Recent Developments of Graphene Oxide-Based Membranes: A Review
Abstract
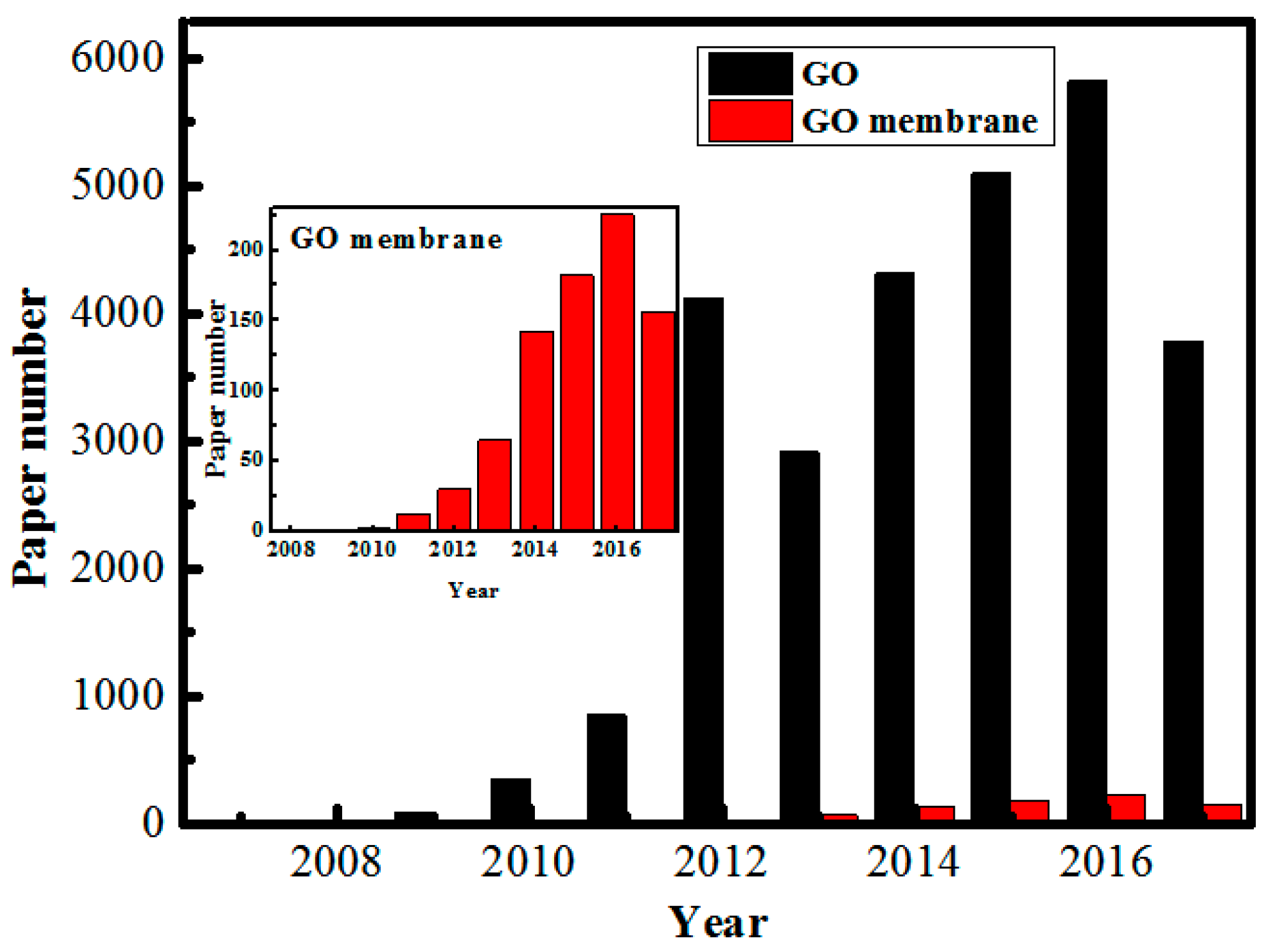
1. Introduction
2. Preparation and Characterization of GO
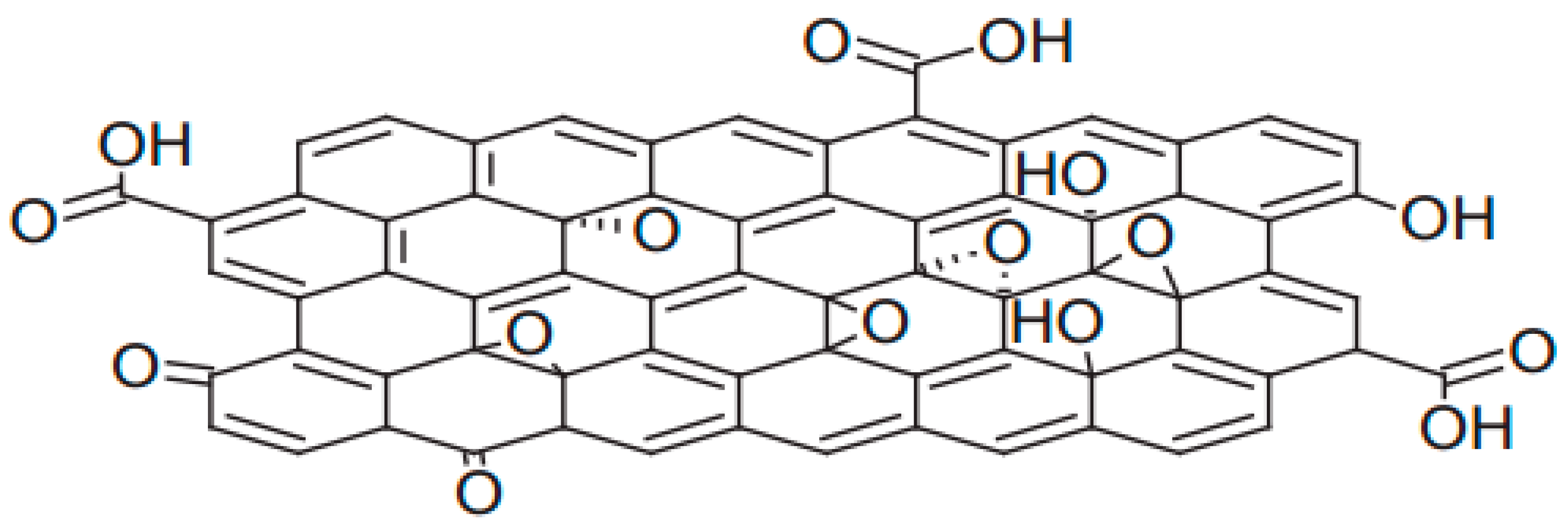
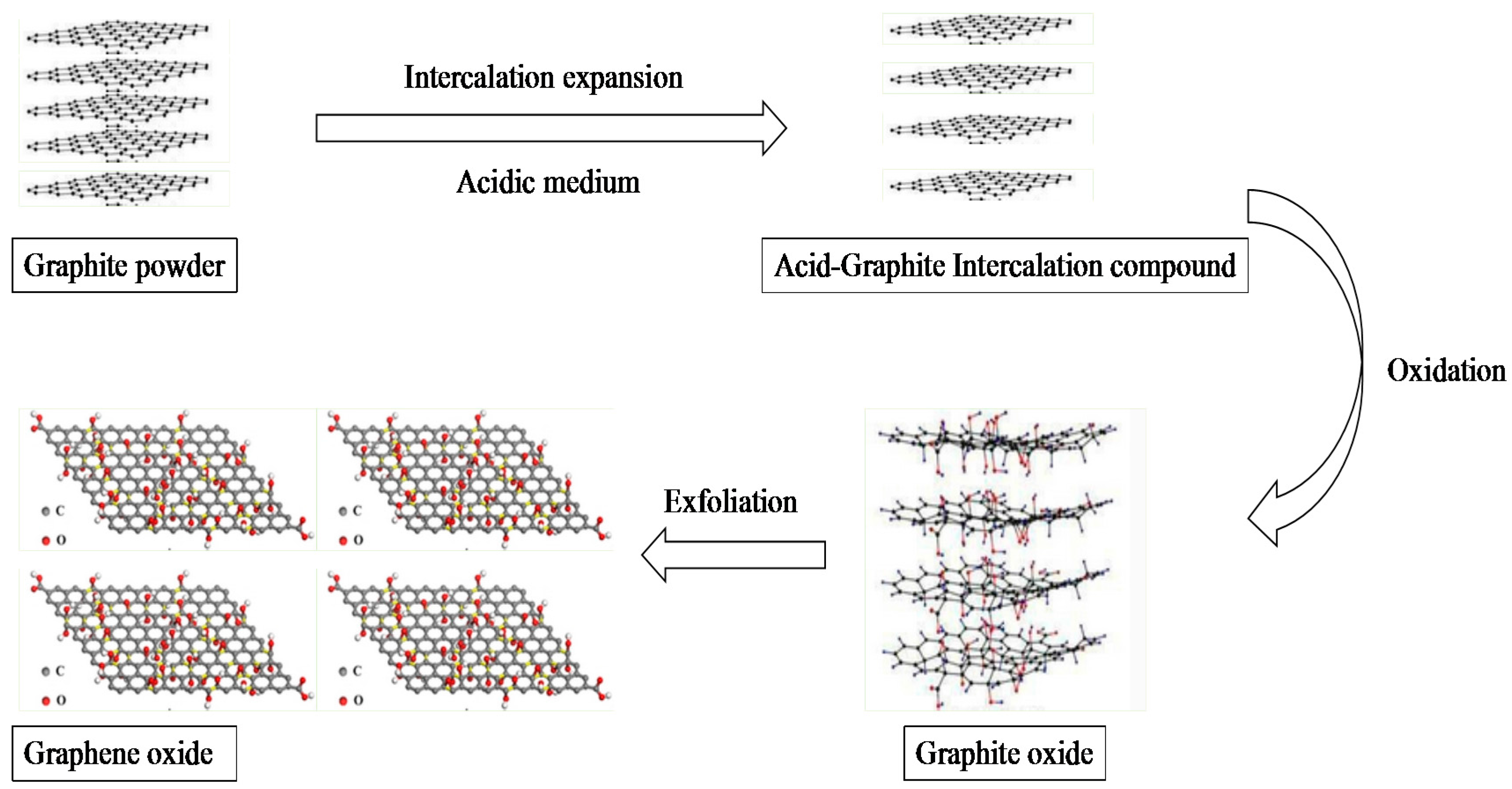
2.1. Preparation of GO
2.2. Characterization of GO
3. GO-Based Membranes
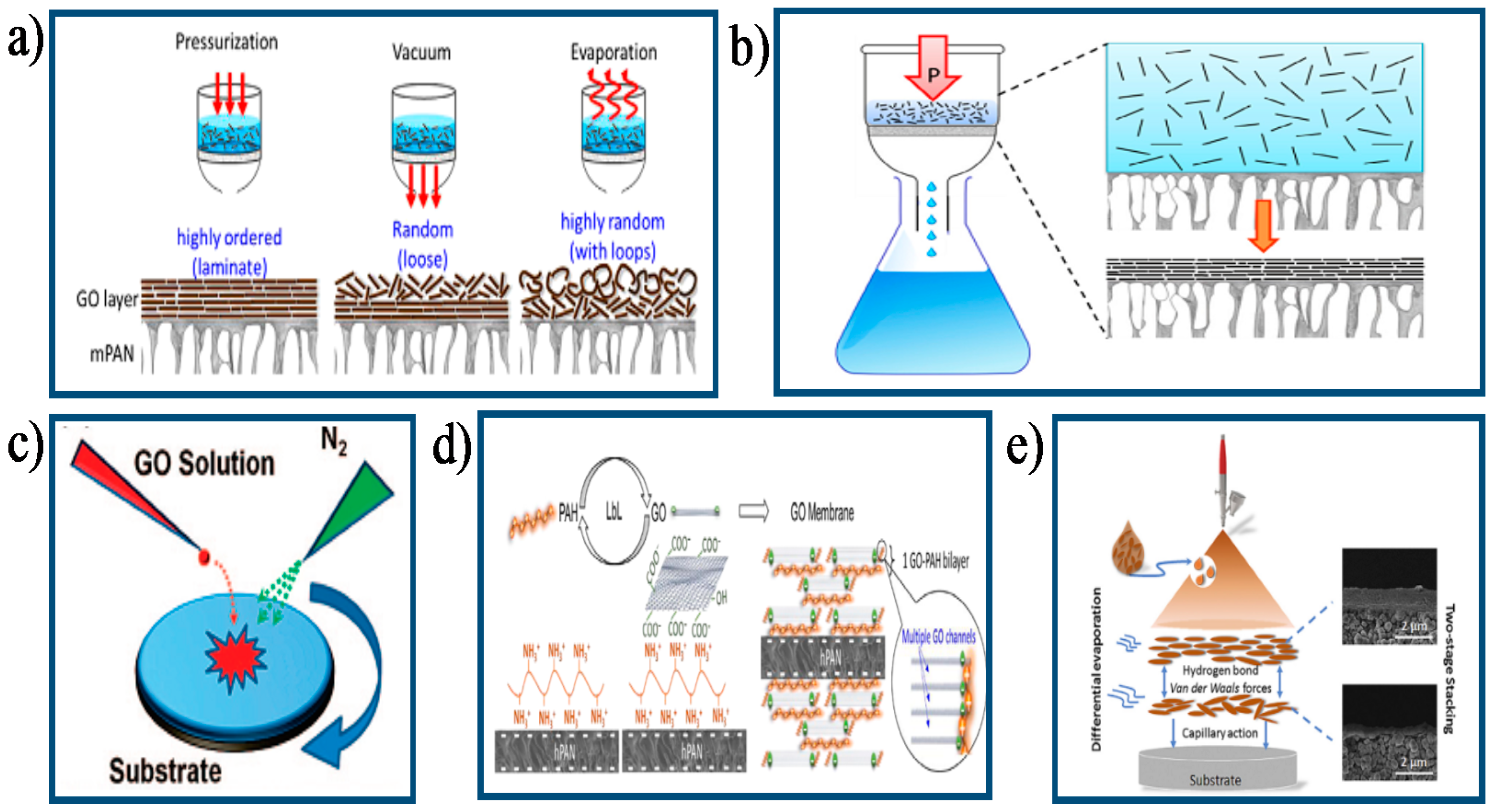
3.1. Preparation Methods of GO Membranes
3.1.1. Filtration-Assisted Method
3.1.2. Casting/Coating-Assisted Method
3.1.3. Layer-by-Layer Assembly Method
3.1.4. Other Methods
3.2. Characterization of GO Membranes
3.3. Types of GO-Based Membranes
3.3.1. Free-Standing GO Membranes
3.3.2. Supported-GO Membranes
3.3.3. GO-Modified Composite Membranes
4. Enhanced Separation Performance of GO Membrane
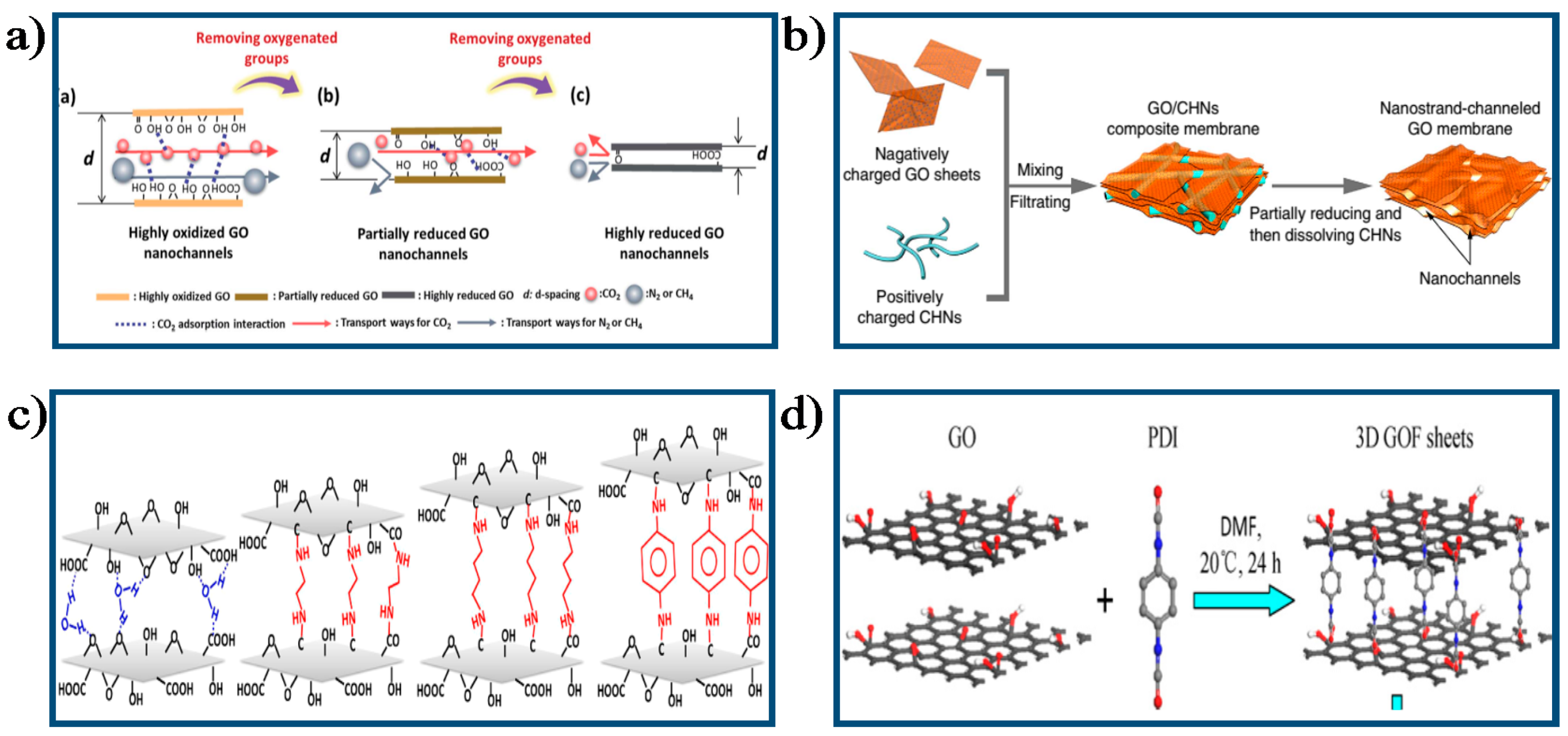
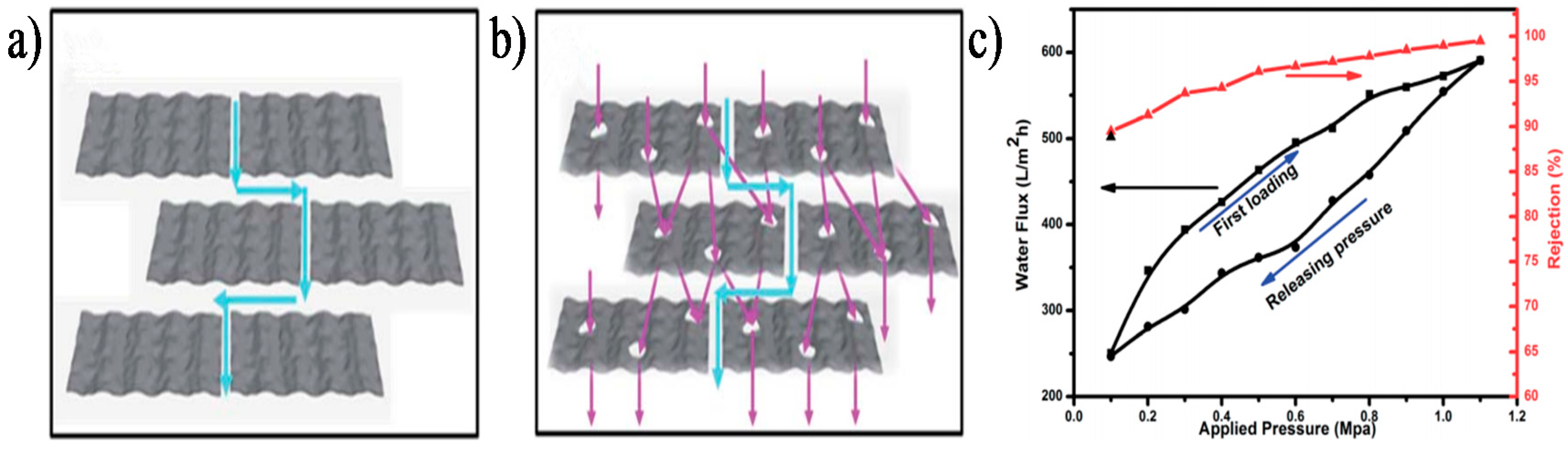
4.1. Physical Approach for Improving Separation Performance of GO Membrane
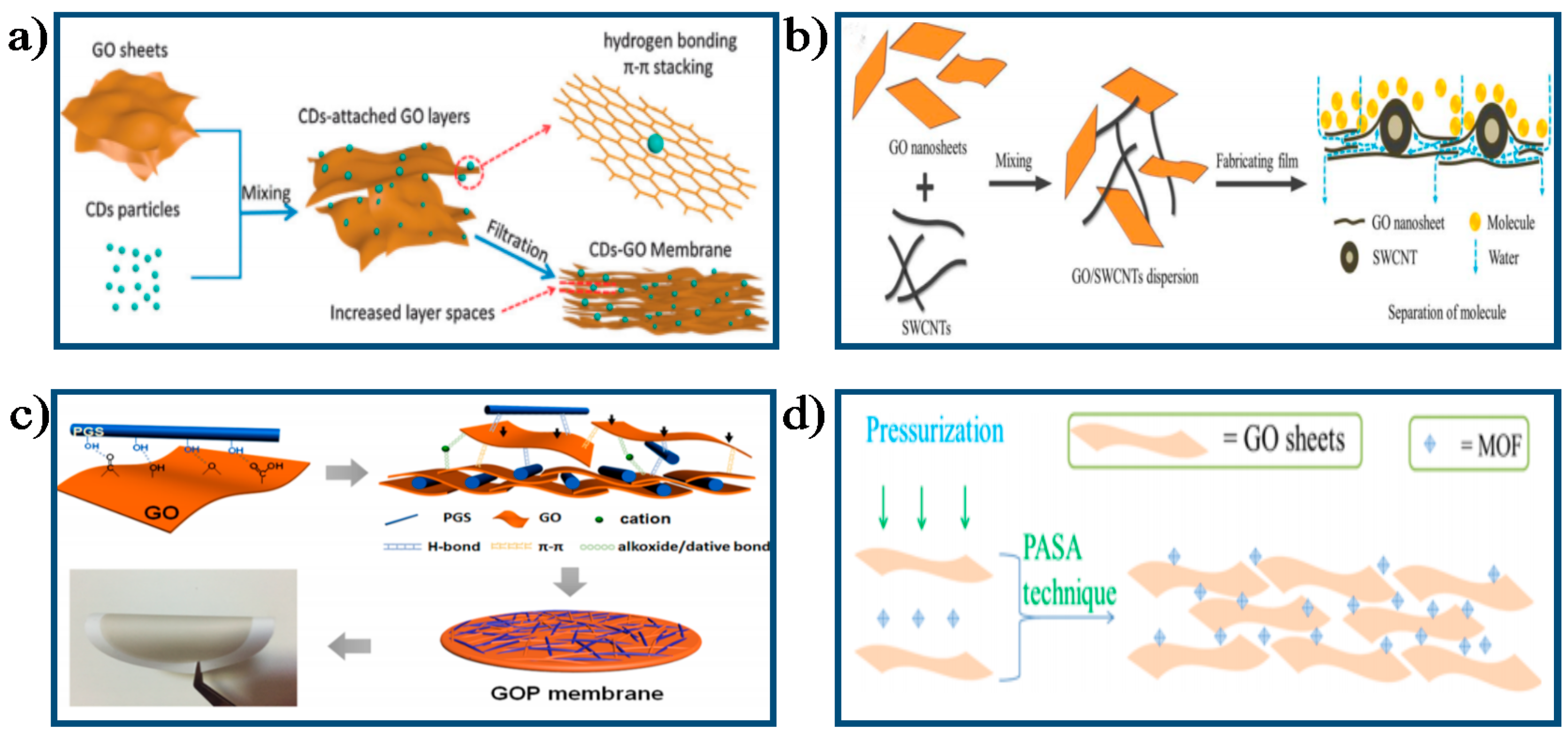
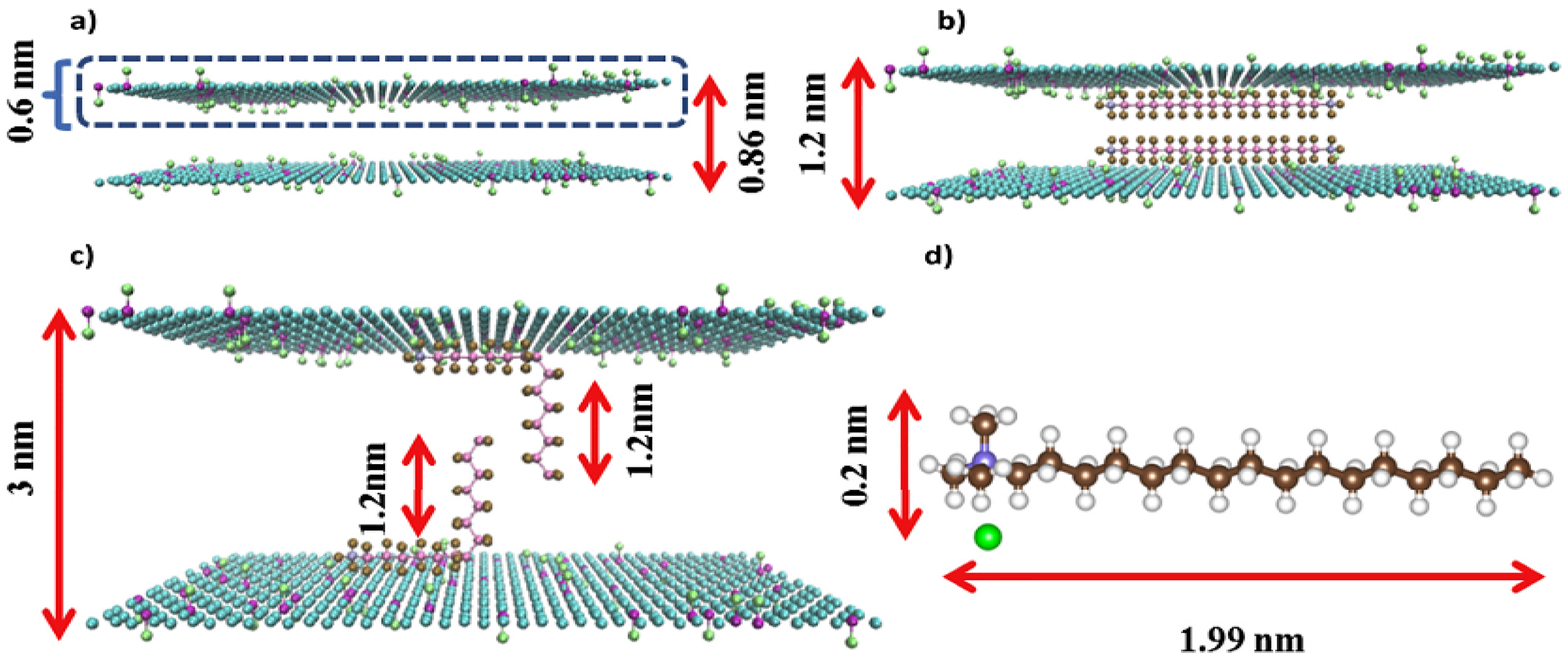
4.2. Chemical Approach for Improving Separation Performance of GO Membrane
4.3. Other Approach
5. Advanced Aqueous Stability and Mechanical Strength of GO Membranes
6. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Gin, D.L.; Noble, R.D. Designing the Next Generation of Chemical Separation Membranes. Science 2011, 332, 674–676. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.P.; Arnot, T.C.; Mattia, D. A review of reverse osmosis membrane materials for desalination—Development to date and future potential. J. Membr. Sci. 2011, 370, 1–22. [Google Scholar] [CrossRef]
- Geise, G.M.; Park, H.B.; Sagle, A.C.; Freeman, B.D.; McGrath, J.E. Water permeability and water/salt selectivity tradeoff in polymers for desalination. J. Membr. Sci. 2011, 369, 130–138. [Google Scholar] [CrossRef]
- Humplik, T.; Lee, J.; O Hern, S.C.; Fellman, B.A.; Baig, M.A.; Hassan, S.F.; Atieh, M.A.; Rahman, F.; Laoui, T.; Karnik, R.; Wang, E.N. Nanostructured materials for water desalination. Nanotechnology 2011, 22, 292001. [Google Scholar] [CrossRef] [PubMed]
- De Volder, M.F.L.; Tawfick, S.H.; Baughman, R.H.; Hart, A.J. Carbon Nanotubes: Present and Future Commercial Applications. Science 2013, 339, 535–539. [Google Scholar] [CrossRef] [PubMed]
- Nair, R.R.; Wu, H.A.; Jayaram, P.N.; Grigorieva, I.V.; Geim, A.K. Unimpeded permeation of water through helium-leak-tight graphene-Based Membranes. Science 2012, 335, 442–444. [Google Scholar] [CrossRef] [PubMed]
- Paul, D.R. Creating New Types of Carbon-Based Membranes. Science 2012, 335, 413–414. [Google Scholar] [CrossRef] [PubMed]
- Kalra, A.; Garde, S.; Hummer, G. Osmotic water transport through carbon nanotube membranes. Proc. Natl. Acad. Sci. USA 2003, 100, 10175–10180. [Google Scholar] [CrossRef] [PubMed]
- Huang, H.; Ying, Y.; Peng, X. Graphene oxide nanosheet: An emerging star material for novel separation membranes. J. Mater. Chem. A 2014, 2, 13772–13782. [Google Scholar] [CrossRef]
- Koenig, S.P.; Wang, L.; Pellegrino, J.; Bunch, J.S. Selective molecular sieving through porous graphene. Nat. Nanotechnol. 2012, 7, 728–732. [Google Scholar] [CrossRef] [PubMed]
- Garaj, S.; Hubbard, W.; Reina, A.; Kong, J.; Branton, D.; Golovchenko, J.A. Graphene as a subnanometre trans-electrode membrane. Nature 2010, 467, 190–193. [Google Scholar] [CrossRef] [PubMed]
- Brodie, B.C. On the Atomic Weight of Graphite. Philos. Trans. R. Soc. Lond. 1859, 149, 249–259. [Google Scholar] [CrossRef]
- Staudenmaier, L. Verfahren zur Darstellung der Graphitsaure. Eur. J. Inorg. Chem. 1898, 31, 1481–1487. [Google Scholar] [CrossRef]
- Hummers, W.S.; Offeman, R.E. Preparation of Graphitic Oxide. J. Am. Chem. Soc. 1958, 80, 1339. [Google Scholar] [CrossRef]
- Peng, L.; Xu, Z.; Liu, Z.; Wei, Y.; Sun, H.; Li, Z.; Zhao, X.; Gao, C. An iron-based green approach to 1-h production of single-layer graphene oxide. Nat. Commun. 2015, 6, 5716. [Google Scholar] [CrossRef] [PubMed]
- Marcano, D.C.; Kosynkin, D.V.; Berlin, J.M.; Sinitskii, A.; Sun, Z.; Slesarev, A.; Alemany, L.B.; Lu, W.; Tour, J.M. Improved Synthesis of Graphene Oxide. ACS Nano 2010, 4, 4806–4814. [Google Scholar] [CrossRef] [PubMed]
- Kovtyukhova, N.I.; Ollivier, P.J.; Martin, B.R.; Mallouk, T.E.; Chizhik, S.A.; Buzaneva, E.V.; Gorchinskiy, A.D. Layer-by-Layer Assembly of Ultrathin Composite Films from Micron-Sized Graphite Oxide Sheets and Polycations. Chem. Mater. 1999, 11, 771–778. [Google Scholar] [CrossRef]
- Guo, H.; Wang, X.; Qian, Q.; Wang, F.; Xia, X. A Green Approach to the Synthesis of Graphene Nanosheets. ACS Nano 2009, 3, 2653–2659. [Google Scholar] [CrossRef] [PubMed]
- Shearer, C.J.; Slattery, A.D.; Stapleton, A.J.; Shapter, J.G.; Gibson, C.T. Accurate thickness measurement of grapheme. Nanotechnology 2016, 27, 125704. [Google Scholar] [CrossRef] [PubMed]
- Lerf, A.; He, H.Y.; Forster, M.; Klinowski, J. Structure of graphite oxide revisited. J. Phys. Chem. B 1998, 102, 4477–4482. [Google Scholar] [CrossRef]
- Szabó, T.; Szeri, A.; Dékány, I. Composite graphitic nanolayers prepared by self-assembly between finely dispersed graphite oxide and a cationic polymer. Carbon N. Y. 2005, 43, 87–94. [Google Scholar] [CrossRef]
- Huang, H.; Mao, Y.; Ying, Y.; Liu, Y.; Sun, L.; Peng, X. Salt concentration, pH and pressure controlled separation of small molecules through lamellar graphene oxide membranes. Chem. Commun. 2013, 49, 5963–5965. [Google Scholar] [CrossRef] [PubMed]
- Obata, S.; Saiki, K.; Taniguchi, T.; Ihara, T.; Kitamura, Y.; Matsumoto, Y. Graphene Oxide: A Fertile Nanosheet for Various Applications. J. Phys. Soc. Jpn. 2015, 84, 1–18. [Google Scholar] [CrossRef]
- Shen, J.; Zhang, M.; Liu, G.; Jin, W. Facile tailoring of the two-dimensional graphene oxide channels for gas separation. RSC Adv. 2016, 6, 54281–54285. [Google Scholar] [CrossRef]
- Kim, H.W.; Yoon, H.W.; Yoon, S.; Yoo, B.M.; Ahn, B.K.; Cho, Y.H.; Shin, H.J.; Yang, H.; Paik, U.; Kwon, S.; et al. Selective Gas Transport Through Few-Layered Graphene and Graphene Oxide Membranes. Science 2013, 342, 91–95. [Google Scholar] [CrossRef] [PubMed]
- Hu, M.; Mi, B. Enabling Graphene Oxide Nanosheets as Water Separation Membranes. Environ. Sci. Technol. 2013, 47, 3715–3723. [Google Scholar] [CrossRef] [PubMed]
- Feng, B.; Xu, K.; Huang, A. Covalent synthesis of three-dimensional graphene oxide framework (GOF) membrane for seawater desalination. Desalination 2016, 394, 123–130. [Google Scholar] [CrossRef]
- Hung, W.; An, Q.; De Guzman, M.; Lin, H.; Huang, S.; Liu, W.; Hu, C.; Lee, K.; Lai, J. Pressure-assisted self-assembly technique for fabricating composite membranes consisting of highly ordered selective laminate layers of amphiphilic graphene oxide. Carbon N. Y. 2014, 68, 670–677. [Google Scholar] [CrossRef]
- Jiang, Y.; Biswas, P.; Fortner, J.D. A review of recent developments in graphene-enabled membranes for water treatment. Environ. Sci. Water Res. Technol. 2016, 2, 915–922. [Google Scholar] [CrossRef]
- Yoon, H.W.; Cho, Y.H.; Park, H.B. Graphene-based membranes: Status and prospects. Philos. Trans. A Math. Phys. Eng. Sci. 2016, 374, 1–23. [Google Scholar] [CrossRef] [PubMed]
- Amadei, C.A.; Vecitis, C.D. How to Increase the Signal-to-Noise Ratio of Graphene Oxide Membrane Research. J. Phys. Chem. Lett. 2016, 7, 3791–3797. [Google Scholar] [CrossRef] [PubMed]
- An, D.; Yang, L.; Wang, T.; Liu, B. Separation Performance of Graphene Oxide Membrane in Aqueous Solution. Ind. Eng. Chem. Res. 2016, 55, 4803–4810. [Google Scholar] [CrossRef]
- Liu, G.; Jin, W.; Xu, N. Two-Dimensional-Material Membranes: A New Family of High-Performance Separation Membranes. Angew. Chem. Int. Ed. 2016, 55, 13384–13397. [Google Scholar] [CrossRef] [PubMed]
- Perreault, F.; Fonseca, D.F.A.; Elimelech, M. Environmental applications of graphene-based nanomaterials. Chem. Soc. Rev. 2015, 44, 5861–5896. [Google Scholar] [CrossRef] [PubMed]
- Mahmoud, K.A.; Mansoor, B.; Mansour, A.; Khraisheh, M. Functional graphene nanosheets: The next generation membranes for water desalination. Desalination 2015, 356, 208–225. [Google Scholar] [CrossRef]
- You, Y.; Sahajwalla, V.; Yoshimura, M.; Joshi, R.K. Graphene and graphene oxide for desalination. Nanoscale 2015, 8, 117–119. [Google Scholar] [CrossRef] [PubMed]
- Hegab, H.M.; Zou, L. Graphene oxide-assisted membranes: Fabrication and potential applications in desalination and water purification. J. Membr. Sci. 2015, 484, 95–106. [Google Scholar] [CrossRef]
- Liu, G.; Jin, W.; Xu, N. Graphene-based membranes. Chem. Soc. Rev. 2015, 44, 516–553. [Google Scholar] [CrossRef] [PubMed]
- Mi, B. Graphene Oxide Membranes for Ionic and Molecular Sieving. Science 2014, 343, 740–742. [Google Scholar] [CrossRef] [PubMed]
- Hofmann, U.; König, E. Untersuchungen uber Graphitoxyd. Z. Anorg. Allg. Chem. 1937, 234, 311–336. [Google Scholar] [CrossRef]
- Joshi, R.K.; Carbone, P.; Wang, F.C.; Kravets, V.G.; Su, Y.; Grigorieva, I.V.; Wu, H.A.; Geim, A.K.; Nair, R.R. Precise and Ultrafast Molecular Sieving Through Graphene Oxide Membranes. Science 2014, 343, 752–754. [Google Scholar] [CrossRef] [PubMed]
- Piner, R.D.; Ruoff, R.S.; Stankovich, S.; Zimney, E.J.; Evmenenko, G.; Dikin, D.A.; Dommett, G.H.B.; Nguyen, S.T. Preparation and characterization of graphene oxide paper. Nature 2007, 448, 457–460. [Google Scholar]
- Tsou, C.; An, Q.; Lo, S.; De Guzman, M.; Hung, W.; Hu, C.; Lee, K.; Lai, J. Effect of microstructure of graphene oxide fabricated through different self-assembly techniques on 1-butanol dehydration. J. Membr. Sci. 2015, 477, 93–100. [Google Scholar] [CrossRef]
- Shen, J.; Zhang, M.; Liu, G.; Guan, K.; Jin, W. Size effects of graphene oxide on mixed matrix membranes for CO2 separation. AIChE J. 2016, 62, 2843–2852. [Google Scholar] [CrossRef]
- Wang, J.; Zhang, P.; Liang, B.; Liu, Y.; Xu, T.; Wang, L.; Cao, B.; Pan, K. Graphene Oxide as an Effective Barrier on a Porous Nanofibrous Membrane for Water Treatment. ACS Appl. Mater. Interfaces 2016, 8, 6211–6218. [Google Scholar] [CrossRef] [PubMed]
- Robinson, J.T.; Zalalutdinov, M.; Baldwin, J.W.; Snow, E.S.; Wei, Z.; Sheehan, P.; Houston, B.H. Wafer-scale Reduced Graphene Oxide Films for Nanomechanical Devices. Nano Lett. 2008, 8, 3441–3445. [Google Scholar] [CrossRef] [PubMed]
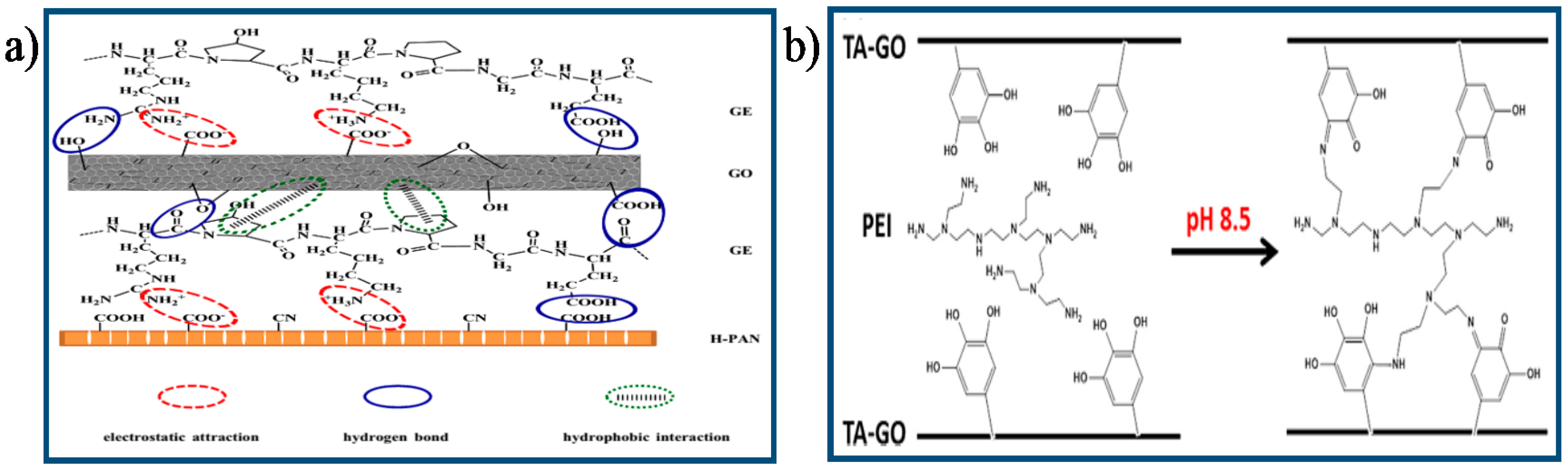
- Hu, M.; Mi, B. Layer-by-layer assembly of graphene oxide membranes via electrostatic interaction. J. Membr. Sci. 2014, 469, 80–87. [Google Scholar] [CrossRef]
- Guan, K.; Shen, J.; Liu, G.; Zhao, J.; Zhou, H.; Jin, W. Spray-evaporation assembled graphene oxide membranes for selective hydrogen transport. Sep. Purif. Technol. 2017, 174, 126–135. [Google Scholar] [CrossRef]
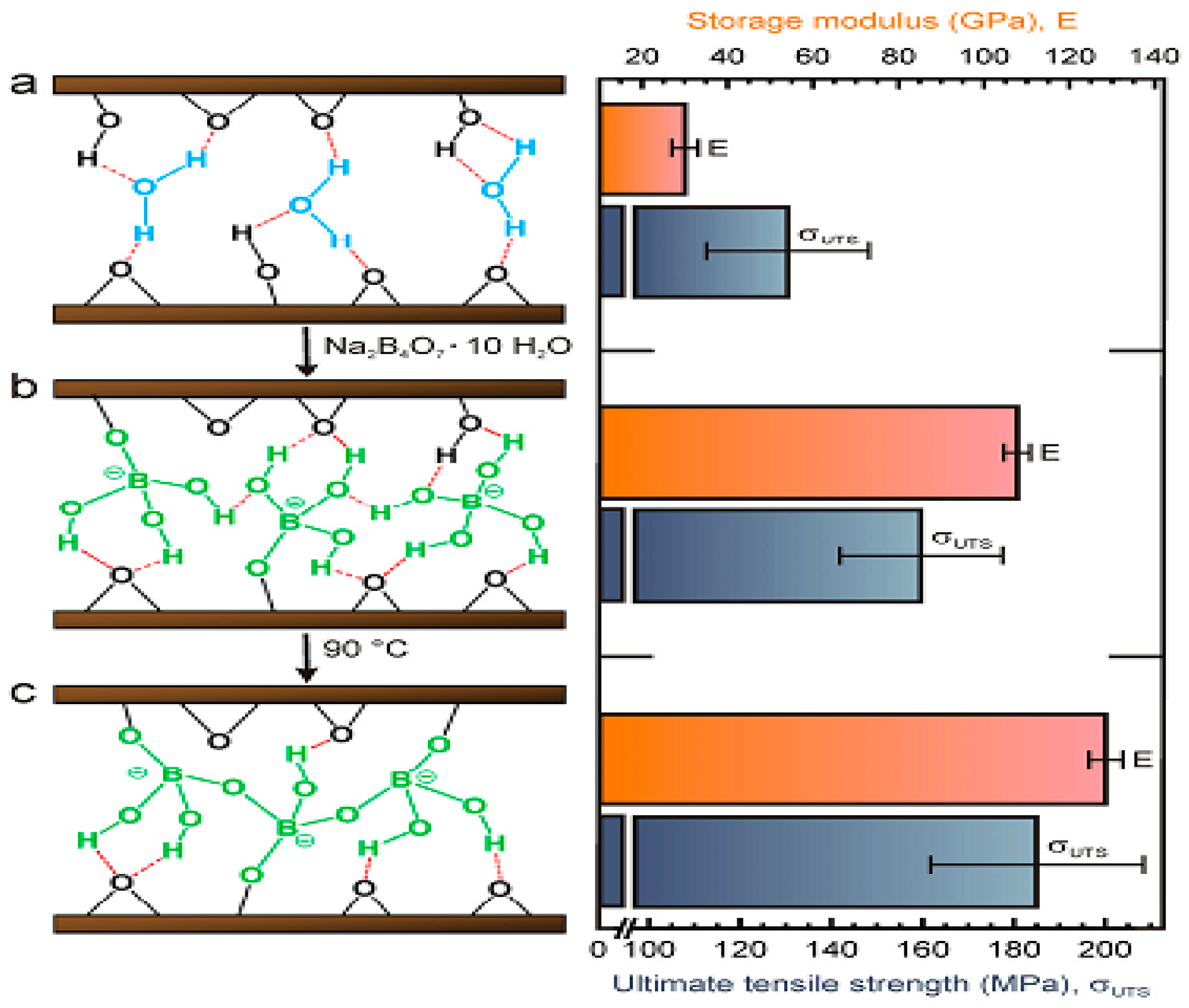
- An, Z.; Compton, O.C.; Putz, K.W.; Brinson, L.C.; Nguyen, S.T. Bio-Inspired Borate Cross-Linking in Ultra-Stiff Graphene Oxide Thin Films. Adv. Mater. 2011, 23, 3842–3846. [Google Scholar] [CrossRef] [PubMed]
- Hu, M.; Zheng, S.; Mi, B. Organic Fouling of graphene oxide membranes and its implications for membrane fouling control in engineered osmosis. Environ. Sci. Technol. 2016, 50, 685–693. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; Yang, Q.; Yang, Y.; Lv, W.; Wen, Y.; Hou, P.; Wang, M.; Cheng, H. Self-Assembled Free-Standing Graphite Oxide Membrane. Adv. Mater. 2009, 21, 3007. [Google Scholar] [CrossRef]
- Wei, Z.; Barlow, D.E.; Sheehan, P.E. The assembly of single-layer graphene oxide and graphene using molecular templates. Nano Lett. 2008, 8, 3141–3145. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Zhang, G.; Bai, X.; Sun, X.; Wang, X.; Wang, E.; Dai, H. Highly conducting graphene sheets and Langmuir–Blodgett films. Nat. Nanotechnol. 2008, 3, 538–542. [Google Scholar] [CrossRef] [PubMed]
- Cote, L.J.; Kim, F.; Huang, J. Langmuir–Blodgett Assembly of Graphite Oxide Single Layers. J. Am. Chem. Soc. 2009, 131, 1043–1049. [Google Scholar] [CrossRef] [PubMed]
- Shen, J.; Liu, G.; Huang, K.; Chu, Z.; Jin, W.; Xu, N. Subnanometer two-dimensional graphene oxide channels for ultrafast gas sieving. ACS Nano 2016, 10, 3398–3409. [Google Scholar] [CrossRef] [PubMed]
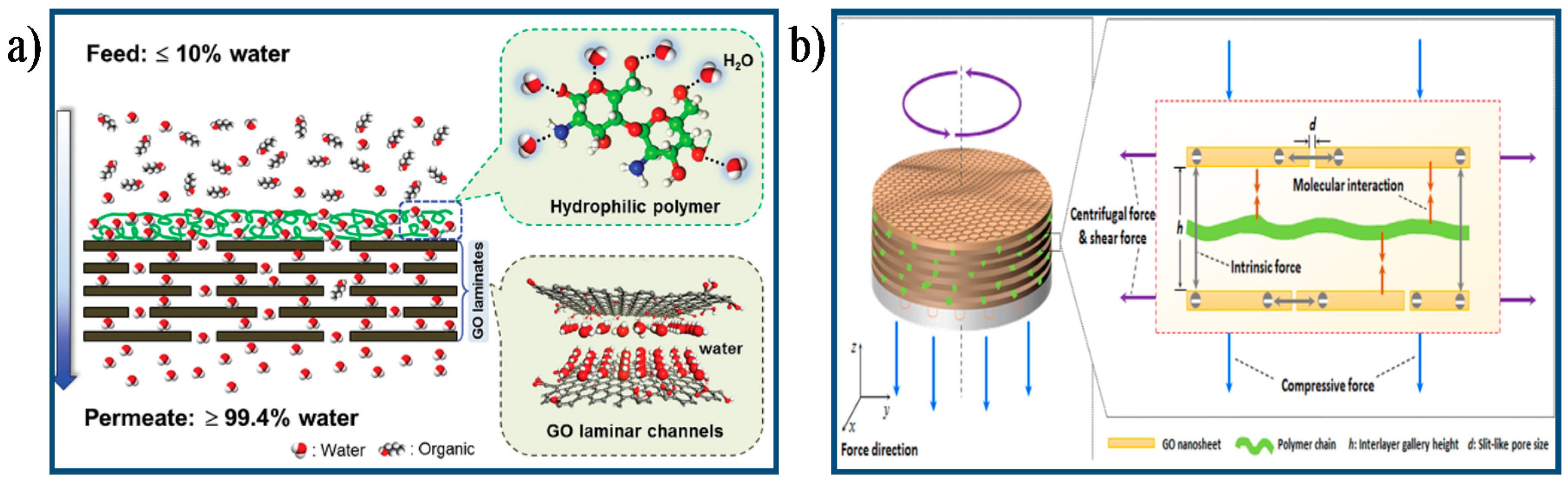
- Akbari, A.; Sheath, P.; Martin, S.T.; Shinde, D.B.; Shaibani, M.; Banerjee, P.C.; Tkacz, R.; Bhattacharyya, D.; Majumder, M. Large-area graphene-based nanofiltration membranes by shear alignment of discotic nematic liquid crystals of graphene oxide. Nat. Commun. 2016, 7, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Zheng, S.; Tu, Q.; Urban, J.J.; Li, S.; Mi, B. Swelling of graphene oxide membranes in aqueous solution: characterization of interlayer spacing and insight into water transport mechanisms. ACS Nano 2017, 11, 6440–6450. [Google Scholar] [CrossRef] [PubMed]
- Huang, K.; Liu, G.; Lou, Y.; Dong, Z.; Shen, J.; Jin, W. A Graphene oxide membrane with highly selective molecular separation of aqueous organic solution. Angew. Chem. Int. Ed. 2014, 53, 6929–6932. [Google Scholar] [CrossRef] [PubMed]
- Jia, Z.; Wang, Y.; Shi, W.; Wang, J. Diamines cross-linked graphene oxide free-standing membranes for ion dialysis separation. J. Membr. Sci. 2016, 520, 139–144. [Google Scholar] [CrossRef]
- Zhao, X.; Su, Y.; Liu, Y.; Li, Y.; Jiang, Z. Free-standing graphene oxide-palygorskite nanohybrid membrane for oil/water separation. ACS Appl. Mater. Interfaces 2016, 8, 8247–8256. [Google Scholar] [CrossRef] [PubMed]
- Cotet, L.C.; Magyari, K.; Todea, M.; Dudescu, M.C.; Danciu, V.; Baia, L. Versatile self-assembled graphene oxide membranes obtained under ambient conditions by using a water-ethanol suspension. J. Mater. Chem. A 2017, 5, 2132–2142. [Google Scholar] [CrossRef]
- Sun, P.; Zhu, M.; Wang, K.; Zhong, M.; Wei, J.; Wu, D.; Xu, Z.; Zhu, H. Selective ion penetration of graphene oxide membranes. ACS Nano 2013, 7, 428–437. [Google Scholar] [CrossRef] [PubMed]
- Tang, Y.P.; Paul, D.R.; Chung, T.S. Free-standing graphene oxide thin films assembled by a pressurized ultrafiltration method for dehydration of ethanol. J. Membr. Sci. 2014, 458, 199–208. [Google Scholar] [CrossRef]
- Wang, S.; Wu, Y.; Zhang, N.; He, G.; Xin, Q.; Wu, X.; Wu, H.; Cao, X.; Guiver, M.D.; Jiang, Z. A highly permeable graphene oxide membrane with fast and selective transport nanochannels for efficient carbon capture. Energy Environ. Sci. 2016, 9, 3107–3112. [Google Scholar] [CrossRef]
- Chu, K.H.; Huang, Y.; Yu, M.; Heo, J.; Flora, J.R.V.; Jang, A.; Jang, M.; Jung, C.; Park, C.M.; Kim, D.Y.H. Evaluation of graphene oxide-coated ultrafiltration membranes for humic acid removal at different pH and conductivity conditions. Sep. Purif. Technol. 2017, 181, 139–147. [Google Scholar] [CrossRef]
- Rao, Z.; Feng, K.; Tang, B.; Wu, P. Surface decoration of amino-functionalized metal-organic framework/graphene oxide composite onto polydopamine-coated membrane substrate for highly efficient heavy metal removal. ACS Appl. Mater. Interfaces 2017, 9, 2594–2605. [Google Scholar] [CrossRef] [PubMed]
- Aba, N.F.D.; Chong, J.Y.; Wang, B.; Mattevi, C.; Li, K. Graphene oxide membranes on ceramic hollow fibers-Microstructural stability and nanofiltration performance. J. Membr. Sci. 2015, 484, 87–94. [Google Scholar] [CrossRef]
- Goh, K.; Setiawan, L.; Wei, L.; Si, R.; Fane, A.G.; Wang, R.; Chen, Y. Graphene oxide as effective selective barriers on a hollow fiber membrane for water treatment process. J. Membr. Sci. 2015, 474, 244–253. [Google Scholar] [CrossRef]
- Lou, Y.; Liu, G.; Liu, S.; Shen, J.; Jin, W. A facile way to prepare ceramic-supported graphene oxide composite membrane via silane-graft modification. Appl. Surf. Sci. 2014, 307, 631–637. [Google Scholar] [CrossRef]
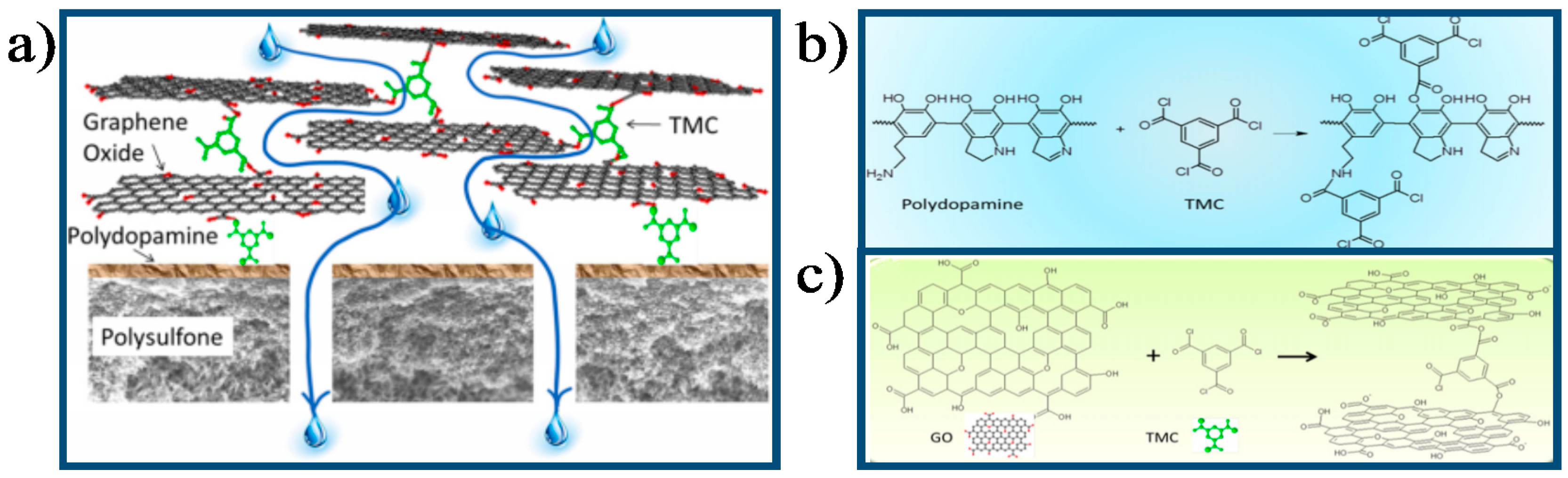
- Xu, K.; Feng, B.; Zhou, C.; Huang, A. Synthesis of highly stable graphene oxide membranes on polydopamine functionalized supports for seawater desalination. Chem. Eng. Sci. 2016, 146, 159–165. [Google Scholar] [CrossRef]
- Salehi, H.; Rastgar, M.; Shakeri, A. Anti-fouling and high water permeable forward osmosis membrane fabricated via layer by layer assembly of chitosan/graphene oxide. Appl. Surf. Sci. 2017, 413, 99–108. [Google Scholar] [CrossRef]
- Lai, G.S.; Lau, W.J.; Goh, P.S.; Ismail, A.F.; Yusof, N.; Tan, Y.H. Graphene oxide incorporated thin film nanocomposite nanofiltration membrane for enhanced salt removal performance. Desalination 2016, 387, 14–24. [Google Scholar] [CrossRef]
- Lee, J.; Chae, H.; Won, Y.J.; Lee, K.; Lee, C.; Lee, H.H.; Kim, I.; Lee, J. Graphene oxide nanoplatelets composite membrane with hydrophilic and antifouling properties for wastewater treatment. J. Membr. Sci. 2013, 448, 223–230. [Google Scholar] [CrossRef]
- Chae, H.; Lee, J.; Lee, C.; Kim, I.; Park, P. Graphene oxide-embedded thin-film composite reverse osmosis membrane with high flux, anti-biofouling, and chlorine resistance. J. Membr. Sci. 2015, 483, 128–135. [Google Scholar] [CrossRef]
- Wang, N.; Ji, S.; Zhang, G.; Li, J.; Wang, L. Self-assembly of graphene oxide and polyelectrolyte complex nanohybrid membranes for nanofiltration and pervaporation. Chem. Eng. J. 2012, 213, 318–329. [Google Scholar] [CrossRef]
- Zhang, Y.; Shen, Q.; Hou, J.; Sutrsna, P.D.; Chen, V. Shear-aligned graphene oxide laminate/Pebax ultrathin composite hollow fiber membranes using a facile dip-coating approach. J. Mater. Chem. A 2017, 17, 7732–7737. [Google Scholar] [CrossRef]
- Yang, M.; Zhao, C.; Zhang, S.; Li, P.; Hou, D. Preparation of graphene oxide modified poly(m-phenylene isophthalamide) nanofiltration membrane with improved water flux and antifouling property. Appl. Surf. Sci. 2017, 394, 149–159. [Google Scholar] [CrossRef]
- Lim, S.; Park, M.J.; Phuntsho, S.; Tijing, L.D.; Nisola, G.M.; Shim, W.G.; Chung, W.J.; Shon, H.K. Dual-layered nanocomposite substrate membrane based on polysulfone/graphene oxide for mitigating internal concentration polarization in forward osmosis. Polymer 2017, 110, 36–48. [Google Scholar] [CrossRef]
- Wang, J.; Wang, Y.; Zhu, J.; Zhang, Y.; Liu, J.; Bruggen, B.V.D. Construction of TiO2@graphene oxide incorporated antifouling nanofiltration membrane with elevated filtration performance. J. Membr. Sci. 2017, 533, 279–288. [Google Scholar] [CrossRef]
- Li, X.; Zhao, C.; Yang, M.; Yang, B.; Hou, D.; Wang, T. Reduced graphene oxide-NH2 modified low pressure nanofiltration composite hollow fiber membranes with improved water flux and antifouling capabilities. Appl. Surf. Sci. 2017, 419, 418–428. [Google Scholar] [CrossRef]
- Qin, D.; Liu, Z.Y.; Bai, H.W.; Sun, D.D. Three-dimensional architecture constructed from a graphene oxide nanosheet-polymer composite for high-flux forward osmosis membranes. J. Mater. Chem. A. 2017, 24, 12183–12192. [Google Scholar] [CrossRef]
- Fan, Y.; Quan, X.; Zhao, H.M.; Chen, S.; Zhang, Y.B.; Zhang, Q. Poly(vinylidene fluoride) hollow-fiber membranes containing silver/graphene oxide dope with excellent filtration performance. J. Appl. Polym. Sci. 2017, 15, 1–9. [Google Scholar] [CrossRef]
- Perreault, F.; Tousley, M.E.; Elimelech, M. Thin-film composite polyamide membranes functionalized with biocidal graphene oxide nanosheets. Environ. Sci. Technol. Lett. 2014, 1, 71–76. [Google Scholar] [CrossRef]
- Zhang, J.Q.; Xue, Q.Z.; Pan, X.L.; Jin, Y.K.; Lu, W.B.; Ding, D.G.; Guo, Q.K. Graphene oxide/polyacrylonitrile fiber hierarchical-structured membrane for ultra-fast microfiltration of oil–water emulsion. Chem. Eng. J. 2017, 307, 643–649. [Google Scholar] [CrossRef]
- Zhang, J.Q.; Pan, X.L.; Xue, Q.Z.; He, D.L.; Zhu, L.; Guo, Q.K. Antifouling hydrolyzed polyacrylonitrile/graphene oxide membrane with spindle-knotted structure for highly effective separation of oil–water emulsion. J. Membr. Sci. 2017, 532, 38–46. [Google Scholar] [CrossRef]
- Zinadini, S.; Zinatizadeh, A.A.; Rahimi, M.; Vatanpour, V.; Zangeneh, H. Preparation of a novel antifouling mixed matrix PES membrane by embedding graphene oxide nanoplates. J. Membr. Sci. 2014, 453, 292–301. [Google Scholar] [CrossRef]
- Ding, R.; Zhang, H.; Li, Y.; Wang, J.; Shi, B.; Mao, H.; Dang, J.; Liu, J. Graphene oxide-embedded nanocomposite membrane for solvent resistant nanofiltration with enhanced rejection ability. Chem. Eng. Sci. 2015, 138, 227–238. [Google Scholar] [CrossRef]
- Huang, H.; Song, Z.; Wei, N.; Shi, L.; Mao, Y.; Ying, Y.; Sun, L.; Xu, Z.; Peng, X. Ultrafast viscous water flow through nanostrand-channelled graphene oxide membranes. Nat. Commun. 2013, 4, 1–2. [Google Scholar] [CrossRef] [PubMed]
- Sun, P.; Wang, K.; Wei, J.; Zhong, M.; Wu, D.; Zhu, H. Effective recovery of acids from iron-based electrolytes using graphene oxide membrane filters. J. Mater. Chem. A. 2014, 2, 7734–7737. [Google Scholar] [CrossRef]
- Sun, P.Z.; Zheng, F.; Zhu, M.; Song, Z.G.; Wang, K.L.; Zhong, M.L.; Wu, D.H.; Little, R.B.; Xu, Z.P.; Zhu, H.W. Selective Trans-Membrane Transport of Alkali and Alkaline Earth Cations through Graphene Oxide Membranes Based on Cation-π Interactions. ACS Nano. 2014, 8, 850–959. [Google Scholar] [CrossRef] [PubMed]
- Tang, M.; Coleman, X. Diffusive transport of two charge equivalent and structurally similar ruthenium complex ions through graphene oxide membranes. Nano Res. 2015, 8, 1128–1138. [Google Scholar]
- Xu, W.L.; Fang, C.; Zhou, F.L.; Song, Z.N.; Liu, Q.L.; Qiao, R.; Yu, M. Self-Assembly: A Facile Way of Forming Ultrathin, High-Performance Graphene Oxide Membranes for Water Purification. Nano Lett. 2017, 5, 2928–2933. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Eftekhari, E.; Zhu, G.; Zhang, X.; Yan, Z.; Li, Q. Graphene oxide membranes with tunable permeability due to embedded carbon dots. Chem. Commun. 2014, 50, 13089–13092. [Google Scholar] [CrossRef] [PubMed]
- Gao, S.-J.; Qin, H.; Liu, P.; Jin, J. SWCNT-intercalated GO ultrathin films for ultrafast separation of molecules. J. Mater. Chem. A 2015, 3, 6649–6654. [Google Scholar] [CrossRef]
- Ying, Y.; Liu, D.; Zhang, W.; Ma, J.; Huang, H.; Yang, Q.; Zhong, C. High-flux graphene oxide membranes intercalated by metal-organic framework with highly selective separation of aqueous organic solution. ACS Appl. Mater. Interfaces 2017, 9, 1710–1718. [Google Scholar] [CrossRef] [PubMed]
- Lian, B.; Deng, J.; Leslie, G.; Bustamante, H.; Sahajwalla, V.; Nishina, Y.; Joshi, R.K. Surfactant modified graphene oxide laminates for filtration. Carbon N. Y. 2017, 116, 240–245. [Google Scholar] [CrossRef]
- Pei, S.; Zhao, J.; Du, J.; Ren, W.; Cheng, H. Direct reduction of graphene oxide films into highly conductive and flexible graphene films by hydrohalic acids. Carbon N. Y. 2010, 48, 4466–4474. [Google Scholar] [CrossRef]
- Liu, H.; Wang, H.; Zhang, X. Facile fabrication of freestanding ultrathin reduced graphene oxide membranes for water purification. Adv. Mater. 2015, 27, 249–254. [Google Scholar] [CrossRef] [PubMed]
- Sun, P.; Chen, Q.; Li, X.; Liu, H.; Wang, K.; Zhong, M.; Wei, J.; Wu, D.; Ma, R.; Sasaki, T.; Zhu, H. Highly efficient quasi-static water desalination using monolayer graphene oxide/titania hybrid laminates. NPG Asia Mater. 2015, 7, 1–8. [Google Scholar] [CrossRef]
- Mei, X.; Meng, X.; Wu, F. Hydrothermal method for the production of reduced graphene oxide. Physica E 2015, 68, 81–86. [Google Scholar] [CrossRef]
- Su, Y.; Kravets, V.G.; Wong, S.L.; Waters, J.; Geim, A.K.; Nair, R.R. Impermeable barrier films and protective coatings based on reduced graphene oxide. Nat. Commun. 2014, 5, 4843. [Google Scholar] [CrossRef] [PubMed]
- Li, D.; Muller, M.B.; Gilje, S.; Kaner, R.B.; Wallace, G.G. Processable aqueous dispersions of graphene nanosheets. Nat. Nanotechnol. 2008, 3, 101–105. [Google Scholar] [CrossRef] [PubMed]
- Zhou, X.; Zhang, J.; Wu, H.; Yang, H.; Zhang, J.; Guo, S. Reducing Graphene Oxide via Hydroxylamine: A Simple and Efficient Route to Graphene. J. Phys. Chem. C 2011, 115, 11957–11961. [Google Scholar] [CrossRef]
- Zhang, J.; Yang, H.; Shen, G.; Cheng, P.; Zhang, J.; Guo, S. Reduction of graphene oxide via l-ascorbic acid. Chem Commun. 2010, 46, 1112–1114. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Gao, B.; Chen, J.; Li, Y.; Creamer, A.E.; Chen, H. Slow-release fertilizer encapsulated by graphene oxide films. Chem. Eng. J. 2014, 255, 107–113. [Google Scholar] [CrossRef]
- Stankovich, S.; Piner, R.D.; Chen, X.; Wu, N.; Nguyen, S.T.; Ruoff, R.S. Stable aqueous dispersions of graphitic nanoplatelets via the reduction of exfoliated graphite oxide in the presence of poly(sodium 4-styrenesulfonate). J. Mater. Chem. 2006, 16, 155–158. [Google Scholar] [CrossRef]
- Stankovich, S.; Dikin, D.A.; Piner, R.D.; Kohlhaas, K.A.; Kleinhammes, A.; Jia, Y.; Wu, Y.; Nguyen, S.T.; Ruoff, R.S. Synthesis of graphene-based nanosheets via chemical reduction of exfoliated graphite oxide. Carbon N. Y. 2007, 45, 1558–1565. [Google Scholar] [CrossRef]
- Wang, L.; Sasaki, T. Titanium Oxide Nanosheets: Graphene Analogues with Versatile Functionalities. Chem. Rev. 2014, 114, 9455–9486. [Google Scholar] [CrossRef] [PubMed]
- Fernandez-Merino, M.J.; Guardia, L.; Paredes, J.I.; Villar-Rodil, S.; Solis-Fernandez, P.; Martinez-Alonso, A.; Tascon, J.M.D. Vitamin C Is an ideal substitute for hydrazine in the reduction of graphene oxide suspensions. J. Phys. Chem. C 2010, 114, 6426–6432. [Google Scholar] [CrossRef]
- Hung, W.; Tsou, C.; De Guzman, M.; An, Q.; Liu, Y.; Zhang, Y.; Hu, C.; Lee, K.; Lai, J. Cross-Linking with Diamine Monomers To Prepare Composite Graphene Oxide-Framework Membranes with Varying d-Spacing. Chem. Mater. 2014, 26, 2983–2990. [Google Scholar] [CrossRef]
- Jia, Z.; Shi, W. Tailoring permeation channels of graphene oxide membranes for precise ion separation. Carbon N. Y. 2016, 101, 290–295. [Google Scholar] [CrossRef]
- Jia, Z.; Wang, Y. Covalently crosslinked graphene oxide membranes by esterification reactions for ions separation. J. Mater. Chem. A 2015, 3, 4405–4412. [Google Scholar] [CrossRef]
- Li, W.; Zhang, Y.; Xu, Z.; Yang, A.; Meng, Q.; Zhang, G. Self-assembled graphene oxide microcapsules with adjustable permeability and yolk-shell superstructures derived from atomized droplets. Chem Commun. 2014, 50, 15867–15869. [Google Scholar] [CrossRef] [PubMed]
- Yuan, Y.; Gao, X.L.; Wei, Y.; Wang, X.Y.; Wang, J.; Zhang, Y.S.; Gao, C.J. Enhanced desalination performance of carboxyl functionalized graphene oxide nanofiltration membranes. Desalination. 2017, 405, 29–39. [Google Scholar] [CrossRef]
- Ying, Y.; Sun, L.; Wang, Q.; Fan, Z.; Peng, X. In-plane mesoporous graphene oxide nanosheet assembled membranes for molecular separation. RSC Adv. 2014, 4, 21425. [Google Scholar] [CrossRef]
- Huang, K.; Liu, G.; Shen, J.; Chu, Z.; Zhou, H.; Gu, X.; Jin, W.; Xu, N. High-efficiency water-transport channels using the synergistic effect of a hydrophilic polymer and graphene Oxide Laminates. Adv. Funct. Mater. 2015, 25, 5809–5815. [Google Scholar] [CrossRef]
- Shen, H.; Wang, N.; Ma, K.; Wang, L.; Chen, G.; Ji, S. Tuning inter-layer spacing of graphene oxide laminates with solvent green to enhance its nanofiltration performance. J. Membr. Sci. 2017, 527, 43–50. [Google Scholar] [CrossRef]
- Yeh, C.N.; Raidongia, K.; Shao, J.; Yang, Q.H.; Huang, J. On the origin of the stability of graphene oxide membranes in water. Nat. Chem. 2014, 7, 166–170. [Google Scholar] [CrossRef] [PubMed]
- Sun, S.; Wang, C.; Chen, M.; Li, M. The mechanism for the stability of graphene oxide membranes in a sodium sulfate solution. Chem. Phys. Lett. 2013, 561–562, 166–169. [Google Scholar] [CrossRef]
- Huang, T.; Zhang, L.; Chen, H.; Gao, C. A cross-linking graphene oxide–polyethyleneimine hybrid film containing ciprofloxacin: One-step preparation, controlled drug release and antibacterial performance. J. Mater. Chem. B 2015, 3, 1605–1611. [Google Scholar] [CrossRef]
- Zhao, J.; Zhu, Y.; Pan, F.; He, G.; Fang, C.; Cao, K.; Xing, R.; Jiang, Z. Fabricating graphene oxide-based ultrathin hybrid membrane for pervaporation dehydration via layer-by-layer self-assembly driven by multiple interactions. J. Membr. Sci. 2015, 487, 162–172. [Google Scholar] [CrossRef]
- Park, S.; Dikin, D.A.; Nguyen, S.T.; Ruoff, R.S. Graphene oxide sheets chemically cross-linked by polyallylamine. J. Phys. Chem. C 2009, 113, 15801–15804. [Google Scholar] [CrossRef]
- Park, S.; Lee, K.; Bozoklu, G.; Cai, W.; Nguyen, S.T.; Ruoff, R.S. Graphene oxide papers modified by divalent ions—Enhancing mechanical properties via chemical cross-linking. ACS Nano 2008, 2, 572–578. [Google Scholar] [CrossRef] [PubMed]
- Lim, M.; Choi, Y.; Kim, J.; Kim, K.; Shin, H.; Kim, J.; Shin, D.M.; Lee, J. Cross-linked graphene oxide membrane having high ion selectivity and antibacterial activity prepared using tannic acid-functionalized graphene oxide and polyethyleneimine. J. Membr. Sci. 2017, 521, 1–9. [Google Scholar] [CrossRef]
- Zheng, S.; Mi, B. Emerging investigators series: Silica-crosslinked graphene oxide membrane and its unique capability in removing neutral organic molecules from water. Environ. Sci. Water Res. Technol. 2016, 2, 717–725. [Google Scholar] [CrossRef]
- Zhao, X.; Zhang, Q.; Hao, Y.; Li, Y.; Fang, Y.; Chen, D. Alternate multilayer films of poly(vinyl alcohol) and exfoliated graphene oxide fabricated via a facial layer-by-layer assembly. Macromolecules 2010, 43, 9411–9416. [Google Scholar] [CrossRef]
- Liu, G.G.; Han, K.; Ye, H.Q.; Zhu, C.Y.; Gao, Y.P.; Liu, Y.; Zhou, Y.H. Graphene oxide/triethanolamine modified titanate nanowires as photocatalytic membrane for water treatment. Chem. Eng. J. 2017, 320, 74–80. [Google Scholar] [CrossRef]
- Zhang, P.; Gong, J.; Zeng, G.; Deng, C.; Yang, H.; Liu, H. Cross-linking to prepare composite graphene oxide-framework membranes with high-flux for dyes and heavy metal ions removal. Chem. Eng. J. 2017, 322, 657–666. [Google Scholar] [CrossRef]
- Yang, E.; Kim, C.M.; Song, J.H.; Ki, H.; Ham, M.H.; Kim, I.S. Enhanced desalination performance of forward osmosis membranes based on reduced graphene oxide laminates coated with hydrophilic polydopamine. Carbon N. Y. 2017, 117, 293–300. [Google Scholar] [CrossRef]
- Kim, S.; Li, X.C.; Ou, R.; Liu, H.; Zhang, X.; Simon, G.P.; Easton, C.D.; Wang, H. Highly crosslinked, chlorine tolerant polymer network entwined graphene oxide membrane for water desalination. J. Mater. Chem. A 2017, 4, 1533–1540. [Google Scholar] [CrossRef]
| Oxidant | Method | Acid | Reaction Time | Interlayer Spacing | C:O Ratio | Note | Reference |
|---|---|---|---|---|---|---|---|
| KClO3 | Brodie | HNO3 | 3–4 days | 5.95 Å | 2.16 | Toxic gas ClO2 | [12] |
| Staudenmaier | HNO3, H2SO4 | 1–10 days | 6.23 Å | 1.85 | Toxic gas ClO2, NOx | [13] | |
| Hofmann | HNO3, H2SO4 | 4 days | – | – | Toxic gas ClO2, NOx | [40] | |
| KMnO4 | Hummers | NaNO3, H2SO4 | ≈2 h | 6.67 Å | 2.25 | Toxic gas NOx, Mn2+ in GO | [14] |
| Modified Hummers | K2S2O8, P2O5, H2SO4 | 8 h | 6.9 Å | 2.3 | – | [17] | |
| Improved Hummers | 9:1 H2SO4/H3PO4 | ≈12 h | 9.3 Å | – | Mn2+ in GO | [16] | |
| K2FeO4 | Iron-based green method | H2SO4 | 1 h | 9.0 Å | 2.2 | Fe3+ in GO | [15] |
| Name | Characterization Method | Characterization Information | Reference |
|---|---|---|---|
| Micromorphology and size of GO | SEM | Lateral size distribution of GO sheets, observe the structural morphology of GO | [15,16] |
| TEM | Morphology of GO (wrinkles), monolayer character of GO sheets | [15,16,17,18] | |
| AFM | Lateral size and thickness of GO sheets | [16,17,18,19] | |
| Thermal stability | TGA | Thermal stability of GO | [15,16] |
| Chemical structure of GO | XPS | Quantitatively analyze the chemistry composition of GO | [15,16,17] |
| ICP-MS | Chemistry composition of GO, identified the metal ion content in GO | [15] | |
| FTIR | Characteristic bands corresponding to oxygen functional groups, confirmed the successful synthesis of GO | [15,16,17,18] | |
| XRD | Crystalline structures of the GO nanosheets, inter-sheet distance of GO, confirmed the successful synthesis of GO | [15,16,17,18] | |
| Raman spectroscopy | Analyze the chemical structure of GO combined with XPS, FTIR, XRD, ICP-MS | [15,16,18] | |
| Electrochemical property | Zeta potential measurement | GO nanosheets are negatively charged over a wide pH range | [22] |
| Method | Description | Note |
|---|---|---|
| Filtration-assisted | Vacuum filtration | Good nanoscale control over the membrane thickness; laminar structure of GO membranes is dictated by the filtration force; highly scalable |
| Pressure filtration | ||
| Casting/coating-based | Spinning-casting/coating | Nonuniform deposition of GO nanosheets; poor control over the membrane thickness; producing highly continuous GO membranes; highly scalable |
| Drop-casting | ||
| Dip-coating | ||
| Spray-coating | ||
| Doctor blade-casting | ||
| LbL assembly | Layer-by-layer assembly | Easily control of the GO layer number, packing, and thickness |
| Others | Hybrid approach | Easily control of the GO assembly, industrial-scalability, rapid throughput. |
| Evaporation-assembled method | Scale-up, easily control of the membrane thickness and size | |
| Templating method | – | |
| Langmuir–Blodgett (LB) assembly | Producing highly uniform, close-packed monolayered GO membrane | |
| Shear-alignment method | Scale-up, industrial-scalability, producing large-area GO membrane, rapid throughput |
| Characterization Method | Characterization Information | Reference |
|---|---|---|
| Surface Zeta potential | Identified the surface charges of membrane | [22] |
| Stress–strain curves | Mechanical stability of the membrane, tensile strength, Young’s modulus | [22] |
| SEM | Surface morphology and cross-section structure | [26] |
| AFM | Surface roughness of membrane, membrane uniformity | [26] |
| CA | Surface hydrophilic or hydrophobic property of membrane | [27] |
| FTIR | Chemical structure of membrane, surface functional groups of membrane | [48] |
| XPS | Quantitatively analyze the elemental compositions of membrane | [48] |
| Raman spectroscopy | Identified the existence of GO in composite membrane | [48] |
| TGA | Thermal stability of membrane | [49] |
| TEM | Surface morphology and cross-section structure | [53] |
| XRD | Crystalline structures, d-spacing of membrane | [58] |
| Integrated quartz crystal microbalance with dissipation and ellipsometry | Accurately measure the d-spacing of GO membranes in an aqueous environment | [57] |
| Types of GO Membrane | Name of GO Membrane | Fabrication Method | Application | Membrane Performance | Reference |
|---|---|---|---|---|---|
| Free-standing | GO membrane | Flow-directed self-assembly | – | Elastic modulus: 32 GPa Tensile strength: 70.7 MPa | [42] |
| GO membrane | Evaporation-driven LbL self-assembly | – | Elastic modulus: 12.7 GPa Tensile strength: 67.7 MPa | [51] | |
| Cross-linked GO membrane | Vacuum filtration | Ion dialysis separation | Elastic modulus: 10.5402 GPa K+/Mg2+ selectivity factor: 7.15 | [59] | |
| GOP nanohybrid membrane | Vacuum filtration | Oil/water separation | Water flux: 1869 L/m2/h Superior anti-oil-fouling | [60] | |
| GO membrane | Self-assembly under ambient condition | – | Tensile strength: 46.20 MPa Elongation: 1.29% Young’s modulus: 5.08 GPa | [61] | |
| GO membrane | Drop-casting | Ion penetration | Entirely blocked heavy-metal salt (e.g., copper sulfate) and organic contaminants (rhodamine B); low rejection of sodium salts | [62] | |
| GO membrane | Pressurized ultrafiltration | Dehydration of 85 wt % ethanol | Water permeability: 13,800 Barrer Selectivity: 227 | [63] | |
| Supported | GO/PES | Spin-casting | Gas separation | CO2 permeability: 8500 Barrer CO2/N2 selectivity: 20 | [25] |
| GOF/Al2O3 | Vacuum filtration | 3.5 wt % seawater desalination | Water flux: 11.4 kg/m2/h Ion rejection: >99.9% | [27] | |
| GO/mPAN | Pressure-assisted | Pervaporation of a 70 wt % IPA/water mixture | Permeation flux: 4137 g/m2/h Separation factor: 1164 | [28] | |
| self-assembly | |||||
| GO/PAN | LbL assembly | Water purification | Water flux: 2.1–5.8 L/m2/h Sucrose rejection: 99% | [47] | |
| GO/Nylon | Shear-alignment method | Water treatment | Water permeability: 71 ± 5 L/m2/bar/h Rejection: organic probe molecules (hydrated radius >5 Å): >90% Monovalent and divalent salts: 30–40% | [56] | |
| GO/PES | Vacuum filtration | Gas separation | CO2 permeance: 650 GPU CO2/CH4 selectivity: 75 | [64] | |
| GO/PES | Vacuum filtration | Humic acid removal | Rejection: 85.3–93.9% Superior antifouling capability | [65] | |
| IRMOF-3/GO/PDA-PSF | Dip-coating | Heavy-metal removal | Water flux: 31 L/m2/h Copper(II) rejection: 90% | [66] | |
| GO/ceramic | Dip-coating | Pervaporation separation of water/ethanol mixtures | Total flux: 461.86 g/m2/h Water recovery: 39.92 wt % | [69] | |
| GO/Al2O3 | Vacuum filtration | 3.5 wt % seawater desalination | Water flux: 48.4 kg/m2/h Ion rejection: ≥99.7% | [70] | |
| GO-modified | GO/PSF | Phase inversion | Water purification | water flux: 353.5 L/m2/bar/h Rejection: Na2SO4 (95.2%); MgSO4 (91.1%); NaCl (59.5%) | [72] |
| GO/PSF | Phase inversion | Water treatment | Water flux: 450 L/m2/h BSA rejection: 99% | [73] | |
| GO/PESc | LbL | Water treatment | Water flux: 7.1 kg/m2/MPa/h Rejection: Mg2+ (92.6%) Na+ (43.2%) | [75] | |
| Self-assembly | |||||
| Pebax/GO/PVDF | Dip-coating | Gas separation | N2 permeance: 9.6 GPU CO2 permeance: 413.3 GPU CO2/N2 seletivity: 43.2 | [76] | |
| GO/H-PAN | Electrospinning | Oil/water separation | Water flux: 3500 L/m2/h Rejection ratio: 99% Superior anti-oil-fouling | [83] | |
| GO/APAN | – | Oil/water separation | Water flux: 10,000 L/m2/h Rejection ratio: ≥98% Superior anti-oil-fouling | [84] | |
| GO/PEI/DPAN | Dip-coating | Solvent resistant NF | Ethanol flux: 10.8 L/m2/h Acetone flux: 15.7 L/m2/h Ethyl acetate flux: 12.9 L/m2/h n-heptane flux: 3.1 L/m2/h PEG(Mw 200) rejection 96.8% | [85] | |
| – | GO/PES | Phase inversion | Water treatment | Water flux: 20.4 kg/m2/h Direct Red 16 rejection: 96% Superior anti-fouling capability | [86] |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ma, J.; Ping, D.; Dong, X. Recent Developments of Graphene Oxide-Based Membranes: A Review. Membranes 2017, 7, 52. https://doi.org/10.3390/membranes7030052
Ma J, Ping D, Dong X. Recent Developments of Graphene Oxide-Based Membranes: A Review. Membranes. 2017; 7(3):52. https://doi.org/10.3390/membranes7030052
Chicago/Turabian StyleMa, Jinxia, Dan Ping, and Xinfa Dong. 2017. "Recent Developments of Graphene Oxide-Based Membranes: A Review" Membranes 7, no. 3: 52. https://doi.org/10.3390/membranes7030052
APA StyleMa, J., Ping, D., & Dong, X. (2017). Recent Developments of Graphene Oxide-Based Membranes: A Review. Membranes, 7(3), 52. https://doi.org/10.3390/membranes7030052




