The Road not Taken: Less Traveled Roads from the TGN to the Plasma Membrane
Abstract
:1. Introduction
2. Traffic along the Secretory Pathway
2.1. Getting to the trans-Golgi
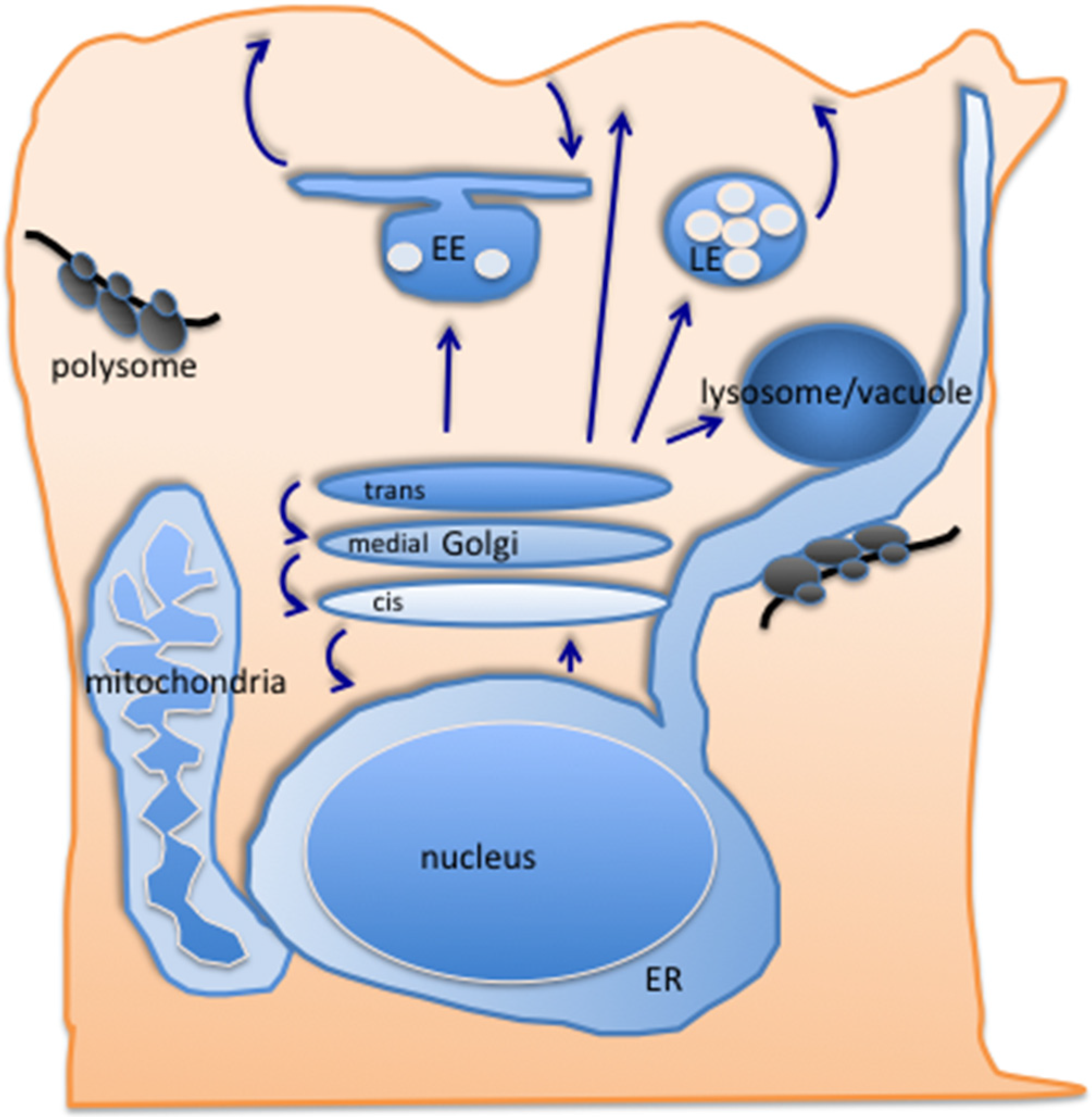
2.2. Exit from the Golgi—The Conventional Routes
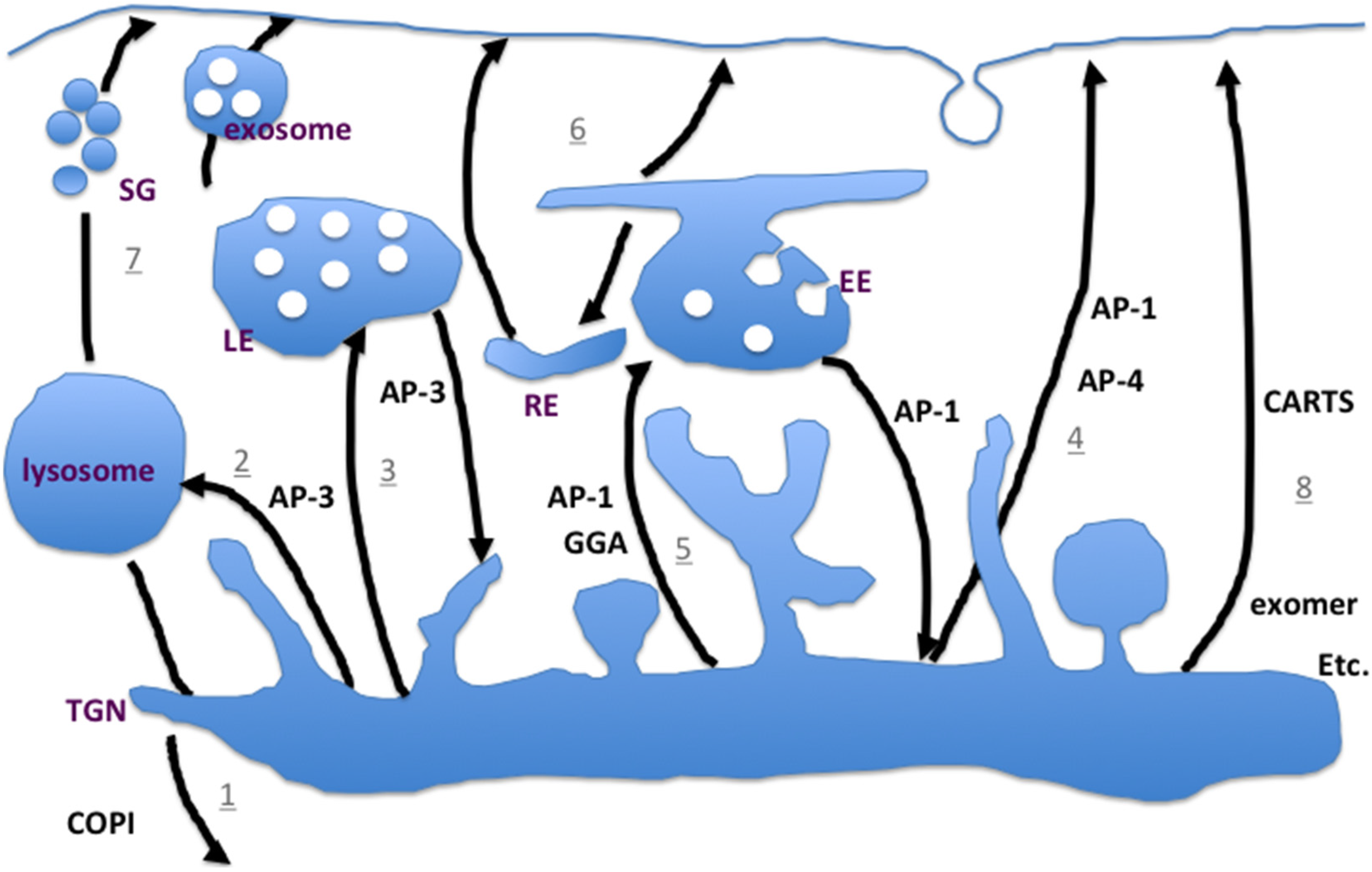
2.3. Less Studied—But not Less Important—Pathways to the Plasma Membrane
2.4. Exomer-Dependent Transport to the Plasma Membrane
2.5. Exomer-Dependent Cargoes
Acknowledgments
Conflicts of Interest
References
- Tomas, A.; Futter, C.E.; Eden, E.R. EGF receptor trafficking: Consequences for signaling and cancer. Trends Cell Biol. 2014, 24, 26–34. [Google Scholar] [CrossRef] [PubMed]
- Ziolkowska, N.E.; Christiano, R.; Walther, T.C. Organized living: Formation mechanisms and functions of plasma membrane domains in yeast. Trends Cell Biol. 2012, 22, 151–158. [Google Scholar] [CrossRef] [PubMed]
- Cohen, A.W.; Combs, T.P.; Scherer, P.E.; Lisanti, M.P. Role of caveolin and caveolae in insulin signaling and diabetes. Am. J. Physiol. Endocrinol. Metab. 2003, 285, E1151–E1160. [Google Scholar] [CrossRef] [PubMed]
- Lehmann, G.L.; Benedicto, I.; Philp, N.J.; Rodriguez-Boulan, E. Plasma membrane protein polarity and trafficking in rpe cells: Past, present and future. Exp. Eye Res. 2014, 126, 5–15. [Google Scholar] [CrossRef] [PubMed]
- Rizzo, V.; Richman, J.; Puthanveettil, S.V. Dissecting mechanisms of brain aging by studying the intrinsic excitability of neurons. Front. Aging Neurosci. 2014, 6, 337. [Google Scholar] [PubMed]
- Free, S.J. Fungal cell wall organization and biosynthesis. Adv. Genet. 2013, 81, 33–82. [Google Scholar] [PubMed]
- Bonnans, C.; Chou, J.; Werb, Z. Remodelling the extracellular matrix in development and disease. Nat. Rev. Mol. Cell Biol. 2014, 15, 786–801. [Google Scholar] [CrossRef] [PubMed]
- Ebel, J.; Beuter, S.; Wuchter, J.; Kriebel, M.; Volkmer, H. Organisation and control of neuronal connectivity and myelination by cell adhesion molecule neurofascin. Adv. Neurobiol. 2014, 8, 231–247. [Google Scholar] [PubMed]
- De Beco, S.; Amblard, F.; Coscoy, S. New insights into the regulation of e-cadherin distribution by endocytosis. Int. Rev. Cell Mol. Biol. 2012, 295, 63–108. [Google Scholar] [PubMed]
- Kupfer, L.; Hinrichs, W.; Groschup, M.H. Prion protein misfolding. Curr. Mol. Med. 2009, 9, 826–835. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.C.; Miller, E.A.; Goldberg, J.; Orci, L.; Schekman, R. Bi-directional protein transport between the ER and Golgi. Annu. Rev. Cell Dev. Biol. 2004, 20, 87–123. [Google Scholar] [CrossRef] [PubMed]
- Kondylis, V.; Rabouille, C. A novel role for dp115 in the organization of tER sites in drosophila. J. Cell Biol. 2003, 162, 185–198. [Google Scholar] [CrossRef] [PubMed]
- Kurokawa, K.; Okamoto, M.; Nakano, A. Contact of cis-Golgi with ER exit sites executes cargo capture and delivery from the ER. Nat. Commun. 2014, 5, 3653. [Google Scholar] [CrossRef] [PubMed]
- Witte, K.; Schuh, A.L.; Hegermann, J.; Sarkeshik, A.; Mayers, J.R.; Schwarze, K.; Yates, J.R., 3rd; Eimer, S.; Audhya, A. Tfg-1 function in protein secretion and oncogenesis. Nat. Cell Biol. 2011, 13, 550–558. [Google Scholar] [CrossRef]
- Barlowe, C.; Orci, L.; Yeung, T.; Hosobuchi, M.; Hamamoto, S.; Salama, N.; Rexach, M.F.; Ravazzola, M.; Amherdt, M.; Schekman, R. COPII: A membrane coat formed by sec proteins that drive vesicle budding from the endoplasmic reticulum. Cell 1994, 77, 895–907. [Google Scholar] [CrossRef] [PubMed]
- Jaschke, A.; Chung, B.; Hesse, D.; Kluge, R.; Zahn, C.; Moser, M.; Petzke, K.J.; Brigelius-Flohe, R.; Puchkov, D.; Koepsell, H.; et al. The GTPase ARFRP1 controls the lipidation of chylomicrons in the Golgi of the intestinal epithelium. Hum. Mol. Genet. 2012, 21, 3128–3142. [Google Scholar] [CrossRef] [PubMed]
- Saito, K.; Chen, M.; Bard, F.; Chen, S.; Zhou, H.; Woodley, D.; Polischuk, R.; Schekman, R.; Malhotra, V. TANGO1 facilitates cargo loading at endoplasmic reticulum exit sites. Cell 2009, 136, 891–902. [Google Scholar] [CrossRef] [PubMed]
- Saito, K.; Yamashiro, K.; Ichikawa, Y.; Erlmann, P.; Kontani, K.; Malhotra, V.; Katada, T. cTAGE5 mediates collagen secretion through interaction with TANGO1 at endoplasmic reticulum exit sites. Mol. Biol. Cell 2011, 22, 2301–2308. [Google Scholar] [CrossRef] [PubMed]
- Siddiqi, S.; Saleem, U.; Abumrad, N.A.; Davidson, N.O.; Storch, J.; Siddiqi, S.A.; Mansbach, C.M., 2nd. A novel multiprotein complex is required to generate the prechylomicron transport vesicle from intestinal ER. J. Lipid Res. 2010, 51, 1918–1928. [Google Scholar] [CrossRef] [PubMed]
- Venditti, R.; Scanu, T.; Santoro, M.; Di Tullio, G.; Spaar, A.; Gaibisso, R.; Beznoussenko, G.V.; Mironov, A.A.; Mironov, A., Jr.; Zelante, L.; et al. Sedlin controls the ER export of procollagen by regulating the Sar1 cycle. Science 2012, 337, 1668–1672. [Google Scholar] [CrossRef] [PubMed]
- Stanley, P. Golgi glycosylation. Cold Spring Harb. Perspect. Biol. 2011, 3. [Google Scholar] [CrossRef]
- Beznoussenko, G.V.; Parashuraman, S.; Rizzo, R.; Polishchuk, R.; Martella, O.; Di Giandomenico, D.; Fusella, A.; Spaar, A.; Sallese, M.; Capestrano, M.G.; et al. Transport of soluble proteins through the Golgi occurs by diffusion via continuities across cisternae. eLife 2014, 3. [Google Scholar] [CrossRef]
- Glick, B.S.; Luini, A. Models for Golgi traffic: A critical assessment. Cold Spring Harb. Perspect. Biol. 2011, 3, a005215. [Google Scholar] [CrossRef] [PubMed]
- Pfeffer, S.R. Hopping rim to rim through the golgi. eLife 2013, 2, e00903. [Google Scholar] [CrossRef] [PubMed]
- Papanikou, E.; Glick, B.S. Golgi compartmentation and identity. Curr. Opin. Cell Biol. 2014, 29, 74–81. [Google Scholar] [CrossRef] [PubMed]
- Hirschberg, K.; Miller, C.M.; Ellenberg, J.; Presley, J.F.; Siggia, E.D.; Phair, R.D.; Lippincott-Schwartz, J. Kinetic analysis of secretory protein traffic and characterization of Golgi to plasma membrane transport intermediates in living cells. J. Cell Biol. 1998, 143, 1485–1503. [Google Scholar] [CrossRef] [PubMed]
- Polishchuk, E.V.; Di Pentima, A.; Luini, A.; Polishchuk, R.S. Mechanism of constitutive export from the Golgi: Bulk flow via the formation, protrusion, and en bloc cleavage of large trans-Golgi network tubular domains. Mol. Biol. Cell 2003, 14, 4470–4485. [Google Scholar] [CrossRef] [PubMed]
- Polishchuk, R.S.; San Pietro, E.; Di Pentima, A.; Tete, S.; Bonifacino, J.S. Ultrastructure of long-range transport carriers moving from the trans-Golgi network to peripheral endosomes. Traffic 2006, 7, 1092–1103. [Google Scholar] [CrossRef] [PubMed]
- Puertollano, R. Clathrin-mediated transport: Assembly required. Workshop on molecular mechanisms of vesicle selectivity. EMBO Rep. 2004, 5, 942–946. [Google Scholar] [CrossRef] [PubMed]
- Anitei, M.; Hoflack, B. Bridging membrane and cytoskeleton dynamics in the secretory and endocytic pathways. Nat. Cell Biol. 2012, 14, 11–19. [Google Scholar] [CrossRef]
- Bard, F.; Malhotra, V. The formation of TGN-to-plasma-membrane transport carriers. Annu. Rev. Cell Dev. Biol. 2006, 22, 439–455. [Google Scholar] [CrossRef] [PubMed]
- De Matteis, M.A.; Luini, A. Exiting the Golgi complex. Nat. Rev. Mol. Cell Biol. 2008, 9, 273–284. [Google Scholar]
- Egea, G.; Lazaro-Dieguez, F.; Vilella, M. Actin dynamics at the golgi complex in mammalian cells. Curr. Opin. Cell Biol. 2006, 18, 168–178. [Google Scholar] [CrossRef] [PubMed]
- Cowles, C.R.; Odorizzi, G.; Payne, G.S.; Emr, S.D. The AP-3 adaptor complex is essential for cargo-selective transport to the yeast vacuole. Cell 1997, 91, 109–118. [Google Scholar] [CrossRef] [PubMed]
- Dell’Angelica, E.C.; Shotelersuk, V.; Aguilar, R.C.; Gahl, W.A.; Bonifacino, J.S. Altered trafficking of lysosomal proteins in Hermansky-Pudlak syndrome due to mutations in the beta 3A subunit of the AP-3 adaptor. Mol. Cell 1999, 3, 11–21. [Google Scholar] [CrossRef] [PubMed]
- Simpson, F.; Peden, A.A.; Christopoulou, L.; Robinson, M.S. Characterization of the adaptor-related protein complex, AP-3. J. Cell Biol. 1997, 137, 835–845. [Google Scholar] [CrossRef] [PubMed]
- Le Borgne, R.; Alconada, A.; Bauer, U.; Hoflack, B. The mammalian AP-3 adaptor-like complex mediates the intracellular transport of lysosomal membrane glycoproteins. J. Biol. Chem. 1998, 273, 29451–29461. [Google Scholar]
- Stepp, J.D.; Huang, K.; Lemmon, S.K. The yeast adaptor protein complex, AP-3, is essential for the efficient delivery of alkaline phosphatase by the alternate pathway to the vacuole. J. Cell Biol. 1997, 139, 1761–1774. [Google Scholar] [CrossRef] [PubMed]
- Kent, H.M.; Evans, P.R.; Schafer, I.B.; Gray, S.R.; Sanderson, C.M.; Luzio, J.P.; Peden, A.A.; Owen, D.J. Structural basis of the intracellular sorting of the SNARE VAMP7 by the AP3 adaptor complex. Dev. Cell 2012, 22, 979–988. [Google Scholar] [CrossRef] [PubMed]
- D’Souza, R.S.; Semus, R.; Billings, E.A.; Meyer, C.B.; Conger, K.; Casanova, J.E. Rab4 orchestrates a small GTPase cascade for recruitment of adaptor proteins to early endosomes. Curr. Biol. 2014, 24, 1187–1198. [Google Scholar] [CrossRef] [PubMed]
- Dores, M.R.; Paing, M.M.; Lin, H.; Montagne, W.A.; Marchese, A.; Trejo, J. AP-3 regulates PAR1 ubiquitin-independent MVB/lysosomal sorting via an ALIX-mediated pathway. Mol. Biol. Cell 2012, 23, 3612–3623. [Google Scholar] [CrossRef] [PubMed]
- Bendor, J.; Lizardi-Ortiz, J.E.; Westphalen, R.I.; Brandstetter, M.; Hemmings, H.C., Jr.; Sulzer, D.; Flajolet, M.; Greengard, P. AGAP1/AP-3-dependent endocytic recycling of M5 muscarinic receptors promotes dopamine release. EMBO J. 2010, 29, 2813–2826. [Google Scholar] [CrossRef] [PubMed]
- Asensio, C.S.; Sirkis, D.W.; Edwards, R.H. Rnai screen identifies a role for adaptor protein AP-3 in sorting to the regulated secretory pathway. J. Cell Biol. 2010, 191, 1173–1187. [Google Scholar] [CrossRef] [PubMed]
- Chapuy, B.; Tikkanen, R.; Muhlhausen, C.; Wenzel, D.; von Figura, K.; Honing, S. AP-1 and AP-3 mediate sorting of melanosomal and lysosomal membrane proteins into distinct post-Golgi trafficking pathways. Traffic 2008, 9, 1157–1172. [Google Scholar] [CrossRef] [PubMed]
- Bonifacino, J.S. Adaptor proteins involved in polarized sorting. J. Cell Biol. 2014, 204, 7–17. [Google Scholar] [CrossRef] [PubMed]
- Farias, G.G.; Cuitino, L.; Guo, X.; Ren, X.; Jarnik, M.; Mattera, R.; Bonifacino, J.S. Signal-mediated, AP-1/clathrin-dependent sorting of transmembrane receptors to the somatodendritic domain of hippocampal neurons. Neuron 2012, 75, 810–823. [Google Scholar] [CrossRef] [PubMed]
- Gravotta, D.; Carvajal-Gonzalez, J.M.; Mattera, R.; Deborde, S.; Banfelder, J.R.; Bonifacino, J.S.; Rodriguez-Boulan, E. The clathrin adaptor AP-1A mediates basolateral polarity. Dev. Cell 2012, 22, 811–823. [Google Scholar] [CrossRef] [PubMed]
- Robinson, M.S. Adaptable adaptors for coated vesicles. Trends Cell Biol. 2004, 14, 167–174. [Google Scholar] [CrossRef] [PubMed]
- Skorobogata, O.; Escobar-Restrepo, J.M.; Rocheleau, C.E. An AGEF-1/Arf GTPase/AP-1 ensemble antagonizes LET-23 EGFR basolateral localization and signaling during C. elegans vulva induction. PLoS Genet. 2014, 10, e1004728. [Google Scholar] [CrossRef] [PubMed]
- Hirst, J.; Borner, G.H.; Antrobus, R.; Peden, A.A.; Hodson, N.A.; Sahlender, D.A.; Robinson, M.S. Distinct and overlapping roles for AP-1 and GGAs revealed by the “knocksideways” system. Curr. Biol. 2012, 22, 1711–1716. [Google Scholar] [CrossRef] [PubMed]
- Valdivia, R.H.; Baggott, D.; Chuang, J.S.; Schekman, R.W. The yeast clathrin adaptor protein complex 1 is required for the efficient retention of a subset of late Golgi membrane proteins. Dev. Cell 2002, 2, 283–294. [Google Scholar] [CrossRef] [PubMed]
- Duncan, M.C.; Costaguta, G.; Payne, G.S. Yeast epsin-related proteins required for Golgi-endosome traffic define a gamma-adaptin ear-binding motif. Nat. Cell Biol. 2003, 5, 77–81. [Google Scholar] [CrossRef] [PubMed]
- Friant, S.; Pecheur, E.I.; Eugster, A.; Michel, F.; Lefkir, Y.; Nourrisson, D.; Letourneur, F. Ent3p is a PtdIns(3,5)P2 effector required for protein sorting to the multivesicular body. Dev. Cell 2003, 5, 499–511. [Google Scholar] [CrossRef] [PubMed]
- Puertollano, R.; van der Wel, N.N.; Greene, L.E.; Eisenberg, E.; Peters, P.J.; Bonifacino, J.S. Morphology and dynamics of clathrin/GGA1-coated carriers budding from the trans-Golgi network. Mol. Biol. Cell 2003, 14, 1545–1557. [Google Scholar] [CrossRef] [PubMed]
- Robinson, M.S.; Bonifacino, J.S. Adaptor-related proteins. Curr. Opin. Cell Biol. 2001, 13, 444–453. [Google Scholar] [CrossRef] [PubMed]
- Bonifacino, J.S.; Traub, L.M. Signals for sorting of transmembrane proteins to endosomes and lysosomes. Annu. Rev. Biochem. 2003, 72, 395–447. [Google Scholar] [CrossRef] [PubMed]
- Misra, S.; Puertollano, R.; Kato, Y.; Bonifacino, J.S.; Hurley, J.H. Structural basis for acidic-cluster-dileucine sorting-signal recognition by VHS domains. Nature 2002, 415, 933–937. [Google Scholar] [CrossRef] [PubMed]
- Owen, D.J.; Collins, B.M.; Evans, P.R. Adaptors for clathrin coats: Structure and function. Annu. Rev. Cell Dev. Biol. 2004, 20, 153–191. [Google Scholar] [CrossRef] [PubMed]
- Scott, P.M.; Bilodeau, P.S.; Zhdankina, O.; Winistorfer, S.C.; Hauglund, M.J.; Allaman, M.M.; Kearney, W.R.; Robertson, A.D.; Boman, A.L.; Piper, R.C. GGA proteins bind ubiquitin to facilitate sorting at the trans-Golgi network. Nat. Cell Biol. 2004, 6, 252–259. [Google Scholar] [CrossRef] [PubMed]
- Daboussi, L.; Costaguta, G.; Payne, G.S. Phosphoinositide-mediated clathrin adaptor progression at the trans-Golgi network. Nat. Cell Biol. 2012, 14, 239–248. [Google Scholar] [CrossRef] [PubMed]
- Godi, A.; Di Campli, A.; Konstantakopoulos, A.; Di Tullio, G.; Alessi, D.R.; Kular, G.S.; Daniele, T.; Marra, P.; Lucocq, J.M.; De Matteis, M.A. FAPPs control Golgi-to-cell-surface membrane traffic by binding to ARF and PtdIns(4)p. Nat. Cell Biol. 2004, 6, 393–404. [Google Scholar] [CrossRef] [PubMed]
- Klemm, R.W.; Ejsing, C.S.; Surma, M.A.; Kaiser, H.J.; Gerl, M.J.; Sampaio, J.L.; de Robillard, Q.; Ferguson, C.; Proszynski, T.J.; Shevchenko, A.; et al. Segregation of sphingolipids and sterols during formation of secretory vesicles at the trans-Golgi network. J. Cell Biol. 2009, 185, 601–612. [Google Scholar] [CrossRef] [PubMed]
- Surma, M.A.; Klose, C.; Simons, K. Lipid-dependent protein sorting at the trans-Golgi network. Biochim. Biophys. Acta 2012, 1821, 1059–1067. [Google Scholar] [CrossRef] [PubMed]
- Von Blume, J.; Alleaume, A.M.; Kienzle, C.; Carreras-Sureda, A.; Valverde, M.; Malhotra, V. Cab45 is required for Ca(2+)-dependent secretory cargo sorting at the trans-Golgi network. J. Cell Biol. 2012, 199, 1057–1066. [Google Scholar]
- Park, Y.; Kim, K.T. Short-term plasticity of small synaptic vesicle (SSV) and large dense-core vesicle (LDCV) exocytosis. Cell. Signal. 2009, 21, 1465–1470. [Google Scholar] [CrossRef] [PubMed]
- Guo, Y.; Zanetti, G.; Schekman, R. A novel GTP-binding protein-adaptor protein complex responsible for export of Vangl2 from the trans Golgi network. eLife 2013, 2, e00160. [Google Scholar] [PubMed]
- Wakana, Y.; van Galen, J.; Meissner, F.; Scarpa, M.; Polishchuk, R.S.; Mann, M.; Malhotra, V. A new class of carriers that transport selective cargo from the trans Golgi network to the cell surface. EMBO J. 2012, 31, 3976–3990. [Google Scholar] [CrossRef] [PubMed]
- Richter, S.; Kientz, M.; Brumm, S.; Nielsen, M.E.; Park, M.; Gavidia, R.; Krause, C.; Voss, U.; Beckmann, H.; Mayer, U.; et al. Delivery of endocytosed proteins to the cell-division plane requires change of pathway from recycling to secretion. eLife 2014, 3, e02131. [Google Scholar] [CrossRef] [PubMed]
- Lakkaraju, A.; Rodriguez-Boulan, E. Itinerant exosomes: Emerging roles in cell and tissue polarity. Trends Cell Biol. 2008, 18, 199–209. [Google Scholar] [CrossRef] [PubMed]
- Yoon, Y.J.; Kim, O.Y.; Gho, Y.S. Extracellular vesicles as emerging intercellular communicasomes. BMB Rep. 2014, 47, 531–539. [Google Scholar] [CrossRef] [PubMed]
- Vlassov, A.V.; Magdaleno, S.; Setterquist, R.; Conrad, R. Exosomes: Current knowledge of their composition, biological functions, and diagnostic and therapeutic potentials. Biochim. Biophys. Acta 2012, 1820, 940–948. [Google Scholar] [CrossRef] [PubMed]
- Colombo, M.; Raposo, G.; Thery, C. Biogenesis, secretion, and intercellular interactions of exosomes and other extracellular vesicles. Annu. Rev. Cell Dev. Biol. 2014, 30, 255–289. [Google Scholar] [CrossRef] [PubMed]
- Colombo, M.; Moita, C.; van Niel, G.; Kowal, J.; Vigneron, J.; Benaroch, P.; Manel, N.; Moita, L.F.; Thery, C.; Raposo, G. Analysis of ESCRT functions in exosome biogenesis, composition and secretion highlights the heterogeneity of extracellular vesicles. J. Cell Sci. 2013, 126, 5553–5565. [Google Scholar] [CrossRef] [PubMed]
- Sanchatjate, S.; Schekman, R. Chs5/6 complex: A multiprotein complex that interacts with and conveys chitin synthase III from the trans-Golgi network to the cell surface. Mol. Biol. Cell 2006, 17, 4157–4166. [Google Scholar] [CrossRef] [PubMed]
- Trautwein, M.; Schindler, C.; Gauss, R.; Dengjel, J.; Hartmann, E.; Spang, A. Arf1p, Chs5p and the chaps are required for export of specialized cargo from the golgi. EMBO J. 2006, 25, 943–954. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.W.; Hamamoto, S.; Orci, L.; Schekman, R. Exomer: A coat complex for transport of select membrane proteins from the trans-Golgi network to the plasma membrane in yeast. J. Cell Biol. 2006, 174, 973–983. [Google Scholar] [CrossRef] [PubMed]
- Matsuoka, K.; Orci, L.; Amherdt, M.; Bednarek, S.Y.; Hamamoto, S.; Schekman, R.; Yeung, T. COPII-coated vesicle formation reconstituted with purified coat proteins and chemically defined liposomes. Cell 1998, 93, 263–275. [Google Scholar] [CrossRef] [PubMed]
- Spang, A.; Matsuoka, K.; Hamamoto, S.; Schekman, R.; Orci, L. Coatomer, Arf1p, and nucleotide are required to bud coat protein complex i-coated vesicles from large synthetic liposomes. Proc. Natl. Acad. Sci. USA 1998, 95, 11199–11204. [Google Scholar] [CrossRef] [PubMed]
- Paczkowski, J.E.; Richardson, B.C.; Strassner, A.M.; Fromme, J.C. The exomer cargo adaptor structure reveals a novel gtpase-binding domain. EMBO J. 2012, 31, 4191–4203. [Google Scholar] [CrossRef] [PubMed]
- Rockenbauch, U.; Ritz, A.M.; Sacristan, C.; Roncero, C.; Spang, A. The complex interactions of Chs5p, the chAPs, and the cargo Chs3p. Mol. Biol. Cell 2012, 23, 4402–4415. [Google Scholar] [CrossRef] [PubMed]
- Chuang, J.S.; Schekman, R.W. Differential trafficking and timed localization of two chitin synthase proteins, Chs2p and Chs3p. J. Cell Biol. 1996, 135, 597–610. [Google Scholar] [CrossRef] [PubMed]
- Zanolari, B.; Rockenbauch, U.; Trautwein, M.; Clay, L.; Barral, Y.; Spang, A. Transport to the plasma membrane is regulated differently early and late in the cell cycle in Saccharomyces cerevisiae. J. Cell Sci. 2011, 124, 1055–1066. [Google Scholar] [CrossRef] [PubMed]
- Ziman, M.; Chuang, J.S.; Tsung, M.; Hamamoto, S.; Schekman, R. Chs6p-dependent anterograde transport of Chs3p from the chitosome to the plasma membrane in Saccharomyces cerevisiae. Mol. Biol. Cell 1998, 9, 1565–1576. [Google Scholar] [CrossRef] [PubMed]
- Sacristan, C.; Manzano-Lopez, J.; Reyes, A.; Spang, A.; Muniz, M.; Roncero, C. Oligomerization of the chitin synthase Chs3 is monitored at the Golgi and affects its endocytic recycling. Mol. Microbiol. 2013, 90, 252–266. [Google Scholar] [PubMed]
- Paczkowski, J.E.; Fromme, J.C. Structural basis for membrane binding and remodeling by the exomer secretory vesicle cargo adaptor. Dev. Cell 2014, 30, 610–624. [Google Scholar] [CrossRef] [PubMed]
- Lam, K.K.; Davey, M.; Sun, B.; Roth, A.F.; Davis, N.G.; Conibear, E. Palmitoylation by the DHHC protein Pfa4 regulates the ER exit of Chs3. J. Cell Biol. 2006, 174, 19–25. [Google Scholar] [CrossRef] [PubMed]
- Reyes, A.; Sanz, M.; Duran, A.; Roncero, C. Chitin synthase III requires Chs4p-dependent translocation of Chs3p into the plasma membrane. J. Cell Sci. 2007, 120, 1998–2009. [Google Scholar] [CrossRef] [PubMed]
- Barfield, R.M.; Fromme, J.C.; Schekman, R. The exomer coat complex transports Fus1p to the plasma membrane via a novel plasma membrane sorting signal in yeast. Mol. Biol. Cell 2009, 20, 4985–4996. [Google Scholar] [CrossRef] [PubMed]
- Ritz, A.M.; Trautwein, M.; Grassinger, F.; Spang, A. The prion-like domain in the exomer-dependent cargo Pin2 serves as a trans-Golgi retention motif. Cell Rep. 2014, 7, 249–260. [Google Scholar] [CrossRef] [PubMed]
- Ren, J.; Wen, L.; Gao, X.; Jin, C.; Xue, Y.; Yao, X. CSS-Palm 2.0: An updated software for palmitoylation sites prediction. Protein Eng. Des. Sel. 2008, 21, 639–644. [Google Scholar] [CrossRef] [PubMed]
- Richardson, B.C.; Fromme, J.C. The exomer cargo adaptor features a flexible hinge domain. Structure 2013, 21, 486–492. [Google Scholar] [CrossRef] [PubMed]
- Hirst, J.; Schlacht, A.; Norcott, J.P.; Traynor, D.; Bloomfield, G.; Antrobus, R.; Kay, R.R.; Dacks, J.B.; Robinson, M.S. Characterization of TSET, an ancient and widespread membrane trafficking complex. eLife 2014, 3, e02866. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Spang, A. The Road not Taken: Less Traveled Roads from the TGN to the Plasma Membrane. Membranes 2015, 5, 84-98. https://doi.org/10.3390/membranes5010084
Spang A. The Road not Taken: Less Traveled Roads from the TGN to the Plasma Membrane. Membranes. 2015; 5(1):84-98. https://doi.org/10.3390/membranes5010084
Chicago/Turabian StyleSpang, Anne. 2015. "The Road not Taken: Less Traveled Roads from the TGN to the Plasma Membrane" Membranes 5, no. 1: 84-98. https://doi.org/10.3390/membranes5010084
APA StyleSpang, A. (2015). The Road not Taken: Less Traveled Roads from the TGN to the Plasma Membrane. Membranes, 5(1), 84-98. https://doi.org/10.3390/membranes5010084




