Recent Advances and Future Perspectives in Polymer-Based Nanovaccines
Abstract
1. Introduction
2. Methodology of Literature Review
3. The Application of Polymers in Nanovaccines
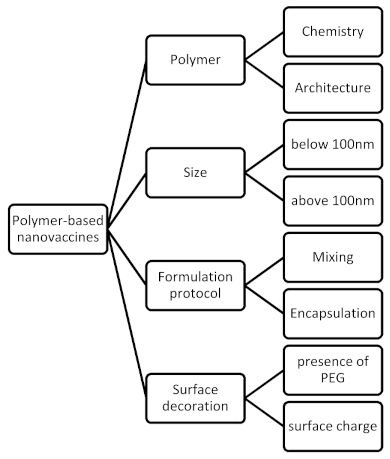
3.1. Polymer-Based Nanovaccines
3.1.1. Micelles
3.1.2. PLGA-Based and Other Biocompatible Nanoparticles
3.2. The Added Value of Polymer-Based Nanovaccines
3.3. The Limitations in the Development of Polymer-Based Nanovaccines
4. Conclusions and Future Perspectives
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Q&As on COVID-19 and Related Health Topics. Available online: https://www.who.int/vaccines/questions-and-answers/q-a-on-vaccines (accessed on 1 June 2013).
- Zhang, Y.; Lin, S.; Wang, X.-Y.; Zhu, G. Nanovaccines for cancer immunotherapy. Wiley Interdiscip. Rev. Nanomed. Nanobiotechnol. 2019, 11, e1559. [Google Scholar] [CrossRef] [PubMed]
- Bernocchi, B.; Carpentier, R.; Betbeder, D. Nasal nanovaccines. Int. J. Pharm. 2017, 530, 128–138. [Google Scholar] [CrossRef] [PubMed]
- Vijayan, V.; Mohapatra, A.; Uthaman, S.; Park, I.-K. Recent Advances in Nanovaccines Using Biomimetic Immunomodulatory Materials. Pharmaceutics 2019, 11, 534. [Google Scholar] [CrossRef]
- Gregory, A.E.; Titball, R.; Williamson, D. Vaccine delivery using nanoparticles. Front. Cell. Infect. Microbiol. 2013, 3, 13. [Google Scholar] [CrossRef] [PubMed]
- Bhardwaj, P.; Bhatia, E.; Sharma, S.; Ahamad, N.; Banerjee, R. Advancements in prophylactic and therapeutic nanovaccines. Acta Biomater. 2020, 108, 1–21. [Google Scholar] [CrossRef] [PubMed]
- Gregory, A.E.; Titball, R.; Williamson, D.; Reichmuth, A.M.; Oberli, M.A.; Jaklenec, A.; Langer, R.; Blankschtein, D. mRNA vaccine delivery using lipid nanoparticles. Ther. Deliv. 2016, 7, 319–334. [Google Scholar] [CrossRef]
- Kheirollahpour, M.; Mehrabi, M.; Dounighi, N.M.; Mohammadi, M.; Masoudi, A. Nanoparticles and Vaccine Development. Pharm. Nanotechnol. 2020, 8, 6–21. [Google Scholar] [CrossRef]
- Kasturi, S.P.; Kozlowski, P.A.; Nakaya, H.I.; Burger, M.C.; Russo, P.; Pham, M.; Kovalenkov, Y.; Silveira, E.L.V.; Haven-ar-Daughton, C.; Burton, S.L.; et al. Adjuvating a Simian Immunodeficiency virus vaccine with Toll-Like Receptor Ligands Encapsulated in Nanoparticles induces Persistent Antibody responses and enhanced protection in TRIM5a restrictive Macaques. J. Virol. 2017, 91, e01844-16. [Google Scholar] [CrossRef]
- Campos, E.V.R.; Pereira, A.E.S.; de Oliveira, J.L.; Carvalho, L.B.; Guilger-Casagrande, M.; de Lima, R.; Fraceto, L.F. How can nanotechnology help to combat COVID-19? Opportunities and urgent need. J. Nanobiotechnol. 2020, 18, 125. [Google Scholar] [CrossRef]
- Chakravarty, M.; Vora, A. Nanotechnology-based antiviral therapeutics. Drug Deliv. Transl. Res. 2021, 11, 748–787. [Google Scholar] [CrossRef]
- Yun, C.-H.; Cho, C.-S. Nanoparticles to Improve the Efficacy of Vaccines. Pharmaceutics 2020, 12, 418. [Google Scholar] [CrossRef]
- Dykman, L.A. Gold nanoparticles for preparation of antibodies and vaccines against infectious diseases. Expert Rev. Vaccines 2020, 19, 465–477. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Guo, J.; Huang, L. Modulation of tumor microenvironment for immunotherapy: Focus on nanomaterial-based strategies. Theranostics 2020, 10, 3099–3117. [Google Scholar] [CrossRef] [PubMed]
- Thomas, C.; Rawat, A.; Hope-Weeks, L.; Ahsan, F. Aerosolized PLA and PLGA Nanoparticles Enhance Humoral, Mucosal and Cytokine Responses to Hepatitis B Vaccine. Mol. Pharm. 2011, 8, 405–415. [Google Scholar] [CrossRef] [PubMed]
- Diwan, M.; Tafaghodi, M.; Samuel, J. Enhancement of immune responses by co-delivery of a CpG oligodeoxynucleotide and tetanus toxoid in biodegradable nanospheres. J. Control. Release 2002, 85, 247–262. [Google Scholar] [CrossRef]
- Manish, M.; Rahi, A.; Kaur, M.; Bhatnagar, R.; Singh, S. A Single-Dose PLGA Encapsulated Protective Antigen Domain 4 Nanoformulation Protects Mice against Bacillus anthracis Spore Challenge. PLoS ONE 2013, 8, e61885. [Google Scholar] [CrossRef]
- Lima, V.M.; Bonato, V.L.; Lima, K.M.; Dos Santos, S.A.; Dos Santos, R.R.; Goncalves, E.D.; Faccioli, L.H.; Brandao, I.T.; Ro-drigues-Junior, J.M.; Silva, C.L. Role of trehalose dimycolate in recruitment of cells and modulation of protection of cytokines and NO in tuberculosis. Infect. Immun. 2001, 69, 5305–5312. [Google Scholar] [CrossRef] [PubMed]
- Borges, O.; Cordeiro-Da-Silva, A.; Tavares, J.; Santarém, N.; de Sousa, A.; Borchard, G.; Junginger, H.E. Immune response by nasal delivery of hepatitis B surface antigen and codelivery of a CpG ODN in alginate coated chitosan nanoparticles. Eur. J. Pharm. Biopharm. 2008, 69, 405–416. [Google Scholar] [CrossRef]
- Li, P.; Luo, Z.; Liu, P.; Gao, N.; Zhang, Y.; Pan, H.; Liu, L.; Wang, C.; Cai, L.; Ma, Y. Bioreducible alginate-poly(ethylenimine) nanogels as an antigen delivery sytems robustly enhance vaccine-elicited humoral and cellular immune responses. J. Control. Release 2013, 168, 271–279. [Google Scholar] [CrossRef]
- Hasegawa, K.; Noguchi, Y.; Koizumi, F.; Uenaka, A.; Tanaka, M.; Shimono, M.; Nakamura, H.; Shiku, H.; Gnjatic, S.; Murphy, R.; et al. In vitro Stimulation of CD8 and CD4 T Cells by Dendritic Cells Loaded with a Complex of Cholesterol-Bearing Hydrophobized Pullulan and NY-ESO-1 Protein: Identification of a New HLA-DR15–Binding CD4 T-Cell Epitope. Clin. Cancer Res. 2006, 12, 1921–1927. [Google Scholar] [CrossRef]
- Saade, F.; Honda-Okubo, Y.; Trec, S.; Petrovsky, N. A novel hepatitis B vaccine containing Advax™, a polysaccharide adjuvant derived from delta inulin, induces robust humoral and cellular immunity with minimal reactogenicity in preclinical testing. Vaccine 2013, 31, 1999–2007. [Google Scholar] [CrossRef] [PubMed]
- Feng, G.; Jiang, Q.; Xia, M.; Lu, Y.; Qiu, W.; Zhao, D.; Lu, L.; Peng, G.; Wang, Y. Enhanced Immune Response and Protective Effects of Nano-chitosan-based DNA Vaccine Encoding T Cell Epitopes of Esat-6 and FL against Mycobacterium Tuberculosis Infection. PLoS ONE 2013, 8, e61135. [Google Scholar] [CrossRef] [PubMed]
- Glaffig, M.; Palitzsch, B.; Hartmann, S.; Schüll, C.; Nuhn, L.; Gerlitzki, B.; Schmitt, E.; Frey, H.; Kunz, H. A Fully Synthetic Glycopeptide Antitumor Vaccine Based on Multiple Antigen Presentation on a Hyperbranched Polymer. Chem. A Eur. J. 2014, 20, 4232–4236. [Google Scholar] [CrossRef] [PubMed]
- Luo, Z.; Li, P.; Deng, J.; Gao, N.; Zhang, Y.; Pan, H.; Liu, L.; Wang, C.; Cai, L.; Ma, Y. Cationic polypeptide micelle-based antigen delivery system: A simple and robust adjuvant to improve vaccine efficacy. J. Control. Release 2013, 170, 259–267. [Google Scholar] [CrossRef]
- Zhang, C.; Shi, G.; Zhang, J.; Song, H.; Niu, J.; Shi, S.; Huang, P.; Wang, Y.; Wang, W.; Li, C.; et al. Targeted antigen delivery to dendritic cell via functionalized alginate nanoparticles for cancer immunotherapy. J. Control. Release 2017, 256, 170–181. [Google Scholar] [CrossRef]
- Yan, Y.; Ding, H. pH-Responsive Nanoparticles for Cancer Immunotherapy: A Brief Review. Nanomaterials 2020, 10, 1613. [Google Scholar] [CrossRef]
- Démoulins, T.; Bassi, I.; Thomann-Harwood, L.; Jandus, C.; Kaeuper, P.; Simon, H.-U.; von Gunten, S.; McCullough, K.C. Alginate-coated chitosan nanogel capacity to modulate the effect of TLR ligands on blood dendritic cells. Nanomed. Nanotechnol. Biol. Med. 2013, 9, 806–817. [Google Scholar] [CrossRef]
- Démoulins, T.; Milona, P.; McCullough, K.C. Alginate-coated chitosan nanogels differentially modulate class-A and class-B CpG-ODN targeting of dendritic cells and intracellular delivery. Nanomedicine 2014, 10, 1739–1749. [Google Scholar] [CrossRef]
- Trimaille, T.; Verrier, B. Micelle-Based Adjuvants for Subunit Vaccine Delivery. Vaccines 2015, 3, 803–813. [Google Scholar] [CrossRef] [PubMed]
- Trimaille, T.; Lacroix, C.; Verrier, B. Self-assembled amphiphilic copolymers as dual delivery system for immunotherapy. Eur. J. Pharm. Biopharm. 2019, 142, 232–239. [Google Scholar] [CrossRef]
- Luo, Z.; Shi, S.; Jin, L.; Xu, L.; Yu, J.; Chen, H.; Li, X. Cationic micelle based vaccine induced potent humoral immune response through enhancing antigen uptake and formation of germinal center. Colloids Surf. B Biointerfaces 2015, 135, 556–564. [Google Scholar] [CrossRef]
- Keller, S.; Wilson, J.T.; Patilea, G.I.; Kern, H.B.; Convertine, A.J.; Stayton, P.S. Neutral polymer micelle carriers with pH-responsive, endosome-releasing activity modulate antigen trafficking to enhance CD8(+) T cell responses. J. Control. Release 2014, 191, 24–33. [Google Scholar] [CrossRef]
- Wilson, J.T.; Keller, S.; Manganiello, M.J.; Cheng, C.; Lee, C.C.; Opara, C.; Convertine, A.; Stayton, P.S. pH-Responsive nanoparticle vaccines for dual-delivery of antigens and immunostimulatory oligonucleotides. ACS Nano 2013, 7, 3912–3925. [Google Scholar] [CrossRef] [PubMed]
- Wusiman, A.; Gu, P.; Liu, Z.; Xu, S.; Zhang, Y.; Hu, Y.; Liu, J.; Wang, D.; Huang, X. Cationic polymer modified PLGA nano-particles encapsulating Alhagi honey polysaccharides as a vaccine delivery system for ovalbumin to improve immune responses. Int. J. Nanomed. 2019, 14, 3221–3234. [Google Scholar] [CrossRef]
- Wusiman, A.; He, J.; Zhu, T.; Liu, Z.; Gu, P.; Hu, Y.; Liu, J.; Wang, D. Macrophage immunomodulatory activity of the cati-onic polymer modified PLGA nanoparticles encapsulating Alhagi honey polysaccharide. Int. J. Biol. Macromol. 2019, 134, 730–739. [Google Scholar] [CrossRef] [PubMed]
- Wusiman, A.; Xu, S.; Ni, H.; Gu, P.; Liu, Z.; Zhang, Y.; Qiu, T.; Hu, Y.; Liu, J.; Wu, Y.; et al. Immunomodulatory effects of Alhagi honey polysaccharides encapsulated into PLGA nanoparticles. Carbohydr. Polym. 2019, 211, 217–226. [Google Scholar] [CrossRef] [PubMed]
- Gu, P.; Liu, Z.; Sun, Y.; Ou, N.; Hu, Y.; Liu, J.; Wu, Y.; Wang, D. Angelica sinensis polysaccharide encapsulated into PLGA nanoparticles as a vaccine delivery and adjuvant system for ovalbumin to promote immune responses. Int. J. Pharm. 2019, 554, 72–80. [Google Scholar] [CrossRef] [PubMed]
- Gu, P.; Wusiman, A.; Wang, S.; Zhang, Y.; Liu, Z.; Hu, Y.; Liu, J.; Wang, D. Polyethylenimine-coated PLGA nanoparticles-encapsulated Angelica sinensis polysaccharide as an adjuvant to enhance immune responses. Carbohydr. Polym. 2019, 223, 115128. [Google Scholar] [CrossRef]
- Gu, P.; Wusiman, A.; Zhang, Y.; Liu, Z.; Bo, R.; Hu, Y.; Liu, J.; Wang, D. Rational Design of PLGA Nanoparticle Vaccine Delivery Systems To Improve Immune Responses. Mol. Pharm. 2019, 16, 5000–5012. [Google Scholar] [CrossRef]
- Rietscher, R.; Schröder, M.; Janke, J.; Czaplewska, J.; Gottschaldt, M.; Scherließ, R.; Hanefeld, A.; Schubert, U.S.; Schneider, M.; Knolle, P.A.; et al. Antigen delivery via hydrophilic PEG-b-PAGE-b-PLGA nanoparticles boosts vac-cination induced T cell immunity. Eur. J. Pharm. Biopharm. 2016, 102, 20–31. [Google Scholar] [CrossRef]
- Lim, Y.T.; Song, C.; Noh, Y.-W. Polymer nanoparticles for cross-presentation of exogenous antigens and enhanced cytotoxic T-lymphocyte immune response. Int. J. Nanomed. 2016, 11, 3753–3764. [Google Scholar] [CrossRef]
- Koerner, J.; Horvath, D.; Groettrup, M. Harnessing Dendritic Cells for Poly (D,L-lactide-co-glycolide) Microspheres (PLGA MS)—Mediated Anti-tumor Therapy. Front. Immunol. 2019, 10, 707. [Google Scholar] [CrossRef]
- Flanary, S.; Hoffman, A.S.; Stayton, P.S. Antigen Delivery with Poly(Propylacrylic Acid) Conjugation Enhances MHC-1 Presentation and T-Cell Activation. Bioconjug. Chem. 2009, 20, 241–248. [Google Scholar] [CrossRef] [PubMed]
- Foster, S.; Duvall, C.L.; Crownover, E.F.; Hoffman, A.S.; Stayton, P.S. Intracellular delivery of a protein antigen with an en-dosomal-releasing polymer enhances CD8 T-cell production and prophylactic vaccine efficacy. Bioconjug. Chem. 2010, 21, 2205–2212. [Google Scholar] [CrossRef]
- Prego, C.; Paolicelli, P.; Díaz, B.; Vicente, S.; Sánchez, A.; González-Fernández, Á.; Alonso, M.J. Chitosan-based nanoparticles for improving immunization against hepatitis B infection. Vaccine 2010, 28, 2607–2614. [Google Scholar] [CrossRef] [PubMed]
- Borges, O.; Silva, M.; de Sousa, A.; Borchard, G.; Junginger, H.E.; Cordeiro-da-Silva, A. Alginate coated chitosan nanoparticles are an effective subcutaneous adjuvant for hepatitis B surface antigen. Int. Immunopharmacol. 2008, 8, 1773–1780. [Google Scholar] [CrossRef] [PubMed]
- Gupta, N.K.; Tomar, P.; Sharma, V.; Dixit, V.K. Development and characterization of chitosan coated poly-(ɛ-caprolactone) nanoparticulate system for effective immunization against influenza. Vaccine 2011, 29, 9026–9037. [Google Scholar] [CrossRef]
- Matsuo, K.; Ishii, Y.; Quan, Y.S.; Kamiyama, F.; Mukai, Y.; Yoshioka, Y.; Okada, N.; Nakagawa, S. Transcutaneous vaccina-tion using a hydrogel patch induces effective immune responses to tetanus and diphtheria toxoid in hairless rat. J. Control. Release 2011, 149, 15–20. [Google Scholar] [CrossRef]
- Hirobe, S.; Matsuo, K.; Quan, Y.-S.; Kamiyama, F.; Morito, H.; Asada, H.; Takaya, Y.; Mukai, Y.; Okada, N.; Nakagawa, S. Clinical study of transcutaneous vaccination using a hydrogel patch for tetanus and diphtheria. Vaccine 2012, 30, 1847–1854. [Google Scholar] [CrossRef]
- De Temmerman, M.-L.; Rejman, J.; Vandenbroucke, R.; De Koker, S.; Libert, C.; Grooten, J.; Demeester, J.; Gander, B.; De Smedt, S.C. Polyelectrolyte LbL microcapsules versus PLGA microparticles for immunization with a protein antigen. J. Control. Release 2012, 158, 233–239. [Google Scholar] [CrossRef]
- San Román, B.; Irache, J.M.; Gómez, S.; Tsapis, N.; Gamazo, C.; Espuelas, M.S. Co-encapsulation of an antigen and CpG oligonucleotides into PLGA microparticles by TROMS technology. Eur. J. Pharm. Biopharm. 2008, 70, 98–108. [Google Scholar] [CrossRef] [PubMed]
- Sarti, F.; Perera, G.; Hintzen, F.; Kotti, K.; Karageorgiou, V.; Kammona, O.; Kiparissides, C.; Bernkop-Schnürch, A. In vivo evidence of oral vaccination with PLGA nanoparticles containing the immunostimulant monophosphoryl lipid A. Biomaterials 2011, 32, 4052–4057. [Google Scholar] [CrossRef] [PubMed]
- Slütter, B.; Bal, S.; Keijzer, C.; Mallants, R.; Hagenaars, N.; Que, I.; Kaijzel, E.; van Eden, W.; Augustijns, P.; Löwik, C.; et al. Nasal vaccination with N-trimethyl chitosan and PLGA based nanoparticles: Nanoparticle charac-teristics determine quality and strength of the antibody response in mice against the encapsulated antigen. Vaccine 2010, 28, 6282–6291. [Google Scholar] [CrossRef]
- Rauta, P.R.; Nayak, B. Parenteral immunization of PLA/PLGA nanoparticle encapsulating outer membrane protein (Omp) from Aeromonas hydrophila: Evaluation of immunostimulatory action in Labeo rohita (rohu). Fish Shellfish Immunol. 2015, 44, 287–294. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Zheng, S.; Luo, L.; Bo, R.; Liu, Z.; Xing, J.; Niu, Y.; Hu, Y.; Liu, J. Evaluation of optimum conditions for pachyman encapsulated in poly(D,L-lactic acid) nanospheres by response surface methodology and results of a related in vitro study. Int. J. Nanomed. 2016, 11, 4891–4904. [Google Scholar] [CrossRef]
- Zheng, S.; Qin, T.; Lu, Y.; Huang, Y.; Luo, L.; Liu, Z.; Bo, R.; Hu, Y.; Liu, J.; Wang, D. Maturation of dendritic cells in vitro and immunological enhancement of mice in vivo by pachyman- and/or OVA-encapsulated poly(D,L-lactic acid) nanospheres. Int. J. Nanomed. 2018, 13, 569–583. [Google Scholar] [CrossRef]
- Han, R.; Zhu, J.; Yang, X.; Xu, H. Surface modification of poly(D,L-lactic-co-glycolic acid) nanoparticles with protamine enhanced cross-presentation of encapsulated ovalbumin by bone marrow-derived dendritic cells. J. Biomed. Mater. Res. A 2011, 96, 142–149. [Google Scholar] [CrossRef]
- Liu, Q.; Chen, X.; Jia, J.; Zhang, W.; Yang, T.; Wang, L.; Ma, G. pH-Responsive Poly(d,l-lactic-co-glycolic acid) Nanoparticles with Rapid Antigen Release Behavior Promote Immune Response. ACS Nano 2015, 9, 4925–4938. [Google Scholar] [CrossRef]
- Heo, M.B.; Cho, M.Y.; Lim, Y.T. Polymer nanoparticles for enhanced immune response: Combined delivery of tumor antigen and small interference RNA for immunosuppressive gene to dendritic cells. Acta Biomater. 2014, 10, 2169–2176. [Google Scholar] [CrossRef]
- Hamdy, S.; Haddadi, A.; Hung, R.W.; Lavasanifar, A. Targeting dendritic cells with nano-particulate PLGA cancer vaccine formulations. Adv. Drug Deliv. Rev. 2011, 63, 943–955. [Google Scholar] [CrossRef]
- Li, L.; Yang, Z.; Chen, X. Recent Advances in Stimuli-Responsive Platforms for Cancer Immunotherapy. Acc. Chem. Res. 2020, 53, 2044–2054. [Google Scholar] [CrossRef]
- Aikins, M.E.; Xu, C.; Moon, J.J. Engineered Nanoparticles for Cancer Vaccination and Immunotherapy. Acc. Chem. Res. 2020, 53, 2094–2105. [Google Scholar] [CrossRef] [PubMed]
- Xia, Y.; Song, T.; Hu, Y.; Ma, G. Synthetic Particles for Cancer Vaccines: Connecting the Inherent Supply Chain. Acc. Chem. Res. 2020, 53, 2068–2080. [Google Scholar] [CrossRef]
- Shen, Y.; Hao, T.; Ou, S.; Hu, C.; Chen, L. Applications and perspectives of nanomaterials in novel vaccine development. MedChemComm 2017, 9, 226–238. [Google Scholar] [CrossRef] [PubMed]
- Silva, A.L.; Soema, P.C.; Slütter, B.; Ossendorp, F.; Jiskoot, W. PLGA particulate delivery systems for subunit vaccines: Linking particle properties to immunogenicity. Hum. Vaccines Immunother. 2016, 12, 1056–1069. [Google Scholar] [CrossRef] [PubMed]
- Jiang, W.; Gupta, R.K.; Deshpande, M.C.; Schwendeman, S.P. Biodegradable poly(lactic-co-glycolic acid) microparticles for injectable delivery of vaccine antigens. Adv. Drug Deliv. Rev. 2005, 57, 391–410. [Google Scholar] [CrossRef]
- Sah, H.; Thoma, L.A.; Desu, H.R.; Sah, E.; Wood, G.C. Concepts and practices used to develop functional PLGA-based na-noparticulate systems. Int. J. Nanomed. 2013, 8, 747–765. [Google Scholar] [CrossRef]
- Perrie, Y.; Kastner, E.; Kaur, R.; Wilkinson, A.; Ingham, A.J. A case-study investigating the physicochemical characteristics that dictate the function of a liposomal adjuvant. Hum. Vaccines Immunother. 2013, 9, 1374–1381. [Google Scholar] [CrossRef]
- Uto, T.; Akagi, T.; Toyama, M.; Nishi, Y.; Shima, F.; Akashi, M.; Baba, M. Comparative activity of biodegradable nanoparticles with aluminum adjuvants: Antigen uptake by dendritic cells and induction of immune response in mice. Immunol. Lett. 2011, 140, 36–43. [Google Scholar] [CrossRef]
- Uto, T.; Wang, X.; Sato, K.; Haragushi, M.; Akagi, T.; Akashi, M.; Baba, M. Targeting of antigen to dendritic cells with poly(γ-glutamic acid) nanoparticles induces antigen specific humoral and cellular immunity. J. Immunol. 2007, 178, 2979–2986. [Google Scholar] [CrossRef]
- Uto, T.; Akagi, T.; Hamasaki, T.; Akashi, M.; Baba, M. Modulation of innate and adaptive immunity by biodegradable nanoparticles. Immunol. Lett. 2009, 125, 46–52. [Google Scholar] [CrossRef]
- Wang, X.; Uto, T.; Akagi, T.; Akashi, M.; Baba, M. Poly(gamma-glutamic acid) nanoparticles as an efficient antigen delivery and adjuvant system: Potential for an AIDS vaccine. J. Med. Virol. 2008, 80, 11–19. [Google Scholar] [CrossRef] [PubMed]
- Uto, T.; Akagi, T.; Yoshinaga, K.; Toyama, M.; Akashi, M.; Baba, M. The induction of innate and adaptive immunity by bio-degradable poly(γ-glutamic acid) nanoparticles via a TLR4 and MyD88 signalling pathway. Biomaterials 2011, 32, 5206–5212. [Google Scholar] [CrossRef] [PubMed]
- Shima, F.; Akagi, T.; Akashi, M. Effect of hydrophobic side chains in the induction o immune responses by nanoparticle ad-juvants consisting of amphiphilic Poly(γ-glutamic acid). Bioconjug. Chem. 2015, 26, 890–898. [Google Scholar] [CrossRef] [PubMed]
- Shima, F.; Akagi, T.; Uto, T.; Akashi, M. Manipulating the antigen-specific immune response by the hydrophobicity of amphiphilic poly-(γ-glutamic acid) nanoparticles. Biomaterials 2013, 34, 9709–9716. [Google Scholar] [CrossRef] [PubMed]
- Shima, F.; Uto, T.; Akagi, T.; Baba, M.; Akashi, M. Size effect of amphiphilic poly(γ-glutamic acid) nanoparticles on cellular uptake and maturation of dendritic cells in vivo. Acta Biomater. 2013, 9, 8894–8901. [Google Scholar] [CrossRef]
- Thomann-Harwood, L.J.; Kaeuper, P.; Rossi, N.; Milona, P.; Herrmann, B.; McCullough, K.C. Nanpgel vaccines targeting dendritic cells: Contributions of the surface decoration and vaccine cargo on cell targeting and activation. J. Control. Release 2012, 166, 95–105. [Google Scholar] [CrossRef]
- Cruz, L.J.; Tacken, P.J.; Fokkink, R.; Figdor, C.G. The influence of PEG chain length and targeting moiety on anti-body-mediated delivery of nanoparticle vaccines to human dendritic cells. Biomaterials 2011, 32, 6791–6803. [Google Scholar] [CrossRef]
- de Oliveira, C.I.; Santos, D.M.; Carneiro, M.W.; de Moura, T.R.; Fukutani, K.; Clarencio, J.; Soto, M.; Espuelas, S.; Brodskyn, C.; Barral, A.; et al. Towards development of novel immunization strategies against leishmaniasis using PLGA nanoparticles loaded with kinetoplastid membrane protein-11. Int. J. Nanomed. 2012, 7, 2115–2127. [Google Scholar] [CrossRef]
- Han, J.; Zhao, D.; Li, D.; Wang, X.; Jin, Z.; Zhao, K. Polymer-Based Nanomaterials and Applications for Vaccines and Drugs. Polymers 2018, 10, 31. [Google Scholar] [CrossRef]
- Zhou, J.; Krishnan, N.; Jiang, Y.; Fang, R.H.; Zhang, L. Nanotechnology for virus treatment. Nano Today 2021, 36, 101031. [Google Scholar] [CrossRef]
- Van Der Weken, H.; Cox, E.; Devriendt, B. Advances in Oral Subunit Vaccine Design. Vaccines 2020, 9, 1. [Google Scholar] [CrossRef] [PubMed]
- Al-Halifa, S.; Gauthier, L.; Arpin, D.; Bourgault, S.; Archambault, D. Nanoparticle-Based Vaccines against Respiratory Viruses. Front. Immunol. 2019, 10, 22. [Google Scholar] [CrossRef] [PubMed]
- Wibowo, D.; Jorritsma, S.H.; Gonzaga, Z.J.; Evert, B.; Chen, S.; Rehm, B.H. Polymeric nanoparticle vaccines to combat emerging and pandemic threats. Biomaterials 2021, 268, 120597. [Google Scholar] [CrossRef]
- Siegrist, S.; Cörek, E.; Detampel, P.; Sandström, J.; Wick, P.; Huwyler, J. Preclinical hazard evaluation strategy for nanomedicines. Nanotoxicology 2019, 13, 73–79. [Google Scholar] [CrossRef]
- Patel, P.; Shah, J. Safety and Toxicological Considerations of Nanomedicines: The Future Directions. Curr. Clin. Pharmacol. 2018, 12, 73–82. [Google Scholar] [CrossRef]
- Swierczewska, M.; Crist, R.M.; McNeil, S.E. Evaluating Nanomedicines: Obstacles and Advancements. Adv. Struct. Saf. Stud. 2017, 1682, 3–16. [Google Scholar] [CrossRef]
- Duncan, R. Polymer therapeutics at a crossroads? Finding the path for improved translation in the twenty-first century. J. Drug Target. 2017, 25, 759–780. [Google Scholar] [CrossRef] [PubMed]
- Stojanowski, J.; Gołębiowski, T. Focus on COVID-19: Antiviral polymers in drugs and vaccines. Polym. Med. 2021, 50, 75–78. [Google Scholar] [CrossRef]
- de Queiroz, N.M.G.P.; Marinho, F.V.; Chagas, M.A.; Leite, L.C.C.; Homan, E.J.; de Magalhães, M.T.Q.; Oliveira, S.C. Vaccines for COVID-19: Perspectives from nucleic acid vaccines to BCG as delivery vector system. Microbes Infect. 2020, 22, 515–524. [Google Scholar] [CrossRef]
- Silveira, M.M.; Moreira, G.M.S.G.; Mendonça, M. DNA vaccines against COVID-19: Perspectives and challenges. Life Sci. 2021, 267, 118919. [Google Scholar] [CrossRef] [PubMed]
- Jiang, X.; Li, Z.; Young, D.J.; Liu, M.; Wu, C.; Loh, X.J. Toward the prevention of coronavirus infection: What role can poly-mers play? Mater. Today Adv. 2021, 10, 100140. [Google Scholar] [CrossRef] [PubMed]
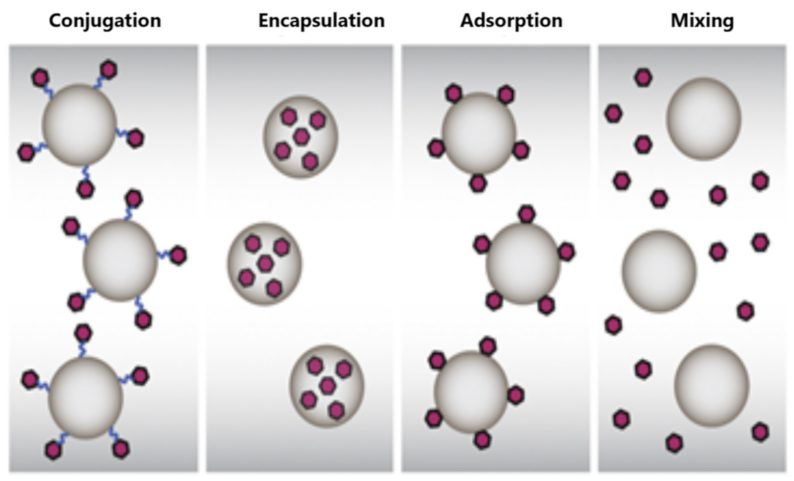
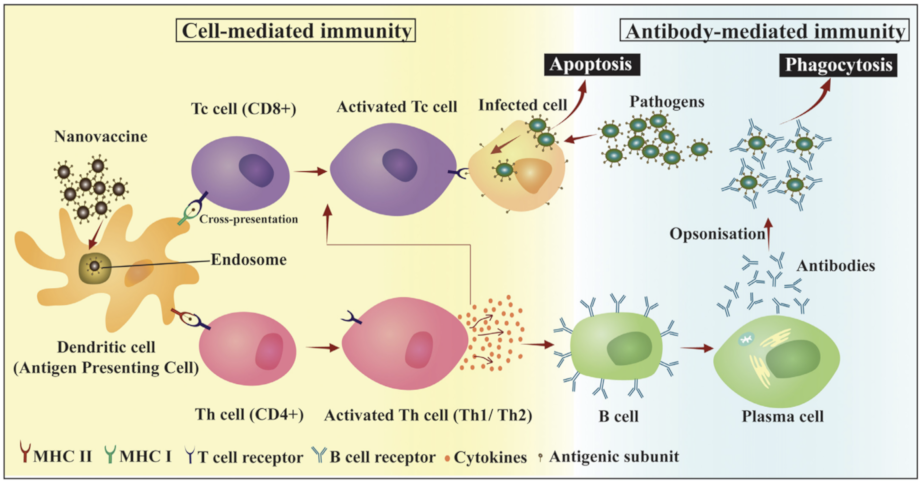

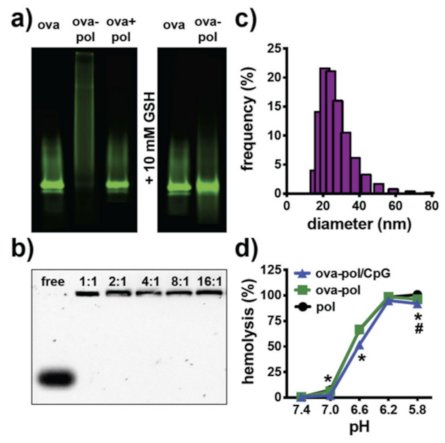
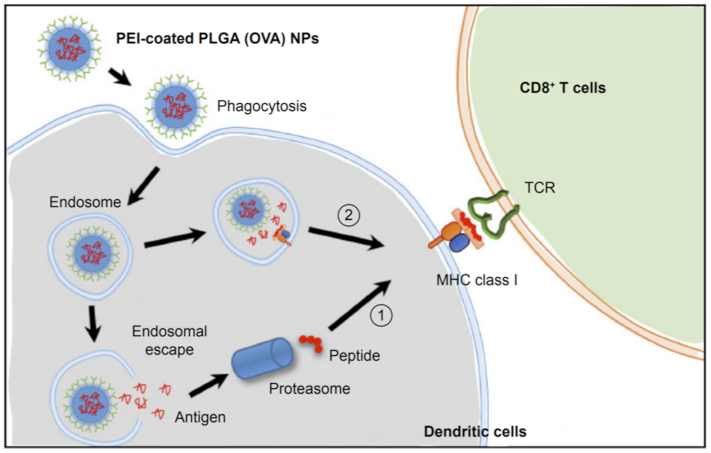
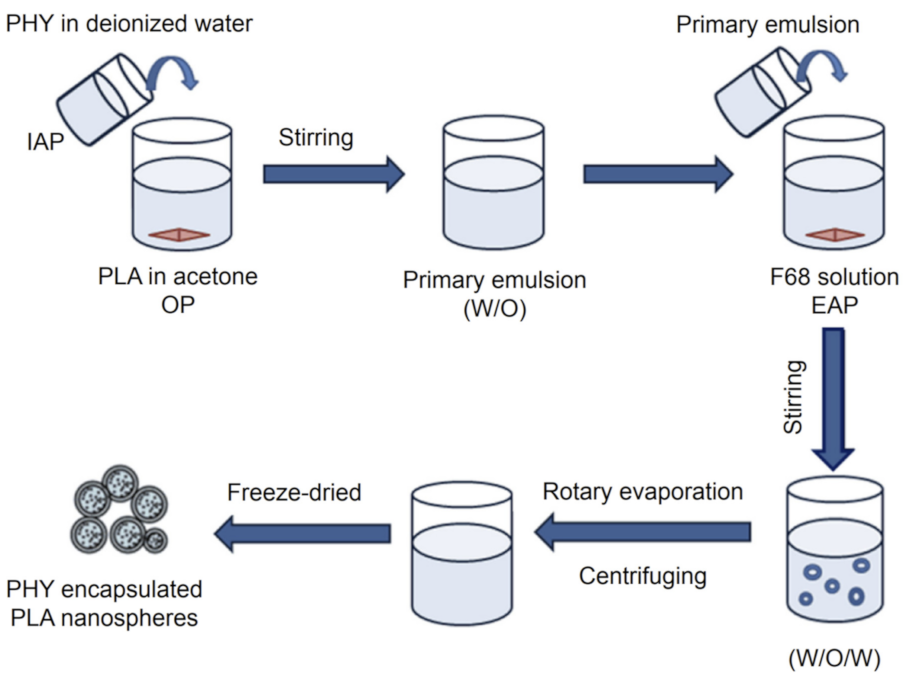
| Types | Size | Shape | Bio-Toxicity | Biocompatibility | Disadvantages |
|---|---|---|---|---|---|
| VPLs | 20–800 nm | Particle | Some unpredictable consequences | Highly cross-protective antibody responses | Low immunogenicity |
| SAP | 10 nm length (70 nm) and wide (40 nm) | Spherical or barrel | - | Reduce the risk of immunogenicity; improve half-life | Creates a new molecular entity; poor solution stability and aggregation |
| CNPs | 30–60 nm length (100–1000 nm) and diameter (0.8–2.0 nm) | Fullerene particle, Nanotube, or mesoporous spheres | Negligible effect on cell viability | Good biocompatibility | Low immune function |
| GNs | 2–150 nm | Particle, rod, spherical, and cubic | Immuno-toxicity | More potent immune response | Immuno-toxicity |
| CNs | 50–100 nm | Calcium phosphate particle | Biocompatible and safe | Biocompatibility and easily biodegradable | - |
| SNs | 50–20 nm | Tunable hollow and mesoporous structure | Toxicity degraded | Biocompatibility and selective tumor targeting, real-time multimodal imaging, vaccine delivery | Toxicity derived from the reducing agents |
| LSs | 25–1000 nm | Spherical | Safely degraded | Stabilize the antigen, biocompatible and stable | - |
| Polymer | 10–2000 nm | Particles | Non-toxicity | Antigen loading into polymeric particles under aqueous conditions via a self-healing process | Loss of antigenicity and immunogenicity during particle synthesis |
| ISCOM | 40 nm | Cage-like particles | Cytotoxicity-mediated immune responses | - | Cytotoxicity-mediated immune responses |
| EN | 50–600 nm | Cytotoxicity-mediated immune responses | Safe | Safe and potent vaccine adjuvant | - |
| Advantages | Disadvantages |
|---|---|
|
|
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pippa, N.; Gazouli, M.; Pispas, S. Recent Advances and Future Perspectives in Polymer-Based Nanovaccines. Vaccines 2021, 9, 558. https://doi.org/10.3390/vaccines9060558
Pippa N, Gazouli M, Pispas S. Recent Advances and Future Perspectives in Polymer-Based Nanovaccines. Vaccines. 2021; 9(6):558. https://doi.org/10.3390/vaccines9060558
Chicago/Turabian StylePippa, Natassa, Maria Gazouli, and Stergios Pispas. 2021. "Recent Advances and Future Perspectives in Polymer-Based Nanovaccines" Vaccines 9, no. 6: 558. https://doi.org/10.3390/vaccines9060558
APA StylePippa, N., Gazouli, M., & Pispas, S. (2021). Recent Advances and Future Perspectives in Polymer-Based Nanovaccines. Vaccines, 9(6), 558. https://doi.org/10.3390/vaccines9060558








