Polydopamine Functionalized Graphene Oxide as Membrane Nanofiller: Spectral and Structural Studies
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
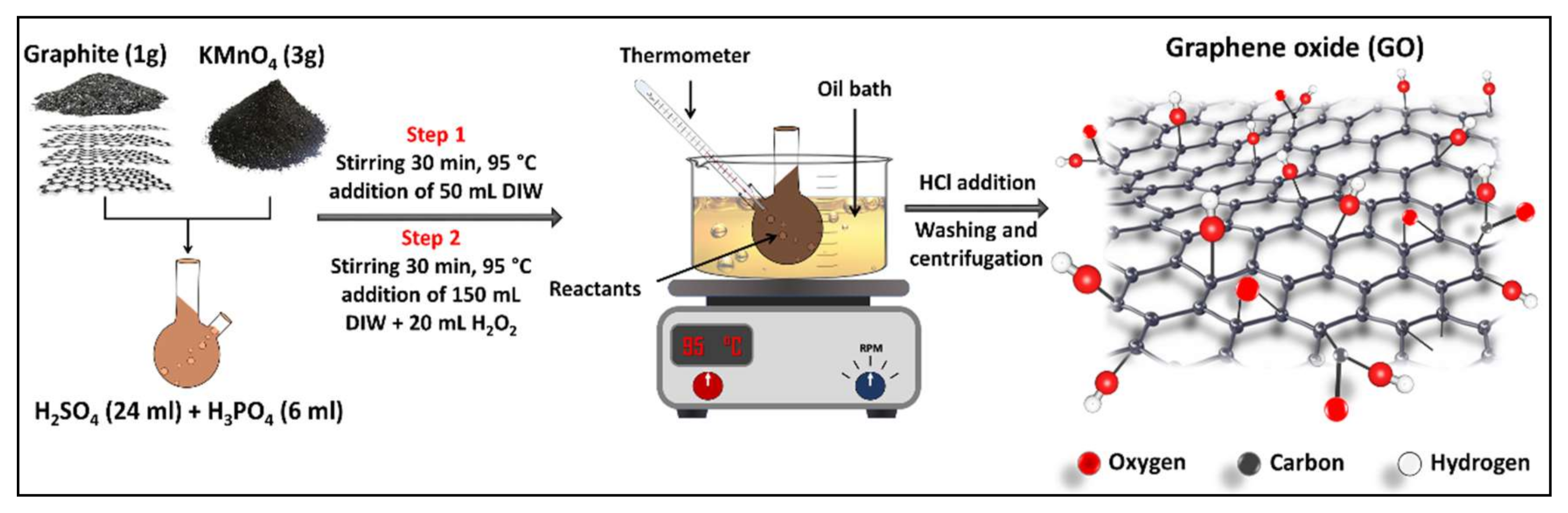
2.2. GO NPs Preparation
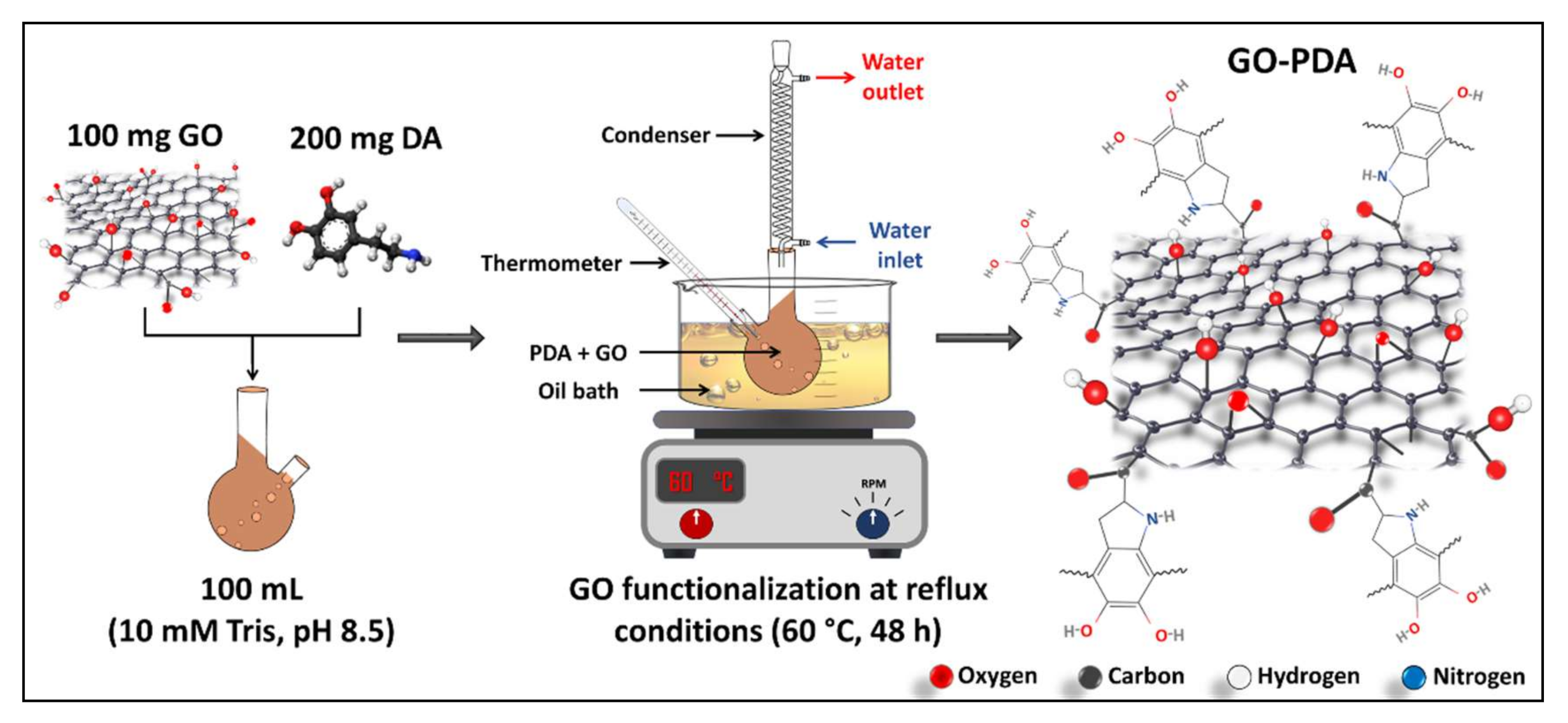
2.3. Functionalization of GO with Dopamine
2.4. Characterization of GO and GO-PDA NPs
2.5. Dispersibility and Hydrophilicity Measurements
3. Results
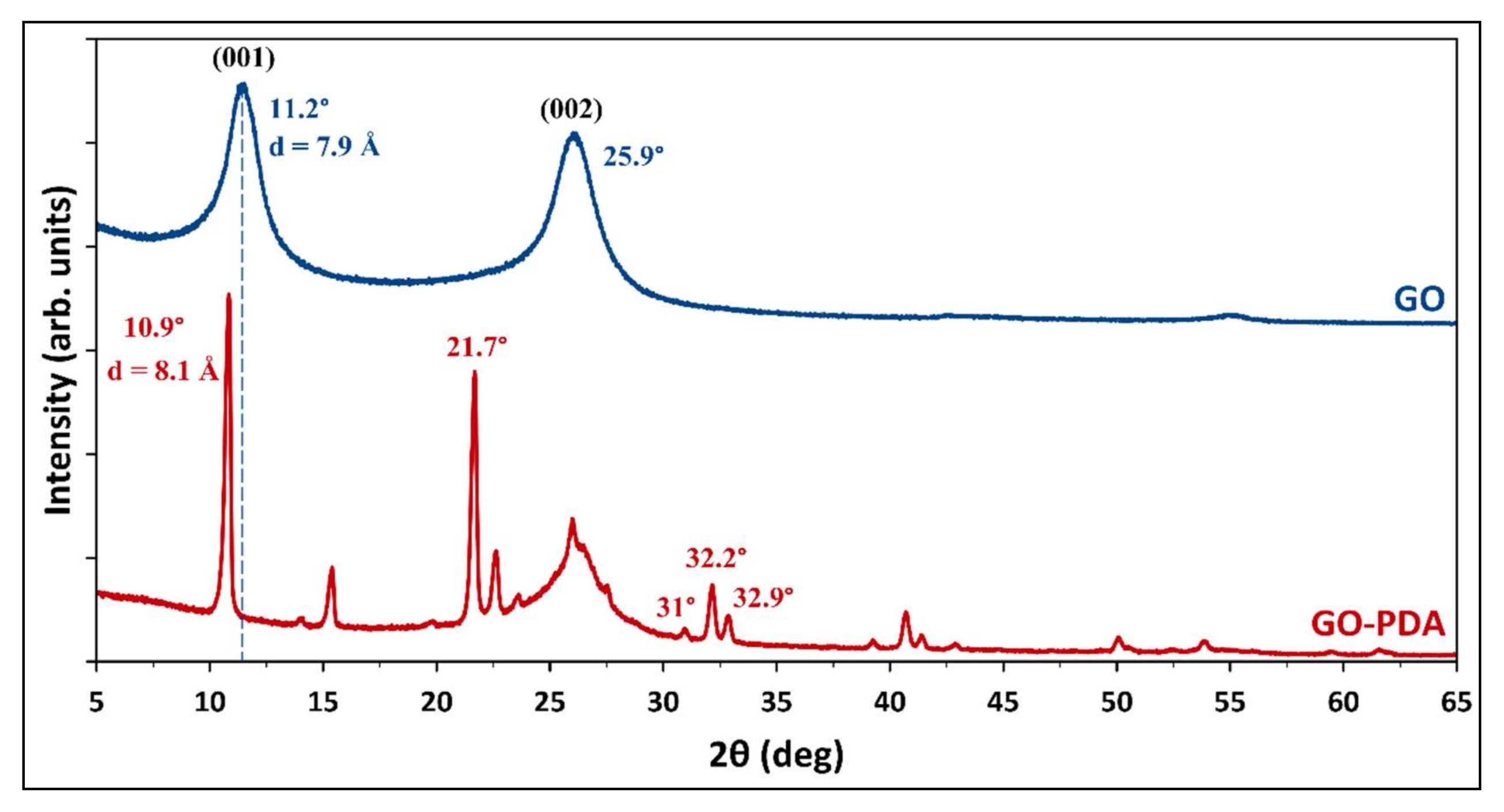
3.1. Structural Properties of GO and GO-PDA
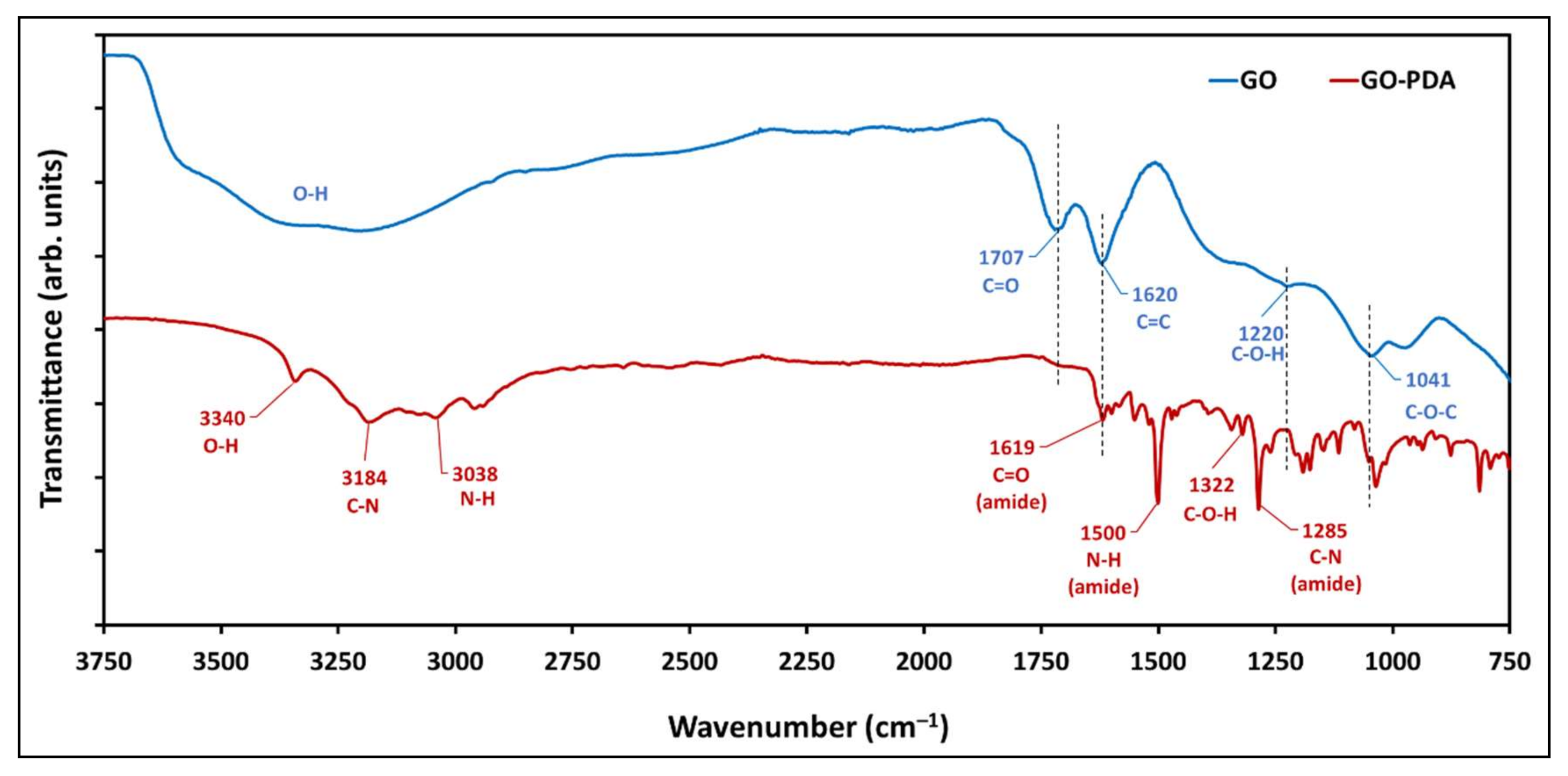
3.2. Compositional Properties of GO and GO-PDA
3.3. Dispersibility and Hydrophilicity
3.4. Thermal Stability
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Stankovich, S.; Dikin, D.A.; Dommett, G.H.B.; Kohlhaas, K.M.; Zimney, E.J.; Stach, E.A.; Piner, R.D.; Nguyen, S.T.; Ruoff, R.S. Graphene-based composite materials. Nat. Cell Biol. 2006, 442, 282–286. [Google Scholar] [CrossRef] [PubMed]
- Hummers, W.S.; Offeman, R.E. Preparation of Graphitic Oxide. J. Am. Chem. Soc. 1958, 80, 1339. [Google Scholar] [CrossRef]
- Lerf, A.; He, H.; Forster, M.; Klinowski, J. Structure of Graphite Oxide Revisited. J. Phys. Chem. B 1998, 102, 4477–4482. [Google Scholar] [CrossRef]
- Al-Gaashani, R.; Najjar, A.; Zakaria, Y.; Mansour, S.; Atieh, M. XPS and structural studies of high quality graphene oxide and reduced graphene oxide prepared by different chemical oxidation methods. Ceram. Int. 2019, 45, 14439–14448. [Google Scholar] [CrossRef]
- Muzyka, R.; Kwoka, M.; Smędowski, Ł.; Díez, N.; Gryglewicz, G. Oxidation of graphite by different modified Hummers methods. New Carbon Mater. 2017, 32, 15–20. [Google Scholar] [CrossRef]
- Chen, J.; Li, Y.; Huang, L.; Li, C.; Shi, G. High-yield preparation of graphene oxide from small graphite flakes via an improved Hummers method with a simple purification process. Carbon 2015, 81, 826–834. [Google Scholar] [CrossRef]
- Ashfaq, M.Y.; Al-Ghouti, M.A.; Da’Na, D.A.; Qiblawey, H.; Zouari, N. Investigating the effect of temperature on calcium sulfate scaling of reverse osmosis membranes using FTIR, SEM-EDX and multivariate analysis. Sci. Total Environ. 2020, 703, 134726. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Ruan, H.; Guo, C.; Liao, J.; Shen, J.; Gao, C. Thin-film nanocomposite reverse osmosis membranes with enhanced antibacterial resistance by incorporating p-aminophenol-modified graphene oxide. Sep. Purif. Technol. 2020, 234, 116017. [Google Scholar] [CrossRef]
- Xue, S.-M.; Ji, C.-H.; Xu, Z.-L.; Tang, Y.-J.; Li, R.-H. Chlorine resistant TFN nanofiltration membrane incorporated with octadecylamine-grafted GO and fluorine-containing monomer. J. Membr. Sci. 2018, 545, 185–195. [Google Scholar] [CrossRef]
- Lu, K.-J.; Zuo, J.; Chung, T.-S. Novel PVDF membranes comprising n-butylamine functionalized graphene oxide for direct contact membrane distillation. J. Membr. Sci. 2017, 539, 34–42. [Google Scholar] [CrossRef]
- Chakraborty, S.; Saha, S.; Dhanak, V.R.; Biswas, K.; Barbezat, M.; Terrasi, G.P.; Chakraborty, A.K. High yield synthesis of amine functionalized graphene oxide and its surface properties. RSC Adv. 2016, 6, 67916–67924. [Google Scholar] [CrossRef]
- Song, M.G.; Choi, J.; Jeong, H.E.; Song, K.; Jeon, S.; Cha, J.; Baeck, S.-H.; Shim, S.E.; Qian, Y. A comprehensive study of various amine-functionalized graphene oxides for room temperature formaldehyde gas detection: Experimental and theoretical approaches. Appl. Surf. Sci. 2020, 529, 147189. [Google Scholar] [CrossRef]
- Ramesh, P.; Bhagavathsingh, J. A facile synthesis of bis-(pththalimidoethyl)-amine functionalized graphene oxide and its dual performance as a supercapacitor electrode and fluorescence sensor. Mater. Chem. Phys. 2019, 222, 45–54. [Google Scholar] [CrossRef]
- Rana, S.; Jonnalagadda, S.B. Cu doped amine functionalized graphene oxide and its scope as catalyst for selective oxidation. Catal. Commun. 2017, 100, 183–186. [Google Scholar] [CrossRef]
- Liu, Y.; Sajjadi, B.; Egiebor, N.O.; Chatterjee, R. Ultrasound-assisted amine functionalized graphene oxide for enhanced CO2 adsorption. Fuel 2019, 247, 10–18. [Google Scholar] [CrossRef]
- Ebrahimi-Tazangi, F.; Hekmatara, H.; Seyed-Yazdi, J.; Hekmatara, H. Synthesis and remarkable microwave absorption properties of amine-functionalized magnetite/graphene oxide nanocomposites. J. Alloys Compd. 2019, 809, 151779. [Google Scholar] [CrossRef]
- Nakagawa, K.; Araya, S.; Kunimatsu, M.; Yoshioka, T.; Shintani, T.; Kamio, E.; Matsuyama, H. Fabrication of Stacked Graphene Oxide Nanosheet Membranes Using Triethanolamine as a Crosslinker and Mild Reducing Agent for Water Treatment. Membranes 2018, 8, 130. [Google Scholar] [CrossRef]
- Kumar, M.; Sreedhar, N.; Jaoude, M.A.; Arafat, H.A. High-Flux, Antifouling Hydrophilized Ultrafiltration Membranes with Tunable Charge Density Combining Sulfonated Poly(ether sulfone) and Aminated Graphene Oxide Nanohybrid. ACS Appl. Mater. Interfaces 2019, 12, 1617–1627. [Google Scholar] [CrossRef]
- Jin, L.; Wang, Z.; Zheng, S.; Mi, B. Polyamide-crosslinked graphene oxide membrane for forward osmosis. J. Membr. Sci. 2018, 545, 11–18. [Google Scholar] [CrossRef]
- Lee, H.; Dellatore, S.M.; Miller, W.M.; Messersmith, P.B. Mussel-Inspired Surface Chemistry for Multifunctional Coatings. Science 2007, 318, 426–430. [Google Scholar] [CrossRef]
- Wang, T.; Qiblawey, H.; Judd, S.; Benamor, A.; Nasser, M.; Mohammadian, M. Fabrication of high flux nanofiltration membrane via hydrogen bonding based co-deposition of polydopamine with poly(vinyl alcohol). J. Membr. Sci. 2018, 552, 222–233. [Google Scholar] [CrossRef]
- Wang, T.; Qiblawey, H.; Sivaniah, E.; Mohammadian, A. Novel methodology for facile fabrication of nanofiltration membranes based on nucleophilic nature of polydopamine. J. Membr. Sci. 2016, 511, 65–75. [Google Scholar] [CrossRef]
- Sun, R.; Renb, F.; Wang, N.; Yao, Y.; Fei, Z.; Wang, H.; Liu, Z.; Xing, R.; Du, Y. Polydopamine functionalized multi-walled carbon nanotubes supported PdAu nanoparticles as advanced catalysts for ethylene glycol oxidation. Colloids Surf. A Physicochem. Eng. Asp. 2019, 578, 123566. [Google Scholar] [CrossRef]
- Kumar, A.; Mishra, B.; Tripathi, B.P. Polydopamine assisted synthesis of ultrafine silver nanoparticles for heterogeneous catalysis and water remediation. Nano-Struct. Nano-Objects 2020, 23, 100489. [Google Scholar] [CrossRef]
- Cui, J.; Zhou, Z.; Xie, A.; Meng, M.; Cui, Y.; Liu, S.; Lu, J.; Zhou, S.; Yan, Y.; Dong, H. Bio-inspired fabrication of superhydrophilic nanocomposite membrane based on surface modification of SiO2 anchored by polydopamine towards effective oil-water emulsions separation. Sep. Purif. Technol. 2019, 209, 434–442. [Google Scholar] [CrossRef]
- Sun, X.; Yan, L.; Xu, R.; Xu, M.; Zhu, Y. Surface modification of TiO2 with polydopamine and its effect on photocatalytic degradation mechanism. Colloids Surf. A Physicochem. Eng. Asp. 2019, 570, 199–209. [Google Scholar] [CrossRef]
- Zhao, Z.; Guo, L.; Feng, L.; Lu, H.; Xu, Y.; Wang, J.; Xiang, B.; Zou, X. Polydopamine functionalized graphene oxide nanocomposites reinforced the corrosion protection and adhesion properties of waterborne polyurethane coatings. Eur. Polym. J. 2019, 120, 109249. [Google Scholar] [CrossRef]
- Faraji, M.; Gharibi, H.; Javaheri, M. High Pt Loading on Polydopamine Functionalized Graphene as a High Performance Cathode Electrocatalyst for Proton Exchange Membrane Fuel Cells. J. Nanostruct. 2016, 6, 156–166. [Google Scholar]
- Palanisamy, S.; Thirumalraj, B.; Chen, S.-M.; Wang, Y.-T.; Velusamy, V.; Ramaraj, S.K. A Facile Electrochemical Preparation of Reduced Graphene Oxide@Polydopamine Composite: A Novel Electrochemical Sensing Platform for Amperometric Detection of Chlorpromazine. Sci. Rep. 2016, 6, 33599. [Google Scholar] [CrossRef]
- Alkhouzaam, A.; Qiblawey, H.; Khraisheh, M.; Atieh, M.; Al-Ghouti, M. Synthesis of graphene oxides particle of high oxidation degree using a modified Hummers method. Ceram. Int. 2020, 46, 23997–24007. [Google Scholar] [CrossRef]
- Shakeri, A.; Salehi, H.; Rastgar, M. Antifouling electrically conductive membrane for forward osmosis prepared by polyaniline/graphene nanocomposite. J. Water Process. Eng. 2019, 32, 100932. [Google Scholar] [CrossRef]
- Panicker, N.J.; Das, J.; Sahu, P. Synthesis of highly oxidized graphene (HOG) by using HNO3 and KMnO4 as oxidizing agents. Mater. Today Proc. 2020. [Google Scholar] [CrossRef]
- Yadav, N.; Lochab, B. A comparative study of graphene oxide: Hummers, intermediate and improved method. FlatChem 2019, 13, 40–49. [Google Scholar] [CrossRef]
- Manwatkar, H.; Gedam, S.; Bhaskar, C.; Dhonde, M.; Thakare, S.R. Synthesis and properties of amino and thiol functionalized graphene oxide. Mater. Today Proc. 2020. [Google Scholar] [CrossRef]
- Xu, L.Q.; Yang, W.J.; Neoh, K.-G.; Kang, E.-T.; Fu, G. Dopamine-Induced Reduction and Functionalization of Graphene Oxide Nanosheets. Macromolecules 2010, 43, 8336–8339. [Google Scholar] [CrossRef]
- Lee, M.; Ku, S.H.; Ryu, J.; Park, C.B. Mussel-inspired functionalization of carbon nanotubes for hydroxyapatite mineralization. J. Mater. Chem. 2010, 20, 8848–8853. [Google Scholar] [CrossRef]
- Liu, Y.; Tu, W.; Chen, M.; Ma, L.; Yang, B.; Liang, Q.; Chen, Y. A mussel-induced method to fabricate reduced graphene oxide/halloysite nanotubes membranes for multifunctional applications in water purification and oil/water separation. Chem. Eng. J. 2018, 336, 263–277. [Google Scholar] [CrossRef]
- Marcano, D.C.; Kosynkin, D.V.; Berlin, J.M.; Sinitskii, A.; Sun, Z.; Slesarev, A.; Alemany, L.B.; Lu, W.; Tour, J.M. Improved Synthesis of Graphene Oxide. ACS Nano 2010, 4, 4806–4814. [Google Scholar] [CrossRef]
- Guerrero-Contreras, J.; Caballero-Briones, F. Graphene oxide powders with different oxidation degree, prepared by synthesis variations of the Hummers method. Mater. Chem. Phys. 2015, 153, 209–220. [Google Scholar] [CrossRef]
- Ma, Y.-R.; Zhang, X.; Zeng, T.; Cao, D.; Zhou, Z.; Li, W.-H.; Niu, H.; Cai, Y. Polydopamine-Coated Magnetic Nanoparticles for Enrichment and Direct Detection of Small Molecule Pollutants Coupled with MALDI-TOF-MS. ACS Appl. Mater. Interfaces 2013, 5, 1024–1030. [Google Scholar] [CrossRef]
- Coskun, H.; Aljabour, A.; Uiberlacker, L.; Strobel, M.; Hild, S.; Cobet, C.; Farka, D.; Stadler, P.; Sariciftci, N.S. Chemical vapor deposition-based synthesis of conductive polydopamine thin-films. Thin Solid Films 2018, 645, 320–325. [Google Scholar] [CrossRef]
- Wang, J.; Zhou, S.; Huang, J.; Zhao, G.; Liu, Y.; Wang, J.; Zhou, S.; Huang, J.; Zhao, G.; Liu, Y. Interfacial modification of basalt fiber filling composites with graphene oxide and polydopamine for enhanced mechanical and tribological properties. RSC Adv. 2018, 8, 12222–12231. [Google Scholar] [CrossRef]
- Malard, L.M.; Pimenta, M.A.; Dresselhaus, G.; Dresselhaus, M.S. Raman spectroscopy in graphene. Phys. Rep. 2009, 473, 51–87. [Google Scholar] [CrossRef]
- Lowe, S.E.; Shi, G.; Zhang, Y.; Qin, J.; Jiang, L.; Jiang, S.; Al-Mamun, M.; Liu, P.; Zhong, Y.L.; Zhao, H. The role of electrolyte acid concentration in the electrochemical exfoliation of graphite: Mechanism and synthesis of electrochemical graphene oxide. Nano Mater. Sci. 2019, 1, 215–223. [Google Scholar] [CrossRef]
- Claramunt, S.; Varea, A.; López-Díaz, D.; Velázquez, M.M.; Cornet, A.; Cirera, A. The Importance of Interbands on the Interpretation of the Raman Spectrum of Graphene Oxide. J. Phys. Chem. C 2015, 119, 10123–10129. [Google Scholar] [CrossRef]
- Sadezky, A.; Muckenhuber, H.; Grothe, H.; Niessner, R.; Pöschl, U. Raman microspectroscopy of soot and related carbonaceous materials: Spectral analysis and structural information. Carbon 2005, 43, 1731–1742. [Google Scholar] [CrossRef]
- López-Díaz, D.; Holgado, M.L.; García-Fierro, J.L.; Velázquez, M.M. Evolution of the Raman Spectrum with the Chemical Composition of Graphene Oxide. J. Phys. Chem. C 2017, 121, 20489–20497. [Google Scholar] [CrossRef]
- Tuinstra, F.; Koenig, J.L. Raman Spectrum of Graphite. J. Chem. Phys. 1970, 53, 1126–1130. [Google Scholar] [CrossRef]
- Ferrari, A.C.; Meyer, J.C.; Scardaci, V.; Casiraghi, C.; Lazzeri, M.; Mauri, F.; Piscanec, S.; Jiang, D.; Novoselov, K.S.; Roth, S.; et al. Raman Spectrum of Graphene and Graphene Layers. Phys. Rev. Lett. 2006, 97, 187401. [Google Scholar] [CrossRef]
- Zhu, Y.; Murali, S.; Cai, W.; Li, X.; Suk, J.W.; Potts, J.R.; Ruoff, R.S. Graphene and Graphene Oxide: Synthesis, Properties, and Applications. Adv. Mater. 2010, 22, 3906–3924. [Google Scholar] [CrossRef]
- Krishnamoorthy, K.; Veerapandian, M.; Yun, K.; Kim, S.-J. The chemical and structural analysis of graphene oxide with different degrees of oxidation. Carbon 2013, 53, 38–49. [Google Scholar] [CrossRef]
- Patel, K.; Singh, N.; Yadav, J.; Nayak, J.M.; Sahoo, S.K.; Lata, J.; Chand, D.; Kumar, S.; Kumar, R. Polydopamine films change their physicochemical and antimicrobial properties with a change in reaction conditions. Phys. Chem. Chem. Phys. 2018, 20, 5744–5755. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Qileng, A.; Huang, J.; Gao, Q.; Liu, Y. Polydopamine as a bridge to decorate monodisperse gold nanoparticles on Fe3O4 nanoclusters for the catalytic reduction of 4-nitrophenol. RSC Adv. 2017, 7, 45545–45551. [Google Scholar] [CrossRef]
- Liu, T.; Kim, K.C.; Lee, B.; Chen, Z.; Noda, S.; Jang, S.S.; Lee, S.W. Self-polymerized dopamine as an organic cathode for Li- and Na-ion batteries. Energy Environ. Sci. 2017, 10, 205–215. [Google Scholar] [CrossRef]
- Li, X.; Shan, H.; Cao, M.; Li, B.; Xipeng, L.; Huiting, S.; Min, C.; Baoan, L. Mussel-inspired modification of PTFE membranes in a miscible THF-Tris buffer mixture for oil-in-water emulsions separation. J. Membr. Sci. 2018, 555, 237–249. [Google Scholar] [CrossRef]
- Tobi, A.R.; Dennis, J.; Zaid, H.; Adekoya, A.; Yar, A.; Fahad, U. Comparative analysis of physiochemical properties of physically activated carbon from palm bio-waste. J. Mater. Res. Technol. 2019, 8, 3688–3695. [Google Scholar] [CrossRef]
- Plata, D.L.; Gschwend, P.M.; Reddy, C.M. Industrially synthesized single-walled carbon nanotubes: Compositional data for users, environmental risk assessments, and source apportionment. Nanotechnology 2008, 19, 185706. [Google Scholar] [CrossRef]
- Braun, E.I.; Pantano, P. The importance of an extensive elemental analysis of single-walled carbon nanotube soot. Carbon 2014, 77, 912–919. [Google Scholar] [CrossRef]
- Pumera, M. Carbon Nanotubes Contain Residual Metal Catalyst Nanoparticles even after Washing with Nitric Acid at Elevated Temperature Because These Metal Nanoparticles Are Sheathed by Several Graphene Sheets. Langmuir 2007, 23, 6453–6458. [Google Scholar] [CrossRef]
- Ren, P.-G.; Wang, H.; Huang, H.-D.; Yan, D.-X.; Li, Z.-M. Characterization and performance of dodecyl amine functionalized graphene oxide and dodecyl amine functionalized graphene/high-density polyethylene nanocomposites: A comparative study. J. Appl. Polym. Sci. 2013, 131. [Google Scholar] [CrossRef]
- Ajdari, F.B.; Kowsari, E.; Ehsani, A.; Chepyga, L.; Schirowski, M.; Jäger, S.; Kasian, O.; Hauke, F.; Ameri, T. Melamine-functionalized graphene oxide: Synthesis, characterization and considering as pseudocapacitor electrode material with intermixed POAP polymer. Appl. Surf. Sci. 2018, 459, 874–883. [Google Scholar] [CrossRef]
- Du, W.; Wu, H.; Chen, H.; Xu, G.; Li, C. Graphene oxide in aqueous and nonaqueous media: Dispersion behaviour and solution chemistry. Carbon 2020, 158, 568–579. [Google Scholar] [CrossRef]
- Shao, W.; Liu, C.; Ma, H.; Hong, Z.; Xie, Q.; Lu, Y. Fabrication of pH-sensitive thin-film nanocomposite nanofiltration membranes with enhanced performance by incorporating amine-functionalized graphene oxide. Appl. Surf. Sci. 2019, 487, 1209–1221. [Google Scholar] [CrossRef]
- Jing, L.-C.; Wang, T.; Cao, W.-W.; Wen, J.-G.; Zhao, H.; Ning, Y.-J.; Yuan, X.-T.; Tian, Y.; Teng, L.-H.; Geng, H. Water-based polyurethane composite anticorrosive barrier coating via enhanced dispersion of functionalized graphene oxide in the presence of acidified multi-walled carbon nanotubes. Prog. Org. Coat. 2020, 146, 105734. [Google Scholar] [CrossRef]
- Konios, D.; Stylianakis, M.M.; Stratakis, E.; Kymakis, E. Dispersion behaviour of graphene oxide and reduced graphene oxide. J. Colloid Interface Sci. 2014, 430, 108–112. [Google Scholar] [CrossRef]
- Aliabadian, E.; Sadeghi, S.; Moghaddam, A.R.; Maini, B.B.; Chen, Z.; Sundararaj, U. Application of graphene oxide nanosheets and HPAM aqueous dispersion for improving heavy oil recovery: Effect of localized functionalization. Fuel 2020, 265, 116918. [Google Scholar] [CrossRef]
- Chang, X.; Wang, Z.; Quan, S.; Xu, Y.; Jiang, Z.; Shao, L. Exploring the synergetic effects of graphene oxide (GO) and polyvinylpyrrodione (PVP) on poly(vinylylidenefluoride) (PVDF) ultrafiltration membrane performance. Appl. Surf. Sci. 2014, 316, 537–548. [Google Scholar] [CrossRef]
- Jiang, Y.; Zeng, Q.; Biswas, P.; Fortner, J. Graphene oxides as nanofillers in polysulfone ultrafiltration membranes: Shape matters. J. Membr. Sci. 2019, 581, 453–461. [Google Scholar] [CrossRef]
- Alkhouzaam, A.; Qiblawey, H. Novel polysulfone ultrafiltration membranes incorporating polydopamine functionalized graphene oxide with enhanced flux and fouling resistance. J. Membr. Sci. 2020, 118900. [Google Scholar] [CrossRef]
- Alkhouzaam, A.; Qiblawey, H.; Khraisheh, M. Synthesis of High-Antifouling and Antibacterial Ultrafiltration Membranes Incorporating Low Concentrations of Graphene Oxide. In Qatar University Annual Research Forum and Exhibition (QUARFE 2020); Qatar University Press: Doha, Qatar, 2020. [Google Scholar] [CrossRef]
- Naeem, H.; Ajmal, M.; Qureshi, R.B.; Muntha, S.T.; Farooq, M.; Siddiq, M. Facile synthesis of graphene oxide–silver nanocomposite for decontamination of water from multiple pollutants by adsorption, catalysis and antibacterial activity. J. Environ. Manag. 2019, 230, 199–211. [Google Scholar] [CrossRef]
- Talyzin, A.V.; Mercier, G.; Klechikov, A.; Hedenström, M.; Johnels, D.; Wei, D.; Cotton, D.; Opitz, A.; Moons, E. Brodie vs Hummers graphite oxides for preparation of multi-layered materials. Carbon 2017, 115, 430–440. [Google Scholar] [CrossRef]








| Aminated-GO | Application | Ref. |
|---|---|---|
| GO-p-aminophenol | RO desalination | [8] |
| GO-ODA | NF desalination | [9] |
| GO-TEOA a | NF desalination | [17] |
| GO-APTS | UF (Natural organic matter removal) | [18] |
| GO-ButA | Membrane distillation (MD) | [10] |
| GO-MXDA b | FO desalination | [19] |
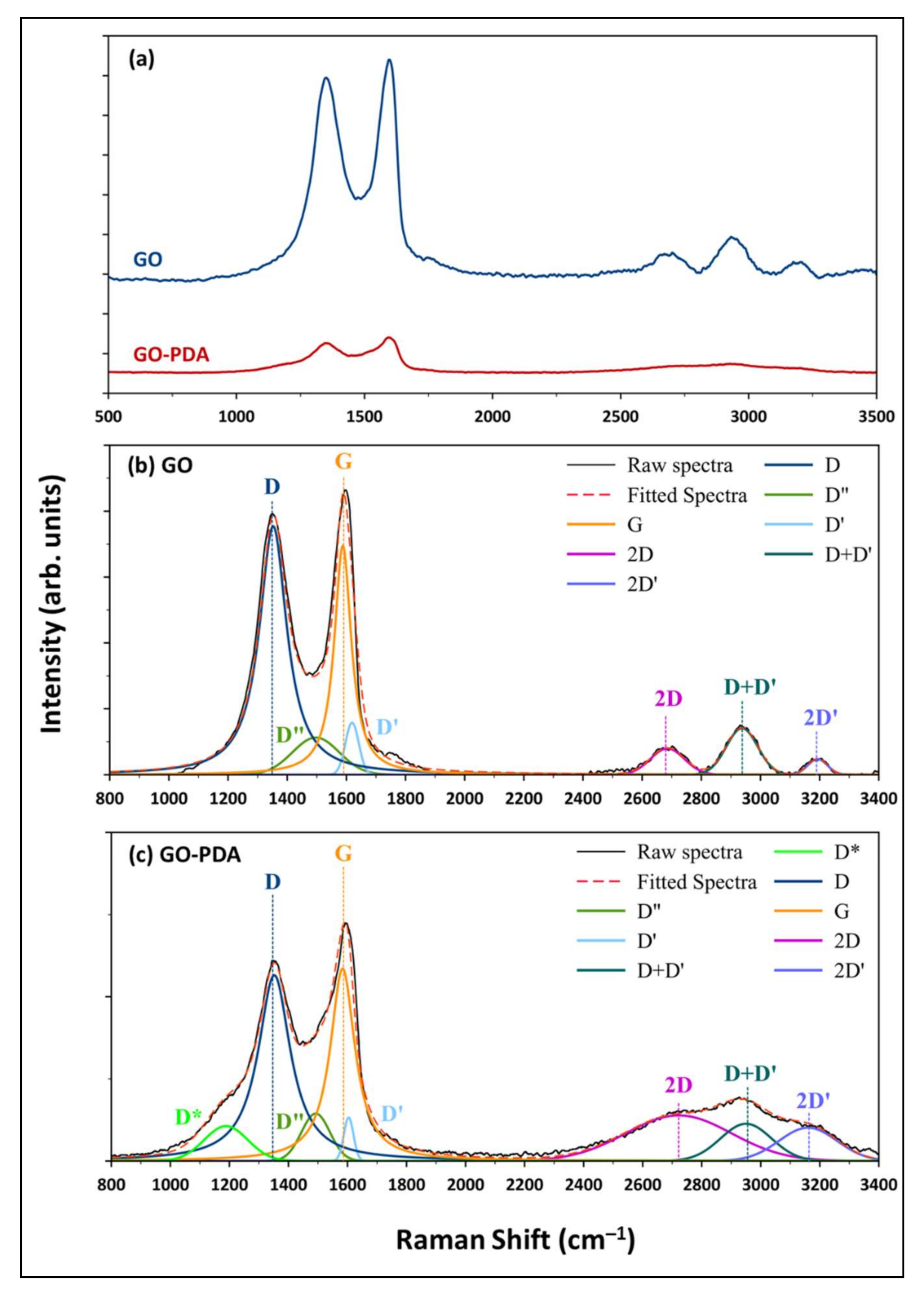
| GO Sample | Curve | Peak Center (cm−1) | Peak Area (arb. unit) | ID/IG | La |
|---|---|---|---|---|---|
| GO | D | 1352 | 334053 | 1.8 | 10.9 |
| G | 1588 | 189857 | |||
| GO-PDA | D | 1352 | 59039 | 1.3 | 15.2 |
| G | 1583 | 46638 |
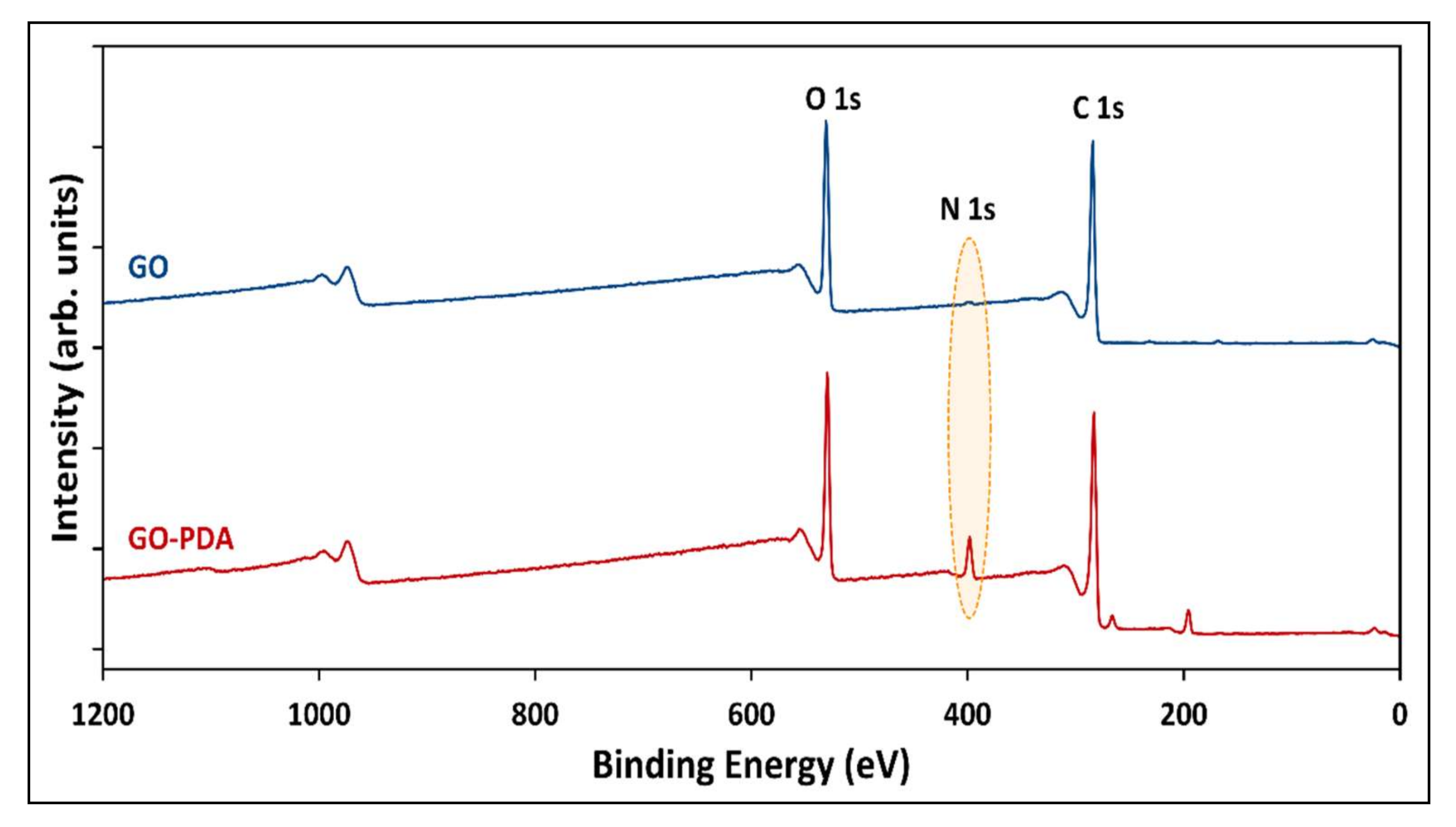
| Sample | Atomic Composition (at.%) | |||||
|---|---|---|---|---|---|---|
| C 1s | O 1s | N 1s | S 2p | Cl 2p | Cl 2s | |
| GO | 74.58 | 24.52 | 0.5 | 0.4 | - | - |
| GO-PDA | 69.94 | 20.61 | 6.61 | 0.11 | 1.82 | 0.91 |
| Sample | Wt.% | At.% | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| N | C | H | S | O | N | C | H | S | O | O/C | |
| GO | 0.3 | 46.8 | 2.6 | 0.3 | 50.0 | 0.2 | 40.3 | 27.1 | 0.1 | 32.3 | 0.80 |
| GO-PDA | 7.8 | 49.8 | 4.8 | 0.0 | 37.7 | 4.7 | 35.2 | 40.1 | 0.0 | 20.0 | 0.6 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alkhouzaam, A.; Qiblawey, H.; Khraisheh, M. Polydopamine Functionalized Graphene Oxide as Membrane Nanofiller: Spectral and Structural Studies. Membranes 2021, 11, 86. https://doi.org/10.3390/membranes11020086
Alkhouzaam A, Qiblawey H, Khraisheh M. Polydopamine Functionalized Graphene Oxide as Membrane Nanofiller: Spectral and Structural Studies. Membranes. 2021; 11(2):86. https://doi.org/10.3390/membranes11020086
Chicago/Turabian StyleAlkhouzaam, Abedalkader, Hazim Qiblawey, and Majeda Khraisheh. 2021. "Polydopamine Functionalized Graphene Oxide as Membrane Nanofiller: Spectral and Structural Studies" Membranes 11, no. 2: 86. https://doi.org/10.3390/membranes11020086
APA StyleAlkhouzaam, A., Qiblawey, H., & Khraisheh, M. (2021). Polydopamine Functionalized Graphene Oxide as Membrane Nanofiller: Spectral and Structural Studies. Membranes, 11(2), 86. https://doi.org/10.3390/membranes11020086








