Effects of Operational Parameters on Mg2+/Li+ Separation Performance in Electrodialysis System
Abstract
1. Introduction
2. Materials and Methods
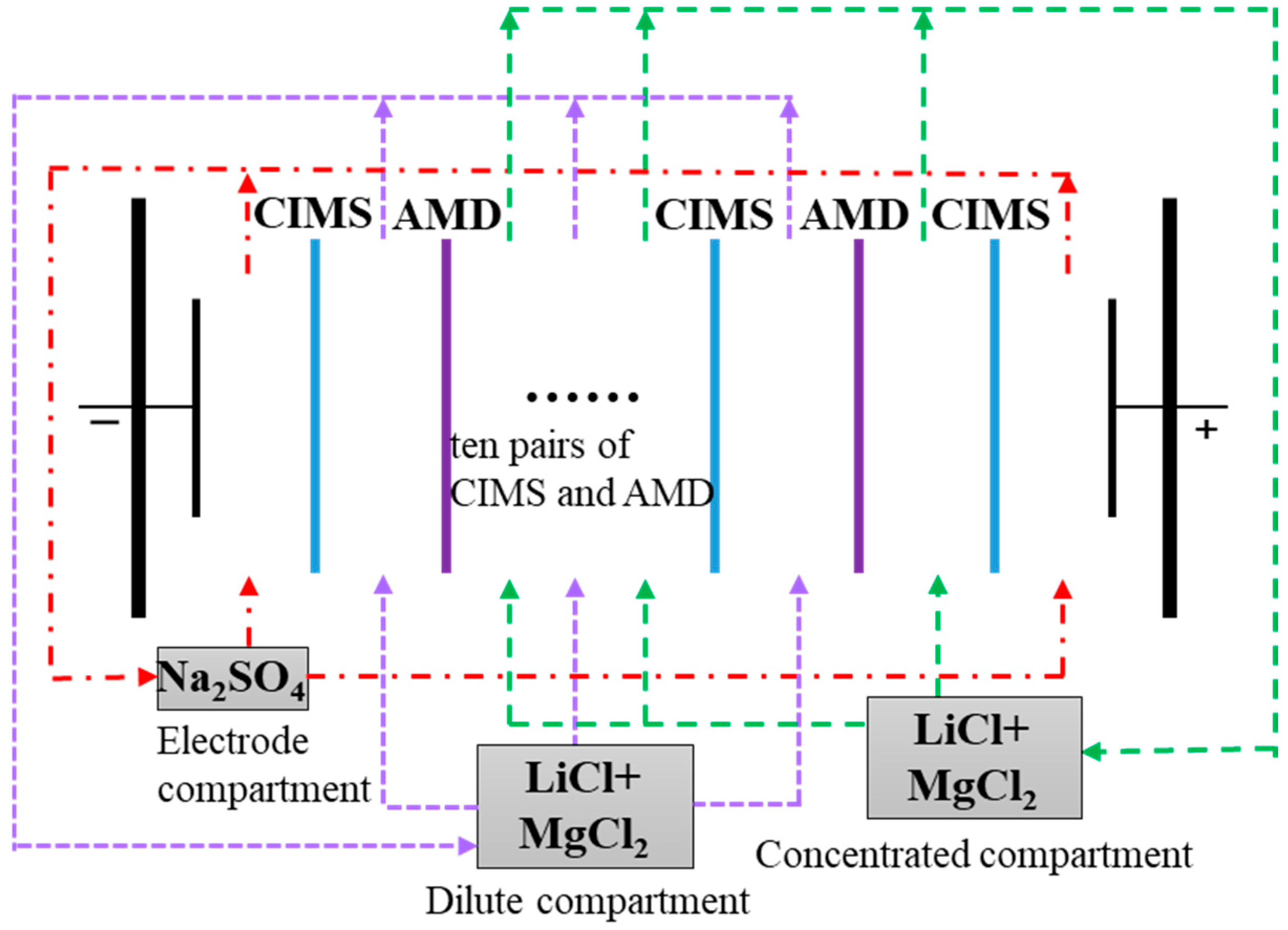
2.1. Experimental Setup and Materials
2.2. Electrodialysis Experiment
2.3. Characterization of Separation Performance
2.3.1. Separation Coefficient
2.3.2. Lithium Recovery Ratio
2.3.3. Permselectivity Coefficient
2.3.4. Ionic Flux
3. Results and Discussion
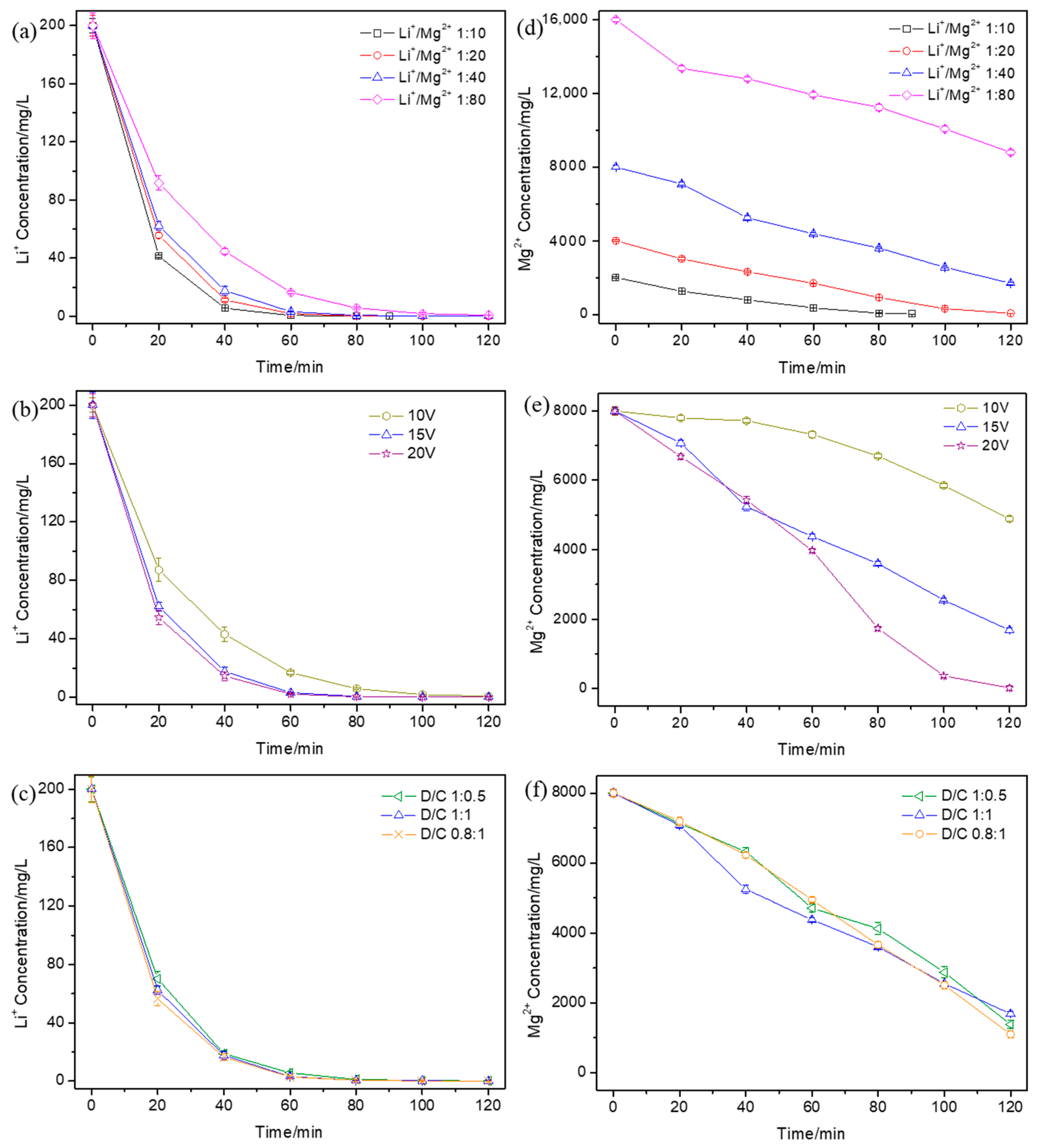
3.1. Effect of Operational Parameters on the Ionic Concentration Variation
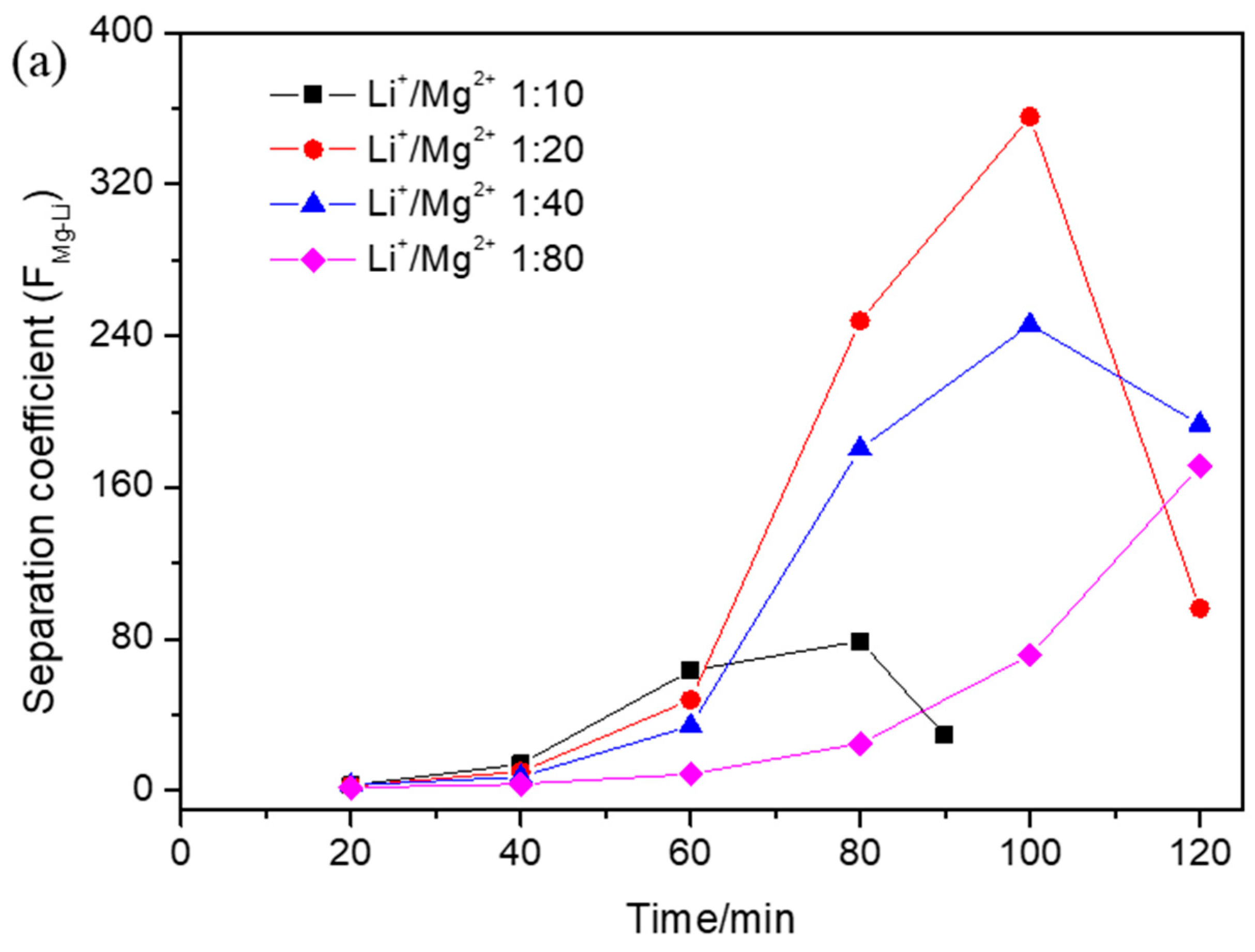
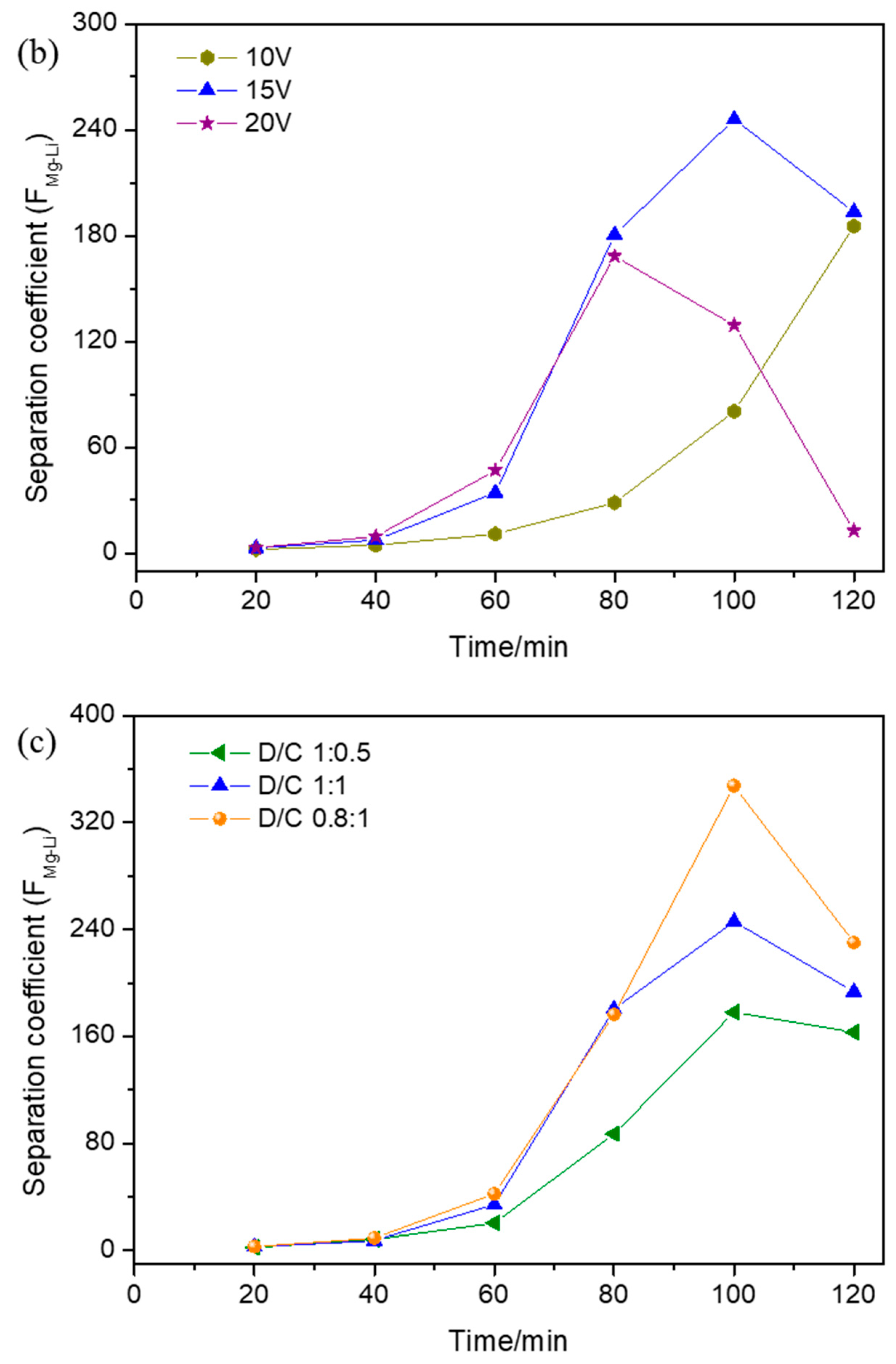
3.2. Effect of Operational Parameters on Separation Coefficient
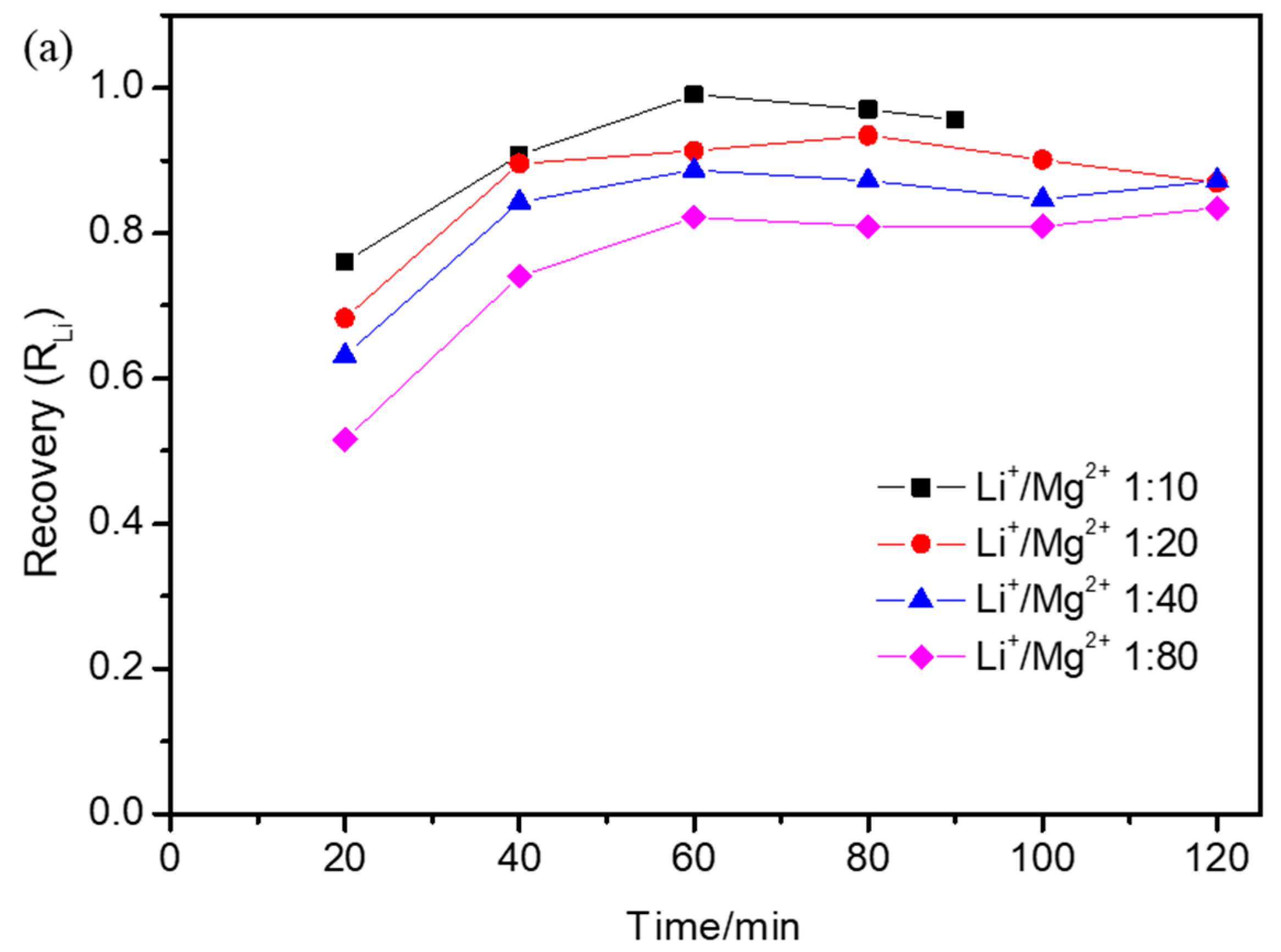
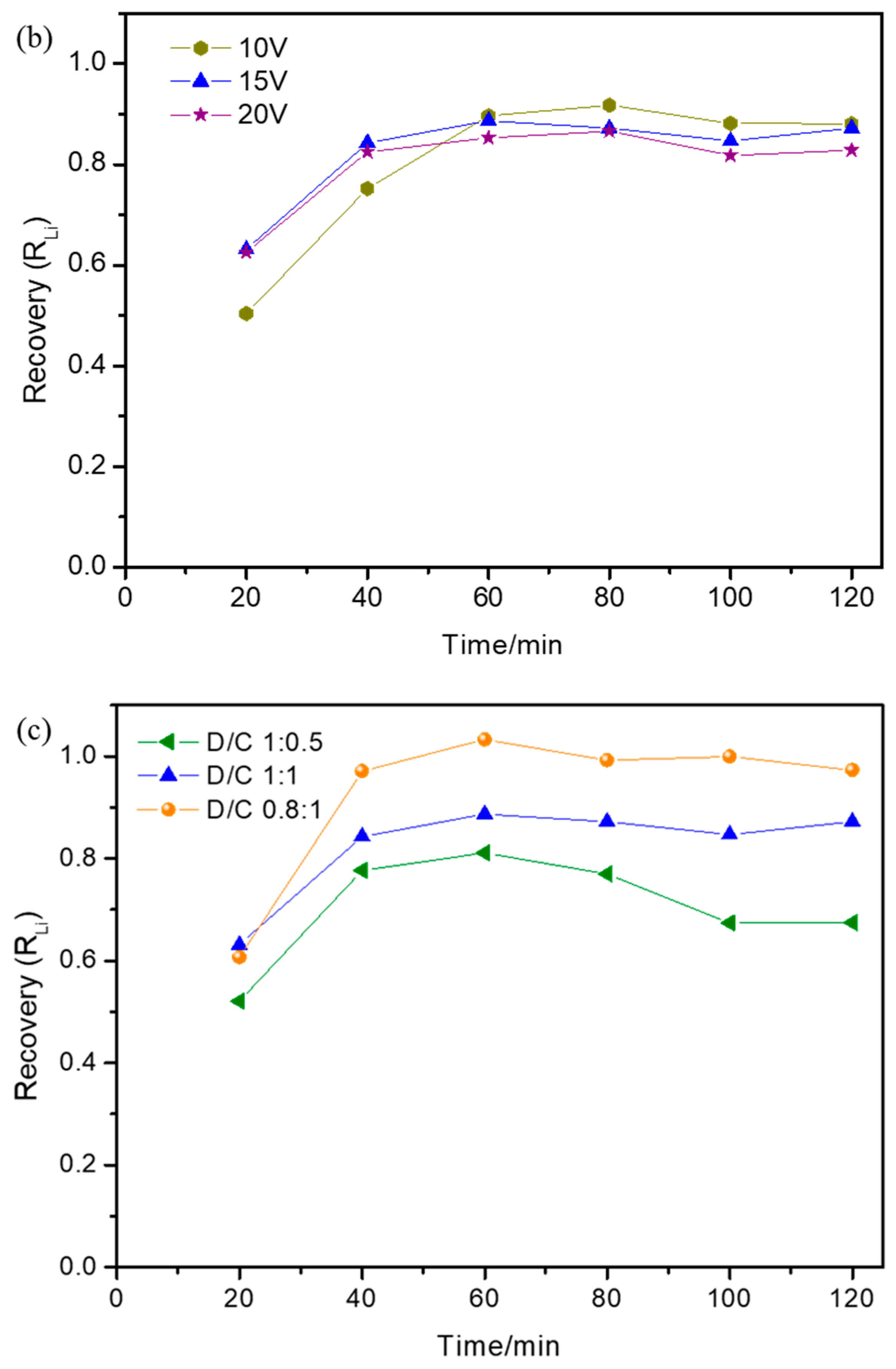
3.3. Effect of Operational Parameters on Lithium Recovery Ratio
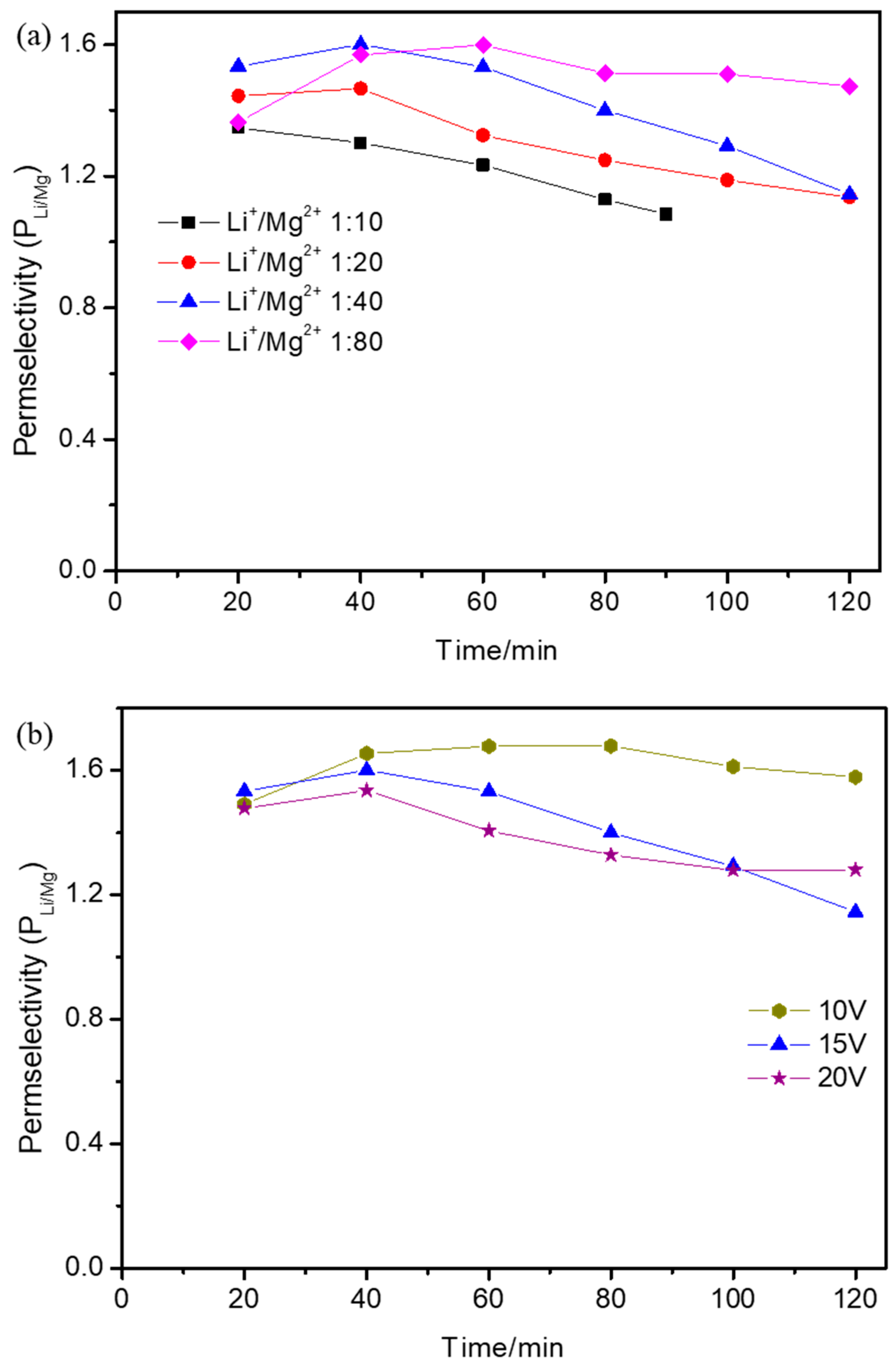
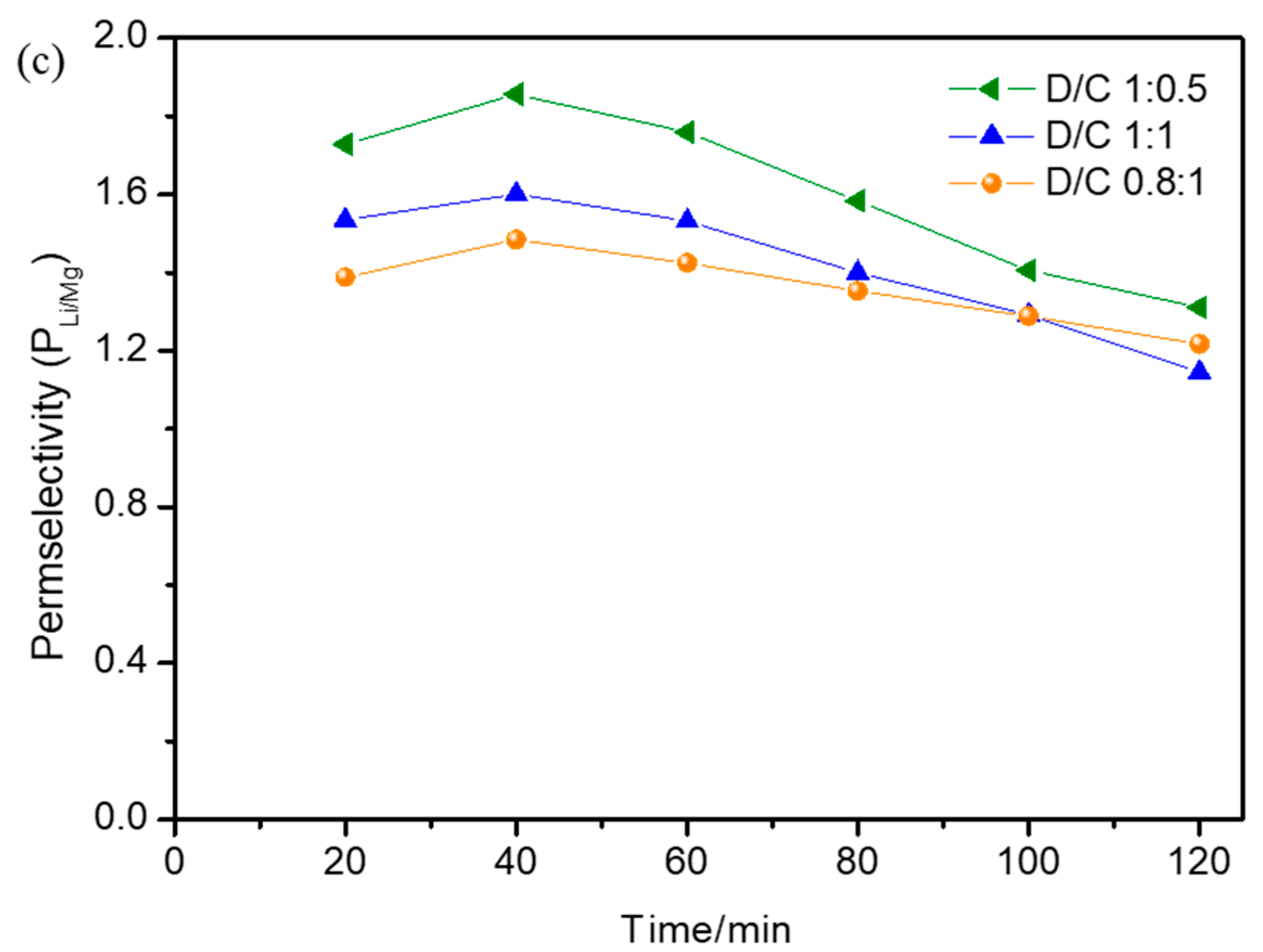
3.4. Effect of Operational Parameters on Permselectivity Coefficient
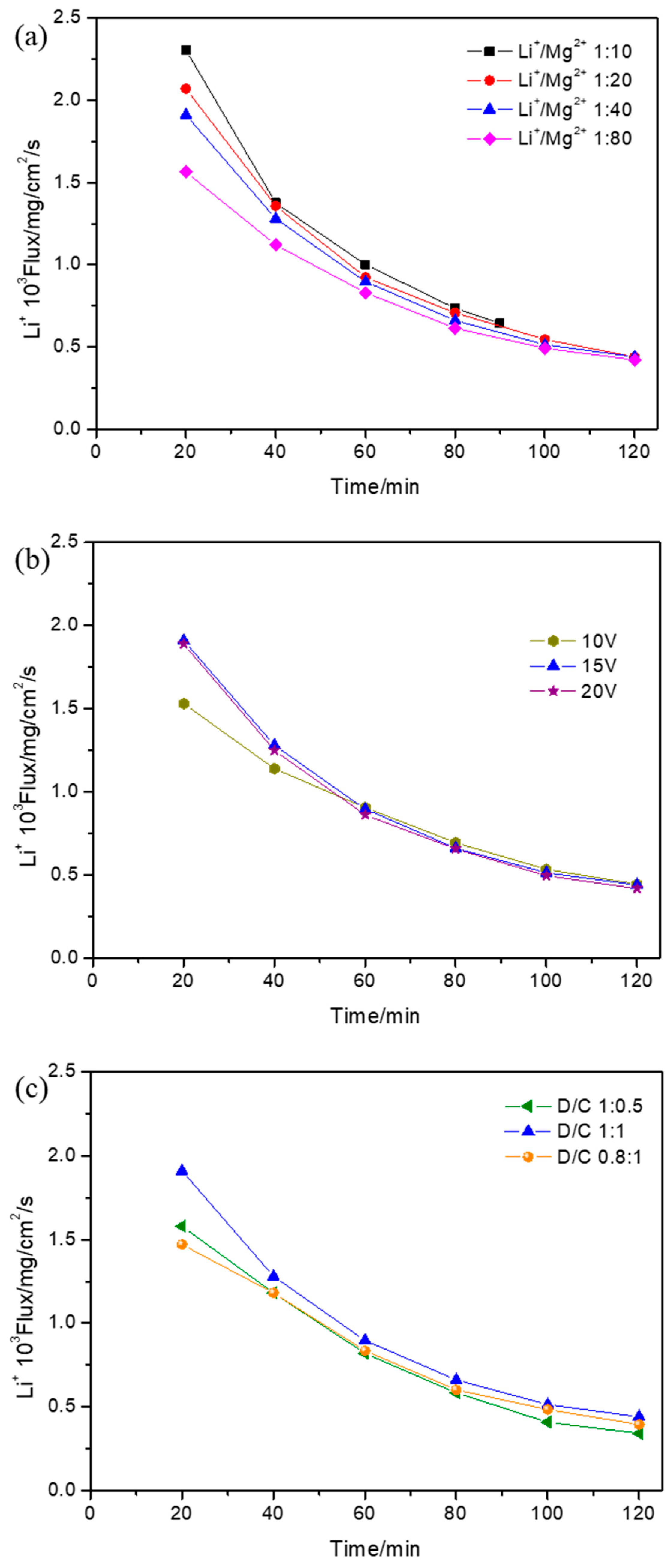
3.5. Effect of Operational Parameters on Li+ Flux
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Speirs, J.; Contestabile, M.; Houari, Y.; Gross, R. The future of lithium availability for electric vehicle batteries. Renew. Sust. Energ. Rev. 2014, 35, 183–193. [Google Scholar] [CrossRef]
- Diouf, B.; Pode, R. Potential of lithium-ion batteries in renewable energy. Renew. Energ. 2015, 76, 375–380. [Google Scholar] [CrossRef]
- Kesler, S.E.; Gruber, P.W.; Medina, P.A.; Keoleian, G.A.; Everson, M.P.; Wallington, T.J. Global lithium resources: Relative importance of pegmatite, brine and other deposits. Ore. Geol. Rev. 2012, 48, 55–69. [Google Scholar] [CrossRef]
- Vikström, H.; Davidsson, S.; Höök, M. Lithium availability and future production outlooks. Appl. Energ. 2013, 110, 252–266. [Google Scholar] [CrossRef]
- Swain, B. Recovery and recycling of lithium: A review. Sep. Purif. Technol. 2017, 172, 388–403. [Google Scholar] [CrossRef]
- Flexer, V.; Baspineiro, C.F.; Galli, C.I. Lithium recovery from brines: A vital raw material for green energies with a potential environmental impact in its mining and processing. Sci. Total. Environ. 2018, 639, 1188–1204. [Google Scholar] [CrossRef]
- Demol, J.; Ho, E.; Soldenhoff, K.; Senanayake, G. The sulfuric acid bake and leach route for processing of rare earth ores and concentrates: A review. Hydrometallurgy 2019, 188, 123–139. [Google Scholar] [CrossRef]
- Xu, S.; Song, J.; Bi, Q.; Chen, Q.; Zhang, W.; Qian, Z.; Zhang, L.; Xu, S.; Tang, N.; He, T. Extraction of lithium from Chinese salt-lake brines by membranes: Design and practice. J. Membr. Sci. 2021, 635, 119441. [Google Scholar] [CrossRef]
- An, J.W.; Kang, D.J.; Tran, K.T.; Kim, M.J.; Lim, T.; Tran, T. Recovery of lithium from Uyuni salar brine. Hydrometallurgy 2012, 117–118, 64–70. [Google Scholar] [CrossRef]
- Meshram, P.; Pandey, B.D.; Mankhand, T.R. Extraction of lithium from primary and secondary sources by pre-treatment, leaching and separation: A comprehensive review. Hydrometallurgy 2014, 150, 192–208. [Google Scholar] [CrossRef]
- Gherasim, C.; Křivčík, J.; Mikulášek, P. Investigation of batch electrodialysis process for removal of lead ions from aqueous solutions. Chem. Eng. J. 2014, 256, 324–334. [Google Scholar] [CrossRef]
- Marder, L.; Bittencourt, S.D.; Zoppas Ferreira, J.; Bernardes, A.M. Treatment of molybdate solutions by electrodialysis: The effect of pH and current density on ions transport behavior. Sep. Purif. Technol. 2016, 167, 32–36. [Google Scholar] [CrossRef]
- Masigol, M.A.; Moheb, A.; Mehrabani-Zeinabad, A. An experimental investigation into batch electrodialysis process for removal of sodium sulfate from magnesium stearate aqueous slurry. Desalination 2012, 300, 12–18. [Google Scholar] [CrossRef]
- Parsa, N.; Moheb, A.; MehrabaniZeinabad, A.; Masigol, M.A. Recovery of lithium ions from sodium-contaminated lithium bromide solution by using electrodialysis process. Chem. Eng. Res. Des. 2015, 98, 81–88. [Google Scholar] [CrossRef]
- Strathmann, H. Electrodialysis, a mature technology with a multitude of new applications. Desalination 2010, 264, 268–288. [Google Scholar] [CrossRef]
- Nie, X.; Sun, S.; Sun, Z.; Song, X.; Yu, J. Ion-fractionation of lithium ions from magnesium ions by electrodialysis using monovalent selective ion-exchange membranes. Desalination 2017, 403, 128–135. [Google Scholar] [CrossRef]
- Tanaka, Y. A computer simulation of continuous ion exchange membrane electrodialysis for desalination of saline water. Desalination 2009, 249, 809–821. [Google Scholar] [CrossRef]
- Hoshino, T. Preliminary studies of lithium recovery technology from seawater by electrodialysis using ionic liquid membrane. Desalination 2013, 317, 11–16. [Google Scholar] [CrossRef]
- Nie, X.-Y.; Sun, S.-Y.; Song, X.; Yu, J.-G. Further investigation into lithium recovery from salt lake brines with different feed characteristics by electrodialysis. J. Membr. Sci. 2017, 530, 185–191. [Google Scholar] [CrossRef]
- Luo, T.; Abdu, S.; Wessling, M. Selectivity of ion exchange membranes: A review. J. Membr. Sci. 2018, 555, 429–454. [Google Scholar] [CrossRef]
- Guo, Z.-Y.; Ji, Z.-Y.; Chen, Q.-B.; Liu, J.; Zhao, Y.-Y.; Li, F.; Liu, Z.-Y.; Yuan, J.-S. Prefractionation of LiCl from concentrated seawater/salt lake brines by electrodialysis with monovalent selective ion exchange membranes. J. Cleaner Prod. 2018, 193, 338–350. [Google Scholar] [CrossRef]
- Zhang, X.-C.; Wang, J.; Ji, Z.-Y.; Ji, P.-Y.; Liu, J.; Zhao, Y.-Y.; Li, F.; Yuan, J.-S. Preparation of Li2CO3 from high Mg2+/Li+ brines based on selective-electrodialysis with feed and bleed mode. J. Environ. Chem. Eng. 2021, 9, 106635. [Google Scholar] [CrossRef]
- Zhang, Y.; Wang, L.; Sun, W.; Hu, Y.; Tang, H. Membrane technologies for Li+/Mg2+ separation from salt-lake brines and seawater: A comprehensive review. J. Ind. Eng. Chem. 2020, 81, 7–23. [Google Scholar] [CrossRef]
- Hosseini, S.M.; Gholami, A.; Madaeni, S.S.; Moghadassi, A.R.; Hamidi, A.R. Fabrication of (polyvinyl chloride/cellulose acetate) electrodialysis heterogeneous cation exchange membrane: Characterization and performance in desalination process. Desalination 2012, 306, 51–59. [Google Scholar] [CrossRef]
- Kariduraganavar, M.Y.; Nagarale, R.K.; Kittur, A.A.; Kulkarni, S.S. Ion-exchange membranes: Preparative methods for electrodialysis and fuel cell applications. Desalination 2006, 197, 225–246. [Google Scholar] [CrossRef]
- Sata, T. Studies on ion exchange membranes with permselectivity for specific ions in electrodialysis. J. Membr. Sci. 1994, 93, 117–135. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhao, Z.; Yang, J.; Kong, D.; Peng, Y.; Jin, D. Effects of Operational Parameters on Mg2+/Li+ Separation Performance in Electrodialysis System. Membranes 2025, 15, 260. https://doi.org/10.3390/membranes15090260
Zhao Z, Yang J, Kong D, Peng Y, Jin D. Effects of Operational Parameters on Mg2+/Li+ Separation Performance in Electrodialysis System. Membranes. 2025; 15(9):260. https://doi.org/10.3390/membranes15090260
Chicago/Turabian StyleZhao, Zhijuan, Jianhua Yang, Dexin Kong, Yunyan Peng, and Dong Jin. 2025. "Effects of Operational Parameters on Mg2+/Li+ Separation Performance in Electrodialysis System" Membranes 15, no. 9: 260. https://doi.org/10.3390/membranes15090260
APA StyleZhao, Z., Yang, J., Kong, D., Peng, Y., & Jin, D. (2025). Effects of Operational Parameters on Mg2+/Li+ Separation Performance in Electrodialysis System. Membranes, 15(9), 260. https://doi.org/10.3390/membranes15090260




