Laboratory Efficacy of Locally Available Backwashing Methods at Removing Fouling in Hollow-Fiber Membrane Filters Used for Household Water Treatment
Abstract
1. Introduction
2. Materials and Methods
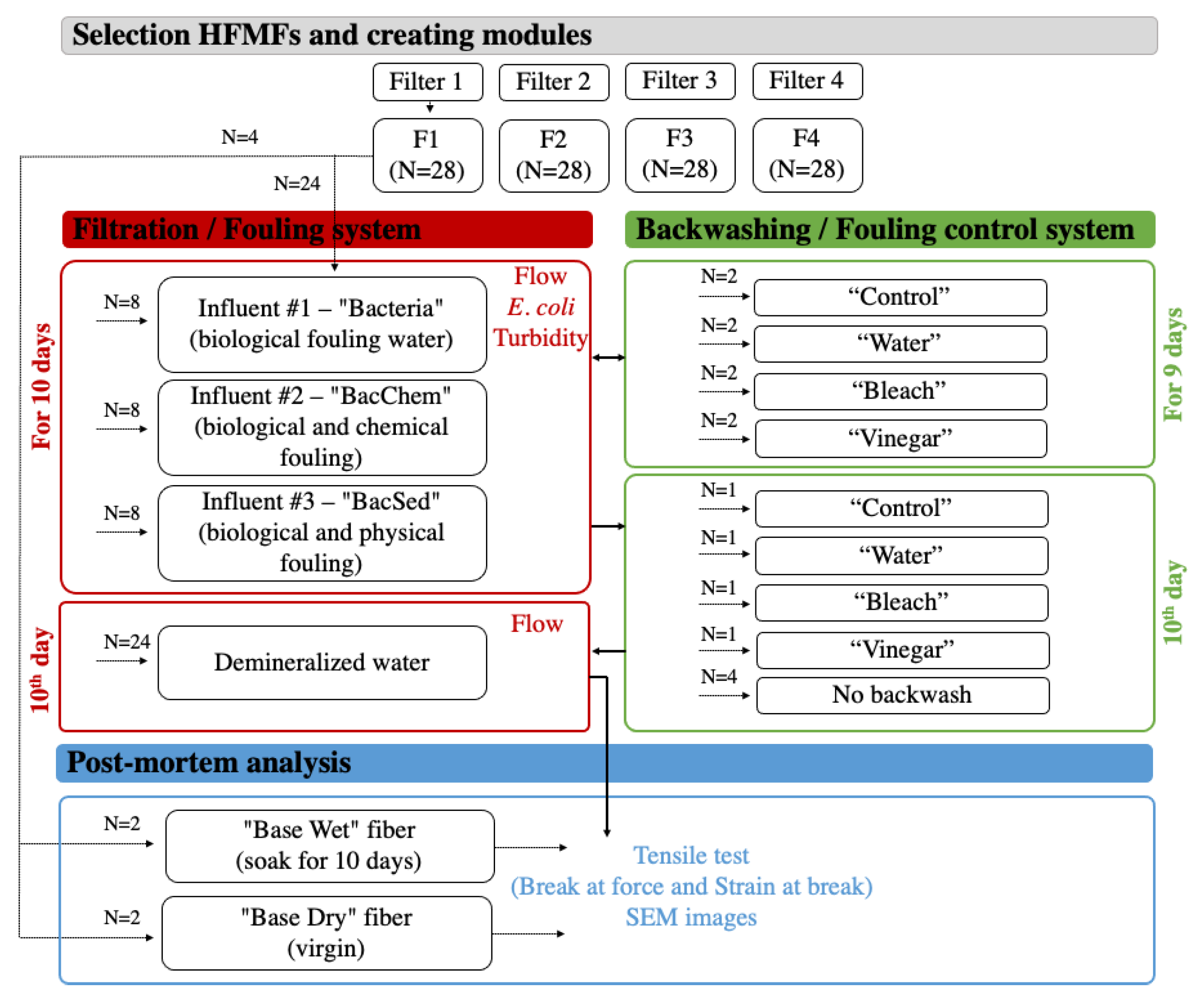
2.1. Selection of Commonly Distributed HFMFs
2.2. Preparation of Modules for Testing
2.3. Filtration System
2.4. Backwashing System
2.5. Data Collection Runs
2.6. Postmortem Analysis
2.7. Data Analysis
3. Results
3.1. Assessing Filter Variation
3.2. Influence of Fiber Wetting Before Testing
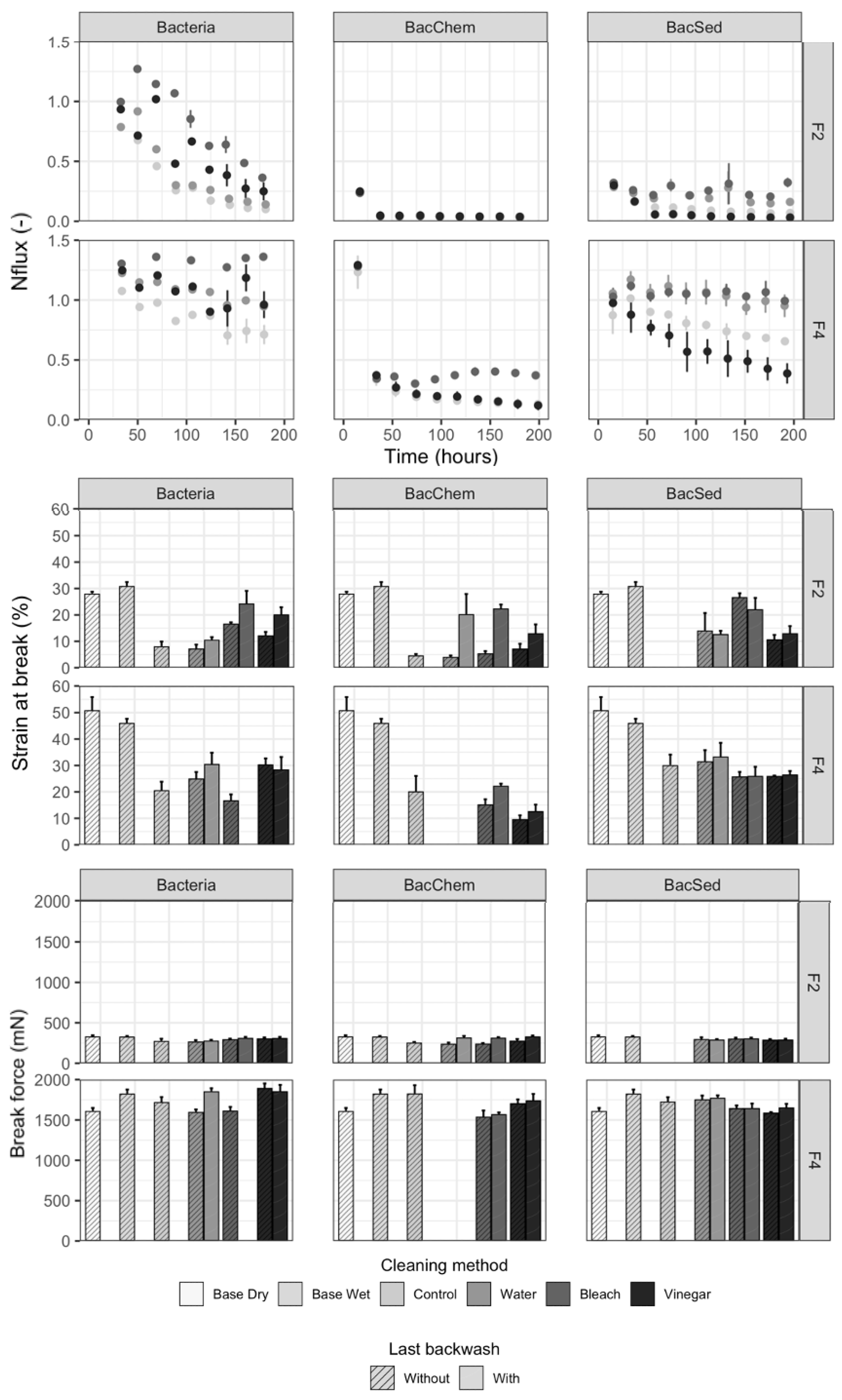
3.3. Development of Fouling on Membranes without Backwashing
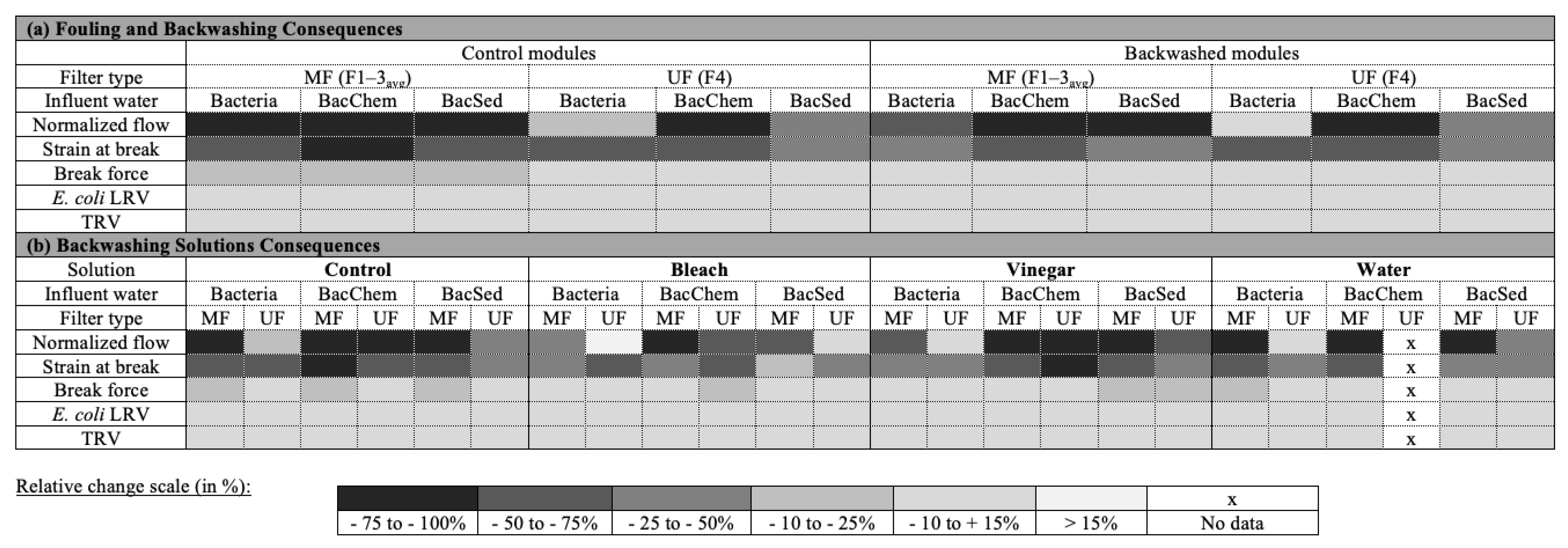
3.4. Performance of Backwashed Modules Compared to Nonbackwashed Modules
3.5. Recovery of Filter Performance by Backwashing Solutions
4. Summary and Recommendations
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- UNICEF; WHO. Progress on Household Drinking Water, Sanitation and Hygiene 2000–2017: Special Focus on Inequalities; UNICEF: New York, NY, USA; WHO: New York, NY, USA, 2019. [Google Scholar]
- WHO. WHO International Scheme to Evaluate Household Water Treatment Technologies; WHO: Geneva, Switzerland, 2016; Available online: https://www.who.int/household_water/scheme/household-water-treatment-report-round-1/en/ (accessed on 20 March 2021).
- Fewtrell, L.; Kaufmann, R.B.; Kay, D.; Enanoria, W.; Haller, L.; Colford, J.M. Water, sanitation, and hygiene interventions to reduce diarrhoea in less developed countries: A systematic review and meta-analysis. Lancet Infect. Dis. 2005, 5, 42–52. [Google Scholar] [CrossRef]
- Clasen, T.F.; Alexander, K.T.; Sinclair, D.; Boisson, S.; Peletz, R.; Chang, H.H.; Majorin, F.; Cairncross, S. Interventions to improve water quality for preventing diarrhoea. Cochrane Database Syst. Rev. 2015. [Google Scholar] [CrossRef] [PubMed]
- WHO. Evaluating Household Water Treatment Options: Health-Based Targets and Microbiological Performance Specifications; World Health Organization: Geneva, Switzerland, 2011; Available online: https://apps.who.int/iris/handle/10665/44693 (accessed on 20 March 2021).
- Obsey, M.D.S.; Tauber, C.E.S.; Asanova, L.M.C.; Brown, J.M.; Elliott, M.A. Point of Use Household Drinking Water Filtration: A Practical, Effective Solution for Providing Sustained Access to Safe Drinking Water in the Developing World. Environ. Sci. Technol. 2008, 42, 4261–4267. [Google Scholar] [CrossRef]
- CAWST. HIF Emergency Household Water Filter Challenge: Evaluation Matrix and Report; CAWST: Calgary, AB, Canada, 2017; Available online: https://www.elrha.org/wp-content/uploads/2017/06/2017-06-01_HIF-Emergency-Household-Water-Filter-Challenge_CAWST-Evaluation-Matrix-and-Report.pdf (accessed on 20 March 2021).
- WHO. Results of Round II of the WHO International Scheme to Evaluate Household Water Treatment Technologies; WHO: Geneva, Switzerland, 2019; Available online: https://www.who.int/water_sanitation_health/publications/highlights-results-of-roundII-of-the-who--scheme-to-evaluate-household-water-treatment-technologies.pdf?ua=1 (accessed on 20 March 2021).
- Kirby, M.A.; Nagel, C.L.; Rosa, G.; Umupfasoni, M.M.; Iyakaremye, L.; Thomas, E.A.; Clasen, T.F. Use, microbiological effectiveness and health impact of a household water filter intervention in rural Rwanda—A matched cohort study. Int. J. Hyg. Environ. Health 2017, 220, 1020–1029. [Google Scholar] [CrossRef] [PubMed]
- Boisson, S.; Kiyombo, M.; Sthreshley, L.; Tumba, S.; Makambo, J.; Clasen, T. Field Assessment of a Novel Household-Based Water Filtration Device: A Randomised, Placebo-Controlled Trial in the Democratic Republic of Congo. PLoS ONE 2010, 5, e12613. [Google Scholar] [CrossRef] [PubMed]
- Pickering, A.J.; Arnold, B.F.; Dentz, H.N.; Colford, J.M.; Null, C. Climate and Health Co-Benefits in Low-Income Countries: A Case Study of Carbon Financed Water Filters in Kenya and a Call for Independent Monitoring. Environ. Health Perspect. 2017, 125, 278–283. [Google Scholar] [CrossRef] [PubMed]
- Goeb, M. Follow-Up on Sawyer Filters in the Community of San Francisco de las Quebradas, San Ramon Nr.2 and Santa Rosa Nr.2, Trojes, Honduras; Pure Water for the World: Rutland, VT, USA, 2013. [Google Scholar]
- Holding, S.; Sadeghi, I.; White, T.; Murray, A.; Ray, J.; Asatekin, A.; Lantagne, D. Acceptability, effectiveness, and fouling of PointOne membrane filters distributed in South Sudan. J. Water Sanit. Hyg. Dev. 2019, 9, 247–257. [Google Scholar] [CrossRef]
- Kohlitz, J.; Hasan, T.; Khatri, K.; Sokota, A.; Iddings, S.; Bera, U.; Psutka, R. Assessing reported use and microbiological performance of a point-of-use household water filter in rural Fiji. J. Water Sanit. Hyg. Dev. 2013, 3, 207–215. [Google Scholar] [CrossRef]
- Fagerli, K.; Hurd, J.; Wells, E.; McAteer, J.; Kim, S.; Seal, L.; Akers, P.; Murphy, J.L.; Quick, R. Evaluation of use, acceptability, and effectiveness of household water filter systems in Honduras, 2016–2017. J. Water Sanit. Hyg. Dev. 2018, 8, 809–816. [Google Scholar] [CrossRef]
- Murray, A.; Goeb, M.; Stewart, B.; Hopper, C.; Peck, J.; Meub, C.; Asatekin, A.; Lantagne, D. Fouling in hollow fiber membrane microfilters used for household water treatment. J. Water Sanit. Hyg. Dev. 2015, 5, 220–228. [Google Scholar] [CrossRef]
- Guo, W.; Ngo, H.-H.; Li, J. A mini-review on membrane fouling. Bioresour. Technol. 2012, 122, 27–34. [Google Scholar] [CrossRef] [PubMed]
- Ayache, C.; Pidou, M.; Croué, J.; Labanowski, J.; Poussade, Y.; Tazi-Pain, A.; Keller, J.; Gernjak, W. Impact of effluent organic matter on low-pressure membrane fouling in tertiary treatment. Water Res. 2013, 47, 2633–2642. [Google Scholar] [CrossRef] [PubMed]
- Amy, G.; Cho, J. Interactions between natural organic matter (nom) and membranes: Rejection and fouling. Water Sci. Technol. 1999, 40, 131–139. [Google Scholar] [CrossRef]
- Akhondi, E.; Zamani, F.; Tng, K.H.; Leslie, G.; Krantz, W.B.; Fane, A.G.; Chew, J.W. The Performance and Fouling Control of Submerged Hollow Fiber (HF) Systems: A Review. Appl. Sci. 2017, 7, 765. [Google Scholar] [CrossRef]
- Nghiem, L.D.; Schäfer, A.I. Fouling autopsy of hollow-fibre MF membranes in wastewater reclamation. Desalination 2006, 188, 113–121. [Google Scholar] [CrossRef]
- Hilal, N.; Ogunbiyi, O.O.; Miles, N.J.; Nigmatullin, R. Methods Employed for Control of Fouling in MF and UF Membranes: A Comprehensive Review. Sep. Sci. Technol. 2005, 40, 1957–2005. [Google Scholar] [CrossRef]
- Zularisam, A.; Ismail, A.; Salim, R. Behaviours of natural organic matter in membrane filtration for surface water treatment—A review. Desalination 2006, 194, 211–231. [Google Scholar] [CrossRef]
- Pooi, C.K.; Ng, H.Y. Review of low-cost point-of-use water treatment systems for developing communities. NPJ Clean Water 2018, 1, 11. [Google Scholar] [CrossRef]
- Gao, W.; Liang, H.; Ma, J.; Han, M.; Chen, Z.-L.; Han, Z.-S.; Li, G.-B. Membrane fouling control in ultrafiltration technology for drinking water production: A review. Desalination 2011, 272, 1–8. [Google Scholar] [CrossRef]
- Lifestraw. Long Term Storage and Maintenance. Available online: https://cdn.shopify.com/s/files/1/2631/0778/files/LifeStraw_Long_Term_Storage_and_Maintenance.pdf?v=12680717380178311691 (accessed on 20 March 2020).
- Sawyer. Cleaning Instructions Sawyer Filters. Available online: https://sawyer.com/wp-content/uploads/2013/11/sawyer_bucket-cleaning_2016-01-15e.pdf (accessed on 23 August 2019).
- Village Water Filters. Bucket Filter Backflush Instructions. Available online: https://villagewaterfilters.org/wp-content/uploads/2016/10/VWF-VF100Bucket-Filter-Backflush-Instructions.pdf (accessed on 23 August 2019).
- Sawyer. Squeeze Water Filtration System | Sawyer Products. Available online: https://sawyer.com/products/sawyer-squeeze-filter/ (accessed on 21 December 2020).
- Village Water Filters Inc. Village Water Filters. Available online: https://villagewaterfilters.org/ (accessed on 21 December 2020).
- APHA; AWWA; WEF. Standard Methods for the Examination of Water and Wastewater, 21st ed.; American Public Health Association: Washington, DC, USA; American Water Works Association: Washington, DC, USA; Water Environment Federation: Washington, DC, USA, 2005. [Google Scholar]
- Gallandat, K.; Lantagne, D. Selection of a Biosafety Level 1 (BSL-1) surrogate to evaluate surface disinfection efficacy in Ebola outbreaks: Comparison of four bacteriophages. PLoS ONE 2017, 12, e0177943. [Google Scholar] [CrossRef]
- Warsinger, D.M.; Chakraborty, S.; Tow, E.W.; Plumlee, M.H.; Bellona, C.; Loutatidou, S.; Karimi, L.; Mikelonis, A.M.; Achilli, A.; Ghassemi, A.; et al. A review of polymeric membranes and processes for potable water reuse. Prog. Polym. Sci. 2018, 81, 209–237. [Google Scholar] [CrossRef]
- Gijsbertsen-Abrahamse, A.; Cornelissen, E.; Hofman, J. Fiber failure frequency and causes of hollow fiber integrity loss. Desalination 2006, 194, 251–258. [Google Scholar] [CrossRef]
- Lee, N.; Amy, G.; Croué, J.-P.; Buisson, H. Identification and understanding of fouling in low-pressure membrane (MF/UF) filtration by natural organic matter (NOM). Water Res. 2004, 38, 4511–4523. [Google Scholar] [CrossRef] [PubMed]
- Fan, L.; Harris, J.L.; Roddick, F.A.; Booker, N.A. Influence of the characteristics of natural organic matter on the fouling of microfiltration membranes. Water Res. 2001, 35, 4455–4463. [Google Scholar] [CrossRef]
- Katsoufidou, K.; Yiantsios, S.; Karabelas, A. A study of ultrafiltration membrane fouling by humic acids and flux recovery by backwashing: Experiments and modeling. J. Membr. Sci. 2005, 266, 40–50. [Google Scholar] [CrossRef]
- Khor, S.L.; Sun, D.D.; Liu, Y.; Leckie, J.O. Biofouling development and rejection enhancement in long SRT MF membrane bioreactor. Process Biochem. 2007, 42, 1641–1648. [Google Scholar] [CrossRef]
- Porcelli, N.; Judd, S. Chemical cleaning of potable water membranes: A review. Sep. Purif. Technol. 2010, 71, 137–143. [Google Scholar] [CrossRef]
- Zhao, Y.; Wu, K.; Wang, Z.; Zhao, L.; Li, S. Fouling and cleaning of membrane—A literature review. J. Environ. Sci. 2000, 12, 241–251. [Google Scholar]
- Mansouri, J.; Harrisson, S.; Chen, V. Strategies for controlling biofouling in membrane filtration systems: Challenges and opportunities. J. Mater. Chem. 2010, 20, 4567–4586. [Google Scholar] [CrossRef]
- Kimura, K.; Hane, Y.; Watanabe, Y.; Amy, G.; Ohkuma, N. Irreversible membrane fouling during ultrafiltration of surface water. Water Res. 2004, 38, 3431–3441. [Google Scholar] [CrossRef]
- Lantagne, D.; Quick, R.; Mintz, E. Household Water Treatment and Safe Storage Options in Developing Countries: A Review of Current Implementation Practices. Wilson Center. 2006. Available online: https://www.wilsoncenter.org/sites/default/files/media/documents/publication/WaterStoriesHousehold.pdf (accessed on 20 March 2021).
- Katadyn. Katadyn First Response Gravity Filter 3L—Fact Sheet. 2018. Available online: https://cdn.katadyngroup.com/assets/Downloads/katadyn/factsheets/filters/8020588_First%20Response%20Gravity%20Filter%203L_en.pdf. (accessed on 20 March 2021).


| Indicator | Definition |
|---|---|
| Strain at Break [%] | Baseline: Average strain at break for “Base Wet” fiber |
| Endline: Average strain at break for fiber at study end | |
| Break Force [mN] | Baseline: Average break force for “Base Wet” fiber |
| Endline: Average break force for fiber at study’s end | |
| Normalized Flow | Baseline: Flow measured before initial filtration run. |
| Endline: Average flow at Days 7–10 of filtration | |
| E. coli [LRV] | Baseline: Average E. coli LRV at Days 1–2 of filtration |
| Endline: Average E. coli LRV at Days 7–10 of filtration | |
| Turbidity [TRV] | Baseline: Average turbidity LRV at Days 1–2 of filtration |
| Endline: Average turbidity LRV at Days 7–10 of filtration |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Heylen, C.; Oliveira Aguiar, A.; String, G.; Domini, M.; Goff, N.; Murray, A.; Asatekin, A.; Lantagne, D. Laboratory Efficacy of Locally Available Backwashing Methods at Removing Fouling in Hollow-Fiber Membrane Filters Used for Household Water Treatment. Membranes 2021, 11, 375. https://doi.org/10.3390/membranes11050375
Heylen C, Oliveira Aguiar A, String G, Domini M, Goff N, Murray A, Asatekin A, Lantagne D. Laboratory Efficacy of Locally Available Backwashing Methods at Removing Fouling in Hollow-Fiber Membrane Filters Used for Household Water Treatment. Membranes. 2021; 11(5):375. https://doi.org/10.3390/membranes11050375
Chicago/Turabian StyleHeylen, Camille, Alice Oliveira Aguiar, Gabrielle String, Marta Domini, Nathaniel Goff, Anna Murray, Ayse Asatekin, and Daniele Lantagne. 2021. "Laboratory Efficacy of Locally Available Backwashing Methods at Removing Fouling in Hollow-Fiber Membrane Filters Used for Household Water Treatment" Membranes 11, no. 5: 375. https://doi.org/10.3390/membranes11050375
APA StyleHeylen, C., Oliveira Aguiar, A., String, G., Domini, M., Goff, N., Murray, A., Asatekin, A., & Lantagne, D. (2021). Laboratory Efficacy of Locally Available Backwashing Methods at Removing Fouling in Hollow-Fiber Membrane Filters Used for Household Water Treatment. Membranes, 11(5), 375. https://doi.org/10.3390/membranes11050375







