A Review on the Progress in Nanoparticle/C Hybrid CMS Membranes for Gas Separation
Abstract
1. Introduction
2. Fe Series Nanoparticles Hybrid CMS Membranes
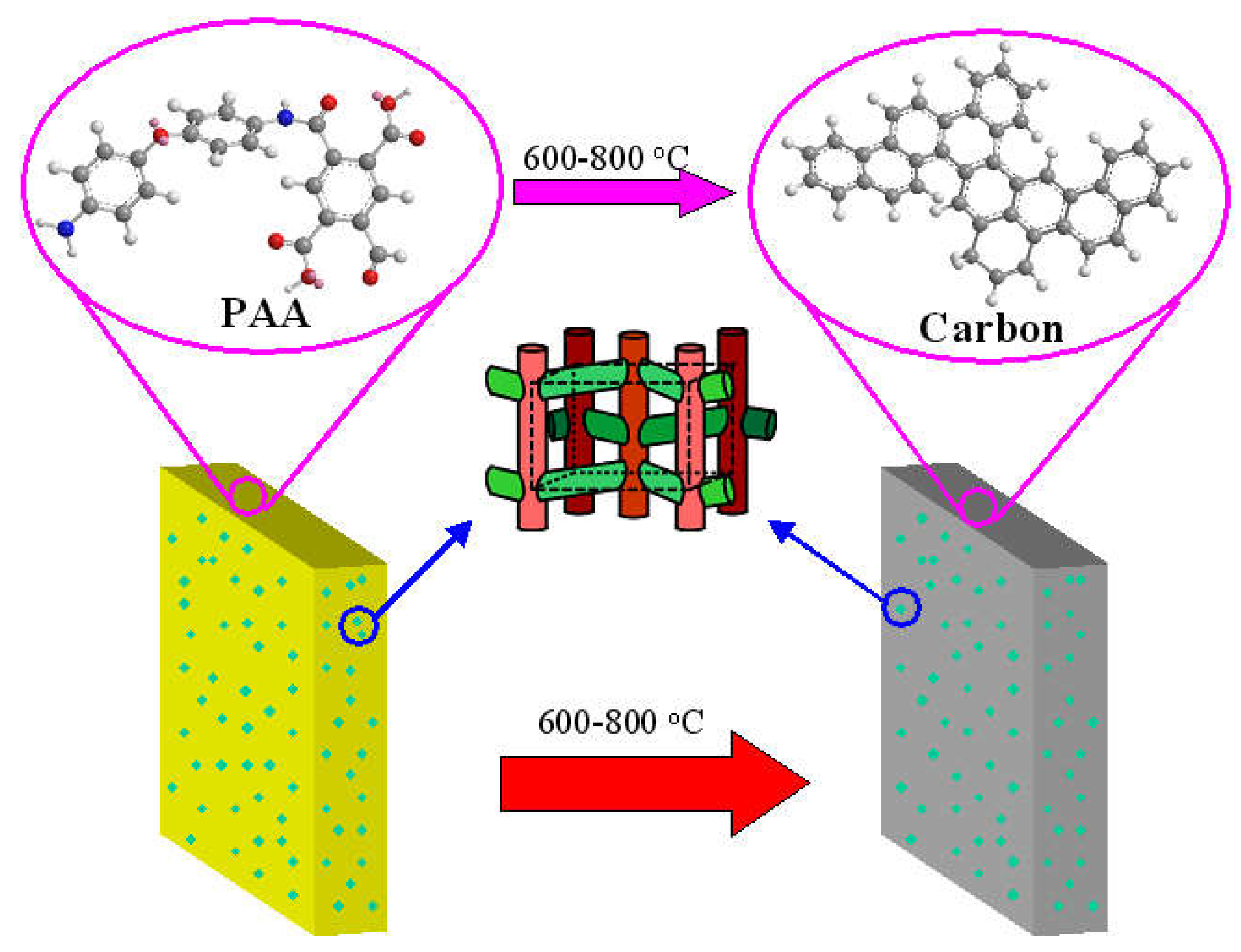
2.1. Preparation of the Hybrid CMS Membranes
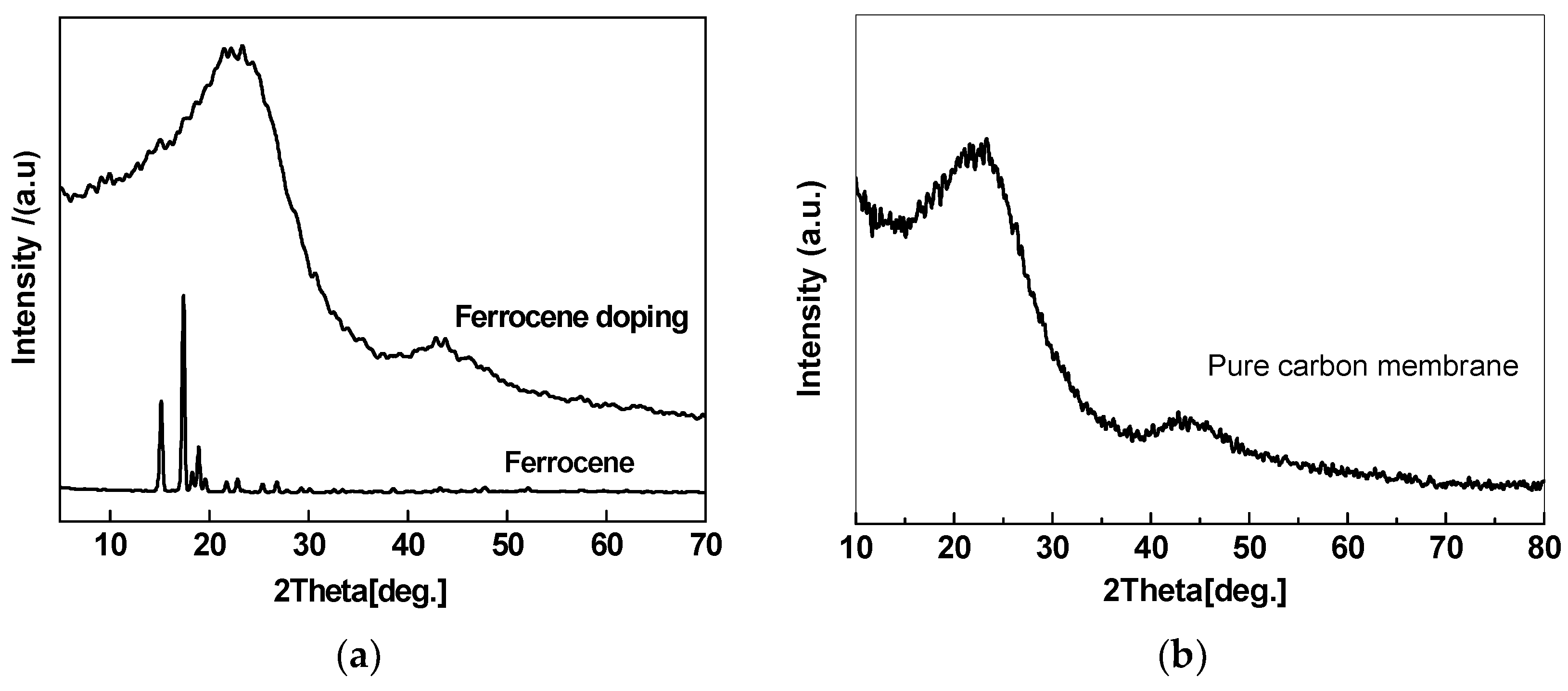
2.2. Ferrocene/PAA Based Hybrid CMS Membrane
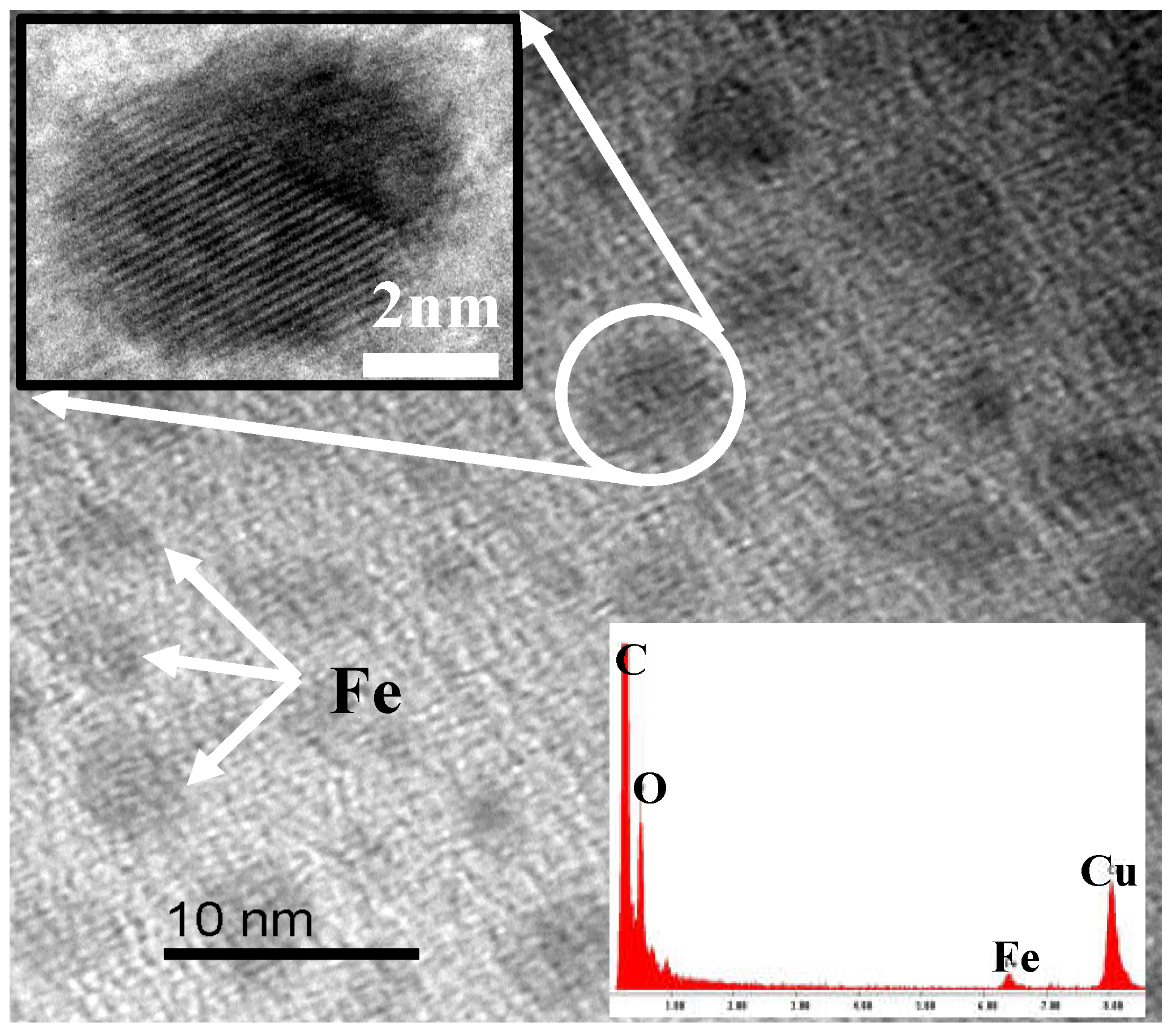
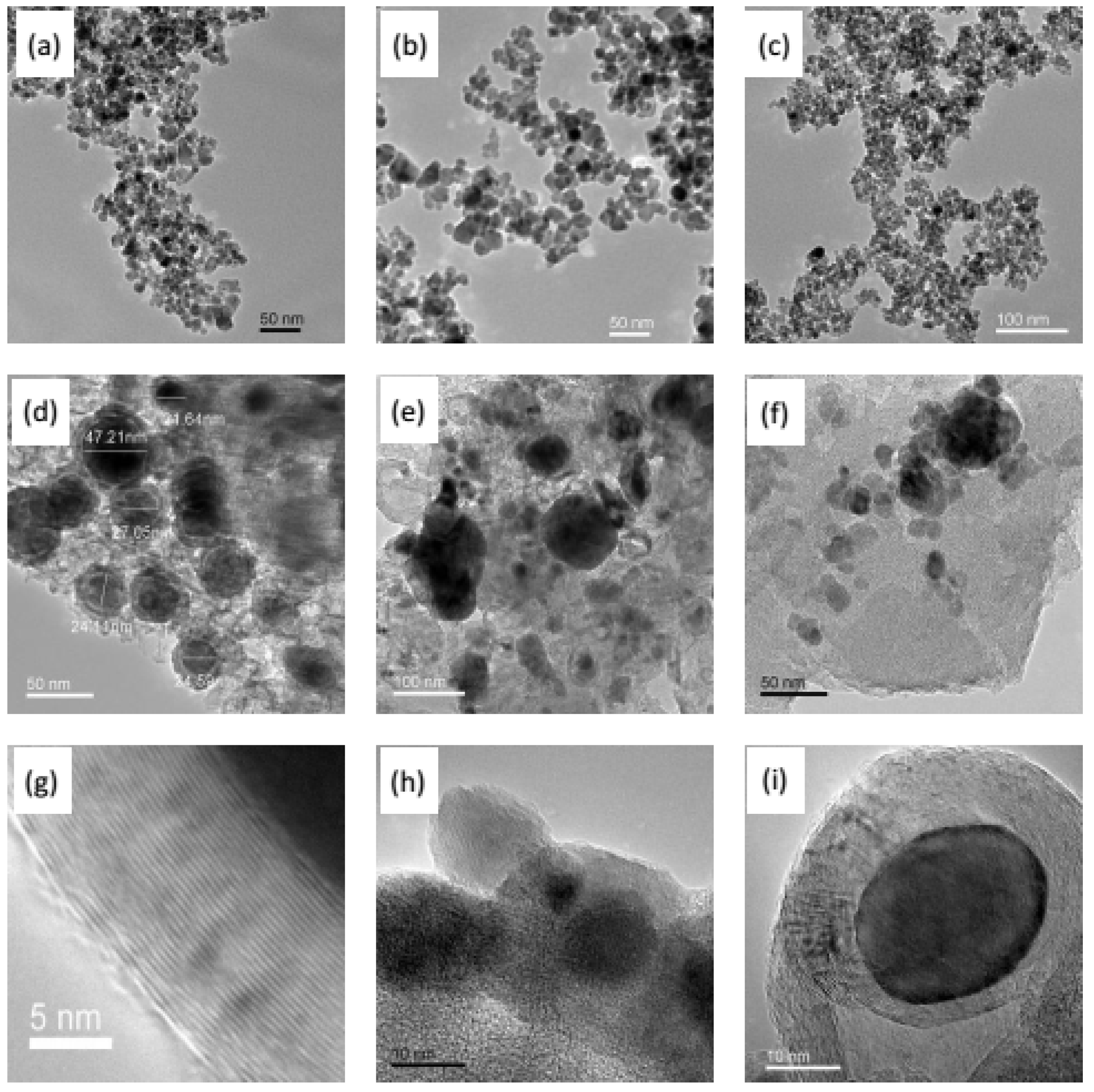
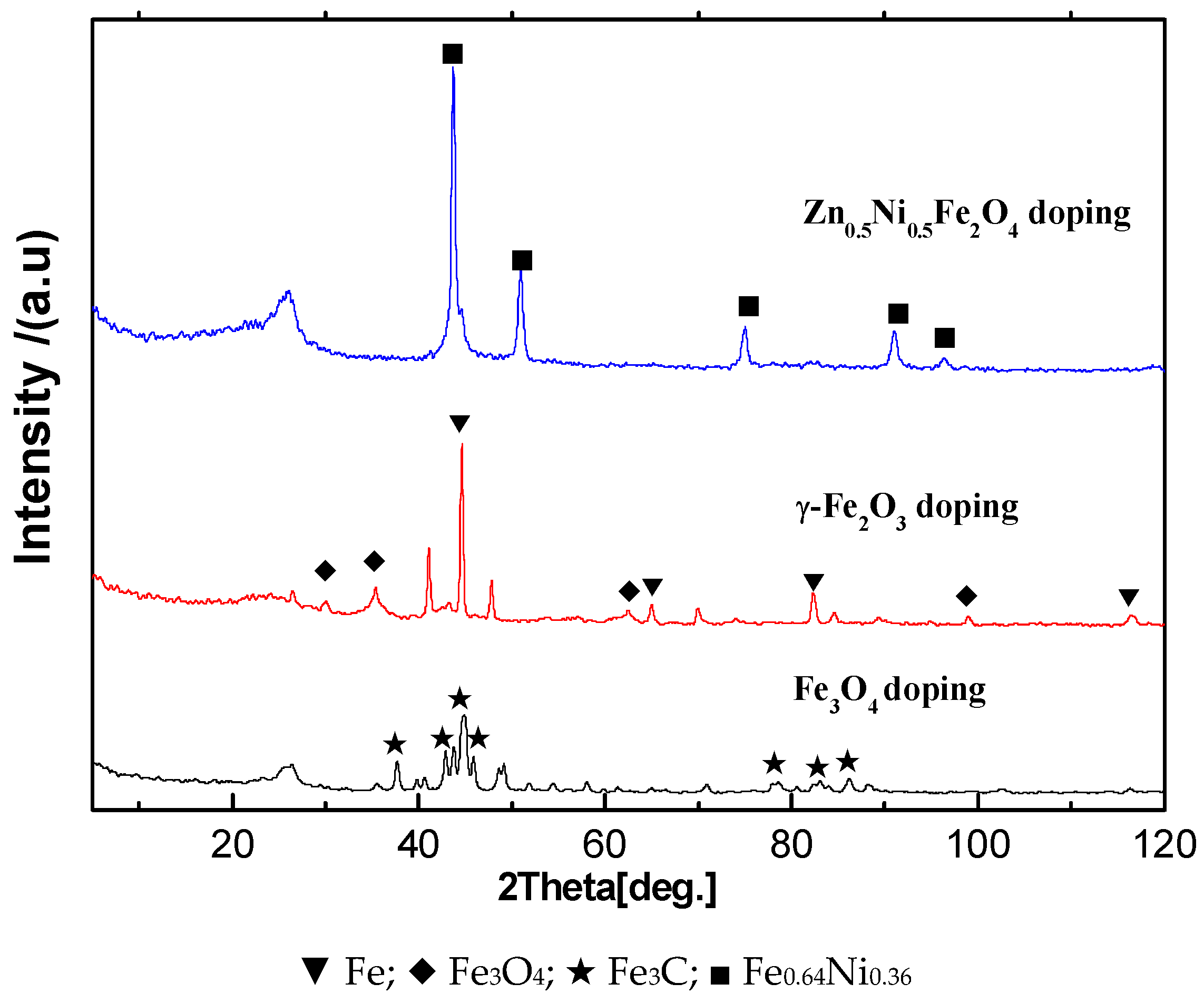
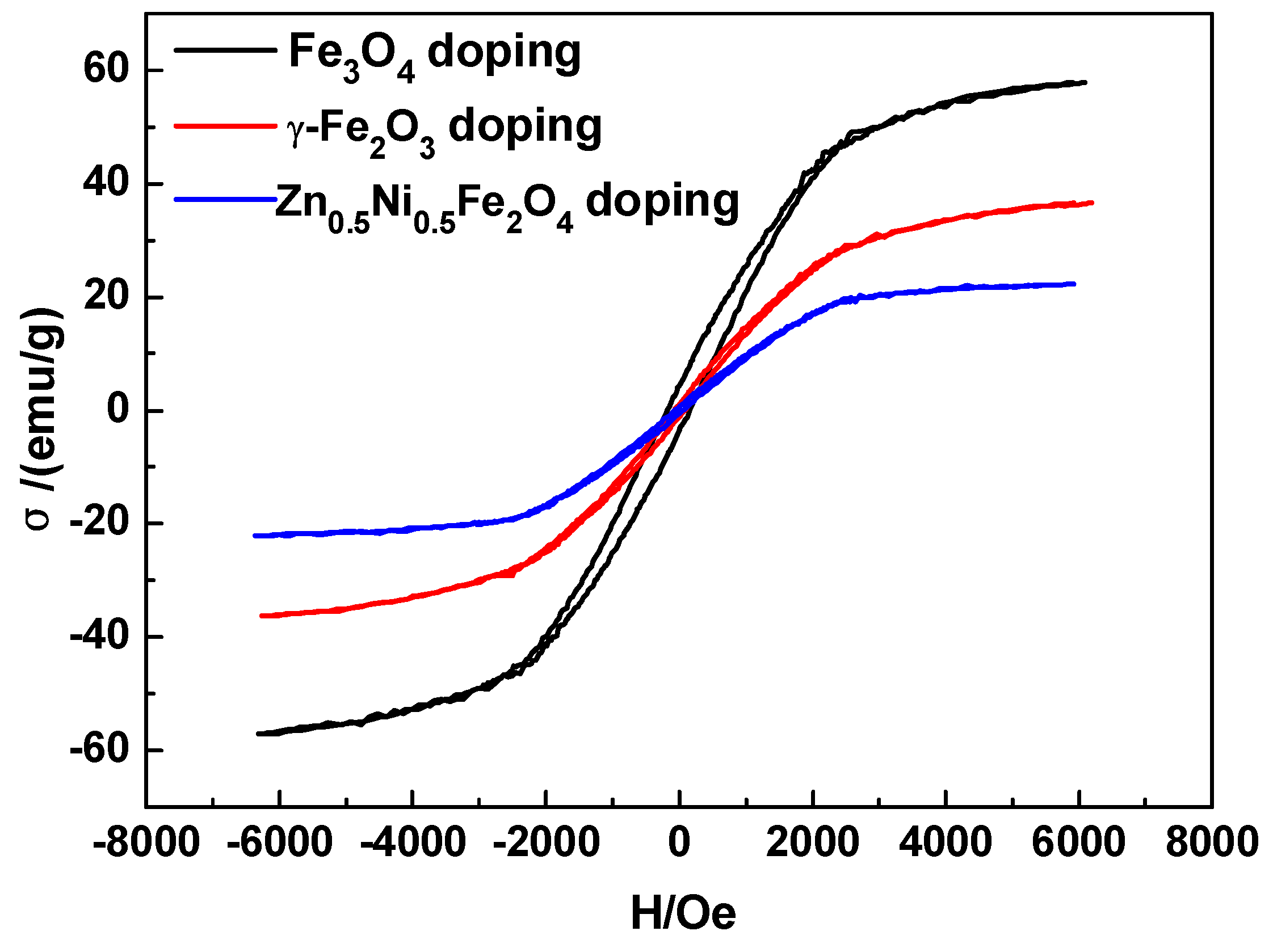
2.3. Fe Series Magnetic Nanoparticle/C Hybrid Membranes
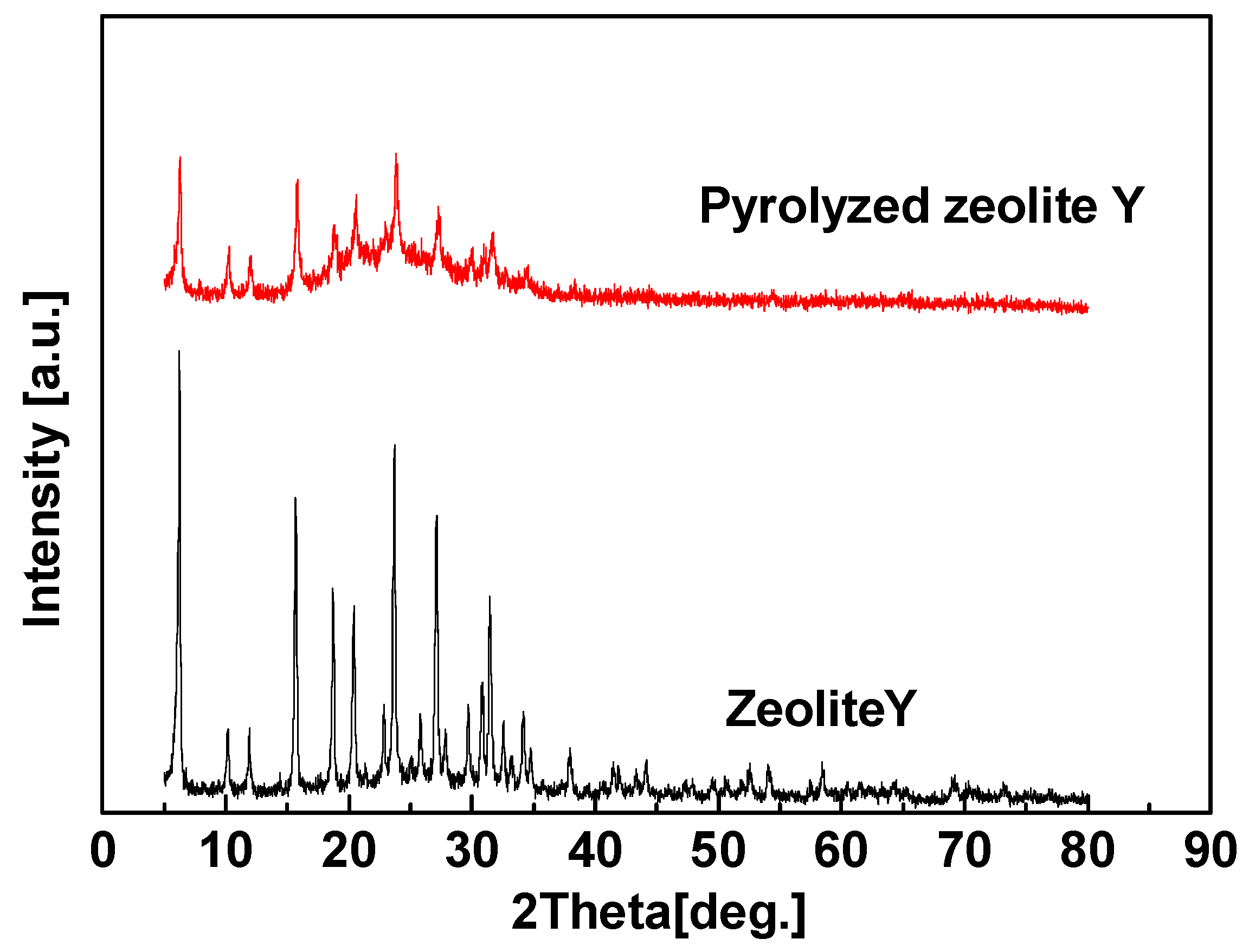
3. Microporous Zeolite/C Hybrid Membranes
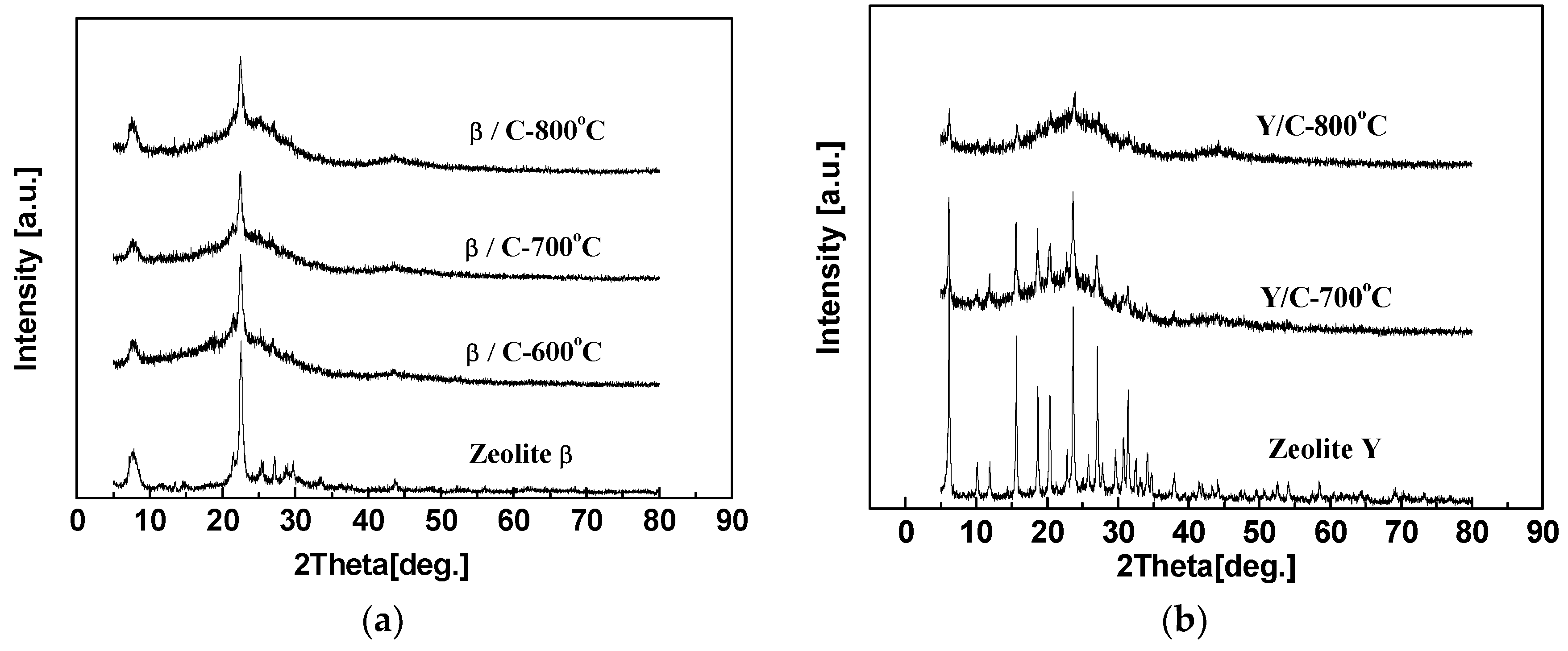
3.1. The Effect of Different Zeolites on the Structure of the Hybrid CMS Membranes
3.2. The Effect of Zeolite Content on the Gas Separation Performance of Hybrid CMS Membranes
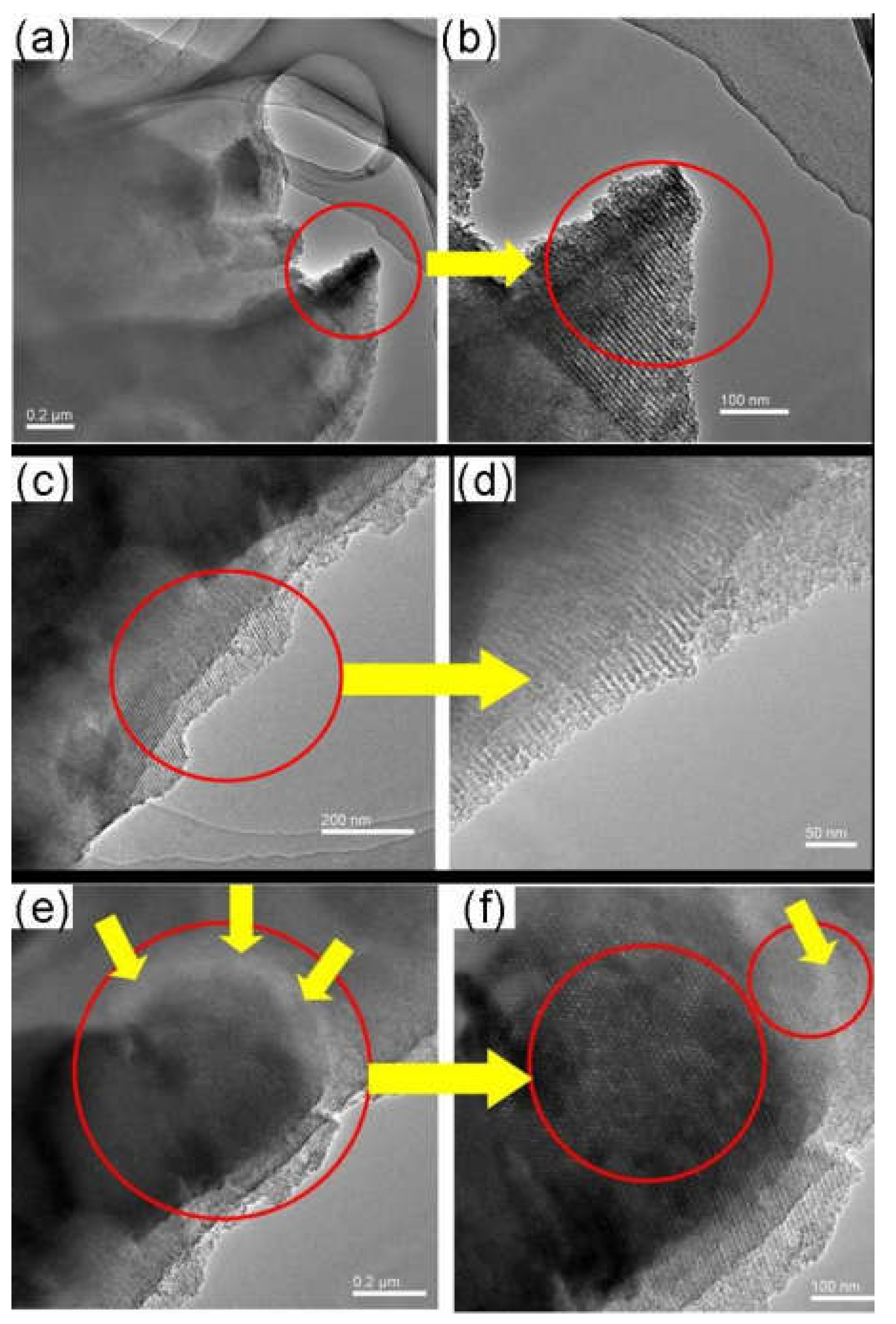
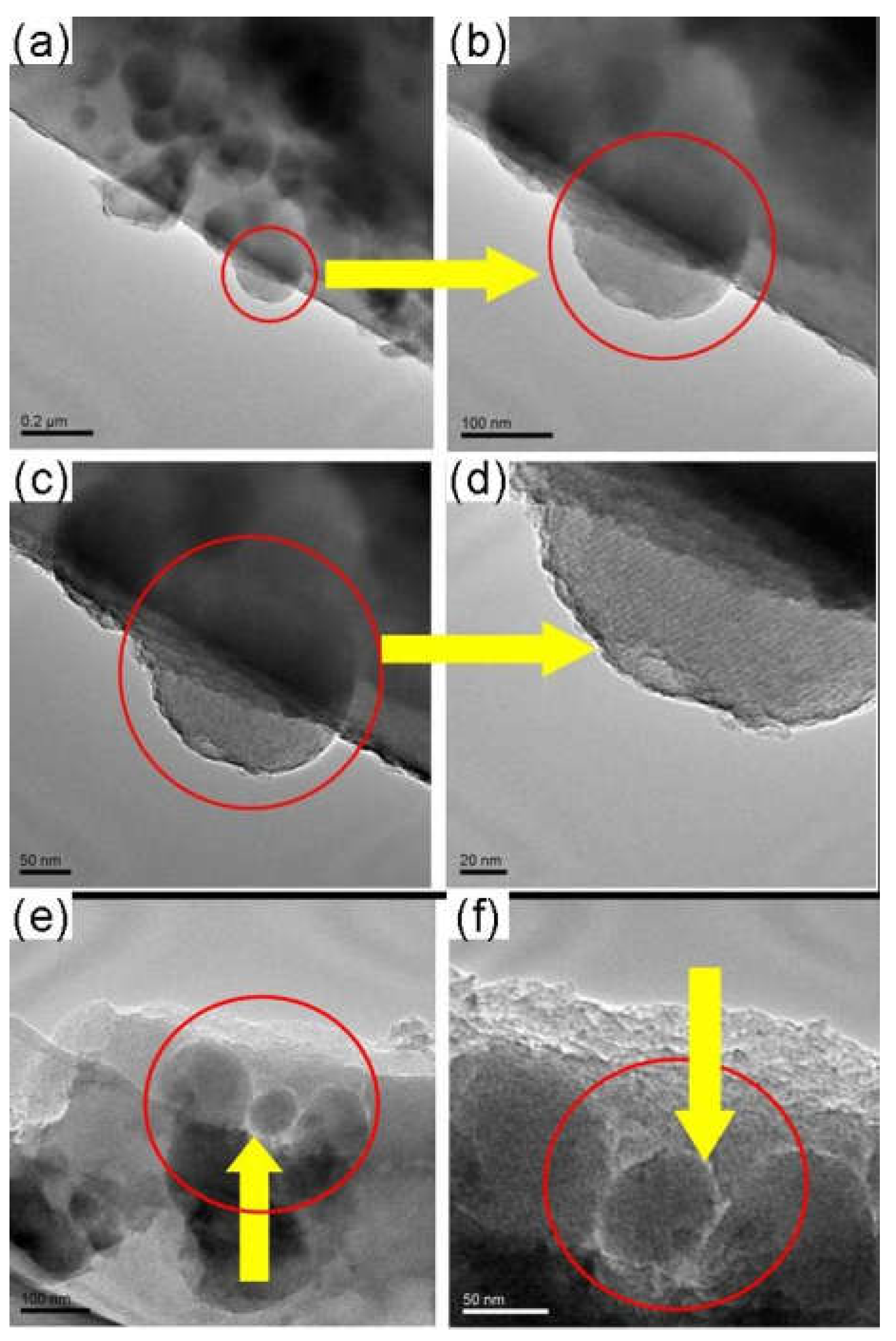
3.3. The Effect of Zeolite Particle Size on the Structure and Property of Hybrid CMS Membranes
3.4. The Effect of Carbonization Temperature and Zeolite Channel Integrity on the Structure and Property of the Zeolite/C Hybrid Membranes
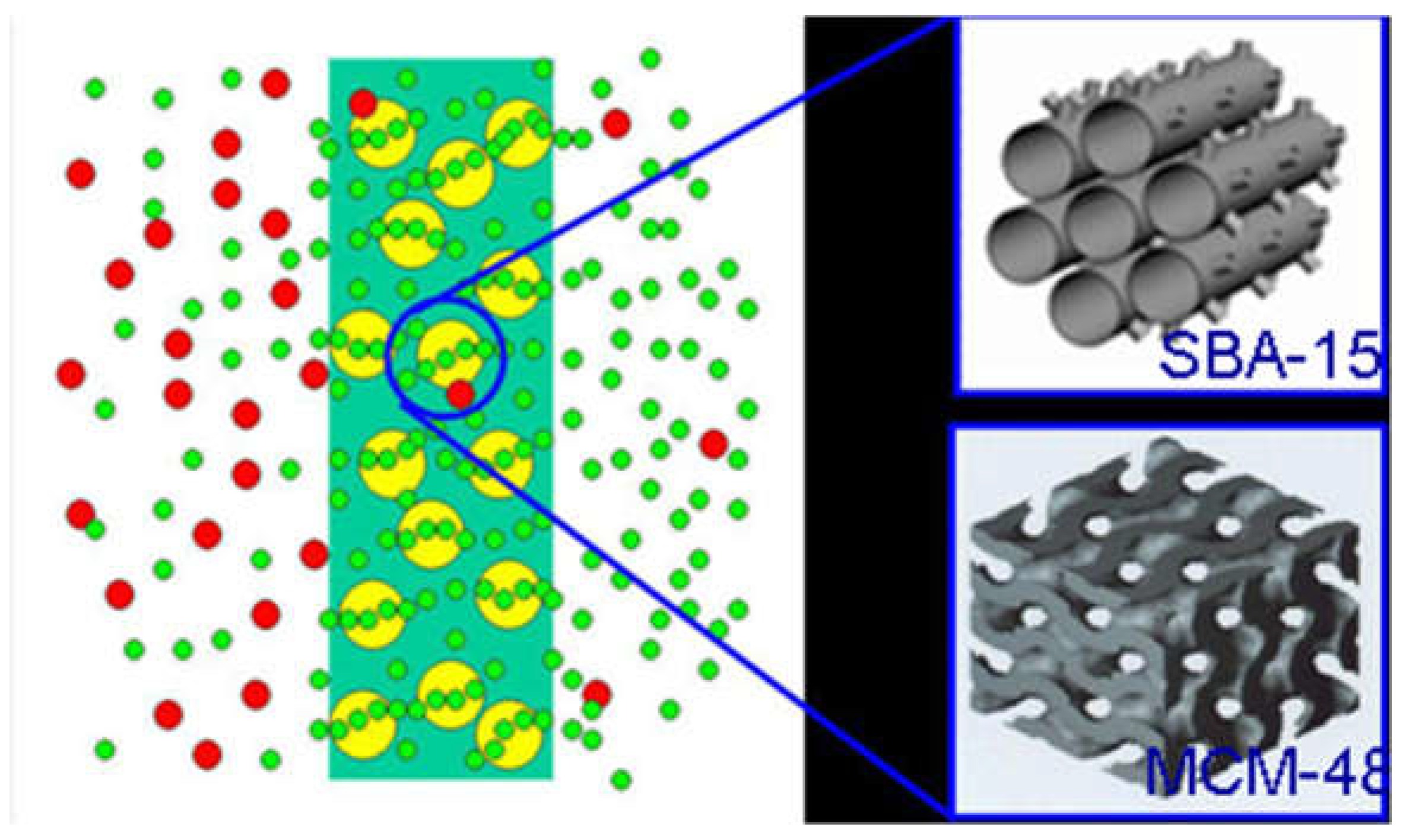
4. Ordered Mesoporous Silica/C Hybrid CMS Membranes and CNTs/C Hybrid CMS Membranes
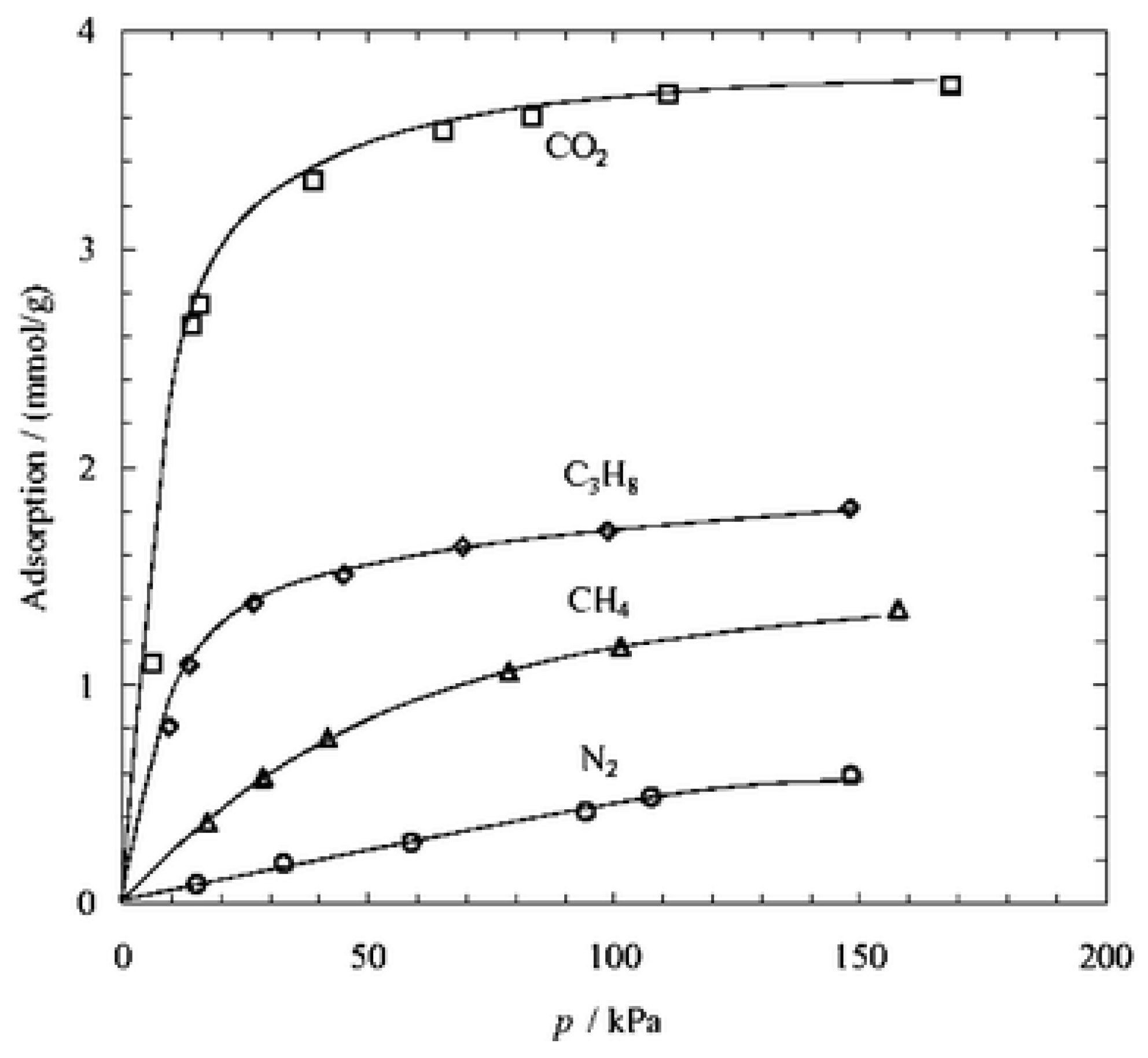
4.1. Ordered Mesoporous Silica/C Hybrid CMS Membranes
4.2. CNTs/C Hybrid CMS Membranes
4.3. Comparison and Analysis of the Gas Separation Property of Hybrid CMS Membranes
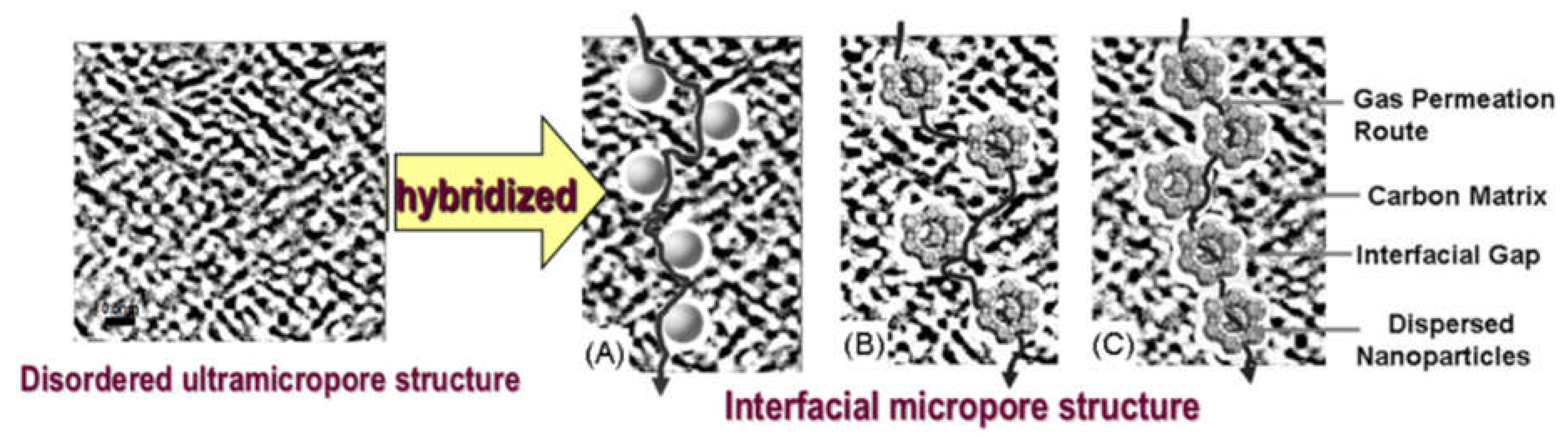
5. The Function of Inorganic Nanoparticles to Enhance the Gas Permeability of Hybrid CMS Membrane
6. Latest Developments in Hybrid CMS Membranes
6.1. MOF/C Hybrid CMS Membrane
6.2. Fe/C hybrid CMS Membrane
6.3. Boehmite/C Hybrid CMS Membrane
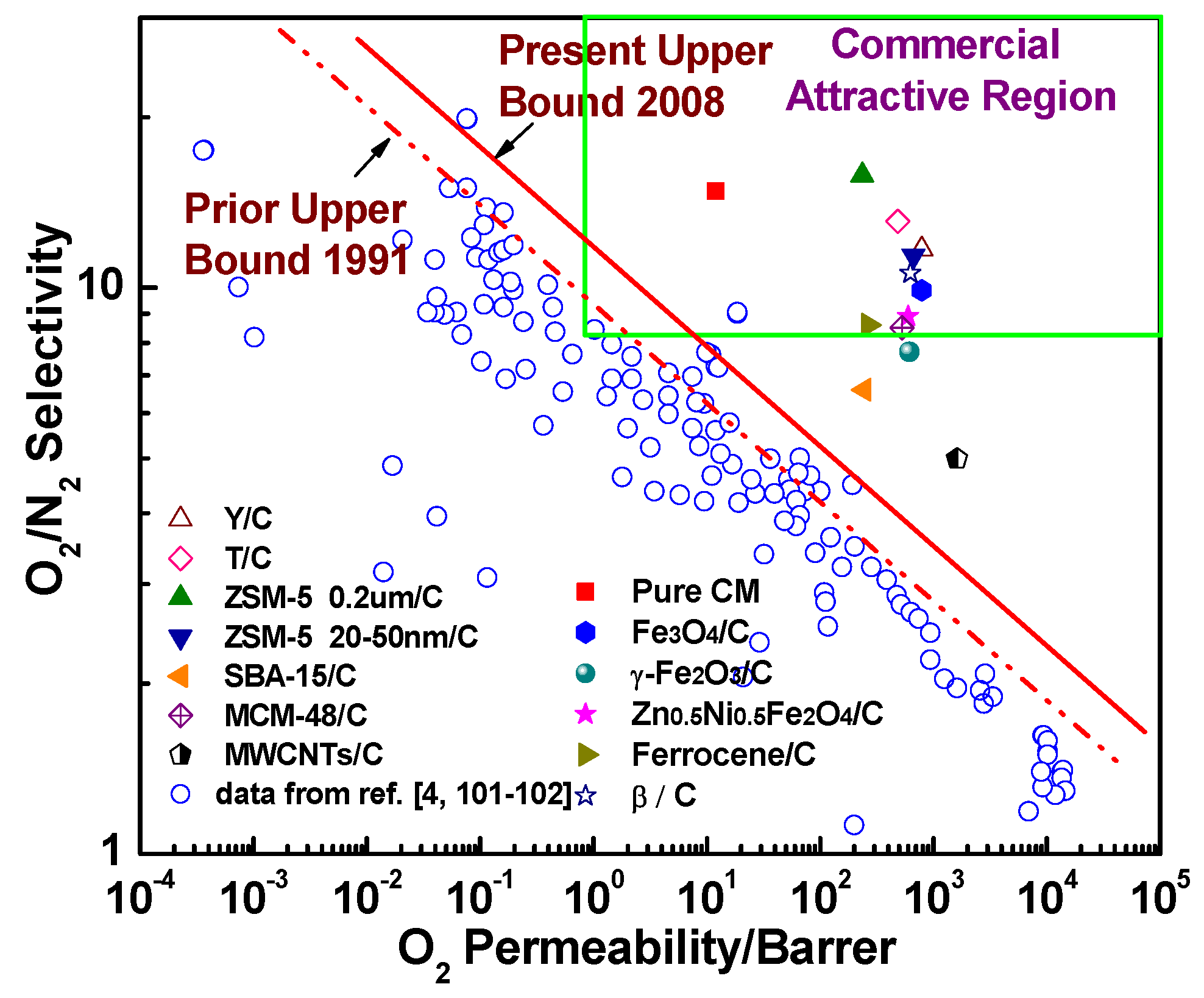
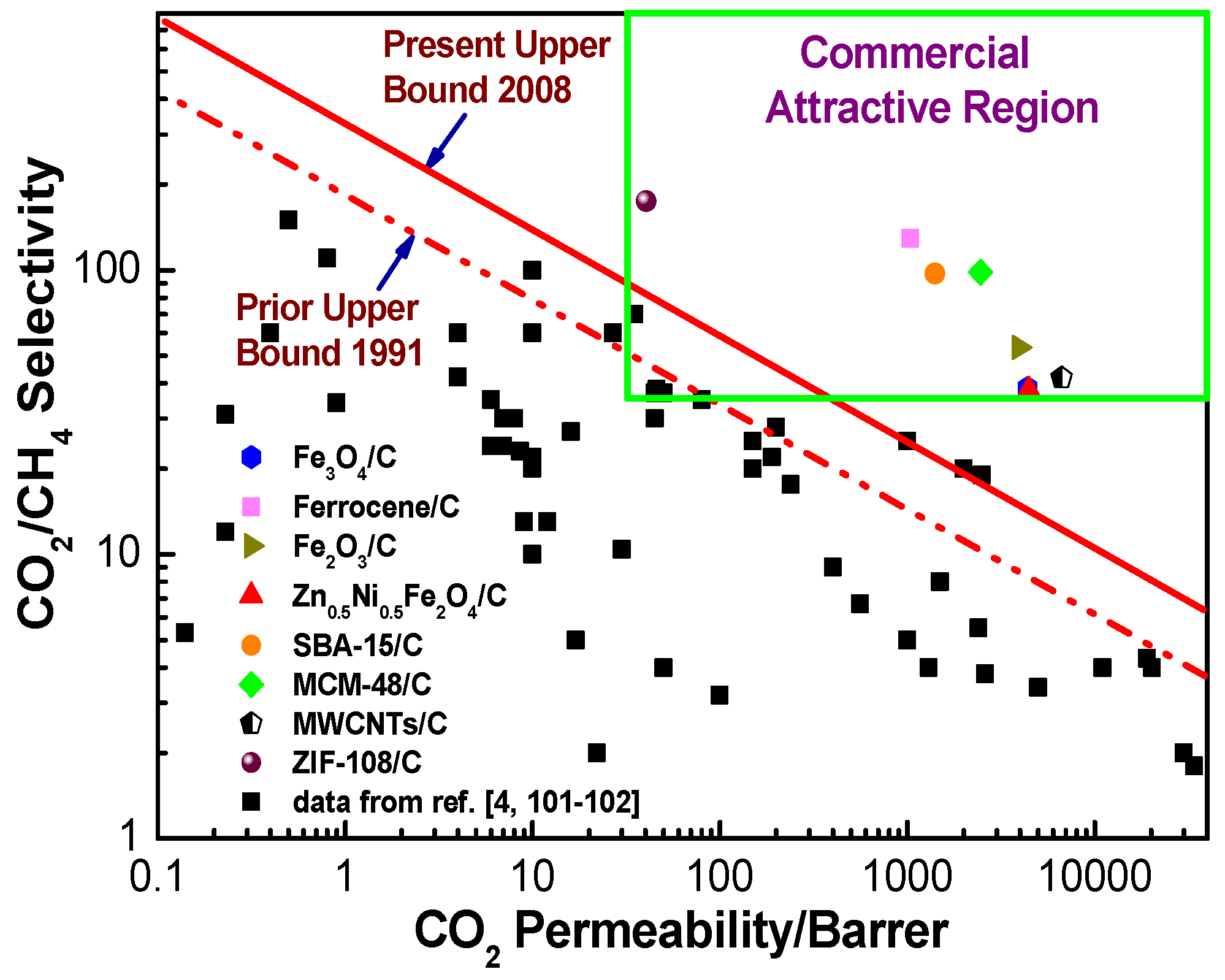
7. The Evaluation of the Gas Separation Performance in the Hybrid CMS Membranes
8. Conclusions and Prospects
- In recent years, some novel precursors, especially the novel polyimides with high fractional free volume and molecular chain rigidity [93,95,104] and PIMs [32,33], were synthesized to prepare CMS membranes with rather high gas permeability. It is necessary to improve the gas permeability further by doping the inorganic particles into these novel precursors to prepare the hybrid CMS membranes. More significantly, the preparation process should be investigated to prevent excessive reduction of selectivity.
- The hybrid CMS membranes related in this review are all self-standing flat membranes in order to study the intrinsic properties of the hybrid CMS membranes. However, coating the hybrid CMS membrane as a separation layer onto supports to fabricate supported CMS membranes is more valuable in practical applications. With a separation layer with a thickness of several micrometers, or even lower than 1 micrometer, the high gas permeability in free-standing hybrid carbon membranes would be transformed into the high gas permeation rate of supported CMS membranes. In particular, to ensure the high selectivity presented by the ultra-thin hybrid CMS membrane separation layer, not only should the proper coating methods be adopted to prevent cracks and pinholes, more importantly, doping particles with as small a size as possible and homogenous dispersion are necessary to prevent the formation of connected gas penetrating channels through the separation layer.
- The hybrid CMS membranes in this review with more outstanding gas permeation properties can be used in all the applied fields of pure CMS membranes. Recently, in addition to conventional gas separation, pure CMS membranes have presented excellent performance in the field of membrane reactors and attracted much attention of researchers. Itoh et al. [10] applied CMS membranes to the dehydrogenation of cyclohexane to remove the produced H2 and improved the conversion rate from 30% to 70%. Zhang et al. [11] used the CMS membrane reactor to the methanol steam reforming reaction to produce H2 with the methanol conversion rate as high as 99.9% and H2 selectivity of 97% at 250 °C. Briceño et al. [105,106] and Zhang et al. [107] also researched the methanol steam reforming reaction with CMS membrane reactor and achieved higher conversions with the CMS membrane reactor than the traditional reactors. Abdollahi et al. [12] took coal-based syngas containing H2S as a raw material and achieved a synchronous separation reaction to obtain H2 in the CMS membrane reactor. Hirota et al. [9] prepared gas-activated CMS membranes with outstanding H2 permeation and selectivity of H2/meth cyclohexane, which presented the potential in H2 storage. Hybrid CMS membranes have better application prospects than pure CMS membranes in the field of membrane reactors because the doping nanoparticles have catalytic activity and can replace some additional catalysts, in addition to the benefit of the excellent gas permeation performance of the hybrid CMS membranes.
Author Contributions
Funding
Conflicts of Interest
References
- Salleh, W.N.W.; Ismail, A.F. Carbon membranes for gas separation processes: Recent progress and future perspective. J. Membr. Sci. Res. 2015, 1, 2–15. [Google Scholar]
- Salleh, W.N.W.; Ismail, A.F.; Matsuura, T.; Abdullah, M.S. Precursor selection and process conditions in the preparation of carbon membrane for gas separation: A review. Sep. Purif. Rev. 2011, 40, 261–311. [Google Scholar] [CrossRef]
- Baker, R.W. Future directions of membrane gas separation technology. Ind. Eng. Chem. Res. 2002, 41, 1393–1411. [Google Scholar] [CrossRef]
- Hägg, M.B.; He, X.Z. Membrane Engineering for the Treatment of Gases; Royal Society of Chemistry: Cambridge, UK, 2011; Volume 2, pp. 162–191. [Google Scholar]
- Ismail, F.A.; Rana, D.; Matsuura, T.; Foley, H. Carbon-Based Membranes for Separation Processes; Springer: New York, NY, USA, 2011. [Google Scholar]
- Ismail, A.F.; David, L.I.B. A review on the latest development of carbon membranes for gas separation. J. Membr. Sci. 2001, 193, 1–18. [Google Scholar] [CrossRef]
- Ockwig, N.W.; Nenoff, T.M. Membranes for hydrogen separation. Chem. Rev. 2007, 107, 4078–4110. [Google Scholar] [CrossRef] [PubMed]
- Llosa Tanco, M.A.; Pacheco Tanaka, D.A. Recent Advances on Carbon Molecular Sieve Membranes (CMSMs) and Reactors. Processes 2016, 4, 29. [Google Scholar] [CrossRef]
- Hirota, Y.; Ishikado, A.; Uchida, Y.; Egashira, Y.; Nishiyama, N. Pore size control of microporous carbon membranes by post-synthesis activation and their use in a membrane reactor for dehydrogenation of methylcyclohexane. J. Membr. Sci. 2013, 440, 134–139. [Google Scholar] [CrossRef]
- Itoh, N.; Haraya, K. A carbon membrane reactor. Catal. Today 2000, 56, 103–111. [Google Scholar] [CrossRef]
- Zhang, X.; Hu, H.; Zhu, Y.; Zhu, S. Methanol steam reforming to hydrogen in a carbon membrane reactor system. Ind. Eng. Chem. Res. 2006, 45, 7997–8001. [Google Scholar] [CrossRef]
- Abdollahi, M.; Yu, J.; Liu, P.K.; Ciora, R.; Sahimi, M.; Tsotsis, T.T. Hydrogen production from coal-derived syngas using a catalytic membrane reactor based process. J. Membr. Sci. 2010, 363, 160–169. [Google Scholar] [CrossRef]
- Hamm, J.B.S.; Ambrosi, A.; Griebeler, J.G.; Marcilio, N.R.; Tessaro, I.C.; Pollo, L.D. Recent advances in the development of supported carbon membranes for gas separation. Int. J. Hydrog. Energy 2017, 42, 24830–24845. [Google Scholar] [CrossRef]
- Tin, P.S.; Xiao, Y.; Chung, T.S. Polyimide-carbonized membranes for gas separation: Structural, composition, and morphological control of precursors. Sep. Purif. Rev. 2006, 35, 285–318. [Google Scholar] [CrossRef]
- Suda, H.; Haraya, K. Gas permeation through micropores of carbon molecular sieve membranes derived from Kapton polyimide. J. Phys. Chem. B 1997, 101, 3988–3994. [Google Scholar] [CrossRef]
- Hatori, H.; Yamada, Y.; Shiraishi, M.; Nakata, H.; Yoshitomi, S. Carbon molecular sieve films from polyimide. Carbon 1992, 30, 719–720. [Google Scholar] [CrossRef]
- Kim, Y.K.; Lee, J.M.; Park, H.B.; Lee, Y.M. The gas separation properties of carbon molecular sieve membranes derived from polyimides having carboxylic acid groups. J. Membr. Sci. 2004, 235, 139–146. [Google Scholar] [CrossRef]
- Hatori, H.; Yamada, Y.; Shiraishi, M. Preparation of macroporous carbon films from polyimide by phase inversion method. Carbon 1992, 30, 303–304. [Google Scholar] [CrossRef]
- Hosseini, S.S.; Peng, N.; Chung, T.S. Gas separation membranes developed through integration of polymer blending and dual-layer hollow fiber spinning process for hydrogen and natural gas enrichments. J. Membr. Sci. 2010, 349, 156–166. [Google Scholar] [CrossRef]
- Shiflett, M.B.; Foley, H.C. Ultrasonic deposition of high-selectivity nanoporous carbon membranes. Science 1999, 285, 1902–1905. [Google Scholar] [CrossRef] [PubMed]
- Steel, K.M.; Koros, W.J. Investigation of porosity of carbon materials and related effects on gas separation properties. Carbon 2003, 41, 253–266. [Google Scholar] [CrossRef]
- Song, C.; Wang, T.; Pan, Y.; Qiu, J. Preparation of coal-based microfiltration carbon membrane and application in oily wastewater treatment. Sep. Purify. Technol. 2006, 51, 80–84. [Google Scholar] [CrossRef]
- Zhou, W.; Yoshino, M.; Kita, H.; Okamoto, K.I. Carbon molecular sieve membranes derived from phenolic resin with a pendant sulfonic acid group. Ind. Eng. Chem. Res. 2001, 40, 4801–4807. [Google Scholar] [CrossRef]
- Fuertes, A.B. Effect of air oxidation on gas separation properties of adsorption-selective carbon membranes. Carbon 2001, 39, 697–706. [Google Scholar] [CrossRef]
- Zhou, W.; Yoshino, M.; Kita, H.; Okamoto, K.I. Preparation and gas permeation properties of carbon molecular sieve membranes based on s ulfonated phenolic resin. J. Membr. Sci. 2003, 217, 55–67. [Google Scholar] [CrossRef]
- Zhang, B.; Wang, T.; Zhang, S.; Qiu, J.; Jian, X. Preparation and characterization of carbon membranes made from poly (phthalazinone ether sulfone ketone). Carbon 2006, 44, 2764–2769. [Google Scholar] [CrossRef]
- Zhang, B.; Wang, T.; Wu, Y.; Qiu, J.; Jian, X. Preparation and gas permeation of composite carbon membranes from poly(phthalazinone ether sulfone ketone). Sep. Purif. Technol. 2008, 60, 259–263. [Google Scholar] [CrossRef]
- David, L.I.B.; Ismail, A.F. Influence of the thermastabilization process and soak time during pyrolysis process on the polyacrylonitrile carbon membranes for O2/N2 separation. J. Membr. Sci. 2003, 213, 285–291. [Google Scholar] [CrossRef]
- Liang, C.; Sha, G.; Guo, S. Carbon membrane for gas separation derived from coal tar pitch. Carbon 1999, 37, 1391–1397. [Google Scholar] [CrossRef]
- Koresh, J.E.; Sofer, A. Molecular sieve carbon permselective membrane. Part I. Presentation of a new device for gas mixture separation. Sep. Sci. Technol. 1983, 18, 723–734. [Google Scholar] [CrossRef]
- Gilron, J.; Soffer, A. Knudsen diffusion in microporous carbon membranes with molecular sieving character. J. Membr. Sci. 2002, 209, 339–352. [Google Scholar] [CrossRef]
- Liao, K.S.; Japip, S.; Lai, J.Y.; Chung, T.S. Boron-embedded hydrolyzed PIM-1 carbon membranes for synergistic ethylene/ethane purification. J. Membr. Sci. 2017, 534, 92–99. [Google Scholar] [CrossRef]
- Liu, J.; Xiao, Y.; Chung, T.S. Flexible thermally treated 3D PIM-CD molecular sieve membranes exceeding the upper bound line for propylene/propane separation. J. Mater. Chem. A 2017, 5, 4583–4595. [Google Scholar] [CrossRef]
- Hosseini, S.S.; Omidkhah, M.R.; Moghaddam, A.Z.; Pirouzfar, V.; Krantz, W.B.; Tan, N.R. Enhancing the properties and gas separation performance of PBI–polyimides blend carbon molecular sieve membranes via optimization of the pyrolysis process. Sep. Purif. Technol. 2014, 122, 278–289. [Google Scholar] [CrossRef]
- Lee, H.J.; Suda, H.; Haraya, K.; Moon, S.H. Gas permeation properties of carbon molecular sieving membranes derived from the polymer blend of polyphenylene oxide (PPO)/polyvinylpyrrolidone (PVP). J. Membr. Sci. 2007, 296, 139–146. [Google Scholar] [CrossRef]
- Linkov, V.M.; Sanderson, R.D.; Jacobs, E.P. Carbon membranes from precursors containing low-carbon residual polymers. Polym. Int. 1994, 35, 239–242. [Google Scholar] [CrossRef]
- Saufi, S.M.; Ismail, A.F. Fabrication of carbon membranes for gas separation—A review. Carbon 2004, 42, 241–259. [Google Scholar] [CrossRef]
- Li, L.; Wang, T.; Liu, Q.; Cao, Y.; Qiu, J. A high CO2 permselective mesoporous silica/carbon composite membrane for CO2 separation. Carbon 2012, 50, 5186–5195. [Google Scholar] [CrossRef]
- Zhang, B.; Wang, T.; Liu, S.; Zhang, S.; Qiu, J.; Chen, Z.; Cheng, H. Structure and morphology of microporous carbon membrane materials derived from poly (phthalazinone ether sulfone ketone). Microporous Mesoporous Mater. 2006, 96, 79–83. [Google Scholar] [CrossRef]
- Yoda, S.; Hasegawa, A.; Suda, H.; Uchimaru, Y.; Haraya, K.; Tsuji, T.; Otake, K. Preparation of a platinum and palladium/polyimide nanocomposite film as a precursor of metal-doped carbon molecular sieve membrane via supercritical impregnation. Chem. Mater. 2004, 16, 2363–2368. [Google Scholar] [CrossRef]
- Zhang, L.; Chen, X.; Zeng, C.; Xu, N. Preparation and gas separation of nano-sized nickel particle-filled carbon membranes. J. Membr. Sci. 2006, 281, 429–434. [Google Scholar] [CrossRef]
- Barsema, J.N.; Balster, J.; Jordan, V.; Van der Vegt, N.F.A.; Wessling, M. Functionalized carbon molecular sieve membranes containing Ag-nanoclusters. J. Membr. Sci. 2003, 219, 47–57. [Google Scholar] [CrossRef]
- Barsema, J.N.; van der Vegt, N.F.A.; Koops, G.H.; Wessling, M. Ag-functionalized carbon molecular-sieve membranes based on polyelectrolyte/polyimide blend precursors. Adv. Funct. Mater. 2005, 15, 69–75. [Google Scholar] [CrossRef]
- Teixeira, M.; Campo, M.; Tanaka, D.A.; Tanco, M.A.; Magen, C.; Mendes, A. Carbon–Al2O3–Ag composite molecular sieve membranes for gas separation. Chem. Eng. Res. Design 2012, 90, 2338–2345. [Google Scholar] [CrossRef]
- Lie, J.A.; Hägg, M.B. Carbon membranes from cellulose and metal loaded cellulose. Carbon 2005, 43, 2600–2607. [Google Scholar] [CrossRef]
- Tanco, M.A.L.; Tanaka, D.A.P.; Rodrigues, S.C.; Texeira, M.; Mendes, A. Composite-alumina-carbon molecular sieve membranes prepared from novolac resin and boehmite. Part I: Preparation, characterization and gas permeation studies. Int. J. Hydrog. Energy 2015, 40, 5653–5663. [Google Scholar] [CrossRef]
- Tanco, M.A.L.; Tanaka, D.A.P.; Mendes, A. Composite-alumina-carbon molecular sieve membranes prepared from novolac resin and boehmite. Part II: Effect of the carbonization temperature on the gas permeation properties. Int. J. Hydrog. Energy 2015, 40, 3485–3496. [Google Scholar] [CrossRef]
- Park, H.B.; Suh, I.Y.; Lee, Y.M. Novel pyrolytic carbon membranes containing silica: Preparation and characterization. Chem. Mater. 2002, 14, 3034–3046. [Google Scholar] [CrossRef]
- Park, H.B.; Lee, Y.M. Pyrolytic carbon–silica membrane: A promising membrane material for improved gas separation. J. Membr. Sci. 2003, 213, 263–272. [Google Scholar] [CrossRef]
- Park, H.B.; Jung, C.H.; Kim, Y.K.; Nam, S.Y.; Lee, S.Y.; Lee, Y.M. Pyrolytic carbon membranes containing silica derived from poly (imide siloxane): The effect of siloxane chain length on gas transport behavior and a study on the separation of mixed gases. J. Membr. Sci. 2004, 235, 87–98. [Google Scholar] [CrossRef]
- Lua, A.C.; Shen, Y. Preparation and characterization of polyimide–silica composite membranes and their derived carbon–silica composite membranes for gas separation. Chem. Eng. J. 2013, 220, 441–451. [Google Scholar] [CrossRef]
- Tin, P.S.; Chung, T.S.; Jiang, L.Y.; Kulprathipanja, S. Carbon–zeolite composite membranes for gas separation. Carbon 2005, 43, 2025–2027. [Google Scholar] [CrossRef]
- Jiang, L.Y.; Chung, T.S.; Rajagopalan, R. Dual-layer hollow carbon fiber membranes for gas separation consisting of carbon and mixed matrix layers. Carbon 2007, 45, 166–172. [Google Scholar] [CrossRef]
- Yin, X.; Wang, J.; Chu, N.; Yang, J.; Lu, J.; Zhang, Y.; Yin, D. Zeolite L/carbon nanocomposite membranes on the porous alumina tubes and their gas separation properties. J. Membr. Sci. 2010, 348, 181–189. [Google Scholar] [CrossRef]
- Liu, Q.; Wang, T.; Qiu, J.; Cao, Y. A novel carbon/ZSM-5 nanocomposite membrane with high performance for oxygen/nitrogen separation. Chem. Commun. 2006, 11, 1230–1232. [Google Scholar] [CrossRef] [PubMed]
- Liu, Q.; Wang, T.; Liang, C.; Zhang, B.; Liu, S.; Cao, Y.; Qiu, J. Zeolite married to carbon: A new family of membrane materials with excellent gas separation performance. Chem. Mater. 2006, 18, 6283–6288. [Google Scholar] [CrossRef]
- Liu, Q.; Wang, T.; Guo, H.; Liang, C.; Liu, S.; Zhang, Z.; Cao, Y.; Su, D.; Qiu, J. Controlled synthesis of high performance carbon/zeolite T composite membrane materials for gas separation. Microporous Mesoporous Mater. 2009, 120, 460–466. [Google Scholar] [CrossRef]
- Jiao, W.; Ban, Y.; Shi, Z.; Jiang, X.; Li, Y.; Yang, W. High performance carbon molecular sieving membranes derived from pyrolysis of metal–organic framework ZIF-108 doped polyimide matrices. Chem. Commun. 2016, 52, 13779–13782. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Wang, C.; Wang, N.; Cao, Y.; Wang, T. The preparation and gas separation properties of zeolite/carbon hybrid membranes. J. Mater. Sci. 2015, 50, 2561–2570. [Google Scholar] [CrossRef]
- Kim, Y.K.; Park, H.B.; Lee, Y.M. Carbon molecular sieve membranes derived from metal-substituted sulfonated polyimide and their gas separation properties. J. Membr. Sci. 2003, 226, 145–158. [Google Scholar] [CrossRef]
- Jang, E.S.; Chang, J.J.; Gwak, J.; Ayral, A.; Rouessac, V.; Cot, L.; Hwang, S.J.; Choy, J.H. Asymmetric high-T c superconducting gas separation membrane. Chem. Mater. 2007, 19, 3840–3844. [Google Scholar] [CrossRef]
- Gwak, J.; Ayral, A.; Rouessac, V.; Cot, L.; Grenier, J.C.; Choy, J.H. Synthesis and characterization of porous ferrimagnetic membranes. Microporous Mesoporous Mater. 2003, 63, 177–184. [Google Scholar] [CrossRef]
- Zhao, X.Y.; Wang, T.H.; Li, L.; Liu, Y.; Cao, Y. Fabrication of Fe/C hybrid carbon membranes for gas separation. CIEST J. 2009, 60, 2232–2236. [Google Scholar]
- Zhao, X.Y.; Wang, T.H.; Li, L.; Liu, Y.; Cao, Y. Polymide/Fe3O4-carbonized Membranes for Gas Separation. J. Inorg. Mater. 2010, 25, 47–52. [Google Scholar] [CrossRef]
- Zhao, X.Y.; Wang, T.H.; Li, L.; Liu, Y.; Cao, Y. Functional carbon membranes fabricated by doping magnetic nanoparticles for gas separation. Modern Chem. Ind. 2009, 29, 257–260. [Google Scholar]
- Bein, T. Synthesis and applications of molecular sieve layers and membranes. Chem. Mater. 1996, 8, 1636–1653. [Google Scholar] [CrossRef]
- Tosheva, L.; Valtchev, V.P. Nanozeolites: Synthesis, crystallization mechanism, and applications. Chem. Mater. 2005, 17, 2494–2513. [Google Scholar] [CrossRef]
- Bu, X.; Feng, P.; Stucky, G.D. Large-cage zeolite structures with multidimensional 12-ring channels. Science 1997, 278, 2080–2085. [Google Scholar] [CrossRef] [PubMed]
- De la Iglesia, Ó.; Mallada, R.; Menéndez, M.; Coronas, J. Continuous zeolite membrane reactor for esterification of ethanol and acetic acid. Chem. Eng. J. 2007, 131, 35–39. [Google Scholar] [CrossRef]
- Weitkamp, J. Zeolites and catalysis. Solid State Ionics 2000, 131, 175–188. [Google Scholar] [CrossRef]
- Duval, J.M.; Folkerts, B.; Mulder, M.H.V. Adsorbent filled membranes for gas separation. Part 1. Improvement of the gas separation properties of polymeric membranes by incorporation of microporous adsorbents. J. Membr. Sci. 1993, 80, 189–198. [Google Scholar] [CrossRef]
- Cui, Y.; Kita, H.; Okamoto, K. Preparation and gas separation performance of zeolite T membrane. J. Mater. Chem. 2004, 14, 924–932. [Google Scholar] [CrossRef]
- Cui, Y.; Kita, H.; Okamoto, K. Preparation and gas separation properties of zeolite T membrane. Chem. Commun. 2003, 17, 2154–2455. [Google Scholar] [CrossRef]
- Kresge, C.T.; Leonowicz, M.E.; Roth, W.J.; Vartuli, J.C.; Beck, J.S. Ordered mesoporous molecular sieves synthesized by a liquid-crystal template mechanism. Nature 1992, 359, 710. [Google Scholar] [CrossRef]
- Beck, J.S.; Vartuli, J.C.; Roth, W.J.; Leonowicz, M.E.; Kresge, C.T.; Schmitt, K.D.; Chu, T.W.; Olson, D.H.; Sheppard, E.W.; Mecullen, S.B.; et al. A new family of mesoporous molecular sieves prepared with liquid crystal templates. J. Am. Chem. Soc. 1992, 114, 10834–10843. [Google Scholar] [CrossRef]
- Tanev, P.T.; Pinnavaia, T.J. A neutral templating route to mesoporous molecular sieves. science 1995, 267, 865–867. [Google Scholar] [CrossRef] [PubMed]
- Zhao, D.; Feng, J.; Huo, Q.; Melosh, N.; Fredrickson, G.H.; Chmelka, B.F.; Stucky, G.D. Triblock copolymer syntheses of mesoporous silica with periodic 50 to 300 angstrom pores. Science 1998, 279, 548–552. [Google Scholar] [CrossRef] [PubMed]
- Lin, H.P.; Mou, C.Y. Structural and morphological control of cationic surfactant-templated mesoporous silica. Acc. Chem. Res. 2002, 35, 927–935. [Google Scholar] [CrossRef] [PubMed]
- Schacht, S.; Huo, Q.; Voigt-Martin, I.G.; Stucky, G.D.; Schüth, F. Oil-water interface templating of mesoporous macroscale structures. Science 1996, 273, 768–771. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.; Kuperman, A.; Coombs, N.; Mamiche-Afara, S.; Ozin, G.A. Synthesis of oriented films of mesoporous silica on mica. Nature 1996, 379, 703. [Google Scholar] [CrossRef]
- Lu, Y.; Ganguli, R.; Drewien, C.A.; Anderson, M.T.; Brinker, C.J.; Gong, W.; Zink, J.I. Continuous formation of supported cubic and hexagonal mesoporous films by sol–gel dip-coating. Nature 1997, 389, 364. [Google Scholar] [CrossRef]
- Zhang, Z.; Han, Y.; Zhu, L.; Wang, R.; Yu, Y.; Qiu, S.; Zhao, D.; Xiao, F.S. Strongly acidic and high-temperature hydrothermally stable mesoporous aluminosilicates with ordered hexagonal structure. Angew. Chem. Int. Ed. 2001, 40, 1258–1262. [Google Scholar] [CrossRef]
- Han, Y.; Li, D.; Zhao, L.; Song, J.; Yang, X.; Li, N.; Di, Y.; Li, C.; Wu, S.; Xu, X.; et al. High-Temperature Generalized Synthesis of Stable Ordered Mesoporous Silica-Based Materials by Using Fluorocarbon–Hydrocarbon Surfactant Mixtures. Angew. Chem. 2003, 115, 3761–3765. [Google Scholar] [CrossRef]
- Hicks, J.C.; Jones, C.W. Controlling the density of amine sites on silica surfaces using benzyl spacers. Langmuir 2006, 22, 2676–2681. [Google Scholar] [CrossRef] [PubMed]
- Zhao, D.; Ren, J.; Li, H.; Li, X.; Deng, M. Gas separation properties of poly(amide-6-b-ethylene oxide)/amino modified multi-walled carbon nanotubes mixed matrix membranes. J. Membr. Sci. 2014, 467, 41–47. [Google Scholar] [CrossRef]
- Habibiannejad, S.A.; Aroujalian, A.; Raisi, A. Pebax-1657 mixed matrix membrane containing surface modified multi-walled carbon nanotubes for gas separation. RSC Adv. 2016, 6, 79563–79577. [Google Scholar] [CrossRef]
- Tseng, H.; Kumar, I.A.; Weng, T.; Lu, C.; Wey, M. Preparation and characterization of carbon molecular sieve membranes for gas separation-the effect of incorporated multi-wall carbon nanotubes. Desalination 2009, 240, 40–45. [Google Scholar] [CrossRef]
- Rao, P.S.; Wey, M.; Tseng, H.; Kumar, I.A.; Weng, T. A comparison of carbon/nanotube molecular sieve membranes with polymer blend carbon molecular sieve membranes for the gas permeation application. Microporous Mesoporous Mater. 2008, 113, 499–510. [Google Scholar] [CrossRef]
- Li, L.; Song, C.; Jiang, D.; Wang, T. Preparation and enhanced gas separation performance of Carbon/Carbon nanotubes (C/CNTs) hybrid membranes. Sep. Purif. Technol. 2017, 188, 73–80. [Google Scholar] [CrossRef]
- Japip, S.; Liao, K.S.; Xiao, Y.; Chung, T.S. Enhancement of molecular-sieving properties by constructing surface nano-metric layer via vapor cross-linking. J. Membr. Sci. 2016, 497, 248–258. [Google Scholar] [CrossRef]
- Japip, S.; Liao, K.S.; Chung, T.S. Molecularly tuned free volume of vapor cross-linked 6FDA-Durene/ZIF-71 MMMs for H2/CO2 separation at 150 °C. Adv. Mater. 2017, 29, 1603833. [Google Scholar] [CrossRef] [PubMed]
- Yang, T.; Xiao, Y.; Chung, T.S. Poly-/metal-benzimidazole nano-composite membranes for hydrogen purification. Energy Environ. Sci. 2011, 4, 4171–4180. [Google Scholar] [CrossRef]
- Koros, W.J.; Zhang, C. Materials for next-generation molecularly selective synthetic membranes. Nat. Mater. 2017, 16, 289. [Google Scholar] [CrossRef] [PubMed]
- Kamath, M.G.; Fu, S.; Itta, A.K.; Qiu, W.; Liu, G.; Swaidan, R.; Koros, W.J. 6FDA-DETDA: DABE polyimide-derived carbon molecular sieve hollow fiber membranes: Circumventing unusual aging phenomena. J. Membr. Sci. 2018, 546, 197–205. [Google Scholar] [CrossRef]
- Bhuwania, N. Engineering the Morphology of Carbon Molecular Sieve (CMS) Hollow Fiber Membranes. Ph.D. Thesis, Georgia Institute of Technology, Atlanta, GA, USA, 2013. Available online: http://hdl.handle.net/1853/53397 (accessed on 20 November 2018).
- Salinas, O.; Ma, X.; Wang, Y.; Han, Y.; Pinnau, I. Carbon molecular sieve membrane from a microporous spirobisindane-based polyimide precursor with enhanced ethylene/ethane mixed-gas selectivity. RSC Adv. 2017, 7, 3265–3272. [Google Scholar] [CrossRef]
- Salinas, O.; Ma, X.; Litwiller, E.; Pinnau, I. High-performance carbon molecular sieve membranes for ethylene/ethane separation derived from an intrinsically microporous polyimide. J. Memb. Sci. 2016, 500, 115–123. [Google Scholar] [CrossRef]
- Zhong, Z.X.; Yao, J.F.; Low, Z.X.; Chen, R.Z.; He, M.; Wang, H.T. Carbon composite membrane derived from a two-dimensional zeolitic imidazolate framework and its gas separation properties. Carbon 2014, 72, 242–249. [Google Scholar] [CrossRef]
- Kumakiri, I.; Tamura, K.; Sasaki, Y.; Tanaka, K.; Kita, H. Influence of Iron Additive on the Hydrogen Separation Properties of Carbon Molecular Sieve Membranes. Ind. Eng. Chem. Res. 2018, 57, 5370–5377. [Google Scholar] [CrossRef]
- Chu, Y.H.; Yancey, D.; Xu, L.; Martinez, M.; Brayden, M.; Koros, W.J. Iron-containing carbon molecular sieve membranes for advanced olefin/paraffin separations. J. Membr. Sci. 2018, 548, 609–620. [Google Scholar] [CrossRef]
- Robeson, L.M. Correlation of separation factor versus permeability for polymeric membranes. J. Membr. Sci. 1991, 62, 165–185. [Google Scholar] [CrossRef]
- Robeson, L.M. The upper bound revisited. J. Membr. Sci. 2008, 320, 390–400. [Google Scholar] [CrossRef]
- Hillock, A.M.W.; Miller, S.J.; Koros, W.J. Crosslinked mixed matrix membranes for the purification of natural gas: Effects of sieve surface modification. J. Membr. Sci. 2008, 314, 193–199. [Google Scholar] [CrossRef]
- Fu, S.; Sanders, E.S.; Kulkarni, S.S.; Koros, W.J. Carbon molecular sieve membrane structure–property relationships for four novel 6FDA based polyimide precursors. J. Membr. Sci. 2015, 487, 60–73. [Google Scholar] [CrossRef]
- Briceño, K.; Iulianelli, A.; Montané, D.; Garcia-Valls, R.; Basile, A. Carbon molecular sieve membranes supported on non-modified ceramic tubes for hydrogen separation in membrane reactors. Int. J. Hydrog. Energy 2012, 37, 13536–13544. [Google Scholar] [CrossRef]
- Briceño, K.; Montané, D.; Garcia-Valls, R.; Iulianelli, A.; Basile, A. Fabrication variables affecting the structure and properties of supported carbon molecular sieve membranes for hydrogen separation. J. Membr. Sci. 2012, 415, 288–297. [Google Scholar] [CrossRef]
- Zhang, B.; Zhao, D.; Wu, Y.; Liu, H.; Wang, T.; Qiu, J. Fabrication and application of catalytic carbon membranes for hydrogen production from methanol steam reforming. Ind. Eng. Chem. Res. 2015, 54, 623–632. [Google Scholar] [CrossRef]







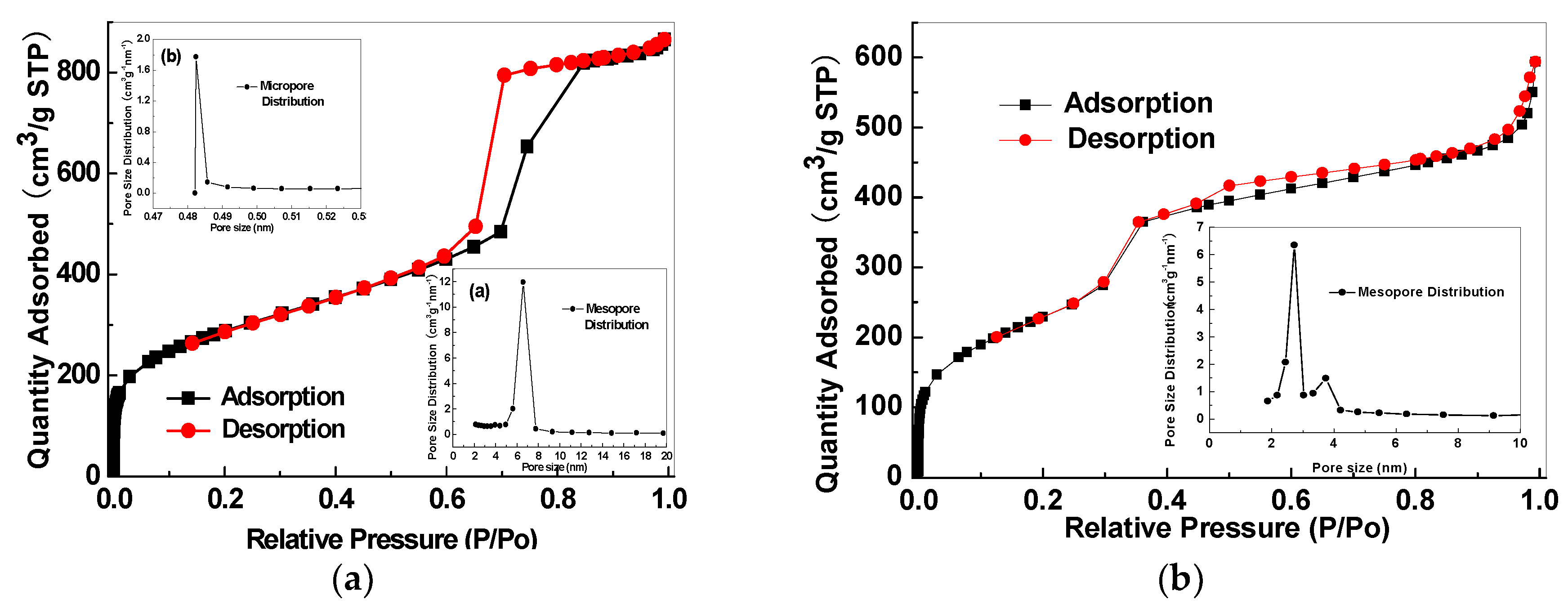
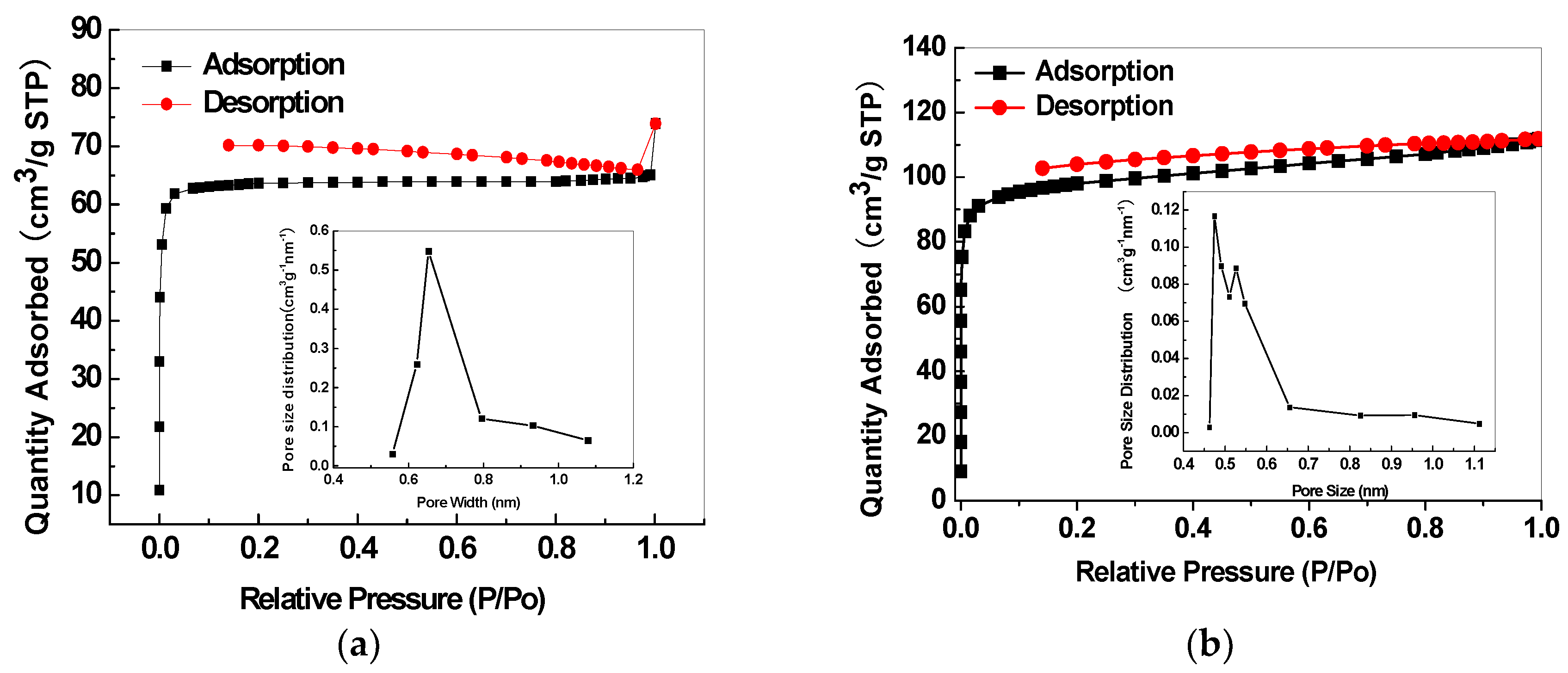

| Sample | Permeability/Barrer a | Ideal Selectivity | |||||||
|---|---|---|---|---|---|---|---|---|---|
| H2 | CO2 | O2 | N2 | CH4 | O2/N2 | CO2/N2 | CO2/CH4 | H2/N2 | |
| Ag/C [42] | - | 290 | 81.3 | 6.7 | - | 12.1 | 43.3 | -- | - |
| K/C [60] | - | - | 6.8 | 1.7 | - | 4 | - | - | - |
| Na/C [60] | - | - | 5.3 | 1.1 | - | 4.8 | - | - | - |
| Pd based [40] | 34.4 | - | - | 0.0061 | - | - | - | - | 5639.3 |
| CaO/C [45] | 860 | 130 | 38 | 3.1 | 3.5 | 12.3 | 41.9 | 37.1 | 277.4 |
| FeO [45] | 280 | 110 | 30 | 8.3 | 4 | 3.6 | 13.3 | 27.5 | 33.7 |
| AgN/C [45] | 1500 | 180 | 53 | 5.1 | 1.4 | 10.4 | 352.9 | 1285.7 | 294.1 |
| Sample | Permeability/Barrer | Ideal Selectivity | |||||||
|---|---|---|---|---|---|---|---|---|---|
| H2 | CO2 | O2 | N2 | CH4 | O2/N2 | CO2/N2 | CO2/CH4 | H2/N2 | |
| CMSM a [59] | 84.4 | 52.7 | 4 | 0.27 | - | 14.8 | 195 | - | 312.6 |
| Ferrocene 10% | 1789 | 634 | 159 | 16 | 3 | 9.9 | 39.6 | 211.0 | 111.8 |
| Ferrocene 15% | 2806 | 1039 | 266 | 31 | 8 | 8.6 | 33.5 | 129.5 | 90.5 |
| Ferrocene 20% | 6997 | 2275 | 1264 | 1001 | 1413 | 1.3 | 1.8 | 1.6 | 7.0 |
| Sample | Permeability/Barrer | Ideal Selectivity | |||||||
|---|---|---|---|---|---|---|---|---|---|
| H2 | CO2 | O2 | N2 | CH4 | O2/N2 | CO2/N2 | CO2/CH4 | H2/N2 | |
| CMSM [59] | 84.4 | 52.7 | 4 | 0.27 | - | 14.8 | 195 | - | 312.6 |
| Fe3O4 10% [64] | 6790 | 2764 | 786 | 79 | 48 | 9.9 | 35.0 | 57.6 | 85.9 |
| Fe3O4 15% [64] | 12,194 | 3433 | 1175 | 136 | 74 | 8.6 | 25.2 | 46.4 | 89.7 |
| Fe3O4 20% [64] | 15,476 | 4385 | 1565 | 193 | 114 | 8.1 | 22.7 | 38.5 | 80.2 |
| γ-Fe2O3 10% | 5415 | 2376 | 616 | 80 | 36 | 7.7 | 29.7 | 66.0 | 67.7 |
| γ-Fe2O3 15% | 7752 | 2790 | 643 | 86 | 48 | 7.5 | 32.4 | 58.1 | 90.1 |
| γ-Fe2O3 20% | 8035 | 3954 | 1187 | 166 | 74 | 7.2 | 23.8 | 53.4 | 48.4 |
| Zn0.5Ni0.5Fe2O4 10% | 5814 | 2401 | 599 | 67 | 27 | 8.9 | 35.8 | 88.9 | 86.8 |
| Zn0.5Ni0.5Fe2O4 15% | 6162 | 2784 | 690 | 85 | 39 | 8.1 | 32.8 | 71.4 | 72.5 |
| Zn0.5Ni0.5Fe2O4 20% | 8191 | 4466 | 1180 | 181 | 118 | 6.5 | 24.7 | 37.8 | 45.3 |
| Pd based [40] | 34.4 | - | - | 0.0061 | - | - | - | - | 5639.3 |
| CMSM in ref. [40] | 49.4 | - | - | 0.15 | - | - | - | - | 329.3 |
| Ag/PFR based [42] | - | 290 | 81.3 | 6.7 | - | 12.1 | 43.3 | - | - |
| CMSM in ref. [42] | - | 64.1 | 16.8 | 2.1 | - | 8 | 30.5 | - | - |
| Sample | Content (wt%) | Permeability (Barrer) | Ideal Selectivity | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| H2 | CO2 | O2 | N2 | CH4 | H2/N2 | CO2/N2 | O2/N2 | CO2/CH4 | ||
| CMSM | 84.4 | 52.7 | 4 | 0.27 | - | 312.6 | 195.1 | 14.8 | - | |
| β/C | 5 | 543 | 255 | 66 | 5.6 | - | 97 | 45.5 | 11.8 | - |
| 10 | 1721 | 810 | 274 | 21.3 | - | 80.8 | 38 | 12.9 | - | |
| 15 | 2108 | 1129 | 382 | 27.9 | - | 75.6 | 40.5 | 13.7 | - | |
| 20 | 2987 | 1360 | 493 | 42.5 | - | 70.3 | 32 | 11.6 | - | |
| 25 | 3996 | 1644 | 628 | 59.4 | - | 67.3 | 27.7 | 10.6 | - | |
| Y/C | 5 | 561 | 250 | 55 | 5.6 | - | 100.2 | 44.6 | 9.8 | - |
| 10 | 1717 | 761 | 236 | 20.7 | - | 82.9 | 36.8 | 11.4 | - | |
| 15 | 2280 | 1022 | 501 | 32.1 | - | 71 | 31.8 | 15.6 | - | |
| 20 | 3090 | 1431 | 605 | 45.5 | - | 67.9 | 31.5 | 13.3 | - | |
| 25 | 4094 | 1783 | 786 | 67.1 | - | 61 | 26.6 | 11.7 | - | |
| Y/C [52] | - | 266 | - | - | 2.15 | - | - | - | 124 | |
| CMSM [52] | - | 611 | - | - | 10 | - | - | - | 61 | |
| Sample | Diameter of Single Crystal | Permeability (Barrer) | Ideal Selectivity | |||||
|---|---|---|---|---|---|---|---|---|
| H2 | CO2 | O2 | N2 | H2/N2 | CO2/N2 | O2/N2 | ||
| T/C [57] | 0.5 μm | 4230 | 1773 | 486 | 37.2 | 113.7 | 47.7 | 13.1 |
| 3 μm | 2474 | 1580 | 341 | 41.5 | 59.6 | 38.1 | 8.2 | |
| 6 μm | 4094 | 1578 | 349 | 46.5 | 88 | 33.9 | 7.5 | |
| 8 μm | 4168 | 2151 | 492 | 107 | 39 | 20.1 | 4.6 | |
| ZSM-5/C [59] | 100 nm | 1179 | 564 | 237 | 15.0 | 78.6 | 37.7 | 15.8 |
| 1 μm | 1366 | 629 | 253 | 23.6 | 57.9 | 26.6 | 10.7 | |
| 5 μm | 1447 | 637 | 254 | 27.0 | 53.6 | 23.7 | 9.4 | |
| 10 μm | 1506 | 660 | 257 | 30.1 | 50 | 22 | 8.5 | |
| Agglomeration Particle Diameter | Permeability (Barrer) | Ideal Selectivity | |||||
|---|---|---|---|---|---|---|---|
| H2 | CO2 | O2 | N2 | H2/ N2 | CO2/N2 | O2/N2 | |
| 20–50 nm 9.09 wt% [56] | 5399 | 3020 | 671 | 59 | 91.5 | 51.2 | 11.4 |
| 0.2 μm [59] | 1179 | 564 | 237 | 15.0 | 78.6 | 37.7 | 15.8 |
| 1 μm [59] | 1190 | 607 | 221 | 18.2 | 65.4 | 33.4 | 12.1 |
| 4 μm [59] | 1224 | 624 | 231 | 21.6 | 56.7 | 28.9 | 10.7 |
| Membrane | Carbonization Temperature (°C) | Permeability (Barrer) | Selectivity | |||||
|---|---|---|---|---|---|---|---|---|
| H2 | CO2 | O2 | N2 | H2/N2 | CO2/N2 | O2/N2 | ||
| CMSM | 600 | 16.5 | 6.57 | 0.74 | 0.05 | 330 | 123 | 13.8 |
| 700 | 84.4 | 52.7 | 4 | 0.27 | 312.6 | 195 | 14.8 | |
| 800 | 36 | 25 | 1.71 | 0.11 | 327.3 | 234 | 16 | |
| zeolite β/C hybrid membrane | 600 | 2234 | 1303 | 357 | 51.9 | 43 | 25.1 | 6.9 |
| 700 | 1721 | 810 | 274 | 21.3 | 80.8 | 38 | 12.8 | |
| 800 | 253 | 158 | 12 | 0.81 | 312.3 | 195 | 14.8 | |
| zeolite Y/C hybrid membrane | 600 | 2304 | 1148 | 298 | 50.8 | 45.4 | 22.6 | 5.86 |
| 700 | 1717 | 761 | 236 | 20.7 | 82.9 | 36.7 | 11.4 | |
| 800 | 102 | 42.6 | 7.5 | 0.7 | 145.7 | 59.3 | 10.5 | |
| Sample | Permeability (Barrer) | Ideal Selectivity | |||||
|---|---|---|---|---|---|---|---|
| H2 | CO2 | O2 | N2 | H2/N2 | CO2/N2 | O2/N2 | |
| Y (pyrolyzed)/carbon | 339 | 204 | 38 | 4 | 84.8 | 51 | 9.6 |
| Y (intact)/carbon | 1717 | 761 | 236 | 20.7 | 82.9 | 36.7 | 11.4 |
| CMSM | 84.4 | 52.7 | 4 | 0.27 | 312.6 | 195.1 | 14.8 |
| Sample | Permeability/Barrer | Ideal Selectivity | ||||||
|---|---|---|---|---|---|---|---|---|
| H2 | CO2 | O2 | N2 | CH4 | O2/N2 | CO2/N2 | CO2/CH4 | |
| CMSM [59] | 84.4 | 52.7 | 4 | 0.27 | - | 14.8 | 195.1 | - |
| 500 nm SBA-15/C [38] | 1807 | 1410 | 246 | 37 | 14.5 | 6.6 | 38.1 | 97.2 |
| 100–200 nm MCM-48/C [38] | 3838 | 2461 | 527 | 62 | 25 | 8.5 | 39.7 | 98.4 |
| Sample | Permeability/Barrer | Ideal Selectivity | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| H2 | CO2 | O2 | N2 | CH4 | O2/N2 | CO2/N2 | H2/N2 | CO2/CH4 | ||
| CMSM | 853 | 321 | 151 | 14 | 6 | 10.79 | 22.93 | 60.93 | 53.50 | |
| SWCNT/C | 2563 | 2626 | 522 | 96 | 46 | 5.4 | 27.4 | 26.7 | 57.1 | |
| MWCNT2/C | 4931 | 6659 | 1307 | 280 | 206 | 4.7 | 23.8 | 17.6 | 32.3 | |
| MWCNT2 1 (Acid treated)/C | 5824 | 6661 | 1259 | 253 | 161 | 5.0 | 26.3 | 23.0 | 41.4 | |
| MWCNT2 Concentration | 0 | 853 | 321 | 151 | 14 | 6 | 10.8 | 22.9 | 60.9 | 53.5 |
| 5% | 2600 | 3407 | 701 | 139 | 107 | 5.1 | 24.3 | 18.7 | 31.8 | |
| 10% | 5824 | 6661 | 1259 | 253 | 161 | 5.0 | 26.3 | 23.0 | 41.4 | |
| 15% | 7071 | 9332 | 1576 | 335 | 256 | 4.7 | 27.9 | 21.1 | 36.5 | |
| MWCNT1 2/C | 4659 | 5425 | 1061 | 234 | 204 | 4.5 | 23.2 | 19.9 | 26.6 | |
| MWCNT3 3/C | 3886 | 4395 | 851 | 164 | 103 | 5.2 | 26.8 | 23.7 | 42.7 | |
| Sample or Hybridized Particle (Content in the Precursor) | Permeability/Barrer a | Ideal Selectivity | |||||||
|---|---|---|---|---|---|---|---|---|---|
| H2 | CO2 | O2 | N2 | CH4 | O2/N2 | CO2/N2 | H2/N2 | CO2/CH4 | |
| CMSM [59] | 84.4 | 52.7 | 4 | 0.27 | - | 14.8 | 195.1 | 312.6 | - |
| MWCNT2 (10%) [89] | 5824 | 6661 | 1259 | 253 | 161 | 5.0 | 26.3 | 23.0 | 41.4 |
| 500 nm SBA-15 (10%) [38] | 1807 | 1410 | 246 | 37 | 14.5 | 6.6 | 38.1 | 48.8 | 97.2 |
| 100–200 nm MCM-48 (10%) [38] | 3838 | 2461 | 527 | 62 | 25 | 8.5 | 39.7 | 61.9 | 98.4 |
| 0.5 μm T (10%) [57] | 4230 | 1773 | 486 | 37.2 | - | 13.1 | 47.7 | 113.7 | - |
| 100 nm ZSM-5 (10%) [59] | 1179 | 564 | 237 | 15.0 | - | 15.8 | 37.6 | 78.6 | - |
| 20–50 nm ZSM-5 (9.09 wt. %) [56] | 5399 | 3020 | 671 | 59 | - | 11.4 | 51.2 | 91.5 | - |
| 20–50 nm Fe3O4 (10%) [64] | 6790 | 2764 | 786 | 79 | 48 | 9.9 | 35.0 | 86.0 | 57.6 |
| Sample | CO2 Permeability/Barrer | CO2/CH4 Separation Factor |
|---|---|---|
| P84-based CMS membrane | 7.1 | 72.4 |
| Hybrid CMS membrane with ZIF-108/P84 = 0.1 | 40.9 | 174 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, L.; Xu, R.; Song, C.; Zhang, B.; Liu, Q.; Wang, T. A Review on the Progress in Nanoparticle/C Hybrid CMS Membranes for Gas Separation. Membranes 2018, 8, 134. https://doi.org/10.3390/membranes8040134
Li L, Xu R, Song C, Zhang B, Liu Q, Wang T. A Review on the Progress in Nanoparticle/C Hybrid CMS Membranes for Gas Separation. Membranes. 2018; 8(4):134. https://doi.org/10.3390/membranes8040134
Chicago/Turabian StyleLi, Lin, Ruisong Xu, Chengwen Song, Bing Zhang, Qingling Liu, and Tonghua Wang. 2018. "A Review on the Progress in Nanoparticle/C Hybrid CMS Membranes for Gas Separation" Membranes 8, no. 4: 134. https://doi.org/10.3390/membranes8040134
APA StyleLi, L., Xu, R., Song, C., Zhang, B., Liu, Q., & Wang, T. (2018). A Review on the Progress in Nanoparticle/C Hybrid CMS Membranes for Gas Separation. Membranes, 8(4), 134. https://doi.org/10.3390/membranes8040134





