Removal of Bacteria and Organic Carbon by an Integrated Ultrafiltration—Nanofiltration Desalination Pilot Plant
Abstract
1. Introduction
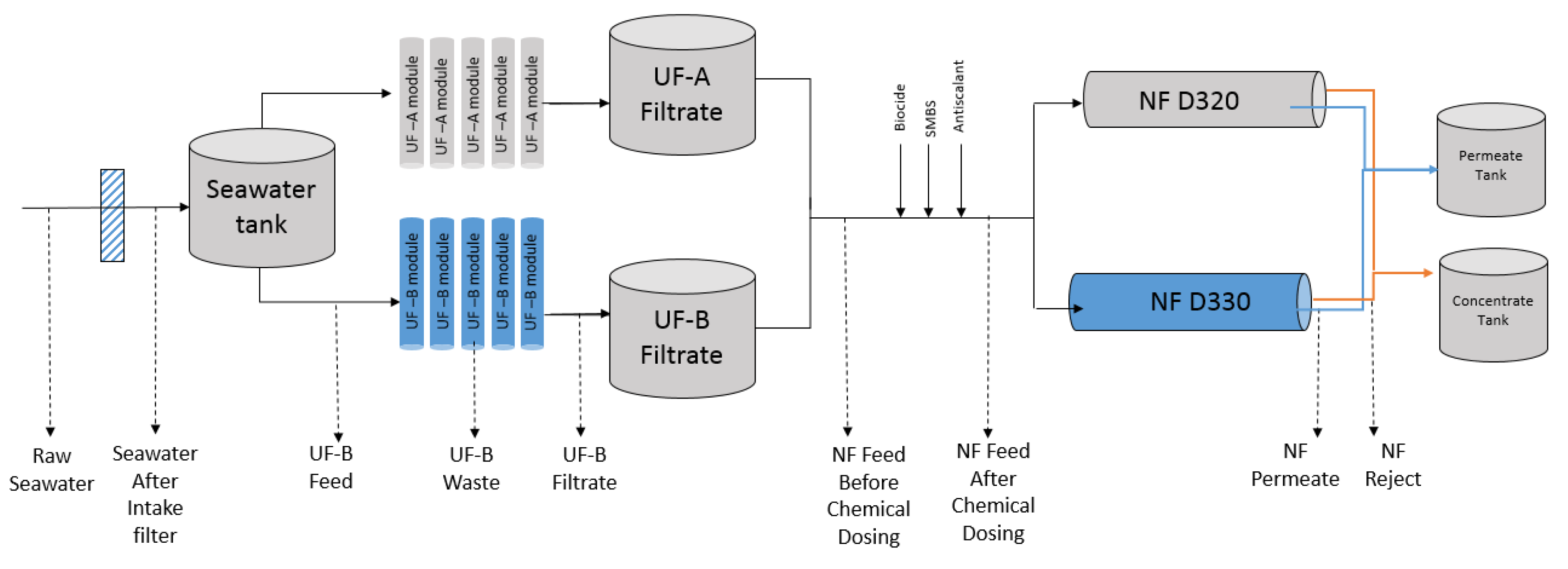
2. Materials and Methods
2.1. Sample Collection
2.2. Membrane Cleaning Procedure
2.3. Water Quality Parameters
2.4. AOC Measurement
2.5. TOC Measurement
2.6. Liquid Chromatography for Organic Carbon Detection (LC-OCD)
2.7. Quantification of Bacteria
3. Results and Discussion
3.1. Physical Parameters
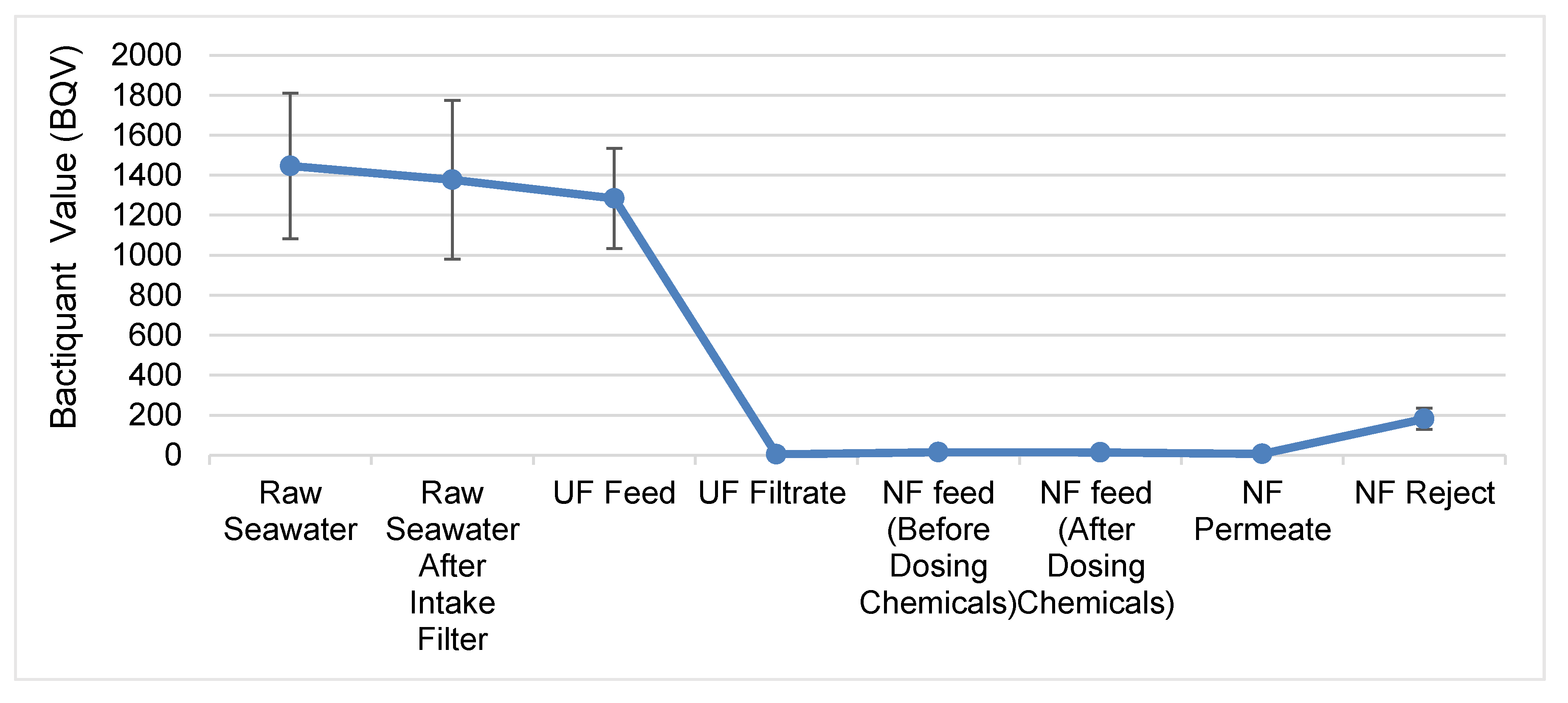
3.2. UF Membranes Remove Bacteria Effectively
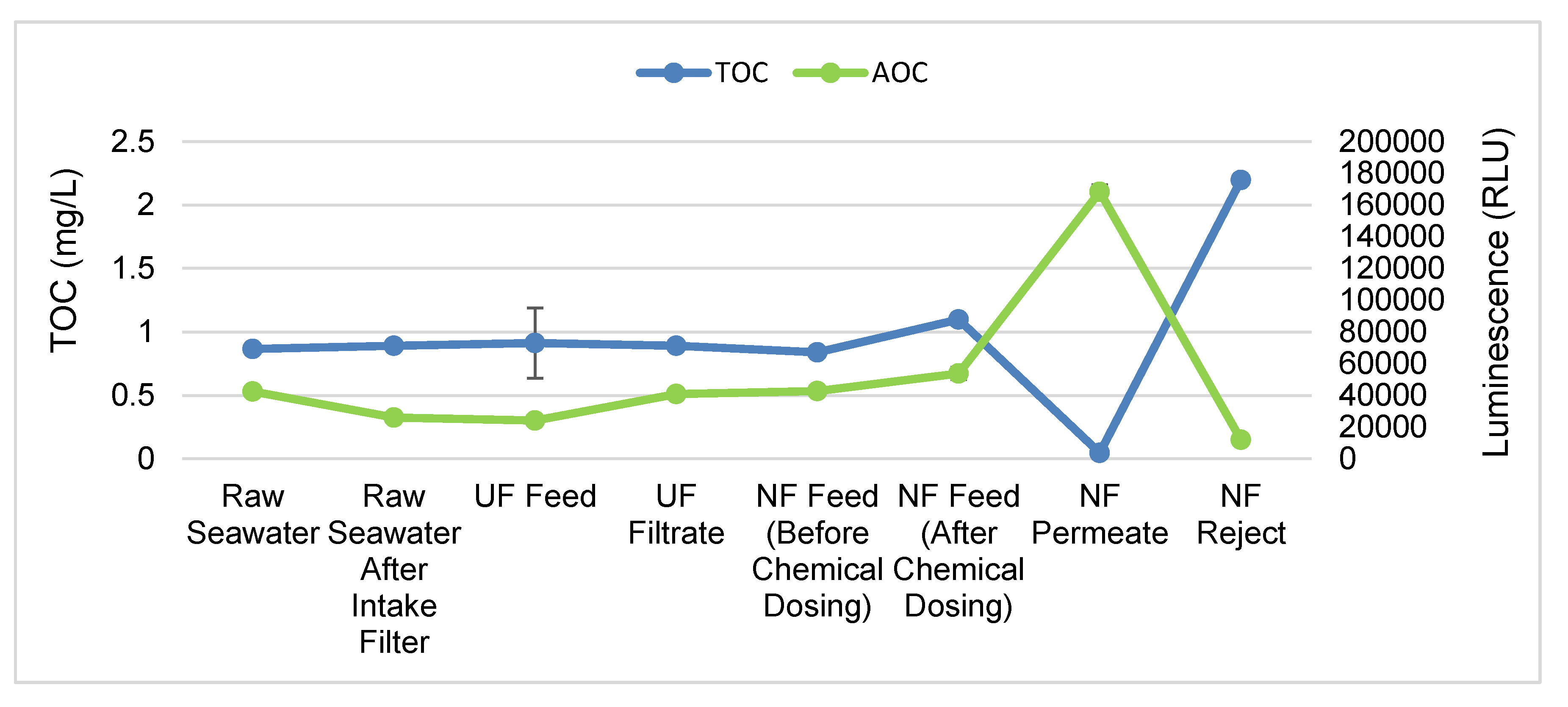
3.3. NF Membrane Removes TOC from Water More Effectively
3.4. Treatment with UF and NF Membranes Leads to Increase in AOC in the Permeate
3.5. CEB of UF Membrane Decreases AOC in UF Permeate
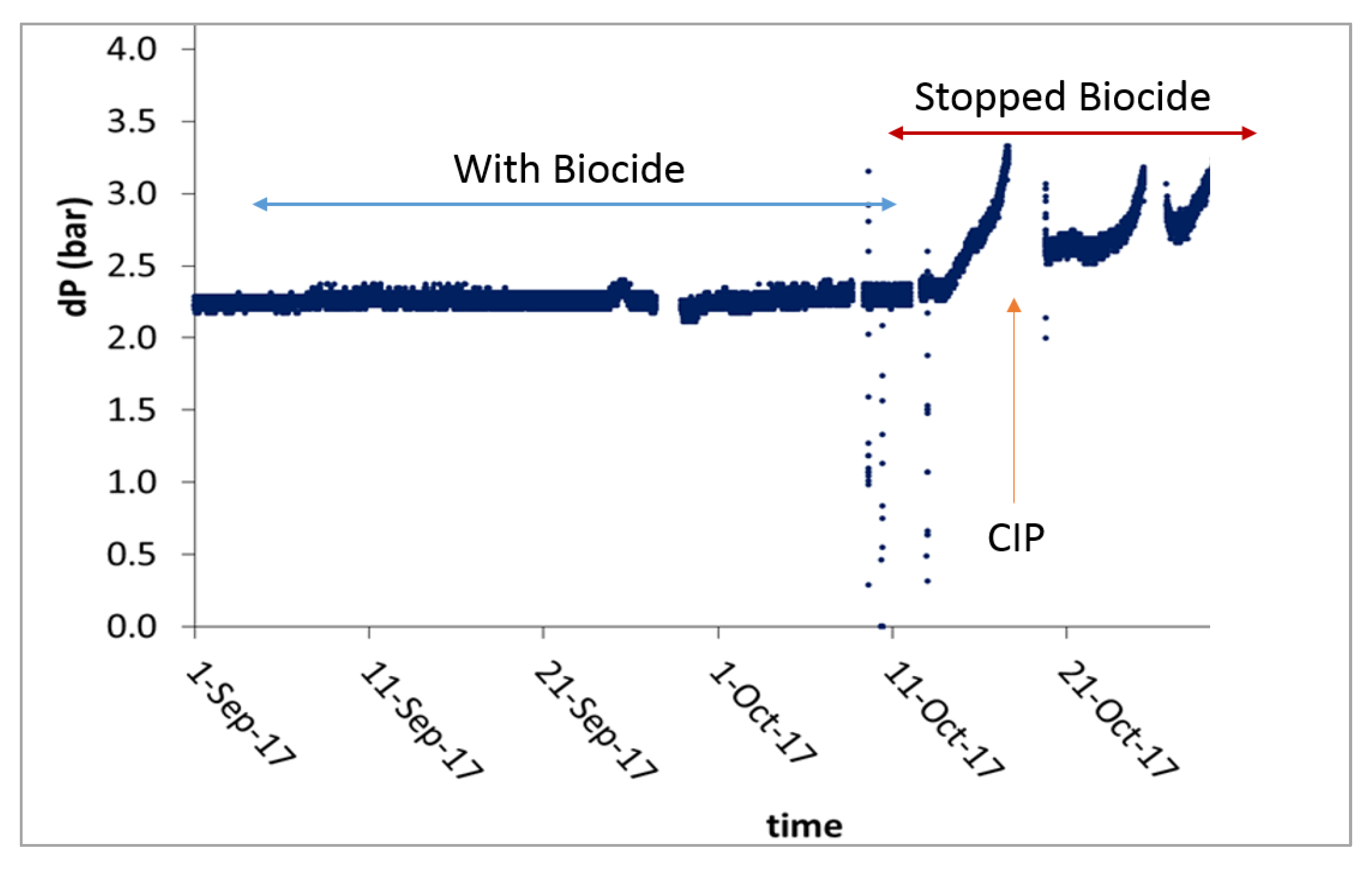
3.6. Biocide DBNPA Is Effective at Killing Bacterial Cells in NF Feed
3.7. LC-OCD Analysis Suggests AOC Is Primarily Composed of LMW Neutral Substances
4. Conclusions
- UF membranes are effective at removing bacterial cells but not organics.
- The antiscalant contributes humic-like organic substances to the water.
- The NF membrane used in this study effectively removes bacteria and TOC but not AOC. We found an increased AOC concentration in the NF permeate.
- LMW neutral sustances constitute a significant component of AOC.
- The use of the biocide DBNPA and CIP improves the performance of the NF membrane by delaying the pressure drop.
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Ghaffour, N.; Missimer, T.M.; Amy, G.L. Technical review and evaluation of the economics of water desalination: Current and future challenges for better water supply sustainability. Desalination 2013, 309, 197–207. [Google Scholar] [CrossRef]
- Weinrich, L.; LeChevallier, M.; Haas, C.N. Contribution of assimilable organic carbon to biological fouling in seawater reverse osmosis membrane treatment. Water Res. 2016, 101, 203–213. [Google Scholar] [CrossRef] [PubMed]
- Rehman, Z.U.; Ali, M.; Iftikhar, H.; Leiknes, T. Genome-resolved metagenomic analysis reveals roles of microbial community members in full-scale seawater reverse osmosis plant. Water Res. 2019, 149, 263–271. [Google Scholar] [CrossRef] [PubMed]
- Vanderkooij, D. Assimilable Organic-Carbon as an Indicator of Bacterial Regrowth. J. Am. Water Work. Assoc. 1992, 84, 57–65. [Google Scholar]
- Huang, S.Q.; Voutchkov, N.; Jiang, S. Balancing carbon, nitrogen and phosphorus concentration in seawater as a strategy to prevent accelerated membrane biofouling. Water Res. 2019, 165, 114978. [Google Scholar] [CrossRef]
- Zhang, W.X.; Jiang, F. Membrane fouling in aerobic granular sludge (AGS)-membrane bioreactor (MBR): Effect of AGS size. Water Res. 2019, 157, 445–453. [Google Scholar] [CrossRef]
- Meng, S.; Meng, X.; Fan, W.; Liang, D.; Wang, L.; Zhang, W.; Liu, Y. The role of transparent exopolymer particles (TEP) in membrane fouling: A critical review. Water Res. 2020, 181, 115930. [Google Scholar] [CrossRef]
- Kucera, J. Biofouling of Polyamide Membranes: Fouling Mechanisms, Current Mitigation and Cleaning Strategies, and Future Prospects. Membranes 2019, 9, 111. [Google Scholar] [CrossRef]
- Escobar, I.C.; Randall, A.A. Case study: Ozonation and distribution system biostability. J. Am. Water Work. Assoc. 2001, 93, 77–89. [Google Scholar] [CrossRef]
- Valavala, R.; Sohn, J.; Han, J.; Her, N.; Yoon, Y. Pretreatment in Reverse Osmosis Seawater Desalination: A Short Review. Environ. Eng. Res. 2011, 16, 205–212. [Google Scholar] [CrossRef]
- Baker, R.W. Membrane Technology and Applications, 3rd ed.; John Wiley & Sons: Hoboken, NJ, USA, 2012. [Google Scholar]
- Pearce, G.K. The case for UF/MF pretreatment to RO in seawater applications. Desalination 2007, 203, 286–295. [Google Scholar] [CrossRef]
- Lorain, O.; Hersant, B.; Persin, F.; Grasmick, A.; Brunard, N.; Espenan, J.M. Ultrafiltration membrane pre-treatment benefits for reverse osmosis process in seawater desalting. Quantification in terms of capital investment cost and operating cost reduction. Desalination 2007, 203, 277–285. [Google Scholar] [CrossRef]
- Semiao, A.J.C.; Gazzola, G.; Habimana, O.; Heffernan, R.; Murphy, C.; Casey, E. Understanding the mechanisms of biofouling on nanofiltration membranes: Effect of the biofilm structure on solute removal. Procedia Eng. 2012, 44, 1557–1560. [Google Scholar] [CrossRef]
- Vrouwenvelder, J.S.; Hinrichs, C.; Van der Meer, W.G.J.; Van Loosdrecht, M.C.M.; Kruithof, J.C. Pressure drop increase by biofilm accumulation in spiral wound RO and NF membrane systems: Role of substrate concentration, flow velocity, substrate load and flow direction. Biofouling 2009, 25, 543–555. [Google Scholar] [CrossRef]
- Meylan, S.; Hammes, F.; Traber, J.; Salhi, E.; von Gunten, U.; Pronk, W. Permeability of low molecular weight organics through nanofiltration membranes. Water Res. 2007, 41, 3968–3976. [Google Scholar] [CrossRef]
- Naz, A.; Sattar, R.; Siddiq, M. Polymer membranes for biofouling mitigation: A review. Polym. Plast. Technol. Mater. 2019, 58, 1829–1854. [Google Scholar] [CrossRef]
- Jeong, S.; Naidu, G.; Vigneswaran, S.; Ma, C.H.; Rice, S.A. A rapid bioluminescence-based test of assimilable organic carbon for seawater. Desalination 2013, 317, 160–165. [Google Scholar] [CrossRef]
- Izadpanah, A.A.; Javidnia, A. The Ability of a Nanofiltration Membrane to Remove Hardness and Ions from Diluted Seawater. Water 2012, 4, 283–294. [Google Scholar] [CrossRef]
- Hong, P.Y.; Moosa, N.; Mink, J. Dynamics of microbial communities in an integrated ultrafiltration-reverse osmosis desalination pilot plant located at theArabian Gulf. Desalin. Water Treat. 2016, 57, 16310–16323. [Google Scholar] [CrossRef]
- Fujioka, T.; Hoang, A.T.; Ueyama, T.; Nghiem, L.D. Integrity of reverse osmosis membrane for removing bacteria: New insight into bacterial passage Electronic supplementary information (ESI) available. Environ. Sci. Water Res. Technol. 2019, 5, 239–245. [Google Scholar] [CrossRef]
- Dehwah, A.H.A.; Li, S.; Al-Mashharawi, S.; Winters, H.; Missimer, T.M. Changes in feedwater organic matter concentrations based on intake type and pretreatment processes at SWRO facilities, Red Sea, Saudi Arabia. Desalination 2015, 360, 19–27. [Google Scholar] [CrossRef]
- Sweity, A.; Ronen, Z.; Herzberg, M. Induced organic fouling with antiscalants in seawater desalination. Desalination 2014, 352, 158–165. [Google Scholar] [CrossRef]
- Schafer, A.I.; Fane, A.G.; Waite, T.D. Cost factors and chemical pretreatment effects in the membrane filtration of waters containing natural organic matter. Water Res. 2001, 35, 1509–1517. [Google Scholar] [CrossRef]
- Lee, J.; Walker, H.W. Effect of process variables and natural organic matter on removal of microcystin-LR by PAC-UF. Environ. Sci. Technol. 2006, 40, 7336–7342. [Google Scholar] [CrossRef] [PubMed]
- Tsujimoto, W.; Kimura, H.; Izu, T.; Irie, T. Membrane filtration and pre-treatment by GAC. Desalination 1998, 119, 323–326. [Google Scholar] [CrossRef]
- Upadhyayula, V.K.K.; Deng, S.G.; Mitchell, M.C.; Smith, G.B. Application of carbon nanotube technology for removal of contaminants in drinking water: A review. Sci. Total Environ. 2009, 408, 1–13. [Google Scholar] [CrossRef]
- Derlon, N.; Mimoso, J.; Klein, T.; Koetzsch, S.; Morgenroth, E. Presence of biofilms on ultrafiltration membrane surfaces increases the quality of permeate produced during ultra-low pressure gravity-driven membrane filtration. Water Res. 2014, 60, 164–173. [Google Scholar] [CrossRef]
- Velten, S.; Boller, M.; Koster, O.; Helbing, J.; Weilenmann, H.U.; Hammes, F. Development of biomass in a drinking water granular active carbon (GAC) filter. Water Res. 2011, 45, 6347–6354. [Google Scholar] [CrossRef]
- Van der Bruggen, B.; Schaep, J.; Wilms, D.; Vandecasteele, C. Influence of molecular size, polarity and charge on the retention of organic molecules by nanofiltration. J. Membr. Sci. 1999, 156, 29–41. [Google Scholar] [CrossRef]
- Lou, J.-C.; Yang, C.-Y.; Chang, C.-J.; Chen, W.-H.; Tseng, W.-B.; Han, J.-Y. Analysis and removal of assimilable organic carbon (AOC) from treated drinking water using a biological activated carbon filter system. J. Environ. Chem. Eng. 2014, 2, 1684–1690. [Google Scholar] [CrossRef]
- Siddiqui, A.; Pinel, I.; Prest, E.I.; Bucs, S.S.; van Loosdrecht, M.C.M.; Kruithof, J.C.; Vrouwenvelder, J.S. Application of DBNPA dosage for biofouling control in spiral wound membrane systems. Desalin. Water Treat. 2017, 68, 12–22. [Google Scholar] [CrossRef]
- Hammes, F.; Meylan, S.; Salhi, E.; Koster, O.; Egli, T.; Von Gunten, U. Formation of assimilable organic carbon (AOC) and specific natural organic matter (NOM) fractions during ozonation of phytoplankton. Water Res. 2007, 41, 1447–1454. [Google Scholar] [CrossRef] [PubMed]

| Ultrafiltration Membrane | Nanofiltration Membrane | |
|---|---|---|
| Membrane Type | Polyvinylidene fluoride(PVDF) | Thin-film composite |
| Nominal Pore Diameter (nm) | 30 | 1 |
| Surface area (m2) | 77 | 40.9 |
| Average Flux (LMH) | 78 | 28 |
| Feed flow (m3/h) | 30 | 13.74 |
| System Pressure (bar) | 13–18 | 4–6 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rehman, Z.U.; Khojah, B.; Leiknes, T.; Alsogair, S.; Alsomali, M. Removal of Bacteria and Organic Carbon by an Integrated Ultrafiltration—Nanofiltration Desalination Pilot Plant. Membranes 2020, 10, 223. https://doi.org/10.3390/membranes10090223
Rehman ZU, Khojah B, Leiknes T, Alsogair S, Alsomali M. Removal of Bacteria and Organic Carbon by an Integrated Ultrafiltration—Nanofiltration Desalination Pilot Plant. Membranes. 2020; 10(9):223. https://doi.org/10.3390/membranes10090223
Chicago/Turabian StyleRehman, Zahid Ur, Bayan Khojah, TorOve Leiknes, Safiya Alsogair, and Mona Alsomali. 2020. "Removal of Bacteria and Organic Carbon by an Integrated Ultrafiltration—Nanofiltration Desalination Pilot Plant" Membranes 10, no. 9: 223. https://doi.org/10.3390/membranes10090223
APA StyleRehman, Z. U., Khojah, B., Leiknes, T., Alsogair, S., & Alsomali, M. (2020). Removal of Bacteria and Organic Carbon by an Integrated Ultrafiltration—Nanofiltration Desalination Pilot Plant. Membranes, 10(9), 223. https://doi.org/10.3390/membranes10090223






