The Influence of Cationic Nitrosyl Iron Complex with Penicillamine Ligands on Model Membranes, Membrane-Bound Enzymes and Lipid Peroxidation
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals and Materials
2.2. Investigated Compound
2.3. Liposomes Study
2.3.1. Vesicle Preparation
2.3.2. Dynamic Light Scattering
2.3.3. Fluorescence Measurements
2.4. Tissue Preparation
2.5. Luminol Chemiluminescence Assay
2.6. TBARS Assay
2.7. MAO Activity Assay
2.8. The Antiglycation Assay
2.9. Acute Toxicity Evaluation in Mice
2.10. Statistical Analysis
3. Results and Discussion
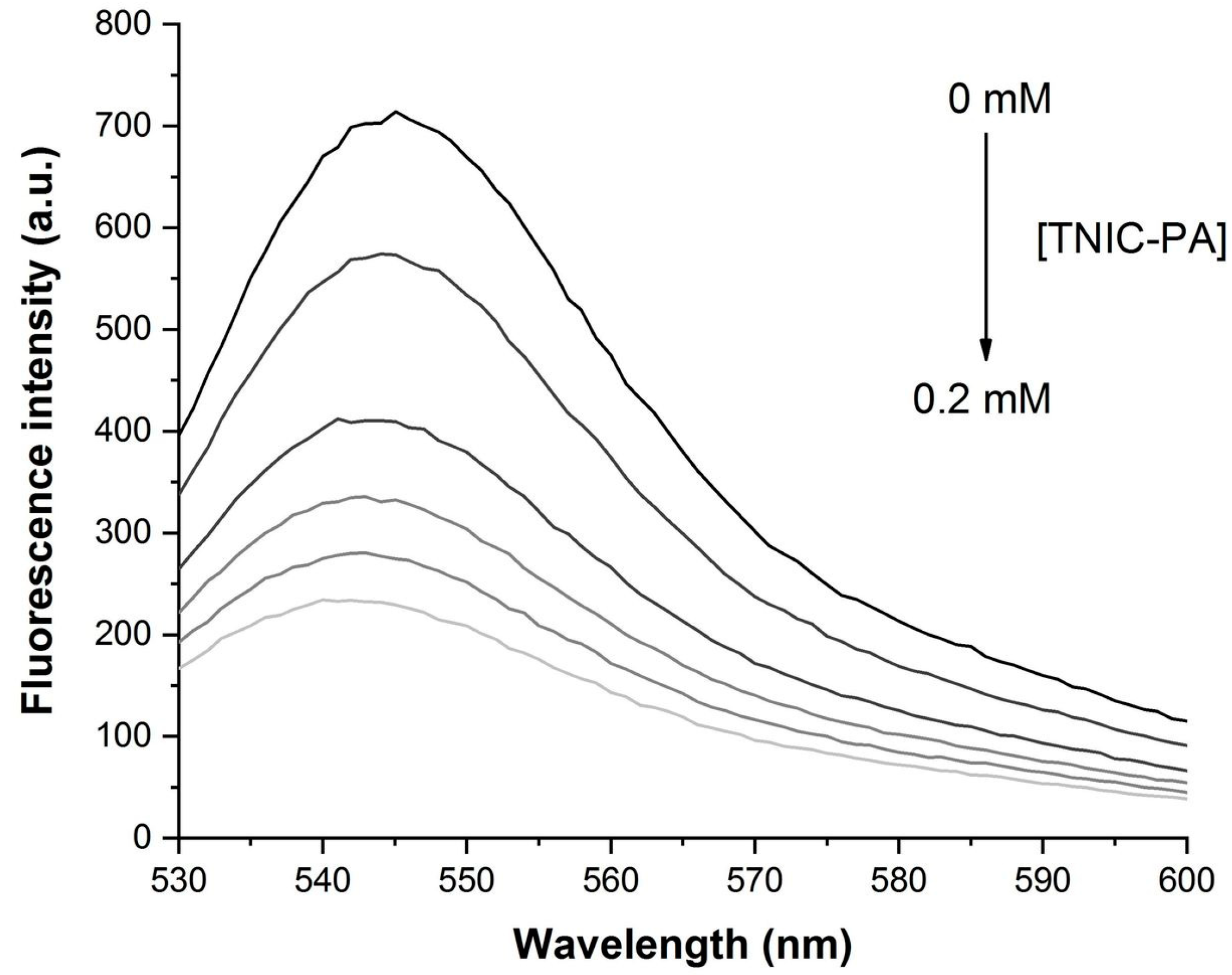
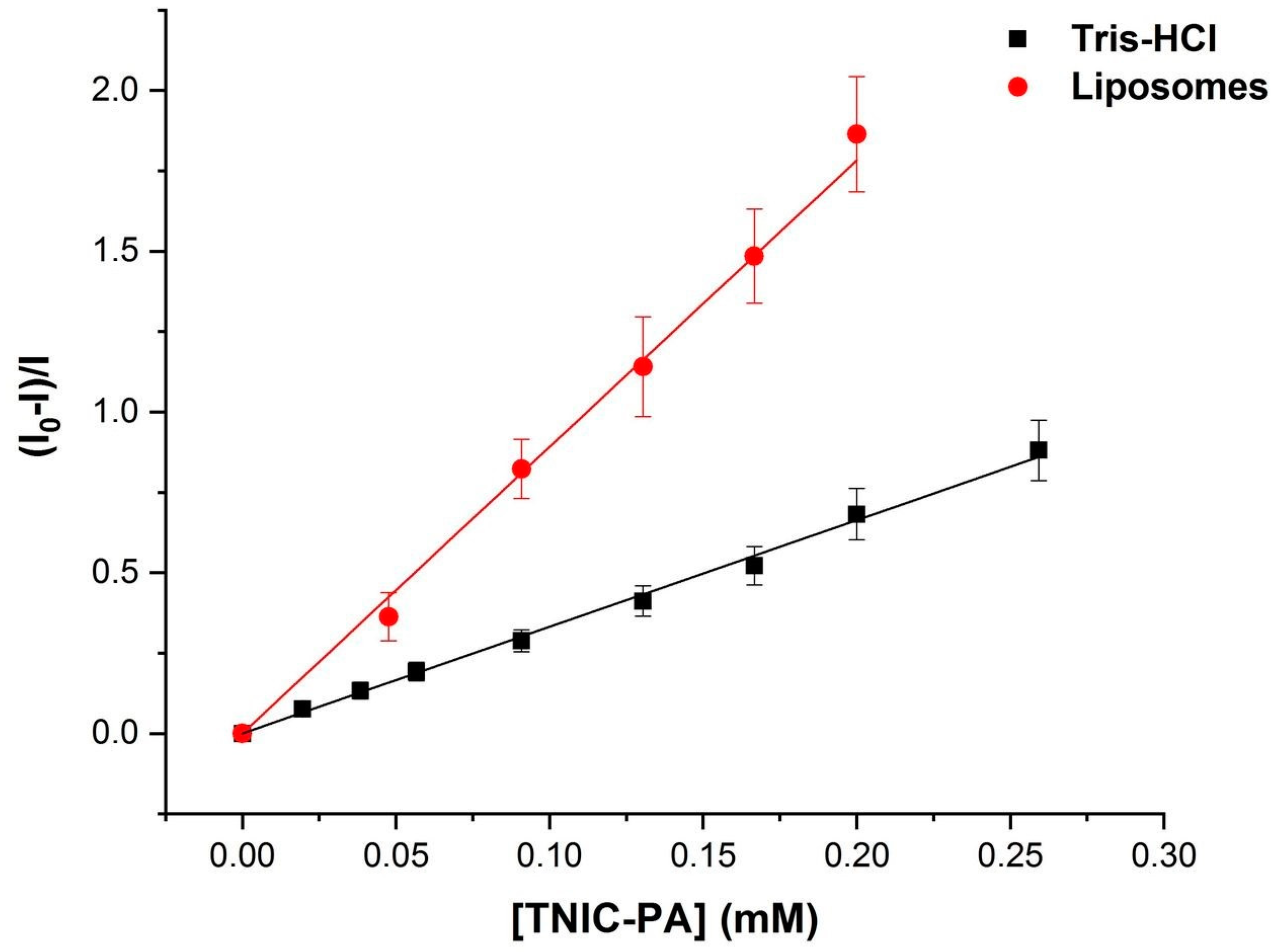
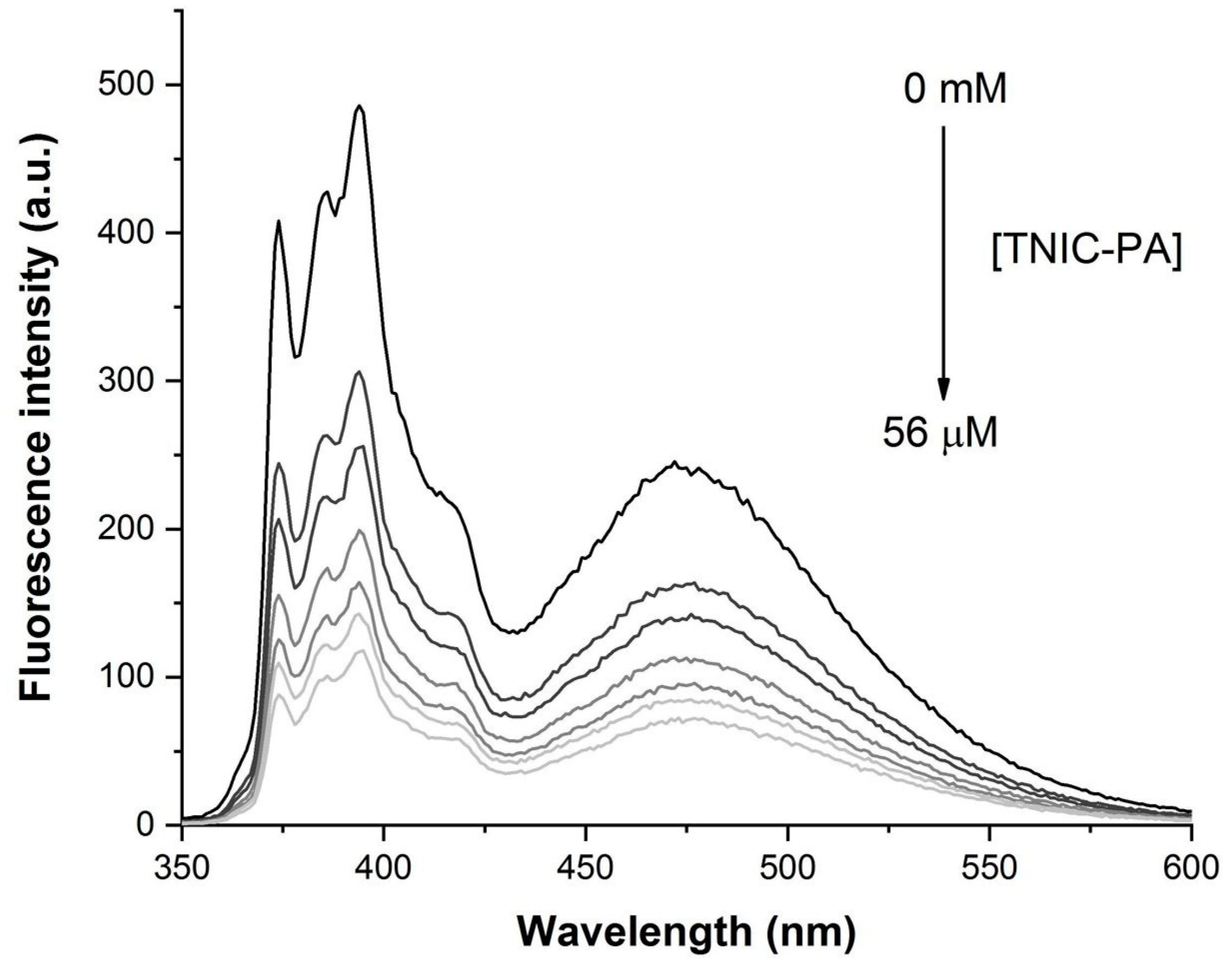
3.1. Fluorescent Probe Studies in Model Membrane
3.2. Antioxidant Activity
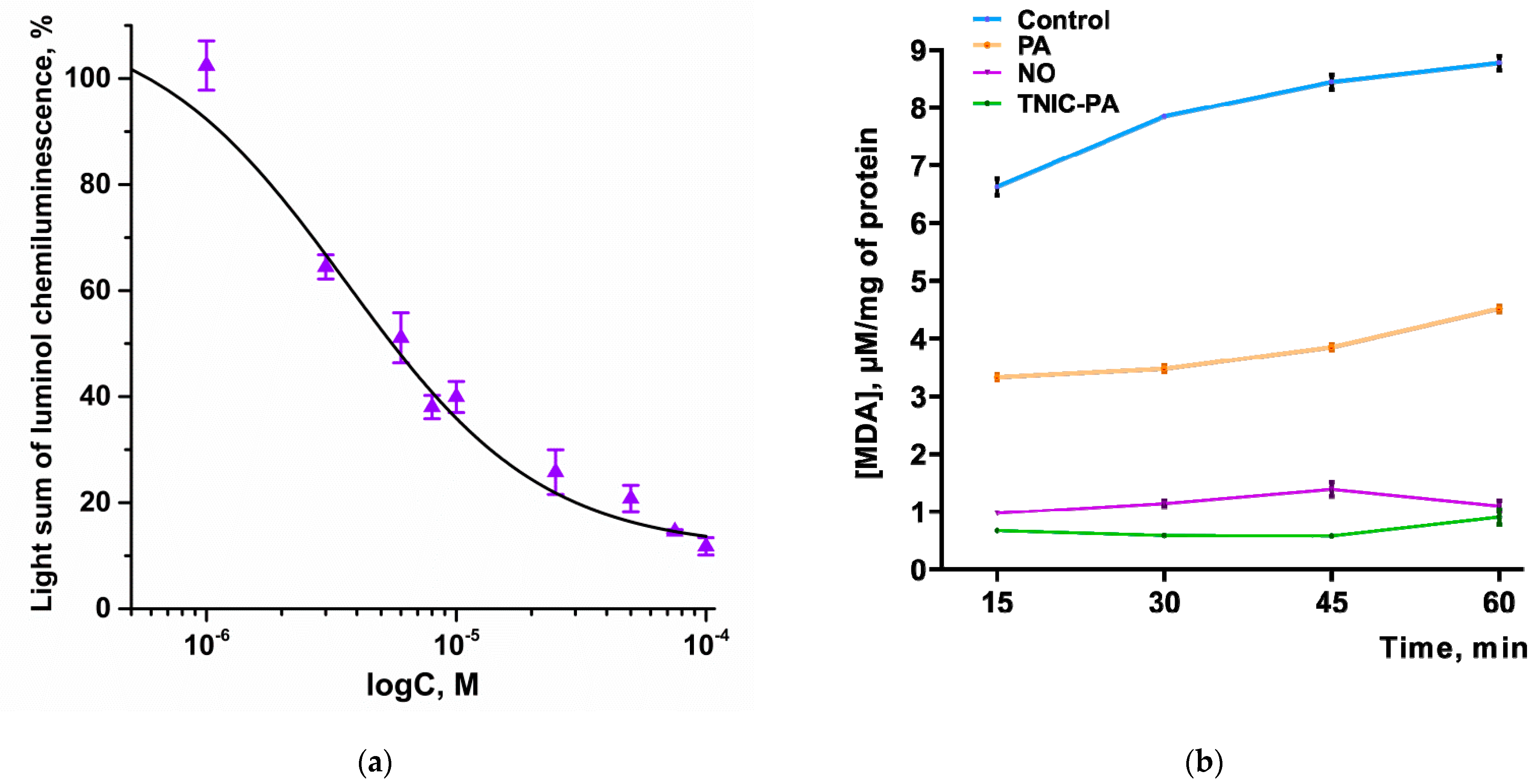
3.2.1. Radical Scavenging Capacity in Luminol Chemiluminescence Assay
3.2.2. TBARS Assay
3.3. Determination of the Inhibitory Effect of TNIC–PA on Membrane-Bound Enzymes MAO-A and B
3.4. Antiglycating Activity
3.5. Acute Toxicity Assessment
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Alberts, B.; Johnson, A.; Lewis, J.; Raff, M.; Roberts, K.; Walter, P. Molecular Biology of the Cell, 4th ed.; Garland Science: New York, NY, USA, 2002; ISBN 0-8153-3218-1. [Google Scholar]
- Cournia, Z.; Allen, T.W.; Andricioaei, I.; Antonny, B.; Baum, D.; Brannigan, G.; Buchete, N.-V.; Deckman, J.T.; Delemotte, L.; del Val, C.; et al. Membrane Protein Structure, Function, and Dynamics: A Perspective from Experiments and Theory. J. Membr. Biol. 2015, 248, 611–640. [Google Scholar] [CrossRef] [PubMed]
- Overington, J.P.; Al-Lazikani, B.; Hopkins, A.L. How Many Drug Targets Are There? Nat. Rev. Drug Discov. 2006, 5, 993–996. [Google Scholar] [CrossRef] [PubMed]
- Vanin, A.F. Dinitrosyl Iron Complexes with Thiolate Ligands: Physico-Chemistry, Biochemistry and Physiology. Nitric Oxide 2009, 21, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Sanina, N.A.; Shmatko, N.Y.; Korchagin, D.V.; Shilov, G.V.; Terent’ev, A.A.; Stupina, T.S.; Balakina, A.A.; Komleva, N.V.; Ovanesyan, N.S.; Kulikov, A.V.; et al. A New Member of the Cationic Dinitrosyl Iron Complexes Family Incorporating N-Ethylthiourea Is Effective against Human HeLa and MCF-7 Tumor Cell Lines. J. Coord. Chem. 2016, 69, 812–825. [Google Scholar] [CrossRef]
- Mumyatova, V.A.; Kozub, G.I.; Kondrat’eva, T.A.; Terent’ev, A.A.; Sanina, N.A. Antibacterial Activity of [1Fe-2S]- and [2Fe-2S]-Nitrosyl Complexes as Nitric Oxide Donors. Russ. Chem. Bull. 2019, 68, 1025–1030. [Google Scholar] [CrossRef]
- Gizatullin, A.R.; Akentieva, N.P.; Sanina, N.A.; Shmatko, N.Y.; Goryachev, N.S.; Shkondina, N.I.; Prichodchenko, T.R.; Zhelev, N.; Aldoshin, S.M. Effect of Dinitrosyl Iron Complexes (NO Donors) on the Metabolic Processes in Human Fibroblasts. Dokl. Biochem. Biophys. 2018, 483, 337–340. [Google Scholar] [CrossRef]
- Akentieva, N.P.; Sanina, N.A.; Prichodchenko, T.R.; Gizatullin, A.R.; Shkondina, N.I.; Shushanov, S.S.; Stupina, T.S.; Aldoshin, S.M. Anticancer Activity of Dinitrosyl Iron Complex (NO Donor) on the Multiple Myeloma Cells. Dokl. Biochem. Biophys. 2019, 486, 238–242. [Google Scholar] [CrossRef]
- Psikha, B.L.; Saratovskikh, E.A.; Sulimenkov, I.V.; Konyukhova, A.S.; Sanina, N.A. Reactions of Water-Soluble Binuclear Tetranitrosyl Iron Complexes of the μ-S Structural Type with Adenosine Triphosphoric Acid: Kinetics and Reaction Mechanism. Inorg. Chim. Acta 2022, 531, 120709. [Google Scholar] [CrossRef]
- Sanina, N.A.; Serebrjakova, L.I.; Shul’zhenko, V.S.; Pisarenko, O.I.; Rudneva, T.N.; Aldoshin, S.M. Use of Binuclear Sulphur-Nitrosyl Cationic Iron Complex as Vasodilatator. RU Patent 2460531 C2 2012, 5 April 2010. [Google Scholar]
- Pokidova, O.V.; Kormukhina, A.Y.; Kotelnikov, A.I.; Rudneva, T.N.; Lyssenko, K.A.; Sanina, N.A. Features of the Decomposition of Cationic Nitrosyl Iron Complexes with N-Ethylthiourea and Penicillamine Ligands in the Presence of Albumin. Inorganica Chim. Acta 2021, 524, 120453. [Google Scholar] [CrossRef]
- Faingold, I.I.; Kotelnikova, R.A.; Smolina, A.V.; Poletaeva, D.A.; Soldatova, Y.V.; Pokidova, O.V.; Sadkov, A.P.; Sanina, N.A.; Aldoshin, S.M. Antioxidant Activity of Tetranitrosyl Iron Complex with Thiosulfate Ligands and Its Effect on Catalytic Activity of Mitochondrial Enzymes In Vitro. Dokl. Biochem. Biophys. 2019, 488, 342–345. [Google Scholar] [CrossRef]
- Rodrigues, C.; Gameiro, P.; Reis, S.; Lima, J.L.F.; de Castro, B. Derivative Spectrophotometry as a Tool for the Determination of Drug Partition Coefficients in Water/Dimyristoyl-l-α-Phosphatidylglycerol (DMPG) Liposomes. Biophys. Chem. 2001, 94, 97–106. [Google Scholar] [CrossRef]
- Sies, H. Oxidative Stress. In Stress: Physiology, Biochemistry, and Pathology; Elsevier: Amsterdam, The Netherlands, 2019; pp. 153–163. [Google Scholar]
- Gianazza, E.; Brioschi, M.; Fernandez, A.M.; Banfi, C. Lipoxidation in Cardiovascular Diseases. Redox Biol. 2019, 23, 101119. [Google Scholar] [CrossRef] [PubMed]
- Ayala, A.; Muñoz, M.F.; Argüelles, S. Lipid Peroxidation: Production, Metabolism, and Signaling Mechanisms of Malondialdehyde and 4-Hydroxy-2-Nonenal. Oxid. Med. Cell. Longev. 2014, 2014, 360438. [Google Scholar] [CrossRef] [PubMed]
- Kaludercic, N.; Mialet-Perez, J.; Paolocci, N.; Parini, A.; Di Lisa, F. Monoamine Oxidases as Sources of Oxidants in the Heart. J. Mol. Cell. Cardiol. 2014, 73, 34–42. [Google Scholar] [CrossRef] [PubMed]
- Bergmann, O.; Bhardwaj, R.D.; Bernard, S.; Zdunek, S.; Barnabé-Heider, F.; Walsh, S.; Zupicich, J.; Alkass, K.; Buchholz, B.A.; Druid, H.; et al. Evidence for Cardiomyocyte Renewal in Humans. Science 2009, 324, 98–102. [Google Scholar] [CrossRef]
- Kennedy, A.L.; Lyons, T.J. Glycation, Oxidation, and Lipoxidation in the Development of Diabetic Complications. Metabolism 1997, 46, 14–21. [Google Scholar] [CrossRef]
- Sanina, N.A.; Syrtsova, L.A.; Shkondina, N.I.; Rudneva, T.N.; Malkova, E.S.; Bazanov, T.A.; Kotel’nikov, A.I.; Aldoshin, S.M. Reactions of Sulfur-Nitrosyl Iron Complexes of “G=2.03” Family with Hemoglobin (Hb): Kinetics of Hb–NO Formation in Aqueous Solutions. Nitric Oxide 2007, 16, 181–188. [Google Scholar] [CrossRef]
- Lowry, O.H.; Rosebrough, N.J.; Farr, A.L.; Randall, R.J. Protein Measurement with the Folin Phenol Reagent. J. Biol. Chem. 1951, 193, 265–275. [Google Scholar] [CrossRef]
- Di Meo, S.; Venditti, P.; Piro, M.C.; De Leo, T. Enhanced Luminescence Study of Liver Homogenate Response to Oxidative Stress. Arch. Physiol. Biochem. 1995, 103, 187–195. [Google Scholar] [CrossRef]
- Ohkawa, H.; Ohishi, N.; Yagi, K. Assay for Lipid Peroxides in Animal Tissues by Thiobarbituric Acid Reaction. Anal. Biochem. 1979, 95, 351–358. [Google Scholar] [CrossRef]
- Veryovkina, I.V.; Abdel Samed, M.M.; Gorkin, V.Z. Mitochondrial Monoamine Oxidase of Rat Liver: Reversible Qualitative Alterations in Catalytic Properties. Biochim. Biophys. Acta-Enzymol. 1972, 258, 56–70. [Google Scholar] [CrossRef]
- Kotel’nikova, R.A.; Grigoriev, V.V.; Smolina, A.V.; Faingold, I.I.; Mishchenko, D.V.; Van’kin, G.I.; Zamoiskii, V.L.; Poletaeva, D.A.; Markova, N.A.; Romanova, V.S.; et al. Design of a Hybrid Nanostructure Based on Fullerene C60 and Biologically Active Substance for Modeling Physiological Properties of Compounds. Russ. Chem. Bull. 2014, 63, 2375–2382. [Google Scholar] [CrossRef]
- Chen, Y.F.; Roan, H.Y.; Lii, C.K.; Huang, Y.C.; Wang, T.-S. Relationship between Antioxidant and Antiglycation Ability of Saponins, Polyphenols, and Polysaccharides in Chinese Herbal Medicines Used to Treat Diabetes. J. Med. Plants Res. 2011, 5, 2322–2331. [Google Scholar]
- Matsuura, N.; Aradate, T.; Sasaki, C.; Kojima, H.; Ohara, M.; Hasegawa, J.; Ubukata, M. Screening System for the Maillard Reaction Inhibitor from Natural Product Extracts. J. Health Sci. 2002, 48, 520–526. [Google Scholar] [CrossRef]
- Séro, L.; Sanguinet, L.; Blanchard, P.; Dang, B.; Morel, S.; Richomme, P.; Séraphin, D.; Derbré, S. Tuning a 96-Well Microtiter Plate Fluorescence-Based Assay to Identify AGE Inhibitors in Crude Plant Extracts. Molecules 2013, 18, 14320–14339. [Google Scholar] [CrossRef] [PubMed]
- Mironov, A.N.; Bunyatyan, N.D.; Vasiliev, A.N.; Verstakova, O.L.; Zhuravleva, M.V.; Lepakhin, V.K.; Korobov, N.V.; Merkulov, V.A.; Orekhov, S.N.; Sakaeva, I.V.; et al. Rukovodstvo Po Provedeniyu Doklinicheskikh Issledovaniy Lekarstvennykh Sredstv.(Guidelines for Conducting Preclinical Studies of Drugs, Part 1); Grif & K.: Moscow, Russia, 2012. (In Russian) [Google Scholar]
- Kachel, K.; Asuncion-Punzalan, E.; London, E. The Location of Fluorescence Probes with Charged Groups in Model Membranes. Biochim. Biophys. Acta-Biomembr. 1998, 1374, 63–76. [Google Scholar] [CrossRef]
- Galla, H.-J.; Sackmann, E. Lateral Diffusion in the Hydrophobic Region of Membranes: Use of Pyrene Excimers as Optical Probes. Biochim. Biophys. Acta-Biomembr. 1974, 339, 103–115. [Google Scholar] [CrossRef]
- Brocklehurst, J.R.; Freedman, R.B.; Hancock, D.J.; Radda, G.K. Membrane Studies with Polarity-Dependant and Excimer-Forming Fluorescent Probes. Biochem. J. 1970, 116, 721–731. [Google Scholar] [CrossRef]
- Mclean, L.; Hagaman, K. Effect of Lipid Physical State on the Rate of Peroxidation of Liposomes. Free Radic. Biol. Med. 1992, 12, 113–119. [Google Scholar] [CrossRef]
- Hinzmann, J.S.; McKenna, R.L.; Pierson, T.S.; Han, F.; Kézdy, F.J.; Epps, D.E. Interaction of Antioxidants with Depth-Dependent Fluorescence Quenchers and Energy Transfer Probes in Lipid Bilayers. Chem. Phys. Lipids 1992, 62, 123–138. [Google Scholar] [CrossRef]
- Kavanagh, R.J.; Kam, P.C.A. Lazaroids: Efficacy and Mechanism of Action of the 21-Aminosteroids in Neuroprotection. Br. J. Anaesth. 2001, 86, 110–119. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.-A.; Tsujita, M.; Hayashi, M.; Yokoyama, S. Probucol Inactivates ABCA1 in the Plasma Membrane with Respect to Its Mediation of Apolipoprotein Binding and High Density Lipoprotein Assembly and to Its Proteolytic Degradation. J. Biol. Chem. 2004, 279, 30168–30174. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, Y.J.; Tsuchiya, M.; Wassall, S.R.; Choo, Y.M.; Govil, G.; Kagan, V.E.; Packer, L. Structural and Dynamic Membrane Properties of .Alpha.-Tocopherol and .Alpha.-Tocotrienol: Implication to the Molecular Mechanism of Their Antioxidant Potency. Biochemistry 1993, 32, 10692–10699. [Google Scholar] [CrossRef] [PubMed]
- Valko, M.; Leibfritz, D.; Moncol, J.; Cronin, M.T.D.; Mazur, M.; Telser, J. Free Radicals and Antioxidants in Normal Physiological Functions and Human Disease. Int. J. Biochem. Cell Biol. 2007, 39, 44–84. [Google Scholar] [CrossRef]
- Frecnh, R.C.; Herbert, K.E.; Scott, D.L.; Perrett, D. Effect of Penicillamine on Luminol-Dependent Chemiluminescence in Human Leucocytes. Biochem. Soc. Trans. 1988, 16, 891. [Google Scholar] [CrossRef]
- Lambert, A.J.; Brand, M.D. Reactive Oxygen Species Production by Mitochondria. Mitochondrial DNA 2009, 554, 165–181. [Google Scholar]
- Maurel, A.; Hernandez, C.; Kunduzova, O.; Bompart, G.; Cambon, C.; Parini, A.; Francés, B. Age-Dependent Increase in Hydrogen Peroxide Production by Cardiac Monoamine Oxidase A in Rats. Am. J. Physiol. Heart Circ. Physiol. 2003, 284, H1460–H1467. [Google Scholar] [CrossRef]
- Umbarkar, P.; Singh, S.; Arkat, S.; Bodhankar, S.L.; Lohidasan, S.; Sitasawad, S.L. Monoamine Oxidase-A Is an Important Source of Oxidative Stress and Promotes Cardiac Dysfunction, Apoptosis, and Fibrosis in Diabetic Cardiomyopathy. Free Radic. Biol. Med. 2015, 87, 263–273. [Google Scholar] [CrossRef]
- Villeneuve, C.; Guilbeau-Frugier, C.; Sicard, P.; Lairez, O.; Ordener, C.; Duparc, T.; De Paulis, D.; Couderc, B.; Spreux-Varoquaux, O.; Tortosa, F.; et al. P53-PGC-1α Pathway Mediates Oxidative Mitochondrial Damage and Cardiomyocyte Necrosis Induced by Monoamine Oxidase-A Upregulation: Role in Chronic Left Ventricular Dysfunction in Mice. Antioxid. Redox Signal. 2013, 18, 5–18. [Google Scholar] [CrossRef]
- Maggiorani, D.; Manzella, N.; Edmondson, D.E.; Mattevi, A.; Parini, A.; Binda, C.; Mialet-Perez, J. Monoamine Oxidases, Oxidative Stress, and Altered Mitochondrial Dynamics in Cardiac Ageing. Oxid. Med. Cell. Longev. 2017, 2017, 3017947. [Google Scholar] [CrossRef]
- Watanabe, K.; Minamiura, N.; Yasunobu, K.T. Thiols Liberate Covalently Bonded Flavin from Monoamine Oxidase. Biochem. Biophys. Res. Commun. 1980, 94, 579–585. [Google Scholar] [CrossRef]
- Azouzi, S.; Santuz, H.; Morandat, S.; Pereira, C.; Côté, F.; Hermine, O.; El Kirat, K.; Colin, Y.; Le Van Kim, C.; Etchebest, C.; et al. Antioxidant and Membrane Binding Properties of Serotonin Protect Lipids from Oxidation. Biophys. J. 2017, 112, 1863–1873. [Google Scholar] [CrossRef] [PubMed]
- Jagdale, A.D.; Bavkar, L.N.; More, T.A.; Joglekar, M.M.; Arvindekar, A.U. Strong Inhibition of the Polyol Pathway Diverts Glucose Flux to Protein Glycation Leading to Rapid Establishment of Secondary Complications in Diabetes Mellitus. J. Diabetes Complicat. 2016, 30, 398–405. [Google Scholar] [CrossRef] [PubMed]
- Ramkissoon, J.S.; Mahomoodally, M.F.; Subratty, A.H.; Ahmed, N. Inhibition of Glucose- and Fructose-Mediated Protein Glycation by Infusions and Ethanolic Extracts of Ten Culinary Herbs and Spices. Asian Pac. J. Trop. Biomed. 2016, 6, 492–500. [Google Scholar] [CrossRef]
- Grzegorczyk-Karolak, I.; Gołąb, K.; Gburek, J.; Wysokińska, H.; Matkowski, A. Inhibition of Advanced Glycation End-Product Formation and Antioxidant Activity by Extracts and Polyphenols from Scutellaria Alpina L. and S. Altissima L. Molecules 2016, 21, 739. [Google Scholar] [CrossRef]
- Moldogazieva, N.T.; Mokhosoev, I.M.; Mel’nikova, T.I.; Porozov, Y.B.; Terentiev, A.A. Oxidative Stress and Advanced Lipoxidation and Glycation End Products (ALEs and AGEs) in Aging and Age-Related Diseases. Oxid. Med. Cell. Longev. 2019, 2019, 3085756. [Google Scholar] [CrossRef]
- Awasthi, S.; Gayathiri, S.K.; Ramya, R.; Duraichelvan, R.; Dhason, A.; Saraswathi, N.T. Advanced Glycation-Modified Human Serum Albumin Evokes Alterations in Membrane and Eryptosis in Erythrocytes. Appl. Biochem. Biotechnol. 2015, 177, 1013–1024. [Google Scholar] [CrossRef]
- Akhter, F.; Chen, D.; Akhter, A.; Sosunov, A.A.; Chen, A.; McKhann, G.M.; Yan, S.F.; Yan, S.S. High Dietary Advanced Glycation End Products Impair Mitochondrial and Cognitive Function. J. Alzheimer’s Dis. 2020, 76, 165–178. [Google Scholar] [CrossRef]
- Almaida-Pagan, P.F.; Ortega-Sabater, C.; Lucas-Sanchez, A.; Gonzalez-Silvera, D.; Martinez-Nicolas, A.; Rol de Lama, M.A.; Mendiola, P.; de Costa, J. Age-Related Changes in Mitochondrial Membrane Composition of Nothobranchius Furzeri.: Comparison with a Longer-Living Nothobranchius Species. Biogerontology 2019, 20, 83–92. [Google Scholar] [CrossRef]
- Hummel, S.G.; Fischer, A.J.; Martin, S.M.; Schafer, F.Q.; Buettner, G.R. Nitric Oxide as a Cellular Antioxidant: A Little Goes a Long Way. Free Radic. Biol. Med. 2006, 40, 501–506. [Google Scholar] [CrossRef]
- Rubbo, H.; Odonnell, V. Nitric Oxide, Peroxynitrite and Lipoxygenase in Atherogenesis: Mechanistic Insights. Toxicology 2005, 208, 305–317. [Google Scholar] [CrossRef] [PubMed]
- Shumaev, K.B.; Kosmachevskaya, O.V.; Grachev, D.I.; Timoshin, A.A.; Topunov, A.F.; Lankin, V.Z.; Ruuge, E.K. A Possible Mechanism of the Antioxidant Action of Dinitrosyl Iron Complexes. Biochem. (Mosc.) Suppl. Ser. B Biomed. Chem. 2021, 15, 313–319. [Google Scholar] [CrossRef]
- Elosta, A.; Ghous, T.; Ahmed, N. Natural Products as Anti-Glycation Agents: Possible Therapeutic Potential for Diabetic Complications. Curr. Diabetes Rev. 2012, 8, 92–108. [Google Scholar] [CrossRef] [PubMed]





Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Poletaeva, D.A.; Soldatova, Y.V.; Smolina, A.V.; Savushkin, M.A.; Klimanova, E.N.; Sanina, N.A.; Faingold, I.I. The Influence of Cationic Nitrosyl Iron Complex with Penicillamine Ligands on Model Membranes, Membrane-Bound Enzymes and Lipid Peroxidation. Membranes 2022, 12, 1088. https://doi.org/10.3390/membranes12111088
Poletaeva DA, Soldatova YV, Smolina AV, Savushkin MA, Klimanova EN, Sanina NA, Faingold II. The Influence of Cationic Nitrosyl Iron Complex with Penicillamine Ligands on Model Membranes, Membrane-Bound Enzymes and Lipid Peroxidation. Membranes. 2022; 12(11):1088. https://doi.org/10.3390/membranes12111088
Chicago/Turabian StylePoletaeva, Darya A., Yuliya V. Soldatova, Anastasiya V. Smolina, Maxim A. Savushkin, Elena N. Klimanova, Nataliya A. Sanina, and Irina I. Faingold. 2022. "The Influence of Cationic Nitrosyl Iron Complex with Penicillamine Ligands on Model Membranes, Membrane-Bound Enzymes and Lipid Peroxidation" Membranes 12, no. 11: 1088. https://doi.org/10.3390/membranes12111088
APA StylePoletaeva, D. A., Soldatova, Y. V., Smolina, A. V., Savushkin, M. A., Klimanova, E. N., Sanina, N. A., & Faingold, I. I. (2022). The Influence of Cationic Nitrosyl Iron Complex with Penicillamine Ligands on Model Membranes, Membrane-Bound Enzymes and Lipid Peroxidation. Membranes, 12(11), 1088. https://doi.org/10.3390/membranes12111088




