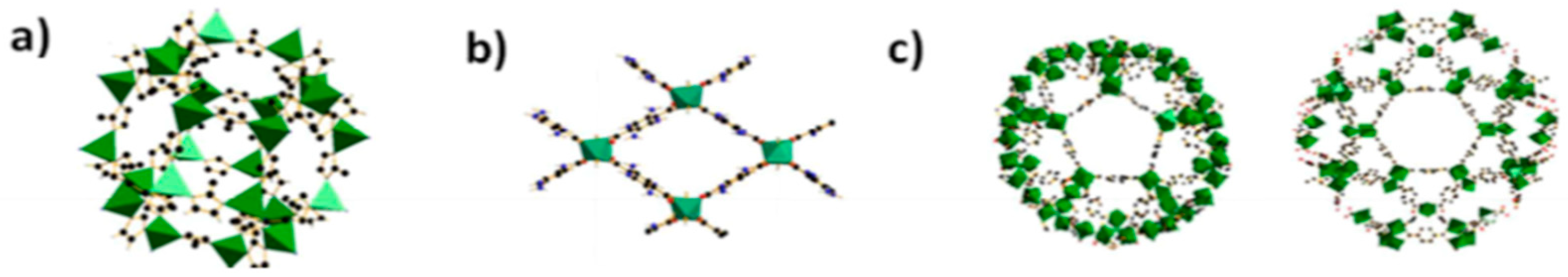
2.4. Metal Organic Frameworks
2.4.1. MIL-101 (Cr)
2.4.2. MIL-53
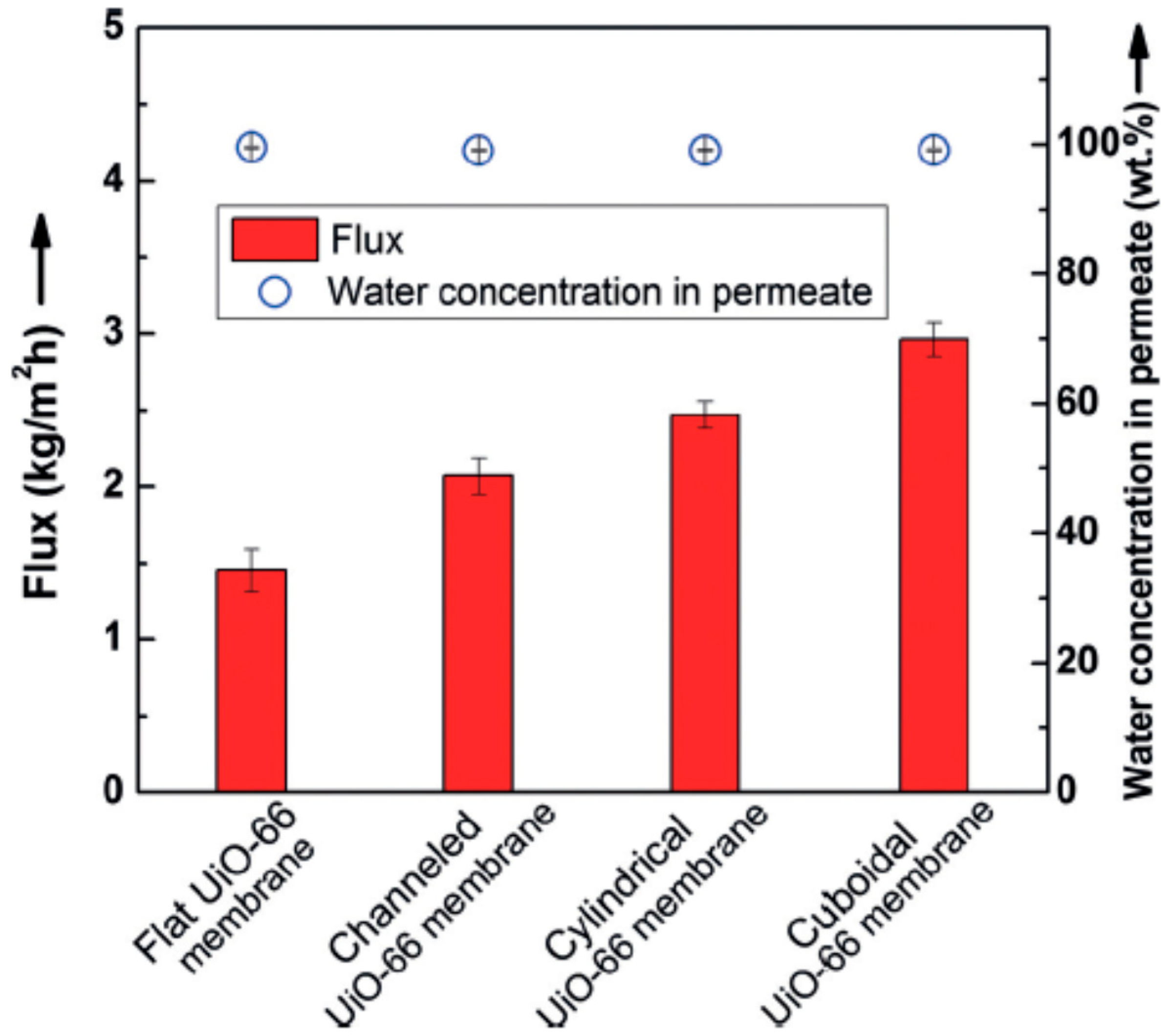
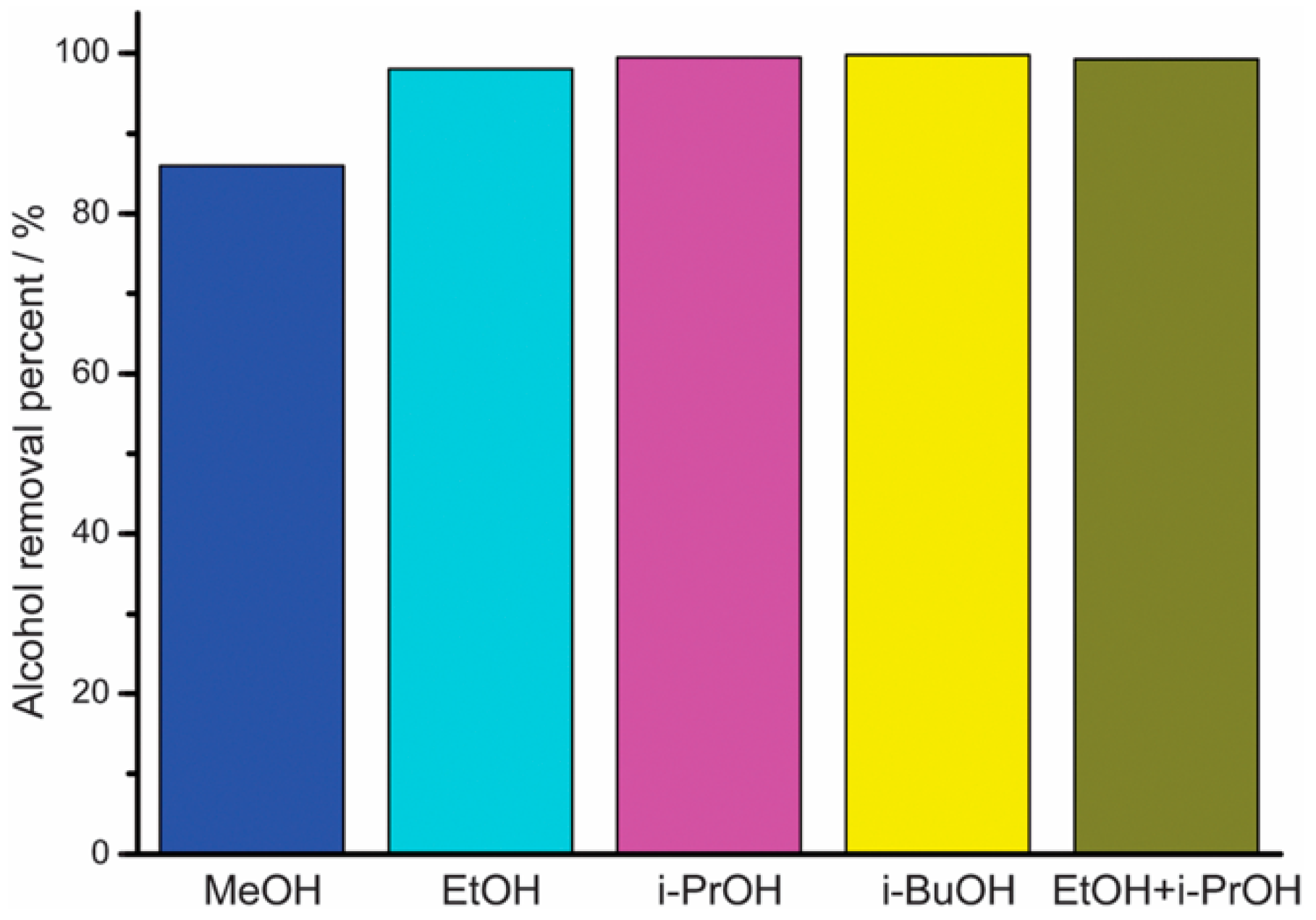
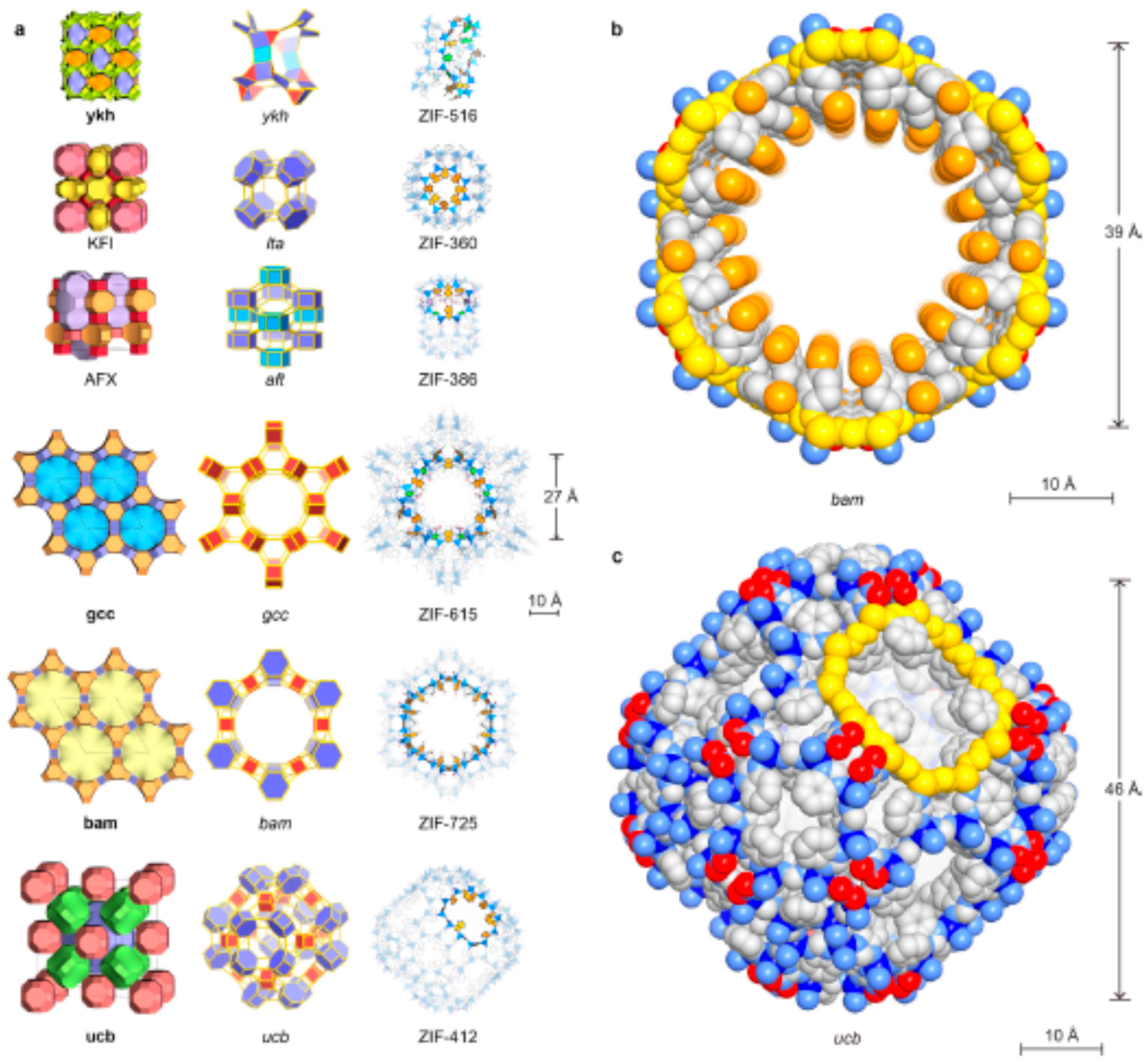
2.4.3. ZIF
Challenges in the Formation of ZIF Membranes
2.4.4. Metal Azolate Framework (MAF-6)
2.5. Grignard Functionalized Ceramic Membranes
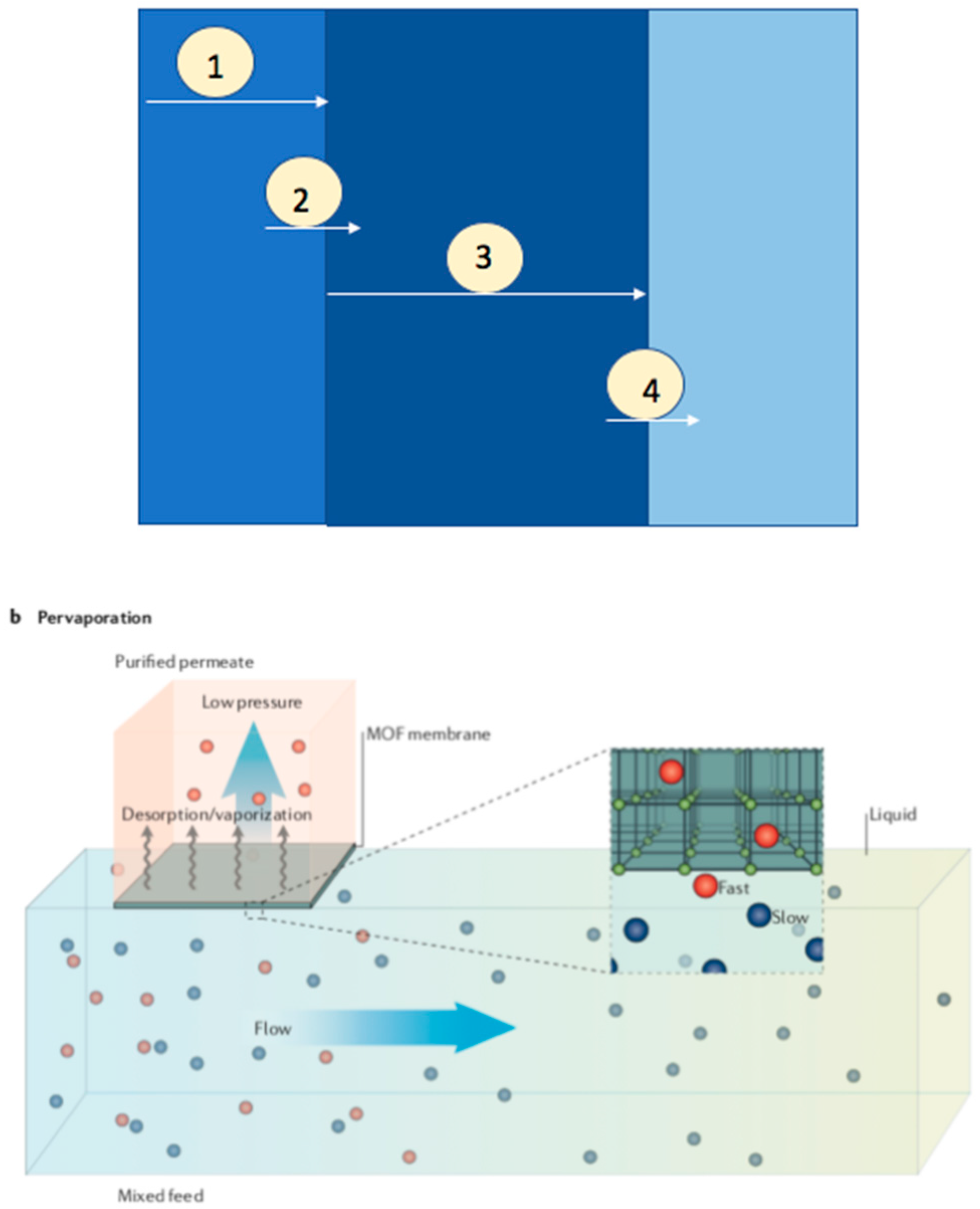
3. Pervaporation
4. Merits of Solvent Recovery
5. Conclusions
Funding
Conflicts of Interest
References
- Kim, J.F.; Székely, G.; Valtcheva, I.B.; Livingston, A.G. Increasing the sustainability of membrane processes through cascade approach and solvent recovery—Pharmaceutical purification case study. Green Chem. 2014, 16, 133–145. [Google Scholar] [CrossRef]
- Sawamura, K.I.; Furuhata, T.; Sekine, Y.; Kikuchi, E.; Subramanian, B.; Matsukata, M. Zeolite Membrane for Dehydration of Isopropylalcohol–Water Mixture by Vapor Permeation. ACS Appl. Mater. Interfaces 2015, 7, 13728–13730. [Google Scholar] [CrossRef] [PubMed]
- Sheldon, R.A. Metrics of Green Chemistry and Sustainability: Past, Present, and Future. ACS Sustain. Chem. Eng. 2018, 6, 32–48. [Google Scholar] [CrossRef]
- Cavanagh, E.J.; Savelski, M.J.; Slater, C.S. Optimization of environmental impact reduction and economic feasibility of solvent waste recovery using a new software tool. Chem. Eng. Res. Des. 2014, 92, 1942–1954. [Google Scholar] [CrossRef]
- Cheng, X.; Jiang, Z.; Cheng, X.; Yang, H.; Tang, L.; Liu, G.; Wang, M.; Wu, H.; Pan, F.; Cao, X. Water-selective permeation in hybrid membrane incorporating multifunctional hollow ZIF-8 nanospheres. J. Membr. Sci. 2018, 555, 146–156. [Google Scholar] [CrossRef]
- Fedosov, D.A.; Smirnov, A.V.; Knyazeva, E.E.; Ivanova, I.I. Zeolite membranes: Synthesis, properties, and application. Pet. Chem. 2011, 51, 657–667. [Google Scholar] [CrossRef]
- Yang, J.; Zhang, Y.B.; Liu, Q.; Trickett, C.A.; Puebla, E.G.; Monge, M.A.; Cong, H.; Aldossary, A.; Deng, H.; Yaghi, O.M. Principles of Designing Extra-large pore openings and cages in Zeolitic Imidazolate Frameworks. J. Am. Chem. Soc. 2017, 139, 6448–6455. [Google Scholar] [CrossRef]
- Solvent Recovery Recycling Units. Available online: https://www.maratek.com/en/solvent-recycling-equipment-sp-bu (accessed on 2 October 2019).
- Jyoti, G.; Keshav, A.; Anandkumar, J. Review on Pervaporation: Theory, Membrane Performance and Application to Intensification of Esterification Reaction. J. Eng. Educ. 2015, 2015, 927068. [Google Scholar] [CrossRef]
- Ciora, R.J.; Liu, P.K.T. Ceramic Membranes for Environmental Related Applications. Fluid/Particle Sep. J. 2003, 15, 51–60. [Google Scholar]
- Wee, S.L.; Tye, C.T.; Bhatia, S. Membrane separation process—Pervaporation through zeolite membrane. Sep. Purif. Technol. 2008, 63, 500–516. [Google Scholar] [CrossRef]
- Lee, P.S.; Hong, D.Y.; Cha, G.Y.; An, H.; Moon, S.Y.; Munsuk, S.; Chang, B.J.; Lee, J.S.; Kim, J.H. Mixed matrix membranes incorporated with three-dimensionally ordered mesopore imprinted (3DOm-i) zeolite. Sep. Purif. Technol. 2019, 210, 29–37. [Google Scholar] [CrossRef]
- Yin, H.; Lau, C.Y.; Rozowski, M.; Howardd, C.; Xu, Y.; Lai, T.; Lind, M.L. Free-standing ZIF-71/PDMS nanocomposite membranes for the recovery of ethanol and 1-butanol from water through pervaporation. J. Membr. Sci. 2017, 529, 286–292. [Google Scholar] [CrossRef]
- Li, Q.; Cheng, L.; Shen, J.; Shi, J.; Chen, G.; Zhao, J.; Jin, W. Improved ethanol recovery through mixed-matrix membrane with hydrophobic MAF-6 as filler. Sep. Purif. Technol. 2017, 178, 105–112. [Google Scholar] [CrossRef]
- Mao, H.; Zhen, H.; Ahmad, A.; Zhang, A.; Zhao, Z. In situ fabrication of MOF nanoparticles in PDMS membrane via interfacial synthesis for enhanced ethanol permselective pervaporation. J. Membr. Sci. 2018, 573, 344–358. [Google Scholar] [CrossRef]
- Yao, J.; Wang, H. Zeolitic imidazolate framework composite membranes and thin films: Synthesis and applications. Chem. Soc. Rev. 2014, 43, 4470–4493. [Google Scholar] [CrossRef]
- Bétard, A.; Fischer, R.A. Metal-organic framework thin films: From fundamentals to applications. Chem. Rev. 2012, 112, 1055–1083. [Google Scholar] [CrossRef]
- Kasik, A.; James, J.; Lin, Y.S. Synthesis of ZIF-68 Membrane on a ZnO Modified α-Alumina Support by a Modified Reactive Seeding Method. Ind. Eng. Chem. Res. 2016, 55, 2831–2839. [Google Scholar] [CrossRef]
- Kim, J.H.; Moon, S.J.; Park, S.H.; Cook, M.; Livingston, A.G.; Lee, Y.M. A robust thin film composite membrane incorporating thermally rearranged polymer support for organic solvent nanofiltration and pressure retarded osmosis. J. Membr. Sci. 2017, 550, 322–331. [Google Scholar] [CrossRef]
- Korelskiy, D.; Ye, P.; Fouladvand, S.; Karimi, S.; Sjöberg, E.; Hedlund, J. Efficient ceramic zeolite membranes for CO2/H2 separation. J. Mater. Chem. A 2015, 3, 12500–12506. [Google Scholar] [CrossRef]
- Liu, Y.; Wang, X.; Zhang, Y.; He, Y.; Gu, X. Scale-p of NaAeote membranes on α-Al2O3 hollow fibers by a secondary growth method with vacuum seeding. Chin. J. Chem. Eng. 2015, 23. [Google Scholar] [CrossRef]
- Zhang, J.; Liu, W. Thin porous metal sheet-supported NaA zeolite membrane for water/ethanol separation. J. Membr. Sci. 2011, 371, 197–210. [Google Scholar] [CrossRef]
- Mastropietro, T.F.; Brunetti, A.; Zit, P.F.; Poerio, T.; Richter, H.; Weyd, M.; Barbieri, G. Study of the separation properties of FAU membranes constituted by hierarchically assembled nanozeolites. Sep. Purif. Technol. 2015, 156, 321–327. [Google Scholar] [CrossRef]
- Okamoto, K.I.; Kita, H.; Horii, K.; Tanaka, K.; Kondo, M. Zeolite NaA membrane: Preparation, single-gas permeation, and pervaporation and vapor permeation of water/organic liquid mixtures. Ind. Eng. Chem. Res. 2001, 40, 163–175. [Google Scholar] [CrossRef]
- Bowen, T.C.; Wyss, J.C.; Noble, R.D.; Falconer, J.L. Measurements of diffusion through a zeolite membrane using isotopic-transient pervaporation. Microporous Mesoporous Mater. 2004, 71, 199–210. [Google Scholar] [CrossRef]
- Algieri, P.; Bernardo, G.; Golemme, G.; Barbieri, E.; Drioli, E. Permeation properties of a thin silicalite-1 (MFI) membrane. J. Membr. Sci. 2003, 222, 181–190. [Google Scholar] [CrossRef]
- Kanezashi, M.; O’Brien, J.; Lin, Y.S. Template-free synthesis of silicate membranes: permeation characterics and thermal stability improvement. J. Membr. Sci. 2006, 286, 213–222. [Google Scholar] [CrossRef]
- Wang, X.; Karakilic, P.; Liu, X.; Shan, M.; Nijmeijer, A.; Winnubst, L.; Gascon, J.; Kapteijn, F. One-Pot Synthesis of High-Flux b-Oriented MFI Zeolite Membranes for Xe Recovery. ACS Appl. Mater. Interfaces 2018, 10, 33574–33580. [Google Scholar] [CrossRef]
- Sels, B.F.; Schoonheydt, R.A. Transition-Metal Ions in Zeolites: Coordination and Activation of Oxygen. Inorg. Chem. 2010, 49, 3573–3583. [Google Scholar]
- Kumakiri, I.; Hashimoto, K.; Nakagawa, Y.; Inoue, Y.; Kanehiro, Y.; Tanaka, K.; Kita, H. Application of FAU zeolite membranes to alcohol/acrylate mixture systems. Catal. Today 2014, 236, 86–91. [Google Scholar] [CrossRef]
- Pina, M.P.; Arruebo, M.; Felipe, M.; Fleta, F.; Bernal, M.P.; Coronas, J.; Menéndez, M.; Santamaría, J. A semi-continuous method for the synthesis of NaA zeolite membranes on tubular supports. J. Membr. Sci. 2004, 244, 141–150. [Google Scholar] [CrossRef]
- Ma, J.; Shao, J.; Wang, Z.; Yan, Y. Preparation of Zeolite NaA Membranes on Macroporous Alumina Supports by Secondary Growth of Gel Layers. Ind. Eng. Chem. Res. 2014, 53, 6121–6130. [Google Scholar] [CrossRef]
- Wang, Z.; Ge, Q.; Gao, J.; Shao, J.; Liu, C.; Yan, Y. High-Performance Zeolite Membranes on Inexpensive Large-Pore Supports: Highly Reproducible Synthesis using a Seed Paste. Chem. Sus. Chem. 2011, 4, 1570–1573. [Google Scholar] [CrossRef]
- Jafari, M.; Bayat, A.; Mohammadi, T.; Kazemimoghadam, M. Dehydration of ethylene glycol by pervaporation using gamma alumina/NaA zeolite composite membrane. Chem. Eng. Res. Des. 2013, 91, 2412–2419. [Google Scholar] [CrossRef]
- Liu, Y.; Yang, Z.; Yu, C.; Gu, X.; Xu, N. Effect of seeding methods on growth of NaA zeolite membranes. Microporous Mesoporous Mater. 2011, 143, 348–356. [Google Scholar] [CrossRef]
- Ji, M.; Gao, X.; Wang, X.; Zhang, Y.; Jiang, J.; Gu, X. An ensemble synthesis strategy for fabrication of hollow fiber T-type zeolite membrane modules. J. Membr. Sci. 2018, 563, 460–469. [Google Scholar] [CrossRef]
- Asghari, M.; Mousavi, S.R.; Mohammadi, T. A comprehensive comparative study on morphology and pervaporative performance of porous-supported mesoporous zeolitic membranes. Microporous Mesoporous Mater. 2019, 280, 174–186. [Google Scholar] [CrossRef]
- Cao, Y.; Li, Y.X.; Wang, M.; Xu, Z.L.; Wei, Y.M.; Shen, B.J.; Zhu, K.K. High-flux NaA zeolite pervaporation membranes dynamically synthesized on the alumina hollow fiber inner-surface in a continuous flow system. J. Membr. Sci. 2019, 570–571, 445–454. [Google Scholar] [CrossRef]
- Zhang, Y.; Chen, S.; Shi, R.; Du, P.; Qiu, X.; Gu, X. Pervaporation dehydration of acetic acid through hollow fiber supported DD3R zeolite membrane. Sep. Purif. Technol. 2018, 204, 234–242. [Google Scholar] [CrossRef]
- Navajas, A.; Mittal, N.; Rangnekar, N.; Zhang, H.; Cornejo, A.; Gandía, L.M.; Tsapatsis, M. Environmental evaluation of the improvements for industrial scaling of zeolite membrane manufacturing by life cycle assessment. ACS Sustain. Chem. Eng. 2018, 6, 15773–15780. [Google Scholar] [CrossRef]
- Guerra, K.; Pellegrino, J. Development of a techno-economic model to compare ceramic and polymeric membranes. Sep. Sci. Technol. 2013, 48, 51–65. [Google Scholar] [CrossRef]
- Titus, M.P.; Llorens, J. Zeolite Membranes for Solvent Dehydration: An Overview and Review of Own Results. In Handbook of Zeolites: Structure, Properties and Applications (Materials Science and Technologies Series); Nova Science Publishers, Inc.: New York, NY, USA, 2019; pp. 523–554. [Google Scholar]
- Kallus, S.; Condre, J.M.; Hahn, A.; Golemme, G.; Algieri, C.; Dieudonne, P.; Timmins, P.; Ramsay, J.D.F. Colloidal zeolites and zeolite membranes. J. Mater. Chem. 2002, 12, 1–8. [Google Scholar] [CrossRef]
- Tsuru, T.; Sudoh, T.; Yoshioka, T.; Asaeda, M. Nanofiltration in non-aqueous solutions by porous silica–zirconia membranes. J. Membr. Sci. 2001, 185, 253–261. [Google Scholar] [CrossRef]
- Kosinov, N.; Hensen, E.J.M. Synthesis and separation properties of an α-alumina-supported high-silica MEL membrane. J. Membr. Sci. 2013, 447, 12–18. [Google Scholar] [CrossRef]
- Ma, Y.; Wang, J.; Tsuru, T. Pervaporation of water/ethanol mixtures through microporus silica membranes. Sep. Sci. Technol. 2009, 66, 479–485. [Google Scholar]
- Boutikos, P.; Pereira, C.S.M.; Silva, V.M.T.M.; Rodrigues, A.E. Performance evaluation of silica membrane for water-n-butanol binary mixture. Sep. Purif. Technol. 2014, 127, 18–28. [Google Scholar] [CrossRef]
- Tago, T.; Nakasaka, Y.; Kayoda, A.; Masuda, T. Preparation of hydrophilic silicate-1 nanocrystal-layered membrane for separation of water from water-acetone solution by pervaporation. Sep. Purif. Technol. 2007, 58, 7–11. [Google Scholar] [CrossRef]
- Wang, J.; Tsuru, T. Cobalt-doped silica membranes for pervaporation dehydration of ethanol/water solutions. J. Membr. Sci. 2011, 369, 13–19. [Google Scholar] [CrossRef]
- Kujawa, J.; Cerneaux, S.; Kujawski, W. Removal of hazardous volatile organic compounds from water by vacuum pervaporation with hydrophobic ceramic membranes. J. Membr. Sci. 2015, 474, 11–19. [Google Scholar] [CrossRef]
- Tres, M.V.; Racoski, J.C.; Luccio, M.D.; Oliveira, J.V.; Treichel, H.; de Oliveira, D.; Mazutti, M.A. Separation of soybean oil/n-hexane and soybean oil/n-butane mixtures using ceramic membranes. Food Res. Int. 2014, 63, 33–41. [Google Scholar] [CrossRef]
- Amelio, A.; Paredes, D.A.F.; Degrève, J.; Luis, P.; Van der Bruggen, B.; Espinosa, J. Conceptual model-based design and environmental evaluation of waste solvent technologies: Application to the separation of the mixture acetone-water. Sep. Purif. Technol. 2018, 53, 1791–1810. [Google Scholar] [CrossRef]
- Nagasawa, H.; Matsuda, N.; Kanezashi, M.; Yoshioka, T.; Tsuru, T. Pervaporation and vapor permeation characteristics of BTESE-derived organo-silica membranes and their long-term stability in a high-water-content IPA/water mixture. J. Membr. Sci. 2016, 498, 336–344. [Google Scholar] [CrossRef]
- Roy, B.; Dey, S.; Sahoo, C.G.; Roy, S.N.; Bandyopadhyay, S. Degumming, Dewaxing and Deacidification of Rice Bran Oil-Hexane Miscella Using Ceramic Membrane: Pilot Plant Study. J. Am. Oil Chem. Soc. 2014, 91, 1453–1460. [Google Scholar] [CrossRef]
- Maitlo, I.; Ali, S.; Shehzad, F.K.; Nie, J. Template-assisted hydrophobic porous silica membrane: A purifier sieve for organic solvents. J. Appl. Polym. Sci. 2018, 135, 45822. [Google Scholar] [CrossRef]
- Wang, H.T.; Mitra, A.; Cao, T.G.; Wang, Z.B.; Huang, L.M.; Li, S.; Li, Z.J.; Yu, Y.S. Synthesis and evaluation of pure-silicazeolite BEA as low dielectric constant material for microprocessors. Ind. Eng. Chem. Res. 2004, 43, 2946–2949. [Google Scholar]
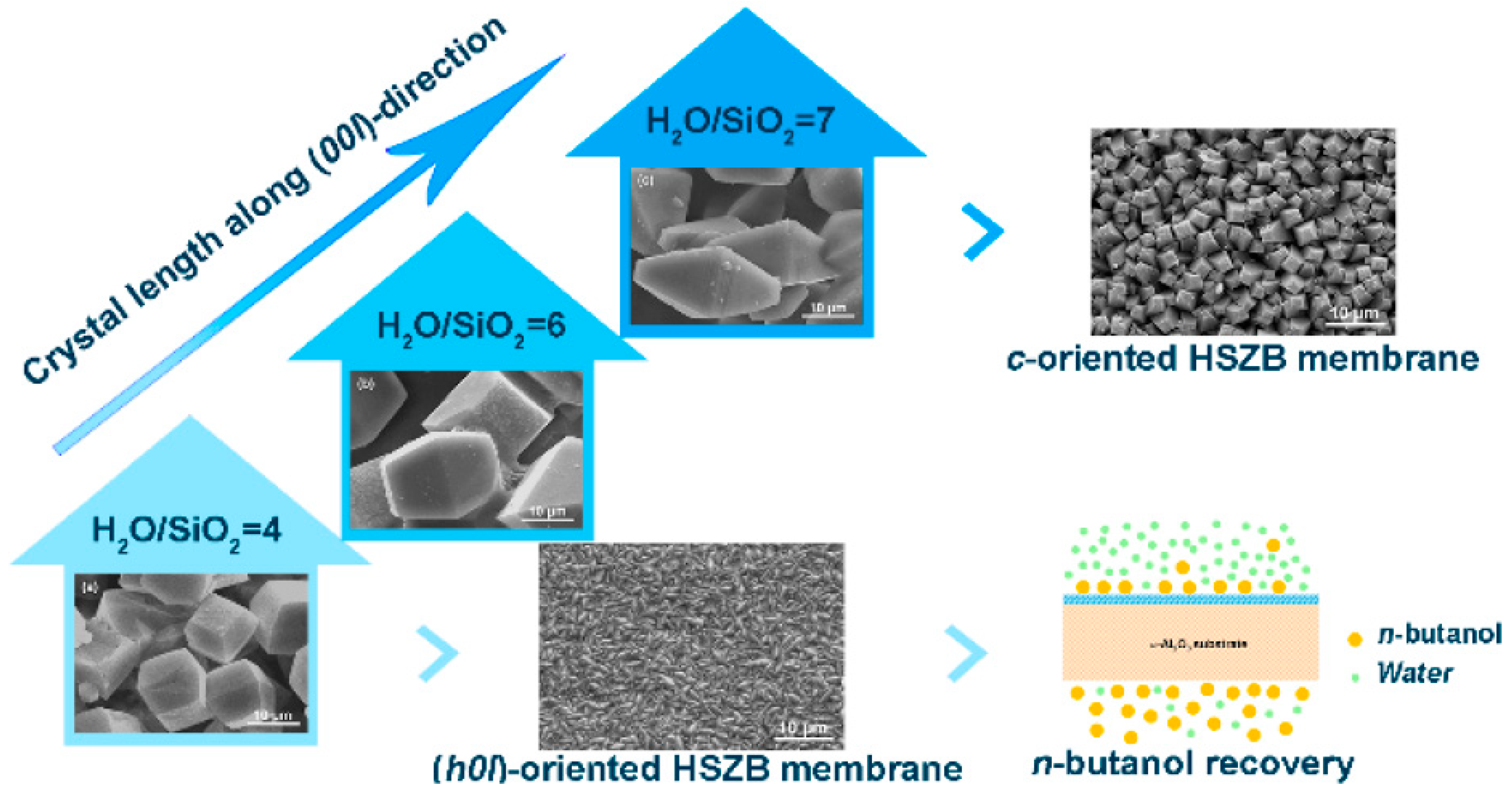
- Li, Y.; Ma, N.; Liu, X.; Zhang, B. Synthesis of Dense High-Silica Zeolite Beta Membranes with Controllable Orientation for n-Butanol Recovery from Dilute Aqueous Solution. Cryst. Growth Des. 2019, 19, 3166–3171. [Google Scholar] [CrossRef]
- Taleb, A.A.; Baraka, N.E.; Saffaj, N.; Laknifil, A.; Mamouni, R.; Fatni, A.; Hammadi, A.E.; Qacimi, N.E. New Tubular Ceramic Membranes from Natural Moroccan Clay for Microfiltration Application. E3S Web Conf. 2018, 37, 01011. [Google Scholar] [CrossRef]
- Vanherck, K.; Vandezande, P.; Aldea, S.O.; Vankelecom, I.F.J. Cross-linked polyimide membranes for solvent resistant nanofiltration in aprotic solvents. J. Membr. Sci. 2008, 320, 468–476. [Google Scholar] [CrossRef]
- Farahani, M.H.D.A.; Hua, D.; Chung, T.S. Cross-linked mixed matrix membranes (MMMs) consisting of amine-functionalized multi-walled carbon nanotubes and P84 polyimide for organic solvent nanofiltration (OSN) with enhanced flux. J. Membr. Sci. 2018, 548, 319–331. [Google Scholar] [CrossRef]
- Farahani, M.H.D.A.; Hua, D.; Chung, T.S. Cross-linked mixed matrix membranes consisting of carboxyl-functionalized multi-walled carbon nanotubes and P84 polyimide for organic solvent nanofiltration (OSN). Sep. Purif. Technol. 2017, 186, 243–254. [Google Scholar] [CrossRef]
- Sorribas, S. Pervaporation and membrane reactor performance of polyimide based mixed matrix membranes containing MOF HKUST-1. Chem. Eng. Sci. 2015, 124, 37–44. [Google Scholar] [CrossRef]
- Sorribas, S.; Gorgojo, P.; Téllez, C.; Coronas, J.; Livingston, A.G. High flux thin film nanocomposite membranes based on metal–organic frameworks for organic solvent nanofiltration. J. Am. Chem. Soc. 2013, 135, 15201–15208. [Google Scholar] [CrossRef] [PubMed]
- Su, P.; Zhang, X.; Li, Y.; Chen, H.; Meng, Q.; Zhang, G. Distillation of alcohol/water solution in hybrid metal–organic framework hollow fibers. AIChE J. 2019, 65, e16693. [Google Scholar] [CrossRef]
- Naik, P.V.; Wee, L.K.; Meledina, M.; Turner, S.; Li, Y.; Tendeloo, G.V.; Martens, J.A.; Vankelecom, I.F.J. PDMS membranes containing ZIF-coated mesoporous silica spheres for efficient ethanol recovery via pervaporation. J. Mater. Chem. A 2016, 4, 12790. [Google Scholar] [CrossRef]
- Fan, H.; Shi, Q.; Yan, H.; Ji, S.; Dong, J.; Zhang, G. Simultaneous Spray Self- Assembly of Highly loaded ZIF-8-PDMS Nanohybrid Membranes exhibiting exceptionally high biobutanol- permselective pervaporation. Angew. Chem. Int. Eng. Ed. 2014, 53, 5578–5582. [Google Scholar] [CrossRef]
- Kasik, A.; Dong, X.; Lin, Y.S. Synthesis and stability of zeolitic imidazolate framework-68 membranes. Microporous Mesoporous Mater. 2014, 204, 99–105. [Google Scholar] [CrossRef]
- Zhu, L.; Yu, H.; Zhang, H.; Shen, J.; Xue, L.; Gao, C.; van der Bruggen, B. Mixed matrix membranes containing MIL-53 (Al) for potential application in organic solvent nanofiltration. RSC Adv. 2015, 5, 73068–73076. [Google Scholar] [CrossRef]
- Huang, K.; Wang, B.; Guo, S.; Li, K. Micropatterned Ultrathin MOF Membranes with Enhanced Molecular Sieving Property. Angew. Chem. Int. Eng Ed. 2018, 57, 13892–13896. [Google Scholar] [CrossRef]
- Zirehpour, A.; Rahimpour, A.; Shamsabadi, A.A.; Gh, M.S.; Soroush, M. Mitigation of Thin-Film Composite Membrane Biofouling via Immobilizing Nano-Sized Biocidal Reservoirs in the Membrane Active Layer. Environ. Sci. Technol. 2017, 51, 5511–5522. [Google Scholar] [CrossRef]
- Satheeshkumar, C.; Yu, H.J.; Park, H.; Kim, M.; Lee, J.S.; Seo, M. Thiol–ene photopolymerization of vinylfunctionalized metal–organic frameworks towards mixed-matrix membranes. J. Mater. Chem. A 2018, 6, 21961–21968. [Google Scholar] [CrossRef]
- Demirel, O.H.; Rijnaarts, T.; de Wit, P.; Wood, J.A.; Benes, N.E. Electroforming of a metal–organic framework on porous copper hollow fibers. J. Mater. Chem. A 2019, 7, 12616–12626. [Google Scholar] [CrossRef]
- Maya, F.; Cabello, C.P.; Clavijo, S.; Palomino, G.T. Zeolitic imidazolate framework dispersions for the fast and highly efficient extraction of organic. RSC Adv. 2015, 5, 28203–28210. [Google Scholar] [CrossRef]
- Xu, S.J.; Shen, Q.; Chen, G.E.; Xu, Z.L. Novel β-CD@ZIF-8 Nanoparticles-Doped Poly (m-phenyleneisophthalamide) (PMIA) Thin-Film Nanocomposite (TFN) Membrane for Organic Solvent Nanofiltration (OSN). ACS Omega 2018, 3, 11770–11787. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Liu, Q.; Huang, A. A superhydrophobic zeolitic imidazolate framework (ZIF-90) with high steam stability for efficient recovery of bioalcohols. Chem. Commun. 2016, 52, 3400–3402. [Google Scholar] [CrossRef] [PubMed]
- Banerjee, R.; Furukawa, H.; Britt, D.; Knobler, C.; Yachi, O.M.; O’Keef, M. Control of pore size and Functionality in IsoreticularZeoliticImidazolate Frameworks and their Carbon Dioxide Selective Capture Properties. J. Am. Chem. Soc. 2009, 131, 3875–3877. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.; Cote, A.P.; Furukawa, H.; O’Keeffe, M.; Yaghi, O.M. Functional metal-organic frameworks: Gas storage, separation and catalysis. Nature 2008, 453, 207–211. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.J.; Xiao, Y.L.; Liu, D.H.; Yang, Q.Y.; Zhong, C.L. A hybrid zeolitic imidazolate framework membrane by mixed-liner synthesis for efficient CO2 capture. Chem. Commun. 2013, 49, 600–602. [Google Scholar] [CrossRef] [PubMed]
- Van Goethem, C.; Mertens, M.; Cirujano, F.G.; Seo, J.W.; de Vos, D.; Vankelecom, I.F.J. Improved MOF nanoparticle recovery and purification using crosslinked PVDF membranes. Chem. Commun. 2018, 54, 7370–7373. [Google Scholar] [CrossRef]
- Dong, X.L.; Lin, Y.S. Synthesis of an organophilic ZIF-71 membrane for pervaporation solvent separation. Chem. Commun. 2013, 12, 1196–1198. [Google Scholar]
- Li, W.; Wu, W.; Li, Z.; Shi, J.; Xia, Y. Sol–gel asynchronous crystallization of ultraselective metal–organic framework membranes for gas separation. J. Mater. Chem. A 2018, 6, 16333–16340. [Google Scholar] [CrossRef]
- Kong, L.; Zhang, X.F.; Liu, H.; Qiu, J. Synthesis of a highly stable ZIF-8 membrane on a macroporous ceramic tube by manual-rubbing ZnO deposition as a multifunctional layer. J. Membr. Sci. 2015, 490, 354–363. [Google Scholar] [CrossRef]
- Hosseinabadi, S.R.; Wyns, K.; Buekenhoudt, A.; van der Bruggen, B.; Ormerod, D. Performance of Grignard functionalized ceramic nanofiltration membranes. Sep. Purif. Technol. 2015, 147, 320–328. [Google Scholar] [CrossRef]


| Separation System | Temp (K) | XW (wt%) | Q (kg/(m2·h)) | α [-] |
|---|---|---|---|---|
| Ethanol | 348 | 10 | 2.15 | 10,000 |
| 348 | 5 | 1.10 | 16,000 | |
| 348 | 0.5 | 0.012 | 5100 | |
| Methanol | 323 | 10 | 0.57 | 2100 |
| 323 | 5 | 0.23 | 2500 | |
| n-propanol | 348 | 10 | 1.91 | 18,000 |
| i-propanol | 348 | 10 | 1.76 | 10,000 |
| Acetone | 323 | 10 | 0.91 | 5600 |
| 323 | 5 | 0.83 | 6800 | |
| Dioxane | 333 | 10 | 1.87 | >9000 |
| DMF | 333 | 10 | 0.95 | >9000 |
| Membrane | Contact Angle (°) | |||
|---|---|---|---|---|
| TFC (without MOF) | - | - | ~73 | - |
| TFN-NH2-MIL-53 (Al) | - | - | ~49 | - |
| TFN-MIL-53 (Al) | - | - | ~54 | - |
| TFN-ZIF-8 | - | - | ~75 | - |
| TFN-MIL-101 (Cr) | ~53 (0.05) | ~52 (0.1) | ~50 (0.2) | ~43 (0.4) |
| Membrane | Permeance Methanol (L·m−2·h−1·bar−1) | Permeance Methanol/PS (Polystyrene) (L·m−2·h−1·bar−1) |
|---|---|---|
| TFC (without MOF) | 1.8 | 1.5 |
| TFN-NH2-MIL-53 (Al) | 2.3 | 1.8 |
| TFN-MIL-53 (Al) | 2.3 | 1.9 |
| TFN-ZIF-8 | 2.5 | 2.1 |
| TFN-MIL-101 (Cr) | 4.2 | 3.9 |
| MOF Content (wt%) | Contact Angle (°C) |
|---|---|
| 0 | 86 ± 2 |
| 0.3 | 79 ± 2 |
| 0.5 | 76 ± 1 |
| 1.0 | 73 ± 1 |
| 1.5 | 71 ± 2 |
| Material | Molecular Name | Pore Size (Å) | References |
|---|---|---|---|
| ZIF-7 | Zn (benzimidazole)2 | 3.0 | [16,76,77] |
| ZIF-8 | Zn(2-methylimidazole)2 | 3.4 | [16,76,77] |
| ZIF-90 | Zn(imidazolate-2-carboxaldehyde) | 3.5 | [16,76,77] |
| ZIF-71 | Zn(4,5-dichloroimidazole)2 | 4.2 | [16,76,77] |
| ZIF-69 | Zn(5chlorobenzimidazole)(2-nitroimidazole) | 4.4 | [16,76,77] |
| ZIF 68 | Zn(benzimidazole)(2-nitroimidazole) | 7.5 | [18] |
| ZIF 22 | Zn(5-azabenzimidazolate)2 | 0.44 | [77] |
| ZIF 78 | Zn(5-nitrobenzimidazole)(2-nitroimidazole) | 0.38 | [78] |
| ZIF 95 | Zn(5-chlorobenzimidazole)2 | 0.37 | [79] |
| ZIF 360 | Zn(bIM)1.00 (nIM)0.70(IM)0.30 | 4.8 | [7] |
| ZIF 365 | Zn(cbIM)0.95 (nIM)0.60(IM)0.45 | 5.0 | [7] |
| ZIF-410 | Zn(cbIM)1.0 (aIM)0.90 | 5.0 | [7] |
| ZIF486 | Zn(nbIM)0.20 (mIM)0.65(IM)1.15 | 6.0 | [7] |
| ZIF412 | Zn(bIM)1.13 (nIM)0.62(IM)0.25 | 8.2 | [7] |
| ZIF413 | Zn(mIM)1.03 (nIM)0.64(IM)0.33 | 6.8 | [7] |
| ZIF414 | Zn(nbIM)0.921 (mIM)0.62(IM)0.47 | 4.6 | [7] |
| ZIF725 | Zn(bbIM)1.35 (nIM)0.40(IM)0.25 | 22.5 | [7] |
| System | Total Flux (g·m−2·h−1) | Separation Factor | Alcohol or DMC Permeance (g·m−2·h−1·kPa−1) | Selectivity |
|---|---|---|---|---|
| EtOH-water | 322.18 | 6.09 | 117.43 | 1.50 |
| MeOH-water | 394.64 | 21.38 | 260.22 | 4.32 |
| DMC-MeOH | 271.21 | 5.34 | 102.89 | 8.08 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ruthusree, S.; Sundarrajan, S.; Ramakrishna, S. Progress and Perspectives on Ceramic Membranes for Solvent Recovery. Membranes 2019, 9, 128. https://doi.org/10.3390/membranes9100128
Ruthusree S, Sundarrajan S, Ramakrishna S. Progress and Perspectives on Ceramic Membranes for Solvent Recovery. Membranes. 2019; 9(10):128. https://doi.org/10.3390/membranes9100128
Chicago/Turabian StyleRuthusree, Senthilnathan, Subramanian Sundarrajan, and Seeram Ramakrishna. 2019. "Progress and Perspectives on Ceramic Membranes for Solvent Recovery" Membranes 9, no. 10: 128. https://doi.org/10.3390/membranes9100128
APA StyleRuthusree, S., Sundarrajan, S., & Ramakrishna, S. (2019). Progress and Perspectives on Ceramic Membranes for Solvent Recovery. Membranes, 9(10), 128. https://doi.org/10.3390/membranes9100128







