Sustainability in Membrane Technology: Membrane Recycling and Fabrication Using Recycled Waste
Abstract
1. Introduction
2. Polymer and Polymeric Membrane Recycling
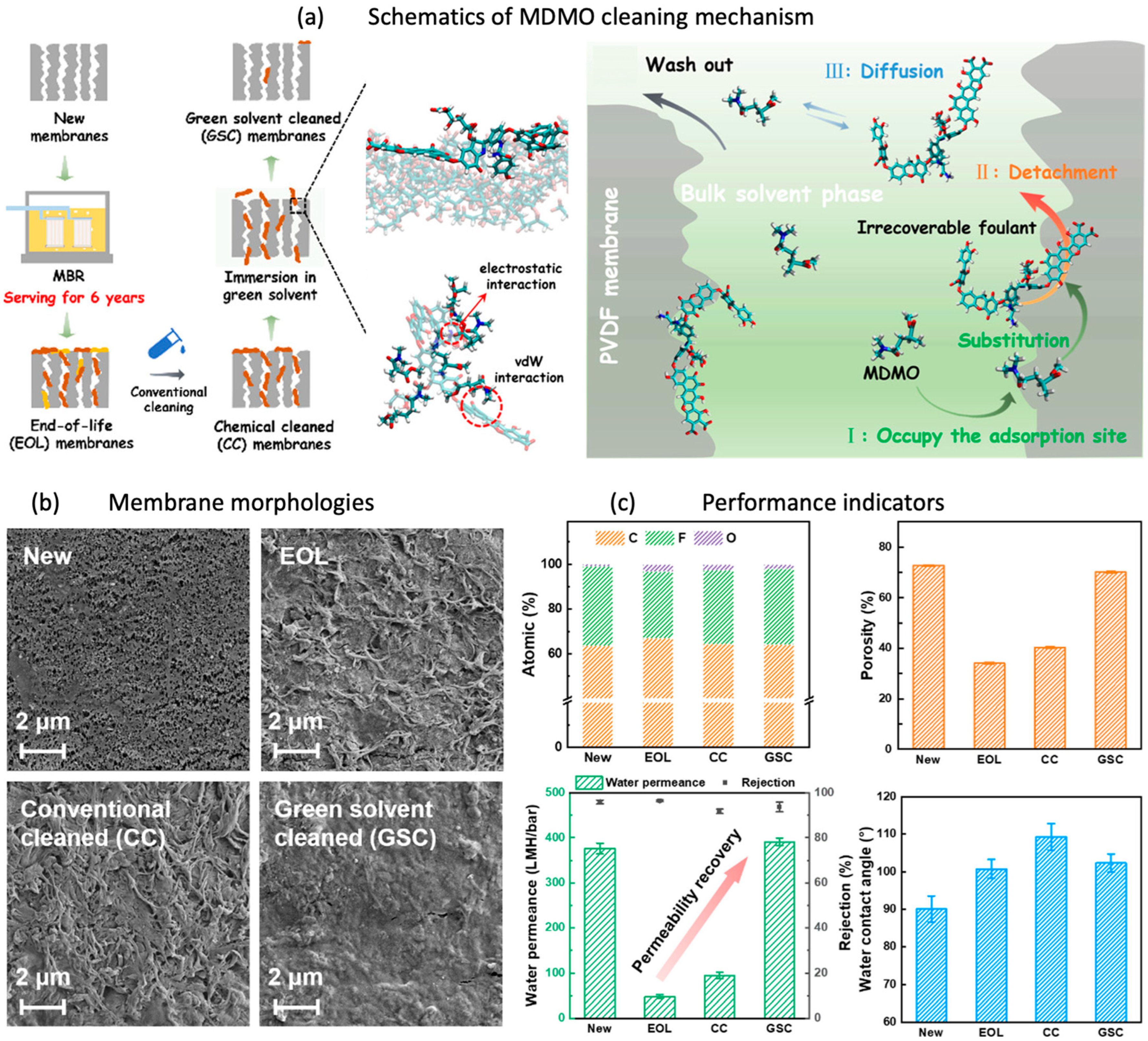
2.1. Polymeric Membrane Regeneration
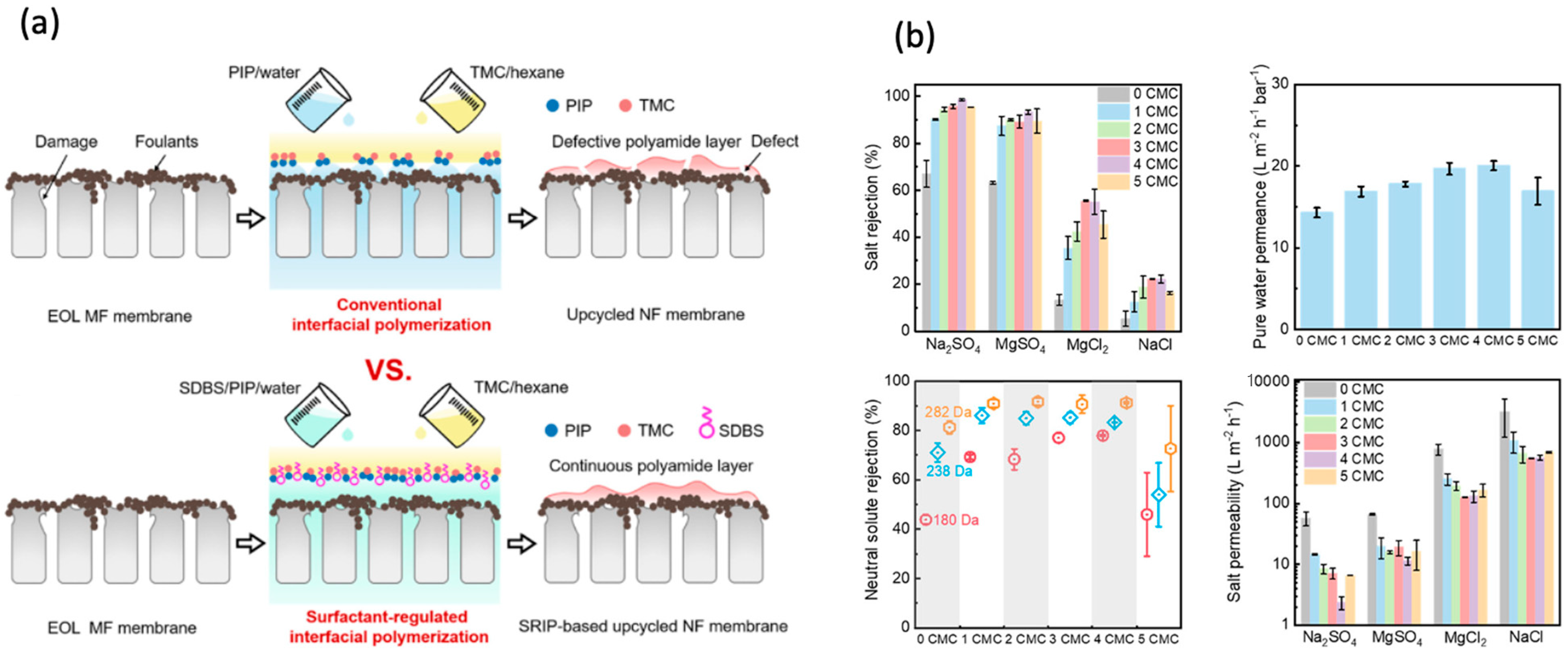
2.2. Polymeric Membrane Upcycling
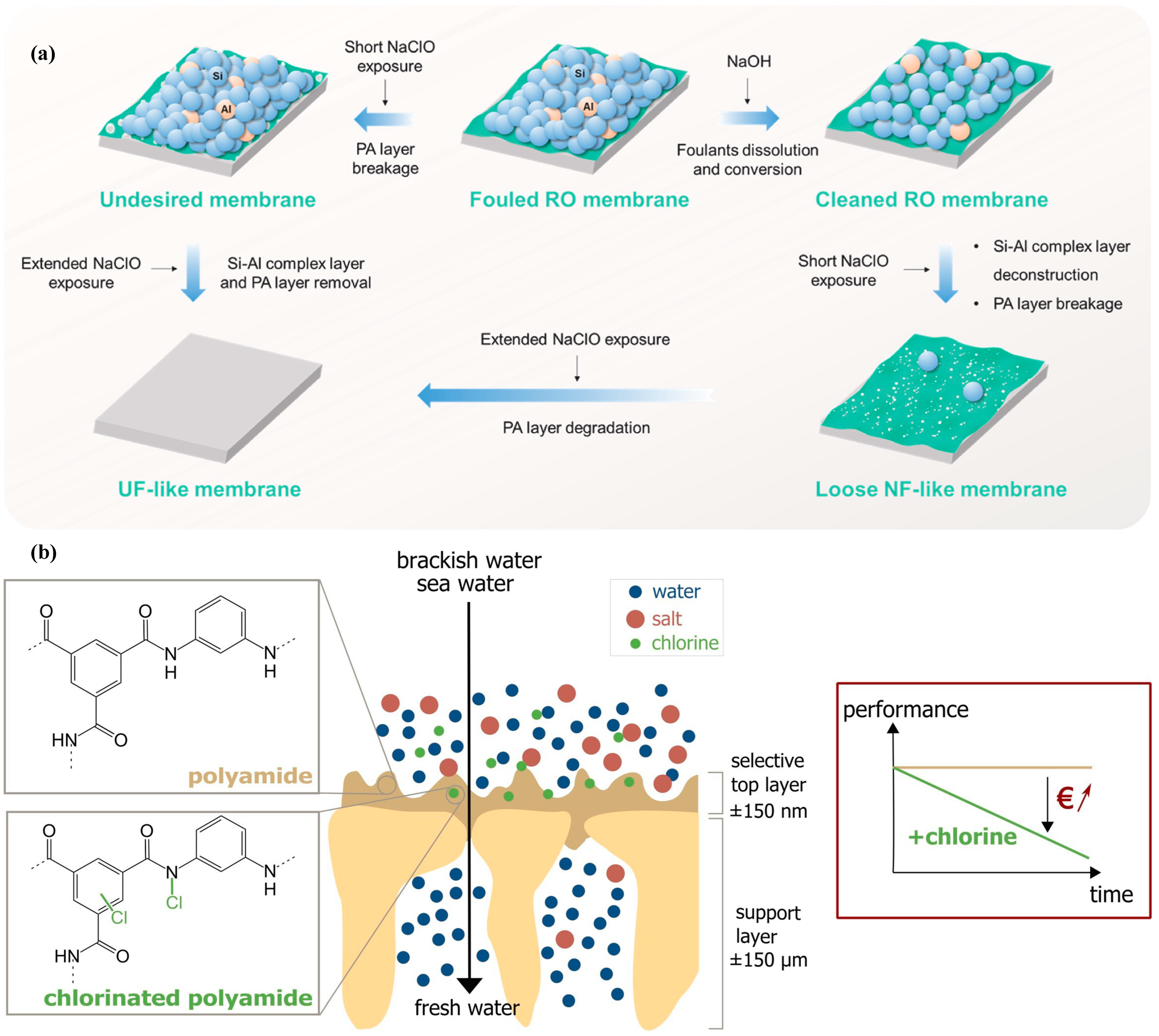
2.3. Polymeric Membrane Downcycling
2.4. Polymeric Membrane Re-Preparation
3. Membrane Fabrication Using Recycled Waste
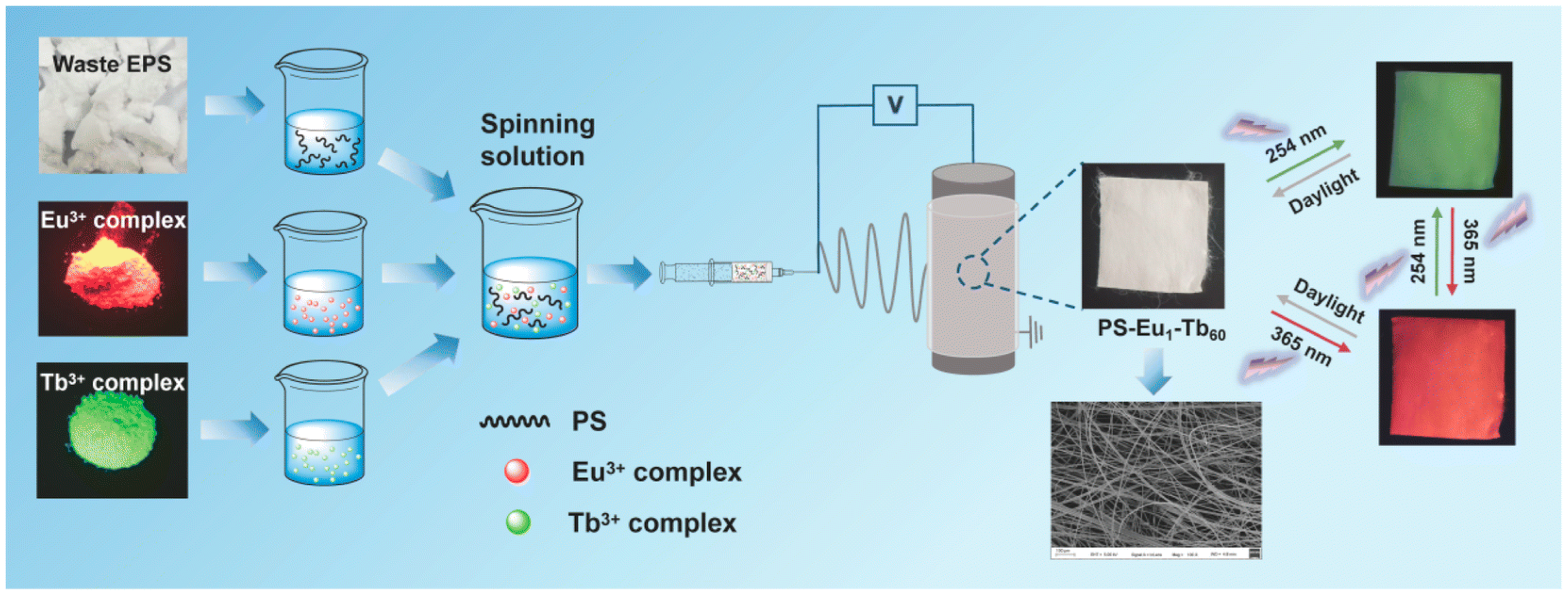
3.1. Recycled Polystyrene
3.2. Recycled Polyethylene Terephthalate
3.3. Recycled Polyvinyl Chloride
3.4. Recycled Tire Rubber
3.5. Recycled Keratin
3.6. Recycled Cellulose and Its Derivatives
4. Challenges, Limitations, and Future Work Directions
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- He, C.; Liu, Z.; Wu, J.; Pan, X.; Fang, Z.; Li, J.; Bryan, B.A. Future global urban water scarcity and potential solutions. Nat. Commun. 2021, 12, 4667. [Google Scholar] [CrossRef]
- Mekonnen, M.M.; Hoekstra, A.Y. Sustainability: Four billion people facing severe water scarcity. Sci. Adv. 2016, 2, 1500323. [Google Scholar] [CrossRef]
- van Vliet, M.T.H.; Jones, E.R.; Flörke, M.; Franssen, W.H.P.; Hanasaki, N.; Wada, Y.; Yearsley, J.R. Global water scarcity including surface water quality and expansions of clean water technologies. Environ. Res. Lett. 2021, 16, 024020. [Google Scholar] [CrossRef]
- Lavrnić, S.; Zapater-Pereyra, M.; Mancini, M.L. Water Scarcity and Wastewater Reuse Standards in Southern Europe: Focus on Agriculture. Water Air Soil Pollut. 2017, 228, 251. [Google Scholar] [CrossRef]
- Farid, M.U.; Kharraz, J.A.; Sun, J.; Boey, M.; Riaz, M.A.; Wong, P.W.; Jia, M.; Zhang, X.; Deka, B.J.; Khanzada, N.K.; et al. Advancements in Nano-enabled Membrane Distillation for A Sustainable Water-Energy-Environment Nexus. Adv. Mater. 2023, 2307950. [Google Scholar] [CrossRef] [PubMed]
- Salehi, M. Global water shortage and potable water safety; Today’s concern and tomorrow’s crisis. Environ. Int. 2022, 158, 106936. [Google Scholar] [CrossRef] [PubMed]
- Alkhudhiri, A.; Hilal, N. Membrane distillation-Principles, applications, configurations, design, and implementation. In Emerging Technologies for Sustainable Desalination Handbook; Elsevier: Amsterdam, The Netherlands, 2018; pp. 55–106. [Google Scholar] [CrossRef]
- Deka, B.J.; Guo, J.; Wong, P.W.; Khanzada, N.K.; Kharraz, J.A.; Tsang, C.W.; An, A.K. A Conductive Hydrophobic Polyaniline Sandwiched Polyvinylidene Fluoride Membrane for Early Detection of Surfactant-Induced Wetting in Membrane Distillation Using Impedance. ACS Appl. Polym. Mater. 2021, 3, 679–690. [Google Scholar] [CrossRef]
- Peng, J.; Deka, B.J.; Wu, S.; Luo, Z.; Kharraz, J.A.; Jia, W. Rational Design of PDA/P-PVDF@PP Janus Membrane with Asymmetric Wettability for Switchable Emulsion Separation. Membranes 2023, 13, 14. [Google Scholar] [CrossRef] [PubMed]
- Jung, B.; Ma, S.; Khor, C.M.; Khanzada, N.K.; Anvari, A.; Wang, X.; Im, S.; Wu, J.; Rao, U.; An, A.K.; et al. Impact of polarity reversal on inorganic scaling on carbon nanotube-based electrically-conducting nanofiltration membranes. Chem. Eng. J. 2023, 452, 139216. [Google Scholar] [CrossRef]
- Khanzada, N.K.; Choi, P.J.; An, A.K. Hybrid forward/reverse osmosis (HFRO): An approach for optimized operation and sustainable resource recovery. In Clean Energy and Resource Recovery: Wastewater Treatment Plants as Biorefineries; Elsevier: Amsterdam, The Netherlands, 2021; Volume 2, pp. 69–94. [Google Scholar] [CrossRef]
- Khanzada, N.K.; Farid, M.U.; Kharraz, J.A.; Choi, J.; Tang, C.Y.; Nghiem, L.D.; Jang, A.; An, A.K. Removal of organic micropollutants using advanced membrane-based water and wastewater treatment: A review. J. Membr. Sci. 2020, 598, 117672. [Google Scholar] [CrossRef]
- Hilal, N.; Kochkodan, V. Surface modified microfiltration membranes with molecularly recognising properties. J. Membr. Sci. 2003, 213, 97–113. [Google Scholar] [CrossRef]
- Hilal, N.; Nigmatullin, R.; Alpatova, A. Immobilization of cross-linked lipase aggregates within microporous polymeric membranes. J. Membr. Sci. 2004, 238, 131–141. [Google Scholar] [CrossRef]
- Alkhudhiri, A.; Hilal, N. Air gap membrane distillation: A detailed study of high saline solution. Desalination 2017, 403, 179–186. [Google Scholar] [CrossRef]
- Seman, M.N.A.; Khayet, M.; Hilal, N. Development of antifouling properties and performance of nanofiltration membranes modified by interfacial polymerisation. Desalination 2011, 273, 36–47. [Google Scholar] [CrossRef]
- Farhat, A.; Ahmad, F.; Hilal, N.; Arafat, H.A. Boron removal in new generation reverse osmosis (RO) membranes using two-pass RO without pH adjustment. Desalination 2013, 310, 50–59. [Google Scholar] [CrossRef]
- Al Aani, S.; Wright, C.J.; Atieh, M.A.; Hilal, N. Engineering nanocomposite membranes: Addressing current challenges and future opportunities. Desalination 2017, 401, 1–15. [Google Scholar] [CrossRef]
- Issaoui, M.; Jellali, S.; Zorpas, A.A.; Dutournie, P. Membrane technology for sustainable water resources management: Challenges and future projections. Sustain. Chem. Pharm. 2022, 25, 100590. [Google Scholar] [CrossRef]
- Khoo, Y.S.; Lau, W.J.; Hasan, S.W.; Salleh, W.N.W.; Ismail, A.F. New approach of recycling end-of-life reverse osmosis membranes via sonication for microfiltration process. J. Environ. Chem. Eng. 2021, 9, 106731. [Google Scholar] [CrossRef]
- Landaburu-Aguirre, J.; García-Pacheco, R.; Molina, S.; Rodríguez-Sáez, L.; Rabadán, J.; García-Calvo, E. Fouling prevention, preparing for re-use and membrane recycling. Towards circular economy in RO desalination. Desalination 2016, 393, 16–30. [Google Scholar] [CrossRef]
- Moradi, M.R.; Pihlajamäki, A.; Hesampour, M.; Ahlgren, J.; Mänttäri, M. End-of-life RO membranes recycling: Reuse as NF membranes by polyelectrolyte layer-by-layer deposition. J. Membr. Sci. 2019, 584, 300–308. [Google Scholar] [CrossRef]
- Lawler, W.; Bradford-Hartke, Z.; Cran, M.J.; Duke, M.; Leslie, G.; Ladewig, B.P.; Le-Clech, P. Towards new opportunities for reuse, recycling and disposal of used reverse osmosis membranes. Desalination 2012, 299, 103–112. [Google Scholar] [CrossRef]
- Senán-Salinas, J.; Landaburu-Aguirre, J.; Contreras-Martinez, J.; García-Calvo, E. Life Cycle Assessment application for emerging membrane recycling technologies: From reverse osmosis into forward osmosis. Resour. Conserv. Recycl. 2022, 179, 106075. [Google Scholar] [CrossRef]
- Rehman, S.; Yang, Y.S.; Patria, R.D.; Zulfiqar, T.; Khanzada, N.K.; Khan, R.J.; Lin, C.S.K.; Lee, D.J.; Leu, S.Y. Substrate-related factors and kinetic studies of Carbohydrate-Rich food wastes on enzymatic saccharification. Bioresour. Technol. 2023, 390, 129858. [Google Scholar] [CrossRef] [PubMed]
- Sharma, S.; Basu, S.; Shetti, N.P.; Kamali, M.; Walvekar, P.; Aminabhavi, T.M. Waste-to-energy nexus: A sustainable development. Environ. Pollut. 2020, 267, 115501. [Google Scholar] [CrossRef] [PubMed]
- Khoshsepehr, Z.; Alinejad, S.; Alimohammadlou, M. Exploring industrial waste management challenges and smart solutions: An integrated hesitant fuzzy multi-criteria decision-making approach. J. Clean Prod. 2023, 420, 138327. [Google Scholar] [CrossRef]
- Abdel-Shafy, H.I.; Mansour, M.S.M. Solid waste issue: Sources, composition, disposal, recycling, and valorization. Egypt J. Pet. 2018, 27, 1275–1290. [Google Scholar] [CrossRef]
- Bhat, R.A.; Singh, D.V.; Qadri, H.; Dar, G.H.; Dervash, M.A.; Bhat, S.A.; Unal, B.T.; Ozturk, M.; Hakeem, K.R.; Yousaf, B. Vulnerability of municipal solid waste: An emerging threat to aquatic ecosystems. Chemosphere 2022, 287, 132223. [Google Scholar] [CrossRef]
- Goh, P.S.; Othman, M.H.D.; Matsuura, T. Waste reutilization in polymeric membrane fabrication: A new direction in membranes for separation. Membranes 2021, 11, 782. [Google Scholar] [CrossRef]
- Sharifian, S.; Asasian-Kolur, N. Polyethylene terephthalate (PET) waste to carbon materials: Theory, methods and applications. J. Anal. Appl. Pyrolysis 2022, 163, 105496. [Google Scholar] [CrossRef]
- Aciu, C.; Manea, D.L.; Molnar, L.M.; Jumate, E. Recycling of Polystyrene Waste in the Composition of Ecological Mortars. Procedia Technol. 2015, 19, 498–505. [Google Scholar] [CrossRef]
- Akan, O.D.; Udofia, G.E.; Okeke, E.S.; Mgbechidinma, C.L.; Okoye, C.O.; Zoclanclounon, Y.A.B.; Atakpa, E.O.; Adebanjo, O.O. Plastic waste: Status, degradation and microbial management options for Africa. J. Environ. Manag. 2021, 292, 112758. [Google Scholar] [CrossRef]
- Rautela, R.; Arya, S.; Vishwakarma, S.; Lee, J.; Kim, K.H.; Kumar, S. E-waste management and its effects on the environment and human health. Sci. Total Environ. 2021, 773, 145623. [Google Scholar] [CrossRef]
- Rehman, S.; Islam, M.K.; Khanzada, N.K.; An, A.K.; Chaiprapat, S.; Leu, S.Y. Whole sugar 2,3-butanediol fermentation for oil palm empty fruit bunches biorefinery by a newly isolated Klebsiella pneumoniae PM2. Bioresour. Technol. 2021, 333, 125206. [Google Scholar] [CrossRef]
- Rehman, S.; Islam, M.K.; Khanzada, N.K.; Zhuang, H.; Wang, H.; Chaiprapat, S.; Leu, S.Y. Sustainability index accounting food and carbon benefits on circular 2,3-butanediol biorefinery with oil palm empty fruit bunches. Appl. Energy 2021, 303, 117667. [Google Scholar] [CrossRef]
- Rehman, Z.U.; Amjad, H.; Khan, S.J.; Yasmeen, M.; Khan, A.A.; Khanzada, N.K. Performance Evaluation of UF Membranes Derived from Recycled RO Membrane, a Step towards Circular Economy in Desalination. Membranes 2023, 13, 628. [Google Scholar] [CrossRef] [PubMed]
- Agyabeng-Mensah, Y.; Tang, L.; Afum, E.; Baah, C.; Dacosta, E. Organisational identity and circular economy: Are inter and intra organisational learning, lean management and zero waste practices worth pursuing? Sustain. Prod. Consum. 2021, 28, 648–662. [Google Scholar] [CrossRef]
- Jin, Y.; Du, J.; He, Y. Optimization of process planning for reducing material consumption in additive manufacturing. J. Manuf. Syst. 2017, 44, 65–78. [Google Scholar] [CrossRef]
- Haber, J.; Ausserwoeger, H.; Lehmann, C.; Pillet, L.; Schenkel, B.; Guélat, B. Minimizing Material Consumption in Flow Process Research and Development: A Novel Approach Toward Robust and Controlled Mixing of Reactants. Org. Process. Res. Dev. 2022, 26, 2456–2463. [Google Scholar] [CrossRef]
- Vezzoli, C. Design for Environmental Sustainability: Life Cycle Design of Products, 2nd ed.; Springer: London, UK, 2018. [Google Scholar] [CrossRef]
- Ghaly, S.T.; Noby, H.; Hayashi, J.I.; El-Shazly, A.H. Various waste polystyrene for useful membrane fabrication: Comparative experimental study. Mater. Today Proc. 2023. [Google Scholar] [CrossRef]
- Mejía, H.F.G.; Toledo-Alarcón, J.; Rodriguez, B.; Cifuentes, J.R.; Porré, F.O.; Haeger, M.P.L.; Ovalle, N.V.; Astudillo, C.L.; García, A. Direct recycling of discarded reverse osmosis membranes for domestic wastewater treatment with a focus on water reuse. Chem. Eng. Res. Des. 2022, 184, 473–487. [Google Scholar] [CrossRef]
- de Aguilar, D.C.B.; de Paula, E.C.; Amaral, M.C.S. Recycled membrane for drinking water treatment with high iron and manganese concentrations. J. Environ. Chem. Eng. 2023, 11, 110466. [Google Scholar] [CrossRef]
- Cote, P.; Alam, Z.; Penny, J. Hollow fiber membrane life in membrane bioreactors (MBR). Desalination 2012, 288, 145–151. [Google Scholar] [CrossRef]
- Xie, W.; Tiraferri, A.; Liu, B.; Tang, P.; Wang, F.; Chen, S.; Figoli, A.; Chu, L.Y. First Exploration on a Poly(vinyl chloride) Ultrafiltration Membrane Prepared by Using the Sustainable Green Solvent PolarClean. ACS Sustain. Chem. Eng. 2020, 8, 91–101. [Google Scholar] [CrossRef]
- Tian, C.; Wang, T.; Han, H.; Dai, R.; Wang, Z. Green Solvent Cleaning Removes Irrecoverable Foulants from End-of-Life Membranes in Membrane Bioreactors: Efficacy and Mechanisms. Environ. Sci. Technol. 2022, 56, 12563–12572. [Google Scholar] [CrossRef] [PubMed]
- Tian, C.; Chen, J.; Li, X.; Dai, R.; Wang, Z. Chemical cleaning−solvent treatment−hydrophilic modification strategy for regenerating end-of-life PVDF membrane. J. Membr. Sci. 2023, 669, 121325. [Google Scholar] [CrossRef]
- Deqian, R. Cleaning and Regeneration of Membranes; Elsevier Science Publishers B.V.: Amsterdam, The Netherlands, 1987. [Google Scholar]
- Pontie, M.; Bilongo, T.G.; Roesink, E.; de Grooth, J.; Dinaux, C.; Hannachi, A.; Charlot, F.; Oubaid, F.; Mrimi, S.; Shabani, M. Confronting two nanofiltration membranes for lithium-calcium selective separation in natural brines. Emergent. Mater. 2022, 5, 1495–1505. [Google Scholar] [CrossRef]
- Ma, B.; Ulbricht, M.; Hu, C.; Fan, H.; Wang, X.; Pan, Y.R.; Hosseini, S.S.; Panglisch, S.; Van der Bruggen, B.; Wang, Z. Membrane Life Cycle Management: An Exciting Opportunity for Advancing the Sustainability Features of Membrane Separations. Environ. Sci. Technol. 2023, 57, 3013–3020. [Google Scholar] [CrossRef] [PubMed]
- Chowdhury, M.R.; Steffes, J.; Huey, B.D.; Mccutcheon, J.R. 3D Printed Polyamide Membranes for Desalination. Science 2018, 361, 682–686. [Google Scholar] [CrossRef] [PubMed]
- Dai, R.; Han, H.; Wang, T.; Li, J.; Tang, C.Y.; Wang, Z. Fouling is the beginning: Upcycling biopolymer-fouled substrates for fabricating high-permeance thin-film composite polyamide membranes. Green Chem. 2021, 23, 1013–1025. [Google Scholar] [CrossRef]
- Dai, R.; Chen, J.; Han, H.; Zhou, H.; Wang, Z. Interfacial Wettability Regulation Enables One-Step Upcycling of the End-of-Life Polymeric Microfiltration Membrane. ACS ES T Eng. 2023, 3, 479–486. [Google Scholar] [CrossRef]
- Dai, R.; Yang, Z.; Qiu, Z.; Long, L.; Tang, C.Y.; Wang, Z. Distinct impact of substrate hydrophilicity on performance and structure of TFC NF and RO polyamide membranes. J. Membr. Sci. 2022, 662, 120966. [Google Scholar] [CrossRef]
- Yang, Z.; Sun, P.F.; Li, X.; Gan, B.; Wang, L.; Song, X.; Park, H.D.; Tang, C.Y. A Critical Review on Thin-Film Nanocomposite Membranes with Interlayered Structure: Mechanisms, Recent Developments, and Environmental Applications. Environ. Sci. Technol. 2020, 54, 15563–15583. [Google Scholar] [CrossRef] [PubMed]
- Dai, R.; Han, H.; Wang, T.; Li, J.; Wu, Z.; Tang, C.Y.; Wang, Z. Cleaning-Healing-Interfacial Polymerization Strategy for Upcycling Real End-of-Life Polyvinylidene Fluoride Microfiltration Membranes. ACS Sustain. Chem. Eng. 2021, 9, 10352–10360. [Google Scholar] [CrossRef]
- Wang, X.; Han, H.; Zhou, H.; Wang, T.; Dai, R.; Wang, Z. Rapid Upcycling of End-of-Life Microfiltration Membrane Mediated by the Healing of Metal-Organic Complex. ACS Sustain. Chem. Eng. 2022, 10, 9841–9849. [Google Scholar] [CrossRef]
- Somrani, A.; Shabani, M.; Mohamed, Z.; Ghaffour, N.; Seibel, F.; Briao, V.B.; Pontié, M. Transforming an end-of-life reverse osmosis membrane in a cationic exchange membrane and its application in a fungal microbial fuel cell. Ionics 2021, 27, 3169–3184. [Google Scholar] [CrossRef]
- Peng, L.E.; Yang, Z.; Long, L.; Zhou, S.; Guo, H.; Tang, C.Y. A critical review on porous substrates of TFC polyamide membranes: Mechanisms, membrane performances, and future perspectives. J. Membr. Sci. 2022, 641, 119871. [Google Scholar] [CrossRef]
- Veza, J.M.; Rodriguez-Gonzalez, J.J. Second use for old reverse osmosis membranes: Wastewater treatment. Desalination 2003, 15, 65–72. [Google Scholar] [CrossRef]
- Khaless, K.; Achiou, B.; Boulif, R.; Benhida, R. Recycling of spent reverse osmosis membranes for second use in the clarification of wet-process phosphoric acid. Minerals 2021, 11, 637. [Google Scholar] [CrossRef]
- Rodriguez, J.J.; Jimhez, V.; Trujillo, O.; Vezab, J.M. Reuse of reverse osmosis membranes wastewater treatment in advanced. Desalination 2002, 150, 219–225. [Google Scholar] [CrossRef]
- Khanzada, N.K.; Jassby, D.; An, A.K. Conductive reverse osmosis membrane for electrochemical chlorine reduction and sustainable brackish water treatment. Chem. Eng. J. 2022, 435, 134858. [Google Scholar] [CrossRef]
- Lawler, W.; Antony, A.; Cran, M.; Duke, M.; Leslie, G.; Le-Clech, P. Production and characterisation of UF membranes by chemical conversion of used RO membranes. J. Membr. Sci. 2013, 447, 203–211. [Google Scholar] [CrossRef]
- García-Pacheco, R.; Landaburu-Aguirre, J.; Molina, S.; Rodríguez-Sáez, L.; Teli, S.B.; García-Calvo, E. Transformation of end-of-life RO membranes into NF and UF membranes: Evaluation of membrane performance. J. Membr. Sci. 2015, 495, 305–315. [Google Scholar] [CrossRef]
- De Paula, E.C.; Gomes, J.C.L.; Amaral, M.C.S. Recycling of end-of-life reverse osmosis membranes by oxidative treatment: A technical evaluation. Water Sci. Technol. 2017, 76, 605–622. [Google Scholar] [CrossRef]
- Zhang, X.; Choi, P.J.; Khanzada, N.K.; Wong, P.W.; An, A.K. Highly permeable chlorine-resistant forward osmosis membrane by grafting novel sulfonamide monomers. Desalination 2023, 564, 116754. [Google Scholar] [CrossRef]
- Zhang, X.; Choi, P.J.; Khanzada, N.K.; Sun, J.; Wong, P.W.; Guo, J.; Ling, L.; Wu, D.; Jang, A.; An, A.K. FO membrane fabricated by layer-by-layer interfacial polymerisation and grafted sulfonamide group for improving chlorine resistance and water permeability. J. Membr. Sci. 2022, 663, 121042. [Google Scholar] [CrossRef]
- Ahmed, J.; Jamal, Y. A pilot application of recycled discarded RO membranes for low strength gray water reclamation. Environ. Sci. Pollut. Res. 2020, 28, 34042–34050. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Xu, Y.; Ma, B.; Zou, W.; Zeng, J.; Dai, R.; Wang, Z. Alkaline pre-treatment enables controllable downcycling of Si-Al fouled end-of-life RO membrane to NF and UF membranes. J. Membr. Sci. 2024, 690, 122209. [Google Scholar] [CrossRef]
- Verbeke, R.; Gómez, V.; Vankelecom, I.F.J. Chlorine-resistance of reverse osmosis (RO) polyamide membranes. Prog. Polym. Sci. 2017, 72, 1–15. [Google Scholar] [CrossRef]
- Sun, J.; Jia, W.; Guo, J.; Khanzada, N.K.; Jin, P.; Wong, P.W.; Zhang, X.; An, A.K. Amino-embedded carbon quantum dots incorporated thin-film nanocomposite membrane for desalination by pervaporation. Desalination 2022, 533, 115742. [Google Scholar] [CrossRef]
- Senán-Salinas, J.; García-Pacheco, R.; Landaburu-Aguirre, J.; García-Calvo, E. Recycling of end-of-life reverse osmosis membranes: Comparative LCA and cost-effectiveness analysis at pilot scale. Resour. Conserv. Recycl. 2019, 150, 104423. [Google Scholar] [CrossRef]
- García-Pacheco, R.; Landaburu-Aguirre, J.; Terrero-Rodríguez, P.; Campos, E.; Molina-Serrano, F.; Rabadán, J.; Zarzo, D.; García-Calvo, E. Validation of recycled membranes for treating brackish water at pilot scale. Desalination 2018, 433, 199–208. [Google Scholar] [CrossRef]
- García-Pacheco, R.; Li, Q.; Comas, J.; Taylor, R.A.; Le-Clech, P. Novel housing designs for nanofiltration and ultrafiltration gravity-driven recycled membrane-based systems. Sci. Total Environ. 2021, 767, 144181. [Google Scholar] [CrossRef]
- Seibel, F.I.; Brião, V.B.; Maxime, P.; Seibel, F.I.; Otávio, G.; Giubel, M.; Brião, V.B.; Shabani, M.; Pontié, M. End-of-life reverse osmosis membranes: Recycle procedure and its applications for the treatment of brackish and surface water. J. Appl. Res. Water Wastewater 2021, 8, 77–87. [Google Scholar]
- de Paula, E.C.; Amaral, M.C.S. Environmental and economic evaluation of end-of-life reverse osmosis membranes recycling by means of chemical conversion. J. Clean Prod. 2018, 194, 85–93. [Google Scholar] [CrossRef]
- Li, B.; Wang, S.; Loh, X.J.; Li, Z.; Chung, T.S. Closed-loop recyclable membranes enabled by covalent adaptable networks for water purification. Proc. Natl. Acad. Sci. USA 2023, 120, e2301009120. [Google Scholar] [CrossRef] [PubMed]
- Wei, C.; Cheng, Q.; Lin, L.; He, Z.; Huang, K.; Ma, S.; Chen, L. One-step fabrication of recyclable polyimide nanofiltration membranes with high selectivity and performance stability by a phase inversion-based process. J. Mater. Sci. 2018, 53, 11104–11115. [Google Scholar] [CrossRef]
- Hardian, R.; Cywar, R.M.; Chen, E.Y.X.; Szekely, G. Sustainable nanofiltration membranes based on biosourced fully recyclable polyesters and green solvents. J. Membr. Sci. Lett. 2022, 2, 100016. [Google Scholar] [CrossRef]
- Xie, W.; Li, T.; Tiraferri, A.; Drioli, E.; Figoli, A.; Crittenden, J.C.; Liu, B. Toward the Next Generation of Sustainable Membranes from Green Chemistry Principles. ACS Sustain. Chem. Eng. 2021, 9, 50–75. [Google Scholar] [CrossRef]
- Patel, R.V.; Raj, G.B.; Chaubey, S.; Yadav, A. Investigation on the feasibility of recycled polyvinylidene difluoride polymer from used membranes for removal of methylene blue: Experimental and DFT studies. Water Sci. Technol. 2022, 86, 194–210. [Google Scholar] [CrossRef] [PubMed]
- Liang, L.; Veksha, A.; Amrad, M.Z.B.M.; Snyder, S.A.; Lisak, G. Upcycling of exhausted reverse osmosis membranes into value-added pyrolysis products and carbon dots. J. Hazard Mater. 2021, 419, 126472. [Google Scholar] [CrossRef]
- Yadav, P.; Ismail, N.; Essalhi, M.; Tysklind, M.; Athanassiadis, D.; Tavajohi, N. Assessment of the environmental impact of polymeric membrane production. J. Membr. Sci. 2021, 622, 118987. [Google Scholar] [CrossRef]
- Zulfi, A.; Munir, M.M.; Hapidin, D.A.; Rajak, A.; Edikresnha, D.; Iskandar, F.; Khairurrijal, K. Air filtration media from electrospun waste high-impact polystyrene fiber membrane. Mater. Res. Express 2018, 5, 035049. [Google Scholar] [CrossRef]
- Arrosyid, B.H.; Zulfi, A.; Nur’aini, S.; Hartati, S.; Rafryanto, A.F.; Noviyanto, A.; Hapidin, D.A.; Feriyanto, D.; Khairurrijal, K. High-Efficiency Water Filtration by Electrospun Expanded Polystyrene Waste Nanofibers. ACS Omega 2023, 8, 23664–23672. [Google Scholar] [CrossRef] [PubMed]
- Fan, Y.; Su, H.; Li, P.; Lin, M.; Liu, D.; Pei, K.; Cao, X. Upcycling waste expanded polystyrene into UV-excited dual-mode multicolor luminescent electrospun fiber membranes for advanced anti-counterfeiting. RSC Adv. 2023, 13, 10123–10134. [Google Scholar] [CrossRef]
- Ajari, H.; Zrelli, A.; Chaouachi, B.; Pontié, M. Preparation and characterization of hydrophobic flat sheet membranes based on a recycled polymer. Int. Polym. Process. 2019, 34, 376–382. [Google Scholar] [CrossRef]
- Li, M.E.; Zhao, H.B.; Cheng, J.B.; Wang, T.; Fu, T.; Zhang, A.N.; Wang, Y.Z. An Effective Green Porous Structural Adhesive for Thermal Insulating, Flame-Retardant, and Smoke-Suppressant Expandable Polystyrene Foam. Engineering 2022, 17, 151–160. [Google Scholar] [CrossRef]
- Chaukura, N.; Gwenzi, W.; Bunhu, T.; Ruziwa, D.T.; Pumure, I. Potential uses and value-added products derived from waste polystyrene in developing countries: A review. Resour. Conserv. Recycl. 2016, 107, 157–165. [Google Scholar] [CrossRef]
- Tajeddin, B.; Ahmadi, B.; Sohrab, F.; Chenarbon, H.A. Polymers for Modified Atmosphere Packaging Applications. In Food Packaging and Preservation; Elsevier: Amsterdam, The Netherlands, 2018; pp. 457–499. [Google Scholar] [CrossRef]
- Hidalgo-Crespo, J.; Moreira, C.M.; Jervis, F.X.; Soto, M.; Amaya, J.L.; Banguera, L. Circular economy of expanded polystyrene container production: Environmental benefits of household waste recycling considering renewable energies. Energy Rep. 2022, 8, 306–311. [Google Scholar] [CrossRef]
- Lithner, D.; Larsson, A.; Dave, G. Environmental and health hazard ranking and assessment of plastic polymers based on chemical composition. Sci. Total Environ. 2011, 409, 3309–3324. [Google Scholar] [CrossRef]
- Ramos-Olmos, A.R.; Rogel-Hernández, A.E.; Flores-López, B.L.Z.; Lin, B.; Wai, S.; Espinoza-Gómez, A.H. Synthesis and characterization of asymmetric ultrafiltration membrane made with recycled polystyrene foam and different additives. J. Chil. Chem. Soc. 2008, 53, 1705–1708. [Google Scholar] [CrossRef]
- Adamczak, M.; Kamińska, G.; Bohdziewicz, J. Application of waste polymers as basic material for ultrafiltration membranes preparation. Water 2020, 12, 179. [Google Scholar] [CrossRef]
- Ke, H.; Feldman, E.; Guzman, P.; Cole, J.; Wei, Q.; Chu, B.; Alkhudhiri, A.; Alrasheed, R.; Hsiao, B.S. Electrospun polystyrene nanofibrous membranes for direct contact membrane distillation. J. Membr. Sci. 2016, 515, 86–97. [Google Scholar] [CrossRef]
- Huan, S.; Liu, G.; Han, G.; Cheng, W.; Fu, Z.; Wu, Q.; Wang, Q. Effect of experimental parameters on morphological, mechanical and hydrophobic properties of electrospun polystyrene fibers. Materials 2015, 8, 2718–2734. [Google Scholar] [CrossRef]
- Sin, L.T.; Tueen, B.S. Plastics and environmental sustainability issues. In Plastics and Sustainability; Elsevier: Amsterdam, The Netherlands, 2023; pp. 1–43. [Google Scholar] [CrossRef]
- Ding, J.; Kong, Y.; Yang, J. Preparation of Polyimide/Polyethylene Terephthalate Composite Membrane for Li-Ion Battery by Phase Inversion. J. Electrochem. Soc. 2012, 159, A1198–A1202. [Google Scholar] [CrossRef]
- Arahman, N.; Fahrina, A.; Amalia, S.; Sunarya, R.; Mulyati, S. Effect of PVP on the characteristic of modified membranes made from waste PET bottles for humic acid removal. F1000Research 2017, 6, 668. [Google Scholar] [CrossRef]
- Passos de Oliveira Santos, R.; Fernanda Rossi, P.; Ramos, L.A.; Frollini, E. Renewable resources and a recycled polymer as raw materials: Mats from electrospinning of lignocellulosic biomass and PET solutions. Polymers 2018, 10, 538. [Google Scholar] [CrossRef]
- Zhang, Z.; Xie, G.; Xiao, K.; Kong, X.Y.; Li, P.; Tian, Y.; Wen, L.; Jiang, L. Asymmetric Multifunctional Heterogeneous Membranes for pH- and Temperature-Cooperative Smart Ion Transport Modulation. Adv. Mater. 2016, 28, 9613–9619. [Google Scholar] [CrossRef]
- Zhou, Z.; Fang, L.F.; Wang, S.Y.; Matsuyama, H. Improving bonding strength between a hydrophilic coating layer and poly(ethylene terephthalate) braid for preparing mechanically stable braid-reinforced hollow fiber membranes. J. Appl. Polym. Sci. 2018, 135, 46104. [Google Scholar] [CrossRef]
- Strain, I.N.; Wu, Q.; Pourrahimi, A.M.; Hedenqvist, M.S.; Olsson, R.T.; Andersson, R.L. Electrospinning of recycled PET to generate tough mesomorphic fibre membranes for smoke filtration. J. Mater. Chem. A Mater. 2015, 3, 1632–1640. [Google Scholar] [CrossRef]
- Espinoza, S.L.S.; Arbeitman, C.R.; Clochard, M.C.; Grasselli, M. Functionalization of nanochannels by radio-induced grafting polymerization on PET track-etched membranes. Radiat. Phys. Chem. 2014, 94, 72–75. [Google Scholar] [CrossRef]
- Wang, P.; Wang, M.; Liu, F.; Ding, S.; Wang, X.; Du, G.; Liu, J.; Apel, P.; Kluth, P.; Trautmann, C.; et al. Ultrafast ion sieving using nanoporous polymeric membranes. Nat. Commun. 2018, 9, 569. [Google Scholar] [CrossRef]
- Rajesh, S.; Murthy, Z.V.P. Ultrafiltration membranes from waste polyethylene terephthalate and additives: Synthesis and characterization. Quim. Nova 2014, 37, 653–657. [Google Scholar] [CrossRef]
- Kusumocahyo, S.P.; Ambani, S.K.; Kusumadewi, S.; Sutanto, H.; Widiputri, D.I.; Kartawiria, I.S. Utilization of used polyethylene terephthalate (PET) bottles for the development of ultrafiltration membrane. J. Environ. Chem. Eng. 2020, 8, 104381. [Google Scholar] [CrossRef]
- Kusumocahyo, S.P.; Ambani, S.K.; Marceline, S. Improved permeate flux and rejection of ultrafiltration membranes prepared from polyethylene terephthalate (PET) bottle waste. Sustain. Environ. Res. 2021, 31, 1–11. [Google Scholar] [CrossRef]
- Zander, N.E.; Gillan, M.; Sweetser, D. Recycled PET nanofibers for water filtration applications. Materials 2016, 9, 247. [Google Scholar] [CrossRef]
- Xu, G.R.; An, X.C.; Das, R.; Xu, K.; Xing, Y.L.; Hu, Y.X. Application of electrospun nanofibrous amphiphobic membrane using low-cost poly (ethylene terephthalate) for robust membrane distillation. J. Water Process Eng. 2020, 36, 101351. [Google Scholar] [CrossRef]
- Pulido, B.A.; Habboub, O.S.; Aristizabal, S.L.; Szekely, G.; Nunes, S.P. Recycled Poly(ethylene terephthalate) for High Temperature Solvent Resistant Membranes. ACS Appl. Polym. Mater. 2019, 1, 2379–2387. [Google Scholar] [CrossRef]
- Kiani, S.; Mousavi, S.M.; Bidaki, A. Preparation of polyethylene terephthalate/xanthan nanofiltration membranes using recycled bottles for removal of diltiazem from aqueous solution. J. Clean. Prod. 2021, 314, 128082. [Google Scholar] [CrossRef]
- Demirel, E.; Zhang, B.; Papakyriakou, M.; Xia, S.; Chen, Y. Fe2O3 nanocomposite PVC membrane with enhanced properties and separation performance. J. Membr. Sci. 2017, 529, 170–184. [Google Scholar] [CrossRef]
- Mishra, G.; Mukhopadhyay, M. Enhanced antifouling performance of halloysite nanotubes (HNTs) blended poly(vinyl chloride) (PVC/HNTs) ultrafiltration membranes: For water treatment. J. Ind. Eng. Chem. 2018, 63, 366–379. [Google Scholar] [CrossRef]
- Fang, L.F.; Jeon, S.; Kakihana, Y.; Kakehi, J.I.; Zhu, B.K.; Matsuyama, H.; Zhao, S. Improved antifouling properties of polyvinyl chloride blend membranes by novel phosphate based-zwitterionic polymer additive. J. Membr. Sci. 2017, 528, 326–335. [Google Scholar] [CrossRef]
- Wang, S.Y.; Fang, L.F.; Cheng, L.; Jeon, S.; Kato, N.; Matsuyama, H. Novel ultrafiltration membranes with excellent antifouling properties and chlorine resistance using a poly(vinyl chloride)-based copolymer. J. Membr. Sci. 2018, 549, 101–110. [Google Scholar] [CrossRef]
- Aji, M.M.; Narendren, S.; Purkait, M.K.; Katiyar, V. Utilization of waste polyvinyl chloride (PVC) for ultrafiltration membrane fabrication and its characterization. J. Environ. Chem. Eng. 2020, 8, 103650. [Google Scholar] [CrossRef]
- Aji, M.M.; Narendren, S.; Purkait, M.K.; Katiyar, V. Biopolymer (gum arabic) incorporation in waste polyvinylchloride membrane for the enhancement of hydrophilicity and natural organic matter removal in water. J. Water Process. Eng. 2020, 38, 101569. [Google Scholar] [CrossRef]
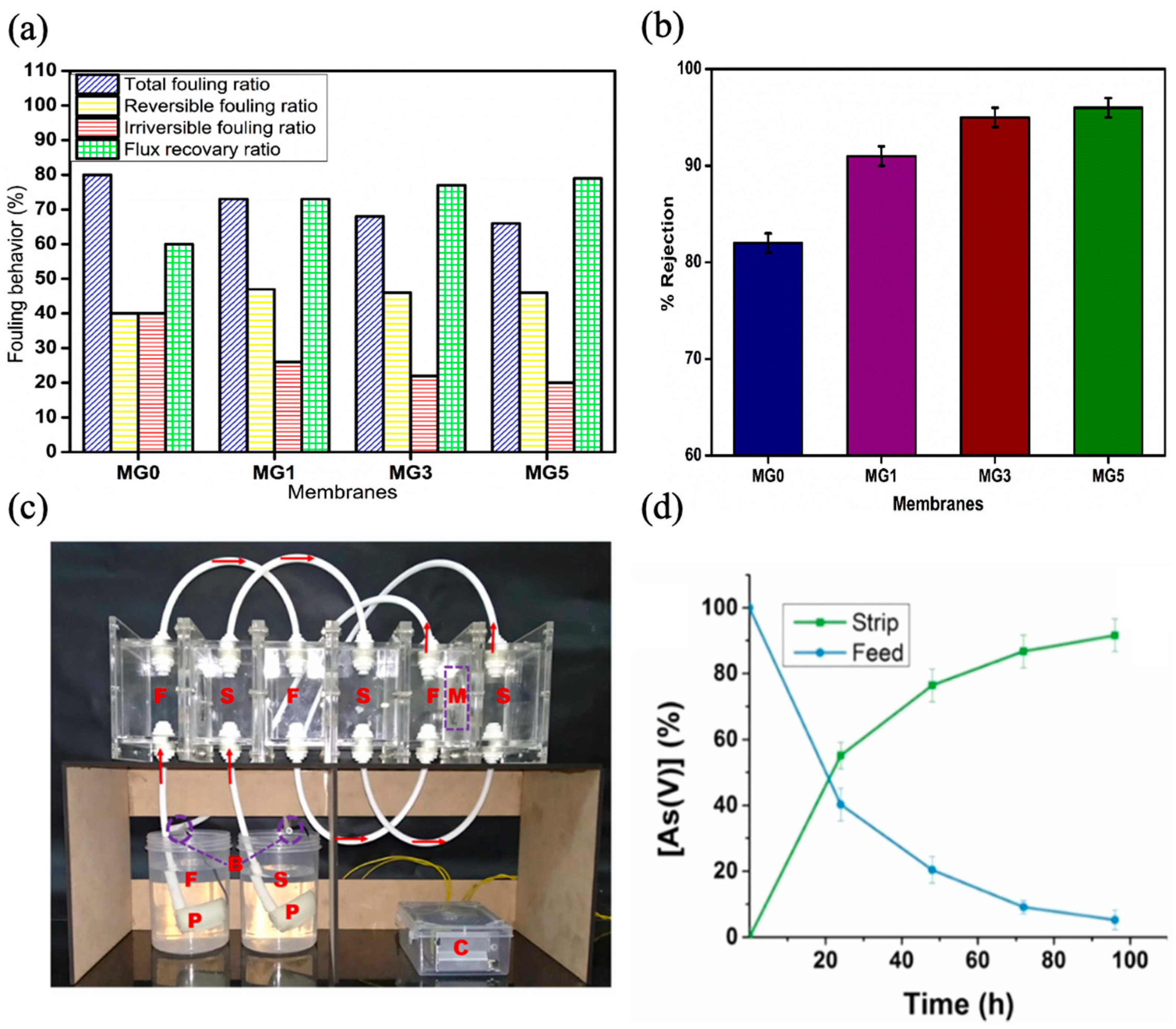
- Govindappa, H.; Bhat, M.P.; Uthappa, U.T.; Sriram, G.; Altalhi, T.; Kumar, S.P.; Kurkuri, M. Fabrication of a novel polymer inclusion membrane from recycled polyvinyl chloride for the real-time extraction of arsenic (V) from water samples in a continuous process. Chem. Eng. Res. Des. 2022, 182, 145–156. [Google Scholar] [CrossRef]
- Dobrotă, D.; Dobrotă, G. An innovative method in the regeneration of waste rubber and the sustainable development. J. Clean. Prod. 2018, 172, 3591–3599. [Google Scholar] [CrossRef]
- Asaro, L.; Gratton, M.; Seghar, S.; Hocine, N.A. Recycling of rubber wastes by devulcanization. Resour. Conserv. Recycl. 2018, 133, 250–262. [Google Scholar] [CrossRef]
- Ghorai, S.; Mondal, D.; Hait, S.; Ghosh, A.K.; Wiessner, S.; Das, A.; De, D. Devulcanization of Waste Rubber and Generation of Active Sites for Silica Reinforcement. ACS Omega 2019, 4, 17623–17633. [Google Scholar] [CrossRef]
- Lin, Y.T.; Kao, F.Y.; Chen, S.H.; Wey, M.Y.; Tseng, H.H. A facile approach from waste to resource: Reclaimed rubber-derived membrane for dye removal. J. Taiwan Inst. Chem. Eng. 2020, 112, 286–295. [Google Scholar] [CrossRef]
- Tseng, H.H.; Lin, Z.Y.; Chen, S.H.; Lai, W.H.; Wey, M.Y. Reuse of reclaimed tire rubber for gas-separation membranes prepared by hot-pressing. J. Clean. Prod. 2019, 237, 117739. [Google Scholar] [CrossRef]
- Zhuang, G.L.; Wey, M.Y.; Tseng, H.H. A novel technique using reclaimed tire rubber for gas separation membranes. J. Membr. Sci. 2016, 520, 314–325. [Google Scholar] [CrossRef]
- Reddy, C.C.; Khilji, I.A.; Gupta, A.; Bhuyar, P.; Mahmood, S.; AL-Japairai, K.A.S.; Chua, G.K. Valorization of keratin waste biomass and its potential applications. J. Water Process. Eng. 2021, 40, 101707. [Google Scholar] [CrossRef]
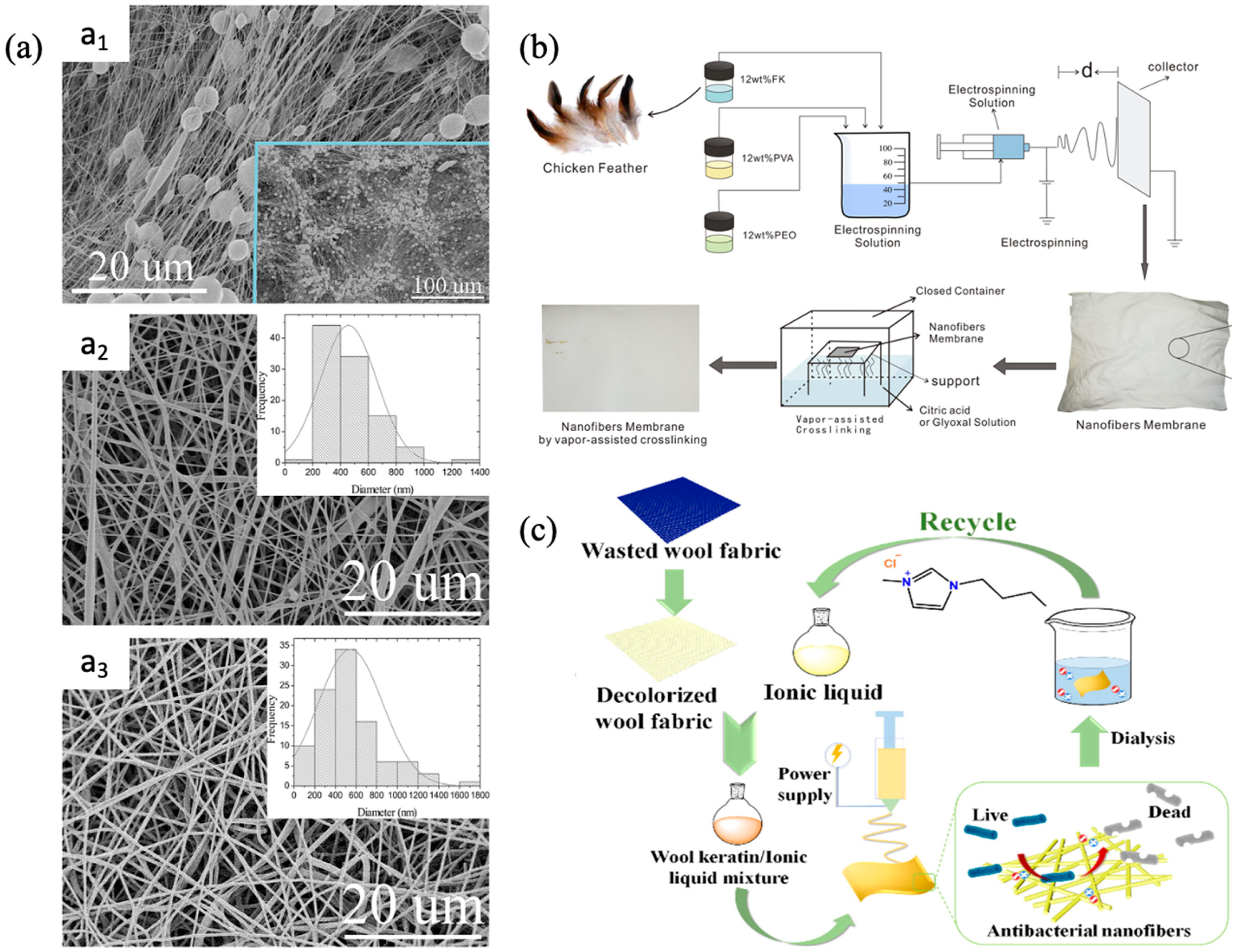
- Ma, H.; Shen, J.; Cao, J.; Wang, D.; Yue, B.; Mao, Z.; Wu, W.; Zhang, H. Fabrication of wool keratin/polyethylene oxide nano-membrane from wool fabric waste. J. Clean. Prod. 2017, 161, 357–361. [Google Scholar] [CrossRef]
- Ding, J.; Chen, M.; Chen, W.; He, M.; Zhou, X.; Yin, G. Vapor-assisted crosslinking of a FK/PVA/PEO nanofiber membrane. Polymers 2018, 10, 747. [Google Scholar] [CrossRef]
- Zhong, X.; Li, R.; Wang, Z.; Wang, W.; Yu, D. Eco-fabrication of antibacterial nanofibrous membrane with high moisture permeability from wasted wool fabrics. Waste Manag. 2020, 102, 404–411. [Google Scholar] [CrossRef]
- David, P.S.; Karunanithi, A.; Fathima, N.N. Improved filtration for dye removal using keratin-polyamide blend nanofibrous membranes. Environ. Sci. Pollut. Res. 2020, 27, 45629–45638. [Google Scholar] [CrossRef]
- Karunanidhi, A.; David, P.S.; Fathima, N.N. Electrospun Keratin-Polysulfone Blend Membranes for Treatment of Tannery Effluents. Water Air Soil Pollut. 2020, 231, 1–11. [Google Scholar] [CrossRef]
- Teow, Y.H.; Amirudin, S.N.; Ho, K.C. Sustainable approach to the synthesis of cellulose membrane from oil palm empty fruit bunch for dye wastewater treatment. J. Water Process Eng. 2020, 34, 101182. [Google Scholar] [CrossRef]
- Mohamed, M.A.; Salleh, W.N.W.; Jaafar, J.; Ismail, A.F.; Mutalib, M.A.; Mohamad, A.B.; Zain, M.F.; Awang, N.A.; Hir, Z.A.M. Physicochemical characterization of cellulose nanocrystal and nanoporous self-assembled CNC membrane derived from Ceiba pentandra. Carbohydr. Polym. 2017, 157, 1892–1902. [Google Scholar] [CrossRef] [PubMed]
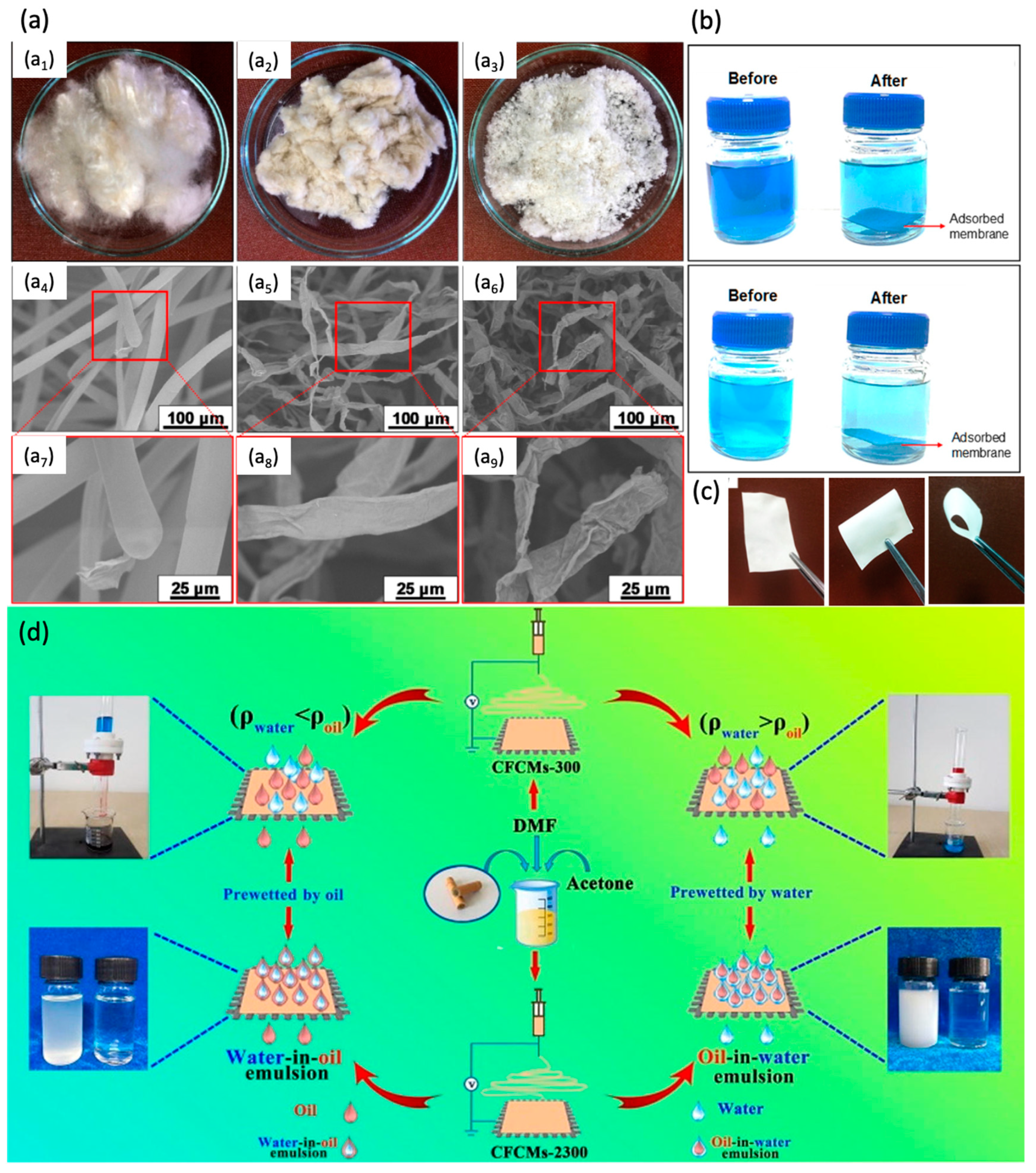
- Lopatina, A.; Anugwom, I.; Blot, H.; Conde, A.S.; Manttari, M.; Kallioinen, M. Re-use of waste cotton textile as an ultrafiltration membrane. J. Environ. Chem. Eng. 2021, 9, 105705. [Google Scholar] [CrossRef]
- Vignesh, N.; Suriyaraj, S.P.; Selvakumar, R.; Chandraraj, K. Facile Fabrication and Characterization of Zn Loaded Cellulose Membrane from Cotton Microdust Waste and its Antibacterial Properties—A Waste to Value Approach. J. Polym. Environ. 2021, 29, 1651–1662. [Google Scholar] [CrossRef]
- Liu, W.; Cui, M.; Shen, Y.; Zhu, G.; Luo, L.; Li, M.; Li, J. Waste cigarette filter as nanofibrous membranes for on-demand immiscible oil/water mixtures and emulsions separation. J. Colloid Interface Sci. 2019, 549, 114–122. [Google Scholar] [CrossRef] [PubMed]
- Mohamed, M.A.; Salleh, W.N.W.; Jaafar, J.; Ismail, A.F.; Mutalib, M.A.; Jamil, S.M. Feasibility of recycled newspaper as cellulose source for regenerated cellulose membrane fabrication. J. Appl. Polym. Sci. 2015, 132, 43. [Google Scholar] [CrossRef]
- Mohamed, M.A.; Salleh, W.W.N.; Jaafar, J.; Ismail, A.F.; Mutalib, M.A.; Sani, N.A.A.; Asri, S.E.A.M.; Ong, C.S. Physicochemical characteristic of regenerated cellulose/N-doped TiO2 nanocomposite membrane fabricated from recycled newspaper with photocatalytic activity under UV and visible light irradiation. Chem. Eng. J. 2016, 284, 202–215. [Google Scholar] [CrossRef]
- Doyan, A.; Leong, C.L.; Bilad, M.R.; Kurnia, K.A.; Susilawati, S.; Prayogi, S.; Narkkun, T.; Faungnawakij, K. Cigarette butt waste as material for phase inverted membrane fabrication used for oil/water emulsion separation. Polymers 2021, 13, 1907. [Google Scholar] [CrossRef]
- Senán-Salinas, J.; Landaburu-Aguirre, J.; García-Pacheco, R.; García-Calvo, E. Recyclability Definition of Recycled Nanofiltration Membranes through a Life Cycle Perspective and Carbon Footprint Indicator. Membranes 2022, 12, 854. [Google Scholar] [CrossRef]
- Senán-Salinas, J.; Blanco, A.; García-Pacheco, R.; Landaburu-Aguirre, J.; García-Calvo, E. Prospective Life Cycle Assessment and economic analysis of direct recycling of end-of-life reverse osmosis membranes based on Geographic Information Systems. J. Clean. Prod. 2021, 282, 124400. [Google Scholar] [CrossRef]



Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Khanzada, N.K.; Al-Juboori, R.A.; Khatri, M.; Ahmed, F.E.; Ibrahim, Y.; Hilal, N. Sustainability in Membrane Technology: Membrane Recycling and Fabrication Using Recycled Waste. Membranes 2024, 14, 52. https://doi.org/10.3390/membranes14020052
Khanzada NK, Al-Juboori RA, Khatri M, Ahmed FE, Ibrahim Y, Hilal N. Sustainability in Membrane Technology: Membrane Recycling and Fabrication Using Recycled Waste. Membranes. 2024; 14(2):52. https://doi.org/10.3390/membranes14020052
Chicago/Turabian StyleKhanzada, Noman Khalid, Raed A. Al-Juboori, Muzamil Khatri, Farah Ejaz Ahmed, Yazan Ibrahim, and Nidal Hilal. 2024. "Sustainability in Membrane Technology: Membrane Recycling and Fabrication Using Recycled Waste" Membranes 14, no. 2: 52. https://doi.org/10.3390/membranes14020052
APA StyleKhanzada, N. K., Al-Juboori, R. A., Khatri, M., Ahmed, F. E., Ibrahim, Y., & Hilal, N. (2024). Sustainability in Membrane Technology: Membrane Recycling and Fabrication Using Recycled Waste. Membranes, 14(2), 52. https://doi.org/10.3390/membranes14020052









