Hydration Fingerprints: A Reproducible Protocol for Accurate Water Uptake in Anion-Exchange Membranes
Abstract
1. Introduction
1.1. Purpose of the Study
1.2. Background and State-of-the-Art
2. Materials and Methods
2.1. Materials
2.2. Water Uptake Determination
- Conventional touch-blotting using Kimtech™ wipes, as routinely described in the literature [14,15,16].
- ○
- Standardized pressure-blotting (Figure 1): Samples were sandwiched between two pre-cut Kimtech™ layers, covered with a rigid pressing cap, and compressed twice for 10 s using a 1 kg weight (~0.44 N cm−2). By fixing both pressure and dwell time, this protocol eliminates operator variability and replicates the uniform compression experienced in full-cell assemblies—preventing local deformation while ensuring consistent surface-water removal. All materials were prepared in advance, and the membrane was weighed immediately after blotting using a balance positioned directly next to the setup, minimizing time-dependent variation due to ambient humidity or temperature. In addition, all handling was performed in a clean room with controlled environmental conditions to limit CO2 exposure, which can affect the ionic form of the membrane.
2.3. Water Uptake Protocol Variations
3. Results and Discussion
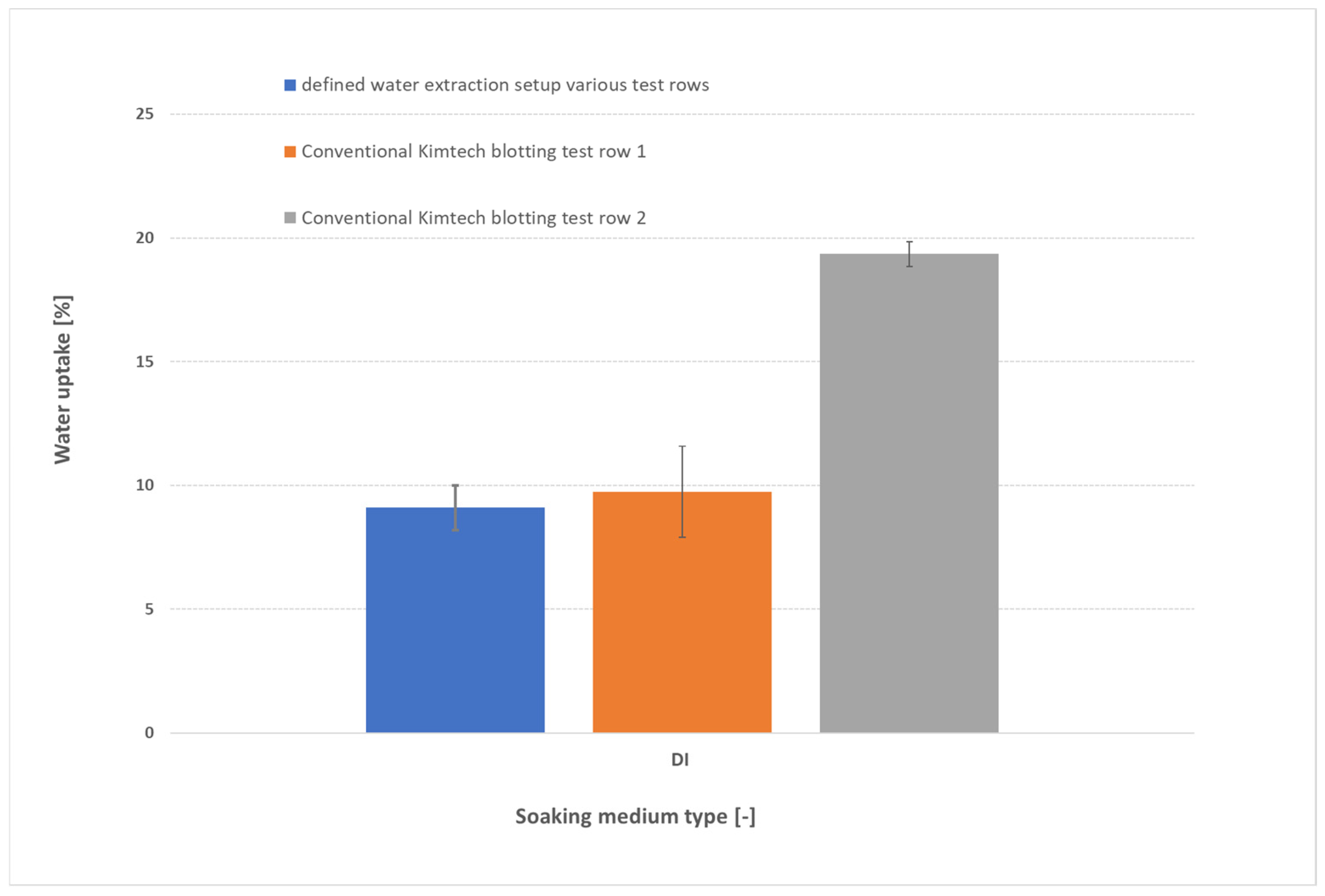
3.1. Influence of Blotting on Water Uptake
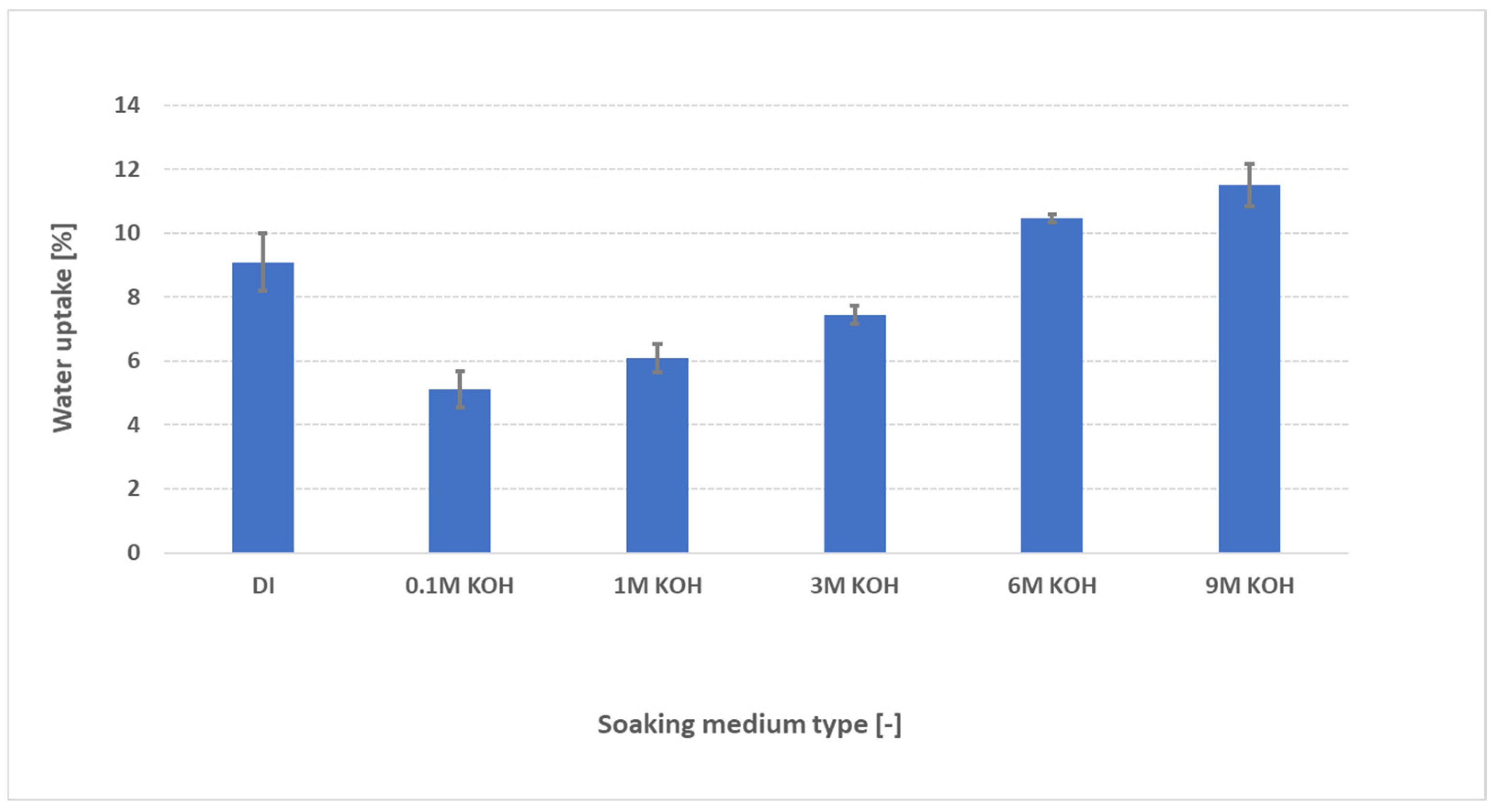
3.2. Evaluation of Effect of KOH Concentration on Water Uptake
3.3. Temperature-Dependent Water Uptake Behavior
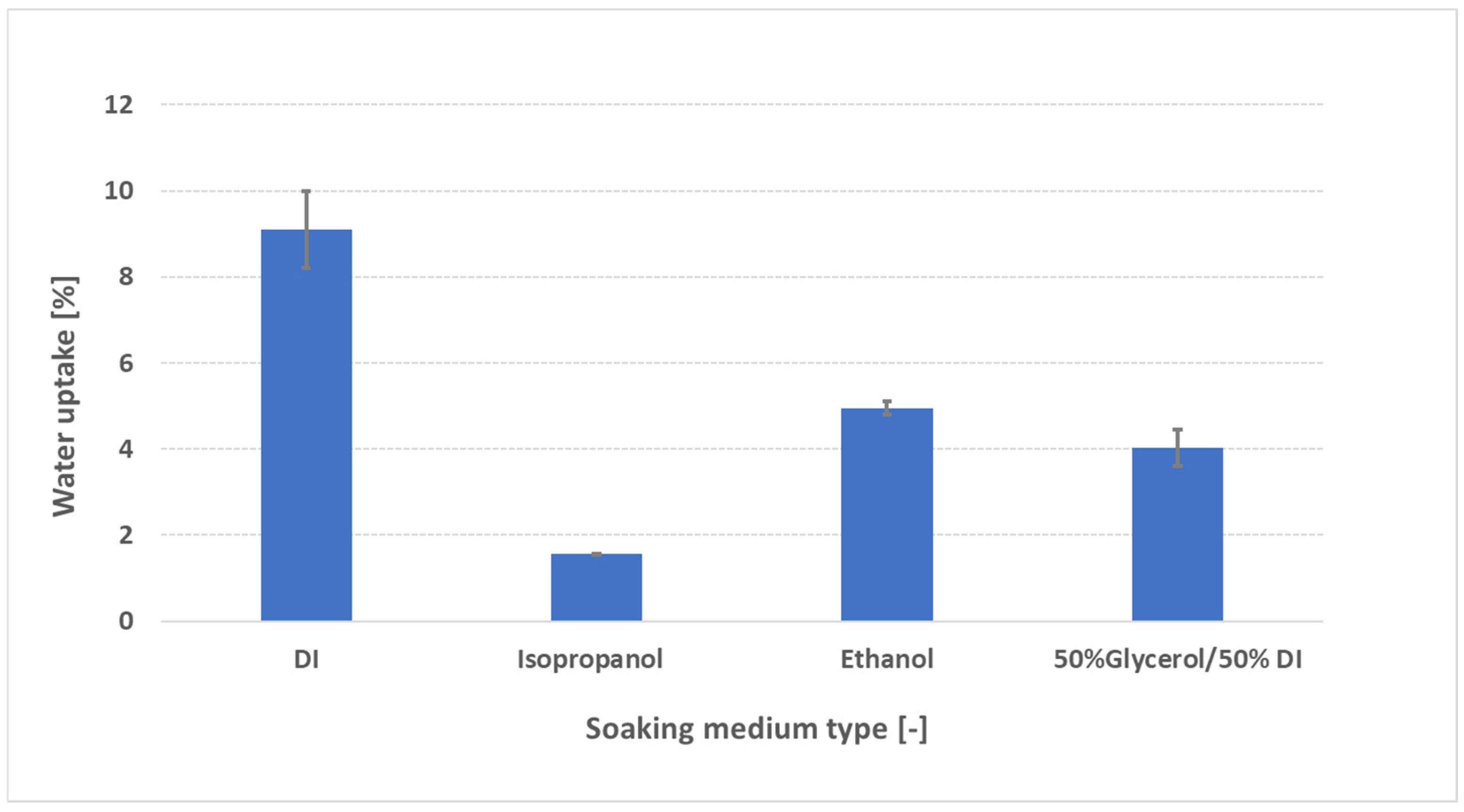
3.4. Effect of Swelling Media on Water Uptake Behavior
- Polarity (dielectric constant ε): DI water (ε ≈ 80) strongly solvates quaternary ammonium sites and OH− counter-ions, generating high osmotic pressure and maximal swelling. EtOH (ε ≈ 24) has lower polarity, so it cannot solvate ionic sites as effectively—yielding moderate uptake (~5 wt%). IPA (ε ≈ 18) is less polar still, so it forms fewer hydrogen bonds with ionic centers, resulting in minimal uptake (~3 wt%). A lower dielectric constant means the solvent cannot screen fixed charges or stabilize ion clusters as well, so its affinity for sulfonic acid (or quaternary ammonium) sites is reduced and swelling is suppressed [26].
- Molecular size and viscosity: IPA’s branched structure and higher viscosity (≈2 cP at 20 °C) hinder its diffusion into sub-nanometer hydrophilic channels, limiting uptake. EtOH (≈1.2 cP) diffuses more readily, while glycerol/DI (ε ≈ 50–55; high viscosity from glycerol’s three –OH groups) swells moderately (~6 wt%) by partially hydrating without full domain expansion [15].
- Hydrogen-bonding affinity: Glycerol’s multiple –OH groups support strong hydrogen bond networks that partially hydrate and plasticize the polymer backbone. EtOH forms fewer hydrogen bonds, and IPA’s single –OH bond (in a branched environment) yields a weaker, less extensive network, reducing its ability to open ionic channels [15]. All relevant parameters of the solvents are summarized in Table S2 of the Supplementary Material. From a practical standpoint, these uptake measurements could guide the selection of ink composition (e.g., ratio of DI to solvent—“dry ink” versus “liquid medium” ratios) or solvent type to minimize swelling and optimize the catalyst–membrane interface. In particular, pretreatment in any solvent immediately before ink application may help preserve membrane planarity, reduce delamination risk, and improve catalyst adhesion. Further studies—examining water uptake at different “DI to solvent ratios”, in different mixed-solvent systems, or for extended exposure times—are necessary to identify a more clear correlation between water uptake and the ideal ink formulation.
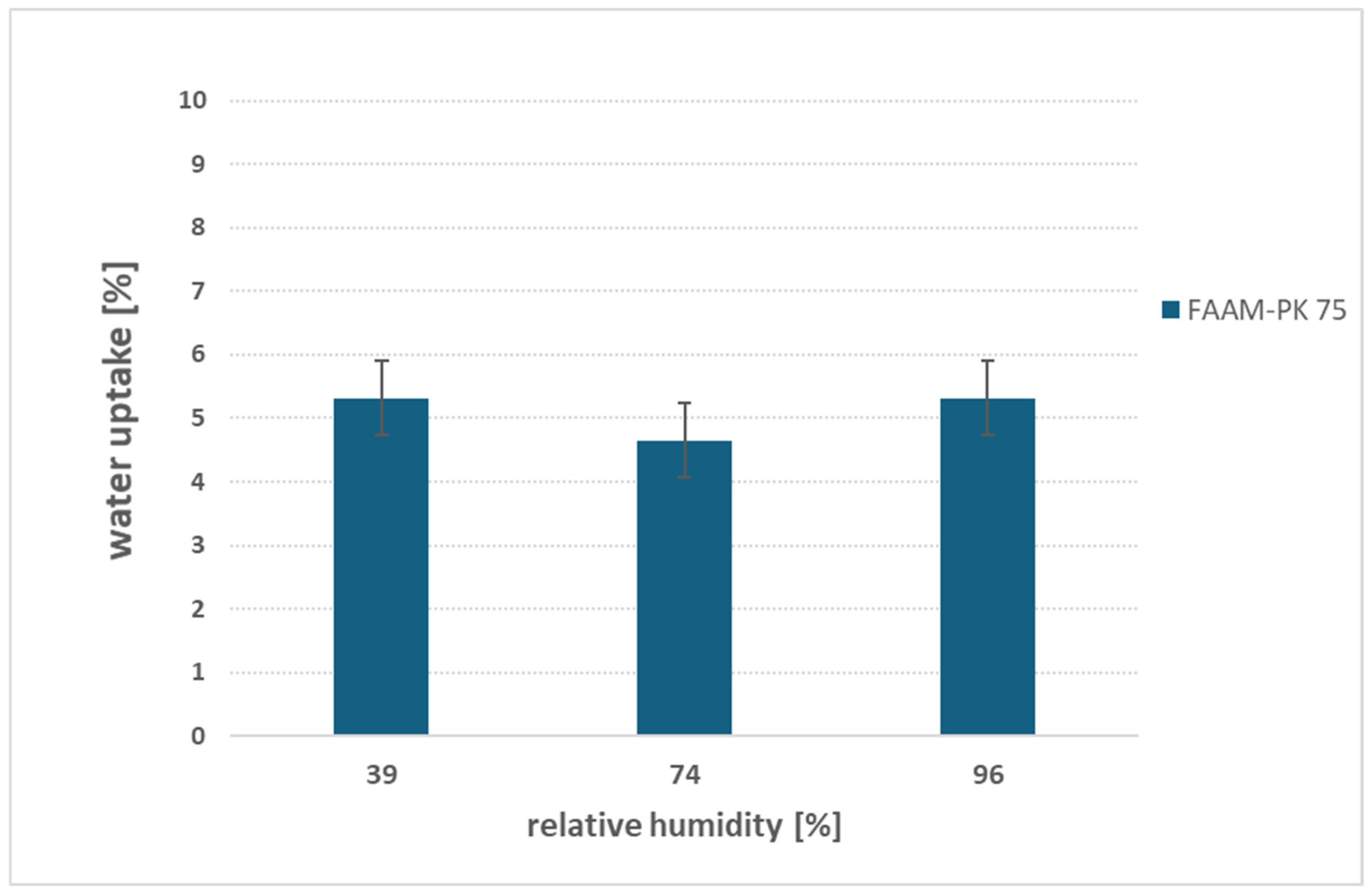
3.5. Effect of Relative Humidity on Water Uptake Behavior
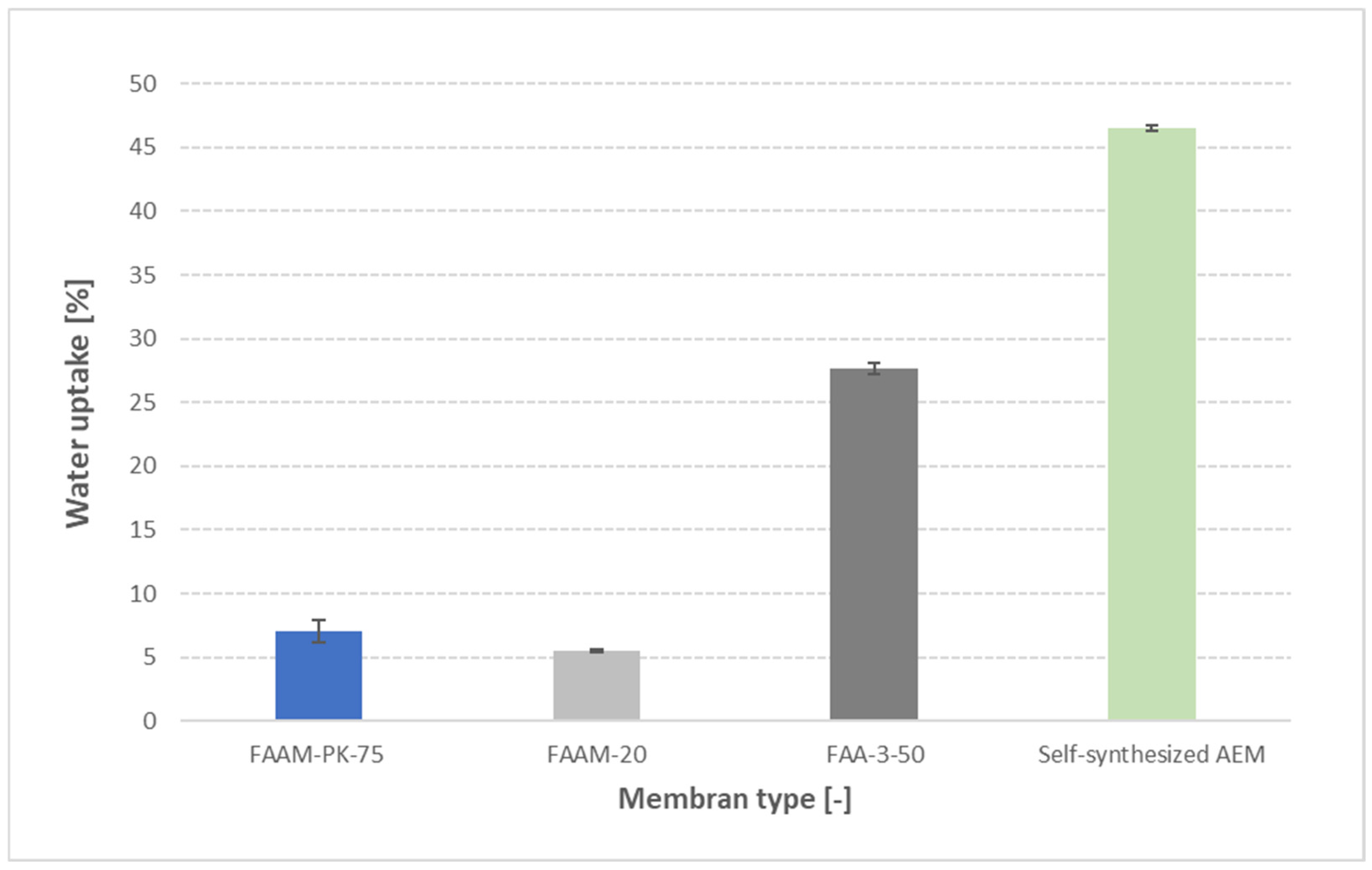
3.6. Cross-Validation with Other AEMs
4. Conclusions
- KOH concentration: Uptake drops from ≈9 wt% in DI water to ≈6 wt% at 1 M KOH (due to OH− screening), then increases to ≈11 wt% at 9 M as osmotic swelling overtakes screening.
- Swelling medium: Organic solvents are ranked as isopropanol (~1.5 wt%) < glycerol/DI (~4 wt%) < ethanol (~5 wt%) < DI water (~9 wt%), showing IPA as a promising low-swelling pre-soaking medium for ink compatibility.
- Temperature: Heating from 23 °C to 60 °C roughly halves uptake in DI water (thermal densification), while in 1 M KOH, a modest +2% increase indicates that external OH− partially counteracts densification.
- Relative humidity: Vapor-phase sorption over 39–96% RH yields a steady ≈5 wt% uptake for FAAM-PK-75, demonstrating that PEEK reinforcement inhibits gas-phase swelling and confirming that saturated salt environments offer a simple, reliable method for controlled-RH measurements.
5. Outlook
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Generative AI Disclosure
Abbreviations
| Abbreviation/Variables | Definition |
| AEM | Anion-exchange membrane |
| ATR-FTIR | Attenuated total reflectance–Fourier transform infrared spectroscopy |
| CCM | Catalyst-coated membrane |
| DFT | Density functional theory |
| DI | Deionized (water) |
| DVS | Dynamic vapor sorption |
| DSC | Differential scanning calorimetry |
| EIS | Electrochemical impedance spectroscopy |
| ETFE | Ethylene tetrafluoroethylene |
| IEC | Ion-exchange capacity |
| IPA | Isopropanol (2-propanol) |
| KOH | Potassium hydroxide |
| MeOH | Methanol |
| MD | Molecular dynamics |
| PEEK | Polyether ether ketone |
| PPO | Poly(phenylene oxide) |
| QPTTP-x | Quaternary phosphonium–trimethylpiperidinium copolymers |
| RH | Relative humidity |
| TGA | Thermogravimetric analysis |
| WU | Water uptake |
| mdry | Dried mass of the membrane |
| mwet | Wet mass of the membrane |
| n | Number of measurements repetition |
References
- Chatenet, M.; Pollet, B.G.; Dekel, D.R.; Dionigi, F.; Deseure, J.; Millet, P.; Braatz, R.D.; Bazant, M.Z.; Eikerling, M.; Staffell, I.; et al. Water Electrolysis: From Textbook Knowledge to the Latest Scientific Strategies and Industrial Developments. Chem. Soc. Rev. 2022, 51, 4583–4762. [Google Scholar] [CrossRef]
- Ashdot, A.; Kattan, M.; Kitayev, A.; Tal-Gutelmacher, E.; Amel, A.; Page, M. Design Strategies for Alkaline Exchange Membrane–Electrode Assemblies: Optimization for Fuel Cells and Electrolyzers. Membranes 2021, 11, 686. [Google Scholar] [CrossRef]
- Henkensmeier, D.; Cho, W.-C.; Jannasch, P.; Stojadinovic, J.; Li, Q.; Aili, D.; Jensen, J.O. Separators and Membranes for Advanced Alkaline Water Electrolysis. Chem. Rev. 2024, 124, 6393–6443. [Google Scholar] [CrossRef] [PubMed]
- ANION CORDIS, Grant Agreement ID: 875024; Anion Exchange Membrane Electrolysis for Renewable Hydrogen Production on a Wide-Scale, Roma, Italy, 2020. [CrossRef]
- Luque Di Salvo, J.; De Luca, G.; Cipollina, A.; Micale, G. Effect of Ion Exchange Capacity and Water Uptake on Hydroxide Transport in PSU-TMA Membranes: A DFT and Molecular Dynamics Study. J. Membr. Sci. 2020, 599, 117837. [Google Scholar] [CrossRef]
- Khalid, H.; Najibah, M.; Park, H.; Bae, C.; Henkensmeier, D. Properties of Anion Exchange Membranes with a Focus on Water Electrolysis. Membranes 2022, 12, 989. [Google Scholar] [CrossRef] [PubMed]
- Wijaya, G.H.A.; Im, K.S.; Nam, S.Y. Advancements in Commercial Anion Exchange Membranes: A Review of Membrane Properties in Water Electrolysis Applications. Desalination Water Treat. 2024, 320, 100605. [Google Scholar] [CrossRef]
- Lee, S.J.; Shin, S.-H.; Cha, M.S.; Yang, S.H.; Kim, T.H.; Cho, H.J.; Oh, K.-H.; Kim, T.-H.; Kim, S.; Lee, J.Y. Anisotropic Polyphenylene-Based Anion-Exchange Membranes with Flexible Side-Chains via Click Reaction for High-Performance Water Electrolysis. Mater. Today Energy 2024, 43, 101602. [Google Scholar] [CrossRef]
- Vandiver, M.A.; Caire, B.R.; Carver, J.R.; Waldrop, K.; Hibbs, M.R.; Varcoe, J.R.; Herring, A.M.; Liberatore, M.W. Mechanical Characterization of Anion Exchange Membranes by Extensional Rheology under Controlled Hydration. J. Electrochem. Soc. 2014, 161, H677–H683. [Google Scholar] [CrossRef]
- Fumasep FAAM-PK-75 Datasheet. Available online: https://www.quintech.de/wp-content/uploads/2020/03/fumasep-FAA-3-PK-75_v2.2.pdf (accessed on 6 June 2025).
- Zheng, Y.; Ash, U.; Pandey, R.P.; Ozioko, A.G.; Ponce-González, J.; Handl, M.; Weissbach, T.; Varcoe, J.R.; Holdcroft, S.; Liberatore, M.W.; et al. Water Uptake Study of Anion Exchange Membranes. Macromolecules 2018, 51, 3264–3278. [Google Scholar] [CrossRef]
- Zhang, S.; Li, X.; Yang, Y.; Li, J.; Zheng, J.; Zhang, S. Microporous and Low Swelling Branched Poly(Aryl Piperidinium) Anion Exchange Membranes for High-Performed Water Electrolyzers. J. Membr. Sci. 2024, 698, 122587. [Google Scholar] [CrossRef]
- Duan, Q.; Ge, S.; Wang, C.-Y. Water Uptake, Ionic Conductivity and Swelling Properties of Anion-Exchange Membrane. J. Power Sources 2013, 243, 773–778. [Google Scholar] [CrossRef]
- Tomasino, E.; Mukherjee, B.; Ataollahi, N.; Scardi, P. Water Uptake in an Anion Exchange Membrane Based on Polyamine: A First-Principles Study. J. Phys. Chem. B 2022, 126, 7418–7428. [Google Scholar] [CrossRef]
- Laín, L.; Barragán, V.M. Swelling Properties of Alkali-Metal Doped Polymeric Anion-Exchange Membranes in Alcohol Media for Application in Fuel Cells. Int. J. Hydrogen Energy 2016, 41, 14160–14170. [Google Scholar] [CrossRef]
- Tham, D.D.; Kim, D. C2 and N3 Substituted Imidazolium Functionalized Poly(Arylene Ether Ketone) Anion Exchange Membrane for Water Electrolysis with Improved Chemical Stability. J. Membr. Sci. 2019, 581, 139–149. [Google Scholar] [CrossRef]
- Ge, Q.; Zhu, X.; Yang, Z. Highly Conductive and Water-Swelling Resistant Anion Exchange Membrane for Alkaline Fuel Cells. IJMS 2019, 20, 3470. [Google Scholar] [CrossRef] [PubMed]
- Rana, M.M.; Rajkumar, P.; Kang, B.-S.; Park, G.; Sun, H.-J.; Kim, S.Y.; Lee, H.-K.; Shim, J. Optimizing Membrane Pretreatment for Improved Performance of Anion Exchange Membrane-Unitized Regenerative Fuel Cells. J. Appl. Electrochem. 2024, 54, 53–63. [Google Scholar] [CrossRef]
- Pandey, T.P.; Maes, A.M.; Sarode, H.N.; Peters, B.D.; Lavina, S.; Vezzù, K.; Yang, Y.; Poynton, S.D.; Varcoe, J.R.; Seifert, S.; et al. Interplay between Water Uptake, Ion Interactions, and Conductivity in an e-Beam Grafted Poly(Ethylene-Co-Tetrafluoroethylene) Anion Exchange Membrane. Phys. Chem. Chem. Phys. 2015, 17, 4367–4378. [Google Scholar] [CrossRef] [PubMed]
- Boström, O.; Choi, S.-Y.; Xia, L.; Meital, S.; Lohmann-Richters, F.; Jannasch, P. Alkali-Stable Polybenzimidazole Anion Exchange Membranes Tethered with N, N -Dimethylpiperidinium Cations for Dilute Aqueous KOH Fed Water Electrolyzers. J. Mater. Chem. A 2023, 11, 21170–21182. [Google Scholar] [CrossRef]
- Najibah, M.; Tsoy, E.; Khalid, H.; Chen, Y.; Li, Q.; Bae, C.; Hnát, J.; Plevová, M.; Bouzek, K.; Jang, J.H.; et al. PBI Nanofiber Mat-Reinforced Anion Exchange Membranes with Covalently Linked Interfaces for Use in Water Electrolysers. J. Membr. Sci. 2021, 640, 119832. [Google Scholar] [CrossRef]
- Ahmed, F.; Sutradhar, S.C.; Ryu, T.; Jang, H.; Choi, K.; Yang, H.; Yoon, S.; Rahman, M.M.; Kim, W. Comparative Study of Sulfonated Branched and Linear Poly(Phenylene)s Polymer Electrolyte Membranes for Fuel Cells. Int. J. Hydrogen Energy 2018, 43, 5374–5385. [Google Scholar] [CrossRef]
- Kusoglu, A.; Weber, A.Z. New Insights into Perfluorinated Sulfonic-Acid Ionomers. Chem. Rev. 2017, 117, 987–1104. [Google Scholar] [CrossRef]
- Godino, M.P.; Barragán, V.M.; Villaluenga, J.P.G.; Izquierdo-Gil, M.A.; Ruiz-Bauzá, C.; Seoane, B. Liquid Transport through Sulfonated Cation-Exchange Membranes for Different Water–Alcohol Solutions. Chem. Eng. J. 2010, 162, 643–648. [Google Scholar] [CrossRef]
- Yi, Y.D.; Bae, Y.C. Swelling Behaviors of Proton Exchange Membranes in Alcohols. Polymer 2017, 130, 112–123. [Google Scholar] [CrossRef]
- Khan, M.I.; Khraisheh, M. Synthesis and Characterization of Stable Anion Exchange Membranes for Desalination Applications. Desalination Water Treat. 2018, 113, 36–44. [Google Scholar] [CrossRef]
- Fumasep-Faa-3-50-Technical-Specifications. Available online: https://www.quintech.de/wp-content/uploads/2020/03/fumasep-FAA-3-50-onPET_v2.2.pdf (accessed on 6 June 2025).
- Fumasep FAAM-20-Technical Specifications. Available online: https://www.bwt.com/en/-/media/bwt/fumatech/datasheets/new/fumasep/awe/fumasep-faam-20-onpet_v2-2.pdf?rev=d6f8385f49b54229b76e0b2feb83f61c (accessed on 6 June 2025).
- PubChem-Isopropyl Alcohol. Available online: https://pubchem.ncbi.nlm.nih.gov/compound/Isopropyl-Alcohol#section=Viscosity (accessed on 30 June 2025).
- PubChem—Ethanol. Available online: https://pubchem.ncbi.nlm.nih.gov/compound/702 (accessed on 30 June 2025).
- CAS Common Chemistry Isopropanol Data Sheet. Available online: https://commonchemistry.cas.org/detail?ref=67-63-0 (accessed on 30 June 2025).
- Properties of Solvents Used in Organic Chemistry. Available online: http://murov.info/orgsolvents.htm (accessed on 30 June 2025).




| Protocol | Membrane Type | Soaking Medium | Duration (h) | T (°C) | rH (%) |
|---|---|---|---|---|---|
| Blotting comparison | FAAM-PK-75 |
| 24 | RT | 100 |
| KOH concentration effect | FAAM-PK-75 | KOH solutions (0.1 M–9 M) | 24 | RT | 100 |
| Temperature-dependent swelling | FAAM-PK-75 |
| 24 | RT and 60 | 100 |
| Alternative immersion media | FAAM-PK-75 |
| 24 | RT | 100 |
| RH-controlled sorption | FAAM-PK-75 | Vapor over salts:
| 48 h | RT | 33/75/93 |
| Cross-validation | -FAAM-20, -FAA-3-50, -self-synthesized AEM | 1 M KOH | 24 | RT | 100 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Temmel, S.E.; Ölschläger, D.; Wörner, R. Hydration Fingerprints: A Reproducible Protocol for Accurate Water Uptake in Anion-Exchange Membranes. Membranes 2025, 15, 257. https://doi.org/10.3390/membranes15090257
Temmel SE, Ölschläger D, Wörner R. Hydration Fingerprints: A Reproducible Protocol for Accurate Water Uptake in Anion-Exchange Membranes. Membranes. 2025; 15(9):257. https://doi.org/10.3390/membranes15090257
Chicago/Turabian StyleTemmel, Sandra Elisabeth, Daniel Ölschläger, and Ralf Wörner. 2025. "Hydration Fingerprints: A Reproducible Protocol for Accurate Water Uptake in Anion-Exchange Membranes" Membranes 15, no. 9: 257. https://doi.org/10.3390/membranes15090257
APA StyleTemmel, S. E., Ölschläger, D., & Wörner, R. (2025). Hydration Fingerprints: A Reproducible Protocol for Accurate Water Uptake in Anion-Exchange Membranes. Membranes, 15(9), 257. https://doi.org/10.3390/membranes15090257






