Seroprevalence of SARS-CoV-2 Antibodies in Symptomatic Individuals Is Higher than in Persons Who Are at Increased Risk Exposure: The Results of the Single-Center, Prospective, Cross-Sectional Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Location of the Study
2.2. Study Design, Population, and Sampling
2.3. Ethical Permission
2.4. Laboratory Tests
2.5. Adjusting Prevalence Estimates
2.6. Positivity Coefficient (CP) Calculations
2.7. Statistical Analysis
3. Results
3.1. Seropositivity Rates for Joint Detection of IgG and IgM in Persons of High-Risk and Symptomatic Groups
3.2. Adjusted Seropositivity Rates for Joint Detection of IgG and IgM in Persons of High-Risk and Symptomatic Groups
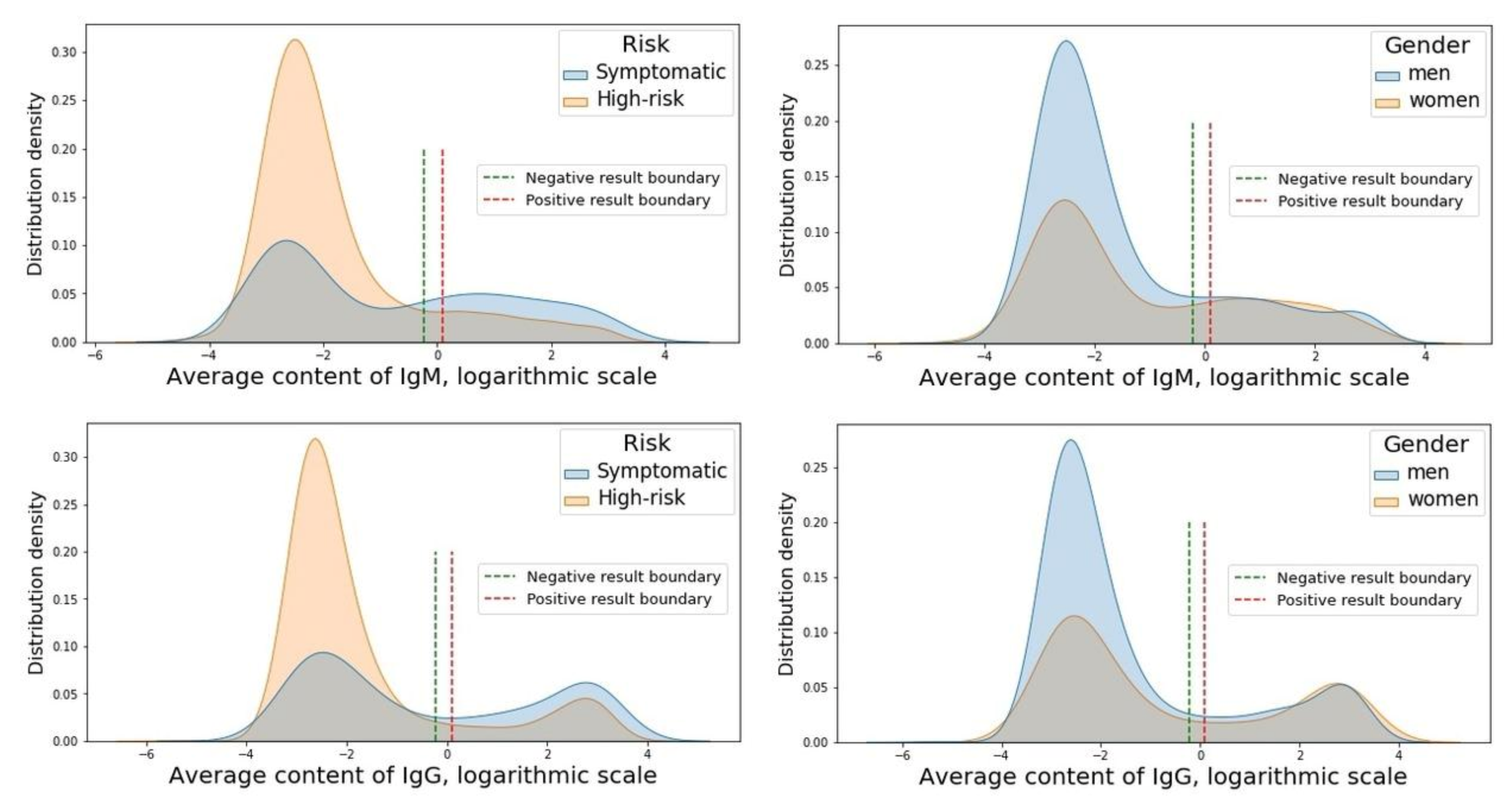
3.3. Antibody Content
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Weekly Epidemiological Update on COVID-19—25 May 2021. Available online: https://www.who.int/publications/m/item/weekly-epidemiological-update-on-covid-19---25-may-2021 (accessed on 31 May 2021).
- Syangtan, G.; Bista, S.; Dawadi, P.; Rayamajhee, B.; Shrestha, L.B.; Tuladhar, R.; Joshi, D.R. Asymptomatic SARS-CoV-2 carriers: A systematic review and meta-analysis. Front. Public Health 2020, 8, 587374. [Google Scholar] [CrossRef] [PubMed]
- Lai, C.-C.; Wang, J.-H.; Hsueh, P.-R. Population-based seroprevalence surveys of anti-SARS-CoV-2 antibody: An up-to-date review. Int. J. Infect. Dis. 2020, 101, 314–322. [Google Scholar] [CrossRef] [PubMed]
- Oran, D.P.; Topol, E.J. Prevalence of asymptomatic SARS-CoV-2 infection: A narrative review. Ann. Intern. Med. 2020, 173, 362–367. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Chen, Z.; Azman, A.S.; Deng, X.; Chen, X.; Lu, W.; Zhao, Z.; Yang, J.; Viboud, C.; Ajelli, M.; et al. Serological evidence of human infection with SARS-CoV-2: A systematic review and meta-analysis. medRxiv 2020. [Google Scholar] [CrossRef]
- CDC Healthcare Workers. Available online: https://www.cdc.gov/coronavirus/2019-ncov/hcp/testing-overview.html (accessed on 9 May 2021).
- Xu, Y.; Xiao, M.; Liu, X.; Xu, S.; Du, T.; Xu, J.; Yang, Q.; Xu, Y.; Han, Y.; Li, T.; et al. Significance of serology testing to assist timely diagnosis of SARS-CoV-2 infections: Implication from a family cluster. Emerg. Microbes. Infect. 2020, 9, 924–927. [Google Scholar] [CrossRef] [PubMed]
- Kissler, S.M.; Tedijanto, C.; Goldstein, E.; Grad, Y.H.; Lipsitch, M. Projecting the transmission dynamics of SARS-CoV-2 through the postpandemic period. Science 2020, 368, 860–868. [Google Scholar] [CrossRef]
- Huang, A.T.; Garcia-Carreras, B.; Hitchings, M.D.T.; Yang, B.; Katzelnick, L.C.; Rattigan, S.M.; Borgert, B.A.; Moreno, C.A.; Solomon, B.D.; Trimmer-Smith, L.; et al. A systematic review of antibody mediated immunity to coronaviruses: Kinetics, correlates of protection, and association with severity. Nat. Commun. 2020, 11, 4704. [Google Scholar] [CrossRef]
- Poustchi, H.; Darvishian, M.; Mohammadi, Z.; Shayanrad, A.; Delavari, A.; Bahadorimonfared, A.; Eslami, S.; Javanmard, S.H.; Shakiba, E.; Somi, M.H.; et al. SARS-CoV-2 antibody seroprevalence in the general population and high-risk occupational groups across 18 cities in Iran: A population-based cross-sectional study. Lancet Infect. Dis. 2021, 21, 473–481. [Google Scholar] [CrossRef]
- Hsueh, P.-R.; Huang, L.-M.; Chen, P.-J.; Kao, C.-L.; Yang, P.-C. Chronological evolution of IgM, IgA, IgG and neutralisation antibodies after infection with SARS-associated coronavirus. Clin. Microbiol. Infect. 2004, 10, 1062–1066. [Google Scholar] [CrossRef]
- Orner, E.P.; Rodgers, M.A.; Hock, K.; Tang, M.S.; Taylor, R.; Gardiner, M.; Olivo, A.; Fox, A.; Prostko, J.; Cloherty, G.; et al. Comparison of SARS-CoV-2 IgM and IgG seroconversion profiles among hospitalized patients in two US cities. Diagn. Microbiol. Infect. Dis. 2021, 99, 115300. [Google Scholar] [CrossRef]
- Long, Q.-X.; Liu, B.-Z.; Deng, H.-J.; Wu, G.-C.; Deng, K.; Chen, Y.-K.; Liao, P.; Qiu, J.-F.; Lin, Y.; Cai, X.-F.; et al. Antibody responses to SARS-CoV-2 in patients with COVID-19. Nat. Med. 2020, 26, 845–848. [Google Scholar] [CrossRef] [PubMed]
- Chvatal-Medina, M.; Mendez-Cortina, Y.; Patiño, P.J.; Velilla, P.A.; Rugeles, M.T. Antibody responses in COVID-19: A review. Front. Immunol. 2021, 12, 633184. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Li, J.; Li, H.; Lei, P.; Shen, G.; Yang, C. Persistence of SARS-CoV-2-specific antibodies in COVID-19 patients. Int. Immunopharmacol. 2021, 90, 107271. [Google Scholar] [CrossRef] [PubMed]
- Luo, C.; Liu, M.; Li, Q.; Zheng, X.; Ai, W.; Gong, F.; Fan, J.; Liu, S.; Wang, X.; Luo, J. Dynamic changes and prevalence of SARS-CoV-2 IgG/IgM antibodies: Multiple factors-based analysis. Int. J. Infect. Dis. 2021. [Google Scholar] [CrossRef]
- Gómez-Ochoa, S.A.; Franco, O.H.; Rojas, L.Z.; Raguindin, P.F.; Roa-Díaz, Z.M.; Wyssmann, B.M.; Guevara, S.L.R.; Echeverría, L.E.; Glisic, M.; Muka, T. COVID-19 in healthcare workers: A living systematic review and meta-analysis of prevalence, risk factors, clinical characteristics, and outcomes. Am. J. Epidemiol. 2020. [Google Scholar] [CrossRef]
- Lan, F.-Y.; Suharlim, C.; Kales, S.N.; Yang, J. Association between SARS-CoV-2 infection, exposure risk and mental health among a cohort of essential retail workers in the USA. Occup. Environ. Med. 2021, 78, 237–243. [Google Scholar] [CrossRef]
- Gaitens, J.; Condon, M.; Fernandes, E.; McDiarmid, M. COVID-19 and essential workers: A narrative review of health outcomes and moral injury. Int. J. Environ. Res. Public Health 2021, 18, 1446. [Google Scholar] [CrossRef]
- Lan, F.-Y.; Wei, C.-F.; Hsu, Y.-T.; Christiani, D.C.; Kales, S.N. Work-related COVID-19 transmission in six Asian countries/areas: A follow-up study. PLoS ONE 2020, 15, e0233588. [Google Scholar] [CrossRef]
- Zhang, J.Z.; Zhou, P.; Han, D.B.; Wang, W.C.; Cui, C.; Zhou, R.; Xu, K.X.; Liu, L.; Wang, X.H.; Bai, X.H.; et al. Investigation on a cluster epidemic of COVID-19 in a supermarket in Liaocheng, Shandong province. Zhonghua Liu Xing Bing Xue Za Zhi 2020, 41, 2024–2028. [Google Scholar] [CrossRef]
- COVID Live Update: 158,369,040 Cases and 3,298,155 Deaths from the Coronavirus—Worldometer. Available online: https://www.worldometers.info/coronavirus/#countries (accessed on 9 May 2021).
- Komissarov, A.B.; Safina, K.R.; Garushyants, S.K.; Fadeev, A.V.; Sergeeva, M.V.; Ivanova, A.A.; Danilenko, D.M.; Lioznov, D.; Shneider, O.V.; Shvyrev, N.; et al. Genomic epidemiology of the early stages of the SARS-CoV-2 outbreak in Russia. Nat. Commun. 2021, 12, 649. [Google Scholar] [CrossRef] [PubMed]
- Barchuk, A.; Skougarevskiy, D.; Titaev, K.; Shirokov, D.; Raskina, Y.; Novkunkskaya, A.; Talantov, P.; Isaev, A.; Pomerantseva, E.; Zhikrivetskaya, S.; et al. Seroprevalence of SARS-CoV-2 antibodies in Saint Petersburg, Russia: A population-based study. medRxiv 2020. [Google Scholar] [CrossRef]
- Popova, A.Y.; Ezhlova, E.B.; Melnikova, A.A.; Trotsenko, O.E.; Zaitseva, T.A.; Lyalina, L.V.; Garbuz, Y.A.; Smirnov, V.S.; Lomonosova, V.I.; Balakhontseva, L.A.; et al. The seroprevalence of SARS-CoV-2 among residents of the Khabarovsk Krai during the COVID-19 epidemic. J. Microbiol. Epidemiol. Immunobiol. 2021, 98, 7–17. [Google Scholar] [CrossRef]
- Popova, A.Y.; Ezhlova, E.B.; Melnikova, A.A.; Stepanova, T.F.; Sharukho, G.V.; Letyushev, A.N.; Folmer, A.Y.; Shepotkova, A.A.; Lyalina, L.V.; Smirnov, V.S.; et al. Distribution of SARS-CoV-2 seroprevalence among residents of the Tyumen Region during the COVID-19 epidemic period. Zhurnal Mikrobiol. Èpidemiologii I Immunobiol. 2020, 97, 392–400. [Google Scholar] [CrossRef]
- Popova, A.Y.; Ezhlova, E.B.; Melnikova, A.A.; Patyashina, M.A.; Sizova, E.P.; Yuzlibaeva, L.R.; Lyalina, L.V.; Smirnov, V.S.; Badamshina, G.G.; Goncharova, A.V.; et al. Distribution of SARS-CoV-2 seroprevalence among residents of the Republic of Tatarstan during the COVID-19 epidemic period. J. Microbiol. Epidemiol. Immunobiol. 2021, 97, 518–528. [Google Scholar] [CrossRef]
- Vector-Best. Available online: https://vector-best.ru/upload/iblock/181/181c954b9aa28cb97b2c2fb4fdb7e46c.pdf (accessed on 9 May 2021).
- Sensitivity and Specificity of Reagent Kits of JSC “Vector-Best” for the Detection of Immunoglobulins of Different Classes to SARS-CoV-2. “Spravochnik Zaveduyushchego KDL”. Available online: https://e.zavkdl.ru/843516 (accessed on 1 April 2021).
- Sempos, C.T.; Tian, L. Adjusting coronavirus prevalence estimates for laboratory test kit error. Am. J. Epidemiol. 2021, 190, 109–115. [Google Scholar] [CrossRef]
- Reagent kit for Enzyme Immunoassay Immunoglobulins Class G to SARS-CoV-2. Available online: http://dias-plus.com/wp-content/uploads/2020/05/%D0%86%D0%BD%D1%81%D1%82%D1%80-%D1%8F-%D0%BD%D0%B0-%D0%86%D0%A4%D0%90-IgG-SARS_CoV-2-%D0%92%D0%91.pdf (accessed on 31 May 2021).
- Rostami, A.; Sepidarkish, M.; Leeflang, M.M.G.; Riahi, S.M.; Nourollahpour Shiadeh, M.; Esfandyari, S.; Mokdad, A.H.; Hotez, P.J.; Gasser, R.B. SARS-CoV-2 seroprevalence worldwide: A systematic review and meta-analysis. Clin. Microbiol. Infect. 2021, 27, 331–340. [Google Scholar] [CrossRef] [PubMed]
- Kalish, H.; Klumpp-Thomas, C.; Hunsberger, S.; Baus, H.A.; Fay, M.P.; Siripong, N.; Wang, J.; Hicks, J.; Mehalko, J.; Travers, J.; et al. Mapping a pandemic: SARS-CoV-2 seropositivity in the United States. medRxiv 2021. [Google Scholar] [CrossRef]
- Wolff, F.; Dahma, H.; Duterme, C.; Van den Wijngaert, S.; Vandenberg, O.; Cotton, F.; Montesinos, I. Monitoring antibody response following SARS-CoV-2 infection: Diagnostic efficiency of 4 automated immunoassays. Diagn. Microbiol. Infect. Dis. 2020, 98, 115140. [Google Scholar] [CrossRef]
- Piccoli, L.; Ferrari, P.; Piumatti, G.; Jovic, S.; Rodriguez, B.F.; Mele, F.; Giacchetto-Sasselli, I.; Terrot, T.; Silacci-Fregni, C.; Cameroni, E.; et al. Risk assessment and seroprevalence of SARS-CoV-2 infection in healthcare workers of COVID-19 and non-COVID-19 hospitals in southern Switzerland. Lancet Reg. Health Eur. 2021, 1, 100013. [Google Scholar] [CrossRef]
- Pasqualotto, A.C.; de Pereira, P.C.; Lana, D.F.D.; Schwarzbold, A.V.; Ribeiro, M.S.; Riche, C.V.W.; Castro, C.P.P.; Korsack, P.L.; Ferreira, P.E.B.; de Domingues, G.C.; et al. COVID-19 seroprevalence in military police force, southern Brazil. PLoS ONE 2021, 16, e0249672. [Google Scholar] [CrossRef]
- Varona, J.F.; Madurga, R.; Peñalver, F.; Abarca, E.; Almirall, C.; Cruz, M.; Ramos, E.; Castellano Vázquez, J.M. Seroprevalence of SARS-CoV-2 antibodies in over 6000 healthcare workers in Spain. Int. J. Epidemiol. 2021. [Google Scholar] [CrossRef] [PubMed]
- Naaber, P.; Hunt, K.; Pesukova, J.; Haljasmägi, L.; Rumm, P.; Peterson, P.; Hololejenko, J.; Eero, I.; Jõgi, P.; Toompere, K.; et al. Evaluation of SARS-CoV-2 IgG antibody response in PCR positive patients: Comparison of nine tests in relation to clinical data. PLoS ONE 2020, 15, e0237548. [Google Scholar] [CrossRef] [PubMed]
- Zeng, F.; Dai, C.; Cai, P.; Wang, J.; Xu, L.; Li, J.; Hu, G.; Wang, Z.; Zheng, F.; Wang, L. A comparison study of SARS-CoV-2 IgG antibody between male and female COVID-19 patients: A possible reason underlying different outcome between sex. J. Med. Virol. 2020, 92, 2050–2054. [Google Scholar] [CrossRef]
- Gudbjartsson, D.F.; Norddahl, G.L.; Melsted, P.; Gunnarsdottir, K.; Holm, H.; Eythorsson, E.; Arnthorsson, A.O.; Helgason, D.; Bjarnadottir, K.; Ingvarsson, R.F.; et al. Humoral immune response to SARS-CoV-2 in Iceland. New Engl. J. Med. 2020, 383, 1724–1734. [Google Scholar] [CrossRef] [PubMed]
- Fink, A.L.; Engle, K.; Ursin, R.L.; Tang, W.-Y.; Klein, S.L. Biological sex affects vaccine efficacy and protection against influenza in mice. Proc. Natl. Acad. Sci. USA 2018, 115, 12477–12482. [Google Scholar] [CrossRef] [PubMed]
- Klein, S.L.; Marriott, I.; Fish, E.N. Sex-based differences in immune function and responses to vaccination. Trans. R. Soc. Trop. Med. Hyg. 2015, 109, 9–15. [Google Scholar] [CrossRef]
- Kadel, S.; Kovats, S. Sex Hormones regulate innate immune cells and promote sex differences in respiratory virus infection. Front. Immunol. 2018, 9, 1653. [Google Scholar] [CrossRef]
- Gadi, N.; Wu, S.C.; Spihlman, A.P.; Moulton, V.R. What’s sex got to do with COVID-19? Gender-based differences in the host immune response to coronaviruses. Front. Immunol. 2020, 11, 2147. [Google Scholar] [CrossRef]
- Bunders, M.J.; Altfeld, M. Implications of sex differences in immunity for SARS-CoV-2 pathogenesis and design of therapeutic interventions. Immunity 2020, 53, 487–495. [Google Scholar] [CrossRef]
- Guan, W.-J.; Ni, Z.-Y.; Hu, Y.; Liang, W.-H.; Ou, C.-Q.; He, J.-X.; Liu, L.; Shan, H.; Lei, C.-L.; Hui, D.S.C.; et al. clinical characteristics of coronavirus disease 2019 in China. N. Engl. J. Med. 2020, 382, 1708–1720. [Google Scholar] [CrossRef]
- Furman, D.; Hejblum, B.P.; Simon, N.; Jojic, V.; Dekker, C.L.; Thiébaut, R.; Tibshirani, R.J.; Davis, M.M. Systems analysis of sex differences reveals an immunosuppressive role for testosterone in the response to influenza vaccination. Proc. Natl. Acad. Sci. USA 2014, 111, 869–874. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, T.; Ellingson, M.K.; Wong, P.; Israelow, B.; Lucas, C.; Klein, J.; Silva, J.; Mao, T.; Oh, J.E.; Tokuyama, M.; et al. Sex differences in immune responses that underlie COVID-19 disease outcomes. Nature 2020, 588, 315–320. [Google Scholar] [CrossRef] [PubMed]
- Robbiani, D.F.; Gaebler, C.; Muecksch, F.; Lorenzi, J.C.C.; Wang, Z.; Cho, A.; Agudelo, M.; Barnes, C.O.; Gazumyan, A.; Finkin, S.; et al. Convergent antibody responses to SARS-CoV-2 in convalescent individuals. Nature 2020, 584, 437–442. [Google Scholar] [CrossRef] [PubMed]
- Shrock, E.; Fujimura, E.; Kula, T.; Timms, R.T.; Lee, I.-H.; Leng, Y.; Robinson, M.L.; Sie, B.M.; Li, M.Z.; Chen, Y.; et al. Viral epitope profiling of COVID-19 patients reveals cross-reactivity and correlates of severity. Science 2020, 370. [Google Scholar] [CrossRef]
- Korte, W.; Buljan, M.; Rösslein, M.; Wick, P.; Golubov, V.; Jentsch, J.; Reut, M.; Peier, K.; Nohynek, B.; Fischer, A.; et al. SARS-CoV-2 IgG and IgA antibody response is gender dependent, and IgG antibodies rapidly decline early on. J. Infect. 2021, 82, e11–e14. [Google Scholar] [CrossRef]
- Gebhard, C.; Regitz-Zagrosek, V.; Neuhauser, H.K.; Morgan, R.; Klein, S.L. Impact of sex and gender on COVID-19 outcomes in Europe. Biol Sex. Differ. 2020, 11, 29. [Google Scholar] [CrossRef]
- Yang, H.S.; Costa, V.; Racine-Brzostek, S.E.; Acker, K.P.; Yee, J.; Chen, Z.; Karbaschi, M.; Zuk, R.; Rand, S.; Sukhu, A.; et al. Association of age with SARS-CoV-2 antibody response. JAMA Netw. Open 2021, 4, e214302. [Google Scholar] [CrossRef]
- Gonzalez-Quintela, A.; Alende, R.; Gude, F.; Campos, J.; Rey, J.; Meijide, L.M.; Fernandez-Merino, C.; Vidal, C. serum levels of immunoglobulins (IgG, IgA, IgM) in a general adult population and their relationship with alcohol consumption, smoking and common metabolic abnormalities. Clin. Exp. Immunol. 2008, 151, 42–50. [Google Scholar] [CrossRef]
- Randolph, H.E.; Barreiro, L.B. Herd immunity: Understanding COVID-19. Immunity 2020, 52, 737–741. [Google Scholar] [CrossRef]
| Risk Group | Gender | Count | Prevalence within the Group | Prevalence | Mean Age |
|---|---|---|---|---|---|
| High-risk | Men | 781 | 72% | 44% | 36.91 |
| Women | 310 | 28% | 17% | 41.79 | |
| Symptomatic | Men | 320 | 46% | 18% | 39.67 |
| Women | 372 | 54% | 21% | 41.39 |
| Variant | Presence [+] or Absence [−] of Igs | Group | n | Prevalence in the Group | Prevalence in the Study Population |
|---|---|---|---|---|---|
| Men vs. Women | |||||
| 1 | IgM [−] and IgG [−] | women | 423 | 62.02% | 23.72% |
| men | 839 | 76.20% | 47.06% | ||
| 2 | IgM [−] and IgG [+] | women | 41 | 6.01% | 2.30% |
| men | 44 | 4.00% | 2.47% | ||
| 3 | IgM [−] and IgG [*] | women | 3 | 0.44% | 0.17% |
| men | 3 | 0.27% | 0.17% | ||
| 4 | IgM [+] and IgG [−] | women | 21 | 3.08% | 1.18% |
| men | 25 | 2.27% | 1.40% | ||
| 5 | IgM [+] and IgG [+] | women | 165 | 24.19% | 9.25% |
| men | 170 | 15.44% | 9.53% | ||
| 6 | IgM [+] and IgG [*] | women | 8 | 1.17% | 0.45% |
| men | 3 | 0.27% | 0.17% | ||
| 7 | IgM [*] and IgG [−] | women | 2 | 0.29% | 0.11% |
| men | 7 | 0.64% | 0.39% | ||
| 8 | IgM [*] and IgG [+] | women | 19 | 2.79% | 1.07% |
| men | 10 | 0.91% | 0.56% | ||
| High−risk vs. Symptomatic | |||||
| 1 | IgM [−] and IgG [−] | high−risk | 895 | 82.03% | 50.20% |
| symptomatic | 367 | 53.03% | 20.58% | ||
| 2 | IgM [−] and IgG [+] | high−risk | 45 | 4.12% | 2.52% |
| symptomatic | 40 | 5.78% | 2.24% | ||
| 3 | IgM [−] and IgG [*] | high−risk | 3 | 0.27% | 0.17% |
| symptomatic | 3 | 0.43% | 0.17% | ||
| 4 | IgM [+] and IgG [−] | high−risk | 21 | 1.92% | 1.18% |
| symptomatic | 25 | 3.61% | 1.40% | ||
| 5 | IgM [+] and IgG [+] | high−risk | 113 | 10.36% | 6.34% |
| symptomatic | 222 | 32.08% | 12.45% | ||
| 6 | IgM [+] and IgG [*] | high−risk | 5 | 0.46% | 0.28% |
| symptomatic | 6 | 0.87% | 0.34% | ||
| 7 | IgM [*] and IgG [−] | high−risk | 5 | 0.46% | 0.28% |
| symptomatic | 4 | 0.58% | 0.22% | ||
| 8 | IgM [*] and IgG [+] | high−risk | 4 | 0.37% | 0.22% |
| symptomatic | 25 | 3.61% | 1.40% | ||
| Gender/Risk Group | 6–12 Days * | 13–20 Days * | ||||
|---|---|---|---|---|---|---|
| Adj. Prev. | Cr. Prev. | Adj. Count | Adj. Prev. | Cr. Prev. | Adj. Count | |
| IgM and IgG positive | ||||||
| All patients | 18.87% | 18.79% | 336 | 18.73% | 18.79% | 334 |
| Women | 24.31% | 24.19% | 166 | 24.14% | 24.19% | 165 |
| Men | 15.49% | 15.44% | 171 | 15.38% | 15.44% | 169 |
| High-risk | 10.37% | 10.36% | 113 | 10.29% | 10.36% | 113 |
| Symptomatic | 32.26% | 32.08% | 223 | 32.03% | 32.08% | 218 |
| IgG positive | ||||||
| All patients | 8.61% | 6.39% | 153 | 6.16% | 6.39% | 110 |
| Women | 11.98% | 8.80% | 82 | 8.57% | 8.80% | 58 |
| Men | 6.52% | 4.90% | 72 | 4.66% | 4.90% | 51 |
| High-risk | 5.94% | 4.49% | 65 | 4.25% | 4.49% | 47 |
| Symptomatic | 12.81% | 9.39% | 89 | 9.17% | 9.39% | 63 |
| IgM positive | ||||||
| All patients | 3.57% | 3.20% | 64 | 3.07% | 3.20% | 55 |
| Women | 4.86% | 4.25% | 33 | 4.17% | 4.25% | 28 |
| Men | 2.77% | 2.54% | 30 | 2.38% | 2.54% | 26 |
| High-risk | 2.57% | 2.38% | 28 | 2.21% | 2.38% | 24 |
| Symptomatic | 5.14% | 4.48% | 36 | 4.42% | 4.48% | 30 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zurochka, A.; Dobrinina, M.; Zurochka, V.; Hu, D.; Solovyev, A.; Ryabova, L.; Kritsky, I.; Ibragimov, R.; Sarapultsev, A. Seroprevalence of SARS-CoV-2 Antibodies in Symptomatic Individuals Is Higher than in Persons Who Are at Increased Risk Exposure: The Results of the Single-Center, Prospective, Cross-Sectional Study. Vaccines 2021, 9, 627. https://doi.org/10.3390/vaccines9060627
Zurochka A, Dobrinina M, Zurochka V, Hu D, Solovyev A, Ryabova L, Kritsky I, Ibragimov R, Sarapultsev A. Seroprevalence of SARS-CoV-2 Antibodies in Symptomatic Individuals Is Higher than in Persons Who Are at Increased Risk Exposure: The Results of the Single-Center, Prospective, Cross-Sectional Study. Vaccines. 2021; 9(6):627. https://doi.org/10.3390/vaccines9060627
Chicago/Turabian StyleZurochka, Alexandr, Maria Dobrinina, Vladimir Zurochka, Desheng Hu, Alexandr Solovyev, Liana Ryabova, Igor Kritsky, Roman Ibragimov, and Alexey Sarapultsev. 2021. "Seroprevalence of SARS-CoV-2 Antibodies in Symptomatic Individuals Is Higher than in Persons Who Are at Increased Risk Exposure: The Results of the Single-Center, Prospective, Cross-Sectional Study" Vaccines 9, no. 6: 627. https://doi.org/10.3390/vaccines9060627
APA StyleZurochka, A., Dobrinina, M., Zurochka, V., Hu, D., Solovyev, A., Ryabova, L., Kritsky, I., Ibragimov, R., & Sarapultsev, A. (2021). Seroprevalence of SARS-CoV-2 Antibodies in Symptomatic Individuals Is Higher than in Persons Who Are at Increased Risk Exposure: The Results of the Single-Center, Prospective, Cross-Sectional Study. Vaccines, 9(6), 627. https://doi.org/10.3390/vaccines9060627






