The Efficacy of COVID-19 Vaccines in Chronic Kidney Disease and Kidney Transplantation Patients: A Narrative Review
Abstract
1. Introduction
2. COVID-19 Pandemic and CKD: The Higher Incidence of Critical Illness and Acute Kidney Injury in Patients with CKD
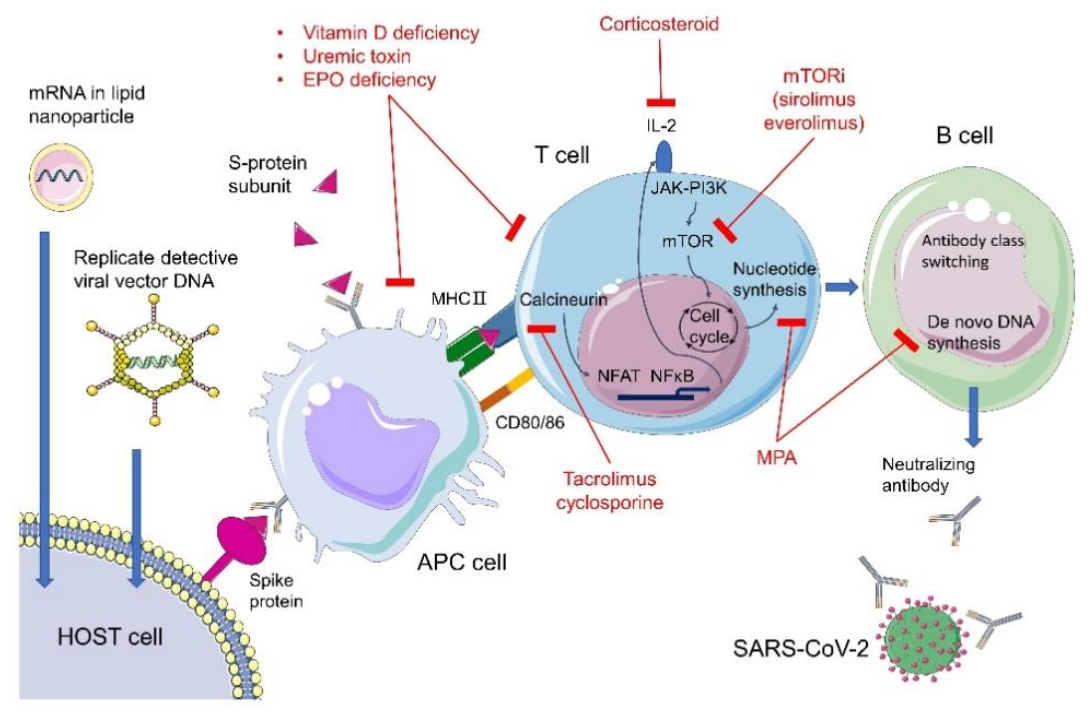
3. COVID-19 Vaccines of Choice in Preventing CKD Patients from Critical Illness
3.1. Replication-Defective Viral Vector Carrying Pathogen Gene(s)
3.2. mRNA Vaccines
3.3. Purified Virus Components
4. COVID-19 Vaccine Efficacy in CKD Patients: From CKD3-5 and CKD-5d to Post Kidney Transplantation
4.1. COVID-19 Vaccination for CKD 3-5 Patients Not on Immunosuppressive Agents
4.2. COVID-19 Vaccination for CKD 3-5 Patients on Immunosuppressive Agents
4.3. COVID-19 Vaccination for CKD-5d Patients
4.4. COVID-19 in Renal Transplantation Patients: The Effect of Immunosuppressants on Vaccine Efficacy
4.4.1. For the Recipients with Maintenance Use of Corticosteroids
4.4.2. For the Recipients with Maintenance Use of Calcineurin Inhibitors
4.4.3. For the Recipients with Maintenance Use of Antimetabolites
4.4.4. For the Recipients with Maintenance Use of mTORis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ACE-2 | angiotensin-converting enzyme 2 |
| AKI | acute kidney injury |
| Akt | protein kinase B (PKB/AKT) |
| CD | Cluster differentiation |
| CKD | chronic kidney disease |
| CNI | Calcineurin inhibitors |
| COVID-19 | coronavirus 19 disease |
| eGFR | estimated glomerular filtration rate |
| EPO | Erythropoietin |
| ESRD | end stage renal disease. |
| GN | glomerulonephritis |
| HD | hemodialysis |
| KT | kidney transplantation |
| MPA | Mycophenolic acid |
| mTOR | mechanistic target of rapamycin |
| NF-kB | nuclear factor kappa-light-chain-enhancer of activated B cells |
| PI3K | phosphatidylinositol 3-kinase |
| RAAS | renin-angiogenin-aldosterone system |
| SARS-CoV-2 | severe acute respiratory syndrome coronavirus 2 |
References
- Zhu, N.; Zhang, D.; Wang, W.; Li, X.; Yang, B.; Song, J.; Zhao, X.; Huang, B.; Shi, W.; Lu, R.; et al. A Novel Coronavirus from Patients with Pneumonia in China, 2019. N. Engl. J. Med. 2020, 382, 727–733. [Google Scholar] [CrossRef]
- Scialo, F.; Daniele, A.; Amato, F.; Pastore, L.; Matera, M.G.; Cazzola, M. ACE2: The Major Cell Entry Receptor for SARS-CoV-2. Lung 2020, 198, 867–877. [Google Scholar] [CrossRef]
- Davidson, A.M.; Wysocki, J.; Batlle, D. Interaction of SARS-CoV-2 and Other Coronavirus With ACE (Angiotensin-Converting Enzyme)-2 as Their Main Receptor. Hypertension 2020, 76, 1339–1349. [Google Scholar] [CrossRef]
- Iwasaki, M.; Saito, J.; Zhao, H.; Sakamoto, A.; Hirota, K.; Ma, D. Inflammation Triggered by SARS-CoV-2 and ACE2 Augment Drives Multiple Organ Failure of Severe COVID-19: Molecular Mechanisms and Implications. Inflammation 2021, 44, 13–34. [Google Scholar] [CrossRef]
- Alam, M.R.; Kabir, M.R.; Reza, S. Comorbidities might be a risk factor for the incidence of COVID-19: Evidence from a web-based survey. Prev. Med. Rep. 2021, 21, 101319. [Google Scholar] [CrossRef]
- Huang, C.; Soleimani, J.; Herasevich, S.; Pinevich, Y.; Pennington, K.M.; Dong, Y.; Pickering, B.W.; Barwise, A.K. Clinical Characteristics, Treatment, and Outcomes of Critically Ill Patients With COVID-19: A Scoping Review. Mayo Clin. Proc. 2021, 96, 183–202. [Google Scholar] [CrossRef]
- Kant, S.; Menez, S.P.; Hanouneh, M.; Fine, D.M.; Crews, D.C.; Brennan, D.C.; Sperati, C.J.; Jaar, B.G. The COVID-19 nephrology compendium: AKI, CKD, ESKD and transplantation. BMC Nephrol. 2020, 21, 449. [Google Scholar] [CrossRef]
- Forni, G.; Mantovani, A.; Forni, G.; Mantovani, A.; Moretta, L.; Rappuoli, R.; Rezza, G.; Bagnasco, A.; Barsacchi, G.; Bussolati, G.; et al. COVID-19 vaccines: Where we stand and challenges ahead. Cell Death Differ. 2021, 28, 626–639. [Google Scholar] [CrossRef]
- Menon, R.; Otto, E.A.; Sealfon, R.; Nair, V.; Wong, A.K.; Theesfeld, C.L.; Chen, X.; Wang, Y.; Boppana, A.S.; Luo, J.; et al. SARS-CoV-2 receptor networks in diabetic and COVID-19-associated kidney disease. Kidney Int. 2020, 98, 1502–1518. [Google Scholar] [CrossRef]
- Singh, A.K.; Gillies, C.L.; Singh, R.; Singh, A.; Chudasama, Y.; Coles, B.; Seidu, S.; Zaccardi, F.; Davies, M.J.; Khunti, K. Prevalence of co-morbidities and their association with mortality in patients with COVID-19: A systematic review and meta-analysis. Diabetes Obes. Metab. 2020, 22, 1915–1924. [Google Scholar] [CrossRef]
- Cai, R.; Zhang, J.; Zhu, Y.; Liu, L.; Liu, Y.; He, Q. Mortality in chronic kidney disease patients with COVID-19: A systematic review and meta-analysis. Int. Urol. Nephrol. 2021, 53, 1623–1629. [Google Scholar] [CrossRef] [PubMed]
- Gansevoort, R.T.; Hilbrands, L.B. CKD is a key risk factor for COVID-19 mortality. Nat. Rev. Nephrol. 2020, 16, 705–706. [Google Scholar] [CrossRef]
- Lin, Y.-C.; Lai, T.-S.; Lin, S.-L.; Chen, Y.-M.; Chu, T.-S.; Tu, Y.-K. Outcomes of coronavirus 2019 infection in patients with chronic kidney disease: A systematic review and meta-analysis. Ther. Adv. Chronic Dis. 2021, 12. [Google Scholar] [CrossRef] [PubMed]
- Pakhchanian, H.; Raiker, R.; Mukherjee, A.; Khan, A.; Singh, S.; Chatterjee, A. Outcomes of COVID-19 in CKD Patients. Clin. J. Am. Soc. Nephrol. 2021, 16, 785. [Google Scholar] [CrossRef]
- Hsu, C.M.; Weiner, D.E.; Aweh, G.; Miskulin, D.C.; Manley, H.J.; Stewart, C.; Ladik, V.; Hosford, J.; Lacson, E.C.; Johnson, D.S.; et al. COVID-19 Among US Dialysis Patients: Risk Factors and Outcomes From a National Dialysis Provider. Am. J. Kidney Dis. 2021, 77, 748–756.e741. [Google Scholar] [CrossRef]
- Valeri, A.M.; Robbins-Juarez, S.Y.; Stevens, J.S.; Ahn, W.; Rao, M.K.; Radhakrishnan, J.; Gharavi, A.G.; Mohan, S.; Husain, S.A. Presentation and Outcomes of Patients with ESKD and COVID-19. J. Am. Soc. Nephrol. 2020, 31, 1409. [Google Scholar] [CrossRef]
- Puelles, V.G.; Lütgehetmann, M.; Lindenmeyer, M.T.; Sperhake, J.P.; Wong, M.N.; Allweiss, L.; Chilla, S.; Heinemann, A.; Wanner, N.; Liu, S.; et al. Multiorgan and Renal Tropism of SARS-CoV-2. N. Engl. J. Med. 2020, 383, 590–592. [Google Scholar] [CrossRef]
- Kosugi, T.; Maeda, K.; Sato, W.; Maruyama, S.; Kadomatsu, K. CD147 (EMMPRIN/Basigin) in kidney diseases: From an inflammation and immune system viewpoint. Nephrol. Dial. Transplant. Off. Publ. Eur. Dial. Transpl. Assoc. Eur. Ren. Assoc. 2015, 30, 1097–1103. [Google Scholar] [CrossRef]
- Lowe, R.; Ferrari, M.; Nasim-Mohi, M.; Jackson, A.; Beecham, R.; Veighey, K.; Cusack, R.; Richardson, D.; Grocott, M.P.W.; Levett, D.Z.H.; et al. Clinical characteristics and outcome of critically ill COVID-19 patients with acute kidney injury: A single centre cohort study. BMC Nephrol. 2021, 22, 92. [Google Scholar] [CrossRef] [PubMed]
- Charytan, D.M.; Parnia, S.; Khatri, M.; Petrilli, C.M.; Jones, S.; Benstein, J.; Horwitz, L.I. Decreasing Incidence of Acute Kidney Injury in Patients with COVID-19 Critical Illness in New York City. Kidney Int. Rep. 2021, 6, 916–927. [Google Scholar] [CrossRef]
- He, Y.; Li, J.; Heck, S.; Lustigman, S.; Jiang, S. Antigenic and immunogenic characterization of recombinant baculovirus-expressed severe acute respiratory syndrome coronavirus spike protein: Implication for vaccine design. J. Virol. 2006, 80, 5757–5767. [Google Scholar] [CrossRef] [PubMed]
- Lundstrom, K. Viral Vectors for COVID-19 Vaccine Development. Viruses 2021, 13, 317. [Google Scholar] [CrossRef]
- Folegatti, P.M.; Ewer, K.J.; Aley, P.K.; Angus, B.; Becker, S.; Belij-Rammerstorfer, S.; Bellamy, D.; Bibi, S.; Bittaye, M.; Clutterbuck, E.A.; et al. Safety and immunogenicity of the ChAdOx1 nCoV-19 vaccine against SARS-CoV-2: A preliminary report of a phase 1/2, single-blind, randomised controlled trial. Lancet 2020, 396, 467–478. [Google Scholar] [CrossRef]
- Sadoff, J.; Gray, G.; Vandebosch, A.; Cárdenas, V.; Shukarev, G.; Grinsztejn, B.; Goepfert, P.A.; Truyers, C.; Fennema, H.; Spiessens, B.; et al. Safety and Efficacy of Single-Dose Ad26.COV2.S Vaccine against Covid-19. N. Engl. J. Med. 2021, 384, 2187–2201. [Google Scholar] [CrossRef] [PubMed]
- Logunov, D.Y.; Dolzhikova, I.V.; Zubkova, O.V.; Tukhvatulin, A.I.; Shcheblyakov, D.V.; Dzharullaeva, A.S.; Grousova, D.M.; Erokhova, A.S.; Kovyrshina, A.V.; Botikov, A.G.; et al. Safety and immunogenicity of an rAd26 and rAd5 vector-based heterologous prime-boost COVID-19 vaccine in two formulations: Two open, non-randomised phase 1/2 studies from Russia. Lancet 2020, 396, 887–897. [Google Scholar] [CrossRef]
- Baden, L.R.; El Sahly, H.M.; Essink, B.; Kotloff, K.; Frey, S.; Novak, R.; Diemert, D.; Spector, S.A.; Rouphael, N.; Creech, C.B.; et al. Efficacy and Safety of the mRNA-1273 SARS-CoV-2 Vaccine. N. Engl. J. Med. 2020, 384, 403–416. [Google Scholar] [CrossRef]
- Polack, F.P.; Thomas, S.J.; Kitchin, N.; Absalon, J.; Gurtman, A.; Lockhart, S.; Perez, J.L.; Pérez Marc, G.; Moreira, E.D.; Zerbini, C.; et al. Safety and Efficacy of the BNT162b2 mRNA Covid-19 Vaccine. N. Engl. J. Med. 2020, 383, 2603–2615. [Google Scholar] [CrossRef] [PubMed]
- Khoury, D.S.; Cromer, D.; Reynaldi, A.; Schlub, T.E.; Wheatley, A.K.; Juno, J.A.; Subbarao, K.; Kent, S.J.; Triccas, J.A.; Davenport, M.P. Neutralizing antibody levels are highly predictive of immune protection from symptomatic SARS-CoV-2 infection. Nat. Med. 2021. [Google Scholar] [CrossRef]
- Pritchard, E.; Matthews, P.C.; Stoesser, N.; Eyre, D.W.; Gethings, O.; Vihta, K.-D.; Jones, J.; House, T.; VanSteenHouse, H.; Bell, I.; et al. Impact of vaccination on new SARS-CoV-2 infections in the United Kingdom. Nat. Med. 2021. [Google Scholar] [CrossRef]
- Tian, J.-H.; Patel, N.; Haupt, R.; Zhou, H.; Weston, S.; Hammond, H.; Lague, J.; Portnoff, A.D.; Norton, J.; Guebre-Xabier, M.; et al. SARS-CoV-2 spike glycoprotein vaccine candidate NVX-CoV2373 elicits immunogenicity in baboons and protection in mice. bioRxiv 2020, 12, 1–4. [Google Scholar] [CrossRef]
- Glenn, D.A.; Hegde, A.; Kotzen, E.; Walter, E.B.; Kshirsagar, A.V.; Falk, R.; Mottl, A. Systematic Review of Safety and Efficacy of COVID-19 Vaccines in Patients With Kidney Disease. Kidney Int. Rep. 2021, 6, 1407–1410. [Google Scholar] [CrossRef]
- Billany, R.E.; Selvaskandan, H.; Adenwalla, S.F.; Hull, K.L.; March, D.S.; Burton, J.O.; Bishop, N.C.; Carr, E.J.; Beale, R.; Tang, J.W.; et al. Seroprevalence of antibody to S1 spike protein following vaccination against COVID-19 in patients receiving hemodialysis: A call to arms. Kidney Int. 2021, 99, 1492–1494. [Google Scholar] [CrossRef]
- Yanay, N.B.; Freiman, S.; Shapira, M.a.; Wishahi, S.; Hamze, M.; Elhaj, M.; Zaher, M.; Armaly, Z. Experience with SARS-CoV-2 BNT162b2 mRNA vaccine in dialysis patients. Kidney Int. 2021, 99, 1496–1498. [Google Scholar] [CrossRef] [PubMed]
- Firket, L.; Descy, J.; Seidel, L.; Bonvoisin, C.; Bouquegneau, A.; Grosch, S.; Jouret, F.; Weekers, L. Serological response to mRNA SARS-CoV-2 BNT162b2 vaccine in kidney transplant recipients depends on prior exposure to SARS-CoV-2. Am. J. Transplant. 2021. [Google Scholar] [CrossRef]
- Kamar, N.; Abravanel, F.; Marion, O.; Couat, C.; Izopet, J.; Del Bello, A. Three Doses of an mRNA Covid-19 Vaccine in Solid-Organ Transplant Recipients. N. Engl. J. Med. 2021. [Google Scholar] [CrossRef]
- Rodríguez-Espinosa, D.; Broseta, J.J.; Maduell, F.; Bedini, J.L.; Vera, M. Humoral response of the mRNA-1273 SARS-CoV-2 vaccine in peritoneal dialysis patients. Kidney Int. 2021. [Google Scholar] [CrossRef] [PubMed]
- Benotmane, I.; Gautier-Vargas, G.; Cognard, N.; Olagne, J.; Heibel, F.; Braun-Parvez, L.; Martzloff, J.; Perrin, P.; Moulin, B.; Fafi-Kremer, S.; et al. Low immunization rates among kidney transplant recipients who received 2 doses of the mRNA-1273 SARS-CoV-2 vaccine. Kidney Int. 2021, 99, 1498–1500. [Google Scholar] [CrossRef] [PubMed]
- Syed-Ahmed, M.; Narayanan, M. Immune Dysfunction and Risk of Infection in Chronic Kidney Disease. Adv. Chronic Kidney Dis. 2019, 26, 8–15. [Google Scholar] [CrossRef]
- Girndt, M.; Sester, M.; Sester, U.; Kaul, H.; Köhler, H. Defective expression of B7-2 (CD86) on monocytes of dialysis patients correlates to the uremia-associated immune defect. Kidney Int. 2001, 59, 1382–1389. [Google Scholar] [CrossRef]
- Ando, M.; Shibuya, A.; Tsuchiya, K.; Akiba, T.; Nitta, K. Reduced expression of Toll-like receptor 4 contributes to impaired cytokine response of monocytes in uremic patients. Kidney Int. 2006, 70, 358–362. [Google Scholar] [CrossRef] [PubMed]
- Eleftheriadis, T.; Antoniadi, G.; Liakopoulos, V.; Kartsios, C.; Stefanidis, I. Basic Science and Dialysis: Disturbances of Acquired Immunity in Hemodialysis Patients. Semin. Dial. 2007, 20, 440–451. [Google Scholar] [CrossRef]
- Kim, K.W.; Chung, B.H.; Jeon, E.J.; Kim, B.M.; Choi, B.S.; Park, C.W.; Kim, Y.S.; Cho, S.G.; Cho, M.L.; Yang, C.W. B cell-associated immune profiles in patients with end-stage renal disease (ESRD). Exp. Mol. Med. 2012, 44, 465–472. [Google Scholar] [CrossRef] [PubMed]
- Pahl, M.V.; Gollapudi, S.; Sepassi, L.; Gollapudi, P.; Elahimehr, R.; Vaziri, N.D. Effect of end-stage renal disease on B-lymphocyte subpopulations, IL-7, BAFF and BAFF receptor expression. Nephrol. Dial. Transplant. Off. Publ. Eur. Dial. Transpl. Assoc. Eur. Ren. Assoc. 2010, 25, 205–212. [Google Scholar] [CrossRef]
- Frasca, D.; Diaz, A.; Romero, M.; Mendez, N.V.; Landin, A.M.; Ryan, J.G.; Blomberg, B.B. Young and elderly patients with type 2 diabetes have optimal B cell responses to the seasonal influenza vaccine. Vaccine 2013, 31, 3603–3610. [Google Scholar] [CrossRef][Green Version]
- Souma, T.; Yamazaki, S.; Moriguchi, T.; Suzuki, N.; Hirano, I.; Pan, X.; Minegishi, N.; Abe, M.; Kiyomoto, H.; Ito, S.; et al. Plasticity of renal erythropoietin-producing cells governs fibrosis. J. Am. Soc. Nephrol. 2013, 24, 1599–1616. [Google Scholar] [CrossRef]
- Cantarelli, C.; Angeletti, A.; Cravedi, P. Erythropoietin, a multifaceted protein with innate and adaptive immune modulatory activity. Am. J. Transplant. 2019, 19, 2407–2414. [Google Scholar] [CrossRef] [PubMed]
- Brines, M.; Patel, N.S.; Villa, P.; Brines, C.; Mennini, T.; De Paola, M.; Erbayraktar, Z.; Erbayraktar, S.; Sepodes, B.; Thiemermann, C.; et al. Nonerythropoietic, tissue-protective peptides derived from the tertiary structure of erythropoietin. Proc. Natl. Acad. Sci. USA 2008, 105, 10925–10930. [Google Scholar] [CrossRef]
- Ehrenreich, H.; Weissenborn, K.; Begemann, M.; Busch, M.; Vieta, E.; Miskowiak, K.W. Erythropoietin as candidate for supportive treatment of severe COVID-19. Mol. Med. 2020, 26, 58. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.H.; Liu, Y.L.; Lin, H.H.; Yang, Y.F.; Kuo, H.L.; Lin, P.W.; Huang, C.C. Intravenous iron attenuates postvaccination anti-HBsAg titres after quadruple hepatitis B vaccination in dialysis patients with erythropoietin therapy. Int. J. Clin. Pract. 2009, 63, 387–393. [Google Scholar] [CrossRef] [PubMed]
- Hou, Y.C.; Liu, W.C.; Zheng, C.M.; Zheng, J.Q.; Yen, T.H.; Lu, K.C. Role of Vitamin D in Uremic Vascular Calcification. BioMed Res. Int. 2017, 2017, 2803579. [Google Scholar] [CrossRef] [PubMed]
- Zheng, J.-Q.; Hou, Y.-C.; Zheng, C.-M.; Lu, C.-L.; Liu, W.-C.; Wu, C.-C.; Huang, M.-T.; Lin, Y.-F.; Lu, K.-C. Cholecalciferol Additively Reduces Serum Parathyroid Hormone and Increases Vitamin D and Cathelicidin Levels in Paricalcitol-Treated Secondary Hyperparathyroid Hemodialysis Patients. Nutrients 2016, 8, 708. [Google Scholar] [CrossRef] [PubMed]
- Zitt, E.; Sprenger-Mähr, H.; Knoll, F.; Neyer, U.; Lhotta, K. Vitamin D deficiency is associated with poor response to active hepatitis B immunisation in patients with chronic kidney disease. Vaccine 2012, 30, 931–935. [Google Scholar] [CrossRef] [PubMed]
- Tangpricha, V.; Wasse, H. Vitamin D Therapy in Kidney Disease: More Vitamin D Is Necessary. Am. J. Kidney Dis. 2014, 64, 667–669. [Google Scholar] [CrossRef]
- Wheeler, D.C.; Winkelmayer, W.C. KDIGO 2017 Clinical Practice Guideline Update for the Diagnosis, Evaluation, Prevention, and Treatment of Chronic Kidney Disease-Mineral and Bone Disorder (CKD-MBD). Kidney Int. Suppl. 2017, 7, 1–59. [Google Scholar] [CrossRef]
- Kliger, A.S.; Foley, R.N.; Goldfarb, D.S.; Goldstein, S.L.; Johansen, K.; Singh, A.; Szczech, L. KDOQI US commentary on the 2012 KDIGO Clinical Practice Guideline for Anemia in CKD. Am. J. Kidney Dis. 2013, 62, 849–859. [Google Scholar] [CrossRef]
- Floege, J.; Barbour, S.J.; Cattran, D.C.; Hogan, J.J.; Nachman, P.H.; Tang, S.C.W.; Wetzels, J.F.M.; Cheung, M.; Wheeler, D.C.; Winkelmayer, W.C.; et al. Management and treatment of glomerular diseases (part 1): Conclusions from a Kidney Disease: Improving Global Outcomes (KDIGO) Controversies Conference. Kidney Int. 2019, 95, 268–280. [Google Scholar] [CrossRef]
- Parikh, S.V.; Almaani, S.; Brodsky, S.; Rovin, B.H. Update on Lupus Nephritis: Core Curriculum 2020. Am. J. Kidney Dis. 2020, 76, 265–281. [Google Scholar] [CrossRef] [PubMed]
- Deepak, P.; Kim, W.; Paley, M.A.; Yang, M.; Carvidi, A.B.; El-Qunni, A.A.; Haile, A.; Huang, K.; Kinnett, B.; Liebeskind, M.J.; et al. Glucocorticoids and B Cell Depleting Agents Substantially Impair Immunogenicity of mRNA Vaccines to SARS-CoV-2. medRxiv 2021. [Google Scholar] [CrossRef]
- La Manna, A.; Polito, C.; Foglia, A.C.; Di Toro, A.; Cafaro, M.R.; Del Gado, R. Reduced response to hepatitis B virus vaccination in boys with steroid-sensitive nephrotic syndrome. Pediatr. Nephrol. 1992, 6, 251–253. [Google Scholar] [CrossRef]
- Negrea, L.; Rovin, B.H. Gross hematuria following vaccination for severe acute respiratory syndrome coronavirus 2 in 2 patients with IgA nephropathy. Kidney Int. 2021, 99, 1487. [Google Scholar] [CrossRef]
- Mancianti, N.; Guarnieri, A.; Tripodi, S.; Salvo, D.P.; Garosi, G. Minimal change disease following vaccination for SARS-CoV-2. J. Nephrol. 2021. [Google Scholar] [CrossRef]
- Zeng, W.; Ma, H.; Ding, C.; Yang, Y.; Sun, Y.; Huang, X.; He, W.; Xiang, Y.; Gao, Y.; Jin, T. Characterization of SARS-CoV-2-specific antibodies in COVID-19 patients reveals highly potent neutralizing IgA. Signal Transduct. Target. Ther. 2021, 6, 35. [Google Scholar] [CrossRef]
- Grupper, A.; Sharon, N.; Finn, T.; Cohen, R.; Israel, M.; Agbaria, A.; Rechavi, Y.; Schwartz, I.F.; Schwartz, D.; Lellouch, Y.; et al. Humoral Response to the Pfizer BNT162b2 Vaccine in Patients Undergoing Maintenance Hemodialysis. Clin. J. Am. Soc. Nephrol. 2021. [Google Scholar] [CrossRef]
- Lesny, P.; Anderson, M.; Cloherty, G.; Stec, M.; Haase-Fielitz, A.; Haarhaus, M.; Santos, C.; Lucas, C.; Macario, F.; Haase, M. Immunogenicity of a first dose of mRNA- or vector-based SARS-CoV-2 vaccination in dialysis patients: A multicenter prospective observational pilot study. J. Nephrol. 2021, 29, 1–9. [Google Scholar] [CrossRef]
- Ma, Y.; Diao, B.; Lv, X.; Zhu, J.; Liang, W.; Liu, L.; Bu, W.; Cheng, H.; Zhang, S.; Shi, M.; et al. 2019 novel coronavirus disease in hemodialysis (HD) patients: Report from one HD center in Wuhan, China. medRxiv 2020. [Google Scholar] [CrossRef]
- Labriola, L.; Scohy, A.; Seghers, F.; Perlot, Q.; De Greef, J.; Desmet, C.; Romain, C.; Morelle, J.; Yombi, J.-C.; Kabamba, B.; et al. A Longitudinal, 3-Month Serologic Assessment of SARS-CoV-2 Infections in a Belgian Hemodialysis Facility. Clin. J. Am. Soc. Nephrol. 2021, 16, 613. [Google Scholar] [CrossRef] [PubMed]
- Sadoff, J.; Le Gars, M.; Shukarev, G.; Heerwegh, D.; Truyers, C.; de Groot, A.M.; Stoop, J.; Tete, S.; Van Damme, W.; Leroux-Roels, I.; et al. Interim Results of a Phase 1–2a Trial of Ad26.COV2.S Covid-19 Vaccine. N. Engl. J. Med. 2021, 384, 1824–1835. [Google Scholar] [CrossRef] [PubMed]
- Halloran, P.F. Immunosuppressive Drugs for Kidney Transplantation. N. Engl. J. Med. 2004, 351, 2715–2729. [Google Scholar] [CrossRef]
- Kidney Disease: Improving Global Outcomes (KDIGO) Transplant Work Group. KDIGO clinical practice guideline for the care of kidney transplant recipients. Am. J. Transplant. Off. J. Am. Soc. Transplant. Am. Soc. Transpl. Surg. 2009, 9, S1–S155. [Google Scholar] [CrossRef]
- Mariat, C.; Alamartine, E.; Diab, N.; de Filippis, J.P.; Laurent, B.; Berthoux, F. A randomized prospective study comparing low-dose OKT3 to low-dose ATG for the treatment of acute steroid-resistant rejection episodes in kidney transplant recipients. Transpl. Int. 1998, 11, 231–236. [Google Scholar] [CrossRef]
- Zarkhin, V.; Li, L.; Kambham, N.; Sigdel, T.; Salvatierra, O.; Sarwal, M.M. A randomized, prospective trial of rituximab for acute rejection in pediatric renal transplantation. Am. J. Transplant. 2008, 8, 2607–2617. [Google Scholar] [CrossRef]
- Xie, P.; Tao, M.; Peng, K.; Zhao, H.; Zhang, K.; Sheng, Y.; Ankawi, G.; Ronco, C. Plasmapheresis Therapy in Kidney Transplant Rejection. Blood Purif. 2019, 47, 73–84. [Google Scholar] [CrossRef]
- Babu, T.M.; Kotton, C.N. Immunizations in Chronic Kidney Disease and Kidney Transplantation. Curr. Treat. Options Infect. Dis. 2021, 17, 1–19. [Google Scholar] [CrossRef]
- Baluch, A.; Humar, A.; Eurich, D.; Egli, A.; Liacini, A.; Hoschler, K.; Campbell, P.; Berka, N.; Urschel, S.; Wilson, L.; et al. Randomized controlled trial of high-dose intradermal versus standard-dose intramuscular influenza vaccine in organ transplant recipients. Am. J. Transplant. 2013, 13, 1026–1033. [Google Scholar] [CrossRef]
- Danziger-Isakov, L.; Cherkassky, L.; Siegel, H.; McManamon, M.; Kramer, K.; Budev, M.; Sawinski, D.; Augustine, J.J.; Hricik, D.E.; Fairchild, R.; et al. Effects of influenza immunization on humoral and cellular alloreactivity in humans. Transplantation 2010, 89, 838–844. [Google Scholar] [CrossRef] [PubMed]
- Ou, M.T.; Boyarsky, B.J.; Motter, J.D.; Greenberg, R.S.; Teles, A.T.; Ruddy, J.A.; Krach, M.R.; Jain, V.S.; Werbel, W.A.; Avery, R.K.; et al. Safety and Reactogenicity of 2 Doses of SARS-CoV-2 Vaccination in Solid Organ Transplant Recipients. Transplantation 2021. [Google Scholar] [CrossRef]
- Lentine, K.L.; Mannon, R.B.; Josephson, M.A. Practicing With Uncertainty: Kidney Transplantation During the COVID-19 Pandemic. Am. J. Kidney Dis. 2021, 77, 777–785. [Google Scholar] [CrossRef] [PubMed]
- Steiner, R.W.; Awdishu, L. Steroids in kidney transplant patients. Semin. Immunopathol. 2011, 33, 157–167. [Google Scholar] [CrossRef]
- Liberman, A.C.; Budziñski, M.L.; Sokn, C.; Gobbini, R.P.; Steininger, A.; Arzt, E. Regulatory and Mechanistic Actions of Glucocorticoids on T and Inflammatory Cells. Front. Endocrinol. 2018. [Google Scholar] [CrossRef] [PubMed]
- Hricik, D.E.; Almawi, W.Y.; Strom, T.B. Trends in the use of glucocorticoids in renal transplantation. Transplantation 1994, 57, 979–989. [Google Scholar] [CrossRef] [PubMed]
- de Roux, A.; Schmidt, N.; Rose, M.; Zielen, S.; Pletz, M.; Lode, H. Immunogenity of the pneumococcal polysaccharide vaccine in COPD patients. The effect of systemic steroids. Respir. Med. 2004, 98, 1187–1194. [Google Scholar] [CrossRef]
- Benotmane, I.; Gautier-Vargas, G.; Cognard, N.; Olagne, J.; Heibel, F.; Braun-Parvez, L.; Martzloff, J.; Perrin, P.; Moulin, B.; Fafi-Kremer, S.; et al. Weak anti-SARS-CoV-2 antibody response after the first injection of an mRNA COVID-19 vaccine in kidney transplant recipients. Kidney Int. 2021, 99, 1487–1489. [Google Scholar] [CrossRef]
- Rozen-Zvi, B.; Yahav, D.; Agur, T.; Zingerman, B.; Ben-Zvi, H.; Atamna, A.; Tau, N.; Mashraki, T.; Nesher, E.; Rahamimov, R. Antibody response to SARS-CoV-2 mRNA vaccine among kidney transplant recipients: A prospective cohort study. Clin. Microbiol. Infect. 2021. [Google Scholar] [CrossRef]
- Tsapepas, D.; Paget, K.; Mohan, S.; Cohen, D.J.; Husain, S.A. Clinically Significant COVID-19 Following SARS-CoV-2 Vaccination in Kidney Transplant Recipients. Am. J. Kidney Dis. 2021. [Google Scholar] [CrossRef]
- Kates, O.S.; Haydel, B.M.; Florman, S.S.; Rana, M.M.; Chaudhry, Z.S.; Ramesh, M.S.; Safa, K.; Kotton, C.N.; Blumberg, E.A.; Besharatian, B.D.; et al. COVID-19 in solid organ transplant: A multi-center cohort study. Clin. Infect. Dis. Off. Publ. Infect. Dis. Soc. Am. 2020. [Google Scholar] [CrossRef]
- Allison, A.C. Mechanisms of action of mycophenolate mofetil. Lupus 2005, 14 (Suppl. 1), 2–8. [Google Scholar] [CrossRef] [PubMed]
- Haneda, M.; Owaki, M.; Kuzuya, T.; Iwasaki, K.; Miwa, Y.; Kobayashi, T. Comparative analysis of drug action on B-cell proliferation and differentiation for mycophenolic acid, everolimus, and prednisolone. Transplantation 2014, 97, 405–412. [Google Scholar] [CrossRef] [PubMed]
- Karnell, J.L.; Karnell, F.G., 3rd; Stephens, G.L.; Rajan, B.; Morehouse, C.; Li, Y.; Swerdlow, B.; Wilson, M.; Goldbach-Mansky, R.; Groves, C.; et al. Mycophenolic acid differentially impacts B cell function depending on the stage of differentiation. J. Immunol. 2011, 187, 3603–3612. [Google Scholar] [CrossRef]
- Gunsilius, E. Immunosuppression and its complications. Mag. Eur. Med. Oncol. 2019, 12, 191. [Google Scholar] [CrossRef]
- Hou, Y.-C.; Chang, Y.-C.; Luo, H.-L.; Lu, K.-C.; Chiang, P.-H. Effect of mechanistic target of rapamycin inhibitors on postrenal transplantation malignancy: A nationwide cohort study. Cancer Med. 2018, 7, 4296–4307. [Google Scholar] [CrossRef] [PubMed]
- Garcia, G., Jr.; Sharma, A.; Ramaiah, A.; Sen, C.; Purkayastha, A.; Kohn, D.B.; Parcells, M.S.; Beck, S.; Kim, H.; Bakowski, M.A.; et al. Antiviral drug screen identifies DNA-damage response inhibitor as potent blocker of SARS-CoV-2 replication. Cell Rep. 2021, 35, 108940. [Google Scholar] [CrossRef]
| Type of Immune Response | Clinical Trial for CKD-5d | Experiences in CKD-5d | Clinical Trial for KT | Experiences in KT | |
|---|---|---|---|---|---|
| Purified virus components | |||||
| NVX-CoV2373 (Novavax) | IgM/IgG | Excluded in phase 2 study (NCT04368988) Not excluded in phase 3 study (NCT04611802) | N/A | Chronic exposure with immunosuppressive agents excluded in phase III (NCT04611802) | N/A |
| Replication-defective viral vector carrying pathogen gene(s) | |||||
| ChAdOx1 nCoV-19 (Oxford-AstraZeneca) | IgM/IgG | For mild to moderate renal disease in phase2/3 study (NCT04400838) | 70.6% antibody response after 1st dose when comparing with BNT162b2 (81.8%, p = 0.3) [32] | Excluded for immunosuppressant medication within the past 6 months in phase 2/3 study | N/A |
| Sputnik V (Gamaleya Research) | IgG, cell mediated immunity | NCT04805632 | N/A | Excluded in phase 3 study (NCT04741061) | N/A |
| Ad26.COV2.S (Janssen) | IgA, cell mediated immunity | Excluded in phase II study (NCT04436276) | N/A | Excluded in phase II study (NCT04436276) | N/A |
| mRNA vaccines | |||||
| BNT162b2 (Pfizer-BioNTech) | IgM/IgG, IgA, cell-mediated immunity | Not excluded in phase III trial | Lower anti–spike antibody level for dialysis patients than health control (116.5 AU/mL vs 176.5 AU/mL, p < 0.01) [33] | Excluded in phase I and III if anticipating the need for immunosuppressive treatment within the next 6 months (NCT04368728, NCT04713553) | |
| mRNA-1273 (Moderna) | IgM/IgG, cell-mediated immunity | Not excluded in phase III trial (NCT04470427) | 97% of seroconversion after 2 doses of the mRNA-1273 vaccine separated by a 28-day interval [36] | Excluded if using for corticosteroids ≥ 20 milligram (mg)/day (NCT04470427) | Patients treated with calcineurin inhibitors, mycophenolate mofetil, or steroids showed significantly lower anti–SARS-CoV-2 antibody titers [37] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hou, Y.-C.; Lu, K.-C.; Kuo, K.-L. The Efficacy of COVID-19 Vaccines in Chronic Kidney Disease and Kidney Transplantation Patients: A Narrative Review. Vaccines 2021, 9, 885. https://doi.org/10.3390/vaccines9080885
Hou Y-C, Lu K-C, Kuo K-L. The Efficacy of COVID-19 Vaccines in Chronic Kidney Disease and Kidney Transplantation Patients: A Narrative Review. Vaccines. 2021; 9(8):885. https://doi.org/10.3390/vaccines9080885
Chicago/Turabian StyleHou, Yi-Chou, Kuo-Cheng Lu, and Ko-Lin Kuo. 2021. "The Efficacy of COVID-19 Vaccines in Chronic Kidney Disease and Kidney Transplantation Patients: A Narrative Review" Vaccines 9, no. 8: 885. https://doi.org/10.3390/vaccines9080885
APA StyleHou, Y.-C., Lu, K.-C., & Kuo, K.-L. (2021). The Efficacy of COVID-19 Vaccines in Chronic Kidney Disease and Kidney Transplantation Patients: A Narrative Review. Vaccines, 9(8), 885. https://doi.org/10.3390/vaccines9080885







