Short Review on Porous Metal Membranes—Fabrication, Commercial Products, and Applications
Abstract
1. Introduction
2. Fabrication of Porous Metal Membranes
3. Commercial Porous Metal Membranes
3.1. Porous Metal Membranes Based on Stainless Steel (SS)
3.2. Porous Metal Membranes Based on Alloys
3.3. Porous Metal Membranes Based on Other Metals
4. Applications of Porous Metal Membranes
4.1. Porous Metal Membrane Filtration
4.2. Porous Metal Membrane Contactors for Membrane Evaporation
4.3. Porous Metal Membrane Bioreactors
4.4. Photocatalytic Metal Membranes
4.5. Catalytic Metal Membranes
5. Research Opportunities
6. Conclusions and Prospects
Funding
Acknowledgments
Conflicts of Interest
References
- Zhu, B.; Li, B.; Zou, L.; Hill, A.J.; Zhao, D.; Lin, J.Y.S.; Duke, M. Functional zeolitic framework membranes for water treatment and desalination. In Functional Nanostructured Materials and Membranes for Water Treatment, 1st ed.; Duke, M., Zhao, D., Semiat, R., Eds.; Wiley-VCH Verlag GmbH & Co. KGaA: Weinheim, Germany, 2013; pp. 217–247. [Google Scholar]
- Introduction to Membrane. Available online: http://www.separationprocesses.com/Membrane/MT_Chp07a.htm (accessed on 16 September 2018).
- Caro, J.; Noack, M. Chapter 1—Zeolite membranes—Status and prospective. In Advances in Nanoporous Materials; Stefan, E., Ed.; Elsevier: Amsterdam, The Netherlands, 2010; Volume 1, pp. 1–96. [Google Scholar]
- He, Y.-H. Metal membranes. In Membrane-Based Separations in Metallurgy: Principles and Applications; Jiang, L.Y., Li, N., Eds.; Elsevier Science: Amsterdam, The Netherlands, 2017; pp. 371–390. [Google Scholar]
- Paganessi, J.; Yuan, J. Use of Porous Metal Membrane for Treating Liquid Foods. U.S. Patent 20060201342 A1, 14 September 2006. [Google Scholar]
- Ahmad, S. Application of membrane technology in food processing. In Food Processing: Strategies for Quality Assessment; Malik, A., Erginkaya, Z., Ahmad, S., Erten, H., Eds.; Springer: New York, NY, USA, 2014; pp. 379–394. [Google Scholar]
- Mannapperuma, J. Design and performance evaluation of membrane systems. In Handbook of Food Engineering Practice; Valentas, K.J., Rotstein, E., Singh, R.P., Eds.; CRC Press: Boca Raton, FL, USA, 1997; pp. 167–210. [Google Scholar]
- Desorbo, W.; Cline, H.E. Metal membranes with uniform submicron-size pores. J. Appl. Phys. 1970, 41, 2099–2105. [Google Scholar] [CrossRef]
- Dumee, L.F.; He, L.; Lin, B.; Ailloux, F.-M.; Lemoine, J.-B.; Velleman, L.; She, F.; Duke, M.C.; Orbell, J.D.; Erskine, G.; et al. The fabrication and surface functionalization of porous metal frameworks—A review. J. Mater. Chem. A 2013, 1, 15185–15206. [Google Scholar] [CrossRef]
- Leiknes, T.; Myklebust, H.; Ødegaard, H. Metal membranes for drinking water treatment. Membr. Technol. 2005, 2005, 6–10. [Google Scholar] [CrossRef]
- Kim, J.-O.; Jung, J.-T.; Chung, J. Treatment performance of metal membrane microfiltration and electrodialysis integrated system for wastewater reclamation. Desalination 2007, 202, 343–350. [Google Scholar] [CrossRef]
- Ouyang, L.; Dotzauer, D.M.; Hogg, S.R.; Macanás, J.; Lahitte, J.-F.; Bruening, M.L. Catalytic hollow fiber membranes prepared using layer-by-layer adsorption of polyelectrolytes and metal nanoparticles. Catal. Today 2010, 156, 100–106. [Google Scholar] [CrossRef]
- Zhang, S.; Qu, Y.; Liu, Y.; Yang, F.; Zhang, X.; Furukawa, K.; Yamada, Y. Experimental study of domestic sewage treatment with a metal membrane bioreactor. Desalination 2005, 177, 83–93. [Google Scholar] [CrossRef]
- Zhang, S.; Yang, F.; Liu, Y.; Zhang, X.; Yamada, Y.; Furukawa, K. Performance of a metallic membrane bioreactor treating simulated distillery wastewater at temperatures of 30 to 45 °C. Desalination 2006, 194, 146–155. [Google Scholar] [CrossRef]
- Xie, Y.H.; Zhu, T.; Xu, C.H.; Nozaki, T.; Furukawa, K. Treatment of domestic sewage by a metal membrane bioreactor. Water Sci. Technol. 2012, 65, 1102–1108. [Google Scholar] [CrossRef] [PubMed]
- Westermann, T.; Melin, T. Flow-through catalytic membrane reactors—Principles and applications. Chem. Eng. Process. 2009, 48, 17–28. [Google Scholar] [CrossRef]
- Ilinich, O.M.; Cuperus, F.P.; van Gemert, R.W.; Gribov, E.N.; Nosova, L.V. Catalytic membrane in denitrification of water: A means to facilitate intraporous diffusion of reactants. Sep. Purif. Technol. 2000, 21, 55–60. [Google Scholar] [CrossRef]
- Ersahin, M.E.; Ozgun, H.; Dereli, R.K.; Ozturk, I.; Roest, K.; van Lier, J.B. A review on dynamic membrane filtration: Materials, applications and future perspectives. Bioresour. Technol. 2012, 122, 196–206. [Google Scholar] [CrossRef] [PubMed]
- Hengl, N.; Mourgues, A.; Pomier, E.; Belleville, M.P.; Paolucci-Jeanjean, D.; Sanchez, J.; Rios, G. Study of a new membrane evaporator with a hydrophobic metallic membrane. J. Membr. Sci. 2007, 289, 169–177. [Google Scholar] [CrossRef]
- Lin, B.; Dumée, L.; Hodgson, P.; Kong, L. De-alloying a nano-porous metal membrane for desalination. In Proceedings of the International Membrane Science and Technology Conference (IMSTEC), Melbourne, Australia, 23–29 November 2013. [Google Scholar]
- Leiknes, T.; Ødegaard, H.; Myklebust, H. Removal of natural organic matter (NOM) in drinking water treatment by coagulation–microfiltration using metal membranes. J. Membr. Sci. 2004, 242, 47–55. [Google Scholar] [CrossRef]
- Banhart, J. Manufacture, characterisation and application of cellular metals and metal foams. Prog. Mater. Sci. 2001, 46, 559–632. [Google Scholar] [CrossRef]
- Liu, P.S.; Liang, K.M. Review functional materials of porous metals made by P/M, electroplating and some other techniques. J. Mater. Sci. 2001, 36, 5059–5072. [Google Scholar] [CrossRef]
- Tong, J.; Su, C.; Kuraoka, K.; Suda, H.; Matsumura, Y. Preparation of thin Pd membrane on CeO2-modified porous metal by a combined method of electroless plating and chemical vapor deposition. J. Membr. Sci. 2006, 269, 101–108. [Google Scholar] [CrossRef]
- Xomeritakis, G.; Lin, Y.S. Fabrication of a thin palladium membrane supported in a porous ceramic substrate by chemical vapor deposition. J. Membr. Sci. 1996, 120, 261–272. [Google Scholar] [CrossRef]
- Anderson, D.G.; Anwar, N.; Aylett, B.J.; Earwaker, L.G.; Nasir, M.I.; Farr, J.P.G.; Stiebahl, K.; Keen, J.M. Chemical vapour deposition of metals and metal silicides on the internal surfaces of porous silicon. J. Organomet. Chem. 1992, 437, C7–C12. [Google Scholar] [CrossRef]
- Rubow, K.L. Advances in filtration technology using sintered metal filters. In Proceedings of the 3rd China International Filtration Exhibition and Conference, Shanghai, China, 16–18 December 2004. [Google Scholar]
- Vida-Simiti, I.; Jumate, N.; Thalmaier, G.; Sechel, N.; Moldovan, V. Metallic membranes obtained with graded structure for microfiltration. Environ. Eng. Manag. J. 2011, 10, 1439–1444. [Google Scholar] [CrossRef]
- Sinterflo® Sintered Metal Filter Cartridges and Elements, Porvair Filtration Group. Available online: https://www.porvairfiltration.com/storage/downloads/sinterflo_brochure_us-1484748277.pdf (accessed on 16 September 2018).
- Duraster© Metallic Microfiltration Membranes, Advanced Material Solutions. Available online: http://www.ams100.com/pdf/AMS%20Crossflow%20Metallic%20Membrane%20Filtration.pdf (accessed on 16 September 2018).
- Data Sheet e78b, Accusep™ Inorganic Membranes, Pall Corporation. Available online: http://www.pall.fr/pdfs/Fuels-and-Chemicals/E78a_AccuSep_Inorganic.pdf (accessed on 16 September 2018).
- PMM® Metal Membrane Filter Cartridges, Pall Corporation. Available online: https://shop.pall.com/us/en/food-beverage/bottled-water/utilities-filtration/pmm-metal-membrane-filter-cartridges-zidgri78ldj?tracking=searchterm:metal+membranes (accessed on 16 September 2018).
- Tappan, B.C.; Steiner, S.A.; Luther, E.P. Nanoporous metal foams. Angew. Chem. Int. Ed. 2010, 49, 4544–4565. [Google Scholar] [CrossRef] [PubMed]
- Jiang, P.; Cizeron, J.; Bertone, J.F.; Colvin, V.L. Preparation of macroporous metal films from colloidal crystals. J. Am. Chem. Soc. 1999, 121, 7957–7958. [Google Scholar] [CrossRef]
- Erlebacher, J.; Aziz, M.J.; Karma, A.; Dimitrov, N.; Sieradzki, K. Evolution of nanoporosity in dealloying. Nature 2001, 410, 450–453. [Google Scholar] [CrossRef] [PubMed]
- Ding, Y.; Erlebacher, J. Nanoporous metals with controlled multimodal pore size distribution. J. Am. Chem. Soc. 2003, 125, 7772–7773. [Google Scholar] [CrossRef] [PubMed]
- Ding, Y.; Chen, M.; Erlebacher, J. Metallic mesoporous nanocomposites for electrocatalysis. J. Am. Chem. Soc. 2004, 126, 6876–6877. [Google Scholar] [CrossRef] [PubMed]
- Lin, B.; Kong, L.; Hodgson, P.; Dumée, L. Impact of the de-alloying kinetics and alloy microstructure on the final morphology of de-alloyed meso-porous metal films. Nanomaterials 2014, 4, 856–878. [Google Scholar] [CrossRef] [PubMed]
- Lin, B.; Kong, L.; Hodgson, P.D.; Mudie, S.; Hawley, A.; Dumee, L.F. Controlled porosity and pore size of nano-porous gold by thermally assisted chemical dealloying—A SAXS study. RSC Adv. 2017, 7, 10821–10830. [Google Scholar] [CrossRef]
- Wu, H.; Hu, L.; Rowell, M.W.; Kong, D.; Cha, J.J.; McDonough, J.R.; Zhu, J.; Yang, Y.; McGehee, M.D.; Cui, Y. Electrospun metal nanofiber webs as high-performance transparent electrode. Nano Lett. 2010, 10, 4242–4248. [Google Scholar] [CrossRef] [PubMed]
- Bognitzki, M.; Becker, M.; Graeser, M.; Massa, W.; Wendorff, J.H.; Schaper, A.; Weber, D.; Beyer, A.; Gölzhäuser, A.; Greiner, A. Preparation of sub-micrometer copper fibers via electrospinning. Adv. Mater. 2006, 18, 2384–2386. [Google Scholar] [CrossRef]
- Hansen, N.S.; Cho, D.; Joo, Y.L. Metal nanofibers with highly tunable electrical and magnetic properties via highly loaded water-based electrospinning. Small 2012, 8, 1510–1514. [Google Scholar] [CrossRef] [PubMed]
- Wu, H.; Zhang, R.; Liu, X.; Lin, D.; Pan, W. Electrospinning of Fe, Co, and Ni nanofibers: Synthesis, assembly, and magnetic properties. Chem. Mater. 2007, 19, 3506–3511. [Google Scholar] [CrossRef]
- Barakat, N.A.M.; Kim, B.; Kim, H.Y. Production of smooth and pure nickel metal nanofibers by the electrospinning technique: Nanofibers possess splendid magnetic properties. J. Phys. Chem. C 2009, 113, 531–536. [Google Scholar] [CrossRef]
- Macías, M.; Chacko, A.; Ferraris, J.P.; Balkus, K.J., Jr. Electrospun mesoporous metal oxide fibers. Microporous Mesoporous Mater. 2005, 86, 1–13. [Google Scholar] [CrossRef]
- Qiu, Y.; Yu, J.; Zhou, X.; Tan, C.; Yin, J. Synthesis of porous NiO and ZnO submicro- and nanofibers from electrospun polymer fiber templates. Nanoscale Res. Lett. 2009, 4, 173–177. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Canfield, N. Development of thin porous metal sheet as micro-filtration membrane and inorganic membrane support. J. Membr. Sci. 2012, 409–410, 113–126. [Google Scholar] [CrossRef]
- Daddis, J.L.; Jernigan, D.A. Sintered Coating for Porous Metallic Filter Surfaces. U.S. Patent US4888114A, 19 December 1989. [Google Scholar]
- Zhan, M.; Li, G.; Wei, Q.; Cui, H.; Lin, L. Preparation of porous TiO2/Ti composite membrane for immunoisolation. Appl. Surf. Sci. 2008, 255, 2256–2258. [Google Scholar]
- Singh, M.; Haverinen, H.M.; Dhagat, P.; Jabbour, G.E. Inkjet printing-process and its applications. Adv. Mater. 2010, 22, 673–685. [Google Scholar] [CrossRef] [PubMed]
- Mott, M.; Song, J.H.; Evans, J.R.G. Microengineering of ceramics by direct ink-jet printing. J. Am. Ceram. Soc. 1999, 82, 1653–1658. [Google Scholar] [CrossRef]
- Calvert, P. Inkjet printing for materials and devices. Chem. Mater. 2001, 13, 3299–3305. [Google Scholar] [CrossRef]
- Chen, Z.; Brandon, N. Inkjet printing and nanoindentation of porous alumina multilayers. Ceram. Int. 2016, 42, 8316–8324. [Google Scholar] [CrossRef]
- Lejeune, M.; Chartier, T.; Dossou-Yovo, C.; Noguera, R. Ink-jet printing of ceramic micro-pillar arrays. J. Eur. Ceram. Soc. 2009, 29, 905–911. [Google Scholar] [CrossRef]
- Ding, X.; Li, Y.; Wang, D.; Yin, Q. Fabrication of BaTiO3 dielectric films by direct ink-jet printing. Ceram. Int. 2004, 30, 1885–1887. [Google Scholar] [CrossRef]
- Mougenot, M.; Lejeune, M.; Baumard, J.F.; Boissiere, C.; Ribot, F.; Grosso, D.; Sanchez, C.; Noguera, R. Ink jet printing of microdot arrays of mesostructured silica. J. Am. Ceram. Soc. 2006, 89, 1876–1882. [Google Scholar] [CrossRef]
- Kanso, A.; Arnaud, E.; Chreim, H.; Monédière, T.; Thévenot, M.; Beaudrouet, E.; Dossou-yovo, C.; Noguéra, R. Design and fabrication of EBG and CWP antennas using inkjet printing technology. Microw. Opt. Technol. Lett. 2013, 55, 1520–1526. [Google Scholar] [CrossRef]
- Yashiro, N.; Usui, T.; Kikuta, K. Application of a thin intermediate cathode layer prepared by inkjet printing for SOFCs. J. Eur. Ceram. Soc. 2010, 30, 2093–2098. [Google Scholar] [CrossRef]
- Fuller, S.B.; Wilhelm, E.J.; Jacobson, J.M. Ink-jet printed nanoparticle microelectromechanical systems. J. Microelectromech. Syst. 2002, 11, 54–60. [Google Scholar] [CrossRef]
- Mark, L.A.; Mikko, A.; Tomi, M.; Ari, A.; Kimmo, O.; Mika, S.; Heikki, S. Electrical sintering of nanoparticle structures. Nanotechnology 2008, 19, 175201. [Google Scholar]
- Reinhold, I.; Hendriks, C.E.; Eckardt, R.; Kranenburg, J.M.; Perelaer, J.; Baumann, R.R.; Schubert, U.S. Argon plasma sintering of inkjet printed silver tracks on polymer substrates. J. Mater. Chem. 2009, 19, 3384–3388. [Google Scholar] [CrossRef]
- Qu, L.; Dai, L. Substrate-enhanced electroless deposition of metal nanoparticles on carbon nanotubes. J. Am. Chem. Soc. 2005, 127, 10806–10807. [Google Scholar] [CrossRef] [PubMed]
- Djokić, S.S.; Cavallotti, P.L. Electroless deposition: Theory and applications. In Electrodeposition: Theory and Practice; Djokic, S.S., Ed.; Springer: New York, NY, USA, 2010; pp. 251–289. [Google Scholar]
- Shu, J.; Adnot, A.; Grandjean, B.P.A.; Kaliaguine, S. Structurally stable composite Pd-Ag alloy membranes: Introduction of a diffusion barrier. Thin Solid Films 1996, 286, 72–79. [Google Scholar] [CrossRef]
- Wu, Z.; Ge, S.; Zhang, M.; Li, W.; Tao, K. Synthesis of nickel nanoparticles supported on metal oxides using electroless plating: Controlling the dispersion and size of nickel nanoparticles. J. Colloid Interface Sci. 2009, 330, 359–366. [Google Scholar] [CrossRef] [PubMed]
- Velleman, L.; Losic, D.; Shapter, J.G. The effects of surface functionality positioning on the transport properties of membranes. J. Membr. Sci. 2012, 411–412, 211–218. [Google Scholar] [CrossRef]
- Wirtz, M.; Parker, M.; Kobayashi, Y.; Martin, C.R. Molecular sieving and sensing with gold nanotube membranes. Chem. Rec. 2002, 2, 259–267. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; He, C.; Xing, W.; Li, F.; Tong, L.; Chen, Z.; Liao, X.; Steinhart, M. Nanoporous metal membranes with bicontinuous morphology from recyclable block-copolymer templates. Adv. Mater. 2010, 22, 2068–2072. [Google Scholar] [CrossRef] [PubMed]
- Arora, H.; Li, Z.; Sai, H.; Kamperman, M.; Warren, S.C.; Wiesner, U. Block copolymer directed nanoporous metal thin films. Macromol. Rapid Commun. 2010, 31, 1960–1964. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Sai, H.; Warren, S.C.; Kamperman, M.; Arora, H.; Gruner, S.M.; Wiesner, U. Metal nanoparticle−block copolymer composite assembly and disassembly. Chem. Mater. 2009, 21, 5578–5584. [Google Scholar] [CrossRef] [PubMed]
- Warren, S.C.; Messina, L.C.; Slaughter, L.S.; Kamperman, M.; Zhou, Q.; Gruner, S.M.; DiSalvo, F.J.; Wiesner, U. Ordered mesoporous materials from metal nanoparticle–block copolymer self-assembly. Science 2008, 320, 1748–1752. [Google Scholar] [CrossRef] [PubMed]
- Orrù, R.; Licheri, R.; Locci, A.M.; Cincotti, A.; Cao, G. Consolidation/synthesis of materials by electric current activated/assisted sintering. Mater. Sci. Eng. 2009, 63, 127–287. [Google Scholar] [CrossRef]
- Schmeda-Lopez, D.R.; Smart, S.; Nunes, E.H.M.; Vasconcelos, D.; Vasconcelos, W.L.; Bram, M.; Meulenberg, W.A.; Diniz da Costa, J.C. Stainless steel hollow fibres—Sintering, morphology and mechanical properties. Sep. Purif. Technol. 2015, 147, 379–387. [Google Scholar] [CrossRef]
- Olevsky, E.; Molinari, A. Instability of sintering of porous bodies. Int. J. Plast. 2000, 16, 1–37. [Google Scholar] [CrossRef]
- Campbell, C.T.; Parker, S.C.; Starr, D.E. The effect of size-dependent nanoparticle energetics on catalyst sintering. Science 2002, 298, 811–814. [Google Scholar] [CrossRef] [PubMed]
- Porous Metal Filter Elements, Mott Corporation. Available online: https://mottcorp.com/sites/default/files/PSELEMENTS_1.pdf (accessed on 16 September 2018).
- Park, J.; Min, S.-H.; Lee, W.-H.; Park, N.-S.; Kim, H.-S.; Kim, J.-O. Properties and filtration performance of microporous metal membranes fabricated by rolling process. J. Water Reuse Desalin. 2016, 7, 11–15. [Google Scholar] [CrossRef]
- Allioux, F.-M.; David, O.; Etxeberria Benavides, M.; Kong, L.; Pacheco Tanaka, D.A.; Dumée, L.F. Preparation of porous stainless steel hollow-fibers through multi-modal particle size sintering towards pore engineering. Membranes 2017, 7, 40. [Google Scholar] [CrossRef] [PubMed]
- Meng, B.; Tan, X.; Meng, X.; Qiao, S.; Liu, S. Porous and dense Ni hollow fibre membranes. J. Alloys Compd. 2009, 470, 461–464. [Google Scholar] [CrossRef]
- Luiten-Olieman, M.W.J.; Winnubst, L.; Nijmeijer, A.; Wessling, M.; Benes, N.E. Porous stainless steel hollow fiber membranes via dry–wet spinning. J. Membr. Sci. 2011, 370, 124–130. [Google Scholar] [CrossRef]
- Luiten-Olieman, M.W.J.; Raaijmakers, M.J.T.; Winnubst, L.; Wessling, M.; Nijmeijer, A.; Benes, N.E. Porous stainless steel hollow fibers with shrinkage-controlled small radial dimensions. Scr. Mater. 2011, 65, 25–28. [Google Scholar] [CrossRef]
- Michielsen, B.; Chen, H.; Jacobs, M.; Middelkoop, V.; Mullens, S.; Thijs, I.; Buekenhoudt, A.; Snijkers, F. Preparation of porous stainless steel hollow fibers by robotic fiber deposition. J. Membr. Sci. 2013, 437, 17–24. [Google Scholar] [CrossRef]
- Wang, M.; Huang, M.-L.; Cao, Y.; Ma, X.-H.; Xu, Z.-L. Fabrication, characterization and separation properties of three-channel stainless steel hollow fiber membrane. J. Membr. Sci. 2016, 515, 144–153. [Google Scholar] [CrossRef]
- Rui, W.; Zhang, C.; Cai, C.; Gu, X. Effects of sintering atmospheres on properties of stainless steel porous hollow fiber membranes. J. Membr. Sci. 2015, 489, 90–97. [Google Scholar] [CrossRef]
- Liu, Y.; Chen, O.Y.; Wei, C.C.; Li, K. Preparation of yttria-stabilised zirconia (YSZ) hollow fibre membranes. Desalination 2006, 199, 360–362. [Google Scholar] [CrossRef]
- Liu, S.; Li, K.; Hughes, R. Preparation of porous aluminium oxide (Al2O3) hollow fibre membranes by a combined phase-inversion and sintering method. Ceram. Int. 2003, 29, 875–881. [Google Scholar] [CrossRef]
- Zhang, X.; Lin, B.; Ling, Y.; Dong, Y.; Fang, D.; Meng, G.; Liu, X. Highly permeable porous YSZ hollow fiber membrane prepared using ethanol as external coagulant. J. Alloys Compd. 2010, 494, 366–371. [Google Scholar] [CrossRef]
- Zhang, X.; Wang, D.K.; Lopez, D.R.S.; Diniz da Costa, J.C. Fabrication of nanostructured TiO2 hollow fiber photocatalytic membrane and application for wastewater treatment. Chem. Eng. J. 2014, 236, 314–322. [Google Scholar] [CrossRef]
- Chong, J.Y.; Wang, B.; Li, K. High performance stainless steel-ceramic composite hollow fibres for microfiltration. J. Membr. Sci. 2017, 541, 425–433. [Google Scholar] [CrossRef]
- Li, Z.; Yang, Z.; Qiu, N.; Yang, G. A sol–gel-derived α-Al2O3 crystal interlayer modified 316l porous stainless steel to support TiO2, SiO2, and TiO2–SiO2 hybrid membranes. J. Mater. Sci. 2011, 46, 3127–3135. [Google Scholar] [CrossRef]
- Wang, J. Studies on the Preparation of Ni Membrane and Pd Membrane Supported on TiAl Intermetallic Alloy. Master’s Thesis, Nanjing Tech University, Nanjing, China, 2006. [Google Scholar]
- Zhou, S.; Fan, Y.; He, Y.; Xu, N. Preparation of titania microfiltration membranes supported on porous Ti–Al alloys. J. Membr. Sci. 2008, 325, 546–552. [Google Scholar] [CrossRef]
- Shen, P.Z.; He, Y.H.; Gao, H.Y.; Zou, J.; Xu, N.P.; Jiang, Y.; Huang, B.Y.; Liu, C.T. Development of a new graded-porosity FeAl alloy by elemental reactive synthesis. Desalination 2009, 249, 29–33. [Google Scholar] [CrossRef]
- Yang, F.; Lin, J.; He, Y.; Du, H.; Chen, G. Innovative fabrication of Ti–48Al–6Nb porous coating by cold gas spraying and reactive sintering. Mater. Lett. 2012, 76, 190–193. [Google Scholar] [CrossRef]
- Wang, Y.H.; Lin, J.P.; He, Y.H.; Wang, Y.L.; Chen, G.L. Effect of Nb on pore structure and tensile property of Ti–48Al cellular alloy. J. Alloys Compd. 2008, 456, 297–303. [Google Scholar] [CrossRef]
- GKN. SIKA-R…IS/AS. Available online: https://www.gkn.com/globalassets/downloads/powder-metallurgy/2017/sika-r-is-as-english.pdf (accessed on 16 September 2018).
- GKN. SIKA-R…AX. Available online: https://www.gkn.com/globalassets/downloads/powder-metallurgy/2017/sika-r-ax-english.pdf (accessed on 16 September 2018).
- GKN. SIKA-FIL. Available online: https://www.gkn.com/globalassets/downloads/powder-metallurgy/2017/sika-fil-english.pdf (accessed on 16 September 2018).
- GKN. SIKA-B. Available online: https://www.gkn.com/globalassets/downloads/powder-metallurgy/2017/sika-b-english.pdf (accessed on 16 September 2018).
- PSS® Porous Metal Filter Cartridges, Pall Corporation. Available online: https://shop.pall.com/us/en/food-beverage/beer-corporate-/particle-filtration/pss-porous-metal-filter-cartridges-zidgri78lxh (accessed on 16 September 2018).
- Accusep™ Inorganic Membranes, Pall Corporation. Available online: http://www.pall.de/pdfs/misc/Accusep_Membrane.pdf (accessed on 16 September 2018).
- Scepter® Stainless Steel Membrane, Graver Technologies. Available online: https://www.gravertech.com/product-lines/crossflow-membrane/scepter-tubular-mf-and-uf-modules/scepter-membrane-module-assemblies/scepter-membrane-module-assemblies-brochure/ (accessed on 16 September 2018).
- Ferrocep®, the Superior Industrial Separation Solution, Hyflux Ltd. Available online: http://www.hyfluxmembranes.com/images/documents/brochures/ferrocep-eng.pdf (accessed on 16 September 2018).
- Metal Membranes, Innovative Membrane Technology. Available online: http://www.metalmembranes.com (accessed on 16 September 2018).
- Silver Membranes, Sterlitech Corporation. Available online: https://www.sterlitech.com/silver-membranes.html (accessed on 16 September 2018).
- AMS Filtration: Titanium Filtration Membranes and Complete Systems. Available online: http://www.apexenvironmental.co.nz/wp-content/uploads/AMS-Wastewater-Brochure.pdf (accessed on 16 September 2018).
- Holmes, M. Mott introduces sterilising grade all-metal filtration membrane. Filtr. Sep. 2013, 50, 18. [Google Scholar]
- Metz, S.J.; Woltersar, H.H. Novel hybrid ceramic metal membrane. In Proceedings of the 15th Aachener Membran Kolloquium (AMK), Aachen, Germany, 12–13 November 2014; pp. 189–194. [Google Scholar]
- Food and Beverage, Pall Corporation. Available online: https://www.pall.com/pdfs/Food-and-Beverage/FBTADIRCROSSEN.pdf (accessed on 16 September 2018).
- Julkowski, J.K. Design and operation of porous metal crossflow microfilters. In Proceedings of the American Filtration and Separation Society Conference, Nashville, TN, USA, 11–12 September 2018; Available online: http://mottcorp.com/sites/default/files/DesignOpPorousMetalCrossflowMicrofilters.pdf (accessed on 16 September 2018).
- Baker, R.W. Membrane Technology and Applications, 2nd ed.; Wiley: Chichester, UK, 2004. [Google Scholar]
- Xie, S.-M.; Zhang, M.; Wang, Z.-Y.; Yuan, L.-M. Porous metal membranes for solid-phase extraction of polycyclic aromatic hydrocarbons. Analyst 2011, 136, 3988–3996. [Google Scholar] [CrossRef] [PubMed]
- Catalyst Recovery Filter. Available online: http://www.gkn.com/sintermetals/products/filter-applications/catalyst-recovery-filter/Pages/default.aspx (accessed on 16 September 2018).
- Kim, R.-H.; Lee, S.; Kim, J.-O. Application of a metal membrane for rainwater utilization: Filtration characteristics and membrane fouling. Desalination 2005, 177, 121–132. [Google Scholar] [CrossRef]
- Kim, R.H.; Lee, S.; Kim, S.-K.; Kim, J.-O. Advanced treatment of rainwater using metal membrane combined with ozonation. In Proceedings of the XI International Rainwater Catchment Systems Association (IRCSA) Conference, Mexico City, Mexico, 25–29 August 2003. [Google Scholar]
- Mostafa, M.G.; Zhu, B.; Cran, M.; Dow, N.; Milne, N.; Desai, D.; Duke, M. Membrane distillation of meat industry effluent with hydrophilic polyurethane coated polytetrafluoroethylene membranes. Membranes 2017, 7, 55. [Google Scholar] [CrossRef] [PubMed]
- Kunz, W.; Benhabiles, A.; Ben-Aïm, R. Osmotic evaporation through macroporous hydrophobic membranes: A survey of current research and applications. J. Membr. Sci. 1996, 121, 25–36. [Google Scholar] [CrossRef]
- Lawson, K.W.; Lloyd, D.R. Membrane distillation. J. Membr. Sci. 1997, 124, 1–25. [Google Scholar] [CrossRef]
- Pomier, E.; Hengl, N.; Belleville, M.-P.; Paolucci-Jeanjean, D.; Sanchez, J.; Rios, G. Study of a new metallic membrane evaporator. Desalination 2006, 199, 185–187. [Google Scholar] [CrossRef]
- Hengl, N.; Mourgues, A.; Belleville, M.P.; Paolucci-Jeanjean, D.; Sanchez, J. Membrane contactor with hydrophobic metallic membranes: 2. Study of operating parameters in membrane evaporation. J. Membr. Sci. 2010, 355, 126–132. [Google Scholar] [CrossRef]
- Mourgues, A.; Hengl, N.; Belleville, M.P.; Paolucci-Jeanjean, D.; Sanchez, J. Membrane contactor with hydrophobic metallic membranes: 1. Modeling of coupled mass and heat transfers in membrane evaporation. J. Membr. Sci. 2010, 355, 112–125. [Google Scholar] [CrossRef]
- Bouhabila, E.H.; Ben Aïm, R.; Buisson, H. Fouling characterisation in membrane bioreactors. Sep. Purif. Technol. 2001, 22–23, 123–132. [Google Scholar] [CrossRef]
- Kraume, M.; Bracklow, U.; Vocks, M.; Drews, A. Nutrients removal in MBRs for municipal wastewater treatment. Water Sci. Technol. 2005, 51, 391–402. [Google Scholar] [CrossRef] [PubMed]
- Zou, L.; Zhu, B. Enhancing the reuse of treated effluent by photocatalytic process. J. Adv. Oxid. Technol. 2007, 10, 273–281. [Google Scholar] [CrossRef]
- Zou, L.; Zhu, B. The synergistic effect of ozonation and photocatalysis on color removal from reused water. J. Photochem. Photobiol. A 2008, 196, 24–32. [Google Scholar] [CrossRef]
- Crittenden, J.C.; Suri, R.P.S.; Perram, D.L.; Hand, D.W. Decontamination of water using adsorption and photocatalysis. Water Res. 1997, 31, 411–418. [Google Scholar] [CrossRef]
- Merenda, A.; Kong, L.; Zhu, B.; Duke, M.C.; Gray, S.R.; Dumée, L.F. Functional nanoporous titanium dioxide for separation applications: Synthesis routes and properties to performance analysis. In Water Scarcity and Ways to Reduce the Impact: Management Strategies and Technologies for Zero Liquid Discharge and Future Smart Cities; Pannirselvam, M., Shu, L., Griffin, G., Philip, L., Natarajan, A., Hussain, S., Eds.; Springer: Cham, Switzerland, 2019; pp. 151–186. [Google Scholar]
- Choi, H.; Stathatos, E.; Dionysiou, D.D. Photocatalytic TiO2 films and membranes for the development of efficient wastewater treatment and reuse systems. Desalination 2007, 202, 199–206. [Google Scholar] [CrossRef]
- Choi, H.; Sofranko, A.C.; Dionysiou, D.D. Nanocrystalline TiO2 photocatalytic membranes with a hierarchical mesoporous multilayer structure: Synthesis, characterization, and multifunction. Adv. Funct. Mater. 2006, 16, 1067–1074. [Google Scholar] [CrossRef]
- Choi, W.-Y.; Chung, J.; Cho, C.-H.; Kim, J.-O. Fabrication and photocatalytic activity of a novel nanostructured TiO2 metal membrane. Desalination 2011, 279, 359–366. [Google Scholar] [CrossRef]
- Daub, K.; Wunder, V.K.; Dittmeyer, R. CVD preparation of catalytic membranes for reduction of nitrates in water. Catal. Today 2001, 67, 257–272. [Google Scholar] [CrossRef]
- Daub, K.; Emig, G.; Chollier, M.J.; Callant, M.; Dittmeyer, R. Studies on the use of catalytic membranes for reduction of nitrate in drinking water. Chem. Eng. Sci. 1999, 54, 1577–1582. [Google Scholar] [CrossRef]
- Kumakiri, I.; Hokstad, J.; Peters, T.A.; Melbye, A.G.; Ræder, H. Oxidation of aromatic components in water and seawater by a catalytic membrane process. J. Pet. Sci. Eng. 2011, 79, 37–44. [Google Scholar] [CrossRef]
- Vorlop, K.D.; Tacke, T. Erste schritte auf dem weg zur edelmetallkatalysierten nitrat- und nitrit-entfernung aus trinkwasser. Chem. Ing. Tech. 1989, 61, 836–837. [Google Scholar] [CrossRef]
- Wehbe, N.; Guilhaume, N.; Fiaty, K.; Miachon, S.; Dalmon, J.A. Hydrogenation of nitrates in water using mesoporous membranes operated in a flow-through catalytic contactor. Catal. Today 2010, 156, 208–215. [Google Scholar] [CrossRef]
- Pera-Titus, M.; Fridmann, M.; Guilhaume, N.; Fiaty, K. Modelling nitrate reduction in a flow-through catalytic membrane contactor: Role of pore confining effects on water viscosity. J. Membr. Sci. 2012, 401–402, 204–216. [Google Scholar]
- Allioux, F.-M.; David, O.; Merenda, A.; Maina, J.W.; Benavides, M.E.; Tanaka, A.P.; Dumee, L.F. Catalytic nickel and nickel-copper alloy hollow-fiber membranes for the remediation of organic pollutants by electrocatalysis. J. Mater. Chem. A 2018, 6, 6904–6915. [Google Scholar] [CrossRef]
- Thorp, J.C.; Sieradzki, K.; Tang, L.; Crozier, P.A.; Misra, A.; Nastasi, M.; Mitlin, D.; Picraux, S.T. Formation of nanoporous noble metal thin films by electrochemical dealloying of PtxSi1−x. Appl. Phys. Lett. 2006, 88, 033110. [Google Scholar] [CrossRef]
- Dumee, L.; Velleman, L.; Sears, K.; Hill, M.; Schutz, J.; Finn, N.; Duke, M.; Gray, S. Control of porosity and pore size of metal reinforced carbon nanotube membranes. Membranes 2011, 1, 25–36. [Google Scholar] [CrossRef] [PubMed]
- Velleman, L.; Shapter, J.G.; Losic, D. Gold nanotube membranes functionalised with fluorinated thiols for selective molecular transport. J. Membr. Sci. 2009, 328, 121–126. [Google Scholar] [CrossRef]
- Cvelbar, U.; Chen, Z.; Levchenko, I.; Sheetz, R.M.; Jasinski, J.B.; Menon, M.; Sunkara, M.K.; Ostrikov, K. Sub-oxide-to-metallic, uniformly-nanoporous crystalline nanowires by plasma oxidation and electron reduction. Chem. Commun. 2012, 48, 11070–11072. [Google Scholar] [CrossRef] [PubMed]
- Teo, W.E.; Ramakrishna, S. A review on electrospinning design and nanofibre assemblies. Nanotechnology 2006, 17, R89–R106. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Liu, Z.; Ma, S.; Zhou, Y. Electroless plating of Ni-Fe alloy on pet fabrics and effects of plating parameters on the alloy coating. In Proceedings of the 2011 IEEE International Conference on Remote Sensing, Environment and Transportation Engineering (RSETE), Nanjing, China, 24–26 June 2011; pp. 7561–7564. [Google Scholar]
- Neouze, M.-A.; Schubert, U. Surface modification and functionalization of metal and metal oxide nanoparticles by organic ligands. Monatsh. Chem. 2008, 139, 183–195. [Google Scholar] [CrossRef]
- Kim, S.Y.; Hong, K.; Kim, K.; Yu, H.K.; Kim, W.-K.; Lee, J.-L. Effect of N2, Ar, and O2 plasma treatments on surface properties of metals. J. Appl. Phys. 2008, 103, 076101. [Google Scholar] [CrossRef]
- Rubow, K.L.; Jha, S. Sintered metal microfiltration media. In Proceedings of the Seventeenth Membrane Technology/Separations Planning Conference, Newton, MA, USA, 6–7 December 1999. [Google Scholar]


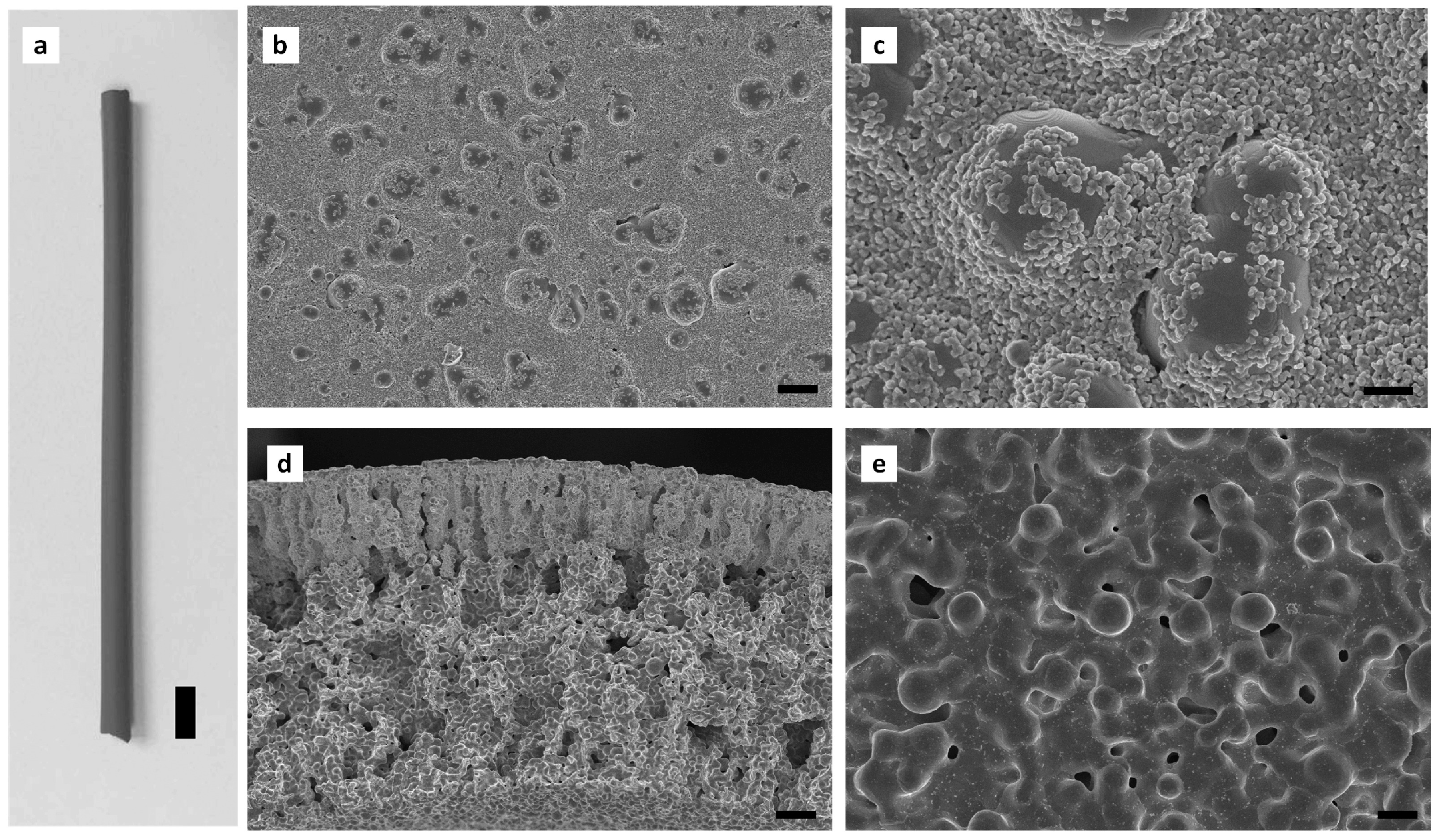

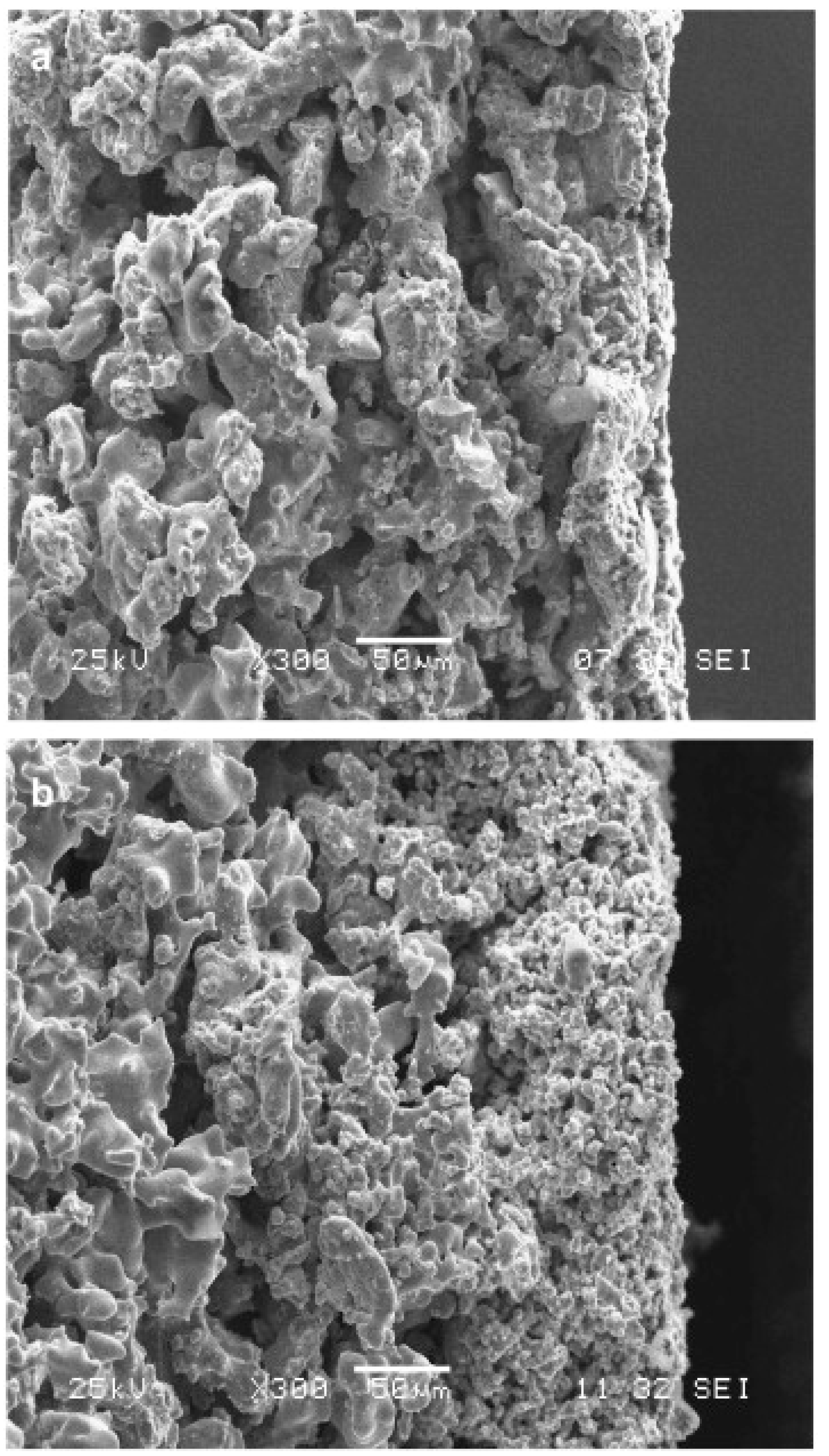



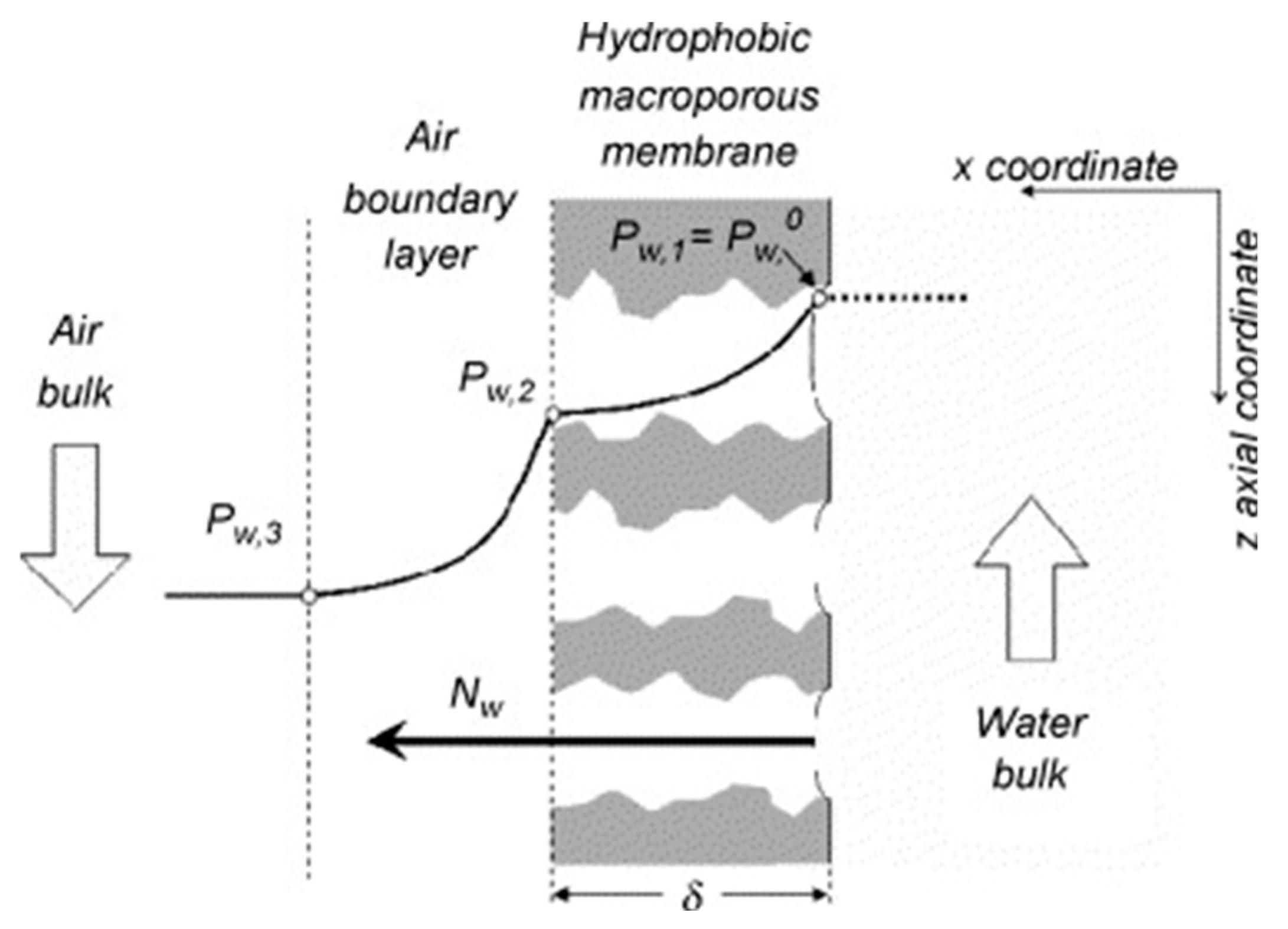

| Pore Size | Techniques | Advantages | Disadvantages |
|---|---|---|---|
| >100 µm | Casting [9,22] | Close control of the pore size distribution. | Inadequate interconnectivity of the pores. |
| Electroplating [22,23] | High efficiency for the fast processing of rough metal coatings. | Required to use the surface as an electrode, which leads to pore filling and clogging within porous structures, thus substantially reducing the surface porosity and pore density. | |
| Chemical vapour deposition (CVD) [9,24,25,26] | Can produce a thin imprint that accurately follows the topography and morphology of the substrate. | Usually only used to coat thin layers of pure metals onto the substrate. | |
| 1–100 µm | Thermal sintering [27,28,29,30,31,32] | Mature technology, easily scale up, cheap to process. | Low pore connectivity and limited porosity. |
| 50 nm–1 µm | Template-directed synthesis [33,34] | Can deposit metals onto a template structure of the desired pore size (e.g., colloidal arrays). | The uniform deposition of metals into colloidal arrays is challenging. |
| De-alloying [8,35,36,37,38,39] | Could lead to very homogeneous structures with narrow pore size distribution. | Difficult to form ultra-thin films of fine grain size alloys. | |
| Electro-spinning [40,41,42,43,44,45,46] | High up-scalability and low cost. | Mechanical strength needs to be enhanced by post-treatments. | |
| Wet casting/coating [47,48,49] | Easy to implement. | Relatively large pores (~1 µm) may exist in the final membranes. | |
| Ink-jet printing [50,51,52,53,54,55,56,57,58,59] | Cost effective, form multi-material components, precisely fabricate intricate layers, able to cover 3D surfaces. | Need post-treatments, still immature. | |
| Electrical sintering [60,61] | Creates finer structures than thermal sintering. | Only can achieve very thin films (<250 nm). | |
| 1 nm–50 nm | Electroless deposition [62,63,64,65,66,67] | Highly controllable structures with nano-scale pore distribution, able to simultaneously co-deposit multiple metals. | Very low deposition rate, a careful analytical control of the plating bath is required, high cost. |
| Block co-polymer (BCP) [68,69,70,71] | Fine control of the nanoparticle distribution, can result in highly crystallised and ordered structures. | Using expensive BCPs prohibits their expansion to a large scale. |
| Manufacturer | Trademark/Brand | Material | Technique | Configuration | Pore Size (µm) | Main Applications |
|---|---|---|---|---|---|---|
| GKN | SIKA-R…IS [96] | SS, nickel-based alloys, Ti | Cold isostatic pressing–sintering | Tubular | 0.5–200 |
|
| GKN | SIKA-R…AS [96] | SS, nickel-based alloys | Coating–sintering | Tubular and disc | 0.1–3 |
|
| GKN | SIKA-R…AX [97] | SS, nickel-based alloys, Ti | Co-axial pressing–sintering | Disc, cylinder, plate, cone | 0.1–200 |
|
| GKN | SIKA-FIL [98] | SS, FeCrAl alloy | Powder metallurgical process–soft sintering | Sheet | 1–100 |
|
| GKN | SIKA-B [99] | Bronze | Moulding–sintering | Disc, cylinder, plate, cone | 8–200 |
|
| Pall | PMM® [32] | SS | Sintering | Tubular | 2–25 |
|
| Pall | PSS® [100] | SS | Sintering | Tubular | 5–55 |
|
| Pall | AccuSep™ [101] | SS, nickel-based alloys, ZrO2 | Sintering or coating-sintering | Tubular | 0.1–5 |
|
| Mott [76] | - | SS, nickel-based alloys, Ti | Sintering | Tubular | 0.2–100 |
|
| Porvair Filtration | Sinterflo® [29] | SS, nickel-based alloys, FeCrAl Alloy, Bronze | Sintering | Cylindrical | 3–50 |
|
| Graver Technologies, LLC | Scepter® [102] | TiO2/SS | Coating–sintering | Tubular | 0.1 or 0.02 |
|
| Hyflux | FerroCep® [103] | TiO2/SS | Coating–sintering | Tubular | 0.1 or 0.02 |
|
| Metalmembranes [104] | - | Metal oxide/Ti or Al | Plasma electrolytic oxidation–electrochemical machining | Plate | 0.01–0.15 |
|
| Sterlitech | Sterlitech™ [105] | Ag | Sintering | Disc | 0.2–5 |
|
| Advanced Material Solutions (AMS) [106] | - | Ti | Coating–sintering | Tubular | 0.05–5 |
|
| AMS [106] | - | Ti | Coating–sintering | Flat sheet | 0.05–20 |
|
| AMS | DuraSter© [30] | SS, high nickel alloys | Coating–sintering | Tubular | - |
|
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhu, B.; Duke, M.; Dumée, L.F.; Merenda, A.; Des Ligneris, E.; Kong, L.; Hodgson, P.D.; Gray, S. Short Review on Porous Metal Membranes—Fabrication, Commercial Products, and Applications. Membranes 2018, 8, 83. https://doi.org/10.3390/membranes8030083
Zhu B, Duke M, Dumée LF, Merenda A, Des Ligneris E, Kong L, Hodgson PD, Gray S. Short Review on Porous Metal Membranes—Fabrication, Commercial Products, and Applications. Membranes. 2018; 8(3):83. https://doi.org/10.3390/membranes8030083
Chicago/Turabian StyleZhu, Bo, Mikel Duke, Ludovic F. Dumée, Andrea Merenda, Elise Des Ligneris, Lingxue Kong, Peter D. Hodgson, and Stephen Gray. 2018. "Short Review on Porous Metal Membranes—Fabrication, Commercial Products, and Applications" Membranes 8, no. 3: 83. https://doi.org/10.3390/membranes8030083
APA StyleZhu, B., Duke, M., Dumée, L. F., Merenda, A., Des Ligneris, E., Kong, L., Hodgson, P. D., & Gray, S. (2018). Short Review on Porous Metal Membranes—Fabrication, Commercial Products, and Applications. Membranes, 8(3), 83. https://doi.org/10.3390/membranes8030083









