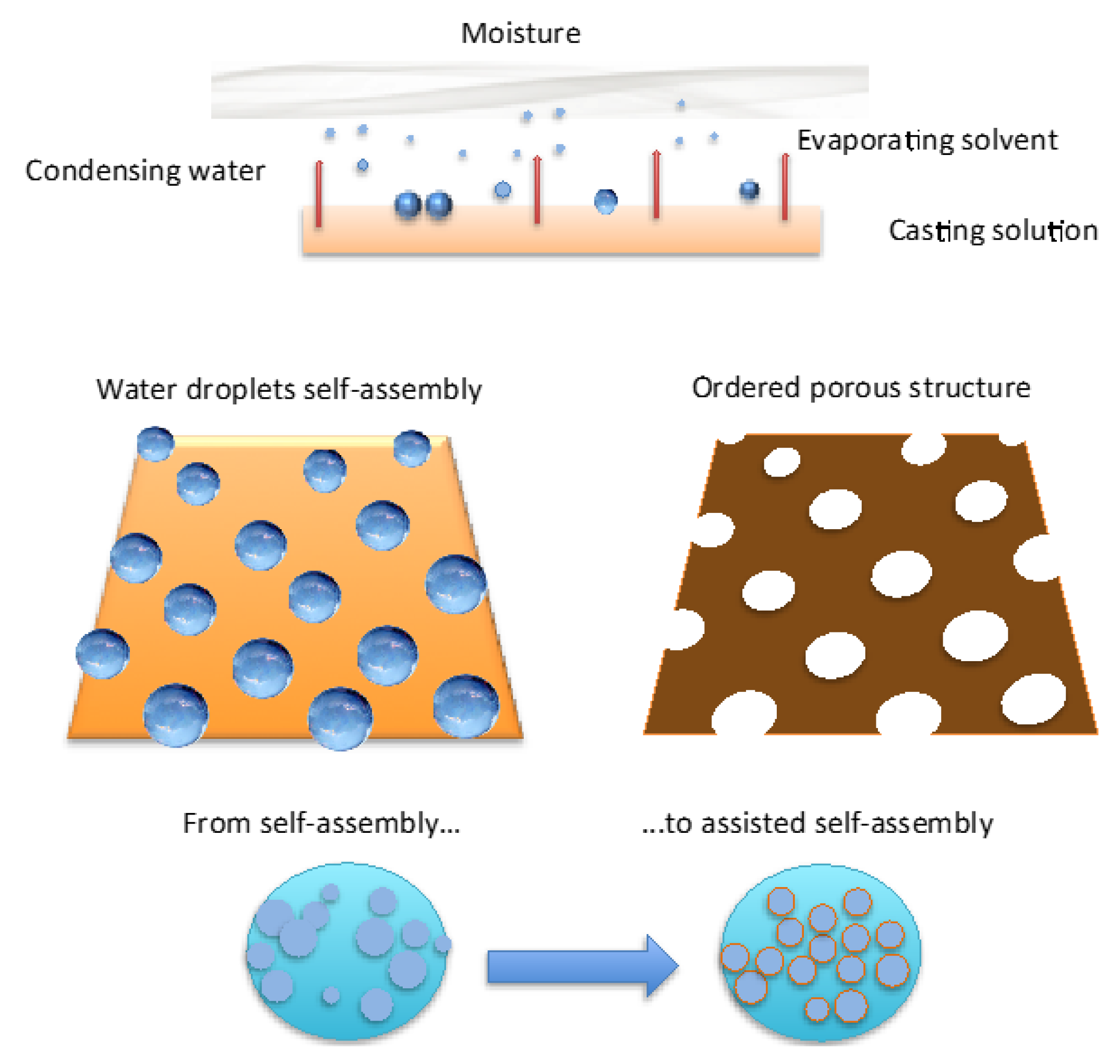
Briefly, when liquid films come in contact with humid air, water condensation on the liquid surface is induced for cooling effects due to solvent evaporation (
Figure 1). After nucleation, floating water droplets form more or less extended domains apart from each other. These domains are dragged by thermo-capillary forces that would direct the self-assembly to the area exposed to the humid air. At the same time the polymer has the ability to envelope the droplets when touching the solution and form a semisolid protective layer around each one. However, as the polymer layer is too weak and/or harsh and virulent collisions occur at the margin of the droplet rafts, coalescence phenomena take place shaping more or less extensive irregular and/or inhomogeneous regions upon the starkness of the impact (
Figure 2).
With this concern, the purpose of this work is to identify one way to reduce and prevent local disorder through controlled kinematic and thermodynamic forces in order to give new inputs to the production of this kind of membrane on a larger scale, especially for polymers exhibiting low ability to stabilize water droplets during flotation through the solution. The possibility to modulate the pore size within packed honeycomb geometries is explored as well.
For this reason, compounds, by virtue of their capability to rearrange and interact with different chemical environments, have been chosen as models to study. A common surfactant such as Tween20 has been used alone and in combination with alcohols, including ethanol, n-propanol, 2-propanol, and n-butanol, in order to increase the local kinematic viscosity of PES solutions, but also to generate chemical microenvironments favorable for hosting and stabilizing water droplets during condensation and flotation events. PES has been demonstrated to be poor in stabilizing droplets in the absence of surfactant, while extensively regular honeycomb textures have been obtained when surface-active molecules have been used to strengthen cooperative interfacial forces [
25].
3.1. Influence of the Surfactant on the Surface Structural Order
Initially, mixtures of PSU in DCM containing 2-propanol at 12 wt % and Tween20 at different content (10
−5–10
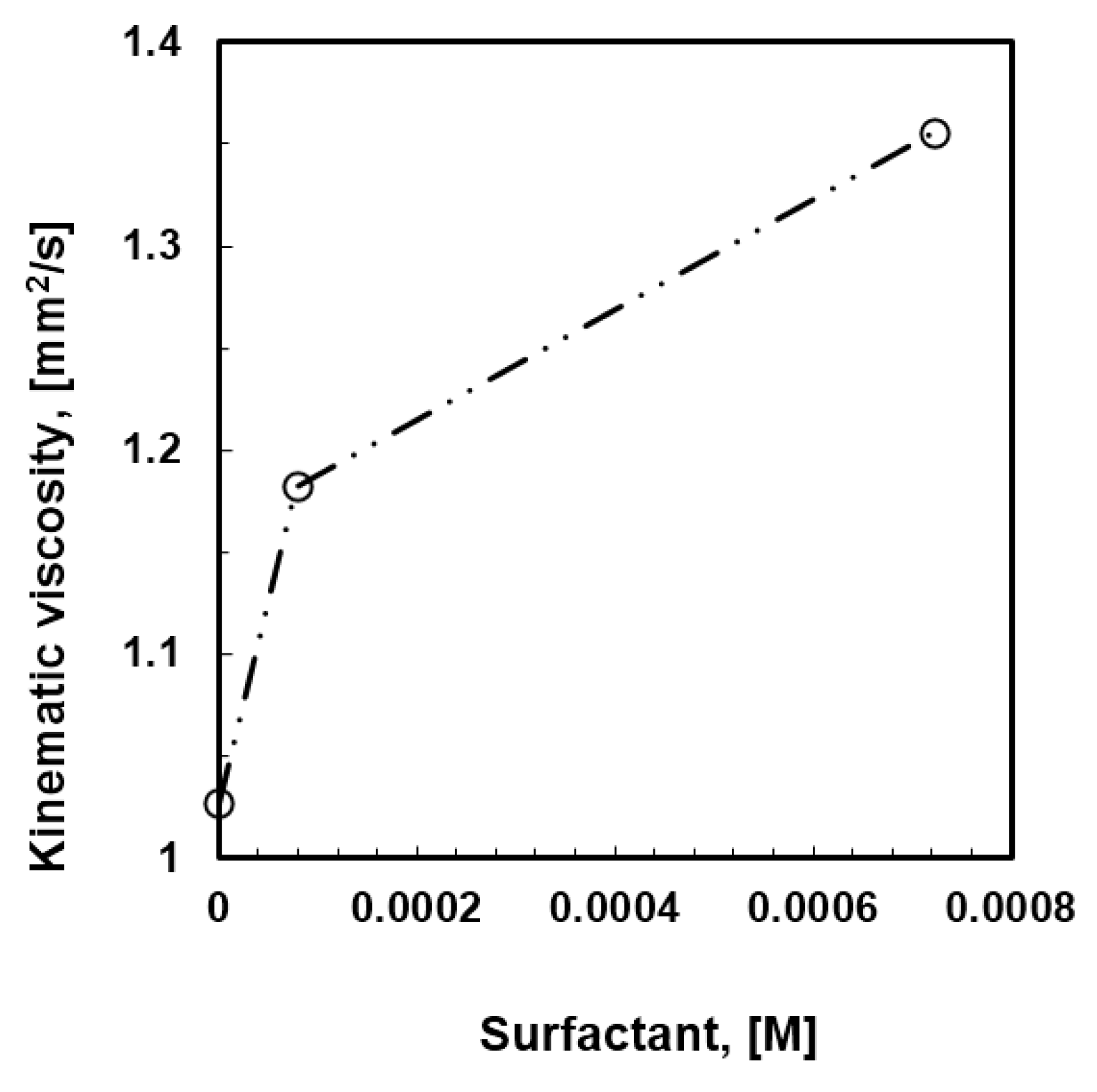
−3 M) were prepared. The behavior of the solutions with increasing content of surfactant has been examined when coming in contact with humid air, while kinetics and thermodynamic aspects have been analyzed as well. As the concentration of the surfactant increases, a local increase in gelation comes through the solution, resulting in an increased kinematic viscosity (
Figure 3).
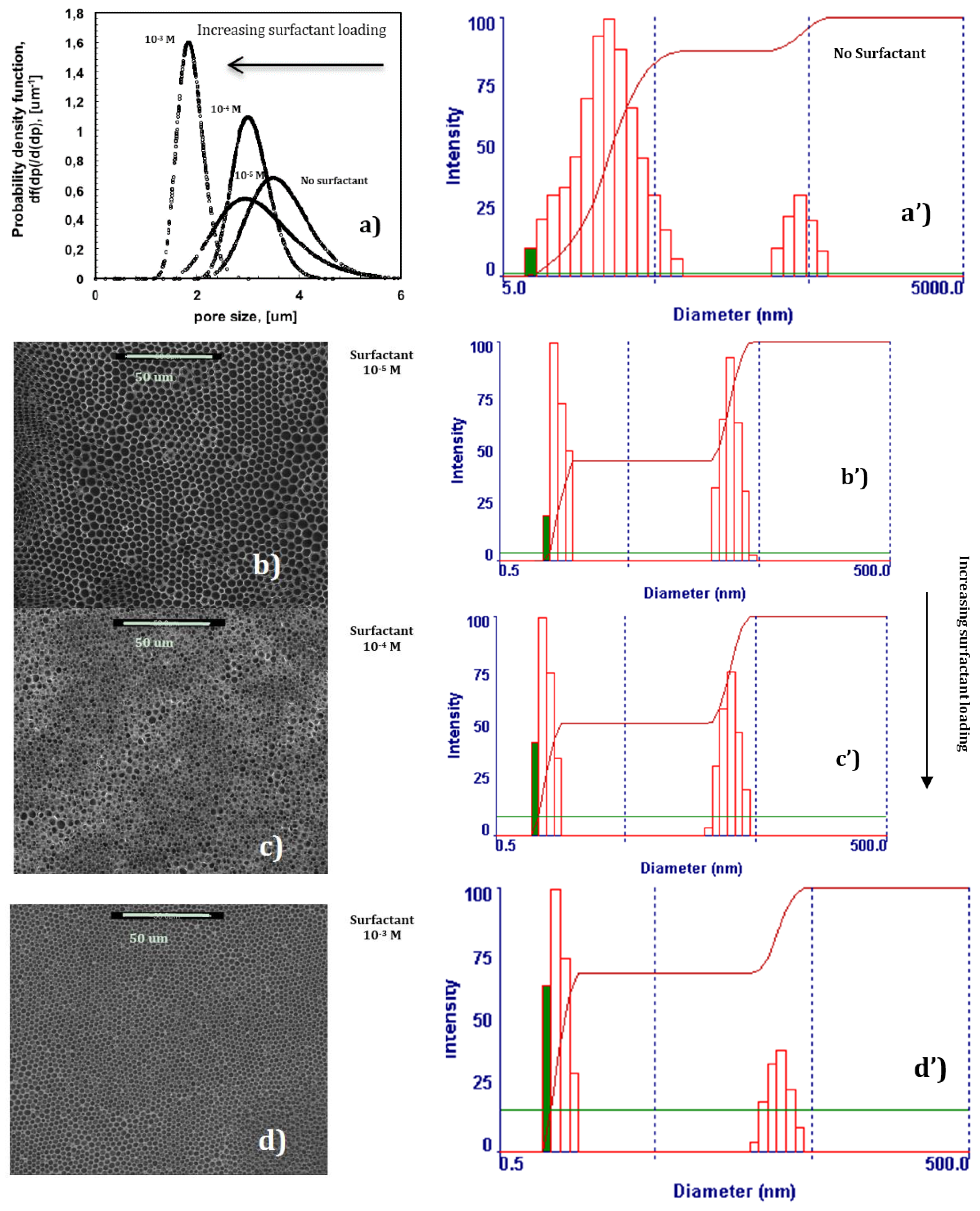
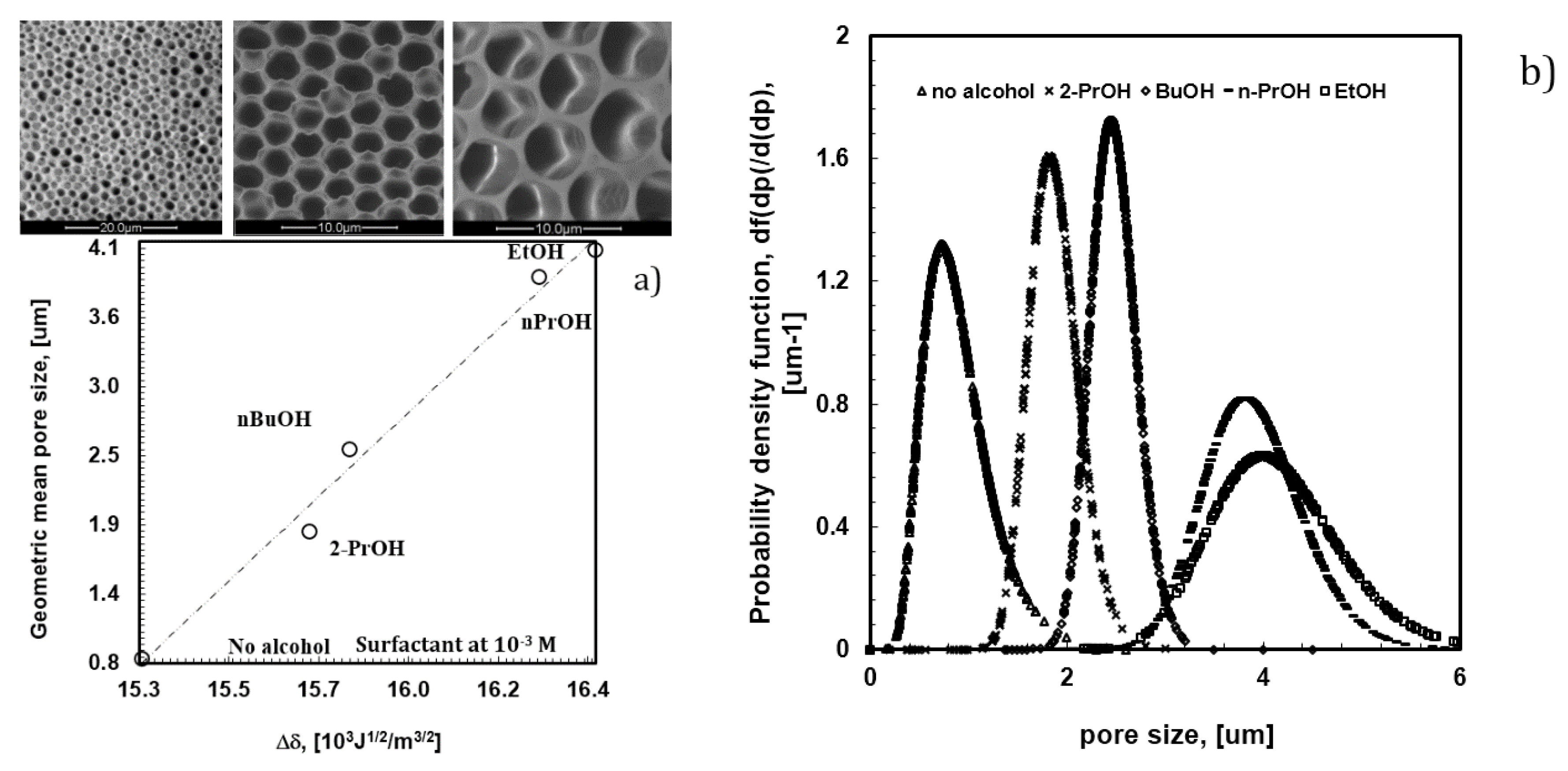
In the absence of surfactant, 2-propanol is unable to provide the assistance necessary to fully prevent local disorder for the polymer PES, as shown in
Figure 2a. Undesired broadness in the pore distribution takes place due to the coexistence of different porous domains, thus resulting in an average pore size of approximately 3.6 um (
Figure 4a).
Instead, a progressive addition of Tween20 to the solution leads to the gradual control of the dynamics of the water droplets, making the collisions softer. SEM micrographs show how larger and geometrically confused air bubbles progressively make space for more defined lattices as the content of the surfactant increases in the mixture (
Figure 4b–d). This suggests a slowing down of the droplets during motion, which avoids coalescence or uncontrolled growth rate at the margin of the single lattices. As a result, an enhanced degree of the order of the texture is obtained (
Figure 4a). In this context, the surfactant assists the polymer in the stabilization of droplets during self-assembly, producing additional fluid viscosity at the droplet-solution-droplet interface. The reduced kinetics prevents the nearest soft particles from merging in bigger bubbles, limiting or, at best, suppressing local disorder at the boundary of the droplet islands. Dynamic Light Scattering (DLS) experiments yield further indications about a cooperative interaction between the surfactant and polymer (
Figure 4a’–d’). In the absence of surfactant, two distinct populations of aggregates can be appreciated in solution within a size range of 5–5000 nm, the first one covering a broader range of heterogeneous assemblies (
Figure 4a’). The gradual addition of the surfactant in solution significantly reduces the broadness of two populations, decreasing the aggregates’ size by one order of magnitude, and leads to a gradual diminution of the second population in favor of the smallest one (
Figure 4b’–d’). Because of the complexity and heterogeneity of the mixtures, establishing the shape and type of the aggregates is not easy at this stage; however, it is undoubtedly due to the relationship between the increasing uniformity of aggregates in solution and the major order of the final texture, which can be regarded as the result of a cooperative action of the various components dispersed in the mixture. It is also relevant to observe how a mono-dispersive pore distribution matches with a gradual reduction of the pore size as the surfactant rises in content (
Figure 4).
Given that the radius of the droplets is time-dependent and proportional to
R <
t1/3 in the beginning, and to
R <
t in the end [
15], very narrow pore size distributions with the formation of a smaller pore size can be regarded as the result of a massive nucleation and a reduced droplet growth rate. There seems to be quick droplet saturation over the liquid surface in contact with moisture, while the boundary of each single droplet island becomes indiscernible at nearly the highest loading of surfactant (
Figure 4d). In this regard, the imprinting action of the droplets is exhausted when the lattice is formed over the entire surface area of the solution touching the moisture. However, this can also be considered a reasonable consequence of a very fast and assisted moisture uptake. Indeed, an increase in the overall surface tension—a value of 28.30 mJ/m
2 against 26.50 mJ/m
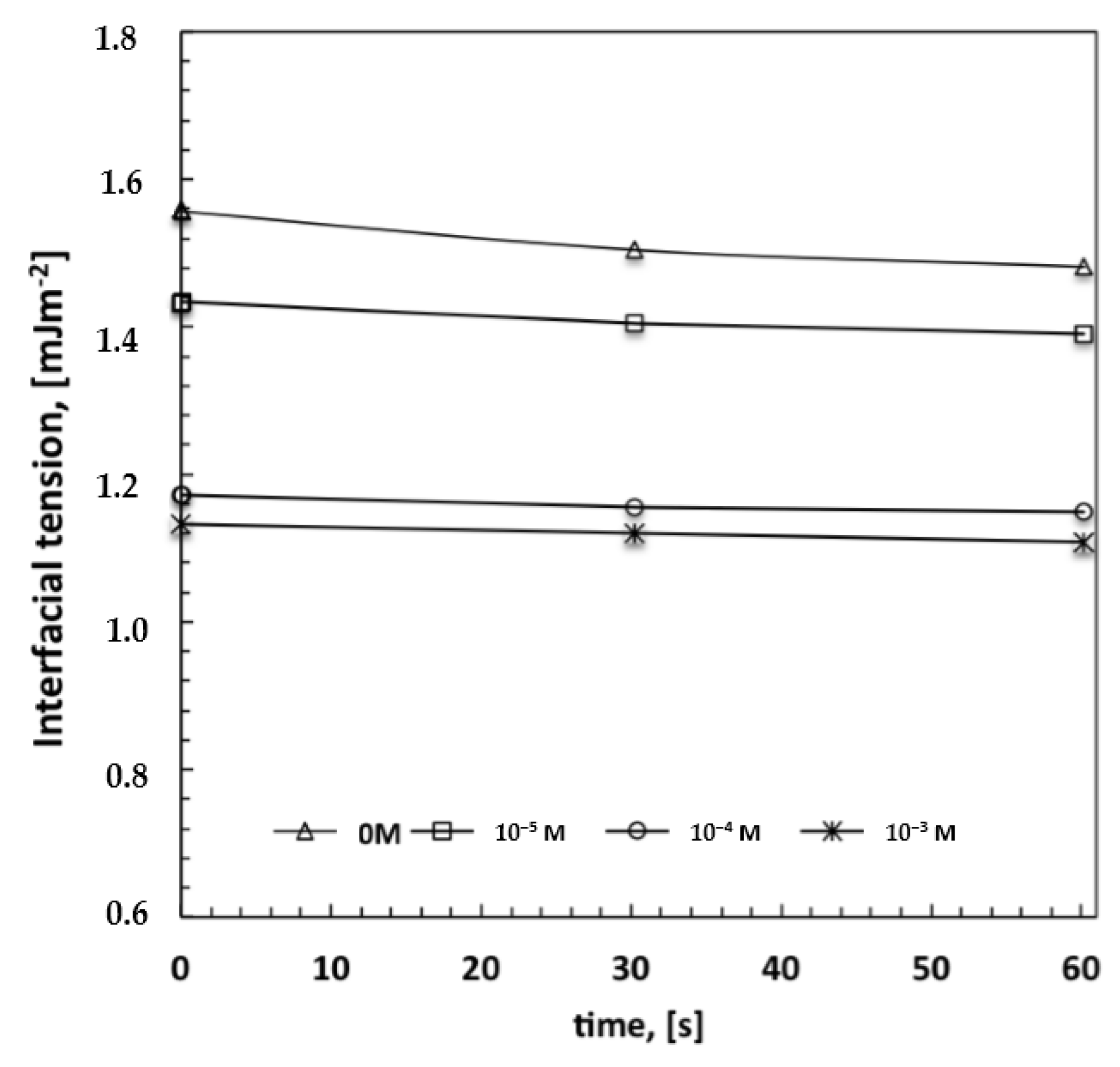
2 estimated for the pure solvent—has been measured for solutions at the highest amount of surfactant. This would suggest that a large number of polar head groups is directed outward from the surface and is prepared to interact with condensing droplets. In order to confirm the ability of the surfactant to interact favorably with the water droplets, interfacial tensions have been measured at the interface established between a single water droplet and the surrounding polymeric solution with increasing content of Tween20. The droplets have been automatically injected into the solution by using a syringe and the surface tension value has been measured according to the pendant drop method. Indeed, the decrease in the interfacial tension values with rising concentration of the surfactant confirms a tendency of the system to reach a minimum of energy (
Figure 5). This means that when the surfactant dissolved in the polymeric solution comes in contact with water droplets, the related polar heads establish attractive hydrophilic interactions at the interface while the hydrophobic tails are pointed towards the rest of the nonpolar solution, thus causing a decrease in the overall interfacial tension. These experimental findings are in full agreement with those found by Kojima
et al. [
23] about the ability of amphiphilic copolymers to establish hydrophilic interfacial forces with water droplets during the formation of honeycomb patterns. Herein, the ability of the surfactant to enhance the process of stabilization of the droplets becomes much stronger at higher concentrations, resulting in a higher uniformity of the structural order as well as in a gradual reduction of the pore size (
Figure 3a–d). This means that the larger availability of the surfactant leads to quicker water uptake and stabilization over the entire surface area of the solution exposed to humid air.
On this basis, there is a clear indication about the necessity to adjust the hydrophilic/hydrophobic balance in solution in order to move water droplets from self-assembly to assisted self-assembly, especially when using polymers with poor ability to rearrange themselves and interact at the interface of local different microenvironments.
It is relevant to observe how the hydrophilic/hydrophobic balance becomes somewhat marked at higher concentrations of surfactant, which is greater than that indicated as a necessary to reach the critical micelle concentration in a binary aqueous solution (CMC, 10
−2 mM) [
26]. In this respect, it must be stressed that the working chemical environment is rather different from the aqueous one, the mixture being nonpolar and containing four different components. This makes it difficult to unequivocally identify the aggregation state of the surfactant, especially in the presence of additional amphiphilic compounds such as alcohols, which allow intermolecular interactions to establish within hydrophilic and hydrophobic domains, causing important changes in CMC as well [
27]. Nevertheless, it is unquestionable that the increase in the concentration of surfactant causes a major number of monomers in proximity to the surface and in the bulk; these monomers could aggregate but also continue to migrate freely towards the surface, making polar heads promptly more available to interact with water and assist the dispersion of the aqueous phase in the continuous oil phase, as clearly confirmed by decreasing interfacial tension values (
Figure 5).
The result is a quicker formation of lattices from a larger number of smaller, stabilized water droplets, which leave an open pore size uniformly distributed over the entire surface area of the film. This clearly implies a predominance of droplet nucleation over the related growth rate.
3.2. Influence of the Alcohol Chain Length on the Pore Size
The pore size can be regarded as the result of a different balance between droplet nucleation and the growth rate steps. A massive droplet nucleation is, in fact, expected to lead to a very fast coverage of the surface with a formation of smaller pores, whereas a lengthy rearrangement extends the growth rate, yielding bigger air bubbles. In this respect, the chemistry of the polymeric mixture has been further changed with the purpose to direct the time-scale of nucleation and the growth rate, respectively. Alcohols with different structure (CH
2)
n = 2–4 containing the OH end group have been added to the mixture, keeping the concentration of the surfactant constant at 10
−4 M, in order to examine the effects of changing hydrophobic/hydrophilic balance on the pore formation. As shown in
Figure 6, the addition of alcohols to the polymeric mixture brings about a marked effect on the value of the overall surface free tension. The latter tends to increase with the length and bulky chain of the alcohol, while the pore size decreases from 4.0 to 0.8 um through the overall film surface.
It is instructive to stress that the increase in the surface free tension affects the scale of affinity, causing inevitably higher values of the solubility parameter (δ) of the solution. The latter is a thermodynamic indicator of the attractive or repulsive interactions established between two systems coming in contact [
28]. The difference between the solubility parameters (Δδ, 10
3J
1/2/m
3/2) of two media yields a clear indication about the level of affinity; thus, small differences indicate a great affinity, whereas large differences suggest a poor attraction. Concerning the systems investigated in this study, smaller differences have been estimated between the solubility parameters of water (72.86 mJ/m
2 at 20 °C) and the composite solutions when the lipophilic component of the alcohol overcomes the hydrophilic one. In this case, a smaller pore size is obtained (
Figure 5a). Differently, a larger pore size is measured for films prepared from solutions containing alcohols with higher polar character (
Figure 6a).
Undeniably, alcohols with shorter tails exhibit increased polarity and have a higher ability to interact with the polar heads of the surfactant, thus reducing their availability towards water droplets. On the contrary, alcohols with longer and bulky chains exhibit more amphiphilic character, taking their dissolution closer to the hydrophobic regions of the surfactant, where dispersive cooperative intermolecular interactions are better established. This implies a larger availability of the polar heads to face water droplets. As a result, a quicker nucleation of droplets with formation of smaller pore size is obtained when polar heads of the surfactant are more available. This can be envisaged as a direct consequence of a higher hydrophobic molecular interaction established into the bulk of the polymeric solution.
In this regard, it is also relevant to observe that such an availability of the head polar groups at the solution surface-air interface becomes much higher in the absence of alcohols. This suggests a decisive role of the alcohol in the rearrangement of the surfactant at the water-solution interface. Indeed, the difference between the solubility parameters of water and the casting solution is somewhat low in absence of alcohol (
Figure 6a). In this case, intermolecular interactions between alcohols and surfactant fail necessarily and a larger number of free monomers in solution orient the polar part outward, yielding major availability to interact with floating droplets. Under these conditions, a larger number of small droplets are formed and stabilized over the entire surface area in contact with humidity. The consequence is a massive nucleation, which leads to highly ordered textures with pores of 0.8 um (
Figure 6b).
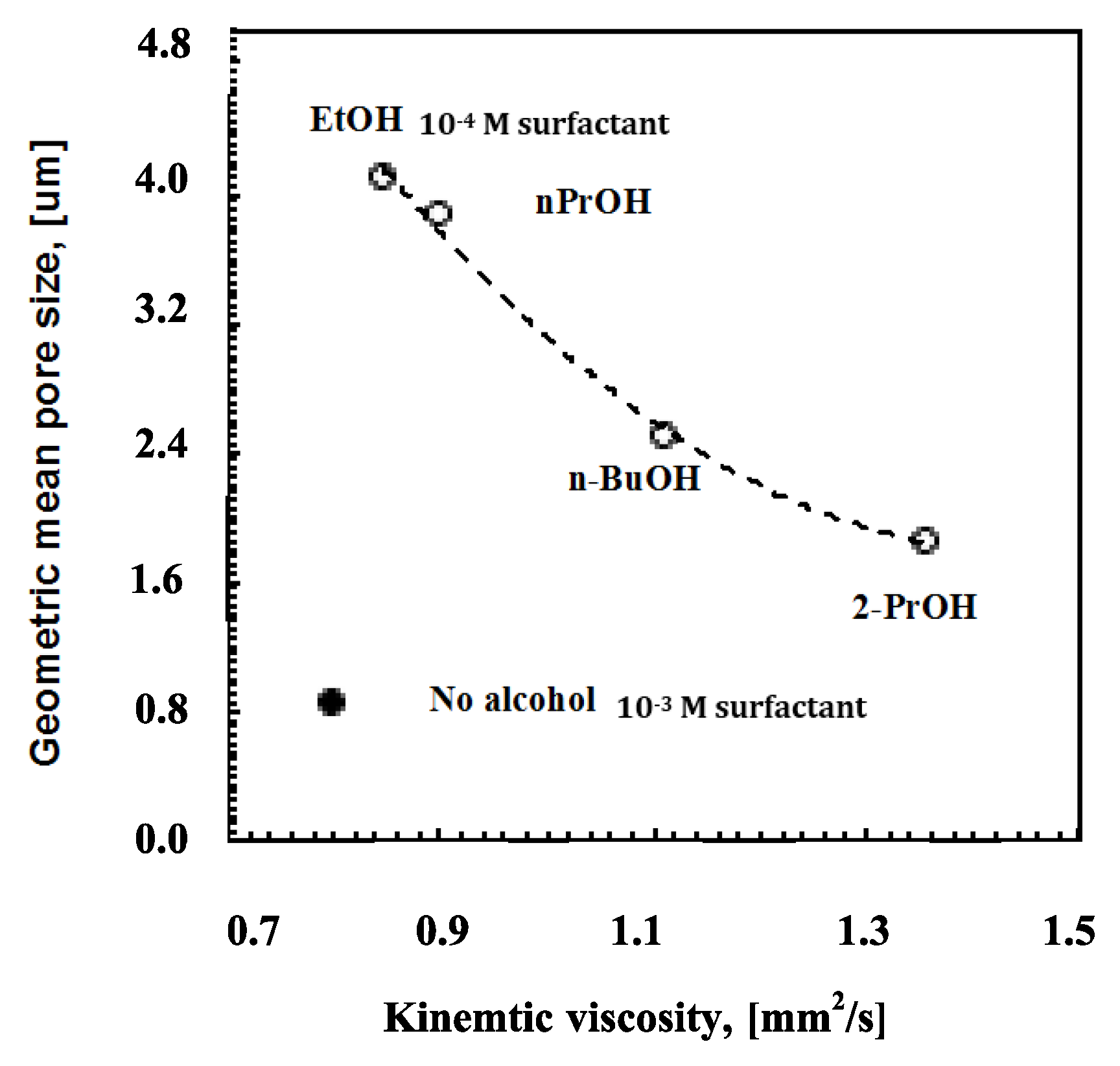
Again, it is also relevant to examine the incidence of the alcohol length chain on the kinematic viscosity.
Figure 7 shows how the rising molecular weight together with the bulky structure of the alcohol causes an effective increase in the solution viscosity (
Figure 6).
This is expected to further enhance the capability of encapsulation of the polymeric solution, thereby reducing the risk of coalescence and harsh collisions.
The viscosity factor becomes, however, non-influential in the absence of alcohols; the rate of moisture uptake and coverage for the entire surface area rather than speedy droplet flotation seems to decide the smaller pore arrangement in ordered textures. Comparing a solution of PSU/DMC containing surfactant at 10
−4 M with ethanol at 12 wt % and a solution of PSU/DMC containing surfactant at 10
−3 M without alcohol, similar values of kinematic viscosity can be appreciated (
Figure 7); however, a significant reduction of around 80% is observed for the pore size as the polymeric solution contains surfactant alone. This suggests that different mechanisms can take place during droplet self-assembly. The surfactant favors moisture uptake and a quicker nucleation rate, whereas the alcohol competes with water in the establishment of intermolecular interactions with the surfactant, reducing the related degree of freedom with the effect of extending the growth rate of the floating droplets depending on the intrinsic polar character of the mixture.
On this basis, the surfactant seems to have a decisive and predominant role in the uptake and stabilization of floating water droplets, whereas the alcohol affects the time scale, resulting in a modular pore size.
It is unquestionable that changes in the hydrophilic-hydrophobic balance cause a competition between droplet nucleation and growth rate steps. Using other classes of amphiphilic compounds, changes in this balance are expected to further modify the time scale with consequences on the final texture of the polymeric porous film [
13].
The intent of this work is to demonstrate the necessity to move from traditional self-assembly to assisted self-assembly approaches as a precondition to make the technique scalable, thus preserving structural order and yielding uniformly modulated pore size at longer range. Of course, the precondition for a successful scale-up has to pass through the adjustment of thermodynamic and kinetic parameters and, consequently, the manipulation of bulk properties of the solutions used, enabling one to contrast undesired effects due to low ability of the polymer to stabilize the droplets but also to the frequent sensitivity of the droplet assembly to little changes in the external environment.