Antibodies Responses to SARS-CoV-2 in a Large Cohort of Vaccinated Subjects and Seropositive Patients
Abstract
:1. Introduction
2. Materials and Methods
- -
- Aged between 18 and 65 years;
- -
- Underwent a serological analysis between 10 and 60 days after SARS-CoV-2 positivity (for the SARS-CoV-2 Cohort) and had not been vaccinated against SARS-CoV-2;
- -
- Underwent a serological analysis between 10 and 60 days after the first dose vaccination (for the Vaccination Cohort) and did not have a positive anamnesis for SARS-CoV-2.
- -
- for the Vaccination Cohort: age, sex, date at first dose vaccination, date at second dose vaccination, date at serological analyses;
- -
- for the SARS-CoV-2 Cohort: age, sex, date at serological analyses, date at first positive SARS-CoV-2 molecular test, worst clinical outcome (codified as asymptomatic, mild, moderate, severe, critical, deceased). SARS-CoV-2 positivity was confirmed by searching for the notification status in the national database, updated daily, by the local health authorities and provided by the Istituto Superiore di Sanità (ISS).
2.1. Laboratory Analyses
2.2. Statistical Analyses
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Stephens, D.S.; McElrath, M.J. COVID-19 and the Path to Immunity. JAMA 2020, 324, 1279. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Guan, X.; Wu, P.; Wang, X.; Zhou, L.; Tong, Y.; Ren, R.; Leung, K.S.M.; Lau, E.H.Y.; Wong, J.Y.; et al. Early Transmission Dynamics in Wuhan, China, of Novel Coronavirus-Infected Pneumonia. N. Engl. J. Med. 2020, 382, 1199–1207. [Google Scholar] [CrossRef] [PubMed]
- Zhu, N.; Zhang, D.; Wang, W.; Li, X.; Yang, B.; Song, J.; Zhao, X.; Huang, B.; Shi, W.; Lu, R.; et al. A Novel Coronavirus from Patients with Pneumonia in China, 2019. N. Engl. J. Med. 2020, 382, 727–733. [Google Scholar] [CrossRef]
- Zhou, P.; Yang, X.L.; Wang, X.G.; Hu, B.; Zhang, L.; Zhang, W.; Si, H.R.; Zhu, Y.; Li, B.; Huang, C.L.; et al. A pneumonia outbreak associated with a new coronavirus of probable bat origin. Nature 2020, 579, 270–273. [Google Scholar] [CrossRef] [Green Version]
- WHO Coronavirus Disease (COVID-19) Dashboard. Available online: https://covid19.who.int (accessed on 29 March 2021).
- WHO. Clinical Management of COVID-19-Living Guidance. 25 January 2021. Available online: https://www.who.int/publications/i/item/WHO-2019-nCoV-clinical-2021-1 (accessed on 20 April 2021).
- Dai, L.; George, F. Gao Viral target for vaccines against Covid 19. Nat. Rev. Immunol. 2021, 21, 73–82. [Google Scholar] [CrossRef]
- Zhou, W.; Xu, X.; Chang, Z.; Wang, H.; Zhong, X.; Tong, X.; Liu, T.; Li, Y. The dynamic changes of serum IgM and IgG against SARS-CoV-2 in patients with COVID-19. J. Med. Virol. 2021, 93, 924–933. [Google Scholar] [CrossRef]
- Gray, C.M.; Peter, J.; Mendelson, M.; Madhi, S.; Blackburn, J.M. COVID-19 antibody testing: From hype to immunological reality. S. Afr. Med. J. 2020, 110, 837. [Google Scholar] [CrossRef]
- Dan, J.M.; Mateus, J.; Kato, Y.; Hastie, K.M.; Yu, E.D.; Faliti, C.E.; Grifoni, A.; Ramirez, S.I.; Haupt, S.; Frazier, A.; et al. Immunological memory to SARS-CoV-2 assessed for up to 8 months after infection. Science 2021, 371, eabf4063. [Google Scholar] [CrossRef]
- Seow, J.; Graham, C.; Merrick, B.; Acors, S.; Pickering, S.; Steel, K.J.A.; Hemmings, O.; O’Byrne, A.; Kouphou, N.; Galao, R.P.; et al. Longitudinal observation and decline of neutralizing antibody responses in the three months following SARS-CoV-2 infection in humans. Nat. Microbiol. 2020, 5, 1598–1607. [Google Scholar] [CrossRef] [PubMed]
- Prévost, J.; Gasser, R.; Beaudoin-Bussières, G.; Richard, J.; Duerr, R.; Laumaea, A.; Anand, S.P.; Goyette, G.; Benlarbi, M.; Ding, S.; et al. Cross-Sectional Evaluation of Humoral Responses against SARS-CoV-2 Spike. Cell Rep. Med. 2020, 1, 100126. [Google Scholar] [CrossRef] [PubMed]
- Okba, N.M.A.; Müller, M.A.; Li, W.; Wang, C.; GeurtsvanKessel, C.H.; Corman, V.M.; Lamers, M.M.; Sikkema, R.S.; De Bruin, E.; Chandler, F.D.; et al. Severe Acute Respiratory Syndrome Coronavirus 2−Specific Antibody Responses in Coronavirus Disease Patients. Emerg. Infect. Dis. 2020, 26, 1478–1488. [Google Scholar] [CrossRef] [PubMed]
- Long, Q.X.; Liu, B.Z.; Deng, H.J.; Wu, G.C.; Deng, K.; Chen, Y.K.; Liao, P.; Qiu, J.F.; Lin, Y.; Cai, X.F.; et al. Antibody responses to SARS-CoV-2 in patients with COVID-19. Nat. Med. 2020, 26, 845–848. [Google Scholar] [CrossRef]
- Widge, A.T.; Rouphael, N.G.; Jackson, L.A.; Anderson, E.J.; Roberts, P.C.; Makhene, M.; Chappell, J.D.; Denison, M.R.; Stevens, L.J.; Pruijssers, A.J.; et al. Durability of Responses after SARS-CoV-2 mRNA-1273 Vaccination. N. Engl. J. Med. 2021, 384, 80–82. [Google Scholar] [CrossRef]
- Italian Medicines Agency-AIFA. Available online: https://www.aifa.gov.it/en/comirnaty (accessed on 23 March 2021).
- Bonelli, F.; Sarasini, A.; Zierold, C.; Calleri, M.; Bonetti, A.; Vismara, C.; Blocki, F.A.; Pallavicini, L.; Chinali, A.; Campisi, D.; et al. Clinical and Analytical Performance of an Automated Serological Test That Identifies S1/S2-Neutralizing IgG in COVID-19 Patients Semiquantitatively. J. Clin. Microbiol. 2020, 58, e01224-20. [Google Scholar] [CrossRef] [PubMed]
- R Core Team. 2020. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2018; Available online: https://www.r-project.org (accessed on 15 April 2021).
- World Health Organization. 2020. mRNA Vaccines against COVID-19: Pfizer-BioNTech COVID-19 Vaccine BNT162b2: Prepared by the Strategic Advisory Group of Experts (SAGE) on Immunization Working Group on COVID-19 Vaccines. Available online: https://apps.who.int/iris/handle/10665/338096 (accessed on 22 December 2020).
- Post, N.; Eddy, D.; Huntley, C.; van Schalkwyk, M.C.I.; Shrotri, M.; Leeman, D.; Rigby, S.; Williams, S.V.; Bermingham, W.H.; Kellam, P.; et al. Antibody response to SARS-CoV-2 infection in humans: A systematic review. PLoS ONE 2020, 15, e0244126. [Google Scholar] [CrossRef] [PubMed]
- Amodio, E.; Battisti, M.; Maida, C.M.; Zarcone, M.; Casuccio, A.; Vitale, F. Socio-demographic factors involved in a low-incidence phase of SARS-CoV-2 spread in Sicily, Italy: An ecological approach. Healthcare 2021. under review. [Google Scholar]
- Klein, S.L.; Morgan, R. The impact of sex and gender on immunotherapy outcomes. Biol. Sex Differ. 2020, 11, 24. [Google Scholar] [CrossRef]
- Takahashi, T.; Ellingson, M.K.; Wong, P.; Israelow, B.; Lucas, C.; Klein, J.; Silva, J.; Mao, T.; Oh, J.E.; Tokuyama, M.; et al. Sex differences in immune responses that underlie COVID-19 disease outcomes. Nature 2020, 588, 315–320. [Google Scholar] [CrossRef] [PubMed]
- Weisberg, S.P.; Connors, T.J.; Zhu, Y.; Baldwin, M.R.; Lin, W.-H.; Wontakal, S.; Szabo, P.A.; Wells, S.B.; Dogra, P.; Gray, J.; et al. Distinct antibody responses to SARS-CoV-2 in children and adults across the COVID-19 clinical spectrum. Nat. Immunol. 2020, 22, 25–31. [Google Scholar] [CrossRef]
- Krammer, F.; Srivastava, K.; Alshammary, H.; Amoako, A.A.; Awawda, M.H.; Beach, K.F.; Bermúdez-González, M.C.; Bielak, D.A.; Carreño, J.M.; Chernet, R.L.; et al. Antibody Responses in Seropositive Persons after a Single Dose of SARS-CoV-2 mRNA Vaccine. N. Engl. J. Med. 2021, 384, 1372–1374. [Google Scholar] [CrossRef]
- Ma, H.; Zeng, W.; He, H.; Zhao, D.; Jiang, D.; Zhou, P.; Cheng, L.; Li, Y.; Ma, X.; Jin, T. Serum IgA, IgM, and IgG responses in COVID-19. Cell. Mol. Immunol. 2020, 17, 773–775. [Google Scholar] [CrossRef] [PubMed]
- Madariaga, M.L.L.; Guthmiller, J.J.; Schrantz, S.; Jansen, M.O.; Christensen, C.; Kumar, M.; Prochaska, M.; Wool, G.; Durkin-Celauro, A.; Oh, W.H.; et al. Clinical predictors of donor antibody titre and correlation with recipient antibody response in a COVID-19 convalescent plasma clinical trial. J. Intern. Med. 2021, 289, 559–573. [Google Scholar] [CrossRef]
- Garcia-Basteiro, A.L.; Moncunill, G.; Tortajada, M.; Vidal, M.; Guinovart, C.; Jiménez, A.; Santano, R.; Sanz, S.; Méndez, S.; Llupià, A.; et al. Seroprevalence of antibodies against SARS-CoV-2 among health care workers in a large Spanish reference hospital. Nat. Commun. 2020, 11, 3500. [Google Scholar] [CrossRef] [PubMed]
- Rogers, T.F.; Zhao, F.; Huang, D.; Beutler, N.; Burns, A.; He, W.T.; Limbo, O.; Smith, C.; Song, G.; Woehl, J.; et al. Isolation of potent SARS-CoV-2 neutralizing antibodies and protection from disease in a small animal model. Science 2020, 369, 956–963. [Google Scholar] [CrossRef] [PubMed]
- Chandrashekar, A.; Liu, J.; Martinot, A.J.; McMahan, K.; Mercado, N.B.; Peter, L.; Tostanoski, L.H.; Yu, J.; Maliga, Z.; Nekorchuk, M.; et al. SARS-CoV-2 infection protects against rechallenge in rhesus macaques. Science 2020, 369, 812–817. [Google Scholar] [CrossRef]
- Hall, V.J.; Foulkes, S.; Charlett, A.; Atti, A.; Monk, E.J.M.; Simmons, R.; Wellington, E.; Cole, M.J.; Saei, A.; Oguti, B.; et al. SARS-CoV-2 infection rates of antibody-positive compared with antibody-negative health-care workers in England: A large, multicentre, prospective cohort study (SIREN). Lancet 2021, 397, 1459–1469. [Google Scholar] [CrossRef]
- Pilz, S.; Chakeri, A.; Ioannidis, J.P.; Richter, L.; Theiler-Schwetz, V.; Trummer, C.; Krause, R.; Allerberger, F. SARS-CoV-2 re-infection risk in Austria. Eur. J. Clin. Investig. 2021, 51, e13520. [Google Scholar] [CrossRef]
- Lumley, S.F.; O’Donnell, D.; Stoesser, N.E.; Matthews, P.C.; Howarth, A.; Hatch, S.B.; Marsden, B.D.; Cox, S.; James, T.; Warren, F.; et al. Antibody Status and Incidence of SARS-CoV-2 Infection in Health Care Workers. N. Engl. J. Med. 2021, 384, 533–540. [Google Scholar] [CrossRef]
- Hansen, C.H.; Michlmayr, D.; Gubbels, S.M.; Mølbak, K.; Ethelberg, S. Assessment of protection against reinfection with SARS-CoV-2 among 4 million PCR-tested individuals in Denmark in 2020: A population-level observational study. Lancet 2021, 397, 1204–1212. [Google Scholar] [CrossRef]
- Harvey, R.A.; Rassen, J.A.; Kabelac, C.A.; Turenne, W.; Leonard, S.; Klesh, R.; Meyer, W.A., 3rd; Kaufman, H.W.; Anderson, S.; Cohen, O.; et al. Association of SARS-CoV-2 Seropositive Antibody Test With Risk of Future Infection. JAMA Intern. Med. 2021, 181, 672–679. [Google Scholar]
- Coughlin, M.M.; Prabhakar, B.S. Neutralizing human monoclonal antibodies to severe acute respiratory syndrome coronavirus: Target, mechanism of action, and therapeutic potential. Rev. Med. Virol. 2011, 22, 2–17. [Google Scholar] [CrossRef] [Green Version]
- Valdivia, A.; Torres, I.; Latorre, V.; Francés-Gómez, C.; Albert, E.; Gozalbo-Rovira, R.; Alcaraz, M.J.; Buesa, J.; Rodríguez-Díaz, J.; Geller, R.; et al. Inference of SARS-CoV-2 spike-binding neutralizing antibody titers in sera from hospitalized COVID-19 patients by using commercial enzyme and chemiluminescent immunoassays. Eur. J. Clin. Microbiol. Infect. Dis. 2021, 40, 485–494. [Google Scholar] [CrossRef] [PubMed]
- Criscuolo, E.; Diotti, R.A.; Strollo, M.; Rolla, S.; Ambrosi, A.; Locatelli, M.; Burioni, R.; Mancini, N.; Clementi, M.; Clementi, N. Weak correlation between antibody titers and neutralizing activity in sera from SARS-CoV-2 infected subjects. J. Med. Virol. 2021, 93, 2160–2167. [Google Scholar] [CrossRef] [PubMed]
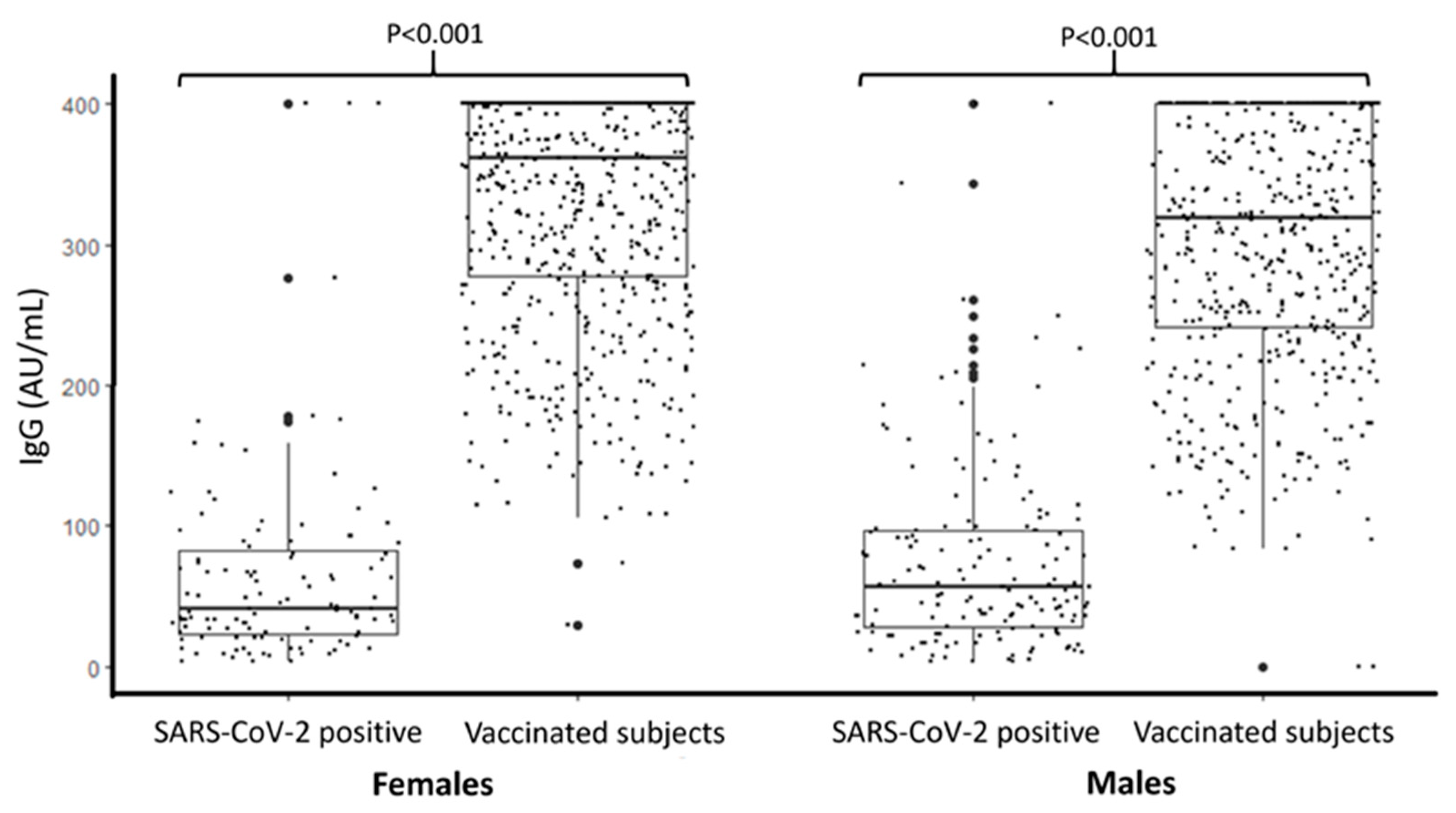
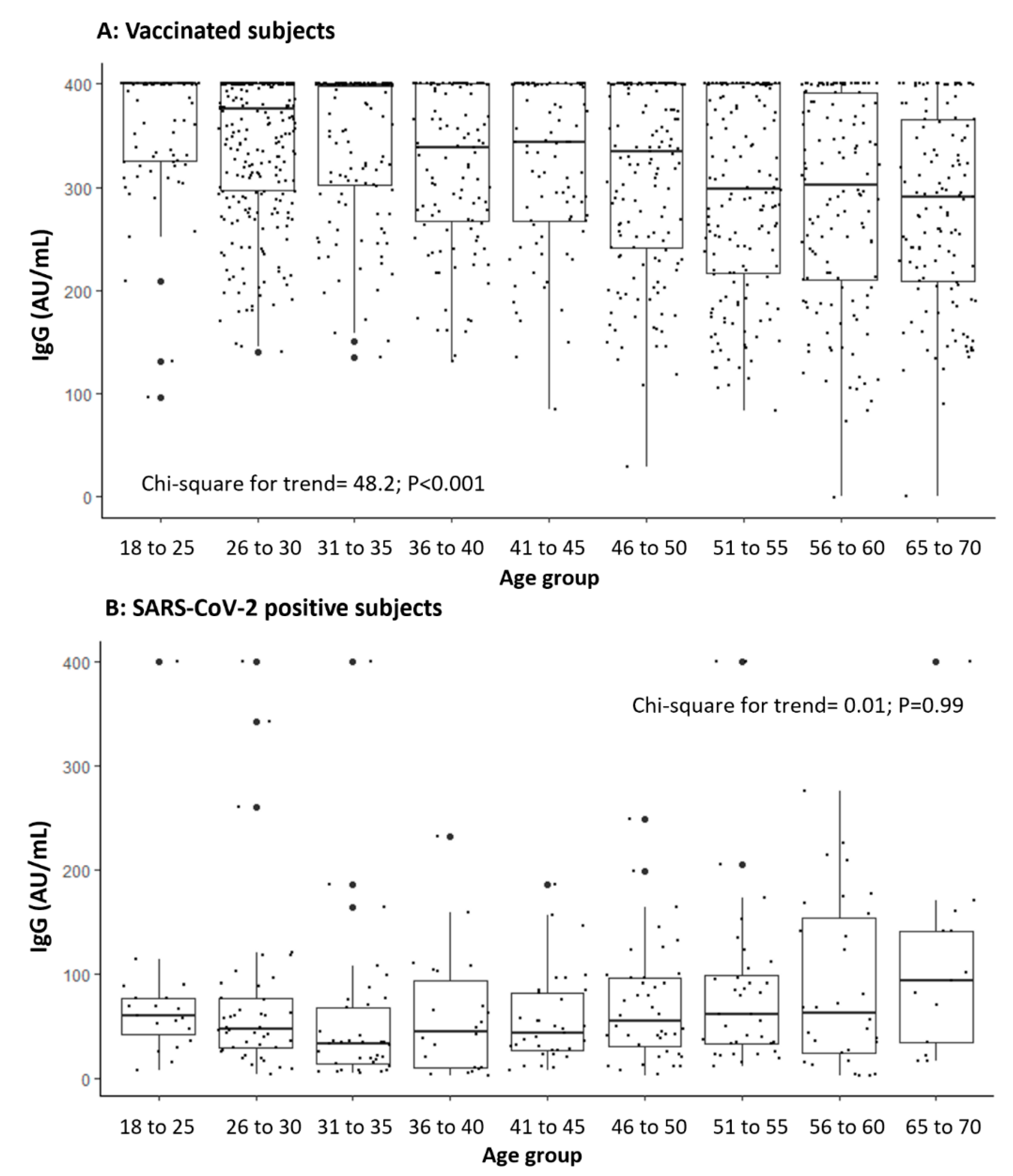
| Investigated Variables | Categories | Vaccination Cohort (SARS-CoV-2 Negative) | SARS-CoV-2 Positive Cohort (Not Vaccinated) | p-Value |
|---|---|---|---|---|
| Total, N (%) | 1256 (82.2) | 272 (17.8) | ||
| Age, median (IQR) | 43 (30–54) | 43 (31–51) | 0.62 | |
| Gender, N (by column %) | ||||
| - F | 688 (54.8) | 115 (42.3) | <0.001 * | |
| - M | 568 (45.2) | 157 (57.7) | ||
| Days after first dose of vaccination or COVID positivity, median (IQR) | 37 (34–41) | 40 (32–49) | 0.13 | |
| IgG (AU/mL) median (IQR) | 342 (259–400) | 48.9 (25.6–92.1) | <0.001 | |
| Worst clinical presentation, IgG median (IQR) | ||||
| - Asymptomatic, Mild | - | 46.4 (24–88.3) | ||
| - Moderate | - | 90.5 (53.7–92.7) | ||
| - Severe, Critical | - | 101.2 (22.3–191.7) | ||
| IgG (AU/mL), N (by column %) | ||||
| -Seronegative | 2 (0.2) | 28 (10.3) | <0.001 * | |
| - 12 to 15 | 0 (0) | 10 (3.7) | ||
| - >15 to 50 | 1 (0.1) | 101 (37.1) | ||
| - 51 to 100 | 7 (0.6) | 77 (28.7) | ||
| - 101 to 150 | 53 (4.2) | 25 (9.2) | ||
| - 151 to 200 | 90 (7.2) | 15 (5.5) | ||
| - 201 to 250 | 130 (10.3) | 6 (2.2) | ||
| - 251 to 300 | 184 (14.6) | 2 (0.7) | ||
| - 301 to 350 | 193 (15.4) | 1 (0.4) | ||
| - 351 to inf | 596 (47.4) | 6 (2.2) |
| Investigated Variables | Coefficient | Standard Error | p-Value |
|---|---|---|---|
| - Vaccination (ref. COVID-19 positivity) | 266.4 | 7.18 | <0.001 |
| - Female sex (ref. male) | −22.3 | 5.7 | <0.001 |
| - Age in years (per unit increase) | −1.96 | 0.22 | <0.001 |
| - Days after vaccination or positivity to COVID-19 (per unit increase) | −1.67 | 0.37 | <0.001 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Amodio, E.; Capra, G.; Casuccio, A.; Grazia, S.D.; Genovese, D.; Pizzo, S.; Calamusa, G.; Ferraro, D.; Giammanco, G.M.; Vitale, F.; et al. Antibodies Responses to SARS-CoV-2 in a Large Cohort of Vaccinated Subjects and Seropositive Patients. Vaccines 2021, 9, 714. https://doi.org/10.3390/vaccines9070714
Amodio E, Capra G, Casuccio A, Grazia SD, Genovese D, Pizzo S, Calamusa G, Ferraro D, Giammanco GM, Vitale F, et al. Antibodies Responses to SARS-CoV-2 in a Large Cohort of Vaccinated Subjects and Seropositive Patients. Vaccines. 2021; 9(7):714. https://doi.org/10.3390/vaccines9070714
Chicago/Turabian StyleAmodio, Emanuele, Giuseppina Capra, Alessandra Casuccio, Simona De Grazia, Dario Genovese, Stefano Pizzo, Giuseppe Calamusa, Donatella Ferraro, Giovanni Maurizio Giammanco, Francesco Vitale, and et al. 2021. "Antibodies Responses to SARS-CoV-2 in a Large Cohort of Vaccinated Subjects and Seropositive Patients" Vaccines 9, no. 7: 714. https://doi.org/10.3390/vaccines9070714
APA StyleAmodio, E., Capra, G., Casuccio, A., Grazia, S. D., Genovese, D., Pizzo, S., Calamusa, G., Ferraro, D., Giammanco, G. M., Vitale, F., & Bonura, F. (2021). Antibodies Responses to SARS-CoV-2 in a Large Cohort of Vaccinated Subjects and Seropositive Patients. Vaccines, 9(7), 714. https://doi.org/10.3390/vaccines9070714












