On the Coupling between Mechanical Properties and Electrostatics in Biological Membranes
Abstract
| Contents | |
| 1 Introduction | 2 |
| 2 Surface Charge Density, Surface and Zeta Potentials | 4 |
| 3 pH Effects | 5 |
| 4 Effects of Ions Different from Protons | 6 |
| 4.1 Metal Cations | 6 |
| 4.2 Cationic Peptides | 9 |
| 5 Potentials across Membranes | 11 |
| 5.1 Electromechanical Coupling | 11 |
| 5.2 External Electric Fields and Electroporation | 13 |
| 5.3 Nerve Impulse Propagation | 15 |
| 6 Summary and Future Perspectives | 17 |
| References | 18 |
1. Introduction
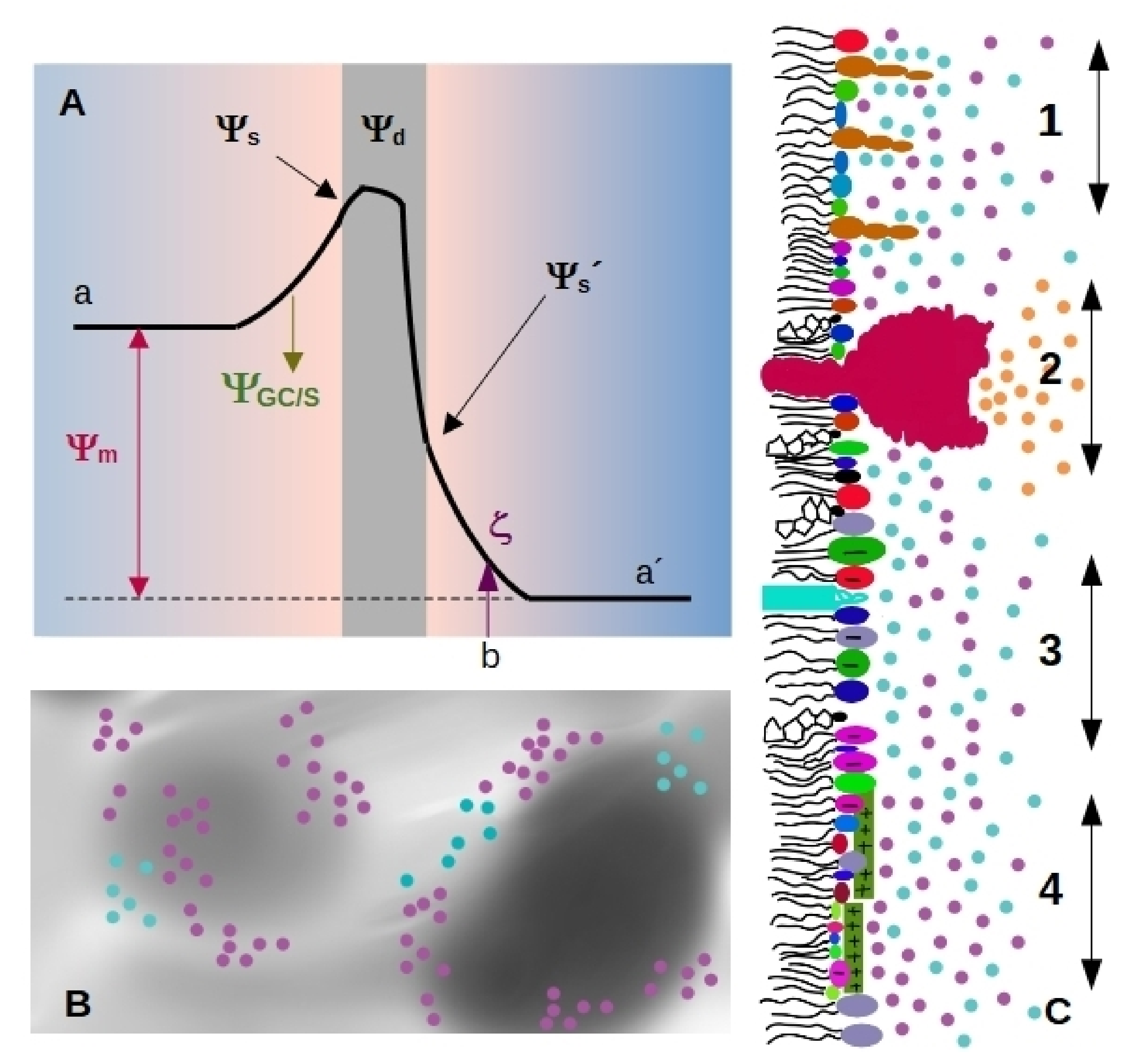
- Membranes separate the interior from the exterior media, having different Volta potential. The difference between these values is called membrane, transmembrane or diffusion potential [18], see Figure 1A (we will use the term membrane potential for throughout the review). The absolute value of this potential difference is about – mV and varies in distances of tens of nm, thus generating high electric fields (of the order of – V/m) through the bilayer.
- The presence of highly ordered molecules with a charge distribution characterized by a dipole or higher order multipoles inside the membrane, as well as a lower dielectric constant in the membrane interior compared to the surface, gives rise to a non-zero potential inside the membrane called dipole potential [19]. This potential is positive in the center of the bilayer (Figure 1A), and leads to very high electric field inside the bilayer (of the order of Vm = 10 V/m).
- Charged moieties on the membrane surface generate a potential difference between the membrane surface and the solution called surface potential (see Figure 1A). These charges in turn interact with small ions or charged molecules from the solution, resulting in an ion cloud around the membrane. The membrane surface charge, together with the ionic composition of the solution modulates the potential drop due to the ion cloud, and it can be modelled using Gouy-Chapman approach (), or with Stern model () in the presence of specific ion-membrane interactions [18], see Figure 1A. Gouy-Chapman model predicts an exponential decay on the potential as we move away from the charged surface, with a characteristic distance (Debye lenght). Stern model predicts a linear drop in the region of adsorbed ions, and a behavior according to Gouy-Chapman model at larger distances. Both models are broadly used in membrane biophysics, working remarkably well despite having coarse approximations. With respecto to this, a good correspondence was found between ion distribution close to a phosphatidylglycerol membrane predicted by Gouy-Chapman model and obtained using molecular dynamics simulations [20].
2. Surface Charge Density, Surface and Zeta Potentials
3. pH Effects
4. Effects of Ions Different from Protons
4.1. Metal Cations
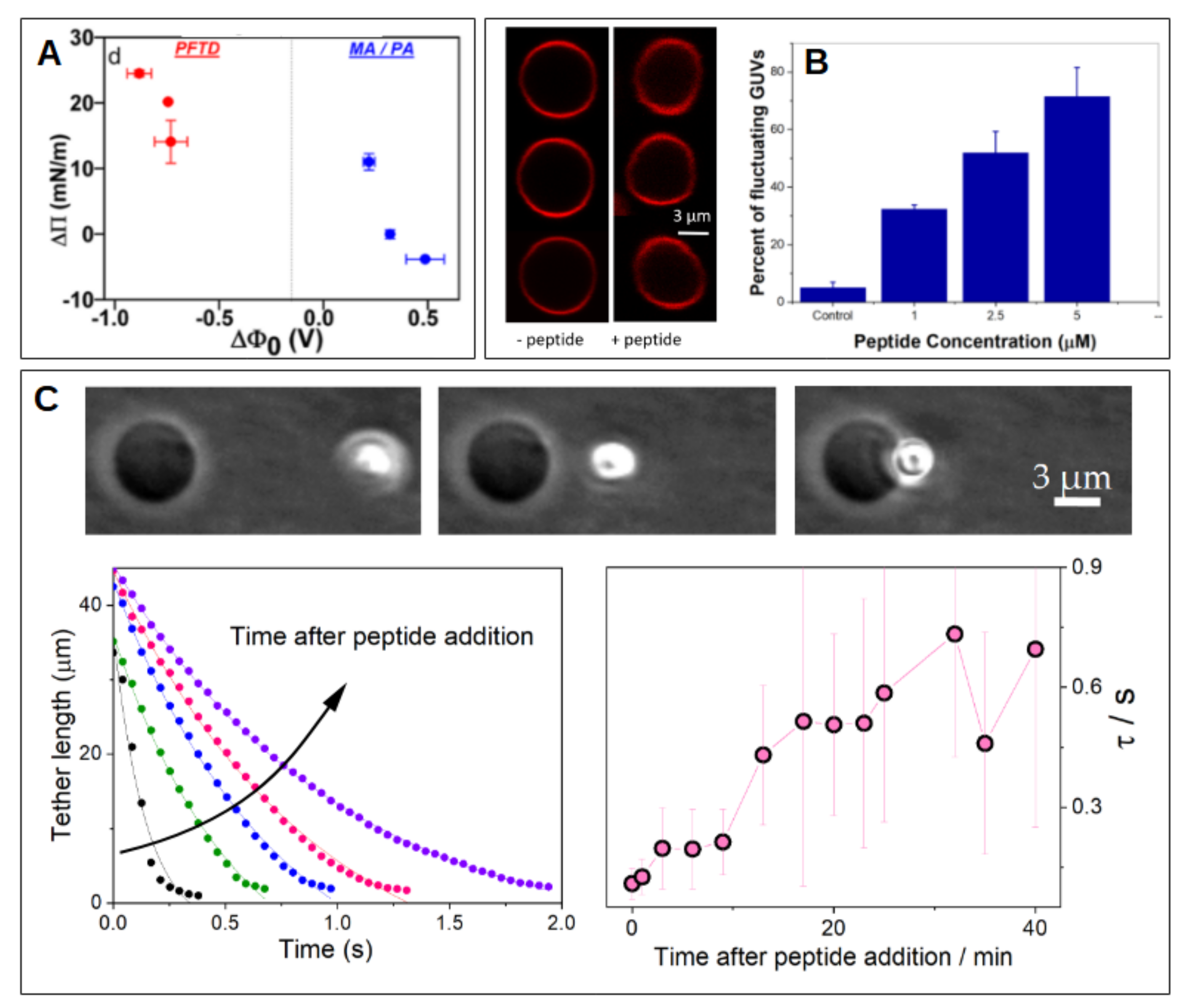
4.2. Cationic Peptides
5. Potentials across Membranes
5.1. Electromechanical Coupling
5.2. External Electric Fields and Electroporation
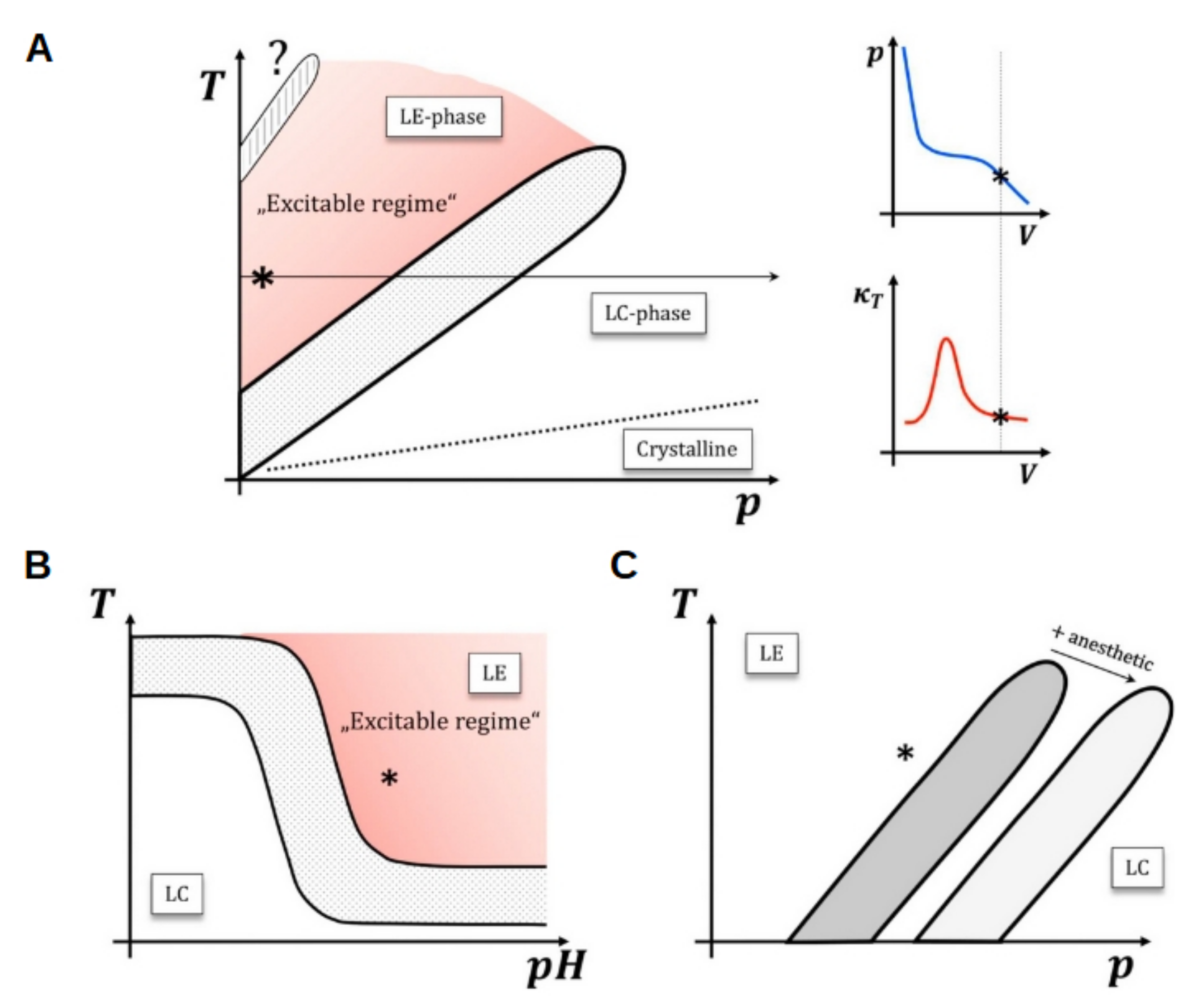
5.3. Nerve Impulse Propagation
6. Summary and Future Perspectives
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Israelachvili, J.N. Intermolecular and Surface Forces; Academic Press: Cambridge, MA, USA, 2011. [Google Scholar]
- Montich, G.G.; Bustos, M.M.; Maggio, B.; Cumar, F.A. Micropolarity of interfaces containing anionic and neutral glycosphingolipids as sensed by Merocyanine 540. Chem. Phys. Lipids 1985, 38, 319–326. [Google Scholar] [CrossRef]
- Cooke, R.; Kuntz, I. The properties of water in biological systems. Annu. Rev. Biophys. Bioeng. 1974, 3, 95–126. [Google Scholar] [CrossRef]
- Mild, K.H.; Løvtrup, S. Movement and structure of water in animal cells. Ideas and experiments. Biochim. Et Biophys. Acta Rev. Biomembr. 1985, 822, 155–167. [Google Scholar] [CrossRef]
- Bagatolli, L.A.; Stock, R.P.; Olsen, L.F. Coupled response of membrane hydration with oscillating metabolism in live cells: An alternative way to modulate structural aspects of biological membranes? Biomolecules 2019, 9, 687. [Google Scholar] [CrossRef]
- Timoshkin, I.; MacGregor, S.; Fouracre, R.; Given, M.; Anderson, J. Forces acting on biological cells in external electrical fields. In Proceedings of the 2006 IEEE Conference on Electrical Insulation and Dielectric Phenomena, Kansas City, MO, USA, 15–18 October 2006; pp. 676–679. [Google Scholar]
- Elia, S.; Lamberti, P.; Tucci, V. A finite element model for the axon of nervous cells. In Proceedings of the COMSOL Conference, Milan, Italy, 14–16 October 2009; pp. 14–16. [Google Scholar]
- Singh, S.; Krishnaswamy, J.A.; Melnik, R. Biological cells and coupled electro-mechanical effects: The role of organelles, microtubules, and nonlocal contributions. J. Mech. Behav. Biomed. Mater. 2020, 110, 103859. [Google Scholar] [CrossRef] [PubMed]
- Honrado, C.; Ciuffreda, L.; Spencer, D.; Ranford-Cartwright, L.; Morgan, H. Dielectric characterization of Plasmodium falciparum-infected red blood cells using microfluidic impedance cytometry. J. R. Soc. Interface 2018, 15, 20180416. [Google Scholar] [CrossRef]
- Gascoyne, P.R.; Noshari, J.; Becker, F.F.; Pethig, R. Use of dielectrophoretic collection spectra for characterizing differences between normal and cancerous cells. IEEE Trans. Ind. Appl. 1994, 30, 829–834. [Google Scholar] [CrossRef]
- Asami, K.; Hanai, T.; Koizumi, N. Dielectric properties of yeast cells. J. Membr. Biol. 1976, 28, 169–180. [Google Scholar] [CrossRef] [PubMed]
- Raicu, V.; Raicu, G.; Turcu, G. Dielectric properties of yeast cells as simulated by the two-shell model. Biochim. Et Biophys. Acta Bioenerg. 1996, 1274, 143–148. [Google Scholar] [CrossRef]
- Haandbæk, N.; Bürgel, S.C.; Heer, F.; Hierlemann, A. Characterization of subcellular morphology of single yeast cells using high frequency microfluidic impedance cytometer. Lab A Chip 2014, 14, 369–377. [Google Scholar] [CrossRef]
- Gagnon, Z.; Gordon, J.; Sengupta, S.; Chang, H.C. Bovine red blood cell starvation age discrimination through a glutaraldehyde-amplified dielectrophoretic approach with buffer selection and membrane cross-linking. Electrophoresis 2008, 29, 2272–2279. [Google Scholar] [CrossRef]
- Huang, Y.; Wang, X.B.; Gascoyne, P.R.; Becker, F.F. Membrane dielectric responses of human T-lymphocytes following mitogenic stimulation. Biochim. Et Biophys. Acta Biomembr. 1999, 1417, 51–62. [Google Scholar] [CrossRef]
- Hughes, M.P.; Morgan, H.; Rixon, F.J.; Burt, J.P.; Pethig, R. Manipulation of herpes simplex virus type 1 by dielectrophoresis. Biochim. Et Biophys. Acta Gen. Subj. 1998, 1425, 119–126. [Google Scholar] [CrossRef]
- Huang, Y.; Wang, X.B.; Holzel, R.; Becker, F.; Gascoyne, P. Electrorotational studies of the cytoplasmic dielectric properties of Friend murine erythroleukaemia cells. Phys. Med. Biol. 1995, 40, 1789. [Google Scholar] [CrossRef] [PubMed]
- Kleijin, J.; Van Leeuwen, H. Electrostatic and Electrodynamic Properties of Biological Interphases; Marcel Dekker: New York, NY, USA, 2000. [Google Scholar]
- Clarke, R.J. The dipole potential of phospholipid membranes and methods for its detection. Adv. Colloid Interface Sci. 2001, 89, 263–281. [Google Scholar] [CrossRef]
- Yi, M.; Nymeyer, H.; Zhou, H.X. Test of the Gouy-Chapman theory for a charged lipid membrane against explicit-solvent molecular dynamics simulations. Phys. Rev. Lett. 2008, 101, 038103. [Google Scholar] [CrossRef] [PubMed]
- Wilke, N. Monomolecular Films of Surfactants with Phase-Coexistence: Distribution of the Phases and Their Consequences. In Comprehensive Guide for Nanocoatings Technology. Vol 2: Characterization and Reliability; NOVA: New York, NY, USA, 2015; pp. 139–158. [Google Scholar]
- Mercado, F.V.; Maggio, B.; Wilke, N. Phase diagram of mixed monolayers of stearic acid and dimyristoylphosphatidylcholine. Effect of the acid ionization. Chem. Phys. Lipids 2011, 164, 386–392. [Google Scholar] [CrossRef]
- Träuble, H. Membrane electrostatics. In Structure of Biological Membranes; Springer: Berlin/Heidelberg, Germany, 1977; pp. 509–550. [Google Scholar]
- Heimburg, T. Thermal Biophysics of Membranes; John Wiley & Sons: Hoboken, NJ, USA, 2008. [Google Scholar]
- Eisenberg, M.; Gresalfi, T.; Riccio, T.; McLaughlin, S. Adsorption of monovalent cations to bilayer membranes containing negative phospholipids. Biochemistry 1979, 18, 5213–5223. [Google Scholar] [CrossRef]
- Egorova, E.; Dukhin, A.; Svetlova, I. Some problems of zeta potential determination in electrophoretic measurements on lipid membranes. Biochim. Et Biophys. Acta Biomembr. 1992, 1104, 102–110. [Google Scholar] [CrossRef]
- McDaniel, R.V.; McLaughlin, A.; Winiski, A.P.; Eisenberg, M.; McLaughlin, S. Bilayer membranes containing the ganglioside GM1: Models for electrostatic potentials adjacent to biological membranes. Biochemistry 1984, 23, 4618–4624. [Google Scholar] [CrossRef]
- Simon, S.; McIntosh, T. Magnitude of the solvation pressure depends on dipole potential. Proc. Natl. Acad. Sci. USA 1989, 86, 9263–9267. [Google Scholar] [CrossRef] [PubMed]
- Li, A.J.; Nussinov, R. A set of van der Waals and coulombic radii of protein atoms for molecular and solvent-accessible surface calculation, packing evaluation, and docking. Proteins Struct. Funct. Bioinform. 1998, 32, 111–127. [Google Scholar] [CrossRef]
- Parsons, D.F.; Ninham, B.W. Ab initio molar volumes and Gaussian radii. J. Phys. Chem. A 2009, 113, 1141–1150. [Google Scholar] [CrossRef]
- Woodle, M.; Collins, L.; Sponsler, E.; Kossovsky, N.; Papahadjopoulos, D.; Martin, F. Sterically stabilized liposomes. Reduction in electrophoretic mobility but not electrostatic surface potential. Biophys. J. 1992, 61, 902–910. [Google Scholar] [CrossRef]
- Freire, J.M.; Domingues, M.M.; Matos, J.; Melo, M.N.; Veiga, A.S.; Santos, N.C.; Castanho, M.A. Using zeta-potential measurements to quantify peptide partition to lipid membranes. Eur. Biophys. J. 2011, 40, 481–487. [Google Scholar] [CrossRef] [PubMed]
- Tocanne, J.F.; Teissié, J. Ionization of phospholipids and phospholipid-supported interfacial lateral diffusion of protons in membrane model systems. Biochim. Et Biophys. Acta Rev. Biomembr. 1990, 1031, 111–142. [Google Scholar] [CrossRef]
- Tsui, F.C.; Ojcius, D.M.; Hubbell, W.L. The intrinsic pKa values for phosphatidylserine and phosphatidylethanolamine in phosphatidylcholine host bilayers. Biophys. J. 1986, 49, 459–468. [Google Scholar] [CrossRef]
- Egorova, E.M. Determination of the apparent pK values of ionizable groups in highly charged lipid membranes. Colloids Surfaces A Physicochem. Eng. Asp. 1996, 110, 47–53. [Google Scholar] [CrossRef]
- Egorova, E.M. Dissociation constants of lipid ionizable groups I. Corrected values for two anionic lipids. Colloids Surfaces A Physicochem. Eng. Asp. 1998, 131, 7–18. [Google Scholar] [CrossRef]
- Egorova, E. Dissociation constants of lipid ionizable groups II. Changes in surface pK at low ionic strengths. Colloids Surfaces A Physicochem. Eng. Asp. 1998, 131, 19–31. [Google Scholar] [CrossRef]
- Kanicky, J.R.; Shah, D.O. Effect of degree, type, and position of unsaturation on the pKa of long-chain fatty acids. J. Colloid Interface Sci. 2002, 256, 201–207. [Google Scholar] [CrossRef] [PubMed]
- Kanicky, J.; Poniatowski, A.; Mehta, N.; Shah, D. Cooperativity among molecules at interfaces in relation to various technological processes: Effect of chain length on the p K a of fatty acid salt solutions. Langmuir 2000, 16, 172–177. [Google Scholar] [CrossRef]
- Pashkovskaya, A.A.; Vazdar, M.; Zimmermann, L.; Jovanovic, O.; Pohl, P.; Pohl, E.E. Mechanism of long-chain free fatty acid protonation at the membrane-water interface. Biophys. J. 2018, 114, 2142–2151. [Google Scholar] [CrossRef]
- Kanicky, J.; Shah, D. Effect of premicellar aggregation on the p K a of fatty acid soap solutions. Langmuir 2003, 19, 2034–2038. [Google Scholar] [CrossRef]
- Le Calvez, E.; Blaudez, D.; Buffeteau, T.; Desbat, B. Effect of cations on the dissociation of arachidic acid monolayers on water studied by polarization-modulated infrared reflection-absorption spectroscopy. Langmuir 2001, 17, 670–674. [Google Scholar] [CrossRef]
- Lakhdar-Ghazal, F.; Tichadou, J.L.; Tocanne, J.F. Effect of pH and Monovalent Cations on the Ionization State of Phosphatidylglycerol in Monolayers: An Experimental (Surface Potential) and Theoretical (Gouy-Chapman) Approach. Eur. J. Biochem. 1983, 134, 531–537. [Google Scholar] [CrossRef] [PubMed]
- Van Dijck, P.; De Kruijff, B.; Verkleij, A.; Van Deenen, L.; De Gier, J. Comparative studies on the effects of pH and Ca2+ on bilayers of various negatively charged phospholipids and their mixtures with phosphatidylcholine. Biochim. Et Biophys. Acta 1978, 512, 84–96. [Google Scholar] [CrossRef]
- Mengistu, D.H.; Kooijman, E.E.; May, S. Ionization properties of mixed lipid membranes: A Gouy–Chapman model of the electrostatic–hydrogen bond switch. Biochim. Et Biophys. Acta Biomembr. 2011, 1808, 1985–1992. [Google Scholar] [CrossRef]
- Loew, S.; Kooijman, E.E.; May, S. Increased pH-sensitivity of protein binding to lipid membranes through the electrostatic-hydrogen bond switch. Chem. Phys. Lipids 2013, 169, 9–18. [Google Scholar] [CrossRef]
- Graber, Z.T.; Thomas, J.; Johnson, E.; Gericke, A.; Kooijman, E.E. Effect of H-bond donor lipids on phosphatidylinositol-3, 4, 5-trisphosphate ionization and clustering. Biophys. J. 2018, 114, 126–136. [Google Scholar] [CrossRef] [PubMed]
- Gilbile, D.; Docto, D.; Kingi, D.; Kurniawan, J.; Monahan, D.; Tang, A.; Kuhl, T. How Well Can You Tailor the Charge of Lipid Vesicles? Langmuir 2019, 35, 15960–15969. [Google Scholar] [CrossRef]
- Pace, C.N.; Grimsley, G.R.; Scholtz, J.M. Protein ionizable groups: PK values and their contribution to protein stability and solubility. J. Biol. Chem. 2009, 284, 13285–13289. [Google Scholar] [CrossRef] [PubMed]
- Pahari, S.; Sun, L.; Alexov, E. PKAD: A database of experimentally measured pKa values of ionizable groups in proteins. Database 2019, 2019, baz024. [Google Scholar] [CrossRef] [PubMed]
- Smith, B.D.; la Celle, P.L. Parallel decrease of erythrocyte membrane deformability and spectrin solubility at low pH. Blood 1979, 53, 15–18. [Google Scholar] [CrossRef]
- Zhou, Y.; Raphael, R.M. Solution pH alters mechanical and electrical properties of phosphatidylcholine membranes: Relation between interfacial electrostatics, intramembrane potential, and bending elasticity. Biophys. J. 2007, 92, 2451–2462. [Google Scholar] [CrossRef]
- Angelova, M.I.; Bitbol, A.F.; Seigneuret, M.; Staneva, G.; Kodama, A.; Sakuma, Y.; Kawakatsu, T.; Imai, M.; Puff, N. pH sensing by lipids in membranes: The fundamentals of pH-driven migration, polarization and deformations of lipid bilayer assemblies. Biochim. Et Biophys. Acta Biomembr. 2018, 1860, 2042–2063. [Google Scholar] [CrossRef] [PubMed]
- Mclaughlin, S.; Mulrine, N.; Gresalfi, T.; Vaio, G.; Mclaughlin, A. Adsorption of divalent cations to bilayer membranes containing phosphatidylserine. J. Gen. Physiol. 1981, 77, 445–473. [Google Scholar] [CrossRef]
- Binder, H.; Zschörnig, O. The effect of metal cations on the phase behavior and hydration characteristics of phospholipid membranes. Chem. Phys. Lipids 2002, 115, 39–61. [Google Scholar] [CrossRef]
- Cevc, G. Isothermal lipid phase transitions. Chem. Phys. Lipids 1991, 57, 293–307. [Google Scholar] [CrossRef]
- Redondo-Morata, L.; Oncins, G.; Sanz, F. Force spectroscopy reveals the effect of different ions in the nanomechanical behavior of phospholipid model membranes: The case of potassium cation. Biophys. J. 2012, 102, 66–74. [Google Scholar] [CrossRef][Green Version]
- Vitkova, V.; Cenova, J.; Finogenova, O.; Mitov, M.; Ermakov, Y.; Bivas, I. Surface charge effect on the bending elasticity of lipid bilayers. C. R. L’Academie Bulg. Sci. 2004, 57, 11–25. [Google Scholar]
- Winterhalter, M.; Helfrich, W. Bending elasticity of electrically charged bilayers: Coupled monolayers, neutral surfaces, and balancing stresses. J. Phys. Chem. 1992, 96, 327–330. [Google Scholar] [CrossRef]
- Claessens, M.; Leermakers, F.; Hoekstra, F.; Cohen Stuart, M. Opposing effects of cation binding and hydration on the bending rigidity of anionic lipid bilayers. J. Phys. Chem. B 2007, 111, 7127–7132. [Google Scholar] [CrossRef] [PubMed]
- Rappolt, M.; Pabst, G.; Amenitsch, H.; Laggner, P. Salt-induced phase separation in the liquid crystalline phase of phosphatidylcholines. Colloids Surfaces A Physicochem. Eng. Asp. 2001, 183, 171–181. [Google Scholar] [CrossRef]
- Böckmann, R.A.; Hac, A.; Heimburg, T.; Grubmüller, H. Effect of sodium chloride on a lipid bilayer. Biophys. J. 2003, 85, 1647–1655. [Google Scholar] [CrossRef]
- Alexiev, U.; Mollaaghababa, R.; Scherrer, P.; Khorana, H.; Heyn, M. Rapid long-range proton diffusion along the surface of the purple membrane and delayed proton transfer into the bulk. Proc. Natl. Acad. Sci. USA 1995, 92, 372–376. [Google Scholar] [CrossRef]
- Heberle, J.; Riesle, J.; Thiedemann, G.; Oesterhelt, D.; Dencher, N.A. Proton migration along the membrane surface and retarded surface to bulk transfer. Nature 1994, 370, 379–382. [Google Scholar] [CrossRef]
- Clarke, R.J.; Lüpfert, C. Influence of anions and cations on the dipole potential of phosphatidylcholine vesicles: A basis for the Hofmeister effect. Biophys. J. 1999, 76, 2614–2624. [Google Scholar] [CrossRef]
- Trewby, W.; Faraudo, J.; Voïtchovsky, K. Long-lived ionic nano-domains can modulate the stiffness of soft interfaces. Nanoscale 2019, 11, 4376–4384. [Google Scholar] [CrossRef]
- Zhao, W.; Róg, T.; Gurtovenko, A.A.; Vattulainen, I.; Karttunen, M. Atomic-scale structure and electrostatics of anionic palmitoyloleoylphosphatidylglycerol lipid bilayers with Na+ counterions. Biophys. J. 2007, 92, 1114–1124. [Google Scholar] [CrossRef] [PubMed]
- Graber, Z.; Shi, Z.; Baumgart, T. Cations induce shape remodeling of negatively charged phospholipid membranes. Phys. Chem. Chem. Phys. 2017, 19, 15285–15295. [Google Scholar] [CrossRef]
- Böckmann, R.A.; Grubmüller, H. Multistep binding of divalent cations to phospholipid bilayers: A molecular dynamics study. Angew. Chem.-Int. Ed. 2004, 43, 1021–1024. [Google Scholar] [CrossRef]
- Margutti, M.P.; Wilke, N.; Villasuso, A.L. Influence of Ca2+ on the surface behavior of phosphatidic acid and its mixture with diacylglycerol pyrophosphate at different pHs. Chem. Phys. Lipids 2020, 228, 104887. [Google Scholar] [CrossRef] [PubMed]
- Laroche, G.; Dufourc, E.J.; Dufourcq, J.; Pezolet, M. Structure and dynamics of dimyristoylphosphatidic acid/calcium complexes by deuterium NMR, infrared, and Raman spectroscopies and small-angle X-ray diffraction. Biochemistry 1991, 30, 3105–3114. [Google Scholar] [CrossRef]
- Villasuso, A.L.; Wilke, N.; Maggio, B.; Machado, E. Zn2+-dependent surface behavior of diacylglycerol pyrophosphate and its mixtures with phosphatidic acid at different pHs. Front. Plant Sci. 2014, 5, 371. [Google Scholar] [CrossRef] [PubMed]
- Ben-Tal, N.; Honig, B.; Peitzsch, R.M.; Denisov, G.; McLaughlin, S. Binding of small basic peptides to membranes containing acidic lipids: Theoretical models and experimental results. Biophys. J. 1996, 71, 561–575. [Google Scholar] [CrossRef]
- Paterson, D.J.; Tassieri, M.; Reboud, J.; Wilson, R.; Cooper, J.M. Lipid topology and electrostatic interactions underpin lytic activity of linear cationic antimicrobial peptides in membranes. Proc. Natl. Acad. Sci. USA 2017, 114, E8324–E8332. [Google Scholar] [CrossRef] [PubMed]
- Derakhshankhah, H.; Jafari, S. Cell penetrating peptides: A concise review with emphasis on biomedical applications. Biomed. Pharmacother. 2018, 108, 1090–1096. [Google Scholar] [CrossRef] [PubMed]
- Ruseska, I.; Zimmer, A. Internalization mechanisms of cell-penetrating peptides. Beilstein J. Nanotechnol. 2020, 11, 101–123. [Google Scholar] [CrossRef] [PubMed]
- Alvares, D.S.; Viegas, T.G.; Neto, J.R. Lipid-packing perturbation of model membranes by pH-responsive antimicrobial peptides. Biophys. Rev. 2017, 9, 669–682. [Google Scholar] [CrossRef]
- Via, M.A.; del Pópolo, M.G.; Wilke, N. Negative Dipole Potentials and Carboxylic Polar Head Groups Foster the Insertion of Cell-Penetrating Peptides into Lipid Monolayers. Langmuir 2018, 34, 3102–3111. [Google Scholar] [CrossRef]
- Via, M.A.; Klug, J.; Wilke, N.; Mayorga, L.S.; del Pópolo, M.G. The interfacial electrostatic potential modulates the insertion of cell-penetrating peptides into lipid bilayers. Phys. Chem. Chem. Phys. 2018, 20, 5180–5189. [Google Scholar] [CrossRef] [PubMed]
- Via, M.A.; Wilke, N.; Mayorga, L.S.; del Pópolo, M.G. Surface charge density and fatty acids enhance the membrane permeation rate of CPP–cargo complexes. Soft Matter 2020, 16, 9890–9898. [Google Scholar] [CrossRef] [PubMed]
- Batta, G.; Karpati, L.; Fulaneto, G.; Tarapcsak, S.; Kovacs, T.; Zakany, F.; Mandity, I.; Nagy, P. Statin-boosted cellular uptake and endosomal escape of penetratin due to reduced membrane dipole potential. Authorea Prepr. 2020. [Google Scholar] [CrossRef]
- Crosio, M.A.; Via, M.A.; Cámara, C.I.; Mangiarotti, A.; del Pópolo, M.G.; Wilke, N. Interaction of a polyarginine peptide with membranes of different mechanical properties. Biomolecules 2019, 9, 625. [Google Scholar] [CrossRef]
- Sharmin, S.; Islam, M.Z.; Karal, M.A.S.; Alam Shibly, S.U.; Dohra, H.; Yamazaki, M. Effects of lipid composition on the entry of cell-penetrating peptide oligoarginine into single vesicles. Biochemistry 2016, 55, 4154–4165. [Google Scholar] [CrossRef]
- Alvares, D.S.; Monti, M.R.; Neto, J.R.; Wilke, N. The antimicrobial peptide Polybia-MP1 differentiates membranes with the hopanoid, diplopterol from those with cholesterol. BBA Adv. 2021, 1, 100002. [Google Scholar] [CrossRef]
- Herce, H.; Garcia, A.; Litt, J.; Kane, R.; Martín, P.; Enrique, N.; Rebolledo, A.; Milesi, V. Arginine-rich peptides destabilize the plasma membrane, consistent with a pore formation translocation mechanism of cell-penetrating peptides. Biophys. J. 2009, 97, 1917–1925. [Google Scholar] [CrossRef]
- Di Pisa, M.; Chassaing, G.; Swiecicki, J.M. Translocation mechanism (s) of cell-penetrating peptides: Biophysical studies using artificial membrane bilayers. Biochemistry 2015, 54, 194–207. [Google Scholar] [CrossRef] [PubMed]
- Kristiansen, J.E.; Hendricks, O.; Delvin, T.; Butterworth, T.S.; Aagaard, L.; Christensen, J.B.; Flores, V.C.; Keyzer, H. Reversal of resistance in microorganisms by help of non-antibiotics. J. Antimicrob. Chemother. 2007, 59, 1271–1279. [Google Scholar] [CrossRef] [PubMed]
- Henriques, S.T.; Melo, M.N.; Castanho, M.A. Cell-penetrating peptides and antimicrobial peptides: How different are they? Biochem. J. 2006, 399, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Jobin, M.L.; Alves, I.D. On the importance of electrostatic interactions between cell penetrating peptides and membranes: A pathway toward tumor cell selectivity? Biochimie 2014, 107, 154–159. [Google Scholar] [CrossRef]
- Akabori, K.; Huang, K.; Treece, B.W.; Jablin, M.S.; Maranville, B.; Woll, A.; Nagle, J.F.; Garcia, A.E.; Tristram-Nagle, S. HIV-1 Tat membrane interactions probed using X-ray and neutron scattering, CD spectroscopy and MD simulations. Biochim. Et Biophys. Acta Biomembr. 2014, 1838, 3078–3087. [Google Scholar] [CrossRef] [PubMed]
- Tristram-Nagle, S.; Chan, R.; Kooijman, E.E.; Qiang, W.; Weliky, D.P.; Nagle, J.F. HIV Fusion Peptide Penetrates, Disorders and Softens T-Cell Membrane Mimics. Biophys. J. 2011, 100, 186a. [Google Scholar] [CrossRef][Green Version]
- Fa, N.; Lins, L.; Courtoy, P.J.; Dufrêne, Y.; van der Smissen, P.; Brasseur, R.; Tyteca, D.; Mingeot-Leclercq, M.P. Decrease of elastic moduli of DOPC bilayers induced by a macrolide antibiotic, azithromycin. Biochim. Et Biophys. Acta Biomembr. 2007, 1768, 1830–1838. [Google Scholar] [CrossRef] [PubMed]
- Pan, J.; Tieleman, D.P.; Nagle, J.F.; Kučerka, N.; Tristram-Nagle, S. Alamethicin in lipid bilayers: Combined use of X-ray scattering and MD simulations. Biochim. Et Biophys. Acta Biomembr. 2009, 1788, 1387–1397. [Google Scholar] [CrossRef]
- Pabst, G.; Danner, S.; Podgornik, R.; Katsaras, J. Entropy-driven softening of fluid lipid bilayers by alamethicin. Langmuir 2007, 23, 11705–11711. [Google Scholar] [CrossRef] [PubMed]
- Häckl, W.; Seifert, U.; Sackmann, E. Effects of fully and partially solubilized amphiphiles on bilayer bending stiffness and temperature dependence of the effective tension of giant vesicles. J. Phys. II 1997, 7, 1141–1157. [Google Scholar] [CrossRef][Green Version]
- Agrawal, H.; Zelisko, M.; Liu, L.; Sharma, P. Rigid proteins and softening of biological membranes—with application to HIV-induced cell membrane softening. Sci. Rep. 2016, 6, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Bouvrais, H.; Méléard, P.; Pott, T.; Jensen, K.J.; Brask, J.; Ipsen, J.H. Softening of POPC membranes by magainin. Biophys. Chem. 2008, 137, 7–12. [Google Scholar] [CrossRef] [PubMed]
- Fournier, J. Nontopological saddle-splay and curvature instabilities from anisotropic membrane inclusions. Phys. Rev. Lett. 1996, 76, 4436. [Google Scholar] [CrossRef]
- Vitkova, V.; Méléard, P.; Pott, T.; Bivas, I. Alamethicin influence on the membrane bending elasticity. Eur. Biophys. J. 2006, 35, 281–286. [Google Scholar] [CrossRef]
- West, A.; Brummel, B.E.; Braun, A.R.; Rhoades, E.; Sachs, J.N. Membrane remodeling and mechanics: Experiments and simulations of α-Synuclein. Biochim. Et Biophys. Acta Biomembr. 2016, 1858, 1594–1609. [Google Scholar] [CrossRef] [PubMed]
- Zemel, A.; Ben-Shaul, A.; May, S. Modulation of the spontaneous curvature and bending rigidity of lipid membranes by interfacially adsorbed amphipathic peptides. J. Phys. Chem. B 2008, 112, 6988–6996. [Google Scholar] [CrossRef] [PubMed]
- Mishra, A.; Gordon, V.D.; Yang, L.; Coridan, R.; Wong, G.C. HIV TAT forms pores in membranes by inducing saddle-splay curvature: Potential role of bidentate hydrogen bonding. Angew. Chem. Int. Ed. 2008, 47, 2986–2989. [Google Scholar] [CrossRef] [PubMed]
- Shchelokovskyy, P.; Tristram-Nagle, S.; Dimova, R. Effect of the HIV-1 fusion peptide on the mechanical properties and leaflet coupling of lipid bilayers. New J. Phys. 2011, 13, 025004. [Google Scholar] [CrossRef] [PubMed]
- Grasso, G.; Muscat, S.; Rebella, M.; Morbiducci, U.; Audenino, A.; Danani, A.; Deriu, M.A. Cell penetrating peptide modulation of membrane biomechanics by Molecular dynamics. J. Biomech. 2018, 73, 137–144. [Google Scholar] [CrossRef]
- Dupuy, F.G.; Pagano, I.; Andenoro, K.; Peralta, M.F.; Elhady, Y.; Heinrich, F.; Tristram-Nagle, S. Selective interaction of colistin with lipid model membranes. Biophys. J. 2018, 114, 919–928. [Google Scholar] [CrossRef] [PubMed]
- Taheri-Araghi, S.; Ha, B.Y. Cationic antimicrobial peptides: A physical basis for their selective membrane-disrupting activity. Soft Matter 2010, 6, 1933–1940. [Google Scholar] [CrossRef]
- Cahill, K. Simple model of the transduction of cell-penetrating peptides. IET Syst. Biol. 2009, 3, 300–306. [Google Scholar] [CrossRef] [PubMed]
- Cohen, L.; Keynes, R.; Hille, B. Light scattering and birefringence changes during nerve activity. Nature 1968, 218, 438–441. [Google Scholar] [CrossRef]
- Zhang, P.C.; Keleshian, A.M.; Sachs, F. Voltage-induced membrane movement. Nature 2001, 413, 428–432. [Google Scholar] [CrossRef] [PubMed]
- Sachs, F.; Brownell, W.; Petrov, A. Membrane electromechanics in biology, with a focus on hearing. MRS Bull. 2009, 34, 665–670. [Google Scholar] [CrossRef] [PubMed]
- Zhang, R.; Qian, F.; Rajagopalan, L.; Pereira, F.A.; Brownell, W.E.; Anvari, B. Prestin modulates mechanics and electromechanical force of the plasma membrane. Biophys. J. 2007, 93, L07–L09. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Petrov, A.G. The Lyotropic State of Matter: Molecular Physics and Living Matter Physics; CRC Press: Boca Raton, FL, USA, 1999. [Google Scholar]
- Vaseashta, A.K.; Mihailescu, I.N. Functionalized Nanoscale Materials, Devices and Systems; Springer Science & Business Media: Berlin/Heidelberg, Germany, 2008. [Google Scholar]
- Rey, A.D.; Servio, P.; Herrera-Valencia, E. Bioinspired model of mechanical energy harvesting based on flexoelectric membranes. Phys. Rev. E 2013, 87, 022505. [Google Scholar] [CrossRef]
- Ahmadpoor, F.; Sharma, P. Flexoelectricity in two-dimensional crystalline and biological membranes. Nanoscale 2015, 7, 16555–16570. [Google Scholar] [CrossRef]
- Vlahovska, P.M. Voltage-morphology coupling in biomimetic membranes: Dynamics of giant vesicles in applied electric fields. Soft Matter 2015, 11, 7232–7236. [Google Scholar] [CrossRef] [PubMed]
- May, S. Curvature elasticity and thermodynamic stability of electrically charged membranes. J. Chem. Phys. 1996, 105, 8314–8323. [Google Scholar] [CrossRef]
- Sarkar, Y.; Majumder, R.; Das, S.; Ray, A.; Parui, P.P. Detection of curvature-radius-dependent interfacial pH/polarity for amphiphilic self-assemblies: Positive versus negative curvature. Langmuir 2017, 34, 6271–6284. [Google Scholar] [CrossRef]
- Pasechnik, V.; Sokolov, V. Change in the permeability of modified bimolecular phospholipid membranes iwth periodic stratching. Biofizika 1973, 18, 655–660. [Google Scholar]
- Ochs, A.L.; Burton, R.M. Electrical response to vibration of a lipid bilayer membrane. Biophys. J. 1974, 14, 473–489. [Google Scholar] [CrossRef][Green Version]
- Petrov, A.G. Physical and Chemical Bases of Biological Information Transfer; Vassileva, J., Ed.; Plenum Press: New York, NY, USA, 1975; p. 167. [Google Scholar]
- Derzhanski, A.; Petrov, A.; Pavloff, Y. Curvature induced conductive and displacement currents through lipid bilayers. J. Phys. Lett. 1981, 42, 119–122. [Google Scholar] [CrossRef][Green Version]
- Petrov, A.G. Flexoelectricity of model and living membranes. Biochim. Et Biophys. Acta Biomembr. 2001, 1561, 1–25. [Google Scholar] [CrossRef]
- Winiski, A.P.; McLaughlin, A.C.; McDaniel, R.V.; Eisenberg, M.; McLaughlin, S. An experimental test of the discreteness-of-charge effect in positive and negative lipid bilayers. Biochemistry 1986, 25, 8206–8214. [Google Scholar] [CrossRef]
- Ambjörnsson, T.; Lomholt, M.A.; Hansen, P.L. Applying a potential across a biomembrane: Electrostatic contribution to the bending rigidity and membrane instability. Phys. Rev. E 2007, 75, 051916. [Google Scholar] [CrossRef]
- Harland, B.; Brownell, W.E.; Spector, A.A.; Sun, S.X. Voltage-induced bending and electromechanical coupling in lipid bilayers. Phys. Rev. E 2010, 81, 031907. [Google Scholar] [CrossRef]
- Bezanilla, F. How membrane proteins sense voltage. Nat. Rev. Mol. Cell Biol. 2008, 9, 323–332. [Google Scholar] [CrossRef] [PubMed]
- Pearlstein, R.A.; Dickson, C.J.; Hornak, V. Contributions of the membrane dipole potential to the function of voltage-gated cation channels and modulation by small molecule potentiators. Biochim. Et Biophys. Acta Biomembr. 2017, 1859, 177–194. [Google Scholar] [CrossRef] [PubMed]
- Weaver, J.C.; Chizmadzhev, Y.A. Theory of electroporation: A review. Bioelectrochem. Bioenerg. 1996, 41, 135–160. [Google Scholar] [CrossRef]
- Dimova, R.; Riske, K.A.; Aranda, S.; Bezlyepkina, N.; Knorr, R.L.; Lipowsky, R. Giant vesicles in electric fields. Soft Matter 2007, 3, 817–827. [Google Scholar] [CrossRef]
- Zimmermann, U.; Vienken, J.; Pilwat, G. Development of drug carrier systems: Electrical field induced effects in cell membranes. J. Electroanal. Chem. Interfacial Electrochem. 1980, 116, 553–574. [Google Scholar] [CrossRef]
- Neumann, E.; Schaefer-Ridder, M.; Wang, Y.; Hofschneider, P. Gene transfer into mouse lyoma cells by electroporation in high electric fields. EMBO J. 1982, 1, 841–845. [Google Scholar] [CrossRef] [PubMed]
- Needham, D.; Hochmuth, R. Electro-mechanical permeabilization of lipid vesicles. Role of membrane tension and compressibility. Biophys. J. 1989, 55, 1001–1009. [Google Scholar] [CrossRef]
- Weaver, J.C. Electroporation of biological membranes from multicellular to nano scales. IEEE Trans. Dielectr. Electr. Insul. 2003, 10, 754–768. [Google Scholar] [CrossRef]
- Martí, J.M.L.; English, N.J.; del Pópolo, M.G. Elucidating mysteries of phase-segregated membranes: Mobile-lipid recruitment facilitates pores’ passage to the fluid phase. Phys. Chem. Chem. Phys. 2018, 20, 19234–19239. [Google Scholar] [CrossRef]
- Kotnik, T.; Rems, L.; Tarek, M.; Miklavčič, D. Membrane electroporation and electropermeabilization: Mechanisms and models. Annu. Rev. Biophys. 2019, 48, 63–91. [Google Scholar] [CrossRef]
- Kirsch, S.A.; Böckmann, R.A. Coupling of membrane nanodomain formation and enhanced electroporation near phase transition. Biophys. J. 2019, 116, 2131–2148. [Google Scholar] [CrossRef]
- Chen, C.; Smye, S.; Robinson, M.; Evans, J. Membrane electroporation theories: A review. Med. Biol. Eng. Comput. 2006, 44, 5–14. [Google Scholar] [CrossRef]
- Sun, S.; Yin, G.; Lee, Y.K.; Wong, J.T.; Zhang, T.Y. Effects of deformability and thermal motion of lipid membrane on electroporation: By molecular dynamics simulations. Biochem. Biophys. Res. Commun. 2011, 404, 684–688. [Google Scholar] [CrossRef]
- Hoejholt, K.; Mužić, T.; Jensen, S.; Dalgaard, L.T.; Bilgin, M.; Nylandsted, J.; Heimburg, T.; Frandsen, S.; Gehl, J. Calcium electroporation and electrochemotherapy for cancer treatment: Importance of cell membrane composition investigated by lipidomics, calorimetry and in vitro efficacy. Sci. Rep. 2019, 9, 1–12. [Google Scholar] [CrossRef]
- Neu, W.K.; Neu, J.C. Theory of electroporation. In Cardiac Bioelectric Therapy; Springer: Berlin/Heidelberg, Germany, 2009; pp. 133–161. [Google Scholar]
- Lewis, T.J. A model for bilayer membrane electroporation based on resultant electromechanical stress. IEEE Trans. Dielectr. Electr. Insul. 2003, 10, 769–777. [Google Scholar] [CrossRef]
- Böckmann, R.A.; de Groot, B.L.; Kakorin, S.; Neumann, E.; Grubmüller, H. Kinetics, statistics, and energetics of lipid membrane electroporation studied by molecular dynamics simulations. Biophys. J. 2008, 95, 1837–1850. [Google Scholar] [CrossRef] [PubMed]
- Breton, M.; Mir, L.M. Investigation of the chemical mechanisms involved in the electropulsation of membranes at the molecular level. Bioelectrochemistry 2018, 119, 76–83. [Google Scholar] [CrossRef]
- Van Uitert, I.; le Gac, S.; van den Berg, A. Determination of the electroporation onset of bilayer lipid membranes as a novel approach to establish ternary phase diagrams: Example of the l-α-PC/SM/cholesterol system. Soft Matter 2010, 6, 4420–4429. [Google Scholar] [CrossRef]
- Mangiarotti, A.; Genovese, D.M.; Naumann, C.A.; Monti, M.R.; Wilke, N. Hopanoids, like sterols, modulate dynamics, compaction, phase segregation and permeability of membranes. Biochim. Et Biophys. Acta Biomembr. 2019, 1861, 183060. [Google Scholar] [CrossRef]
- Ahamed, M.K.; Karal, M.A.S.; Ahmed, M.; Ahammed, S. Kinetics of irreversible pore formation under constant electrical tension in giant unilamellar vesicles. Eur. Biophys. J. 2020, 49, 371–381. [Google Scholar] [CrossRef]
- Sengel, J.T.; Wallace, M.I. Imaging the dynamics of individual electropores. Proc. Natl. Acad. Sci. USA 2016, 113, 5281–5286. [Google Scholar] [CrossRef]
- Tasaki, I.; Byrne, P. Volume expansion of nonmyelinated nerve fibers during impulse conduction. Biophys. J. 1990, 57, 633. [Google Scholar] [CrossRef]
- Qian, F.; Ermilov, S.; Murdock, D.; Brownell, W.E.; Anvari, B. Combining optical tweezers and patch clamp for studies of cell membrane electromechanics. Rev. Sci. Instrum. 2004, 75, 2937–2942. [Google Scholar] [CrossRef] [PubMed]
- Brownell, W.E.; Qian, F.; Anvari, B. Cell membrane tethers generate mechanical force in response to electrical stimulation. Biophys. J. 2010, 99, 845–852. [Google Scholar] [CrossRef] [PubMed]
- Akkin, T.; Joo, C.; de Boer, J.F. Depth-resolved measurement of transient structural changes during action potential propagation. Biophys. J. 2007, 93, 1347–1353. [Google Scholar] [CrossRef]
- Akkin, T.; Landowne, D.; Sivaprakasam, A. Optical coherence tomography phase measurement of transient changes in squid giant axons during activity. J. Membr. Biol. 2009, 231, 35–46. [Google Scholar] [CrossRef] [PubMed]
- Oh, S.; Fang-Yen, C.; Choi, W.; Yaqoob, Z.; Fu, D.; Park, Y.; Dassari, R.R.; Feld, M.S. Label-free imaging of membrane potential using membrane electromotility. Biophys. J. 2012, 103, 11–18. [Google Scholar] [CrossRef]
- Nguyen, T.D.; Deshmukh, N.; Nagarah, J.M.; Kramer, T.; Purohit, P.K.; Berry, M.J.; McAlpine, M.C. Piezoelectric nanoribbons for monitoring cellular deformations. Nat. Nanotechnol. 2012, 7, 587–593. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Liu, X.W.; Wang, H.; Yu, H.; Guan, Y.; Wang, S.; Tao, N. Imaging action potential in single mammalian neurons by tracking the accompanying sub-nanometer mechanical motion. ACS Nano 2018, 12, 4186–4193. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Liu, X.; Wang, S.; Tao, N. Plasmonic imaging of subcellular electromechanical deformation in mammalian cells. J. Biomed. Opt. 2019, 24, 066007. [Google Scholar] [CrossRef] [PubMed]
- Ling, T.; Boyle, K.C.; Zuckerman, V.; Flores, T.; Ramakrishnan, C.; Deisseroth, K.; Palanker, D. High-speed interferometric imaging reveals dynamics of neuronal deformation during the action potential. Proc. Natl. Acad. Sci. USA 2020, 117, 10278–10285. [Google Scholar] [CrossRef] [PubMed]
- Fillafer, C.; Mussel, M.; Muchowski, J.; Schneider, M.F. Cell surface deformation during an action potential. Biophys. J. 2018, 114, 410–418. [Google Scholar] [CrossRef]
- Tasaki, I.; Carnay, L.; Sandlin, R.; Watanabe, A. Fluorescence changes during conduction in nerves stained with acridine orange. Science 1969, 163, 683–685. [Google Scholar] [CrossRef]
- Abbott, B.; Hill, A.V.; Howarth, J. The positive and negative heat production associated with a nerve impulse. Proc. R. Soc. Lond. Ser. B-Biol. Sci. 1958, 148, 149–187. [Google Scholar]
- Heimburg, T.; Jackson, A.D. On soliton propagation in biomembranes and nerves. Proc. Natl. Acad. Sci. USA 2005, 102, 9790–9795. [Google Scholar] [CrossRef] [PubMed]
- Heimburg, T. The capacitance and electromechanical coupling of lipid membranes close to transitions: The effect of electrostriction. Biophys. J. 2012, 103, 918–929. [Google Scholar] [CrossRef]
- Fillafer, C.; Paeger, A.; Schneider, M.F. The living state: How cellular excitability is controlled by the thermodynamic state of the membrane. Prog. Biophys. Mol. Biol. 2020, 162, 57–68. [Google Scholar] [CrossRef]
- Overton, C.E. Studies of Narcosis; Springer Science & Business Media: Berlin/Heidelberg, Germany, 1991. [Google Scholar]
- Wang, T.; Mužić, T.; Jackson, A.D.; Heimburg, T. The free energy of biomembrane and nerve excitation and the role of anesthetics. Biochim. Et Biophys. Acta Biomembr. 2018, 1860, 2145–2153. [Google Scholar] [CrossRef] [PubMed]
- Tamayo-Elizalde, M.; Chen, H.; Malboubi, M.; Ye, H.; Jerusalem, A. Action potential alterations induced by single F11 neuronal cell loading. Prog. Biophys. Mol. Biol. 2021, 162, 141–153. [Google Scholar] [CrossRef]
- Prieto, M.L.; Oralkan, Ö.; Khuri-Yakub, B.T.; Maduke, M.C. Dynamic response of model lipid membranes to ultrasonic radiation force. PLoS ONE 2013, 8, e77115. [Google Scholar] [CrossRef] [PubMed]
- Jerusalem, A.; Al-Rekabi, Z.; Chen, H.; Ercole, A.; Malboubi, M.; Tamayo-Elizalde, M.; Verhagen, L.; Contera, S. Electrophysiological-mechanical coupling in the neuronal membrane and its role in ultrasound neuromodulation and general anaesthesia. Acta Biomater. 2019, 97, 116–140. [Google Scholar] [CrossRef] [PubMed]
- Engelbrecht, J.; Peets, T.; Tamm, K.; Laasmaa, M.; Vendelin, M. On the complexity of signal propagation in nerve fibres. Proc. Est. Acad. Sci. 2018, 67. [Google Scholar] [CrossRef]
- Chen, H.; Garcia-Gonzalez, D.; Jérusalem, A. Computational model of the mechanoelectrophysiological coupling in axons with application to neuromodulation. Phys. Rev. E 2019, 99, 032406. [Google Scholar] [CrossRef]
- Holland, L.; de Regt, H.W.; Drukarch, B. Thinking about the nerve impulse: The prospects for the development of a comprehensive account of nerve impulse propagation. Front. Cell. Neurosci. 2019, 13, 208. [Google Scholar] [CrossRef]
- Shrivastava, S.; Schneider, M.F. Evidence for two-dimensional solitary sound waves in a lipid controlled interface and its implications for biological signalling. J. R. Soc. Interface 2014, 11, 20140098. [Google Scholar] [CrossRef] [PubMed]
- Tamagawa, H.; Ikeda, K. Another interpretation of the Goldman–Hodgkin–Katz equation based on Ling’s adsorption theory. Eur. Biophys. J. 2018, 47, 869–879. [Google Scholar] [CrossRef] [PubMed]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Galassi, V.V.; Wilke, N. On the Coupling between Mechanical Properties and Electrostatics in Biological Membranes. Membranes 2021, 11, 478. https://doi.org/10.3390/membranes11070478
Galassi VV, Wilke N. On the Coupling between Mechanical Properties and Electrostatics in Biological Membranes. Membranes. 2021; 11(7):478. https://doi.org/10.3390/membranes11070478
Chicago/Turabian StyleGalassi, Vanesa Viviana, and Natalia Wilke. 2021. "On the Coupling between Mechanical Properties and Electrostatics in Biological Membranes" Membranes 11, no. 7: 478. https://doi.org/10.3390/membranes11070478
APA StyleGalassi, V. V., & Wilke, N. (2021). On the Coupling between Mechanical Properties and Electrostatics in Biological Membranes. Membranes, 11(7), 478. https://doi.org/10.3390/membranes11070478






