Effects of Ca Substitution in Single-Phase Sr1-xCaxTi0.8Fe0.2O3-ẟ Oxygen Transport Membranes and in Dual-Phase Sr1-xCaxTi0.8Fe0.2O3-ẟ-Ce0.8Gd0.2O2 Membranes
Abstract
1. Introduction
2. Materials and Methods
2.1. Sample Preparation
2.2. Characterization
3. Results and Discussion
3.1. Single-Phase Materials
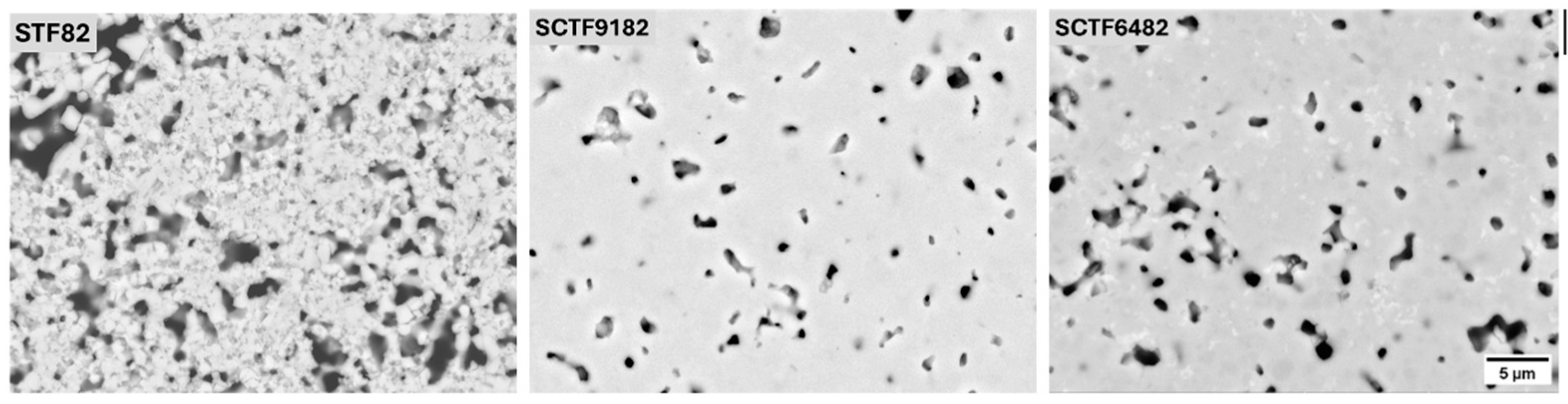
3.1.1. Microstructure
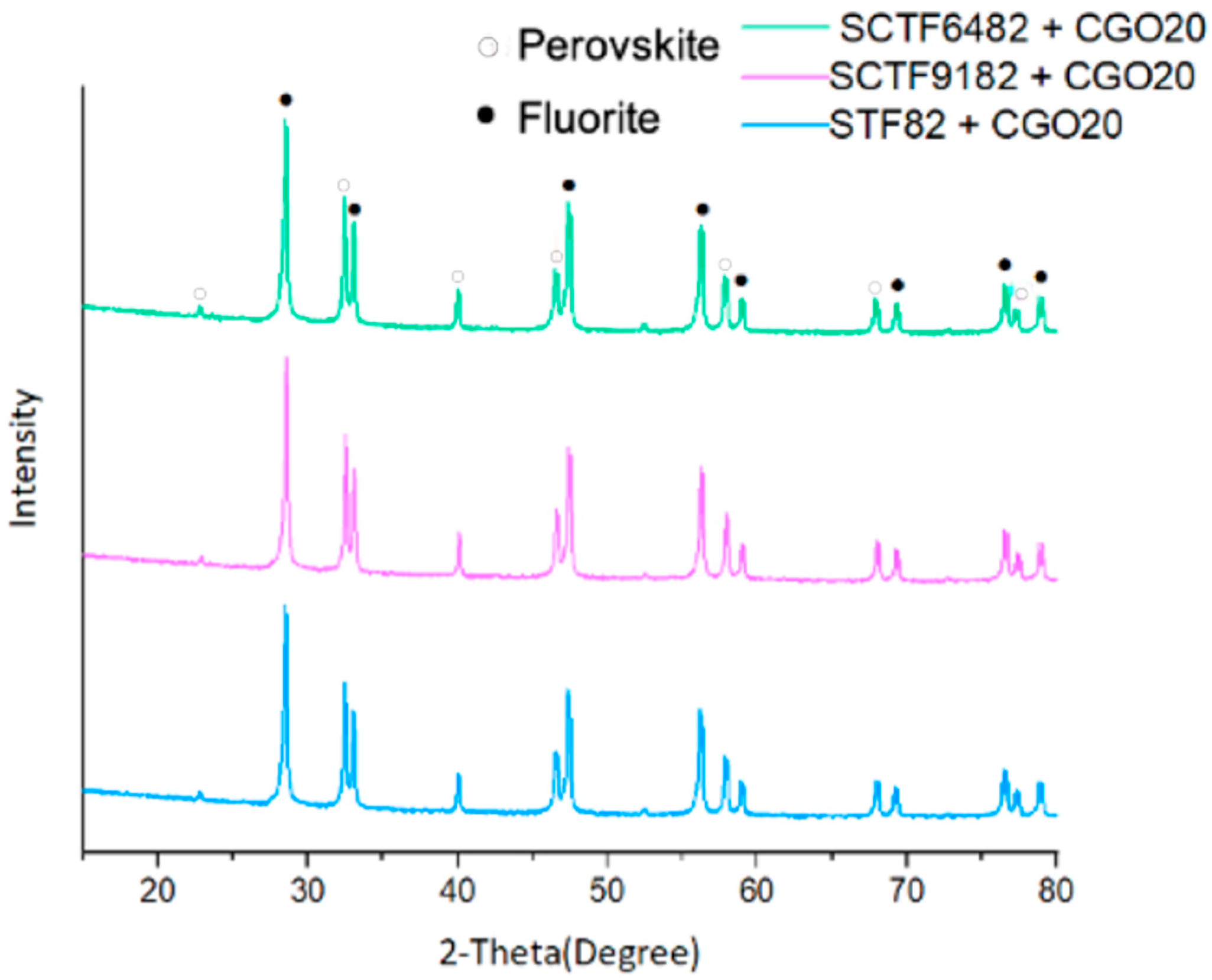
3.1.2. Crystal Structure
3.1.3. Oxygen Permeation Performance
3.2. Dual-Phase Materials
3.2.1. Microstructure
3.2.2. Oxygen Permeation Performance
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Bai, W.; Feng, J.; Luo, C.; Zhang, P.; Wang, H.; Yang, Y.; Zhao, Y.; Fan, H. A Comprehensive Review on Oxygen Transport Membranes: Development History, Current Status, and Future Directions. Int. J. Hydrog. Energy 2021, 46, 36257–36290. [Google Scholar] [CrossRef]
- Zhang, K.; Sunarso, J.; Shao, Z.; Zhou, W.; Sun, C.; Wanga, S.; Liu, S. Research progress and materials selection guidelines on mixed conducting perovskite-type ceramic membranes for oxygen production. RSC Adv. 2011, 1, 1661–1676. [Google Scholar] [CrossRef]
- Teraoka, Y.; Zhang, H.M.; Furukawa, S.; Yamazoe, N. Oxygen Permeation Through Perovskite-Type Oxide. Chem. Lett. 1985, 11, 1743–1746. [Google Scholar] [CrossRef]
- Gupta, S.; Mahapatra, M.K.; Singh, P. Lanthanum Chromite Based Perovskites for Oxygen Transport Membrane. Mater. Sci. Eng. R Rep. 2015, 90, 1–36. [Google Scholar] [CrossRef]
- Schulze-Küppers, F.; Drago, F.; Ferravante, L.; Herzog, S.; Baumann, S.; Pinacci, P.; Meulenberg, W.A. Design and Fabrication of Large-Sized Planar Oxygen Transport Membrane Components for Direct Integration in Oxy-Combustion Processes. Sep. Purif. Technol. 2019, 220, 89–101. [Google Scholar] [CrossRef]
- Betz, M.; Schulze-Küppers, F.; Baumann, S.; Meulenberg, W.A.; Stöver, D. Supported Oxygen Transport Membranes for Oxyfuel Power Plants. Adv. Sci. Technol. 2010, 72, 93–98. [Google Scholar] [CrossRef]
- Drago, F.; Fedeli, P.; Cavaliere, A.; Cammi, A.; Passoni, S.; Mereu, R.; De La Pierre, S.; Smeacetto, F.; Ferraris, M. Development of a Membrane Module Prototype for Oxygen Separation in Industrial Applications. Membranes 2022, 12, 167. [Google Scholar] [CrossRef] [PubMed]
- Julbe, A.; Farrusseng, D.; Guizard, C. Limitations and Potentials of Oxygen Transport Dense and Porous Ceramic Membranes for Oxidation Reactions. Catal. Today 2005, 104, 102–113. [Google Scholar] [CrossRef]
- Arratibel Plazaola, A.; Cruellas Labella, A.; Liu, Y.; Badiola Porras, N.; Pacheco Tanaka, D.; Sint Annaland, M.; Gallucci, F. Mixed Ionic-Electronic Conducting Membranes (MIEC) for Their Application in Membrane Reactors: A Review. Processes 2019, 7, 128. [Google Scholar] [CrossRef]
- Deibert, W.; Ivanova, M.E.; Baumann, S.; Guillon, O.; Meulenberg, W.A. Ion-Conducting Ceramic Membrane Reactors for High-Temperature Applications. J. Membr. Sci. 2017, 543, 79–97. [Google Scholar] [CrossRef]
- Li, W.; Zhu, X.; Cao, Z.; Wang, W.; Yang, W. Mixed Ionic-Electronic Conducting (MIEC) Membranes for Hydrogen Production from Water Splitting. Int. J. Hydrog. Energy 2015, 40, 3452–3461. [Google Scholar] [CrossRef]
- Zhu, X.; Shi, C.; Li, K.; Zhai, K.; Wang, H.; Wei, Y.; Tian, D.; Zeng, C. Water Splitting for Hydrogen Generation over Lanthanum-Calcium-Iron Perovskite-Type Membrane Driven by Reducing Atmosphere. Int. J. Hydrog. Energy 2017, 42, 19776–19787. [Google Scholar] [CrossRef]
- Wu, X.-Y.; Chang, L.; Uddi, M.; Kirchen, P.; Ghoniem, A.F. Toward Enhanced Hydrogen Generation from Water Using Oxygen Permeating LCF Membranes. Phys. Chem. Chem. Phys. 2015, 17, 10093–10107. [Google Scholar] [CrossRef]
- Balachandran, U.B.; Lee, T.H.; Dorris, S.E. Hydrogen Production by Water Dissociation Using Mixed Conducting Dense Ceramic Membranes. Int. J. Hydrog. Energy 2007, 32, 451–456. [Google Scholar] [CrossRef]
- Balachandran, U.; Lee, T.H.; Wang, S.; Dorris, S.E. Use of mixed conducting membranes to produce hydrogen by water dissociation. Int. J. Hydrog. Energy 2004, 29, 291–296. [Google Scholar] [CrossRef]
- Fang, W.; Steinbach, F.; Cao, Z.; Zhu, X.; Feldhoff, A. A Highly Efficient Sandwich-Like Symmetrical Dual-Phase Oxygen Transporting Membrane Reactor for Hydrogen Production by Water Splitting. Angew. Chem. 2016, 128, 8648–8651. [Google Scholar] [CrossRef]
- He, G.; Ling, Y.; Jiang, H.; Toghan, A. Barium Titanate as a Highly Stable Oxygen Permeable Membrane Reactor for Hydrogen Production from Thermal Water Splitting. ACS Sustain. Chem. Eng. 2021, 9, 11147–11154. [Google Scholar] [CrossRef]
- Prosser, N.M.; Shah, M.M. Current and Future Oxygen (O2) Supply Technologies for Oxy-Fuel Combustion. In Oxy-Fuel Combustion for Power Generation and Carbon Dioxide (CO2) Capture; Elsevier: Amsterdam, The Netherlands, 2011; pp. 195–227. [Google Scholar] [CrossRef]
- Zhang, S.; Li, T.; Wang, B.; Zhou, Z.; Meng, X.; Yang, N.; Zhu, X.; Liu, S. Coupling water splitting and partial oxidation of methane (POM) in Ag modified La0.8Ca0.2Fe0.94O3-δ hollow fiber membrane reactors for co-production of H2 and syngas. J. Membr. Sci. 2022, 659, 120772. [Google Scholar] [CrossRef]
- Son, S.J.; Lee, H.J.; Kim, S.K.; Lee, J.-H.; Park, H.J.; Joo, J.H. Exceptional performance of water splitting coupled with methane partial oxidation by oxygen-permeable membrane reactor. Chem. Eng. J. 2023, 466, 143031. [Google Scholar] [CrossRef]
- Park, J.H.; Kwon, Y.; Nam, G.D.; Joo, J.H. Simultaneous Conversion of Carbon Dioxide and Methane to Syngas Using an Oxygen Transport Membrane in Pure CO2 and CH4 Atmospheres. J. Mater. Chem. A 2018, 6, 14246–14254. [Google Scholar] [CrossRef]
- Chen, G.; Feldhoff, A.; Weidenkaff, A.; Li, C.; Liu, S.; Zhu, X.; Sunarso, J.; Huang, K.; Wu, X.; Ghoniem, A.F.; et al. Roadmap for Sustainable Mixed Ionic-Electronic Conducting Membranes. Adv. Funct. Mater. 2022, 32, 2105702. [Google Scholar] [CrossRef]
- Wu, X.-Y.; Ghoniem, A.F. Mixed Ionic-Electronic Conducting (MIEC) Membranes for Thermochemical Reduction of CO2: A Review. Prog. Energy Combust. Sci. 2019, 74, 1–30. [Google Scholar] [CrossRef]
- Lemes-Rachadel, P.; Garcia, G.S.; Machado, R.A.F.; Hotza, D.; Diniz da Costa, J.C. Current Developments of Mixed Conducting Membranes on Porous Substrates. Mater. Res. 2014, 17, 242–249. [Google Scholar] [CrossRef]
- Kharton, V.V.; Yaremchenko, A.A.; Kovalevsky, A.V.; Viskup, A.P.; Naumovich, E.N.; Kerko, P.F. Perovskite-type oxides for high-temperature oxygen separation membranes. J. Membr. Sci. 1999, 163, 307–317. [Google Scholar] [CrossRef]
- Xu, S.J.; Thompson, W.J. Oxygen permeation rates through ion-conducting perovskite membranes. Chem. Eng. Sci. 1999, 54, 17, 3839–3850. [Google Scholar] [CrossRef]
- Zhu, X.; Li, M.; Liu, H.; Zhang, T.; Cong, Y.; Yang, W. Design and Experimental Investigation of Oxide Ceramic Dual-Phase Membranes. J. Membr. Sci. 2012, 394–395, 120–130. [Google Scholar] [CrossRef]
- Zhu, X.; Li, Q.; He, Y.; Cong, Y.; Yang, W. Oxygen Permeation and Partial Oxidation of Methane in Dual-Phase Membrane Reactors. J. Membr. Sci. 2010, 360, 454–460. [Google Scholar] [CrossRef]
- Aliyatulmuna, A.; Utomo, W.P.; Burhan, R.Y.P.; Fansuri, H.; Murwani, I.K. Thermal Expansion, Microhardness and Oxygen Permeation of La1-xSrxCo0.8Fe0.2O3+δ Membranes. Asian J. Chem. 2017, 29, 2191–2196. [Google Scholar] [CrossRef]
- Baumann, S.; Schulze-Küppers, F.; Roitsch, S.; Betz, M.; Zwick, M.; Pfaff, E.M.; Meulenberg, W.A.; Mayer, J.; Stöver, D. Influence of Sintering Conditions on Microstructure and Oxygen Permeation of Ba0.5Sr0.5Co0.8Fe0.2O3−δ (BSCF) Oxygen Transport Membranes. J. Membr. Sci. 2010, 359, 102–109. [Google Scholar] [CrossRef]
- Serra, J.M.; Garcia-Fayos, J.; Baumann, S.; Schulze-Küppers, F.; Meulenberg, W.A. Oxygen Permeation through Tape-Cast Asymmetric Al-La0.6Sr0.4Co0.2Fe0.8O3−δ Membranes. J. Membr. Sci. 2013, 447, 297–305. [Google Scholar] [CrossRef]
- Schulze-Küppers, F.; Baumann, S.; Meulenberg, W.A.; Stöver, D.; Buchkremer, H.-P. Manufacturing and Performance of Advanced Supported Ba0.5Sr0.5Co0.8Fe0.2O3−δ (BSCF) Oxygen Transport Membranes. J. Membr. Sci. 2013, 433, 121–125. [Google Scholar] [CrossRef]
- Bucher, E.; Egger, A.; Caraman, G.B.; Sitte, W. Stability of the SOFC Cathode Material (Ba,Sr)(Co,Fe)O[Sub 3−δ] in CO2-Containing Atmospheres. J. Electrochem. Soc. 2008, 155, B1218. [Google Scholar] [CrossRef]
- Lein, H.; Wiik, K.; Grande, T. Kinetic Demixing and Decomposition of Oxygen Permeable Membranes. Solid State Ion. 2006, 177, 1587–1590. [Google Scholar] [CrossRef]
- Ovenstone, J.; Jung, J.-I.; White, J.S.; Edwards, D.D.; Misture, S.T. Phase Stability of BSCF in Low Oxygen Partial Pressures. J. Solid State Chem. 2008, 181, 576–586. [Google Scholar] [CrossRef]
- Schulze-Küppers, F.; ten Donkelaar, S.F.P.; Baumann, S.; Prigorodov, P.; Sohn, Y.J.; Bouwmeester, H.J.M.; Meulenberg, W.A.; Guillon, O. Structural and Functional Properties of SrTi1-xFexO3−δ (0 ≤ x ≤ 1) for the Use as Oxygen Transport Membrane. Sep. Purif. Technol. 2015, 147, 414–421. [Google Scholar] [CrossRef]
- Liu, Y.; Baumann, S.; Schulze-Küppers, F.; Mueller, D.N.; Guillon, O. Co and Fe Co-Doping Influence on Functional Properties of SrTiO3 for Use as Oxygen Transport Membranes. J. Eur. Ceram. Soc. 2018, 38, 5058–5066. [Google Scholar] [CrossRef]
- Tang, Y.; Baumann, S.; Müller, M.; Sebold, D.; Nijmeijer, A.; Guillon, O.; Meulenberg, W.A. Ni-Doping Influence on Functional Properties of SrTi0.65Fe0.35O3-δ for Use as Oxygen Transport Membranes. J. Eur. Ceram. Soc. 2024, 44, 116742. [Google Scholar] [CrossRef]
- Zhang, Z.; Chen, D.; Dong, F.; Xu, X.; Hao, Y.; Shao, Z. Understanding the doping effect toward the design of CO2-tolerant perovskite membranes with enhanced oxygen permeability. J. Membr. Sci. 2016, 519, 11–21. [Google Scholar] [CrossRef]
- Xi, X.; Liu, X.; Huang, L.; Liu, J.; Zhang, B.-W.; Rothenberg, G.; Fu, X.-Z.; Luo, J.-L. Unraveling the relationship between Sr stoichiometry in SrxFe1. 5Mo0.5O6−σ and its catalytic performance for high-temperature CO2 electrolysis, Mater. Rep. Energy 2023, 3, 100179. [Google Scholar] [CrossRef]
- Fedeli, P.; Nigroni, V.; Malgrati, E.; Cavaliere, A.; Cammi, A.; Drago, F. Ca Substitution in Ca 1-xSrxTi0.8Fe0.2O3-δ Perovskites for Oxygen Transport Membranes: A Promising Strategy to Improve CO2 Tolerance. J. Am. Ceram. Soc. 2024, 107, 5668–5681. [Google Scholar] [CrossRef]
- Ghyngazov, S.A.; Shevelev, S.A. Effect of Additives on Sintering of Zirconia Ceramics. J. Therm. Anal. Calorim. 2018, 134, 45–49. [Google Scholar] [CrossRef]
- Hayashi, H.; Inaba, H.; Matsuyama, M.; Lan, N.G.; Dokiya, M.; Tagawa, H. Structural consideration on the ionic conductivity of perovskite-type oxides. Solid State Ion. 1999, 122, 1–15. [Google Scholar] [CrossRef]
- Kharton, V.V.; Kovalevsky, A.V.; Viskup, A.P.; Jurado, J.R.; Figueiredo, F.M.; Naumovich, E.N.; Frade, J.R. Transport Properties and Thermal Expansion of Sr0. 97Ti1-xFexO3−δ (x = 0.2 − 0.8), J. Solid State Chem. 2001, 156, 437–444. [Google Scholar] [CrossRef]
- Mogensen, M.; Lybye, D.; Bonanos, N.; Hendriksen, P.V.; Poulsen, F.W. Factors controlling the oxide ion conductivity of fluorite and perovskite structured oxides. Solid State Ion. 2004, 174, 279–286. [Google Scholar] [CrossRef]
- Brisotto, M.; Cernuschi, F.; Drago, F.; Lenardi, C.; Rosa, P.; Meneghini, C.; Merlini, M.; Rinaldi, C. High temperature stability of Ba0.5Sr0.5Co0.8Fe0.2O3−δ and La0.6Sr0.4Co1-yFeyO3−δ oxygen separation perovskite membranes. J. Eur. Ceram. Soc. 2016, 36, 1679–1690. [Google Scholar] [CrossRef]
- Shannon, R.D. Revised effective ionic radii and systematic studies of interatomic distances in halides and chalcogenides. Acta Crystallogr. 1976, A32, 751–767. [Google Scholar] [CrossRef]
- Joo, J.H.; Yun, K.S.; Lee, Y.; Jung, J.; Yoo, C.-Y.; Yu, J.H. Dramatically Enhanced Oxygen Fluxes in Fluorite-Rich Dual-Phase Membrane by Surface Modification. Chem. Mater. 2014, 26, 4387–4394. [Google Scholar] [CrossRef]
- Fischer, L.; Neuhaus, K.; Schmidt, C.; Ran, K.; Behr, P.; Baumann, S.; Mayer, J.; Meulenberg, W.A. Phase Formation and Performance of Solid State Reactive Sintered Ce0.8Gd0.2O2−δ–FeCo2O4 Composites. J. Mater. Chem. A 2022, 10, 2412–2420. [Google Scholar] [CrossRef]







| Membranes | Temperature | Sintering Time |
|---|---|---|
| STF82 | 1450 °C | 5 h |
| SCTF9182 | 1400 °C | 5 h |
| SCTF6482 | 1350 °C | 5 h |
| STF82 + CGO | 1400 °C | 5 h |
| SCTF9182 + CGO | 1400 °C | 5 h |
| SCTF6482 + CGO | 1400 °C | 5 h |
| Membranes | Activation Energy kJ/mol |
|---|---|
| STF82 | 78 |
| SCTF9182 | 87 |
| SCTF6482 | 74 |
| Membranes | Activation Energy kJ/mol |
|---|---|
| STF82 + CGO | 81 |
| SCTF9182 + CGO | 78 |
| SCTF6482 + CGO | 95 |
| STF82 + CGO * | 61 |
| SCTF9182 + CGO * | 57 |
| SCTF6482 + CGO * | 61 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nigroni, V.; Tang, Y.; Baumann, S.; Sebold, D.; Malgrati, E.; Fedeli, P. Effects of Ca Substitution in Single-Phase Sr1-xCaxTi0.8Fe0.2O3-ẟ Oxygen Transport Membranes and in Dual-Phase Sr1-xCaxTi0.8Fe0.2O3-ẟ-Ce0.8Gd0.2O2 Membranes. Membranes 2025, 15, 258. https://doi.org/10.3390/membranes15090258
Nigroni V, Tang Y, Baumann S, Sebold D, Malgrati E, Fedeli P. Effects of Ca Substitution in Single-Phase Sr1-xCaxTi0.8Fe0.2O3-ẟ Oxygen Transport Membranes and in Dual-Phase Sr1-xCaxTi0.8Fe0.2O3-ẟ-Ce0.8Gd0.2O2 Membranes. Membranes. 2025; 15(9):258. https://doi.org/10.3390/membranes15090258
Chicago/Turabian StyleNigroni, Veronica, Yuning Tang, Stefan Baumann, Doris Sebold, Enrico Malgrati, and Paolo Fedeli. 2025. "Effects of Ca Substitution in Single-Phase Sr1-xCaxTi0.8Fe0.2O3-ẟ Oxygen Transport Membranes and in Dual-Phase Sr1-xCaxTi0.8Fe0.2O3-ẟ-Ce0.8Gd0.2O2 Membranes" Membranes 15, no. 9: 258. https://doi.org/10.3390/membranes15090258
APA StyleNigroni, V., Tang, Y., Baumann, S., Sebold, D., Malgrati, E., & Fedeli, P. (2025). Effects of Ca Substitution in Single-Phase Sr1-xCaxTi0.8Fe0.2O3-ẟ Oxygen Transport Membranes and in Dual-Phase Sr1-xCaxTi0.8Fe0.2O3-ẟ-Ce0.8Gd0.2O2 Membranes. Membranes, 15(9), 258. https://doi.org/10.3390/membranes15090258







