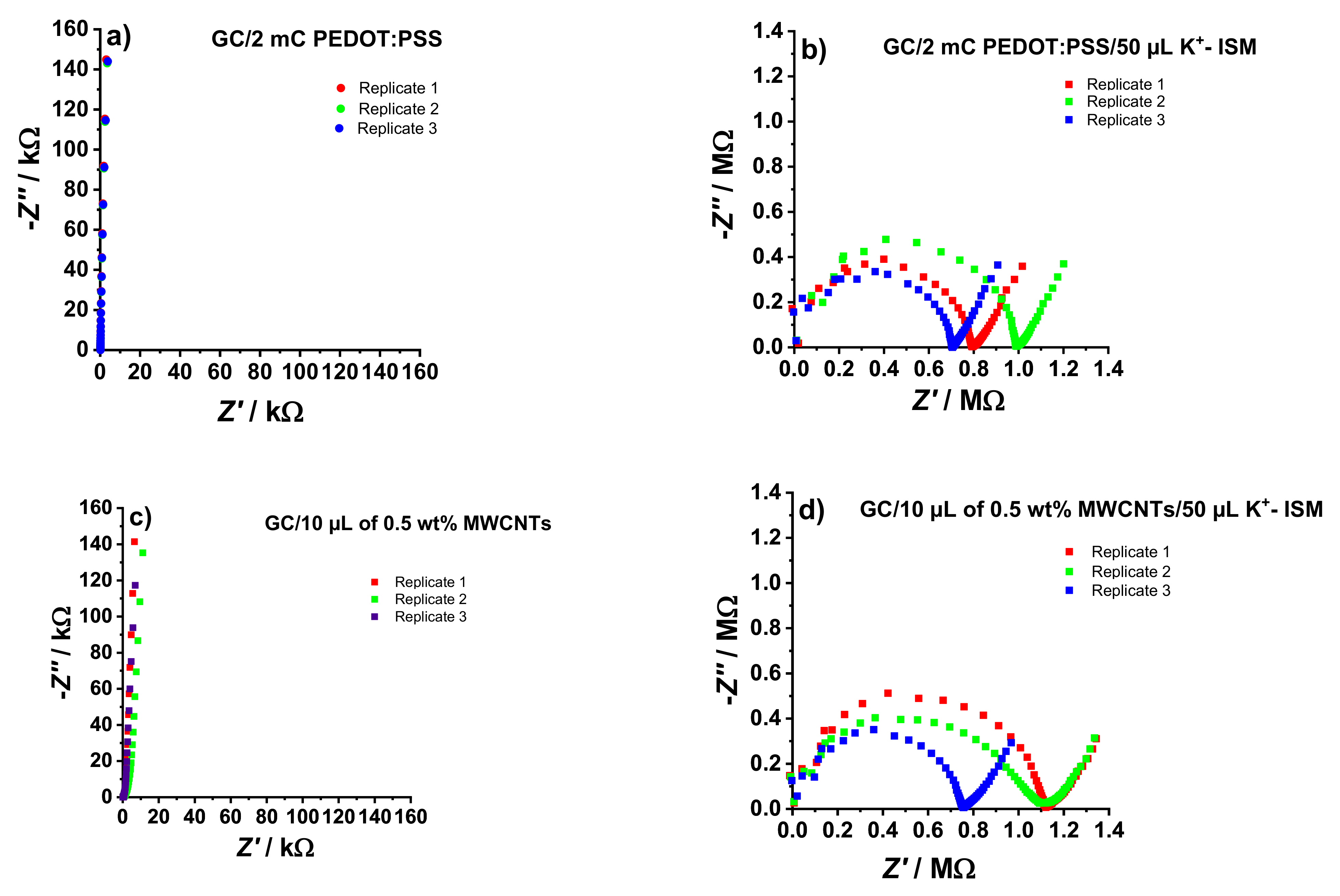
3.1. Electrochemical Impedance Spectroscopy Measurements
The impedance spectra of the K
+-SCISEs were measured before and after drop-casting the K
+- ISM. As shown in
Figure 1, a close to vertical line can be seen in the impedance spectra of both GC/PEDOT:PSS (
Figure 1a) and GC/MWCNTs (
Figure 1c). This is typical for PEDOT:PSS [
16] and MWCNTs [
17] electrodes indicating a capacitive mechanism related to the bulk redox capacitance and double-layer capacitance of the PEDOT:PSS and MWCNTs films, respectively.
While the 90° capacitive line in the GC/PEDOT:PSS electrodes (
Figure 1a) extends to the low-frequency region of the spectra, a slight deviation is noticeable for the GC/MWCNTs (
Figure 1c). This indicates that there is a small diffusion contribution in the transduction process of MWCNTs, which can be attributed to the inhomogeneous quality of the MWCNTs film due to manual deposition [
17]. Moreover, good repeatability was observed in the impedance spectra of the electrodes with galvanostatically polymerized PEDOT:PSS (
Figure 1a), compared to the manually added MWCNTs film (
Figure 1c).
The low-frequency capacitance (
CLF) of GC/PEDOT:PSS and GC/MWCNTs electrodes were estimated using Equation (1) below, where
f is the lowest frequency value used in the EIS measurement (0.01 Hz) and -
Z″ is the corresponding imaginary impedance.
The obtained CLF values for GC/PEDOT:PSS and GC/MWCNTs were 110.6 ± 0.7 µF and 122.0 ± 12.2 µF, respectively, confirming the excellent repeatability of the GC/PEDOT:PSS electrodes.
Compared to the spectra shown in
Figure 1a,c, which are dominated by a capacitive line, the spectra in
Figure 1b,d are dominated by a high-frequency semicircle. This is related to the geometric capacitance and the bulk resistance of the ISM (
RISM), determined by estimating the diameter of the semicircle [
11,
18]. The poor repeatability of the calculated
RISM for the prepared K
+-SCISEs is caused by the manual drop-casting of the K
+- ISM. The
RISM values from the EIS measurements are summarized in
Table 1.
A low-frequency line with an angle of ca. 45° (Warburg impedance) was seen in the impedance spectra of the electrodes after adding the K
+- ISM, i.e., GC/PEDOT:PSS/K
+- ISM (
Figure 1b) and GC/MWCNTs/K
+- ISM (
Figure 1d). This corresponds to the diffusion-limited ion-to-electron transduction mechanism in the electrodes. Estimating the low-frequency capacitance (
CLF) from −
Z″ at the lowest frequency by using Equation (1) gives the values summarized in
Table 1. The
CLF values are significantly lower after the PEDOT:PSS and MWCNTs are coated with the K
+- ISM, in good agreement with earlier observations [
19].
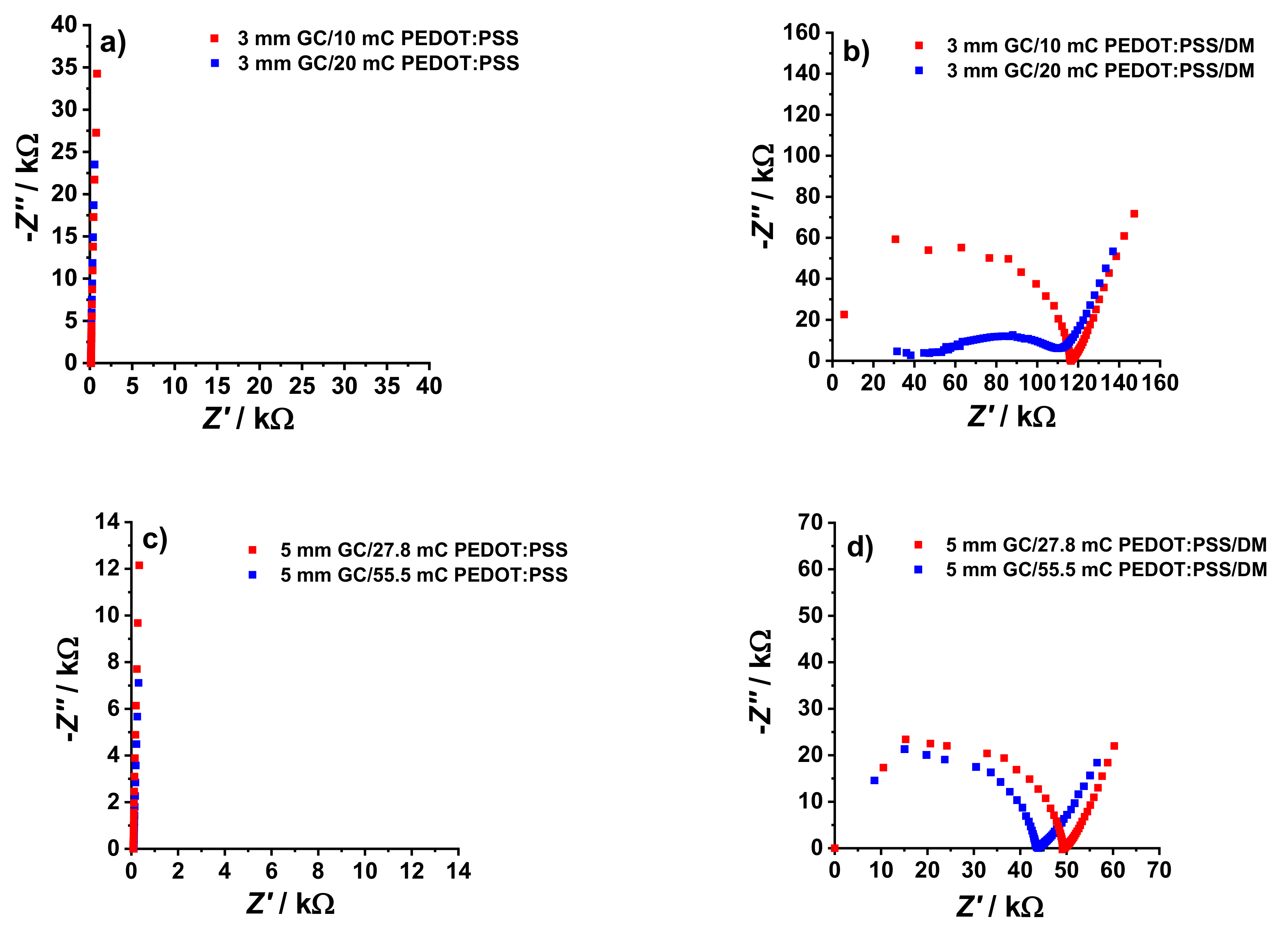
The impedance spectra of the GC/PEDOT:PSS electrodes before and after the deposition of the DM are shown in
Figure 2. The spectra for GC/PEDOT:PSS are dominated by the 90° capacitive line (
Figure 2a,c), while a high-frequency semicircle dominates after the deposition of the dummy membrane (
Figure 2b,d).
The
CLF and
RDM values shown in
Table 2 were estimated in the same way as for the K
+-SCISEs. The redox capacitance of the PEDOT:PSS film was varied in two ways: one is by increasing the film thickness and another is by using electrodes with larger
ØGC, and corresponding area (
AGC). Ideally, the redox capacitance of the film is directly proportional to
Qpolym [
16] which was generally observed in the results of the EIS experiments. As shown from the results in
Table 2, doubling the
Qpolym (and thus the PEDOT:PSS film thickness) at constant
ØGC and DM thickness resulted in a less than two times (ca. 1.4 times) increase in
CLF. Interestingly, ca. 2.8 times increase in electrode area (
AGC) while keeping the thickness of the PEDOT:PSS film and the dummy membrane constant, resulted in ca. 2.8 times increase in
CLF. This shows that there is a better correlation between
AGC and
CLF compared to the correlation between
Qpolym and
CLF.
It has been proven in previous studies [
18] that increasing the electrode area with constant thickness of the CP film and low membrane resistance increases the accessibility of the redox capacitance of the solid-contact layer, and that is why the ratio between the change in
CLF and in
AGC is close to one. A different behavior is seen when the thickness of the PEDOT:PSS film is increased at constant
AGC. The redox capacitance of thicker CP films becomes less accessible, so it is difficult to fully utilize the material as ion-to-electron transducer. This could be the reason why doubling the film thickness only resulted in only ca. 1.4 times the
CLF value. Lastly, since the results from the impedance spectra is dominated by the high-frequency semicircle, the
CLF did not have much effect on the time constant
τ compared to
RDM. The time constant,
τ, which is the product of
RDM and
CLF, falls within a relatively narrow range (
τ = 22–31 s) for all the GC/PEDOT:PSS/DM electrodes presented in
Table 2.
3.2. Potentiometric Measurements
The results from the calibrations of K
+- ISEs in KCl solution, with and without 10
−1 M NaCl as BGE, are shown in
Table 3. In both cases, the electrodes showed similar and close to Nernstian behavior. Except for the conventional K
+- ISEs, which are known to have stable standard potential (
E°), a large variation in the standard potential was observed for the other studied electrodes.
The use of a single electrochemical cell in the proposed novel experimental setup was made possible by the presence of a constant BGE in the sample solution. This eliminated the need for having separate sample and detection cells connected by a salt bridge, which is an advantage over previous studies employing the ISE as the RE [
13,
14,
15]. However, it is important to ensure that the analyte recognition takes place only at the K
+- ISE.
To highlight the difference in response of K
+- ISEs and GC/PEDOT:PSS/DM electrodes to changes in K
+ ion concentration, the potential of these electrodes was measured in the presence of 10
−1 M NaCl as BGE (
Figure 3a). As expected, the presence of a BGE does not affect the performance of the K
+- ISEs. On the other hand, the GC/PEDOT:PSS/DM electrodes was not sensitive to the change in K
+ ion concertation due to the high Na
+ concentration in the BGE.
Earlier studies revealed that conducting polymer films used as solid-contacts have shown sensitivity to gases such as O
2 and CO
2 [
20,
21].
Figure 3b shows results from the gas sensitivity measurement conducted in 10
−3 M KCl solution using GC/PEDOT:PSS electrodes with and without the DM. Introducing O
2 gas in the solution resulted in ca. 10 mV increase in potential of the GC/PEDOT:PSS electrodes. The presence of CO
2 gas in the solution has the most distinct effect on the potential readings (ca. 50 mV) due to the accompanying pH change [
20] as a consequence of the dissolution of CO
2 gas in the solution. As can be seen in
Figure 3b, adding the DM almost eliminated the sensitivity of the electrode to the presence of dissolved gases and pH changes in the sample.
3.3. Chronoamperometric and Chronocoulometric Measurements
In the original setup for constant-potential coulometry a SCISE was connected as the WE, a glassy carbon rod and a conventional Ag/AgCl/3M KCl were used as the counter and reference electrode, respectively [
10,
22].
Recently, a modified set-up was prosed using a two-compartment electrochemical cell. In one compartment containing a redox couple, the counter and working electrodes were placed. In the other compartment, a SCISE was placed and connected as reference electrode. The two compartments were connected by a salt-bridge [
14,
15] or Ag/AgCl wire [
13]. In this work, a single-cell approach is presented which is simple and easy to miniaturize. The ISE was connected as the reference electrode, the glassy carbon rod as the counter electrode, and GC/PEDOT:PSS/DM as the working electrode. The advantage of using the GC/PEDOT:PSS/DM is the easy control and adjustment of the electrode capacitance and resistance which influence the time constant and thus the response time of the electrode. In addition, the presence of the dummy membrane almost eliminates the sensitivity of the electrode to the presence of dissolved gases and pH changes in the sample.
The comparison between the chronoamperometric and chronocoulometric plots obtained using the original and the novel experimental setups is shown in
Figure 4. The GC/2 mC PEDOT:PSS/50 µL K
+- ISM electrode was connected as the WE with a double junction Ag/AgCl/3 M KCl/1 M LiOAc as the RE in the original setup (
Figure 4a). Alternatively, the K
+- ISE was connected as the RE in the new setup and a GC/10 mC PEDOT:PSS/25 µL DM electrode was connected as the WE (
Figure 4b). In both cases a GC rod was used as CE.
It should be noted that the direction of the current changes is opposite in these two cases because the ISE was connected as working electrode in the original setup (
Figure 4a) and as reference electrode in our new setup (
Figure 4b).
As shown in
Figure 4a,b, both setups showed good reversibility and signal repeatability after two consecutive addition/dilution steps. The new setup (
Figure 4b) shows an improvement (at least ca. five times increase) of the current and charge signals, without sacrificing the equilibration time, when compared with the original setup (
Figure 4a). For both setups there is a drift in the charge curve (0–5 min), which must be related to minor potential drifts in either WE or RE (or both) that results in a small bias current in the chronoamperometric curve and, hence, a drift in the charge.
Similar results were obtained using other ISEs, as reference electrode, such as the GC/10 µL of 0.5 wt% MWCNTs/50 µL K+- ISM and conventional K+- ISE (results not shown). The results are independent of the type of the ISE used as reference electrode as long as it has a Nernstian behavior.
3.3.1. Effect of Increasing the Thickness of the Dummy Membrane in the WEs
The effect of increasing the thickness of dummy membrane in the WE was studied using electrodes with similar
ØGC and PEDOT:PSS film loading but with different DM thicknesses. As shown in
Figure 5, electrodes with thicker DM resulted in current peaks with smaller amplitude and longer equilibration time greater than 15 min, as expected. Consequently, only one addition/dilution step was performed for the working electrodes with thicker dummy membranes.
It has been previously revealed through theoretical modelling that the peak current is inversely affected by the cell resistance [
23]. From the results of the EIS measurements, it was confirmed that the addition of the dummy membrane provided a certain resistance
RDM to the electrode connected as WE. Moreover,
RDM influences the time constant (
τ). In addition to the capacitance and resistance, mass transport across the membrane could also affect the response time, which explains why thinner membranes are typically preferable [
11,
23]
The results show that the cumulated charge Q is independent of the DM thickness as Q is the integration of the current with time [
11,
23]. Although reversible, it can be seen in
Figure 5b that the current did not return to the baseline and the residual current affected the magnitude of the reverse charge peak. The same results were obtained using other ISEs as reference electrode. For this reason, the rest of the experiments were performed only with the working electrodes having a thinner DM.
3.3.2. Effect of Increasing the Redox Capacitance of the PEDOT:PSS Film in the WEs
The influence of the redox capacitance of the PEDOT:PSS film in GC/PEDOT:PSS/DM electrode on the signal amplification and equilibration time was investigated. Working electrodes with equivalently thin dummy membrane but with different
ØGC and
Qpolym were examined in the addition/dilution experiments with Δ
CK+ = 5%. In general, increasing the
Qpolym (and consequently the redox capacitance of PEDOT:PSS film) at constant
ØGC resulted in signal amplification. From the EIS experiments, it was observed that the time constant τ is also influenced by the redox capacitance of the conducting polymer films although not as prominently as the effect of
RDM. Increasing the amount of PEDOT:PSS film means that more material participates in the redox reaction to achieve the desired ∆
E [
22,
23], resulting in longer equilibration time.
From the experimental results shown in
Figure 6, it is notable that increasing the thickness of the PEDOT:PSS layer in WEs with the same
ØGC and DM thickness did not influence the amplitude of the current peak. Instead, it resulted in peak broadening and longer equilibration time, which has been consistently observed in both theoretical and experimental studies [
11,
18,
23]. Increasing the
ØGC from 3 mm to 5 mm with constant PEDOT:PSS film thickness with
Qpolym 10 mC and 27.8 mC, respectively seemed to be a better approach. Increasing the redox capacitance accompanied by larger electrode area, compared to just making the CP film thicker, allowed for better access to the electroactive material. This resulted in higher and sharper current peaks without any significant increase in the response time (
Figure 6a).
Integration of the current with time resulted in a more pronounced relationship between the total cumulated charge
Q and the redox capacitance of the PEDOT:PSS film. At constant DM thickness, it is expected that at higher sensitivity is achieved with increased redox capacitance, accompanied by a longer equilibration time [
11]. As shown in
Figure 6b, the change in
Q values after each addition/dilution step increases with increasing the redox capacitance of the PEDOT:PSS film. Moreover, the electrode with lowest
Qpolym (3 mm GC/10 mC PEDOT:PSS/DM), showed the shortest response time, indicated by the appearance of a plateau-like region in the graph. Indeed, it is important to find a compromise between the amplitude of the current peak, response time and
Q to achieve useful results. The same results and interpretations were obtained for the setups with GC/10 µL 0.5 wt% MWCNTs/50 µL K
+- ISM and conventional K
+- ISE connected as the RE (results not shown).
3.3.3. Calibration Experiment
Based on the observed response times, electrodes with thin DM were used in this experiment. The electrochemical cell consisted of GC/2 mC PEDOT:PSS/50 µL K
+- ISM electrode connected as the RE, a GC rod connected as the CE. As the WEs, GC/PEDOT:PSS/DM electrodes with
ØGC = 3 and 5 mm, and various
Qpolym were used. The results obtained from the successive dilution performed from 10
−2 M to 10
−3.4 M KCl solutions with 10
−1 M NaCl as BGE with ∆ log
CK+ = 0.2 decade/ step are shown in
Figure 7.
The results show that increasing the redox capacitance at constant
ØGC and DM thickness resulted in broader peaks and longer response times. Because of the larger dilution steps, the measurement time was extended from 5 to 10 min for each concentration step.
Figure 7a shows that the current peaks are quite reproducible throughout all the dilution steps. The drifting current baseline, most pronounced in the large electrode with the thicker PEDOT:PSS film and highest redox capacitance, is due to the insufficient time for equilibration in between the dilution steps.
The coulometric response and the resulting calibration curves in
Figure 7b,c, respectively, show good linear relationship between the
Q and log
aK+ values. As shown in
Figure 7d, there is a linear relationship between
Qpolym and the slopes from the calibration curves in
Figure 7c. However, the curve is not perfectly linear, due to the difference in
ØGC of the electrodes. Doubling the PEDOT:PSS film thickness resulted in ca. 1.7 times increase in the slope value. Moreover, enlargement of
ØGC by ca. 2.8 times, at constant PEDOT:PSS film thickness, resulted in ca. 3.1 times increase in slope value. This supports the results from EIS measurements, that spreading out the conducting polymer film over a larger area gives better accessibility to its redox capacitance as compared to depositing a thicker film on the same electrode area.
Similar results were obtained using a GC/10 µL 0.5 wt% MWCNTs/50 µL K
+- ISM WE and a conventional K
+- ISE connected as the RE (results not shown).
Figure 8 shows that the obtained results are independent of the type of K
+- ISE, whether all-solid-state or conventional, used as RE. This means that the novel experimental setup can be assembled using different electrode combinations to suit the demands of an experiment.
3.3.4. Detection of Small Concentration Changes
The electrode responses in the new setup were further investigated using addition/dilution experiments with Δ
CK+ = 5%, 2.5%, and 1%, in the presence of 0.1 M NaCl as BGE.
Figure 9 shows results obtained using the 3 mm GC/10 mC PEDOT:PSS/25 µL DM electrode as the WE and GC/2 mC PEDOT:PSS/50 µL K
+- ISM as the RE. As expected, smaller changes in K
+ concentration resulted in smaller current peaks that decay faster as shown in
Figure 9a. Naturally, a larger concentration change results in a larger coulometric response, as shown in
Figure 9b.
As seen in
Figure 9a, a 1% change in concentration resulted in signals with magnitude that are comparable to the 5% change in concentration using the original setup (
Figure 4a). This is in good agreement with the fact that the novel experimental setup produced signals that are at least five times larger than the ones resulted from the original setup, as mentioned earlier in
Section 3.3.