Electrospun Antibacterial Nanomaterials for Wound Dressings Applications
Abstract
1. Introduction
2. Electrospinning Process (Parameters and Biomedical Applications)
3. Antibacterial Nanofibers for Wound Dressing
- Mimicry of the composition.
- Mimicry of the structure.
- Incorporation of bioactive materials.
- Mechanical mimicry.
- Regulation of the skin cell response [48].
3.1. Mimicry of the Composition
3.2. Mimicry of the Structure
3.3. Incorporation of Bioactive Materials
3.4. Mechanical Mimicry
3.5. Regulation of the Skin Cell Response
4. Biopolymeric Nanofibrous Antibacterial Wound Dressings
- -
- Low antigenicity and inherent biocompatibility.
- -
- Increase in fibroblast production and permeation.
- -
- Helps to preserve leukocytes, macrophages, fibroblasts, and epithelial cells.
- -
- Attracts fibroblasts and encourages the deposition of new collagen to the wound bed.
5. Nanoparticle Containing Nanocomposite Antibacterial Nanofibers
6. Biofunctionalized Antibacterial Nanofibers
7. Conclusions
- Nontoxic to mammal cells.
- Nonantigenic.
- Good mechanical resistance.
- Elastic and flexible.
- Antibacterial.
- Permeable for gas exchange.
- Inexpensive.
- Long shelf-life.
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Boer, M.; Duchnik, E.; Maleszka, R.; Marchlewicz, M. Structural and Biophysical Characteristics of Human Skin in Maintaining Proper Epidermal Barrier Function. Postepy Dermatol. Alergol. 2016, 33, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Wysocki, A.B. Skin Anatomy, Physiology, and Pathophysiology. Nurs. Clin. N. Am. 1999, 34, 777–797. [Google Scholar]
- Kolarsick, P.A.J.; Kolarsick, M.A.; Goodwin, C. Anatomy and Physiology of the Skin. J. Dermatol. Nurses’ Assoc. 2011, 3, 203–213. [Google Scholar] [CrossRef]
- Chrintz, H.; Vibits, H.; Cordtz, T.O.; Harreby, J.S.; Waaddegaard, P.; Larsen, S.O. Need for Surgical Wound Dressing. Br. J. Surg. 1989, 76, 204–205. [Google Scholar] [CrossRef]
- Simões, D.; Miguel, S.P.; Ribeiro, M.P.; Coutinho, P.; Mendonça, A.G.; Correia, I.J. Recent Advances on Antimicrobial Wound Dressing: A Review. Eur. J. Pharm. Biopharm. 2018, 127, 130–141. [Google Scholar] [CrossRef]
- Chen, K.; Wang, F.; Liu, S.; Wu, X.; Xu, L.; Zhang, D. In Situ Reduction of Silver Nanoparticles by Sodium Alginate to Obtain Silver-Loaded Composite Wound Dressing with Enhanced Mechanical and Antimicrobial Property. Int. J. Biol. Macromol. 2020, 148, 501–509. [Google Scholar] [CrossRef]
- Wang, S.; Wang, Z.; Xu, C.; Cui, L.; Meng, G.; Yang, S.; Wu, J.; Liu, Z.; Guo, X. PEG-α-CD/AM/Liposome @amoxicillin Double Network Hydrogel Wound Dressing—Multiple Barriers for Long-Term Drug Release. J. Biomater. Appl. 2021, 35, 1085–1095. [Google Scholar] [CrossRef]
- Li, H.; Wei, X.; Yi, X.; Tang, S.; He, J.; Huang, Y.; Cheng, F. Antibacterial, Hemostasis, Adhesive, Self-Healing Polysaccharides-Based Composite Hydrogel Wound Dressing for the Prevention and Treatment of Postoperative Adhesion. Mater. Sci. Eng. C 2021, 123, 111978. [Google Scholar] [CrossRef]
- Zhang, J.; Fang, W.; Zhang, F.; Gao, S.; Guo, Y.; Li, J.; Zhu, Y.; Zhang, Y.; Jin, J. Ultrathin Microporous Membrane with High Oil Intrusion Pressure for Effective Oil/Water Separation. J. Membr. Sci. 2020, 608, 118201. [Google Scholar] [CrossRef]
- Li, D.; Fei, X.; Wang, K.; Xu, L.; Wang, Y.; Tian, J.; Li, Y. A Novel Self-Healing Triple Physical Cross-Linked Hydrogel for Antibacterial Dressing. J. Mater. Chem. B 2021, 9, 6844–6855. [Google Scholar] [CrossRef]
- Shalumon, K.T.; Sheu, C.; Chen, C.-H.; Chen, S.-H.; Jose, G.; Kuo, C.-Y.; Chen, J.-P. Multi-Functional Electrospun Antibacterial Core-Shell Nanofibrous Membranes for Prolonged Prevention of Post-Surgical Tendon Adhesion and Inflammation. Acta Biomater. 2018, 72, 121–136. [Google Scholar] [CrossRef]
- Pan, H.; Fan, D.; Duan, Z.; Zhu, C.; Fu, R.; Li, X. Non-Stick Hemostasis Hydrogels as Dressings with Bacterial Barrier Activity for Cutaneous Wound Healing. Mater. Sci. Eng. C 2019, 105, 110118. [Google Scholar] [CrossRef]
- Alberti, T.; Coelho, D.S.; Voytena, A.; Pitz, H.; de Pra, M.; Mazzarino, L.; Kuhnen, S.; Ribeiro-do-Valle, R.M.; Maraschin, M.; Veleirinho, B. Nanotechnology: A Promising Tool towards Wound Healing. Curr. Pharm. Des. 2017, 23, 3515–3528. [Google Scholar] [CrossRef]
- Newman, M.D.; Stotland, M.; Ellis, J.I. The Safety of Nanosized Particles in Titanium Dioxide–and Zinc Oxide–Based Sunscreens. J. Am. Acad. Dermatol. 2009, 61, 685–692. [Google Scholar] [CrossRef]
- Kalashnikova, I.; Das, S.; Seal, S. Nanomaterials for Wound Healing: Scope and Advancement. Nanomedicine 2015, 10, 2593–2612. [Google Scholar] [CrossRef]
- Hubbell, J.A. Chapter 21—Matrix Effects. In Principles of Tissue Engineering, 4th ed.; Lanza, R., Langer, R., Vacanti, J., Eds.; Academic Press: Boston, MA, USA, 2014; pp. 407–421. ISBN 978-0-12-398358-9. [Google Scholar]
- Rosenberg, M.D. Cell Guidance by Alterations in Monomolecular Films. Science 1963, 139, 411–412. [Google Scholar] [CrossRef]
- Laurencin, C.T.; Ambrosio, A.M.A.; Borden, M.D.; Cooper, J.A., Jr. Tissue Engineering: Orthopedic Applications. Annu. Rev. Biomed. Eng. 1999, 1, 19–46. [Google Scholar] [CrossRef]
- Tanzli, E.; Ehrmann, A. Electrospun Nanofibrous Membranes for Tissue Engineering and Cell Growth. Appl. Sci. 2021, 11, 6929. [Google Scholar] [CrossRef]
- Younes, P.-S.; Mehdi, D.; Abbas, M.; Amir, F.; Roghayeh, S.; Nosratollah, Z. An Overview on Application of Natural Substances Incorporated with Electrospun Nanofibrous Scaffolds to Development of Innovative Wound Dressings. Mini-Rev. Med. Chem. 2018, 18, 414–427. [Google Scholar]
- Tiyek, I.; Gunduz, A.; Yalcinkaya, F.; Chaloupek, J. Influence of Electrospinning Parameters on the Hydrophilicity of Electrospun Polycaprolactone Nanofibres. J. Nanosci. Nanotechnol. 2019, 19, 7251–7260. [Google Scholar] [CrossRef]
- Abrigo, M.; McArthur, S.L.; Kingshott, P. Electrospun Nanofibers as Dressings for Chronic Wound Care: Advances, Challenges, and Future Prospects. Macromol. Biosci. 2014, 14, 772–792. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Li, T.; Han, Y.; Li, F.; Liu, Y. Recent Development of Electrospun Wound Dressing. Curr. Opin. Biomed. Eng. 2021, 17, 100247. [Google Scholar] [CrossRef]
- Dodero, A.; Brunengo, E.; Alloisio, M.; Sionkowska, A.; Vicini, S.; Castellano, M. Chitosan-Based Electrospun Membranes: Effects of Solution Viscosity, Coagulant and Crosslinker. Carbohydr. Polym. 2020, 235, 115976. [Google Scholar] [CrossRef] [PubMed]
- Olkhov, A.A.; Staroverova, O.V.; Kuherenko, E.L.; Iordanskii, A.L. Effect of Electrospinning Solution Parameters on the Properties of Nonvolven Fibrous Material Based on Polyhydroxibutyrate. J. Phys. Conf. Ser. 2020, 1431, 012029. [Google Scholar] [CrossRef]
- Fallahi, D.; Rafizadeh, M.; Mohammadi, N.; Vahidi, B. Effects of Feed Rate and Solution Conductivity on Jet Current and Fiber Diameter in Electrospinning of Polyacrylonitrile Solutions. E-Polymers 2009, 9. [Google Scholar] [CrossRef]
- Angammana, C.; Jayaram, S. Analysis of the Effects of Solution Conductivity on Electrospinning Process and Fiber Morphology. IEEE Trans. Ind. Appl. 2011, 47, 1109–1117. [Google Scholar] [CrossRef]
- Afifi, A.M.; Yamane, H.; Kimura, Y. Effect of Polymer Molecular Weight on the Electrospinning of Polylactides in Entangled and Aligned Fiber Forms. Sen’i Gakkaishi 2010, 66, 35–42. [Google Scholar] [CrossRef][Green Version]
- Park, B.K.; Um, I.C. Effect of Molecular Weight on Electro-Spinning Performance of Regenerated Silk. Int. J. Biol. Macromol. 2018, 106, 1166–1172. [Google Scholar] [CrossRef]
- Liu, Y.; Dong, L.; Fan, J.; Wang, R.; Yu, J.-Y. Effect of Applied Voltage on Diameter and Morphology of Ultrafine Fibers in Bubble Electrospinning. J. Appl. Polym. Sci. 2011, 120, 592–598. [Google Scholar] [CrossRef]
- Yang, G.Z.; Li, H.P.; Yang, J.H.; Wan, J.; Yu, D.G. Influence of Working Temperature on the Formation of Electrospun Polymer Nanofibers. Nanoscale Res. Lett. 2017, 12, 1–10. [Google Scholar] [CrossRef]
- Levitt, A.; Vallett, R.; Dion, G.; Schauer, C. Effect of Electrospinning Processing Variables on Polyacrylonitrile Nanoyarns. J. Appl. Polym. Sci. 2018, 135, 46404. [Google Scholar] [CrossRef]
- Nezarati, R.M.; Eifert, M.B.; Cosgriff-Hernandez, E. Effects of Humidity and Solution Viscosity on Electrospun Fiber Morphology. Tissue Eng. Part C Methods 2013, 19, 810–819. [Google Scholar] [CrossRef]
- Baykara, T.; Taylan, G. Coaxial Electrospinning of PVA/Nigella Seed Oil Nanofibers: Processing and Morphological Characterization. Mater. Sci. Eng. B 2021, 265, 115012. [Google Scholar] [CrossRef]
- Daenicke, J.; Lämmlein, M.; Steinhübl, F.; Schubert, D.W. Revealing Key Parameters to Minimize the Diameter of Polypropylene Fibers Produced in the Melt Electrospinning Process. E-Polymers 2019, 19, 330–340. [Google Scholar] [CrossRef]
- Deitzel, J.M.; Kleinmeyer, J.; Harris, D.; Beck Tan, N.C. The Effect of Processing Variables on the Morphology of Electrospun Nanofibers and Textiles. Polymer 2001, 42, 261–272. [Google Scholar] [CrossRef]
- Geng, X.; Kwon, O.-H.; Jang, J. Electrospinning of Chitosan Dissolved in Concentrated Acetic Acid Solution. Biomaterials 2005, 26, 5427–5432. [Google Scholar] [CrossRef]
- Thompson, C.J.; Chase, G.G.; Yarin, A.L.; Reneker, D.H. Effects of Parameters on Nanofiber Diameter Determined from Electrospinning Model. Polymer 2007, 48, 6913–6922. [Google Scholar] [CrossRef]
- Beachley, V.; Wen, X. Effect of Electrospinning Parameters on the Nanofiber Diameter and Length. Mater. Sci. Eng. C Mater. Biol. Appl. 2009, 29, 663–668. [Google Scholar] [CrossRef]
- Jabur, A.R.; Aldain, S.M.M. Effects of Ambient Temperature and Needle to Collector Distance on PVA Nanofibers Diameter Obtained from Electrospinning Technique. Available online: https://www.semanticscholar.org/paper/Effects-of-Ambient-Temperature-and-Needle-to-on-PVA-Jabur-Aldain/0104988ab6c79c6e95c15d1b3ea589858caa1ed7 (accessed on 18 October 2021).
- Zhang, S.; Liu, H.; Tang, N.; Yu, J.; Ding, B. Chapter 8—Electronetting. In Electrospinning: Nanofabrication and Applications; Ding, B., Wang, X., Yu, J., Eds.; Micro and Nano Technologies; William Andrew Publishing: Norwich, NY, USA, 2019; pp. 249–282. ISBN 978-0-323-51270-1. [Google Scholar]
- De Vrieze, S.; Van Camp, T.; Nelvig, A.; Hagström, B.; Westbroek, P.; De Clerck, K. The Effect of Temperature and Humidity on Electrospinning. J. Mater. Sci. 2009, 44, 1357–1362. [Google Scholar] [CrossRef]
- Karaman, O.; Şen, M.; Demirci, E.A. 11—Electrospun scaffolds for vascular tissue engineering. In Electrospun Materials for Tissue Engineering and Biomedical Applications; Uyar, T., Kny, E., Eds.; Woodhead Publishing: Sawston, UK, 2017; pp. 261–287. ISBN 978-0-08-101022-8. [Google Scholar]
- Torres-Martínez, E.J.; Bravo, J.M.C.; Medina, A.S.; González, G.L.P.; Gómez, L.J.V. A Summary of Electrospun Nanofibers as Drug Delivery System: Drugs Loaded and Biopolymers Used as Matrices. Curr. Drug Deliv. 2018, 15, 1360–1374. [Google Scholar] [CrossRef]
- Eren Boncu, T.; Ozdemir, N.; Uskudar Guclu, A. Electrospinning of Linezolid Loaded PLGA Nanofibers: Effect of Solvents on Its Spinnability, Drug Delivery, Mechanical Properties, and Antibacterial Activities. Drug Dev. Ind. Pharm. 2020, 46, 109–121. [Google Scholar] [CrossRef] [PubMed]
- Khalf, A.; Madihally, S.V. Recent Advances in Multiaxial Electrospinning for Drug Delivery. Eur. J. Pharm. Biopharm. 2017, 112, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Akhmetova, A.; Heinz, A. Electrospinning Proteins for Wound Healing Purposes: Opportunities and Challenges. Pharmaceutics 2021, 13, 4. [Google Scholar] [CrossRef] [PubMed]
- Bjarnsholt, T. The role of bacterial biofilms in chronic infections. Apmis 2013, 121, 1–58. [Google Scholar] [CrossRef]
- Wang, L.; Hu, C.; Shao, L. The Antimicrobial Activity of Nanoparticles: Present Situation and Prospects for the Future. Int. J. Nanomed. 2017, 12, 1227–1249. [Google Scholar] [CrossRef]
- Inzana, J.A.; Schwarz, E.M.; Kates, S.L.; Awad, H.A. Biomaterials Approaches to Treating Implant-Associated Osteomyelitis. Biomaterials 2016, 81, 58–71. [Google Scholar] [CrossRef]
- Tan, X.W.; Goh, T.W.; Saraswathi, P.; Nyein, C.L.; Setiawan, M.; Riau, A.; Lakshminarayanan, R.; Liu, S.; Tan, D.; Beuerman, R.W.; et al. Effectiveness of Antimicrobial Peptide Immobilization for Preventing Perioperative Cornea Implant-Associated Bacterial Infection. Antimicrob. Agents Chemother. 2014, 58, 5229–5238. [Google Scholar] [CrossRef]
- Wei, Z.; Wang, L.; Zhang, S.; Chen, T.; Yang, J.; Long, S.; Wang, X. Electrospun Antibacterial Nanofibers for Wound Dressings and Tissue Medicinal Fields: A Review. J. Innov. Opt. Health Sci. 2020, 13, 2030012. [Google Scholar] [CrossRef]
- Ragelle, H.; Danhier, F.; Préat, V.; Langer, R.; Anderson, D.G. Nanoparticle-Based Drug Delivery Systems: A Commercial and Regulatory Outlook as the Field Matures. Expert Opin. Drug Deliv. 2017, 14, 851–864. [Google Scholar] [CrossRef]
- Huang, Z.-M.; Zhang, Y.-Z.; Kotaki, M.; Ramakrishna, S. A Review on Polymer Nanofibers by Electrospinning and Their Applications in Nanocomposites. Compos. Sci. Technol. 2003, 63, 2223–2253. [Google Scholar] [CrossRef]
- Jones, J.R. Review of Bioactive Glass: From Hench to Hybrids. Acta Biomater. 2013, 9, 4457–4486. [Google Scholar] [CrossRef]
- Reneker, D.H.; Yarin, A.L.; Zussman, E.; Xu, H. Electrospinning of Nanofibers from Polymer Solutions and Melts. In Advances in Applied Mechanics; Elsevier: Amsterdam, The Netherlands, 2007; Volume 41, pp. 43–346. ISBN 978-0-12-002057-7. [Google Scholar]
- Liu, Z.-Y.; Wei, Z.-M.; Wang, X.-J.; Zhang, G.; Long, S.-R.; Yang, J. Preparation and Characterization of Multi-Layer Poly (Arylene Sulfide Sulfone) Nanofibers Membranes for Liquid Filtration. Chin. J. Polym. Sci. 2019, 37, 1248–1256. [Google Scholar] [CrossRef]
- Wehlage, D.; Blattner, H.; Sabantina, L.; Böttjer, R.; Grothe, T.; Rattenholl, A.; Gudermann, F.; Lütkemeyer, D.; Ehrmann, A. Sterilization of PAN/Gelatin Nanofibrous Mats for Cell Growth. Tekstilec 2019, 62, 78–88. [Google Scholar] [CrossRef]
- Percival, N.J. Classification of Wounds and Their Management. Surgery 2002, 20, 114–117. [Google Scholar] [CrossRef]
- Chen, S.; Zhang, M.; Shao, X.; Wang, X.; Zhang, L.; Xu, P.; Zhong, W.; Zhang, L.; Xing, M.; Zhang, L. A Laminin Mimetic Peptide SIKVAV-Conjugated Chitosan Hydrogel Promoting Wound Healing by Enhancing Angiogenesis, Re-Epithelialization and Collagen Deposition. J. Mater. Chem. B 2015, 3, 6798–6804. [Google Scholar] [CrossRef]
- Kuna, V.K.; Padma, A.M.; Håkansson, J.; Nygren, J.; Sjöback, R.; Petronis, S.; Sumitran-Holgersson, S. Significantly Accelerated Wound Healing of Full-Thickness Skin Using a Novel Composite Gel of Porcine Acellular Dermal Matrix and Human Peripheral Blood Cells. Cell Transplant. 2017, 26, 293–307. [Google Scholar] [CrossRef]
- Murali, R.; Thanikaivelan, P. Bionic, Porous, Functionalized Hybrid Scaffolds with Vascular Endothelial Growth Factor Promote Rapid Wound Healing in Wistar Albino Rats. RSC Adv. 2016, 6, 19252–19264. [Google Scholar] [CrossRef]
- Meyer, U.; Handschel, J.; Wiesmann, H.P.; Meyer, T. (Eds.) Fundamentals of Tissue Engineering and Regenerative Medicine; Springer: Berlin/Heidelberg, Germany, 2009; ISBN 978-3-540-77754-0. [Google Scholar]
- Greiner, A.; Wendorff, J.H. Electrospinning: A Fascinating Method for the Preparation of Ultrathin Fibers. Angew. Chem. Int. Ed. 2007, 46, 5670–5703. [Google Scholar] [CrossRef]
- Schultz, G.; Ladwig, G.; Wysocki, A. Extracellular Matrix: Review of Its Roles in Acute and Chronic Wounds. World Wide Wounds 2005, 2005, 1–18. [Google Scholar]
- Matthews, J.A.; Wnek, G.E.; Simpson, D.G.; Bowlin, G.L. Electrospinning of Collagen Nanofibers. Biomacromolecules 2002, 3, 232–238. [Google Scholar] [CrossRef]
- Rho, K.S.; Jeong, L.; Lee, G.; Seo, B.-M.; Park, Y.J.; Hong, S.-D.; Roh, S.; Cho, J.J.; Park, W.H.; Min, B.-M. Electrospinning of Collagen Nanofibers: Effects on the Behavior of Normal Human Keratinocytes and Early-Stage Wound Healing. Biomaterials 2006, 27, 1452–1461. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.-P.; Chang, G.-Y.; Chen, J.-K. Electrospun Collagen/Chitosan Nanofibrous Membrane as Wound Dressing. Colloids Surf. A Physicochem. Eng. Asp. 2008, 313–314, 183–188. [Google Scholar] [CrossRef]
- Venugopal, J.R.; Zhang, Y.; Ramakrishna, S. In Vitro Culture of Human Dermal Fibroblasts on Electrospun Polycaprolactone Collagen Nanofibrous Membrane. Artif. Organs 2006, 30, 440–446. [Google Scholar] [CrossRef] [PubMed]
- Lin, J.; Li, C.; Zhao, Y.; Hu, J.; Zhang, L.-M. Co-Electrospun Nanofibrous Membranes of Collagen and Zein for Wound Healing. ACS Appl. Mater. Interfaces 2012, 4, 1050–1057. [Google Scholar] [CrossRef]
- Buttafoco, L.; Kolkman, N.G.; Engbers-Buijtenhuijs, P.; Poot, A.A.; Dijkstra, P.J.; Vermes, I.; Feijen, J. Electrospinning of Collagen and Elastin for Tissue Engineering Applications. Biomaterials 2006, 27, 724–734. [Google Scholar] [CrossRef]
- Neal, R.A.; McClugage, S.G.; Link, M.C.; Sefcik, L.S.; Ogle, R.C.; Botchwey, E.A. Laminin Nanofiber Meshes that Mimic Morphological Properties and Bioactivity of Basement Membranes. Tissue Eng. Part C Methods 2009, 15, 11–21. [Google Scholar] [CrossRef]
- Chong, E.J.; Phan, T.T.; Lim, I.J.; Zhang, Y.Z.; Bay, B.H.; Ramakrishna, S.; Lim, C.T. Evaluation of Electrospun PCL/Gelatin Nanofibrous Scaffold for Wound Healing and Layered Dermal Reconstitution. Acta Biomater. 2007, 3, 321–330. [Google Scholar] [CrossRef]
- Duan, H.; Feng, B.; Guo, X.; Wang, J.; Zhao, L.; Zhou, G.; Liu, W.; Cao, Y.; Zhang, W.J. Engineering of Epidermis Skin Grafts Using Electrospun Nanofibrous Gelatin/Polycaprolactone Membranes. Int. J. Nanomed. 2013, 8, 2077. [Google Scholar] [CrossRef]
- Powell, H.M.; Boyce, S.T. Fiber Density of Electrospun Gelatin Scaffolds Regulates Morphogenesis of Dermal–Epidermal Skin Substitutes. J. Biomed. Mater. Res. 2008, 84, 1078–1086. [Google Scholar] [CrossRef]
- Kim, S.E.; Heo, D.N.; Lee, J.B.; Kim, J.R.; Park, S.H.; Jeon, S.H.; Kwon, I.K. Electrospun Gelatin/Polyurethane Blended Nanofibers for Wound Healing. Biomed. Mater. 2009, 4, 044106. [Google Scholar] [CrossRef]
- Heo, D.N.; Yang, D.H.; Lee, J.B.; Bae, M.S.; Kim, J.H.; Moon, S.H.; Chun, H.J.; Kim, C.H.; Lim, H.-N.; Kwon, I.K. Burn-Wound Healing Effect of Gelatin/Polyurethane Nanofiber Scaffold Containing Silver-Sulfadiazine. J. Biomed. Nanotechnol. 2013, 9, 511–515. [Google Scholar] [CrossRef]
- Ji, Y.; Ghosh, K.; Shu, X.Z.; Li, B.; Sokolov, J.C.; Prestwich, G.D.; Clark, R.A.F.; Rafailovich, M.H. Electrospun Three-Dimensional Hyaluronic Acid Nanofibrous Scaffolds. Biomaterials 2006, 27, 3782–3792. [Google Scholar] [CrossRef]
- Chutipakdeevong, J.; Ruktanonchai, U.R.; Supaphol, P. Process Optimization of Electrospun Silk Fibroin Fiber Mat for Accelerated Wound Healing. J. Appl. Polym. Sci. 2013, 130, 3634–3644. [Google Scholar] [CrossRef]
- Schneider, A.; Wang, X.Y.; Kaplan, D.L.; Garlick, J.A.; Egles, C. Biofunctionalized Electrospun Silk Mats as a Topical Bioactive Dressing for Accelerated Wound Healing. Acta Biomater. 2009, 5, 2570–2578. [Google Scholar] [CrossRef]
- Noh, H.K.; Lee, S.W.; Kim, J.-M.; Oh, J.-E.; Kim, K.-H.; Chung, C.-P.; Choi, S.-C.; Park, W.H.; Min, B.-M. Electrospinning of Chitin Nanofibers: Degradation Behavior and Cellular Response to Normal Human Keratinocytes and Fibroblasts. Biomaterials 2006, 27, 3934–3944. [Google Scholar] [CrossRef]
- Zhou, Y.; Yang, D.; Chen, X.; Xu, Q.; Lu, F.; Nie, J. Electrospun Water-Soluble Carboxyethyl Chitosan/Poly(Vinyl Alcohol) Nanofibrous Membrane as Potential Wound Dressing for Skin Regeneration. Biomacromolecules 2008, 9, 349–354. [Google Scholar] [CrossRef]
- Dhandayuthapani, B.; Krishnan, U.M.; Sethuraman, S. Fabrication and Characterization of Chitosan-Gelatin Blend Nanofibers for Skin Tissue Engineering. J. Biomed. Mater. Res. 2010, 94, 264–272. [Google Scholar] [CrossRef]
- Kumbar, S.G.; Nukavarapu, S.P.; James, R.; Nair, L.S.; Laurencin, C.T. Electrospun Poly (Lactic Acid-Co-Glycolic Acid) Scaffolds for Skin Tissue Engineering. Biomaterials 2008, 29, 4100–4107. [Google Scholar] [CrossRef]
- Liu, S.-J.; Kau, Y.-C.; Chou, C.-Y.; Chen, J.-K.; Wu, R.-C.; Yeh, W.-L. Electrospun PLGA/Collagen Nanofibrous Membrane as Early-Stage Wound Dressing. J. Membr. Sci. 2010, 355, 53–59. [Google Scholar] [CrossRef]
- Xie, Z.; Paras, C.B.; Weng, H.; Punnakitikashem, P.; Su, L.-C.; Vu, K.; Tang, L.; Yang, J.; Nguyen, K.T. Dual Growth Factor Releasing Multi-Functional Nanofibers for Wound Healing. Acta Biomater. 2013, 9, 9351–9359. [Google Scholar] [CrossRef]
- Vargas, E.A.T.; do Vale Baracho, N.C.; de Brito, J.; de Queiroz, A.A.A. Hyperbranched Polyglycerol Electrospun Nanofibers for Wound Dressing Applications. Acta Biomater. 2010, 6, 1069–1078. [Google Scholar] [CrossRef] [PubMed]
- Carr, K.E. Scanning Electron Microscope Studies of Human Skin. Br. J. Plast. Surg. 1970, 23, 66–72. [Google Scholar] [CrossRef]
- Van Zuijlen, P.P.M.; Ruurda, J.J.B.; van Veen, H.A.; van Marle, J.; van Trier, A.J.M.; Groenevelt, F.; Kreis, R.W.; Middelkoop, E. Collagen Morphology in Human Skin and Scar Tissue: No Adaptations in Response to Mechanical Loading at Joints. Burns 2003, 29, 423–431. [Google Scholar] [CrossRef]
- Rawlins, J.M.; Lam, W.L.; Karoo, R.O.; Naylor, I.L.; Sharpe, D.T. Quantifying Collagen Type in Mature Burn Scars: A Novel Approach Using Histology and Digital Image Analysis. J. Burn. Care Res. 2006, 27, 60–65. [Google Scholar] [CrossRef] [PubMed]
- Osman, O.S.; Selway, J.L.; Harikumar, P.E.; Stocker, C.J.; Wargent, E.T.; Cawthorne, M.A.; Jassim, S.; Langlands, K. A Novel Method to Assess Collagen Architecture in Skin. BMC Bioinform. 2013, 14, 260. [Google Scholar] [CrossRef] [PubMed]
- Abbasipour, M.; Khajavi, R. Nanofiber Bundles and Yarns Production by Electrospinning: A Review. Adv. Polym. Technol. 2013, 32. [Google Scholar] [CrossRef]
- Yousefzadeh, M.; Latifi, M.; Teo, W.-E.; Amani-Tehran, M.; Ramakrishna, S. Producing Continuous Twisted Yarn from Well-Aligned Nanofibers by Water Vortex. Polym. Eng. Sci. 2011, 51, 323–329. [Google Scholar] [CrossRef]
- Ali, U.; Niu, H.; Abbas, A.; Shao, H.; Lin, T. Online Stretching of Directly Electrospun Nanofiber Yarns. RSC Adv. 2016, 6, 30564–30569. [Google Scholar] [CrossRef]
- Khil, M.-S.; Bhattarai, S.R.; Kim, H.-Y.; Kim, S.-Z.; Lee, K.-H. Novel Fabricated Matrix via Electrospinning for Tissue Engineering. J. Biomed. Mater. Res. Part B Appl. Biomater. 2005, 72, 117–124. [Google Scholar] [CrossRef]
- Ravandi, S.A.H.; Tork, R.B.; Dabirian, F.; Gharehaghaji, A.A.; Sajjadi, A. Characteristics of Yarn and Fabric Made out of Nanofibers. Mater. Sci. Appl. 2015, 6, 103. [Google Scholar] [CrossRef]
- Xie, J.; Ma, B.; Michael, P.L. Fabrication of Novel 3D Nanofiber Scaffolds with Anisotropic Property and Regular Pores and Their Potential Applications. Adv. Healthc. Mater. 2012, 1, 674–678. [Google Scholar] [CrossRef]
- Chakraborty, S.; Liao, I.-C.; Adler, A.; Leong, K.W. Electrohydrodynamics: A Facile Technique to Fabricate Drug Delivery Systems. Adv. Drug Deliv. Rev. 2009, 61, 1043–1054. [Google Scholar] [CrossRef]
- Weng, L.; Xie, J. Smart Electrospun Nanofibers for Controlled Drug Release: Recent Advances and New Perspectives. Curr. Pharm. Des. 2015, 21, 1944–1959. [Google Scholar] [CrossRef]
- Said, S.S.; El-Halfawy, O.M.; El-Gowelli, H.M.; Aloufy, A.K.; Boraei, N.A.; El-Khordagui, L.K. Bioburden-Responsive Antimicrobial PLGA Ultrafine Fibers for Wound Healing. Eur. J. Pharm. Biopharm. 2012, 80, 85–94. [Google Scholar] [CrossRef]
- Chou, S.-F.; Carson, D.; Woodrow, K.A. Current Strategies for Sustaining Drug Release from Electrospun Nanofibers. J. Control. Release 2015, 220, 584–591. [Google Scholar] [CrossRef]
- Kim, K.; Luu, Y.K.; Chang, C.; Fang, D.; Hsiao, B.S.; Chu, B.; Hadjiargyrou, M. Incorporation and Controlled Release of a Hydrophilic Antibiotic Using Poly(Lactide-Co-Glycolide)-Based Electrospun Nanofibrous Scaffolds. J. Control. Release 2004, 98, 47–56. [Google Scholar] [CrossRef]
- Ayodeji, O.; Graham, E.; Kniss, D.; Lannutti, J.; Tomasko, D. Carbon Dioxide Impregnation of Electrospun Polycaprolactone Fibers. J. Supercrit. Fluids 2007, 41, 173–178. [Google Scholar] [CrossRef]
- Geiger, B.C.; Nelson, M.T.; Munj, H.R.; Tomasko, D.L.; Lannutti, J.J. Dual Drug Release from CO2-Infused Nanofibers via Hydrophobic and Hydrophilic Interactions. J. Appl. Polym. Sci. 2015, 132. [Google Scholar] [CrossRef]
- Brandl, F.; Sommer, F.; Goepferich, A. Rational Design of Hydrogels for Tissue Engineering: Impact of Physical Factors on Cell Behavior. Biomaterials 2007, 28, 134–146. [Google Scholar] [CrossRef]
- Discher, D.E.; Janmey, P.; Wang, Y. Tissue Cells Feel and Respond to the Stiffness of Their Substrate. Science 2005, 310, 1139–1143. [Google Scholar] [CrossRef]
- Yao, J.; Bastiaansen, C.W.M.; Peijs, T. High Strength and High Modulus Electrospun Nanofibers. Fibers 2014, 2, 158–186. [Google Scholar] [CrossRef]
- Mohammadzadehmoghadam, S.; Dong, Y.; Davies, I.J. Recent Progress in Electrospun Nanofibers: Reinforcement Effect and Mechanical Performance. J. Polym. Sci. Part B Polym. Phys. 2015, 53, 1171–1212. [Google Scholar] [CrossRef]
- Ravandi, S.A.H.; Sadrjahani, M. Mechanical and Structural Characterizations of Simultaneously Aligned and Heat Treated PAN Nanofibers. J. Appl. Polym. Sci. 2012, 124, 3529–3537. [Google Scholar] [CrossRef]
- Xie, J.; Michael, P.L.; Zhong, S.; Ma, B.; MacEwan, M.R.; Lim, C.T. Mussel Inspired Protein-Mediated Surface Modification to Electrospun Fibers and Their Potential Biomedical Applications. J. Biomed. Mater. Res. Part A 2012, 100, 929–938. [Google Scholar] [CrossRef]
- Xie, J.; Zhong, S.; Ma, B.; Shuler, F.D.; Lim, C.T. Controlled Biomineralization of Electrospun Poly (ε-Caprolactone) Fibers to Enhance Their Mechanical Properties. Acta Biomater. 2013, 9, 5698–5707. [Google Scholar] [CrossRef]
- Ma, K.; Chan, C.K.; Liao, S.; Hwang, W.Y.K.; Feng, Q.; Ramakrishna, S. Electrospun Nanofiber Scaffolds for Rapid and Rich Capture of Bone Marrow-Derived Hematopoietic Stem Cells. Biomaterials 2008, 29, 2096–2103. [Google Scholar] [CrossRef]
- Gümüşderelioğlu, M.; Dalkıranoğlu, S.; Aydın, R.S.T.; Çakmak, S. A Novel Dermal Substitute Based on Biofunctionalized Electrospun PCL Nanofibrous Matrix. J. Biomed. Mater. Res. Part A 2011, 98, 461–472. [Google Scholar] [CrossRef]
- Lee, E.J.; Lee, J.H.; Jin, L.; Jin, O.S.; Shin, Y.C.; Oh, S.J.; Lee, J.; Hyon, S.-H.; Han, D.-W. Hyaluronic Acid/Poly (Lactic-Co-Glycolic Acid) Core/Shell Fiber Meshes Loaded with Epigallocatechin-3-O-Gallate as Skin Tissue Engineering Scaffolds. J. Nanosci. Nanotechnol. 2014, 14, 8458–8463. [Google Scholar] [CrossRef]
- Choi, J.S.; Leong, K.W.; Yoo, H.S. In Vivo Wound Healing of Diabetic Ulcers Using Electrospun Nanofibers Immobilized with Human Epidermal Growth Factor (EGF). Biomaterials 2008, 29, 587–596. [Google Scholar] [CrossRef]
- Pelipenko, J.; Kocbek, P.; Govedarica, B.; Rošic, R.; Baumgartner, S.; Kristl, J. The Topography of Electrospun Nanofibers and Its Impact on the Growth and Mobility of Keratinocytes. Eur. J. Pharm. Biopharm. 2013, 84, 401–411. [Google Scholar] [CrossRef]
- Ehrmann, A. Non-Toxic Crosslinking of Electrospun Gelatin Nanofibers for Tissue Engineering and Biomedicine—A Review. Polymers 2021, 13, 1973. [Google Scholar] [CrossRef]
- Homaeigohar, S.; Boccaccini, A.R. Antibacterial Biohybrid Nanofibers for Wound Dressings. Acta Biomater. 2020, 107, 25–49. [Google Scholar] [CrossRef]
- Hakkarainen, T.; Koivuniemi, R.; Kosonen, M.; Escobedo-Lucea, C.; Sanz-Garcia, A.; Vuola, J.; Valtonen, J.; Tammela, P.; Mäkitie, A.; Luukko, K.; et al. Nanofibrillar Cellulose Wound Dressing in Skin Graft Donor Site Treatment. J. Control. Release 2016, 244, 292–301. [Google Scholar] [CrossRef]
- Czaja, W.; Krystynowicz, A.; Bielecki, S.; Brown, R.M. Microbial Cellulose—the Natural Power to Heal Wounds. Biomaterials 2006, 27, 145–151. [Google Scholar] [CrossRef]
- Czaja, W.K.; Young, D.J.; Kawecki, M.; Brown, R.M. The Future Prospects of Microbial Cellulose in Biomedical Applications. Biomacromolecules 2007, 8, 1–12. [Google Scholar] [CrossRef]
- Mogoşanu, G.D.; Grumezescu, A.M. Natural and Synthetic Polymers for Wounds and Burns Dressing. Int. J. Pharm. 2014, 463, 127–136. [Google Scholar] [CrossRef]
- Wutticharoenmongkol, P.; Hannirojram, P.; Nuthong, P. Gallic Acid-Loaded Electrospun Cellulose Acetate Nanofibers as Potential Wound Dressing Materials. Polym. Adv. Technol. 2019, 30, 1135–1147. [Google Scholar] [CrossRef]
- Liu, Y.; Ma, G.; Fang, D.; Xu, J.; Zhang, H.; Nie, J. Effects of Solution Properties and Electric Field on the Electrospinning of Hyaluronic Acid. Carbohydr. Polym. 2011, 83, 1011–1015. [Google Scholar] [CrossRef]
- Eng, D.; Caplan, M.; Preul, M.; Panitch, A. Hyaluronan Scaffolds: A Balance between Backbone Functionalization and Bioactivity. Acta Biomater. 2010, 6, 2407–2414. [Google Scholar] [CrossRef]
- Leach, J.B.; Bivens, K.A.; Patrick, C.W., Jr.; Schmidt, C.E. Photocrosslinked Hyaluronic Acid Hydrogels: Natural, Biodegradable Tissue Engineering Scaffolds. Biotechnol. Bioeng. 2003, 82, 578–589. [Google Scholar] [CrossRef]
- Chen, W. Preparation of Hyaluronan-DNA Matrices and Films. Cold Spring Harb. Protoc. 2012, 2012, pdb.prot071522. [Google Scholar] [CrossRef] [PubMed]
- Uppal, R.; Ramaswamy, G.N.; Arnold, C.; Goodband, R.; Wang, Y. Hyaluronic Acid Nanofiber Wound Dressing—Production, Characterization, and in Vivo Behavior. J. Biomed. Mater. Res. Part B Appl. Biomater. 2011, 97, 20–29. [Google Scholar] [CrossRef] [PubMed]
- Séon-Lutz, M.; Couffin, A.-C.; Vignoud, S.; Schlatter, G.; Hébraud, A. Electrospinning in Water and in Situ Crosslinking of Hyaluronic Acid/Cyclodextrin Nanofibers: Towards Wound Dressing with Controlled Drug Release. Carbohydr. Polym. 2019, 207, 276–287. [Google Scholar] [CrossRef] [PubMed]
- Alavarse, A.C.; de Oliveira Silva, F.W.; Colque, J.T.; da Silva, V.M.; Prieto, T.; Venancio, E.C.; Bonvent, J.-J. Tetracycline Hydrochloride-Loaded Electrospun Nanofibers Mats Based on PVA and Chitosan for Wound Dressing. Mater. Sci. Eng. C 2017, 77, 271–281. [Google Scholar] [CrossRef]
- Sarhan, W.A.; Azzazy, H.M.E.; El-Sherbiny, I.M. Honey/Chitosan Nanofiber Wound Dressing Enriched with Allium Sativum and Cleome Droserifolia: Enhanced Antimicrobial and Wound Healing Activity. ACS Appl. Mater. Interfaces 2016, 8, 6379–6390. [Google Scholar] [CrossRef]
- Homaeigohar, S.; Tsai, T.-Y.; Young, T.-H.; Yang, H.J.; Ji, Y.-R. An Electroactive Alginate Hydrogel Nanocomposite Reinforced by Functionalized Graphite Nanofilaments for Neural Tissue Engineering. Carbohydr. Polym. 2019, 224, 115112. [Google Scholar] [CrossRef]
- Lee, K.Y.; Mooney, D.J. Alginate: Properties and Biomedical Applications. Prog. Polym. Sci. 2012, 37, 106–126. [Google Scholar] [CrossRef]
- Tang, Y.; Lan, X.; Liang, C.; Zhong, Z.; Xie, R.; Zhou, Y.; Miao, X.; Wang, H.; Wang, W. Honey Loaded Alginate/PVA Nanofibrous Membrane as Potential Bioactive Wound Dressing. Carbohydr. Polym. 2019, 219, 113–120. [Google Scholar] [CrossRef]
- Zhou, T.; Wang, N.; Xue, Y.; Ding, T.; Liu, X.; Mo, X.; Sun, J. Electrospun Tilapia Collagen Nanofibers Accelerating Wound Healing via Inducing Keratinocytes Proliferation and Differentiation. Colloids Surf. B Biointerfaces 2016, 143, 415–422. [Google Scholar] [CrossRef]
- Wen, X.; Zheng, Y.; Wu, J.; Wang, L.-N.; Yuan, Z.; Peng, J.; Meng, H. Immobilization of Collagen Peptide on Dialdehyde Bacterial Cellulose Nanofibers via Covalent Bonds for Tissue Engineering and Regeneration. Int. J. Nanomed. 2015, 10, 4623–4637. [Google Scholar] [CrossRef]
- Yao, C.-H.; Lee, C.-Y.; Huang, C.-H.; Chen, Y.-S.; Chen, K.-Y. Novel Bilayer Wound Dressing Based on Electrospun Gelatin/Keratin Nanofibrous Mats for Skin Wound Repair. Mater. Sci. Eng. C 2017, 79, 533–540. [Google Scholar] [CrossRef]
- Melke, J.; Midha, S.; Ghosh, S.; Ito, K.; Hofmann, S. Silk Fibroin as Biomaterial for Bone Tissue Engineering. Acta Biomater. 2016, 31, 1–16. [Google Scholar] [CrossRef]
- Fan, L.; Cai, Z.; Zhang, K.; Han, F.; Li, J.; He, C.; Mo, X.; Wang, X.; Wang, H. Green Electrospun Pantothenic Acid/Silk Fibroin Composite Nanofibers: Fabrication, Characterization and Biological Activity. Colloids Surf. B Biointerfaces 2014, 117, 14–20. [Google Scholar] [CrossRef]
- Song, D.W.; Kim, S.H.; Kim, H.H.; Lee, K.H.; Ki, C.S.; Park, Y.H. Multi-Biofunction of Antimicrobial Peptide-Immobilized Silk Fibroin Nanofiber Membrane: Implications for Wound Healing. Acta Biomater. 2016, 39, 146–155. [Google Scholar] [CrossRef]
- Selvaraj, S.; Fathima, N.N. Fenugreek Incorporated Silk Fibroin Nanofibers-A Potential Antioxidant Scaffold for Enhanced Wound Healing. ACS Appl. Mater. Interfaces 2017, 9, 5916–5926. [Google Scholar] [CrossRef]
- GhavamiNejad, A.; Rajan Unnithan, A.; Kurup Sasikala, A.R.; Samarikhalaj, M.; Thomas, R.G.; Jeong, Y.Y.; Nasseri, S.; Murugesan, P.; Wu, D.; Hee Park, C.; et al. Mussel-Inspired Electrospun Nanofibers Functionalized with Size-Controlled Silver Nanoparticles for Wound Dressing Application. ACS Appl. Mater. Interfaces 2015, 7, 12176–12183. [Google Scholar] [CrossRef]
- Wang, J.; Windbergs, M. Functional Electrospun Fibers for the Treatment of Human Skin Wounds. Eur. J. Pharm. Biopharm. 2017, 119, 283–299. [Google Scholar] [CrossRef]
- Kalantari, K.; Mostafavi, E.; Afifi, A.M.; Izadiyan, Z.; Jahangirian, H.; Rafiee-Moghaddam, R.; Webster, T.J. Wound Dressings Functionalized with Silver Nanoparticles: Promises and Pitfalls. Nanoscale 2020, 12, 2268–2291. [Google Scholar] [CrossRef]
- Shankar, P.D.; Shobana, S.; Karuppusamy, I.; Pugazhendhi, A.; Ramkumar, V.S.; Arvindnarayan, S.; Kumar, G. A Review on the Biosynthesis of Metallic Nanoparticles (Gold and Silver) Using Bio-Components of Microalgae: Formation Mechanism and Applications. Enzym. Microb. Technol. 2016, 95, 28–44. [Google Scholar] [CrossRef]
- Kumar, P.T.S.; Lakshmanan, V.-K.; Biswas, R.; Nair, S.V.; Jayakumar, R. Synthesis and Biological Evaluation of Chitin Hydrogel/Nano ZnO Composite Bandage as Antibacterial Wound Dressing. J. Biomed. Nanotechnol. 2012, 8, 891–900. [Google Scholar] [CrossRef]
- Stoica, E.; Chircov, C.; Grumezescu, A. Nanomaterials for Wound Dressings: An Up-to-Date Overview. Molecules 2020, 25, 2699. [Google Scholar] [CrossRef] [PubMed]
- Jatoi, A.W.; Jo, Y.K.; Lee, H.; Oh, S.-G.; Hwang, D.S.; Khatri, Z.; Cha, H.J.; Kim, I.S. Antibacterial Efficacy of Poly (Vinyl Alcohol) Composite Nanofibers Embedded with Silver-Anchored Silica Nanoparticles. J. Biomed. Mater. Res. Part B Appl. Biomater. 2018, 106, 1121–1128. [Google Scholar] [CrossRef] [PubMed]
- Fouda, M.M.G.; El-Aassar, M.R.; Al-Deyab, S.S. Antimicrobial Activity of Carboxymethyl Chitosan/Polyethylene Oxide Nanofibers Embedded Silver Nanoparticles. Carbohydr. Polym. 2013, 92, 1012–1017. [Google Scholar] [CrossRef] [PubMed]
- Hebeish, A.A.; Ramadan, M.A.; Montaser, A.S.; Farag, A.M. Preparation, Characterization and Antibacterial Activity of Chitosan-g-Poly Acrylonitrile/Silver Nanocomposite. Int. J. Biol. Macromol. 2014, 68, 178–184. [Google Scholar] [CrossRef]
- Maharjan, B.; Joshi, M.K.; Tiwari, A.P.; Park, C.H.; Kim, C.S. In-Situ Synthesis of AgNPs in the Natural/Synthetic Hybrid Nanofibrous Scaffolds: Fabrication, Characterization and Antimicrobial Activities. J. Mech. Behav. Biomed. Mater. 2017, 65, 66–76. [Google Scholar] [CrossRef]
- Hashmi, M.; Ullah, S.; Ullah, A.; Akmal, M.; Saito, Y.; Hussain, N.; Ren, X.; Kim, I.S. Optimized Loading of Carboxymethyl Cellulose (CMC) in Tri-Component Electrospun Nanofibers Having Uniform Morphology. Polymers 2020, 12, 2524. [Google Scholar] [CrossRef]
- Eghbalifam, N.; Shojaosadati, S.A.; Hashemi-Najafabadi, S.; Khorasani, A.C. Synthesis and Characterization of Antimicrobial Wound Dressing Material Based on Silver Nanoparticles Loaded Gum Arabic Nanofibers. Int. J. Biol. Macromol. 2020, 155, 119–130. [Google Scholar] [CrossRef]
- Alven, S.; Buyana, B.; Feketshane, Z.; Aderibigbe, B.A. Electrospun Nanofibers/Nanofibrous Scaffolds Loaded with Silver Nanoparticles as Effective Antibacterial Wound Dressing Materials. Pharmaceutics 2021, 13, 964. [Google Scholar] [CrossRef]
- Bharathi, B.S.; Stalin, T. Cerium Oxide and Peppermint Oil Loaded Polyethylene Oxide/Graphene Oxide Electrospun Nanofibrous Mats as Antibacterial Wound Dressings. Mater. Today Commun. 2019, 21, 100664. [Google Scholar] [CrossRef]
- One-Pot Synthesis of ZnO Nanobelt-like Structures in Hyaluronan Hydrogels for Wound Dressing Applications—ScienceDirect. Available online: https://www.sciencedirect.com/science/article/pii/S014486171930791X?via%3Dihub (accessed on 17 October 2021).
- Kalaycıoğlu, Z.; Kahya, N.; Adımcılar, V.; Kaygusuz, H.; Torlak, E.; Akın-Evingür, G.; Erim, F.B. Antibacterial Nano Cerium Oxide/Chitosan/Cellulose Acetate Composite Films as Potential Wound Dressing. Eur. Polym. J. 2020, 133, 109777. [Google Scholar] [CrossRef]
- In Vivo Evaluation of Chitosan–PVP–Titanium Dioxide Nanocomposite as Wound Dressing Material—ScienceDirect. Available online: https://www.sciencedirect.com/science/article/pii/S0144861713002725?via%3Dihub (accessed on 17 October 2021).
- Archana, D.; Dutta, J.; Dutta, P.K. Evaluation of Chitosan Nano Dressing for Wound Healing: Characterization, in Vitro and in Vivo Studies. Int. J. Biol. Macromol. 2013, 57, 193–203. [Google Scholar] [CrossRef]
- Cai, N.; Li, C.; Han, C.; Luo, X.; Shen, L.; Xue, Y.; Yu, F. Tailoring Mechanical and Antibacterial Properties of Chitosan/Gelatin Nanofiber Membranes with Fe3O4 Nanoparticles for Potential Wound Dressing Application. Appl. Surf. Sci. 2016, 369, 492–500. [Google Scholar] [CrossRef]
- Madhumathi, K.; Sudheesh Kumar, P.T.; Abhilash, S.; Sreeja, V.; Tamura, H.; Manzoor, K.; Nair, S.V.; Jayakumar, R. Development of Novel Chitin/Nanosilver Composite Scaffolds for Wound Dressing Applications. J. Mater. Sci. Mater. Med. 2010, 21, 807–813. [Google Scholar] [CrossRef]
- Nguyen, N.; Nguyen, L.; Thanh, N.; Vo, T.; Quyen, T.; Tran, P.; Nguyen, T.H. Stabilization of Silver Nanoparticles in Chitosan and Gelatin Hydrogel and Its Applications. Mater. Lett. 2019, 248, 241–245. [Google Scholar] [CrossRef]
- Haseeb, M.T.; Hussain, M.A.; Abbas, K.; Youssif, B.G.; Bashir, S.; Yuk, S.H.; Bukhari, S.N.A. Linseed Hydrogel-Mediated Green Synthesis of Silver Nanoparticles for Antimicrobial and Wound-Dressing Applications. Int. J. Nanomed. 2017, 12, 2845–2855. [Google Scholar] [CrossRef]
- Felgueiras, H.P.; Amorim, M.T.P. Functionalization of Electrospun Polymeric Wound Dressings with Antimicrobial Peptides. Colloids Surf. B Biointerfaces 2017, 156, 133–148. [Google Scholar] [CrossRef]
- Costa, F.; Carvalho, I.F.; Montelaro, R.C.; Gomes, P.; Martins, M.C.L. Covalent Immobilization of Antimicrobial Peptides (AMPs) onto Biomaterial Surfaces. Acta Biomater. 2011, 7, 1431–1440. [Google Scholar] [CrossRef]
- Deshmukh, K.; Sankaran, S.; Basheer Ahamed, M.; Khadheer Pasha, S.K. Biomedical Applications of Electrospun Polymer Composite Nanofibres. In Polymer Nanocomposites in Biomedical Engineering; Sadasivuni, K.K., Ponnamma, D., Rajan, M., Ahmed, B., Al-Maadeed, M.A.S.A., Eds.; Lecture Notes in Bioengineering; Springer International Publishing: Cham, Switzerland, 2019; pp. 111–165. ISBN 978-3-030-04741-2. [Google Scholar]
- Peschel, A. How Do Bacteria Resist Human Antimicrobial Peptides? Trends Microbiol. 2002, 10, 179–186. [Google Scholar] [CrossRef]
- Goy, R.C.; Morais, S.T.B.; Assis, O.B.G. Evaluation of the Antimicrobial Activity of Chitosan and Its Quaternized Derivative on E. Coli and S. Aureus Growth. Rev. Bras. Farmacogn. 2016, 26, 122–127. [Google Scholar] [CrossRef]
- Saharan, V.; Mehrotra, A.; Khatik, R.; Rawal, P.; Sharma, S.S.; Pal, A. Synthesis of Chitosan Based Nanoparticles and Their in Vitro Evaluation against Phytopathogenic Fungi. Int. J. Biol. Macromol. 2013, 62, 677–683. [Google Scholar] [CrossRef]
- Arkoun, M.; Daigle, F.; Heuzey, M.-C.; Ajji, A. Mechanism of Action of Electrospun Chitosan-Based Nanofibers against Meat Spoilage and Pathogenic Bacteria. Molecules 2017, 22, 585. [Google Scholar] [CrossRef] [PubMed]
- Park, S.-C.; Choi, C.; Jeong, G.-W.; Lee, H.-S.; Choi, S.-J.; Kim, W.-S.; Nah, J.-W. Algicidal Effects of Free-Amine Water-Soluble Chitosan to Marine Harmful Algal Species. J. Ind. Eng. Chem. 2016, 34, 139–145. [Google Scholar] [CrossRef]
- Chirkov, S.N. The Antiviral Activity of Chitosan. Appl. Biochem. Microbiol. 2002, 38, 1–8. [Google Scholar] [CrossRef]
- Chen, S.; Liu, B.; Carlson, M.A.; Gombart, A.F.; Reilly, D.A.; Xie, J. Recent Advances in Electrospun Nanofibers for Wound Healing. Nanomedicine 2017, 12, 1335–1352. [Google Scholar] [CrossRef] [PubMed]
- Jeon, Y.; Kim, S. Effect of Antimicrobial Activity by Chitosan Oligosaccharide N-Conjugated with Asparagine. J. Microbiol. Biotechnol. 2001, 11, 281–286. [Google Scholar]
- Xiao, B.; Wan, Y.; Zhao, M.; Liu, Y.; Zhang, S. Preparation and Characterization of Antimicrobial Chitosan-N-Arginine with Different Degrees of Substitution. Carbohydr. Polym. 2011, 83, 144–150. [Google Scholar] [CrossRef]
- Antunes, B.P.; Moreira, A.F.; Gaspar, V.M.; Correia, I.J. Chitosan/Arginine–Chitosan Polymer Blends for Assembly of Nanofibrous Membranes for Wound Regeneration. Carbohydr. Polym. 2015, 130, 104–112. [Google Scholar] [CrossRef]
- Unnithan, A.R.; Gnanasekaran, G.; Sathishkumar, Y.; Lee, Y.S.; Kim, C.S. Electrospun Antibacterial Polyurethane–Cellulose Acetate–Zein Composite Mats for Wound Dressing. Carbohydr. Polym. 2014, 102, 884–892. [Google Scholar] [CrossRef]
- He, M.; Chen, M.; Dou, Y.; Ding, J.; Yue, H.; Yin, G.; Chen, X.; Cui, Y. Electrospun Silver Nanoparticles-Embedded Feather Keratin/Poly(Vinyl Alcohol)/Poly(Ethylene Oxide) Antibacterial Composite Nanofibers. Polymers 2020, 12, 305. [Google Scholar] [CrossRef]
- Sun, L.; Han, J.; Liu, Z.; Wei, S.; Su, X.; Zhang, G. The Facile Fabrication of Wound Compatible Anti-Microbial Nanoparticles Encapsulated Collagenous Chitosan Matrices for Effective Inhibition of Poly-Microbial Infections and Wound Repairing in Burn Injury Care: Exhaustive in Vivo Evaluations. J. Photochem. Photobiol. B Biol. 2019, 197, 111539. [Google Scholar] [CrossRef]
- Stie, M.B.; Corezzi, M.; Juncos Bombin, A.D.; Ajalloueian, F.; Attrill, E.; Pagliara, S.; Jacobsen, J.; Chronakis, I.S.; Nielsen, H.M.; Fodera, V. Waterborne Electrospinning of α-Lactalbumin Generates Tunable and Biocompatible Nanofibers for Drug Delivery. ACS Appl. Nano Mater. 2020, 3, 1910–1921. [Google Scholar] [CrossRef]
- Jao, W.-C.; Yang, M.-C.; Lin, C.-H.; Hsu, C.-C. Fabrication and Characterization of Electrospun Silk Fibroin/TiO2 Nanofibrous Mats for Wound Dressings. Polym. Adv. Technol. 2012, 23, 1066–1076. [Google Scholar] [CrossRef]
- Antimicrobial Electrospun Silk Fibroin Mats with Silver Nanoparticles for Wound Dressing Application. Available online: https://www.semanticscholar.org/paper/Antimicrobial-electrospun-silk-fibroin-mats-with-Uttayarat-Jetawattana/bc673765a402c9ed556fad65274d13493864488a (accessed on 17 October 2021).
- Padrão, J.; Machado, R.; Casal, M.; Lanceros-Méndez, S.; Rodrigues, L.R.; Dourado, F.; Sencadas, V. Antibacterial Performance of Bovine Lactoferrin-Fish Gelatine Electrospun Membranes. Int. J. Biol. Macromol. 2015, 81, 608–614. Available online: https://ro.uow.edu.au/eispapers/4614/ (accessed on 17 October 2021). [CrossRef]
- Chen, J.; Liu, Z.; Chen, M.; Zhang, H.; Li, X. Electrospun Gelatin Fibers with a Multiple Release of Antibiotics Accelerate Dermal Regeneration in Infected Deep Burns. Macromol. Biosci. 2016, 16, 1368–1380. [Google Scholar] [CrossRef]
- Yıldız, A.; Kara, A.A.; Acartürk, F. Peptide-Protein Based Nanofibers in Pharmaceutical and Biomedical Applications. Int. J. Biol. Macromol. 2020, 148, 1084–1097. [Google Scholar] [CrossRef]
- Azimi, B.; Maleki, H.; Zavagna, L.; De la Ossa, J.G.; Linari, S.; Lazzeri, A.; Danti, S. Bio-Based Electrospun Fibers for Wound Healing. J. Funct. Biomater. 2020, 11, 67. [Google Scholar] [CrossRef]
- Khabbaz, B.; Solouk, A.; Mirzadeh, H. Polyvinyl alcohol/soy protein isolate nanofibrous patch for wound-healing applications. Prog. Biomater. 2019, 8, 185–186. [Google Scholar] [CrossRef]
- Akshaykumar, K.P.; Zare, E.N.; Torres-Mendieta, R.; Wacławek, S.; Makvandi, P.; Černík, M.; Padil, V.V.; Varma, R.S. Electrospun fibers based on botanical, seaweed, microbial, and animal sourced biomacromolecules and their multidimensional applications. Int. J. Biol. Macromol. 2021, 171, 130–149. [Google Scholar] [CrossRef]
- Wilkinson, L.J.; White, R.J.; Chipman, J.K. Silver and Nanoparticles of Silver in Wound Dressings: A Review of Efficacy and Safety. J. Wound Care 2011, 20, 543–549. [Google Scholar] [CrossRef]
- White, R.; Cooper, R. Silver Sulphadiazine: A Review of the Evidence. Wounds UK 2005, 1, 51. [Google Scholar]
- Lansdown, A.B.G. A Pharmacological and Toxicological Profile of Silver as an Antimicrobial Agent in Medical Devices. Adv. Pharmacol. Sci. 2010, 2010, 910686. [Google Scholar] [CrossRef] [PubMed]
- Samberg, M.E.; Oldenburg, S.J.; Monteiro-Riviere, N.A. Evaluation of Silver Nanoparticle Toxicity in Skin in Vivo and Keratinocytes in Vitro. Environ. Health Perspect. 2010, 118, 407–413. [Google Scholar] [CrossRef]
- Wijnhoven, S.W.P.; Peijnenburg, W.J.G.M.; Herberts, C.A.; Hagens, W.I.; Oomen, A.G.; Heugens, E.H.W.; Roszek, B.; Bisschops, J.; Gosens, I.; Van De Meent, D.; et al. Nano-Silver—A Review of Available Data and Knowledge Gaps in Human and Environmental Risk Assessment. Nanotoxicology 2009, 3, 109–138. [Google Scholar] [CrossRef]
- Chen, X.; Schluesener, H.J. Nanosilver: A Nanoproduct in Medical Application. Toxicol. Lett. 2008, 176, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Drake, P.L.; Hazelwood, K.J. Exposure-Related Health Effects of Silver and Silver Compounds: A Review. Ann. Occup. Hyg. 2005, 49, 575–585. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Lin, T.; Gao, Y.; Xu, Z.; Huang, C.; Yao, G.; Jiang, L.; Tang, Y.; Wang, X. Antimicrobial Electrospun Nanofibers of Cellulose Acetate and Polyester Urethane Composite for Wound Dressing. J. Biomed. Mater. Res. Part B Appl. Biomater. 2012, 100, 1556–1565. [Google Scholar] [CrossRef]
- Tang, Y.; Wong, C.; Wang, H.; Sutti, A.; Kirkland, M.; Wang, X.; Lin, T. Three-Dimensional Tissue Scaffolds from Interbonded Poly (ε-Caprolactone) Fibrous Matrices with Controlled Porosity. Tissue Eng. Part C Methods 2011, 17, 209–218. [Google Scholar] [CrossRef]
- Balusamy, B.; Senthamizhan, A.; Uyar, T. In Vivo Safety Evaluations of Electrospun Nanofibers for Biomedical Applications. In Electrospun Materials for Tissue Engineering and Biomedical Applications; Uyar, T., Kny, E., Eds.; Woodhead Publishing: Sawston, UK, 2017; pp. 101–113. ISBN 978-0-08-101022-8. [Google Scholar]
- Home—ClinicalTrials.Gov. Available online: https://clinicaltrials.gov/ (accessed on 15 November 2021).
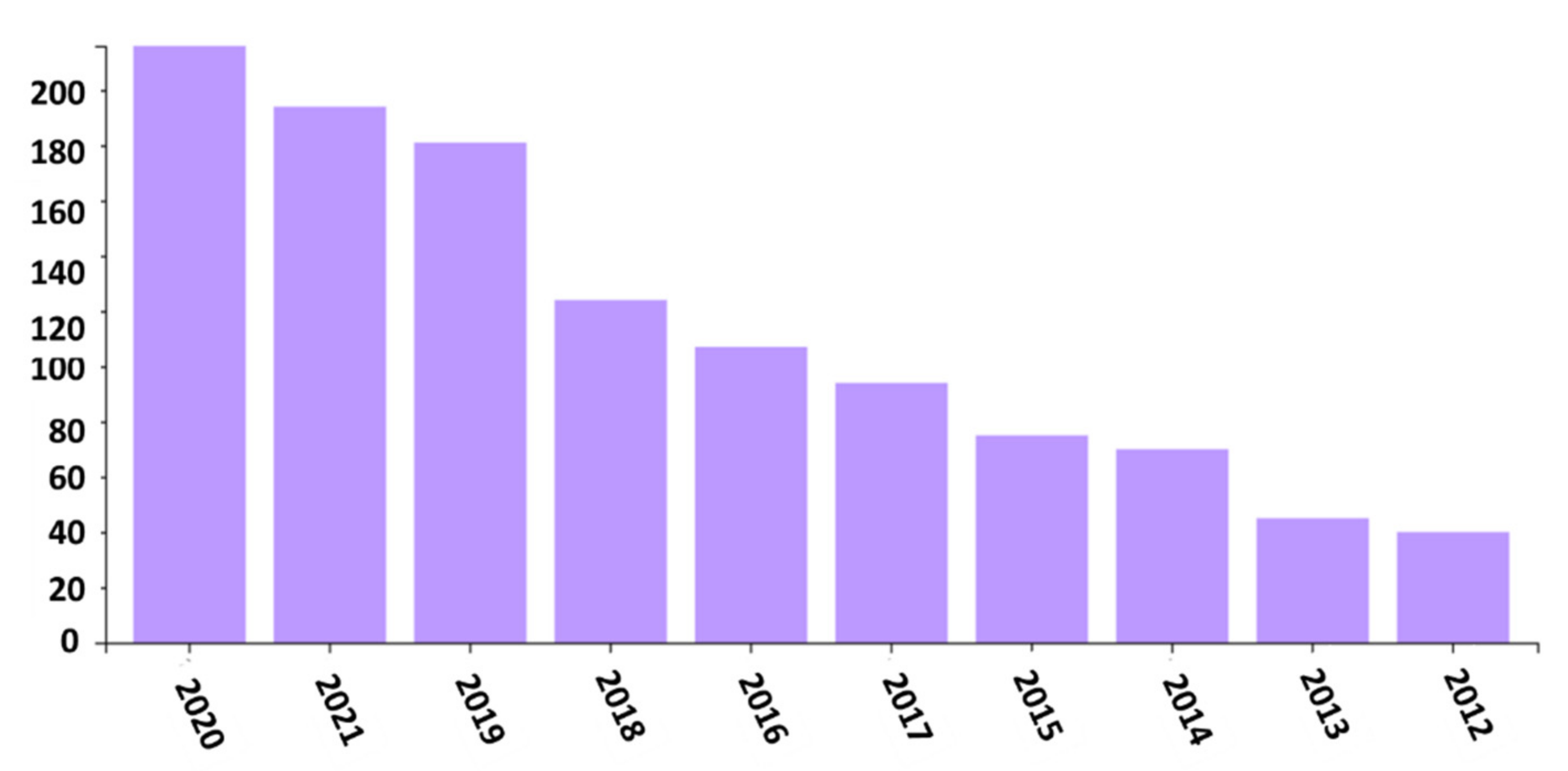
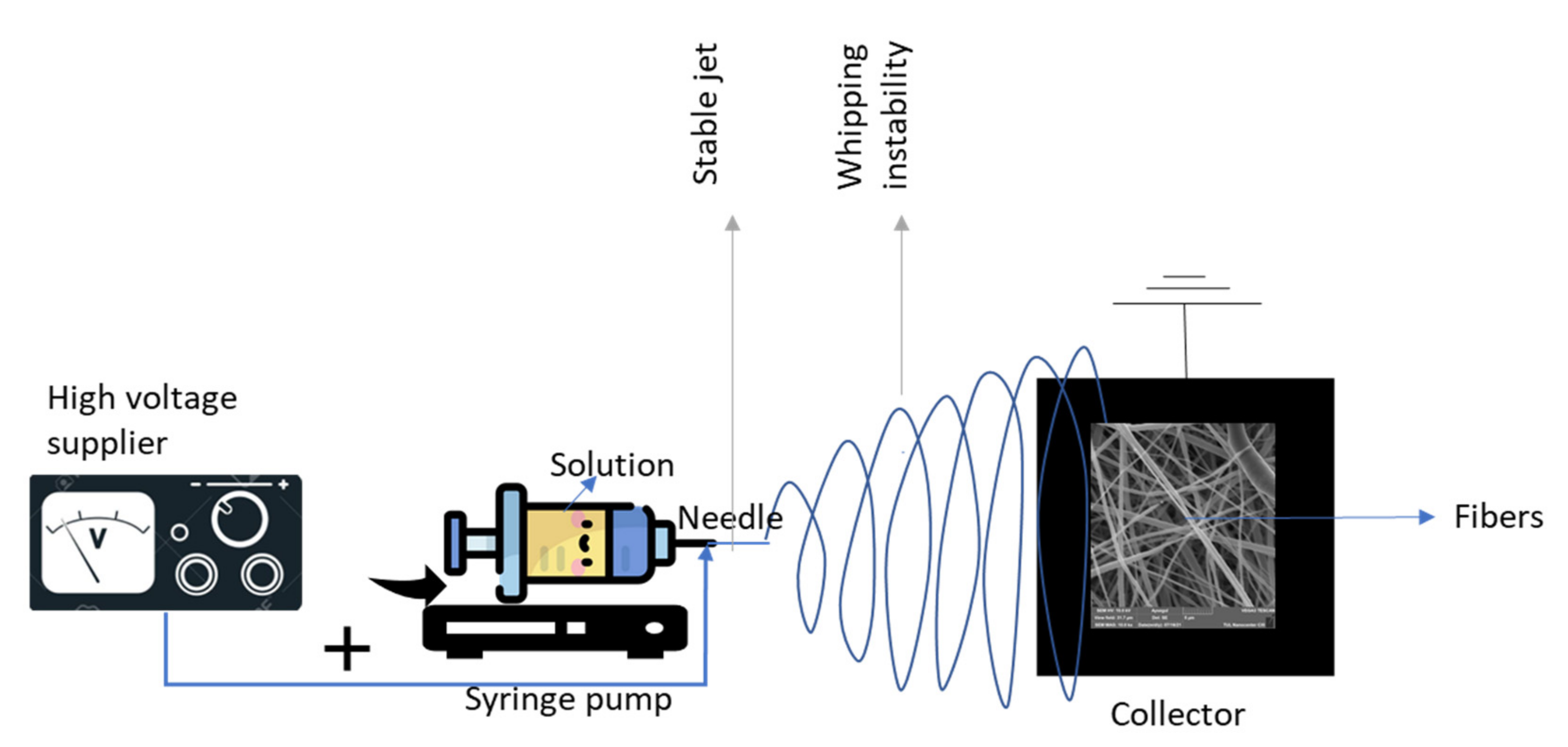
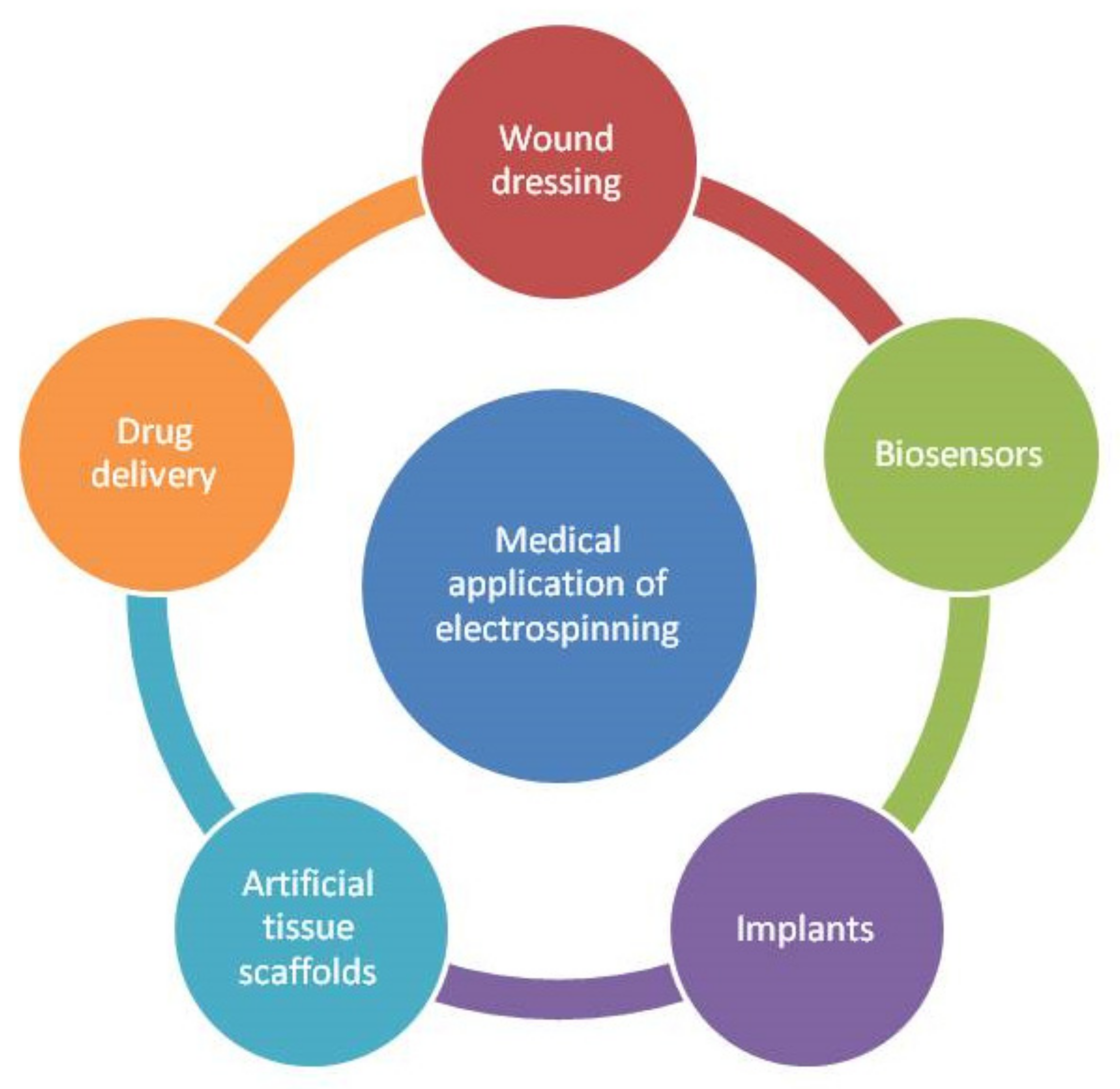
| Parameters | Effect on Fibers | References |
|---|---|---|
| Solution Parameters | ||
| Viscosity | A higher viscosity results in a large fiber diameter. If the viscosity is very low, there will be no continuous fiber formation; if the viscosity is too high, the jet will be difficult to eject from the needle tip. | [24,32,33] |
| Solution Concentration | A minimum solution concentration is required for fiber formation in the electrospinning process. Increased concentration leads to larger diameters. | [34] |
| Molecular weight | Low molecular weight solutions tend to form beads rather than fibers, whereas high molecular weight nanofiber solutions produce fibers with a larger average diameter. | [25] |
| Solution electrical conductivity | When the electrical conductivity of the solution increases, the diameter of the electrospun nanofibers decreases significantly. Beads may also be observed due to the solution’s low conductivity, which results in insufficient elongation of a jet by electrical force to produce uniform fiber. | [27,35] |
| Surface tension | The surface tension of the solution can drive droplets, beads, and fibers and the solution’s low surface tension ensures that spinning occurs with a lower electric field requirement. | [36,37,38] |
| Process Parameters | ||
| Applied voltage | It has been discovered that increasing the electrostatic potential leads to thinner fibers. However, if too much voltage is applied, the jet may become unstable, and the fiber diameters may increase. | [39] |
| Distance from needle to the collector | The traveling time of the polymeric jet is affected. Traveling time should be long enough for complete evaporation of the solvent. | [40,41] |
| Volume feed rate | Increasing the feed rate resulted in an increase in fiber diameter and the formation of a bead structure. | [36,37] |
| Environmental Parameters | ||
| Humidity | High humidity can cause pores on the surface of the fiber. | [33,42] |
| Temperature | Temperature increases cause a decrease in fiber diameter due to a decrease in viscosity. | [42] |
| Composition | Approximate Diameter | Reference |
|---|---|---|
| Collagen | 460 nm | [66,67] |
| Collagen/chitosan | (134 ± 42) nm | [68] |
| Collagen/PCL | (170 ± 0.075) nm | [69] |
| Collagen/Zein | (423–910) nm | [70] |
| Collagen/elastin/PEO | (220–600) nm | [71] |
| Laminin I | (90–300) nm | [72] |
| PCL/gelatin | (470 ± 120) nm; (409 ± 88) nm | [73,74] |
| Gelatin | (570 ± 10) nm | [75] |
| Polyurethane/gelatin | (0.4–2.1) μm | [76,77] |
| HA/PEO | (70–110) nm | [78] |
| Silk fibroin/chitosan | (185.5–249.7) nm | [48] |
| Silk fibroin/PEO | (414 ± 73) nm; 1 μm | [79,80] |
| Chitin | 163 nm | [81] |
| Carboxyethyl chitosan/PVA | (131–456) nm | [82] |
| Chitosan/gelatin | (120–220) nm | [83] |
| PLGA | (150–225) nm | [84] |
| PLGA/collagen | (170–650) nm | [85] |
| Chitosan/PEO | (130–150) nm | [86] |
| Hyperbranched polyglycerol | (58–80) nm | [87] |
| Human Skin | PCL/Collagen | HA/PLGA | PLGA/Collagen | |
|---|---|---|---|---|
| Tensile modulus (MPa) | 15–150 | 21.42 ± 0.04 | 28.0 | 40.43 ± 3.53 |
| Ultimate tensile stress (MPa) | 1–32 | 8.63 ± 1.44 | 1.52 | 1.22 ± 0.12 |
| Ultimate tensile strain (%) | 35–115 | 24.0 ± 7.16 | 60.07 | 96 ± 13 |
| Reference | [112] | [113] | [114] | [112] |
| Material | Nanoparticles | Bacterial Species | Ref. |
|---|---|---|---|
| Carboxymethyl Chitosan/Polyethylene Oxide Nanofibers (CMCTS–PEO) | Ag (12–18 nm) | S. aureus, P. aeruginosa, E. coli, fungus Candida albicans | [149] |
| Alginate/Nicotinamide Nanocomposites | Ag (20–80 nm) | S. aureus and E. coli | [150] |
| Nanofibrous Poly vinyl alcohol, chitosan | Ag | S. aureus and E. coli. | [151] |
| Nanofibrous mats from cellulose acetate | Ag | S. aureus and E. coli. | [152] |
| Nanofibrous membrane from Gum Arabic, polycaprolactone, polyvinyl alcohol | Ag | S. aureus, E. coli, P. aeruginosa and C. albicans | [153] |
| PVA-co-PE nanofibrous membrane | Ag | S. aureus and E. coli. | [154] |
| Electrospun peppermint oil on polyethylene oxide/Graphene oxide | CeO2 | S. aureus and E. coli. | [155] |
| Hyaluronic acid | ZnO | S. aureus, B. subtilis, E. coli, P. aeruginosa, and V. cholerae | [156] |
| Chitosan/cellulose acetate | CeO2 | S. aureus and E. coli. | [157] |
| Chitosan/poly(N-vinylpyrrolidone) | TiO2 | E. coli, S. aureus, B. subtilis and P. aeruginosa | [158] |
| Chitosan/pectin | TiO2 | E. coli, S. aereus, A. niger, B. subtilis, P. aeruginosa | [159] |
| Electrospun Chitosan/Gelatin | Fe3O4 | S. aureus and E. coli. | [160] |
| Β-Chitin Hydrogel | Ag (4–8 nm) | S. aureus and E. coli. | [161] |
| Chitosan/Polyvinyl Alcohol Hydrogel, Collagen | Ag (4–19 nm) | P. aeruginosa and S. aureus | [162] |
| Linseed hydrogel | Ag (10–35 nm) | E. coli, S. mutans, A. niger, S. epidermidis, P. aeruginosa, S. aureus, acillus subtilis, Actinomyces odontolyticus | [163] |
| Protein | Co-Polymer | Antimicrobial Agent | Bacterial Species | Ref. |
|---|---|---|---|---|
| Zein | PU | Ag NPs | E. coli, S. aureus | [151] |
| Zein | PU/CA | Streptomycin | V. vulnificus, S. aureus, B. subtilis | [177] |
| Keratin | PVA, PEO | Ag NPs | E. coli, S. aureus | [178] |
| Collagen | CS | ZnO | S. aureus, E. coli | [179] |
| α-lactoglobulin | PEO | Ampicillin | E. coli, P. aeruginosa, B. thailandensis | [180] |
| Silk fibroin | PEO | TiO2 NPs | E. coli | [181] |
| Silk fibroin | - | Ag NP coating | S. aureus, P. aeruginosa | [182] |
| Silk fibroin | PEO | Cu2O NPs | S. aureus, E. coli | [47] |
| Lactoferrin | Gelatin | - | E. coli, S. aureus | [183] |
| Gelatin | Alginatedialdehyde | Ciprofloxacin, gentamicin | P. aeruginosa, S. epidermidis | [184] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gul, A.; Gallus, I.; Tegginamath, A.; Maryska, J.; Yalcinkaya, F. Electrospun Antibacterial Nanomaterials for Wound Dressings Applications. Membranes 2021, 11, 908. https://doi.org/10.3390/membranes11120908
Gul A, Gallus I, Tegginamath A, Maryska J, Yalcinkaya F. Electrospun Antibacterial Nanomaterials for Wound Dressings Applications. Membranes. 2021; 11(12):908. https://doi.org/10.3390/membranes11120908
Chicago/Turabian StyleGul, Aysegul, Izabela Gallus, Akshat Tegginamath, Jiri Maryska, and Fatma Yalcinkaya. 2021. "Electrospun Antibacterial Nanomaterials for Wound Dressings Applications" Membranes 11, no. 12: 908. https://doi.org/10.3390/membranes11120908
APA StyleGul, A., Gallus, I., Tegginamath, A., Maryska, J., & Yalcinkaya, F. (2021). Electrospun Antibacterial Nanomaterials for Wound Dressings Applications. Membranes, 11(12), 908. https://doi.org/10.3390/membranes11120908








