Biochip for the Detection of Bacillus anthracis Lethal Factor and Therapeutic Agents against Anthrax Toxins
Abstract
:1. Introduction
2. Results
2.1. Biosensor Fabrication
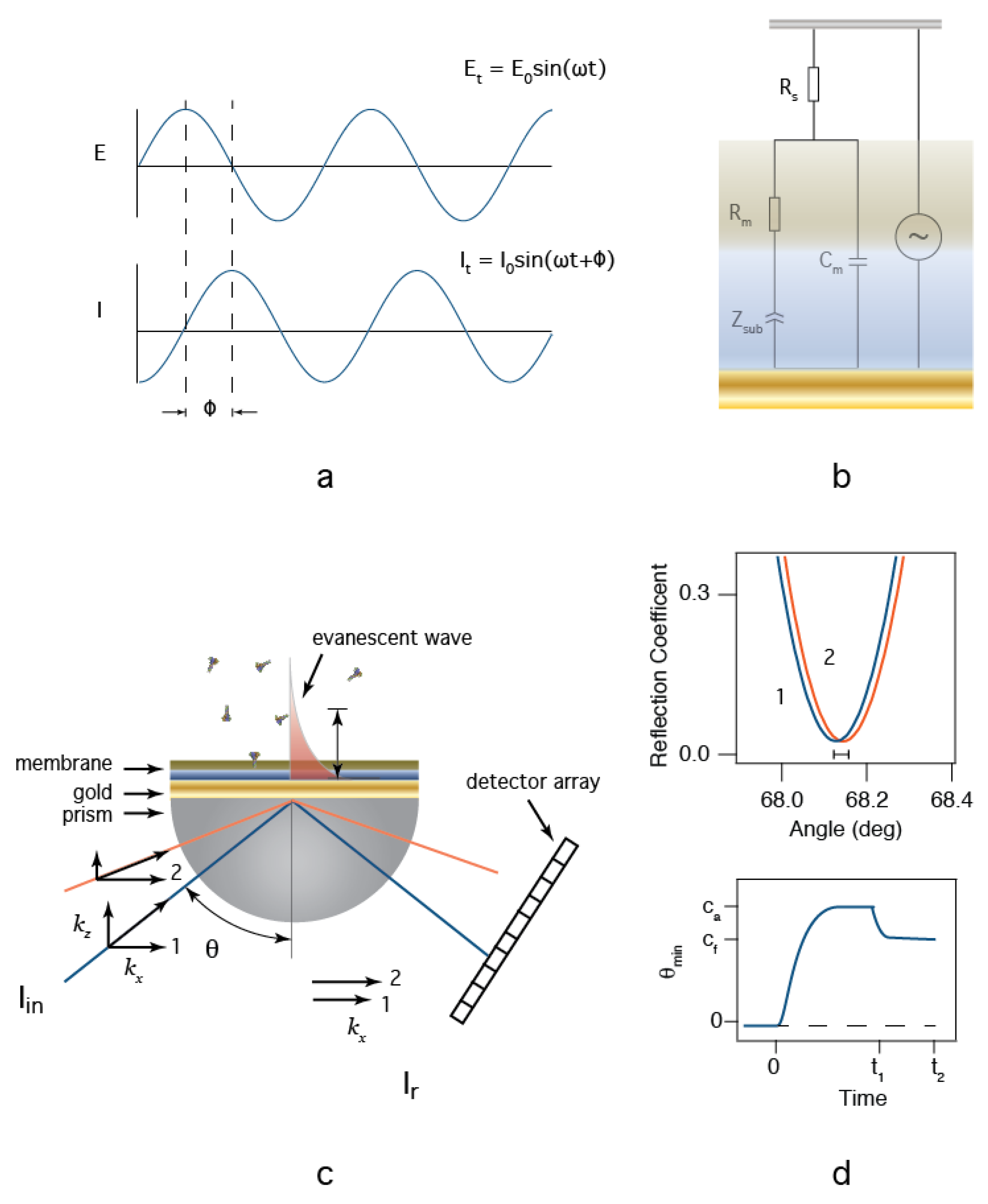
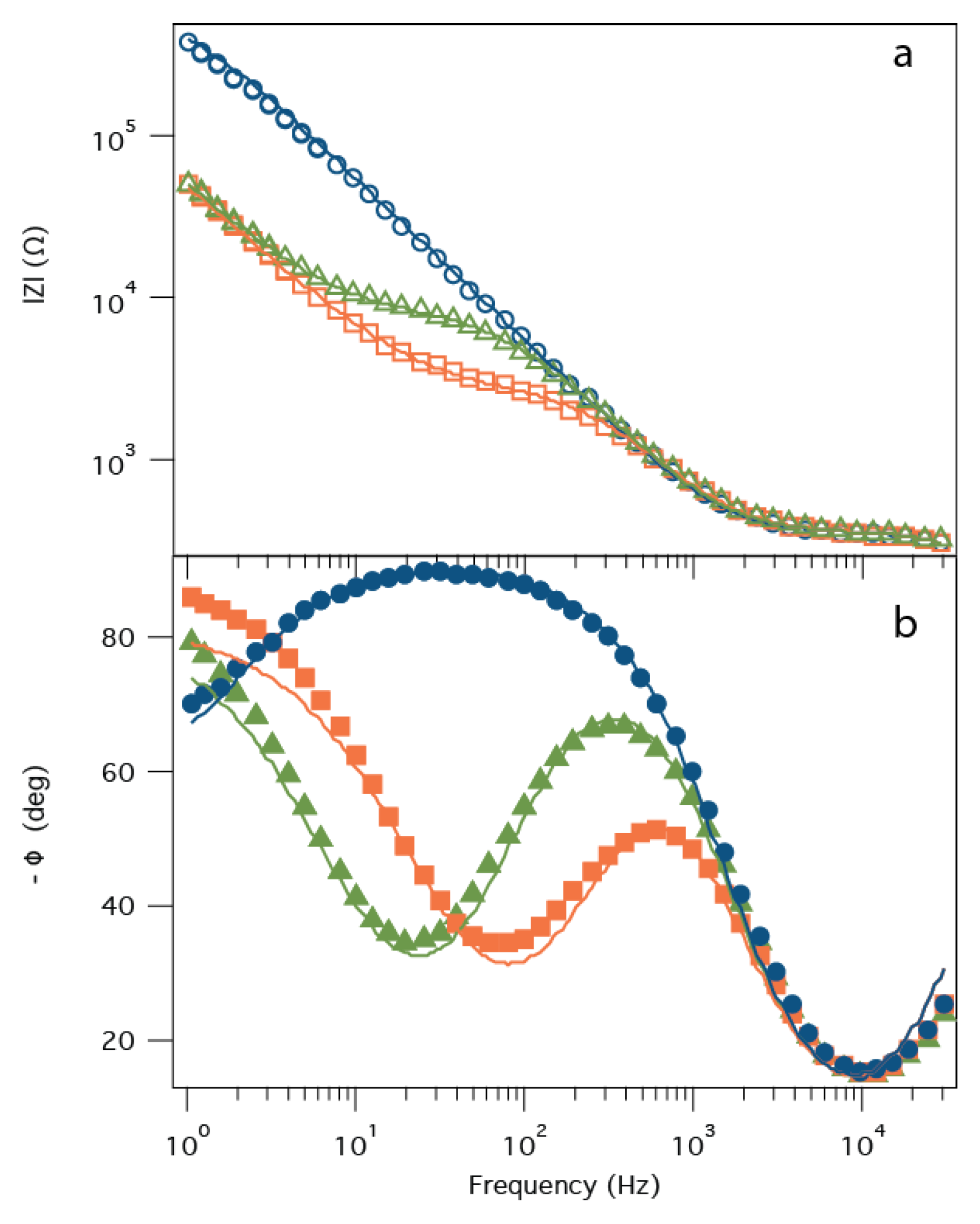
2.2. Electrochemical Impedance Spectroscopy
2.3. Surface Plasmon Resonance
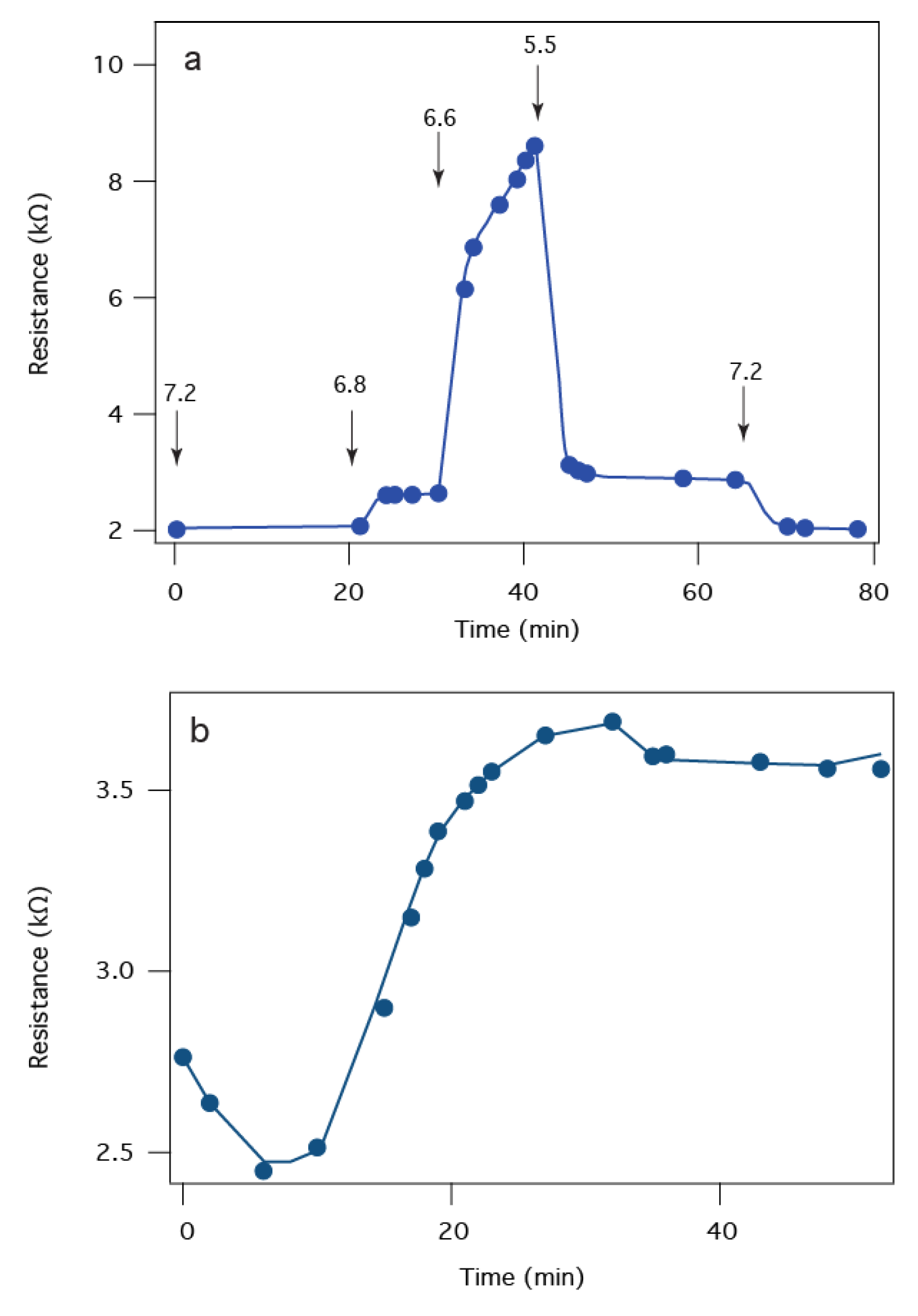
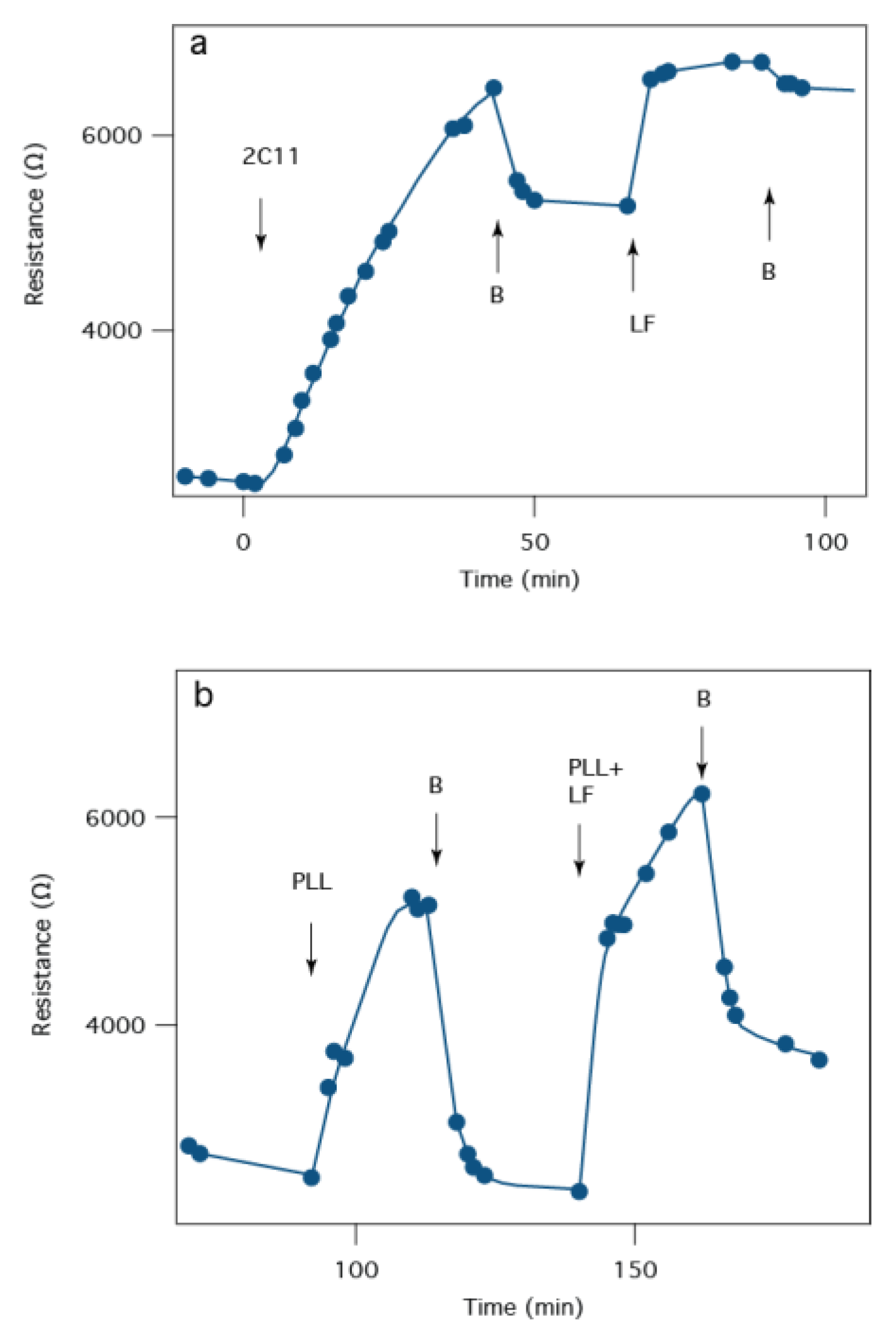
2.4. B. anthracis Toxin Biochip Formation
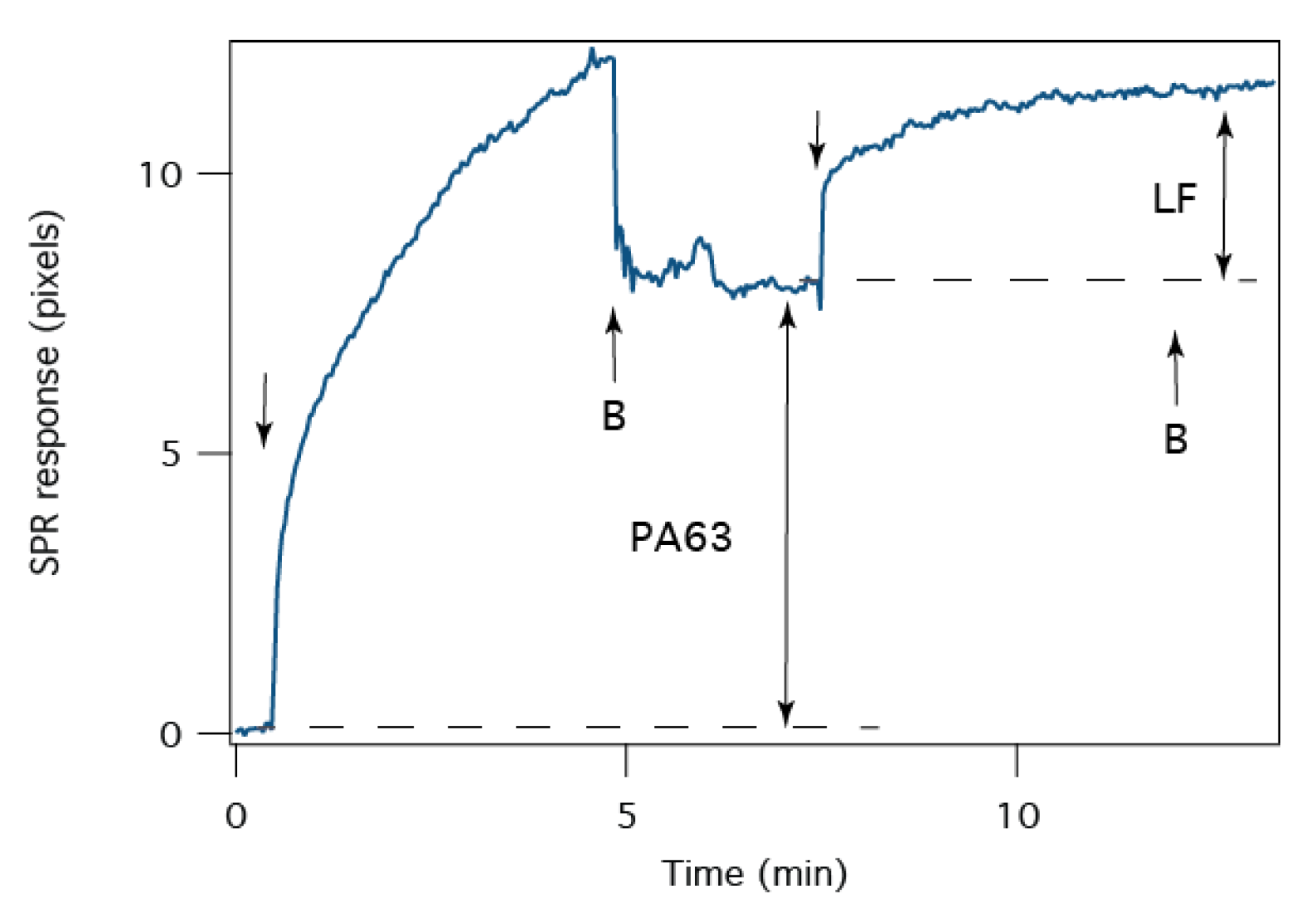
2.5. Detection of B. anthracis Toxin LF
2.6. Screening of Therapeutic Agents against B anthracis Toxins
3. Discussion
4. Materials and Methods
4.1. Electrolyte Solutions and Analytes
4.2. SPR-EIS Measurements
4.3. SPR Sample Preparation
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Sackmann, E. Supported membranes: Scientific and practical applications. Science 1996, 271, 43–48. [Google Scholar] [CrossRef] [PubMed]
- Boxer, S.G. Molecular transport and organization in supported lipid membranes. Curr. Opin. Chem. Biol. 2000, 4, 704–709. [Google Scholar] [CrossRef]
- Richter, R.P.; Him, J.; Brisson, A. Supported lipid membranes. Mater. Today 2003, 6, 32–37. [Google Scholar] [CrossRef]
- Castellana, E.T.; Cremer, P.S. Solid supported lipid bilayers: From biophysical studies to sensor design. Surf. Sci. Rep. 2006, 61, 429–444. [Google Scholar] [CrossRef]
- McConnell, H.M.; Watts, T.H.; Weis, R.M.; Brian, A.A. Supported planar membranes in studies of cell-cell recognition in the immune system. Biochim. Biophys. Acta 1986, 864, 95–106. [Google Scholar] [CrossRef]
- Groves, J. Supported planar bilayers in studies on immune cell adhesion and communication. J. Immunol. Methods 2003, 278, 19–32. [Google Scholar] [CrossRef]
- Sleytr, U.B.; Sára, M.; Pum, D.; Schuster, B. Characterization and use of crystalline bacterial cell surface layers. Prog. Surf. Sci. 2001, 68, 231–278. [Google Scholar] [CrossRef]
- Gufler, P.C.; Pum, D.; Sleytr, U.B.; Schuster, B. Highly robust lipid membranes on crystalline S-layer supports investigated by electrochemical impedance spectroscopy. Biochim. Biophys. Acta 2004, 1661, 154–165. [Google Scholar] [CrossRef] [PubMed]
- Sleytr, U.B.; Huber, C.; Ilk, N.; Pum, D.; Schuster, B.; Egelseer, E.M. S-layers as a tool kit for nanobiotechnological applications. FEMS Microbiol. Lett. 2007, 267, 131–144. [Google Scholar] [CrossRef] [PubMed]
- Damiati, S.; Schrems, A.; Sinner, E.-K.; Sleytr, U.; Schuster, B. Probing Peptide and Protein Insertion in a Biomimetic S-Layer Supported Lipid Membrane Platform. IJMS 2015, 16, 2824–2838. [Google Scholar] [CrossRef] [PubMed]
- Florin, E.L.; Gaub, H.E. Painted supported lipid membranes. Biophys. J. 1993, 64, 375–383. [Google Scholar] [CrossRef]
- Glazier, S.; Vanderah, D.; Plant, A.; Bayley, H.; Valincius, G.; Kasianowicz, J.J. Reconstitution of the pore-forming toxin alpha-hemolysin in phospholipid/18-octadecyl-1-thiahexa(ethylene oxide) and phospholipid/n-octadecanethiol supported bilayer membranes. Langmuir 2000, 16, 10428–10435. [Google Scholar] [CrossRef]
- Cornell, B.; Braach-Maksvytis, V.; King, L.; Osman, P.; Raguse, B.; Wieczorek, L.; Pace, R. A biosensor that uses ion-channel switches. Nature 1997, 387, 580–583. [Google Scholar] [CrossRef] [PubMed]
- Knoll, W.; Naumann, R.; Friedrich, M.; Robertson, J.W.F.; Losche, M.; Heinrich, F.; McGillivray, D.J.; Schuster, B.; Gufler, P.C.; Pum, D.; et al. Solid supported lipid membranes: New concepts for the biomimetic functionalization of solid surfaces. Biointerphases 2008, 3, FA125–FA135. [Google Scholar] [CrossRef] [PubMed]
- Jackman, J.A.; Knoll, W.; Cho, N.-J. Biotechnology Applications of Tethered Lipid Bilayer Membranes. Materials 2012, 5, 2637–2657. [Google Scholar] [CrossRef]
- Woodhouse, G.; King, L.; Wieczorek, L.; Cornell, B. Kinetics of the competitive response of receptors immobilised to ion-channels which have been incorporated into a tethered bilayer. Faraday Discuss. 1998, 111, 247–258. [Google Scholar] [CrossRef] [PubMed]
- Schiller, S.M.; Naumann, R.; Lovejoy, K.; Kunz, H.; Knoll, W. Archaea analogue thiolipids for tethered bilayer lipid membranes on ultrasmooth gold surfaces. Angew. Chem. Int. Ed. 2003, 42, 208–211. [Google Scholar] [CrossRef] [PubMed]
- Atanasov, V.; Atanasova, P.P.; Vockenroth, I.K.; Knorr, N.; Köper, I. A molecular toolkit for highly insulating tethered bilayer lipid membranes on various substrates. Bioconjugate Chem. 2006, 17, 631–637. [Google Scholar] [CrossRef] [PubMed]
- He, L.; Robertson, J.W.F.; Li, J.; Kärcher, I.; Schiller, S.M.; Knoll, W.; Naumann, R. Tethered bilayer lipid membranes based on monolayers of thiolipids mixed with a complementary dilution molecule. 1. Incorporation of channel peptides. Langmuir 2005, 21, 11666–11672. [Google Scholar] [CrossRef] [PubMed]
- McGillivray, D.J.; Valincius, G.; Vanderah, D.J.; Febo-Ayala, W.; Woodward, J.T.; Heinrich, F.; Kasianowicz, J.J.; Losche, M. Molecular-scale structural and functional characterization of sparsely tethered bilayer lipid membranes. Biointerphases 2007, 2, 21–33. [Google Scholar] [CrossRef] [PubMed]
- Heinrich, F.; Ng, T.; Vanderah, D.J.; Shekhar, P.; Mihailescu, M.; Nanda, H.; Losche, M. A New Lipid Anchor for Sparsely Tethered Bilayer Lipid Membranes. Langmuir 2009, 25, 4219–4229. [Google Scholar] [CrossRef] [PubMed]
- Jenkins, A.; Bushby, R.J.; Boden, N.; Evans, S.D.; Knowles, P.F.; Liu, Q.Y.; Miles, R.E.; Ogier, S.D. Ion-selective lipid bilayers tethered to microcontact printed self-assembled monolayers containing cholesterol derivatives. Langmuir 1998, 14, 4675–4678. [Google Scholar] [CrossRef]
- Giess, F.; Friedrich, M.G.; Heberle, J.; Naumann, R.L.; Knoll, W. The protein-tethered lipid bilayer: A novel mimic of the biological membrane. Biophys. J. 2004, 87, 3213–3220. [Google Scholar] [CrossRef] [PubMed]
- Friedrich, M.G.; Robertson, J.W.F.; Walz, D.; Knoll, W.; Naumann, R.L.C. Electronic wiring of a multi-redox site membrane protein in a biomimetic surface architecture. Biophys. J. 2008, 94, 3698–3705. [Google Scholar] [CrossRef] [PubMed]
- Junghans, A.; Champagne, C.; Cayot, P.; Loupiac, C.; Köper, I. Probing Protein-Membrane Interactions Using Solid Supported Membranes. Langmuir 2011, 27, 2709–2716. [Google Scholar] [CrossRef] [PubMed]
- Cornell, B.A.; Krishna, G.; Osman, P.D.; Pace, R.D.; Wieczorek, L. Tethered-bilayer lipid membranes as a support for membrane-active peptides. Biochem. Soc. Trans. 2001, 29, 613–617. [Google Scholar] [CrossRef] [PubMed]
- Vockenroth, I.K.; Atanasova, P.P.; Jenkins, A.T.A.; Köper, I. Incorporation of alpha-hemolysin in different tethered bilayer lipid membrane architectures. Langmuir 2008, 24, 496–502. [Google Scholar] [CrossRef] [PubMed]
- McGillivray, D.J.; Valincius, G.; Heinrich, F.; Robertson, J.W.F.; Vanderah, D.J.; Febo-Ayala, W.; Ignatjev, I.; Losche, M.; Kasianowicz, J.J. Structure of functional Staphylococcus aureus alpha-hemolysin channels in tethered bilayer lipid membranes. Biophys. J. 2009, 96, 1547–1553. [Google Scholar] [CrossRef] [PubMed]
- Schick, S.; Chen, L.; Li, E.; Lin, J.; Köper, I.; Hristova, K. Assembly of the M2 tetramer is strongly modulated by lipid chain length. Biophys. J. 2010, 99, 1810–1817. [Google Scholar] [CrossRef] [PubMed]
- Weiss, S.A.; Bushby, R.J.; Evans, S.D.; Jeuken, L.J.C. A study of cytochrome bo3 in a tethered bilayer lipid membrane. Biochim. Biophys. Acta 2010, 1797, 1917–1923. [Google Scholar] [CrossRef] [PubMed]
- McMillan, D.G.G.; Marritt, S.J.; Firer-Sherwood, M.A.; Shi, L.; Richardson, D.J.; Evans, S.D.; Elliott, S.J.; Butt, J.N.; Jeuken, L.J.C. Protein-protein interaction regulates the direction of catalysis and electron transfer in a redox enzyme complex. J. Am. Chem. Soc. 2013, 135, 10550–10556. [Google Scholar] [CrossRef] [PubMed]
- Jeuken, L.J.C. Electrodes for integral membrane enzymes. Nat. Prod. Rep. 2009, 26, 1234–1240. [Google Scholar] [CrossRef] [PubMed]
- Robertson, J.W.F.; Kasianowicz, J.J.; Banerjee, S. Analytical Approaches for Studying Transporters, Channels and Porins. Chem. Rev. 2012, 112, 6227–6249. [Google Scholar] [CrossRef] [PubMed]
- Robelek, R.; Lemker, E.S.; Wiltschi, B.; Kirste, V.; Naumann, R.L.C.; Oesterhelt, D.; Sinner, E.-K. Incorporation of in vitro Synthesized GPCR into a Tethered Artificial Lipid Membrane System. Angew. Chem. Int. Ed. 2007, 46, 605–608. [Google Scholar] [CrossRef] [PubMed]
- Zieleniecki, J.L.; Nagarajan, Y.; Waters, S.; Rongala, J.; Thompson, V.; Hrmova, M.; Köper, I. Cell-Free Synthesis of a Functional Membrane Transporter into a Tethered Bilayer Lipid Membrane. Langmuir 2016, 32, 2445–2449. [Google Scholar] [CrossRef] [PubMed]
- Clements, B.W.; Casani, J. Disasters and Public Health; Butterworth-Heinemann: Oxford, UK, 2016. [Google Scholar]
- Paddle, B.M. Biosensors for chemical and biological agents of defence interest. Biosens. Bioelectron. 1996, 11, 1079–1113. [Google Scholar] [CrossRef]
- Mabry, R.; Brasky, K.; Geiger, R.; Carrion, R.; Hubbard, G.B.; Leppla, S.; Patterson, J.L.; Georgiou, G.; Iverson, B.L. Detection of Anthrax Toxin in the Serum of Animals Infected with Bacillus anthracis by Using Engineered Immunoassays. Clin. Vaccine Immunol. 2006, 13, 671–677. [Google Scholar] [CrossRef] [PubMed]
- Barth, H.; Aktories, K.; Popoff, M.R.; Stiles, B.G. Binary bacterial toxins: Biochemistry, biology, and applications of common Clostridium and Bacillus proteins. Microbiol. Mol. Biol. Rev. 2004, 68, 373–402. [Google Scholar] [CrossRef] [PubMed]
- Young, J.A.T.; Collier, R.J. Anthrax toxin: Receptor binding, internalization, pore formation, and translocation. Annu. Rev. Biochem. 2007, 76, 243–265. [Google Scholar] [CrossRef] [PubMed]
- Jiang, J.; Pentelute, B.L.; Collier, R.J.; Zhou, Z.H. Atomic structure of anthrax protective antigen pore elucidates toxin translocation. Nature 2015, 521, 545–549. [Google Scholar] [CrossRef] [PubMed]
- Kintzer, A.F.; Thoren, K.L.; Sterling, H.J.; Dong, K.C.; Feld, G.K.; Tang, I.I.; Zhang, T.T.; Williams, E.R.; Berger, J.M.; Krantz, B.A. The Protective Antigen Component of Anthrax Toxin Forms Functional Octameric Complexes. J. Mol. Biol. 2009, 392, 614–629. [Google Scholar] [CrossRef] [PubMed]
- Nablo, B.J.; Panchal, R.G.; Bavari, S.; Nguyen, T.L.; Gussio, R.; Ribot, W.; Friedlander, A.; Chabot, D.; Reiner, J.E.; Robertson, J.W.F.; et al. Anthrax toxin-induced rupture of artificial lipid bilayer membranes. J. Chem. Phys. 2013, 139. [Google Scholar] [CrossRef] [PubMed]
- Halverson, K.M.; Panchal, R.G.; Nguyen, T.L.; Gussio, R.; Little, S.F.; Misakian, M.; Bavari, S.; Kasianowicz, J.J. Anthrax biosensor, protective antigen ion channel asymmetric blockade. J. Biol. Chem. 2005, 280, 34056–34062. [Google Scholar] [CrossRef] [PubMed]
- Nestorovich, E.M.; Karginov, V.A.; Berezhkovskii, A.M.; Bezrukov, S.M. Blockage of anthrax PA63 pore by a multicharged high-affinity toxin inhibitor. Biophys. J. 2010, 99, 134–143. [Google Scholar] [CrossRef] [PubMed]
- Karginov, V.A.; Nestorovich, E.M.; Schmidtmann, F.; Robinson, T.M.; Yohannes, A.; Fahmi, N.E.; Bezrukov, S.M.; Hecht, S.M. Inhibition of S-aureus alpha-hemolysin and B-anthracis lethal toxin by beta-cyclodextrin derivatives. Bioorgan. Med. Chem. 2007, 15, 5424–5431. [Google Scholar] [CrossRef] [PubMed]
- Beitzinger, C.; Bronnhuber, A.; Duscha, K.; Riedl, Z.; Huber-Lang, M.; Benz, R.; Hajós, G.; Barth, H. Designed azolopyridinium salts block protective antigen pores in vitro and protect cells from anthrax toxin. PLoS ONE 2013, 8. [Google Scholar] [CrossRef] [PubMed]
- Vockenroth, I.K.; Ohm, C.; Robertson, J.W.F.; McGillivray, D.J.; Losche, M.; Köper, I. Stable insulating tethered bilayer lipid membranes. Biointerphases 2008, 3. [Google Scholar] [CrossRef] [PubMed]
- Hoiles, W.; Krishnamurthy, V.; Cranfield, C.G.; Cornell, B. An Engineered Membrane to Measure Electroporation: Effect of Tethers and Bioelectronic Interface. Biophys. J. 2014, 107, 1339–1351. [Google Scholar] [CrossRef] [PubMed]
- Valincius, G.; Meskauskas, T.; Ivanauskas, F. Electrochemical Impedance Spectroscopy of Tethered Bilayer Membranes. Langmuir 2012, 28, 977–990. [Google Scholar] [CrossRef] [PubMed]
- Jennings-Antipov, L.D.; Song, L.; Collier, R.J. Interactions of anthrax lethal factor with protective antigen defined by site-directed spin labeling. Proc. Natl. Acad. Sci. USA 2011, 108, 1868–1873. [Google Scholar] [CrossRef] [PubMed]
- Pimental, R.; Christensen, K.; Krantz, B.; Collier, R.J. Anthrax toxin complexes: Heptameric protective antigen can bind lethal factor and edema factor simultaneously. Biochem. Biophys. Res. Commun. 2004, 322, 258–262. [Google Scholar] [CrossRef] [PubMed]
- Pannifer, A.; Wong, T.; Schwarzenbacher, R.; Renatus, M.; Petosa, C.; Bienkowska, J.; Lacy, D.; Collier, R.J.; Park, S.; Leppla, S.; et al. Crystal structure of the anthrax lethal factor. Nature 2001, 414, 229–233. [Google Scholar] [CrossRef] [PubMed]
- Katz, E.; Willner, I. Probing biomolecular interactions at conductive and semiconductive surfaces by impedance spectroscopy: Routes to impedimetric immunosensors, DNA-Sensors, and enzyme biosensors. Electroanal. 2003, 15, 913–947. [Google Scholar] [CrossRef]
- Macdonald, J.R.; Barsoukov, E. Impedance Spectroscopy: Theory, Experiment and Applications, 2nd ed.; Wiley-Interscience: New York, NY, USA, 2005. [Google Scholar]
- Valincius, G.; Mickevicius, M. Tethered Phospholipid Bilayer Membranes. In Advances in Planar Lipid Bilayers and Liposomes; Elsevier: Amsterdam, The Netherlands, 2015; Volume 21, pp. 27–61. [Google Scholar]
- Knoll, W. Interfaces and thin films as seen by bound electromagnetic waves. Annu. Rev. Phys. Chem. 1998, 49, 569–638. [Google Scholar] [CrossRef] [PubMed]
- Homola, J. Surface plasmon resonance sensors for detection of chemical and biological species. Chem. Rev. 2008, 108, 462–493. [Google Scholar] [CrossRef] [PubMed]
- Hansen, W.N. Electric Fields Produced by Propagation of Plane Coherent Electromagnetic Radiation in a Stratified Medium. J. Opt. Soc. Am. 1968, 58, 380–390. [Google Scholar] [CrossRef]
- Corn, R.M. Complex Fresnel Calculations. Available online: http://corninfo.ps.uci.edu/calculations.html (accessed on 6 May 2016).
- Gutknecht, J. Proton/hydroxide conductance and permeability through phospholipid bilayer membranes. Proc. Natl. Acad. Sci. USA 1987, 84, 6443–6446. [Google Scholar] [CrossRef] [PubMed]
- Krantz, B.; Finkelstein, A.; Collier, R.J. Protein translocation through the anthrax toxin transmembrane pore is driven by a proton gradient. J. Mol. Biol. 2006, 355, 968–979. [Google Scholar] [CrossRef] [PubMed]
- Sprenkle, M.D.; Griffith, J.; Marinelli, W.; Boyer, A.E.; Quinn, C.P.; Pesik, N.T.; Hoffmaster, A.; Keenan, J.; Juni, B.A.; Blaney, D.D. Lethal Factor and Anti-Protective Antigen IgG Levels Associated with Inhalation Anthrax, Minnesota, USA. Emerg. Infect. Dis. 2014, 20, 310–314. [Google Scholar] [CrossRef] [PubMed]
- Krantz, B.; Trivedi, A.; Cunningham, K.; Christensen, K.; Collier, R.J. Acid-induced unfolding of the amino-terminal domains of the lethal and edema factors of anthrax toxin. J. Mol. Biol. 2004, 344, 739–756. [Google Scholar] [CrossRef] [PubMed]
- Panchal, R.G.; Halverson, K.M.; Ribot, W.; Lane, D.; Kenny, T.; Abshire, T.G.; Ezzell, J.W.; Hoover, T.A.; Powell, B.; Little, S.; et al. Purified Bacillus anthracis lethal toxin complex formed in vitro and during infection exhibits functional and biological activity. J. Biol. Chem. 2005, 280, 10834–10839. [Google Scholar] [CrossRef] [PubMed]
- Naik, S.; Brock, S.; Akkaladevi, N.; Tally, J.; McGinn-Straub, W.; Zhang, N.; Gao, P.; Gogol, E.P.; Pentelute, B.L.; Collier, R.J.; et al. Monitoring the Kinetics of the pH-Driven Transition of the Anthrax Toxin Prepore to the Pore by Biolayer Interferometry and Surface Plasmon Resonance. Biochemistry 2013, 52, 6335–6347. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, T.T.; Rembert, K.; Conboy, J.C. Label-Free Detection of Drug-Membrane Association Using Ultraviolet-Visible Sum-Frequency Generation. J. Am. Chem. Soc. 2009, 131, 1401–1403. [Google Scholar] [CrossRef] [PubMed]
- Mogridge, J.; Cunningham, K.; Collier, R.J. Stoichiometry of anthrax toxin complexes. Biochemistry 2002, 41, 1079–1082. [Google Scholar] [CrossRef] [PubMed]
- Little, S.F.; Leppla, S.H.; Friedlander, A.M. Production and characterization of monoclonal antibodies against the lethal factor component of Bacillus anthracis lethal toxin. Infect. Immun. 1990, 58, 1606–1613. [Google Scholar] [PubMed]
- Little, S.F.; Leppla, S.H.; Cora, E. Production and characterization of monoclonal antibodies to the protective antigen component of Bacillus anthracis toxin. Infect. Immun. 1988, 56, 1807–1813. [Google Scholar] [PubMed]
- Maynard, J.A.; Maassen, C.B.M.; Leppla, S.H.; Brasky, K.; Patterson, J.L.; Iverson, B.L.; Georgiou, G. Protection against anthrax toxin by recombinant antibody fragments correlates with antigen affinity. Nat. Biotechnol. 2002, 20, 597–601. [Google Scholar] [CrossRef] [PubMed]
- Radjainia, M.; Hyun, J.-K.; Leysath, C.E.; Leppla, S.H.; Mitra, A.K. Anthrax toxin-neutralizing antibody reconfigures the protective antigen heptamer into a supercomplex. Proc. Natl. Acad. Sci. USA 2010, 107, 14070–14074. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Moayeri, M.; Purcell, R. Monoclonal antibody therapies against anthrax. Toxins 2011, 3, 1004–1019. [Google Scholar] [CrossRef] [PubMed]
- Little, S.F.; Novak, J.M.; Lowe, J.R.; Leppla, S.H.; Singh, Y.; Klimpel, K.R.; Lidgerding, B.C.; Friedlander, A.M. Characterization of lethal factor binding and cell receptor binding domains of protective antigen of Bacillus anthracis using monoclonal antibodies. Microbiology (Read. Engl.) 1996, 142, 707–715. [Google Scholar] [CrossRef] [PubMed]
- Moayeri, M.; Wiggins, J.F.; Leppla, S.H. Anthrax protective antigen cleavage and clearance from the blood of mice and rats. Infect. Immun. 2007, 75, 5175–5184. [Google Scholar] [CrossRef] [PubMed]
- Little, S.F.; Webster, W.M.; Fisher, D.E. Monoclonal antibodies directed against protective antigen of Bacillus anthracis enhance lethal toxin activityin vivo. FEMS Immunol. Med. Microbiol. 2011, 62, 11–22. [Google Scholar] [CrossRef] [PubMed]
- Nassi, S.; Collier, R.J.; Finkelstein, A. PA(63) channel of anthrax toxin: An extended beta-barrel. Biochemistry 2002, 41, 1445–1450. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, T.L. Three-dimensional model of the pore form of anthrax protective antigen. Structure and biological implications. J. Biomol. Struct. Dyn. 2004, 22, 253–265. [Google Scholar] [CrossRef] [PubMed]
- Yin, P.; Burns, C.J.; Osman, P.D.J.; Cornell, B.A. A tethered bilayer sensor containing alamethicin channels and its detection of amiloride based inhibitors. Biosens. Bioelectron. 2003, 18, 389–397. [Google Scholar] [CrossRef]
- Tun, T.N.; Jenkins, A.T.A. An electrochemical impedance study of the effect of pathogenic bacterial toxins on tethered bilayer lipid membrane. Electrochem. Commun. 2010, 12, 1411–1415. [Google Scholar] [CrossRef]
- Keizer, H.M.; Dorvel, B.R.; Andersson, M.; Fine, D.; Price, R.B.; Long, J.R.; Dodabalapur, A.; Köper, I.; Knoll, W.; Anderson, P.A.V.; et al. Functional ion channels in tethered bilayer membranes—Implications for biosensors. ChemBioChem 2007, 8, 1246–1250. [Google Scholar] [CrossRef] [PubMed]
- Kumar, P.; Colston, J.T.; Chambers, J.P.; Rael, E.D.; Valdes, J.J. Detection of botulinum toxin using an evanescent wave immunosensor. Biosens. Bioelectron. 1994, 9, 57–63. [Google Scholar] [CrossRef]
- Manich, M.; Knapp, O.; Gibert, M.; Maier, E.; Jolivet-Reynaud, C.; Geny, B.; Benz, R.; Popoff, M.R. Clostridium perfringens Delta Toxin Is Sequence Related to Beta Toxin, NetB, and Staphylococcus Pore-Forming Toxins, but Shows Functional Differences. PLoS ONE 2008, 3. [Google Scholar] [CrossRef] [PubMed]
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Silin, V.; Kasianowicz, J.J.; Michelman-Ribeiro, A.; Panchal, R.G.; Bavari, S.; Robertson, J.W.F. Biochip for the Detection of Bacillus anthracis Lethal Factor and Therapeutic Agents against Anthrax Toxins. Membranes 2016, 6, 36. https://doi.org/10.3390/membranes6030036
Silin V, Kasianowicz JJ, Michelman-Ribeiro A, Panchal RG, Bavari S, Robertson JWF. Biochip for the Detection of Bacillus anthracis Lethal Factor and Therapeutic Agents against Anthrax Toxins. Membranes. 2016; 6(3):36. https://doi.org/10.3390/membranes6030036
Chicago/Turabian StyleSilin, Vitalii, John J. Kasianowicz, Ariel Michelman-Ribeiro, Rekha G. Panchal, Sina Bavari, and Joseph W. F. Robertson. 2016. "Biochip for the Detection of Bacillus anthracis Lethal Factor and Therapeutic Agents against Anthrax Toxins" Membranes 6, no. 3: 36. https://doi.org/10.3390/membranes6030036
APA StyleSilin, V., Kasianowicz, J. J., Michelman-Ribeiro, A., Panchal, R. G., Bavari, S., & Robertson, J. W. F. (2016). Biochip for the Detection of Bacillus anthracis Lethal Factor and Therapeutic Agents against Anthrax Toxins. Membranes, 6(3), 36. https://doi.org/10.3390/membranes6030036