Correlation between Nafion Morphology in Various Dispersion Liquids and Properties of the Cast Membranes
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Obtaining Dispersions
2.3. Membrane Formation by Casting Method
2.4. Methods
3. Results and Discussions
3.1. Properties of Nafion Dispersions
3.2. Membrane Properties
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Connolly, D.J.; Gresham, W.F. Fluorocarbon Vinyl Ether Polymers. U.S. Patent No. 3,282,875, 1 November 1966. [Google Scholar]
- Kreuer, K.-D.; Paddison, S.J.; Spohr, E.; Schuster, M. Transport in Proton Conductors for Fuel-Cell Applications: Simulations, Elementary Reactions, and Phenomenology. Chem. Rev. 2004, 104, 4637–4678. [Google Scholar] [CrossRef] [Green Version]
- Prykhodko, Y.; Fatyeyeva, K.; Hespel, L.; Marais, S. Progress in hybrid composite Nafion®-based membranes for proton exchange fuel cell application. Chem. Eng. J. 2021, 409, 127329. [Google Scholar] [CrossRef]
- Filippov, S.P.; Yaroslavtsev, A.B. Hydrogen energy: Development prospects and materials. Russ. Chem. Rev. 2021, 90, 627–643. [Google Scholar] [CrossRef]
- Curtin, D.E.; Lousenberg, R.D.; Henry, T.J.; Tangeman, P.C.; Tisack, M.E. Advanced materials for improved PEMFC performance and life. J. Power Sources 2004, 131, 41–48. [Google Scholar] [CrossRef]
- Primachenko, O.N.; Marinenko, E.A.; Odinokov, A.S.; Kononova, S.V.; Kulvelis, Y.V.; Lebedev, V.T. State of the art and prospects in the development of proton-conducting perfluorinated membranes with short side chains: A review. Polym. Adv. Technol. 2020, 32, 1386–1408. [Google Scholar] [CrossRef]
- Kusoglu, A.; Weber, A.Z. New Insights into Perfluorinated Sulfonic-Acid Ionomers. Chem. Rev. 2017, 117, 987–1104. [Google Scholar] [CrossRef]
- Yue, M.; Jemei, S.; Zerhouni, N.; Gouriveau, R. Proton exchange membrane fuel cell system prognostics and decision-making: Current status and perspectives. Renew. Energy 2021, 179, 2277–2294. [Google Scholar] [CrossRef]
- Tsampas, M.N.; Pikos, A.; Brosda, S.; Katsaounis, A.; Vayenas, C.G. The effect of membrane thickness on the conductivity of Nafion. Electrochim. Acta 2006, 51, 2743–2755. [Google Scholar] [CrossRef]
- Seo, S.H.; Lee, C.S. The Effects of Membrane Thickness on the Performance and Impedance of the Direct Methanol Fuel Cell. Proc. Inst. Mech. Eng. Part C J. Mech. Eng. Sci. 2010, 224, 2211–2221. [Google Scholar] [CrossRef]
- Slade, S.M.; Ralph, T.R.; de León, C.P.; Campbell, S.A.; Walsh, F.C. The Ionic Conductivity of a Nafion® 1100 Series of Proton-exchange Membranes Re-cast from Butan-1-ol and Propan-2-ol. Fuel Cells 2010, 10, 567–574. [Google Scholar] [CrossRef]
- Slade, S.; Campbell, S.A.; Ralph, T.R.; Walsh, F.C. Ionic Conductivity of an Extruded Nafion 1100 EW Series of Membranes. J. Electrochem. Soc. 2002, 149, A1556. [Google Scholar] [CrossRef] [Green Version]
- Siroma, Z.; Kakitsubo, R.; Fujiwara, N.; Ioroi, T.; Yamazaki, S.; Yasuda, K. Depression of proton conductivity in recast Nafion® film measured on flat substrate. J. Power Sources 2009, 189, 994–998. [Google Scholar] [CrossRef]
- Moore, R.B.; Martin, C.R. Procedure for Preparing Solution-Cast Perfluorosulfonate Ionomer Films and Membranes. Anal. Chem. 1986, 58, 2569–2570. [Google Scholar] [CrossRef]
- Welch, C.; Labouriau, A.; Hjelm, R.; Orler, B.; Johnston, C.; Kim, Y.S. Nafion in dilute solvent systems: Dispersion or solution? ACS Macro Lett. 2012, 1, 1403–1407. [Google Scholar] [CrossRef] [PubMed]
- Collette, F.M.; Thominette, F.; Mendil-Jakani, H.; Gebel, G. Structure and transport properties of solution-cast Nafion® membranes subjected to hygrothermal aging. J. Membr. Sci. 2013, 435, 242–252. [Google Scholar] [CrossRef]
- Loppinet, B.; Gebel, G.; Williams, C.E. Small-angle scattering study of perfluorosulfonated ionomer solutions. J. Phys. Chem. B 1997, 101, 1884–1892. [Google Scholar] [CrossRef]
- Mabuchi, T.; Tokumasu, T. Relationship between Proton Transport and Morphology of Perfluorosulfonic Acid Membranes: A Reactive Molecular Dynamics Approach. J. Phys. Chem. B 2018, 122, 5922–5932. [Google Scholar] [CrossRef]
- Berlinger, S.A.; Dudenas, P.J.; Bird, A.; Chen, X.; Freychet, G.; Mccloskey, B.D.; Kusoglu, A.; Weber, A.Z. Impact of Dispersion Solvent on Ionomer Thin Films and Membranes. ACS Appl. Polym. Mater. 2020, 2, 5824–5834. [Google Scholar] [CrossRef]
- Kim, Y.S.; Welch, C.F.; Hjelm, R.P.; Mack, N.H.; Labouriau, A.; Orler, E.B. Origin of Toughness in Dispersion-Cast Nafion Membranes. Macromolecules 2015, 48, 2161–2172. [Google Scholar] [CrossRef] [Green Version]
- Crothers, A.R.; Radke, C.J.; Weber, A.Z. Impact of Nano- and Mesoscales on Macroscopic Cation Conductivity in Perfluorinated-Sulfonic-Acid Membranes. J. Phys. Chem. C 2017, 121, 28262–28274. [Google Scholar] [CrossRef]
- Lin, H.-L.; Yu, T.L.; Huang, C.-H.; Lin, T.-L. Morphology study of Nafion membranes prepared by solutions casting. J. Polym. Sci. Part B Polym. Phys. 2005, 43, 3044–3057. [Google Scholar] [CrossRef]
- Hsu, W.Y.; Gierke, T.D. Ion transport and clustering in nafion perfluorinated membranes. J. Membr. Sci. 1983, 13, 307–326. [Google Scholar] [CrossRef]
- Gebel, G. Structural evolution of water swollen perfluorosulfonated ionomers from dry membrane to solution. Polymer 2000, 41, 5829–5838. [Google Scholar] [CrossRef]
- Rebrov, A.V.; Ozerin, A.N.; Yakunin, A.N.; Dreiman, N.A.; Timofeyev, S.V.; Popkov, Y.M.; Bakeyev, N.F. Low angle X-ray study of the phase state of water in perfluorinated ion-exchange membranes. Polym. Sci. U.S.S.R. 1987, 29, 1597–1601. [Google Scholar] [CrossRef]
- Berlinger, S.A.; McCloskey, B.D.; Weber, A.Z. Inherent Acidity of Perfluorosulfonic Acid Ionomer Dispersions and Implications for Ink Aggregation. J. Phys. Chem. B 2018, 122, 7790–7796. [Google Scholar] [CrossRef] [Green Version]
- Tarokh, A.; Karan, K.; Ponnurangam, S. Atomistic MD Study of Nafion Dispersions: Role of Solvent and Counterion in the Aggregate Structure, Ionic Clustering, and Acid Dissociation. Macromolecules 2020, 53, 288–301. [Google Scholar] [CrossRef]
- Kuo, A.T.; Urata, S.; Nakabayashi, K.; Watabe, H.; Honmura, S. Coarse-Grained Molecular Dynamics Simulation of Perfluorosulfonic Acid Polymer in Water-Ethanol Mixtures. Macromolecules 2021, 54, 609–620. [Google Scholar] [CrossRef]
- Yang, D.; Zhu, S.; Yu, G.; Tang, H.; Yang, D.; Zhang, C.; Ming, P.; Li, B. Control of Cluster Structures in Catalyst Inks by a Dispersion Medium. ACS Omega 2021, 6, 32960–32969. [Google Scholar] [CrossRef] [PubMed]
- Kim, T.-H.; Yi, J.-Y.; Jung, C.-Y.; Jeong, E.; Yi, S.-C. Solvent effect on the Nafion agglomerate morphology in the catalyst layer of the proton exchange membrane fuel cells. Int. J. Hydrogen Energy 2017, 42, 478–485. [Google Scholar] [CrossRef]
- Gao, X.; Yamamoto, K.; Hirai, T.; Ohta, N.; Uchiyana, T.; Watanabe, T.; Imai, H.; Sugawara, S.; Shinohara, K.; Uchimoto, Y. Impact of the Composition of Alcohol/Water Dispersion on the Proton Transport and Morphology of Cast Perfluorinated Sulfonic Acid Ionomer Thin Films. ACS Omega 2021, 6, 14130–14137. [Google Scholar] [CrossRef]
- Majlan, E.H.; Rohendi, D.; Daud, W.R.W.; Husaini, T.; Haque, M.A. Electrode for proton exchange membrane fuel cells: A review. Renew. Sustain. Energy Rev. 2018, 89, 117–134. [Google Scholar] [CrossRef]
- Takahashi, I.; Kocha, S.S. Examination of the activity and durability of PEMFC catalysts in liquid electrolytes. J. Power Sources 2010, 195, 6312–6322. [Google Scholar] [CrossRef]
- Adamski, M.; Peressin, N.; Holdcroft, S.; Pollet, B.G. Does power ultrasound affect Nafion® dispersions? Ultrason. Sonochem. 2020, 60, 104758. [Google Scholar] [CrossRef] [PubMed]
- Ngo, T.T.; Yu, T.L.; Lin, H.L. Nafion-based membrane electrode assemblies prepared from catalyst inks containing alcohol/water solvent mixtures. J. Power Sources 2013, 238, 1–10. [Google Scholar] [CrossRef]
- Sharma, R.; Grahl-Madsen, L.; Andersen, S.M. Influence of dispersion media on Nafion® ionomer distribution in proton exchange membrane fuel cell catalyst carbon support. Mater. Chem. Phys. 2019, 226, 66–72. [Google Scholar] [CrossRef]
- Tesfaye, M.; Kushner, D.I.; Kusoglu, A. Interplay between Swelling Kinetics and Nanostructure in Perfluorosulfonic Acid Thin-Films: Role of Hygrothermal Aging. ACS Appl. Polym. Mater. 2019, 1, 631–635. [Google Scholar] [CrossRef] [Green Version]
- Gao, X.; Yamamoto, K.; Hirai, T.; Uchiyama, T.; Ohta, N.; Takao, N.; Matsumoto, M.; Imai, H.; Sugawara, S.; Shinohara, K.; et al. Morphology Changes in Perfluorosulfonated Ionomer from Thickness and Thermal Treatment Conditions. Langmuir 2020, 36, 3871–3878. [Google Scholar] [CrossRef]
- Berlinger, S.A.; McCloskey, B.D.; Weber, A.Z. Probing Ionomer Interactions with Electrocatalyst Particles in Solution. ACS Energy Lett. 2021, 6, 2275–2282. [Google Scholar] [CrossRef]
- Devivaraprasad, R.; Masuda, T. Solvent-Dependent Adsorption of Perfluorosulfonated Ionomers on a Pt(111) Surface Using Atomic Force Microscopy. Langmuir 2020, 36, 13793–13798. [Google Scholar] [CrossRef]
- Berezina, N.P.; Timofeev, S.V.; Kononenko, N.A. Effect of conditioning techniques of perfluorinated sulphocationic membranes on their hydrophylic and electrotransport properties. J. Membr. Sci. 2002, 209, 10. [Google Scholar] [CrossRef]
- Lebo, R.B. Properties of mixtures of isopropyl alcohol and water. J. Am. Chem. Soc. 1921, 43, 1005–1011. [Google Scholar] [CrossRef] [Green Version]
- Golubenko, D.V.; Van der Bruggen, B.; Yaroslavtsev, A.B. Ion exchange membranes based on radiation-induced grafted functionalized polystyrene for high-performance reverse electrodialysis. J. Power Sources 2021, 511, 230460. [Google Scholar] [CrossRef]
- Güler, E.; Elizen, R.; Vermaas, D.A.; Saakes, M.; Nijmeijer, K. Performance-determining membrane properties in reverse electrodialysis. J. Membr. Sci. 2013, 446, 266–276. [Google Scholar] [CrossRef]
- Collette, F.M.; Lorentz, C.; Gebel, G.; Thominette, F. Hygrothermal aging of Nafion®. J. Membr. Sci. 2009, 330, 21–29. [Google Scholar] [CrossRef]
- Chen, Q.; Schmidt-Rohr, K. 19F and 13C NMR Signal Assignment and Analysis in a Perfluorinated Ionomer (Nafion) by Two-Dimensional Solid-State NMR. Macromolecules 2004, 37, 5995–6003. [Google Scholar] [CrossRef]
- Safronova, E.; Golubenko, D.; Pourcelly, G.; Yaroslavtsev, A. Mechanical properties and influence of straining on ion conductivity of perfluorosulfonic acid Nafion®-type membranes depending on water uptake. J. Membr. Sci. 2015, 473, 218–225. [Google Scholar] [CrossRef]
- Stenina, I.A.; Yaroslavtsev, A.B. Ionic Mobility in Ion-Exchange Membranes. Membranes 2021, 11, 198. [Google Scholar] [CrossRef]
- Kreuer, K.-D. Proton Conduction in Fuel Cells. In Hydrogen-Transfer Reactions; Wiley-VCH Verlag GmbH & Co. KGaA: Weinheim, Germany, 2006; pp. 709–736. [Google Scholar] [CrossRef]
- Safronova, E.Y.; Voropaeva, D.V.; Lysova, A.A.; Korchagin, O.V.; Bogdanovskaya, V.A.; Yaroslavtsev, A.B. On the properties of Nafion membranes recast from dispersion in N-Methyl-2-Pyrrolidone. Polymers 2022, 14, 5275. [Google Scholar] [CrossRef]
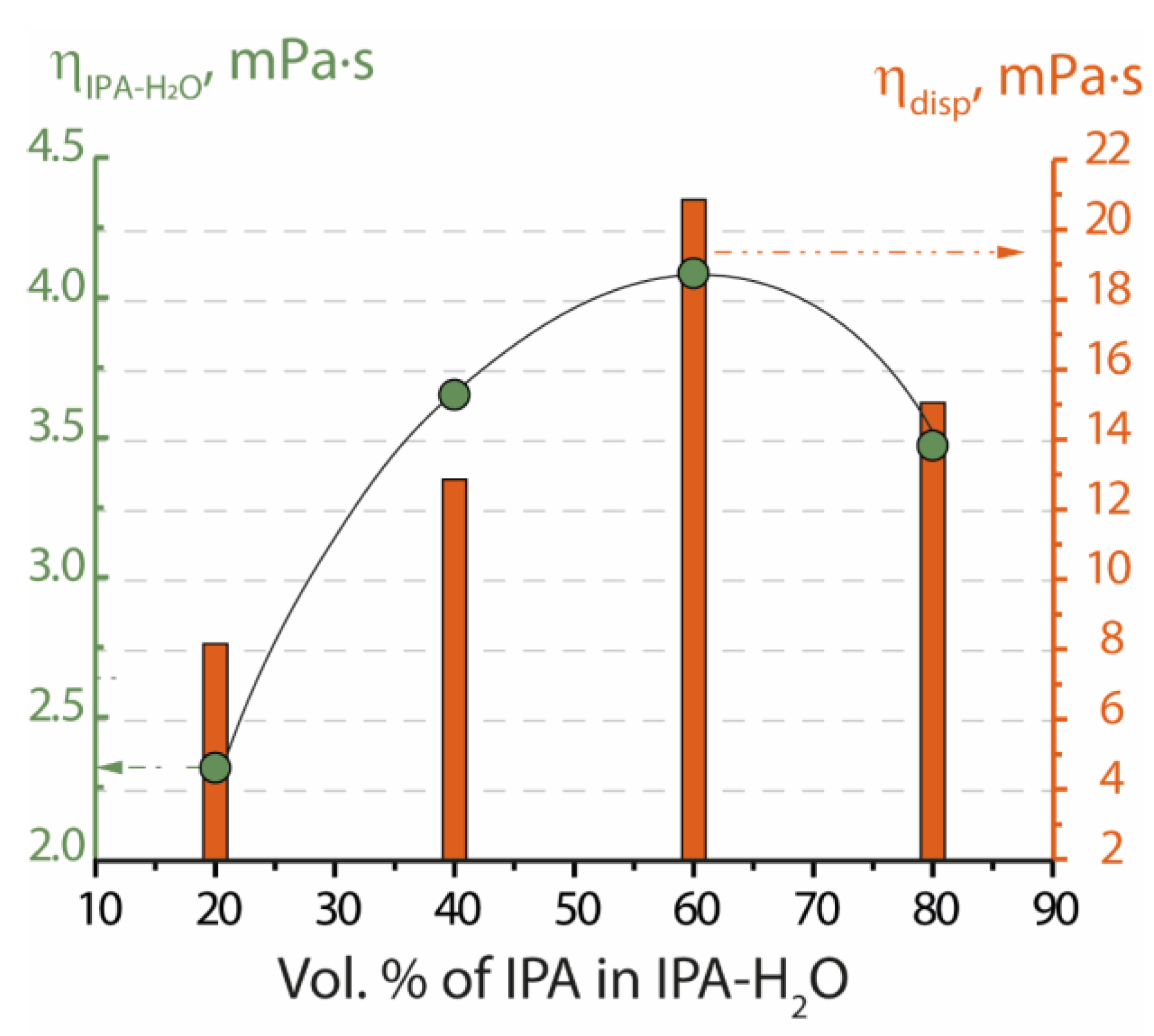


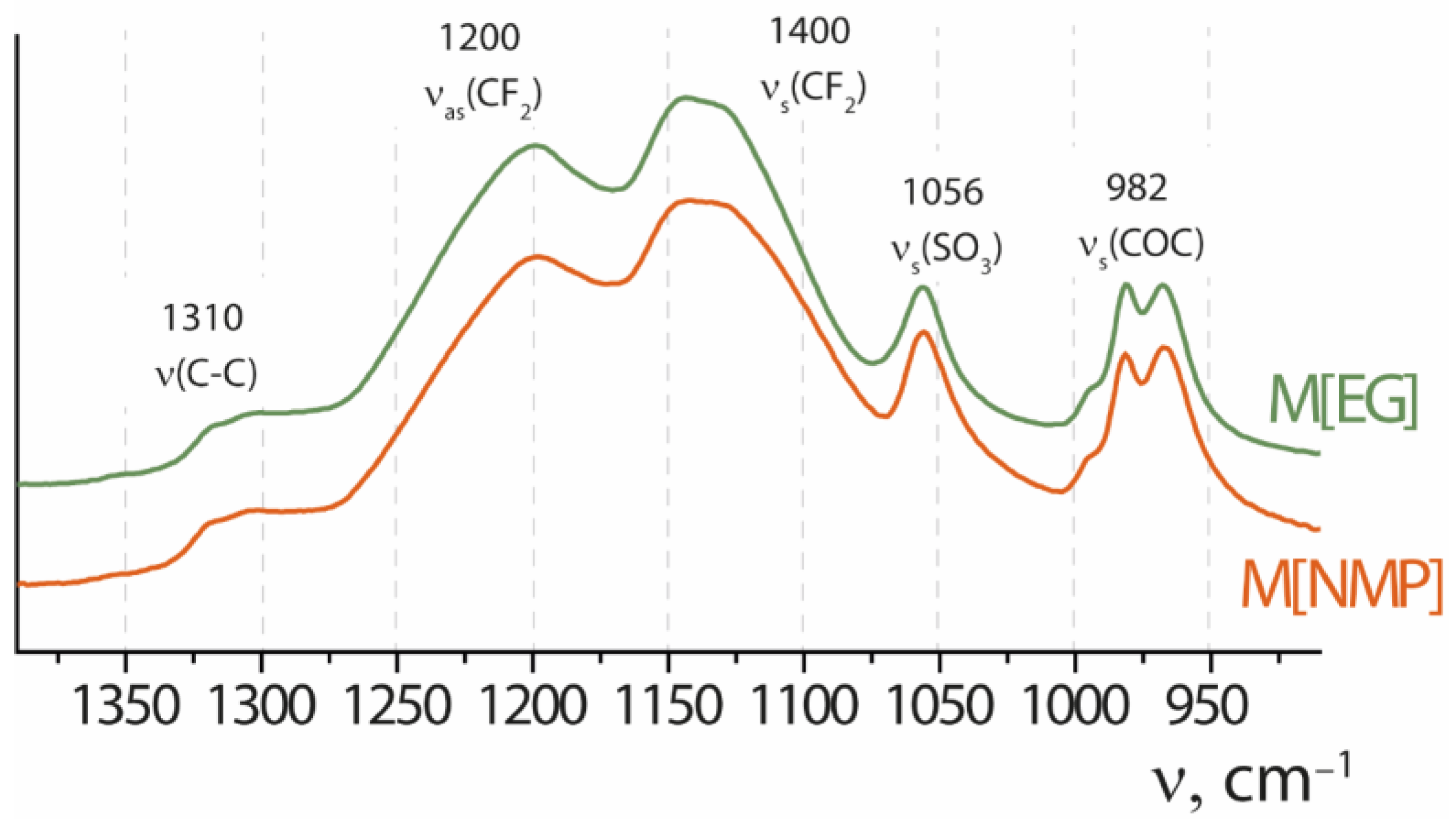
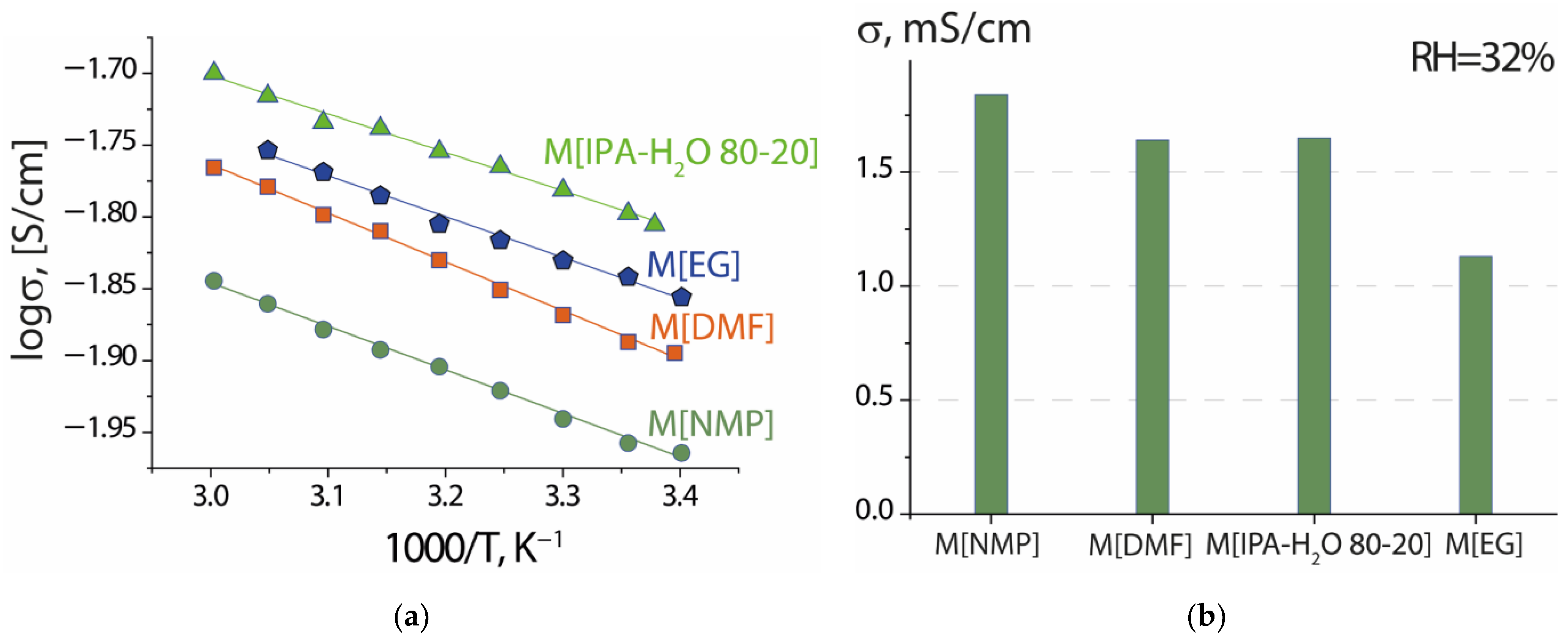
| Dispersing Liquid | Boiling Point, °C | ηdl, mPa·s | Counterion | Conditions for Obtaining the Dispersion ** | ηdisp., mPa·s | Membrane Formation Conditions | Membrane Description *** |
|---|---|---|---|---|---|---|---|
| IPA-H2O 100–0 | 82.2 * | 2.1 | H+, Li+ | Does not disperse when processed in the STC up to 230 °C 6 h | - | - | - |
| IPA-H2O 80–20 | 80.8 * | 3.5 | H+ | 80 °C in RBF with constant stirring for 2 h | 24.6 | 40 °C 3 h in air then 80 °C 3 h in vacuum oven | Mechanically stable, transparent membrane, l > 70 µm |
| Li+ | 15.1 | ||||||
| Na+ | 17.4 | ||||||
| IPA-H2O 60–40 | 81.0 * | 4.1 | Li+ | 80 °C in RBF with constant stirring for 2 h | 20.9 | 40 °C 3 h in air, then 80 °C 3 h in vacuum oven | Poor mechanical properties, it is possible to obtain samples of small size |
| IPA-H2O 40–60 | 81.4 * | 3.7 | Li+ | 150 °C in STC for 3 h | 12.9 | 40 °C 3 h in air, then 80 °C 3 h in vacuum oven | The membrane does not form |
| IPA-H2O 20–80 | 82.6 * | 2.3 | Li+ | 150 °C in STC for 3 h | 8.2 | 40 °C 3 h in air, then 80 °C 3 h in vacuum oven | The membrane forms |
| DMF | 153 | 0.8 | Li+ | 120 °C in STC for 2 h | 5.5 | 40 °C 3 h in air, 60 °C 3 h in air, 120 °C 6 h in vacuum oven | Mechanically stable, transparent membrane, l > 70 µm |
| Na+ | 120 °C in STC for 2 h | 5.7 | 40 °C 3 h in air, 60 °C 3 h in air, 120 °C 6 h in vacuum oven | Mechanically stable, transparent membrane, l > 70 µm | |||
| Ca2+ | 200 °C in STC for 12 h | 14.5 | - | - | |||
| EG | 195–198 | 18.3 | Li+ | 180 °C in STC for 6 h | 35.1 | 140 °C 6 h in a vacuum oven | Transparent brown membrane, l > 100 µm |
| NMP | 202 | 2.8 | Li+ | 100 °C in STC for 2 h | 11.6 | 60 °C 3 h in air, 120 °C 6 h in vacuum | Transparent membrane, l > 50 µm |
| Membrane | Young’s Modulus, MPa | Proportional Limit Stress, MPa | Tensile Strength, MPa | Tensile Strain, % |
|---|---|---|---|---|
| M[IPA-H2O 80–20] | 228 ± 20 | 6.7 ± 1.3 | 7.7 ± 1.4 | 11 ± 2 |
| M[NMP] | 268 ± 10 | 11.6 ± 0.4 | 19.8 ± 0.6 | 241 ± 11 |
| M[DMF] | 319 ± 14 | 12.9 ± 0.5 | 17.0 ± 0.6 | 102 ± 5 |
| Membrane | In Contact with Water, % | RH = 95%, % | RH = 32%, % |
|---|---|---|---|
| M[IPA-H2O 80–20] | 25.1 | 17.6 | 4.8 |
| M[DMF] | 25.2 | 19.5 | 5.6 |
| M[NMP] | 24.9 | 16.6 | 4.8 |
| M[EG] | 15.3 | 11.7 | 4.0 |
| Membrane | P·107, cm2/s | |
|---|---|---|
| M[IPA-H2O 80–20] | 83.1 ± 1.3 | 1.92 ± 0.04 |
| M[DMF] | 85.0 ± 1.2 | 1.26 ± 0.04 |
| M[NMP] | 85.3 ± 1.4 | 1.27 ± 0.04 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Safronova, E.Y.; Voropaeva, D.Y.; Safronov, D.V.; Stretton, N.; Parshina, A.V.; Yaroslavtsev, A.B. Correlation between Nafion Morphology in Various Dispersion Liquids and Properties of the Cast Membranes. Membranes 2023, 13, 13. https://doi.org/10.3390/membranes13010013
Safronova EY, Voropaeva DY, Safronov DV, Stretton N, Parshina AV, Yaroslavtsev AB. Correlation between Nafion Morphology in Various Dispersion Liquids and Properties of the Cast Membranes. Membranes. 2023; 13(1):13. https://doi.org/10.3390/membranes13010013
Chicago/Turabian StyleSafronova, Ekaterina Yu., Daria Yu. Voropaeva, Dmitry V. Safronov, Nastasia Stretton, Anna V. Parshina, and Andrey B. Yaroslavtsev. 2023. "Correlation between Nafion Morphology in Various Dispersion Liquids and Properties of the Cast Membranes" Membranes 13, no. 1: 13. https://doi.org/10.3390/membranes13010013






