Perfluorinated Compounds as Test Media for Porous Membranes
Abstract
:1. Introduction
2. Results and Discussion
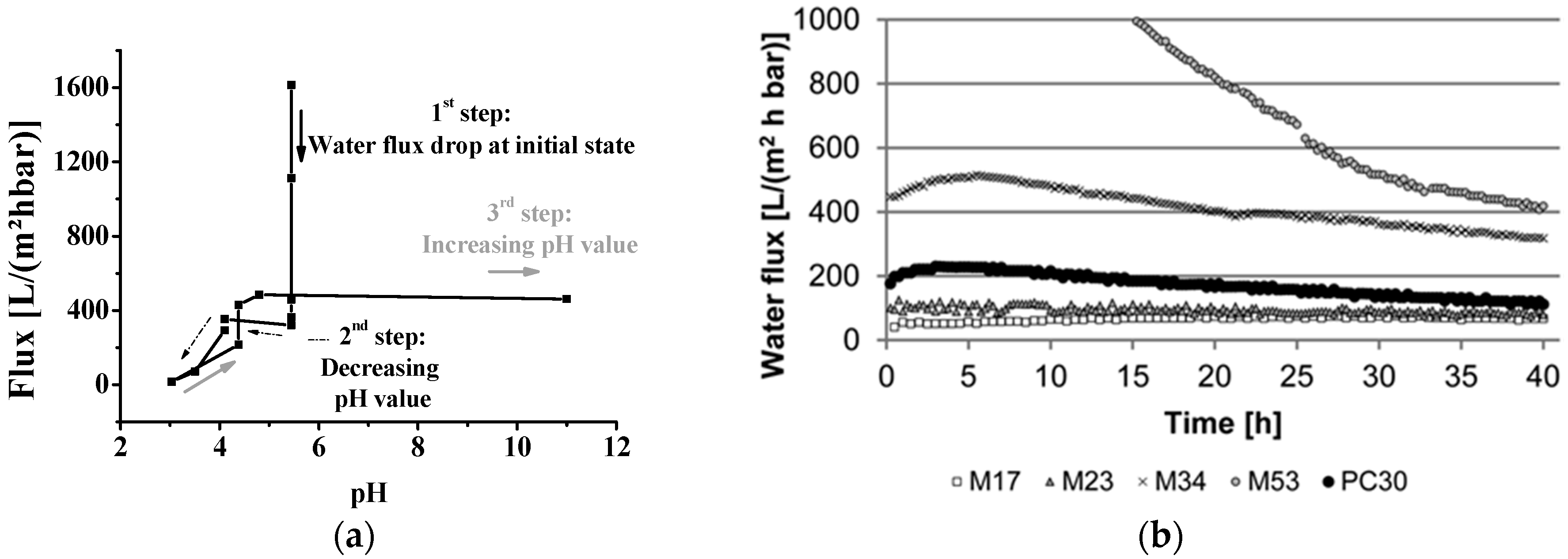
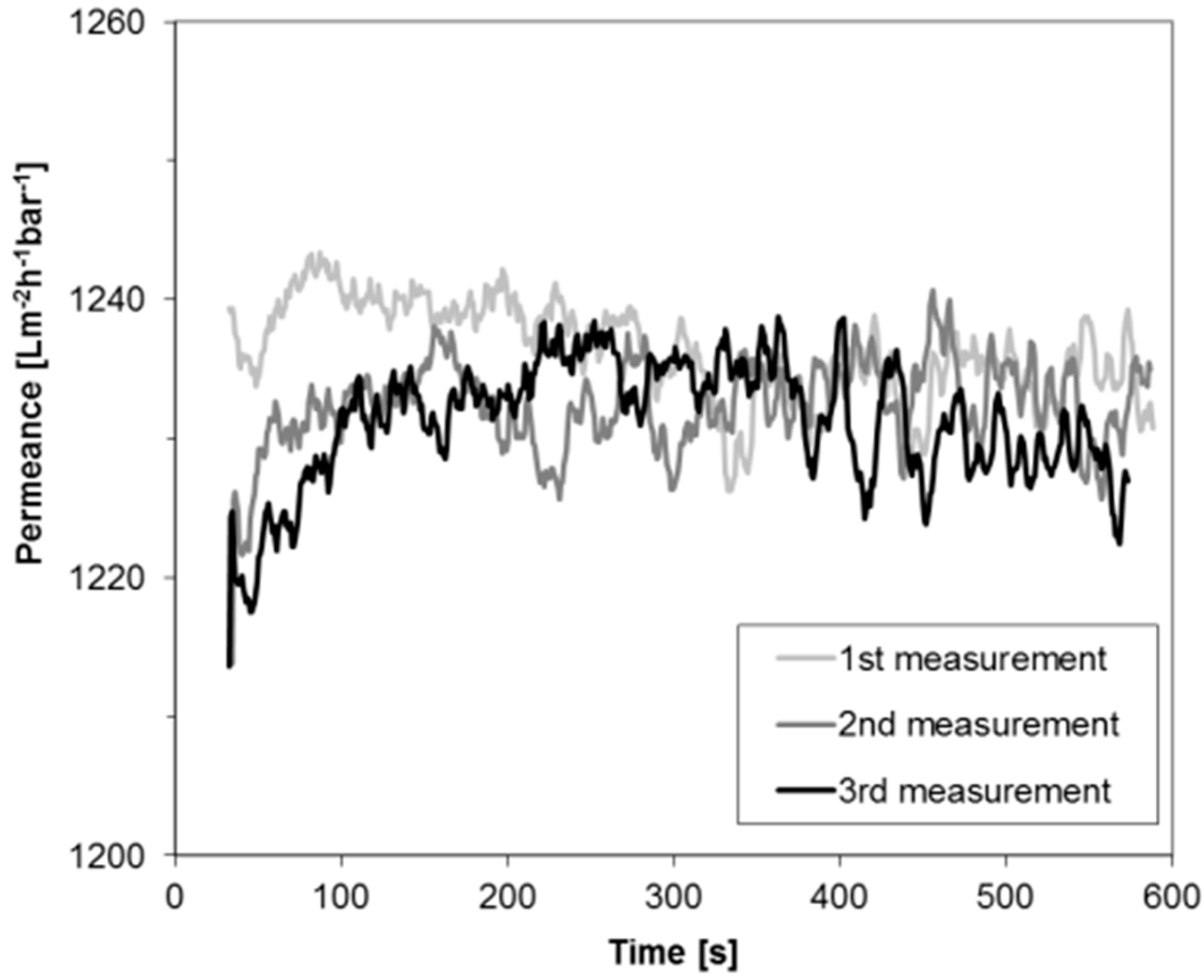
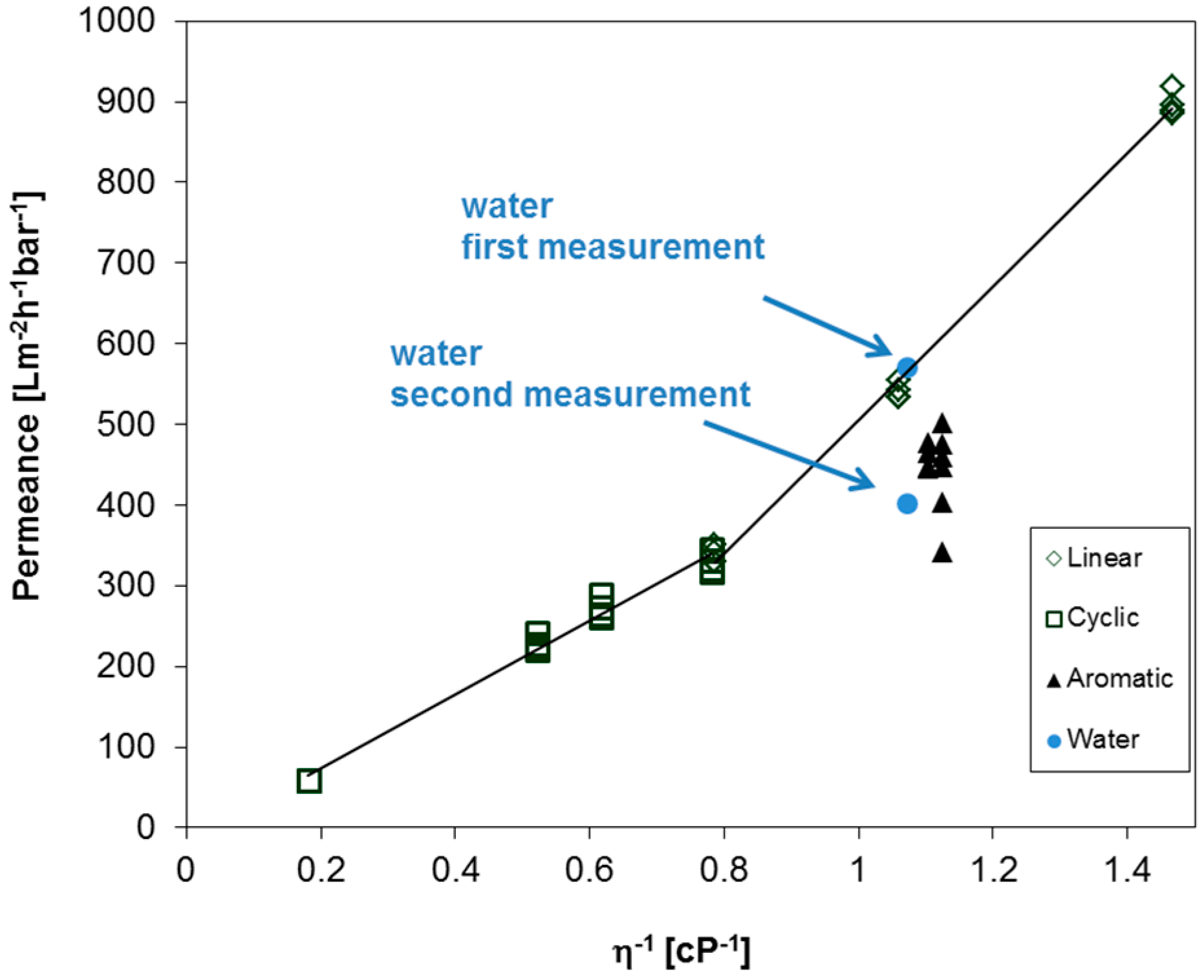
2.1. Verification of the Experimental Method for Sensitive Membranes
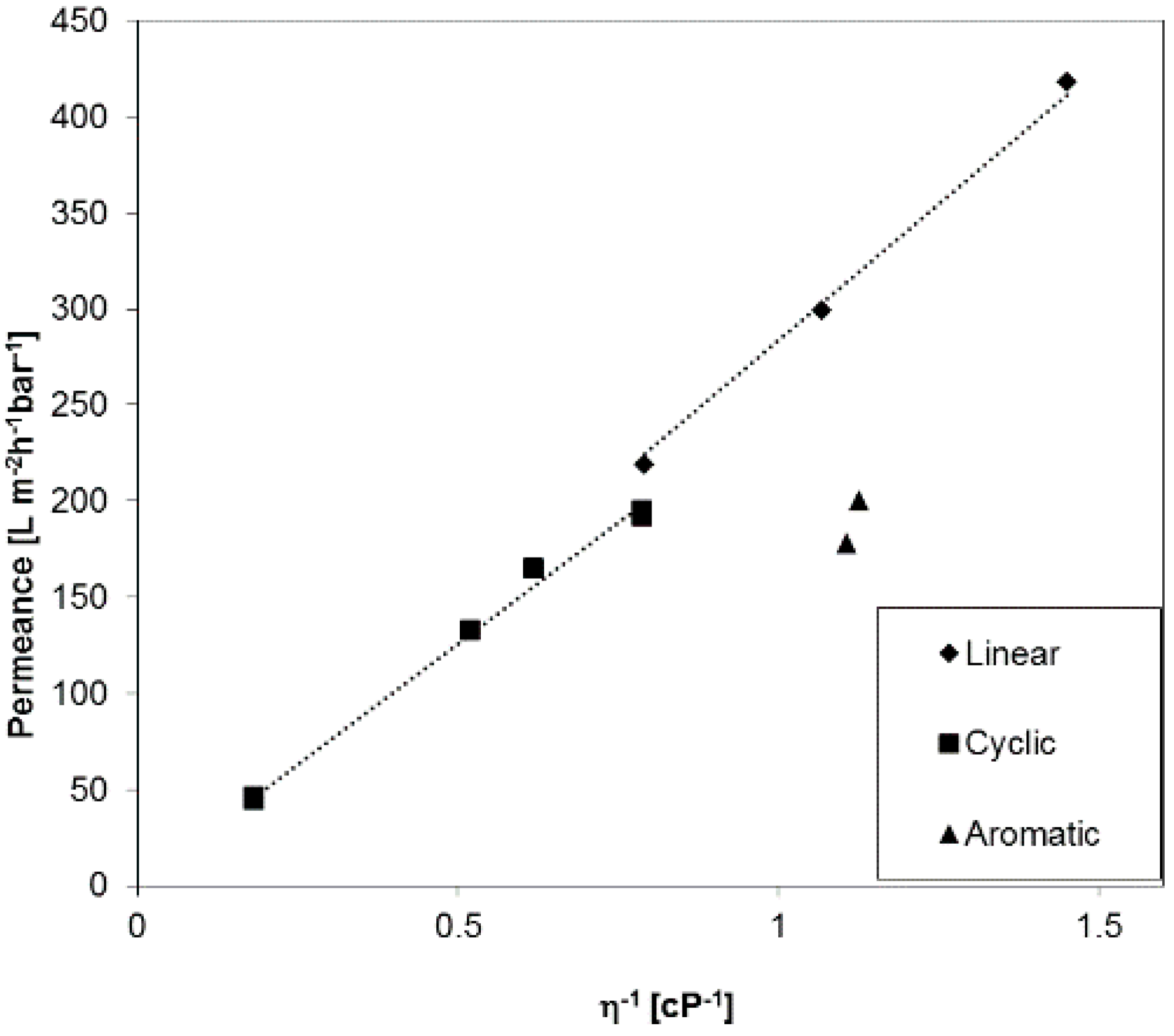
2.2. Permeance of Ultrafiltration Membranes
3. Materials and Methods
3.1. Membranes Used in This Work
3.2. Perfluorinated Compounds Used in This Work and Their Physical Data
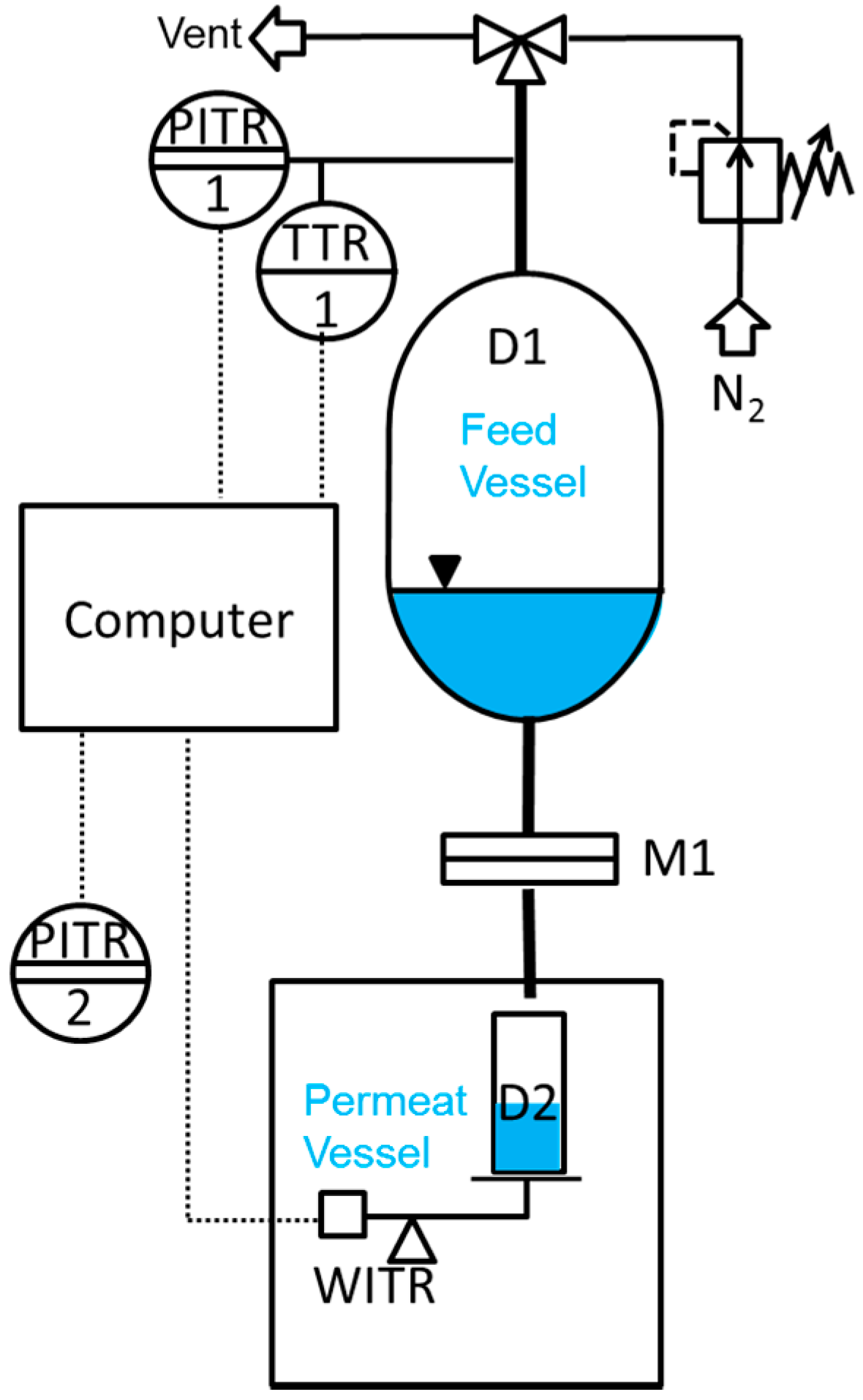
3.3. Liquid Permeance Measurement
4. Conclusions
- PF compounds can be used to study the permeances of sensitive membranes made from water or other solvent swellable materials.
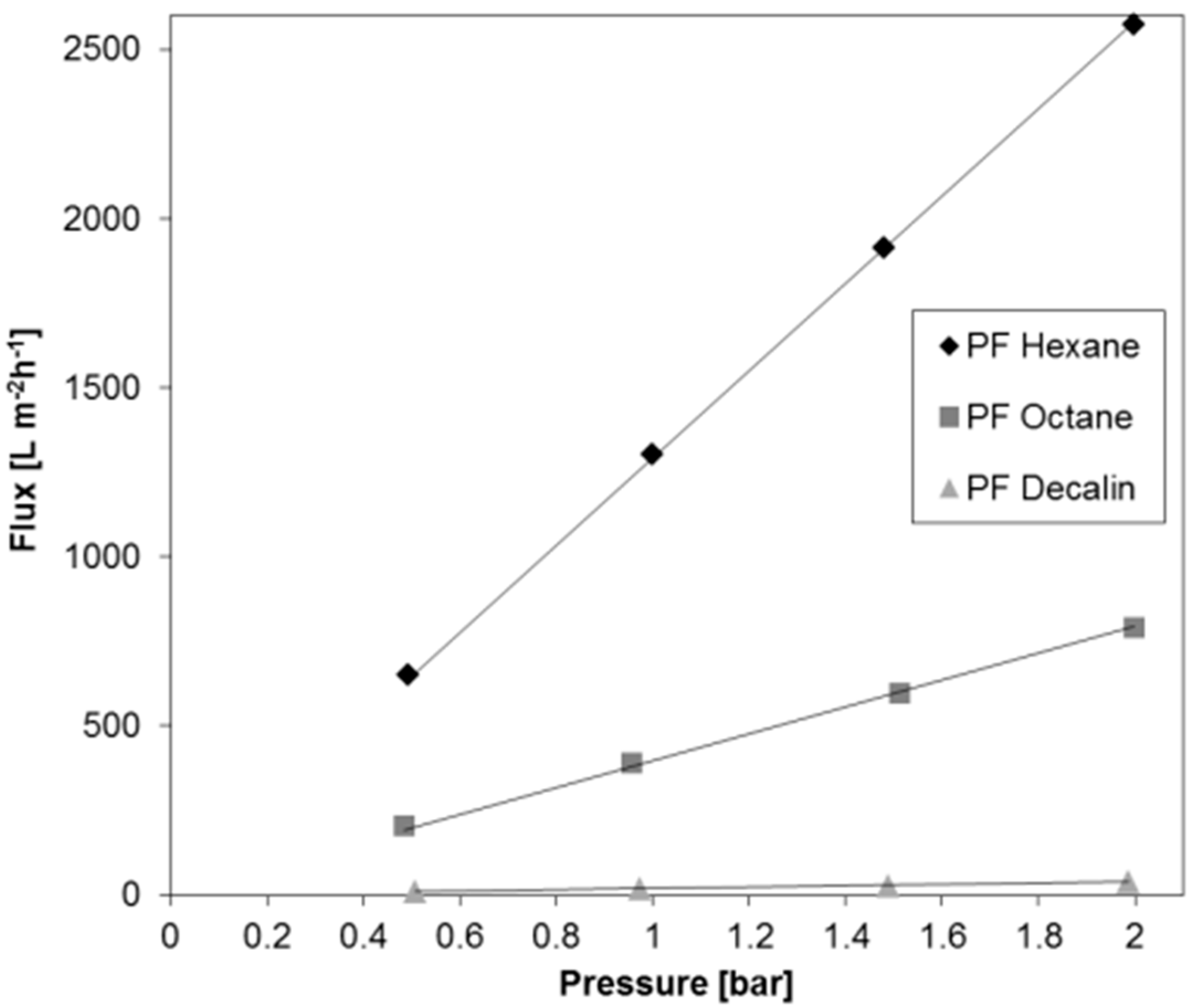
- Pressure dependent measurements show that the permeances of PF compounds are stable in the pressure difference range 0.5 to 2 bar, and the flux does not change significantly with time.
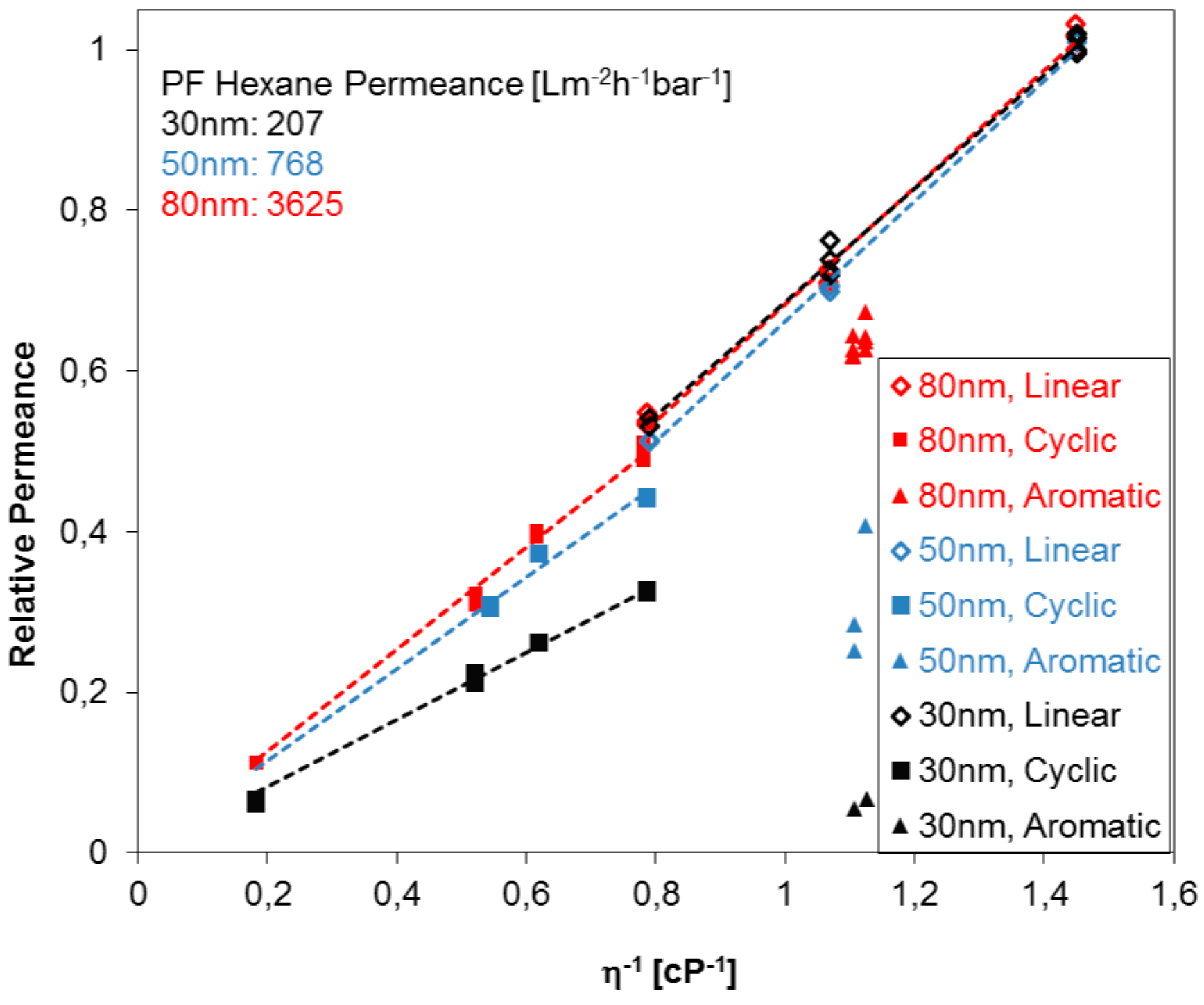
- All studied PF aliphatic compounds, both linear and cyclic, show permeance gradually changing with the viscosity in full agreement with the Hagen–Poiseuille equation.
- The Hagen–Poiseuille trends for linear and cyclic aliphatic compounds deviated increasingly with decreasing average pore diameter: The slope for the cyclic compounds is smaller than that for linear compounds and this observation may possibly be used for estimating the pore size of the membrane without microscopic study each time.
- The permeances of aromatic PF compounds through ultrafiltration membranes made of materials of different nature are lower than the fluxes of aliphatic compounds presumably due to supramolecular interaction between aromatic PF molecules and pore walls.
- Taking into account the considerations discussed above, one can conclude that most suitable PF compounds for the membrane characterization are perfluorooctane and FC-77 by 3M Company (St. Paul, MN, USA). These two compounds have very similar density and viscosity, while one is linear aliphatic and another cyclic ether.
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Van Reis, R.; Zydney, A. Membrane separations in biotechnology. Curr. Opin. Biotechnol. 2001, 12, 208–211. [Google Scholar] [CrossRef]
- Savage, N.; Diallo, M. Nanomaterials and water purification: Opportunities and challenges. J. Nanopart. Res. 2005, 7, 331–342. [Google Scholar] [CrossRef]
- Van Der Bruggen, B.; Vandecasteele, C.; Van Gestel, T.; Doyen, W.; Leysen, R. A review of pressure-driven membrane processes in wastewater treatment and drinking water production. Environ. Prog. 2003, 22, 46–56. [Google Scholar] [CrossRef]
- Saxena, A.; Tripathi, B.P.; Kumar, M.; Shahi, V.K. Membrane-based techniques for the separation and purification of proteins: An overview. Adv. Colloid Interface Sci. 2009, 145, 1–22. [Google Scholar] [CrossRef] [PubMed]
- Jeon, G.; Yang, S.Y.; Kim, J.K. Functional nanoporous membranes for drug delivery. J. Mater. Chem. 2012, 22, 14814–14834. [Google Scholar] [CrossRef]
- Stroeve, P.; Ileri, N. Biotechnical and other applications of nanoporous membranes. Trends Biotechnol. 2011, 29, 259–266. [Google Scholar] [CrossRef] [PubMed]
- Clark, W.M.; Bansal, A.; Sontakke, M.; Ma, Y.H. Protein adsorption and fouling in ceramic ultrafiltration membranes. J. Membr. Sci. 1991, 55, 21–38. [Google Scholar] [CrossRef]
- Robertson, B.C.; Zydney, A.L. Protein adsorption in asymmetric ultrafiltration membranes with highly constricted pores. J. Colloid Interface Sci. 1990, 134, 563–575. [Google Scholar] [CrossRef]
- Freeman, B. Not all membrane pores are made equal; some are more equal than others. Nature 2008, 454, 671. [Google Scholar] [CrossRef] [PubMed]
- Peinemann, K.V.; Abetz, V.; Simon, P.F. Asymmetric superstructure formed in a block copolymer via phase separation. Nat. Mater. 2007, 6, 992–996. [Google Scholar] [CrossRef] [PubMed]
- Jung, A.; Rangou, S.; Abetz, C.; Filiz, V.; Abetz, V. Structure formation of integral asymmetric composite membranes of polystyrene-block-poly(2-vinylpyridine) on a nonwoven. Macromol. Mater. Eng. 2012, 297, 790–798. [Google Scholar] [CrossRef]
- Rangou, S.; Buhr, K.; Filiz, V.; Clodt, J.I.; Lademann, B.; Hahn, J.; Jung, A.; Abetz, V. Self-organized isoporous membranes with tailored pore sizes. J. Membr. Sci. 2014, 451, 266–275. [Google Scholar] [CrossRef]
- Abetz, V. Isoporous block copolymer membranes. Macromol. Rapid Commun. 2015, 36, 10–22. [Google Scholar] [CrossRef] [PubMed]
- Höhme, C.; Hahn, J.; Lademann, B.; Meyer, A.; Bajer, B.; Abetz, C.; Filiz, V.; Abetz, V. Formation of high thermally stable isoporous integral asymmetric block copolymer membranes. Eur. Polym. J. 2016, 85, 72–81. [Google Scholar] [CrossRef]
- Hahn, J.; Clodt, J.I.; Abetz, C.; Filiz, V.; Abetz, V. Thin isoporous block copolymer membranes: It is all about the process. ACS Appl. Mater. Interfaces 2015, 7, 21130–21137. [Google Scholar] [CrossRef] [PubMed]
- Radjabian, M.; Abetz, C.; Fischer, B.; Meyer, A.; Abetz, V. Influence of solvent on the structure of an amphiphilic block copolymer in solution and in formation of an integral asymmetric membrane. ACS Appl. Mater. Interfaces 2017. [Google Scholar] [CrossRef] [PubMed]
- Radjabian, M.; Abetz, V. Tailored pore sizes in integral asymmetric membranes formed by blends of block copolymers. Adv. Mater. 2015, 27, 352–355. [Google Scholar] [CrossRef] [PubMed]
- Hahn, J.; Filiz, V.; Rangou, S.; Clodt, J.; Jung, A.; Buhr, K.; Abetz, C.; Abetz, V. Structure formation of integral-asymmetric membranes of polystyrene-block-poly(ethylene oxide). J. Polym. Sci. Polym. Phys. 2013, 51, 281–290. [Google Scholar] [CrossRef]
- Hahn, J.; Filiz, V.; Rangou, S.; Lademann, B.; Buhr, K.; Clodt, J.I.; Jung, A.; Abetz, C.; Abetz, V. PtBS-b-P4VP and PTMASS-b- P4VP isoporous integral-asymmetric membranes with high thermal and chemical stability. Macromol. Mater. Eng. 2013, 298, 1315–1321. [Google Scholar] [CrossRef]
- Hahn, J.; Clodt, J.I.; Filiz, V.; Abetz, V. Protein separation performance of self-assembled block copolymer membranes. RSC Adv. 2014, 4, 10252–10260. [Google Scholar] [CrossRef]
- Shukla, R.; Cheryan, M. Performance of ultrafiltration membranes in ethanol-water solutions: Effect of membrane conditioning. J. Membr. Sci. 2002, 198, 75–85. [Google Scholar] [CrossRef]
- Miller, D.J.; Dreyer, D.R.; Bielawski, C.W.; Paul, D.R.; Freeman, B.D. Surface modification of water purification membranes. Angew. Chem. Int. Ed. Engl. 2017, 56, 4662–4711. [Google Scholar] [CrossRef] [PubMed]
- Sun, W.; Liu, J.; Chu, H.; Dong, B. Pretreatment and membrane hydrophilic modification to reduce membrane fouling. Membranes 2013, 3, 226–241. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Mo, Y.; Li, J.; Guo, W.; Ngo, H.H. In-situ monitoring techniques for membrane fouling and local filtration characteristics in hollow fiber membrane processes: A critical review. J. Membr. Sci. 2017, 528, 187–200. [Google Scholar] [CrossRef]
- Bitsanis, I.; Somers, S.A.; Davis, H.T.; Tirrell, M. Microscopic dynamics of flow in molecularly narrow pores. J. Chem. Phys. 1990, 93, 3427–3431. [Google Scholar] [CrossRef]
- Clodt, J.I.; Bajer, B.; Buhr, K.; Hahn, J.; Filiz, V.; Abetz, V. Performance study of isoporous membranes with tailored pore sizes. J. Membr. Sci. 2015, 495, 334–340. [Google Scholar] [CrossRef]
- Hernandez, A.; Calvo, J.I.; Pradanos, P.; Tejerina, F. Pore size distributions of track-etched membranes; comparison of surface and bulk porosities. Colloid Surface A 1998, 138, 391–401. [Google Scholar] [CrossRef]
- Calvo, J.I.; Hernández, A.; Caruana, G.; Martínez, L. Pore size distributions in microporous membranes. J. Colloid Interface Sci. 1995, 175, 138–150. [Google Scholar] [CrossRef]
- Grünauer, J.; Filiz, V.; Shishatskiy, S.; Abetz, C.; Abetz, V. Scalable application of thin film coating techniques for supported liquid membranes for gas separation made from ionic liquids. J. Membr. Sci. 2016, 518, 178–191. [Google Scholar] [CrossRef]
- Grünauer, J.; Shishatskiy, S.; Abetz, C.; Abetz, V.; Filiz, V. Ionic liquids supported by isoporous membranes for CO2/N2 gas separation applications. J. Membr. Sci. 2015, 494, 224–233. [Google Scholar] [CrossRef]
- Abetz, V.; Brinkmann, T.; Dijkstra, M.; Ebert, K.; Fritsch, D.; Ohlrogge, K.; Paul, D.; Peinemann, K.V.; Pereira-Nunes, S.; Scharnagl, N.; et al. Developments in membrane research: From material via process design to industrial application. Adv. Eng. Mater. 2006, 8, 328–358. [Google Scholar] [CrossRef]
- Scharnagl, N.; Buschatz, H. Polyacrylonitrile (pan) membranes for ultra- and microfiltration. Desalination 2001, 139, 191–198. [Google Scholar] [CrossRef]

| Membrane | Abbr. Used in This Work | Average Pore Diameter (nm) | Porosity (%) |
|---|---|---|---|
| Polycarbonate track-etched PCN8CP04700 | PC80 | 80 a | 2.0 a |
| Polycarbonate track-etched PCN5CP04700 | PC50 | 50 a | 1.2 a |
| Polycarbonate track-etched PCN3CP04700 | PC30 | 30 a | 0.4 a |
| Polyethylene UPZP05205 | PE50 | 50 a | – c |
| Anodized alumina Anodisc® | Anodisc | 20 a | 40 b |
| Polyacrylonitrile | PAN | 22 b | 12.3 b |
| Block copolymer membranes | BCPM | 42 b | 29.5 b |
| Substance | Abbreviation | Mwa (g/mol) | Density a (g/cm−3) | BP a (°C) | Dyn. Viscosity (cP) at 23 °C |
|---|---|---|---|---|---|
| Perfluorohexane | PF Hexane | 339.04 | 1.686 | 59 | 0.690 |
| Hexadecafluoroheptane | PF Heptane | 388.05 | 1.731 | 83 | 0.936 |
| Perfluorooctane | PF Octane | 438.06 | 1.757 | 103 | 1.27 |
| Perfluoro(methylcyclohexane) | PF MCH | 350.05 | 1.784 | 76 | 1.62 |
| Perfluoro-1,3-imethylcyclohexane | PF DMCH | 400.06 | 1.838 | 101 | 1.93 |
| Perfluorodecalin | PF Decalin | 462.08 | 1.926 | 142 | 5.50 |
| Perfluorocycloether | FC-77 | 416 | 1.767 | 97 | 1.27 |
| Perfluorobenzene | PF Benzene | 186.05 | 1.613 | 81 | 0.889 |
| Perfluorotoluene | PF Toluene | 236.06 | 1.664 | 104 | 0.903 |
| Water | Water | 18.02 | 0.995 | 100 | 0.932 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Clodt, J.I.; Filiz, V.; Shishatskiy, S. Perfluorinated Compounds as Test Media for Porous Membranes. Membranes 2017, 7, 51. https://doi.org/10.3390/membranes7030051
Clodt JI, Filiz V, Shishatskiy S. Perfluorinated Compounds as Test Media for Porous Membranes. Membranes. 2017; 7(3):51. https://doi.org/10.3390/membranes7030051
Chicago/Turabian StyleClodt, Juliana I., Volkan Filiz, and Sergey Shishatskiy. 2017. "Perfluorinated Compounds as Test Media for Porous Membranes" Membranes 7, no. 3: 51. https://doi.org/10.3390/membranes7030051
APA StyleClodt, J. I., Filiz, V., & Shishatskiy, S. (2017). Perfluorinated Compounds as Test Media for Porous Membranes. Membranes, 7(3), 51. https://doi.org/10.3390/membranes7030051








