Adjuvant Selection for Influenza and RSV Prefusion Subunit Vaccines
Abstract
1. Introduction
2. Materials and Methods
2.1. Antigen Production
2.2. Adjuvant Formulation and Mouse Immunizations
2.3. T-Cell Cytokine Profiling
2.4. ELISAs
2.5. Plaque Reduction Neutralisation Tests (PRNTs)
2.6. Statistics
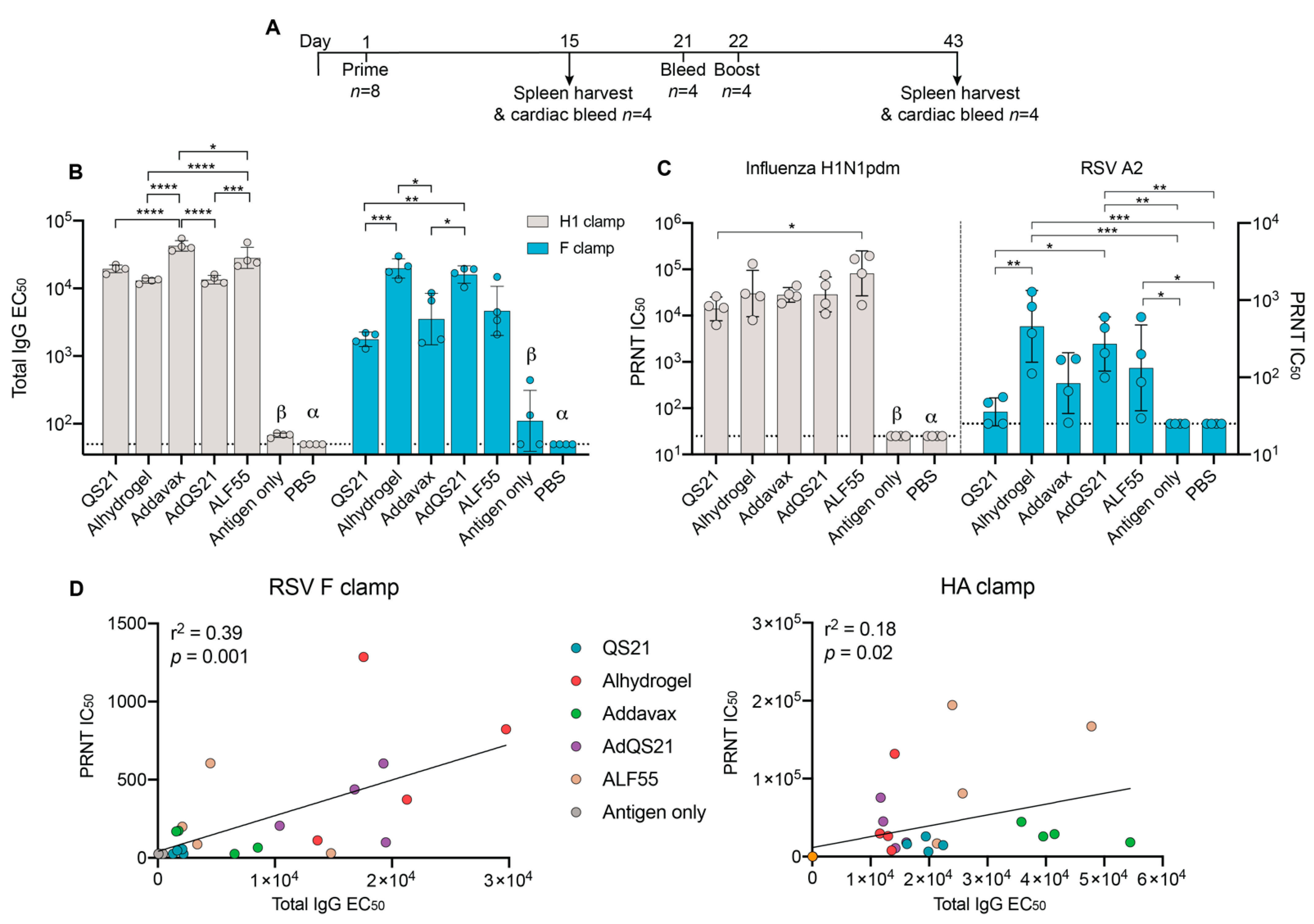
3. Results
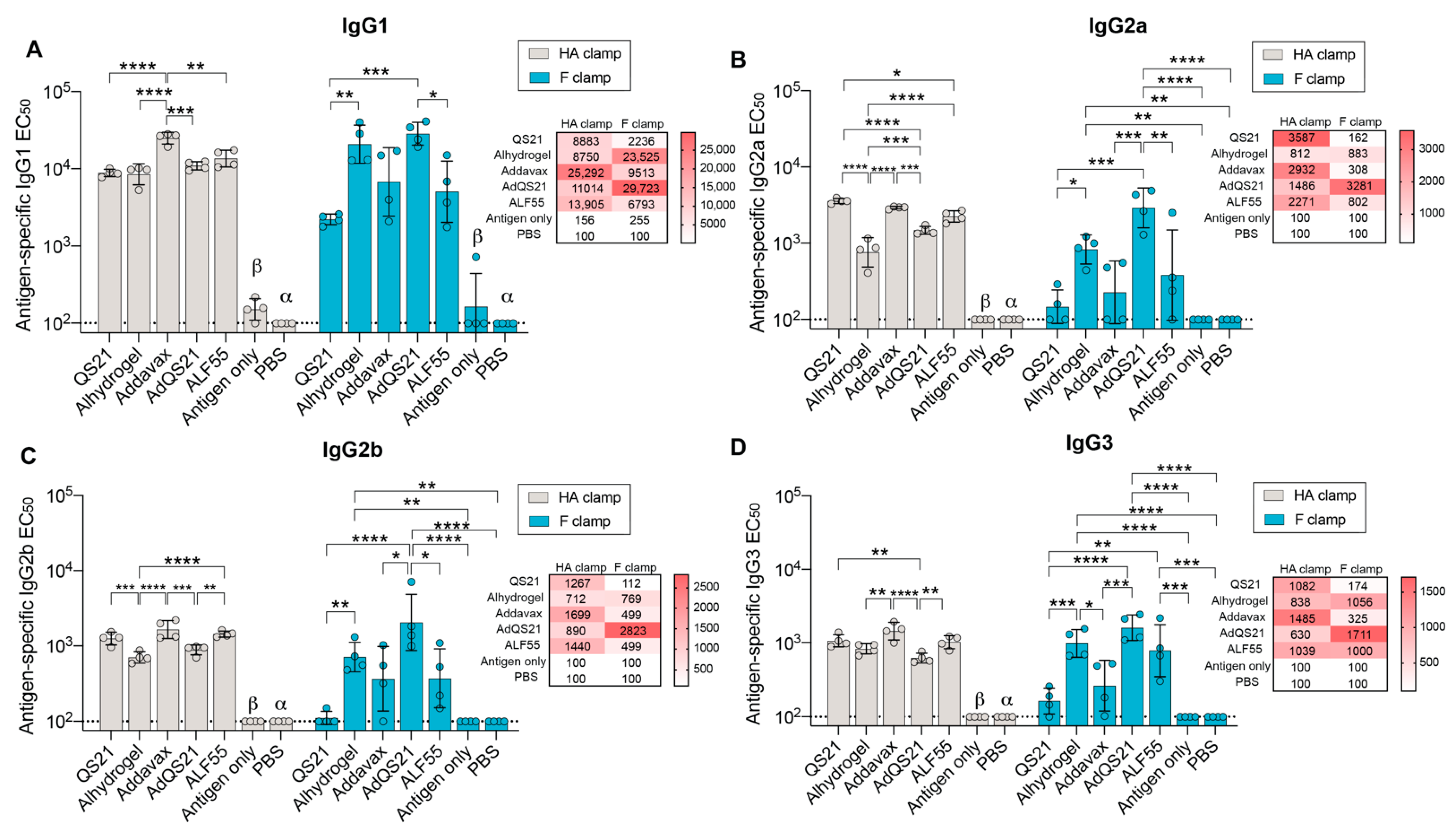
3.1. Humoral Responses
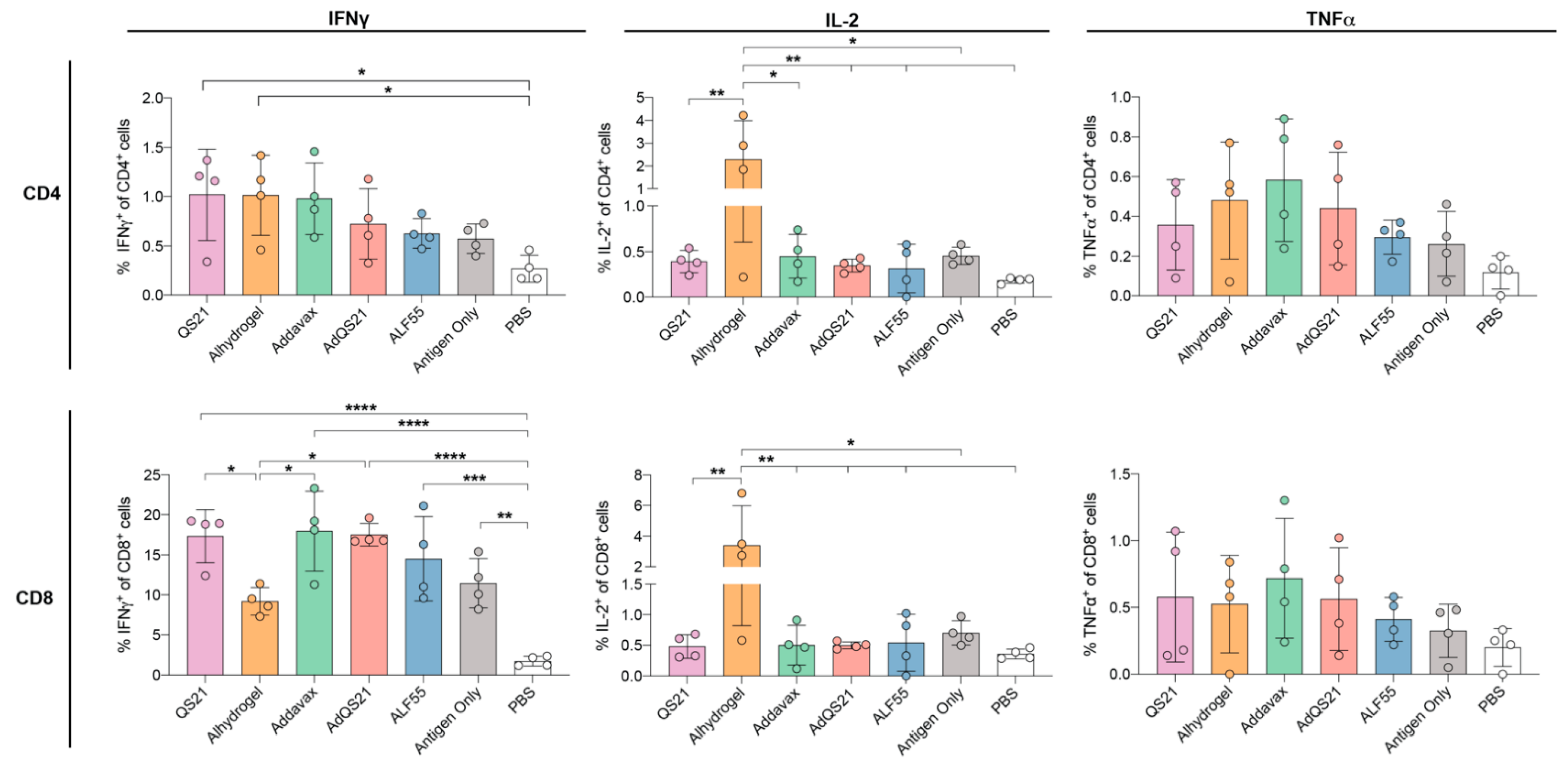
3.2. T-Cell Responses
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Krammer, F. SARS-CoV-2 vaccines in development. Nature 2020, 586, 516–527. [Google Scholar] [CrossRef] [PubMed]
- Moyle, P.M.; Toth, I. Modern Subunit Vaccines: Development, Components, and Research Opportunities. Chem. Med. Chem. 2013, 8, 360–376. [Google Scholar] [CrossRef] [PubMed]
- George, P.J.; Tai, W.; Du, L.; Lustigman, S. The Potency of an Anti-MERS Coronavirus Subunit Vaccine Depends on a Unique Combinatorial Adjuvant Formulation. Vaccines 2020, 8, 251. [Google Scholar] [CrossRef] [PubMed]
- Del Giudice, G.; Rappuoli, R.; Didierlaurent, A.M. Correlates of adjuvanticity: A review on adjuvants in licensed vaccines. Semin. Immunol. 2018, 39, 14–21. [Google Scholar] [CrossRef] [PubMed]
- Knudsen, N.P.H.; Olsen, A.; Buonsanti, C.; Follmann, F.; Zhang, Y.; Coler, R.N.; Fox, C.B.; Meinke, A.; D´Oro, U.; Casini, D.; et al. Different human vaccine adjuvants promote distinct antigen-independent immunological signatures tailored to different pathogens. Sci. Rep. 2016, 6, 19570. [Google Scholar] [CrossRef]
- Reed, S.G.; Orr, M.T.; Fox, C.B. Key roles of adjuvants in modern vaccines. Nat. Med. 2013, 19, 1597–1608. [Google Scholar] [CrossRef]
- Younis, S.Y.; Barnier-Quer, C.; Heuking, S.; Sommandas, V.; Brunner, L.; Vd.Werff, N.; Dubois, P.; Friede, M.; Kocken, C.; Collin, N.; et al. Down selecting adjuvanted vaccine formulations: A comparative method for harmonized evaluation. BMC Immunol. 2018, 19, 6. [Google Scholar] [CrossRef]
- Hawken, J.; Troy, S.B. Adjuvants and inactivated polio vaccine: A systematic review. Vaccine 2012, 30, 6971–6979. [Google Scholar] [CrossRef]
- De Gregorio, E.; Tritto, E.; Rappuoli, R. Alum adjuvanticity: Unraveling a century old mystery. Eur. J. Immunol. 2008, 38, 2068–2071. [Google Scholar] [CrossRef]
- Gupta, R.K. Aluminum compounds as vaccine adjuvants. Adv. Drug Deliv. Rev. 1998, 32, 155–172. [Google Scholar] [CrossRef]
- Brewer, J.M. (How) do aluminium adjuvants work? Immunol. Lett. 2006, 102, 10–15. [Google Scholar] [CrossRef] [PubMed]
- Oleszycka, E.; Lavelle, E.C. Immunomodulatory properties of the vaccine adjuvant alum. Curr. Opin. Immunol. 2014, 28, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Mori, A.; Oleszycka, E.; Sharp, F.A.; Coleman, M.; Ozasa, Y.; Singh, M.; O’Hagan, D.T.; Tajber, L.; Corrigan, O.I.; McNeela, E.A.; et al. The vaccine adjuvant alum inhibits IL-12 by promoting PI3 kinase signaling while chitosan does not inhibit IL-12 and enhances Th1 and Th17 responses. Eur. J. Immunol. 2012, 42, 2709–2719. [Google Scholar] [CrossRef] [PubMed]
- Didierlaurent, A.M.; Morel, S.; Lockman, L.; Giannini, S.L.; Bisteau, M.; Carlsen, H.; Kielland, A.; Vosters, O.; Vanderheyde, N.; Schiavetti, F.; et al. AS04, an Aluminum Salt- and TLR4 Agonist-Based Adjuvant System, Induces a Transient Localized Innate Immune Response Leading to Enhanced Adaptive Immunity. J. Immunol. 2009, 183, 6186. [Google Scholar] [CrossRef]
- Casella, C.R.; Mitchell, T.C. Putting endotoxin to work for us: Monophosphoryl lipid A as a safe and effective vaccine adjuvant. Cell Mol. Life Sci. 2008, 65, 3231. [Google Scholar] [CrossRef]
- Baldridge, J.R.; McGowan, P.; Evans, J.T.; Cluff, C.; Mossman, S.; Johnson, D.; Persing, D. Taking a Toll on human disease: Toll-like receptor 4 agonists as vaccine adjuvants and monotherapeutic agents. Expert Opin. Biol. Ther. 2004, 4, 1129–1138. [Google Scholar] [CrossRef]
- Garcon, N.; Chomez, P.; Van Mechelen, M. GlaxoSmithKline Adjuvant Systems in vaccines: Concepts, achievements and perspectives. Expert Rev. Vaccines 2007, 6, 723–739. [Google Scholar] [CrossRef]
- Didierlaurent, A.M.; Laupèze, B.; Di Pasquale, A.; Hergli, N.; Collignon, C.; Garçon, N. Adjuvant system AS01: Helping to overcome the challenges of modern vaccines. Expert Rev. Vaccines 2017, 16, 55–63. [Google Scholar] [CrossRef]
- Coccia, M.; Collignon, C.; Hervé, C.; Chalon, A.; Welsby, I.; Detienne, S.; van Helden, M.J.; Dutta, S.; Genito, C.J.; Waters, N.C.; et al. Cellular and molecular synergy in AS01-adjuvanted vaccines results in an early IFNγ response promoting vaccine immunogenicity. NPJ Vaccines 2017, 2, 25. [Google Scholar] [CrossRef]
- Garçon, N.; Di Pasquale, A. From discovery to licensure, the Adjuvant System story. Hum. Vaccines Immunother. 2017, 13, 19–33. [Google Scholar] [CrossRef]
- Garçon, N.; Vaughn, D.W.; Didierlaurent, A.M. Development and evaluation of AS03, an Adjuvant System containing α-tocopherol and squalene in an oil-in-water emulsion. Expert Rev. Vaccines 2012, 11, 349–366. [Google Scholar] [CrossRef] [PubMed]
- O’Hagan, D.T.; Ott, G.S.; Nest, G.V.; Rappuoli, R.; Giudice, G.D. The history of MF59((R)) adjuvant: A phoenix that arose from the ashes. Expert Rev. Vaccines 2013, 12, 13–30. [Google Scholar] [CrossRef] [PubMed]
- O’Hagan, D.T. MF59 is a safe and potent vaccine adjuvant that enhances protection against influenza virus infection. Expert Rev. Vaccines 2007, 6, 699–710. [Google Scholar] [CrossRef] [PubMed]
- Schultze, V.; D’Agosto, V.; Wack, A.; Novicki, D.; Zorn, J.; Hennig, R. Safety of MF59™ adjuvant. Vaccine 2008, 26, 3209–3222. [Google Scholar] [CrossRef] [PubMed]
- Chappell, K.J.; Watterson, D.; Young, P.R. Chimeric Molecules and Uses Thereof 2018.
- Osterholm, M.T.; Kelley, N.S.; Sommer, A.; Belongia, E.A. Efficacy and effectiveness of influenza vaccines: A systematic review and meta-analysis. Lancet. Infect. Dis. 2012, 12, 36–44. [Google Scholar] [CrossRef]
- Mameli, C.; D’auria, E.; Erba, P.; Nannini, P.; Zuccotti, G.V. Influenza vaccine response: Future perspectives. Expert Opin. Biol. Ther. 2018, 18, 1–5. [Google Scholar] [CrossRef]
- Kumar, A.; Meldgaard, T.S.; Bertholet, S. Novel Platforms for the Development of a Universal Influenza Vaccine. Front. Immunol. 2018, 9, 600. [Google Scholar] [CrossRef]
- Gross, P.A.; Ennis, F.A.; Gaerlan, P.F.; Denson, L.J.; Denning, C.R.; Schiffman, D. A controlled double-blind comparison of reactogenicity, immunogenicity, and protective efficacy of whole-virus and split-product influenza vaccines in children. J. Infect. Dis. 1977, 136, 623–632. [Google Scholar] [CrossRef]
- Wong, S.-S.; Webby, R.J. Traditional and new influenza vaccines. Clin. Microbiol. Rev. 2013, 26, 476–492. [Google Scholar] [CrossRef]
- Acosta, P.L.; Caballero, M.T.; Polack, F.P. Brief History and Characterization of Enhanced Respiratory Syncytial Virus Disease. Clin. Vaccine Immunol. 2016, 23, 189. [Google Scholar] [CrossRef]
- Murphy, B.R.; Walsh, E.E. Formalin-inactivated respiratory syncytial virus vaccine induces antibodies to the fusion glycoprotein that are deficient in fusion-inhibiting activity. J. Clin. Microbiol. 1988, 26, 1595–1597. [Google Scholar] [CrossRef] [PubMed]
- ClinicalTrials.gov. A Study to Test GlaxoSmithKline’s (GSK) Respiratory Syncytial Virus RSV Candidate Vaccine’s Safety and Immune Response in Japanese Older Adults. Available online: https://clinicaltrials.gov/ct2/show/NCT04090658 (accessed on 15 December 2020).
- ClinicalTrials.gov. Study of Safety, Reactogenicity and Immunogenicity of GlaxoSmithKline’s (GSK)Respiratory Syncytial Virus (RSV)Maternal Unadjuvanted Vaccine in Healthy Pregnant Women (Aged 18 to 40 Years) and Their Infants. Available online: https://clinicaltrials.gov/ct2/show/NCT04126213 (accessed on 15 December 2020).
- ClinicalTrials.gov. Dose, Safety, Tolerability and Immunogenicity of a Stabilized Prefusion RSV F Subunit Protein Vaccine, VRC-RSVRGP084-00-VP (DS-Cav1), Alone or With Alum Adjuvant, in Healthy Adults. Available online: https://clinicaltrials.gov/ct2/show/NCT03049488 (accessed on 15 December 2020).
- ClinicalTrials.gov. A Study of an Ad26.RSV.preF-based Regimen in the Prevention of Reverse Transcriptase Polymerase Chain Reaction (RT-PCR)-Confirmed Respiratory Syncytial Virus (RSV)-Mediated Lower Respiratory Tract Disease in Adults Aged 65 Years and Older. Available online: https://clinicaltrials.gov/ct2/show/NCT03982199 (accessed on 15 December 2020).
- ClinicalTrials.gov. Evaluating the Infectivity, Safety, and Immunogenicity of a Recombinant Live-Attenuated Respiratory Syncytial Virus Vaccine in RSV-Seronegative Infants and Children 6 to 24 Months of Age. Available online: https://clinicaltrials.gov/ct2/show/NCT02952339 (accessed on 15 December 2020).
- Wijesundara, D.K.; Avumegah, M.S.; Lackenby, J.; Modhiran, N.; Isaacs, A.; Young, P.R.; Watterson, D.; Chappell, K.J. Rapid Response Subunit Vaccine Design in the Absence of Structural Information. Front. Immunol. 2020, 11, 592370. [Google Scholar] [CrossRef] [PubMed]
- Watterson, D.; Wijesundara, D.; Modhiran, N.; Mordant, F.; Li, Z.; Avumegah, M.; McMillan, C.; Lackenby, J.; Guilfoyle, K.; van Amerongen, G.; et al. Molecular clamp stabilised Spike protein for protection against SARS-CoV-2. Res. Sq. Prepr. 2020. [Google Scholar] [CrossRef]
- Corti, D.; Voss, J.; Gamblin, S.J.; Codoni, G.; Macagno, A.; Jarrossay, D.; Vachieri, S.G.; Pinna, D.; Minola, A.; Vanzetta, F.; et al. A neutralizing antibody selected from plasma cells that binds to group 1 and group 2 influenza A hemagglutinins. Science 2011, 333, 850–856. [Google Scholar] [CrossRef] [PubMed]
- Magro, M.; Mas, V.; Chappell, K.; Vázquez, M.; Cano, O.; Luque, D.; Terrón, M.C.; Melero, J.A.; Palomo, C. Neutralizing antibodies against the preactive form of respiratory syncytial virus fusion protein offer unique possibilities for clinical intervention. Proc. Natl. Acad. Sci. USA 2012, 109, 3089–3094. [Google Scholar] [CrossRef] [PubMed]
- Ranasinghe, C.; Turner, S.J.; McArthur, C.; Sutherland, D.B.; Kim, J.H.; Doherty, P.C.; Ramshaw, I.A. Mucosal HIV-1 pox virus prime-boost immunization induces high-avidity CD8+ T cells with regime-dependent cytokine/granzyme B profiles. J. Immunol. 2007, 178, 2370–2379. [Google Scholar] [CrossRef]
- DiLillo, D.J.; Tan, G.S.; Palese, P.; Ravetch, J.V. Broadly neutralizing hemagglutinin stalk-specific antibodies require FcγR interactions for protection against influenza virus in vivo. Nat. Med. 2014, 20, 143–151. [Google Scholar] [CrossRef]
- Jegaskanda, S.; Luke, C.; Hickman, H.D.; Sangster, M.Y.; Wieland-Alter, W.F.; McBride, J.M.; Yewdell, J.W.; Wright, P.F.; Treanor, J.; Rosenberger, C.M.; et al. Generation and Protective Ability of Influenza Virus-Specific Antibody-Dependent Cellular Cytotoxicity in Humans Elicited by Vaccination, Natural Infection, and Experimental Challenge. J. Infect. Dis. 2016, 214, 945–952. [Google Scholar] [CrossRef]
- Jegerlehner, A.; Schmitz, N.; Storni, T.; Bachmann, M.F. Influenza A vaccine based on the extracellular domain of M2: Weak protection mediated via antibody-dependent NK cell activity. J. Immunol. 2004, 172, 5598–5605. [Google Scholar] [CrossRef]
- Van Erp, E.A.; Luytjes, W.; Ferwerda, G.; van Kasteren, P.B. Fc-Mediated Antibody Effector Functions During Respiratory Syncytial Virus Infection and Disease. Front. Immunol. 2019, 178, 2370–2379. [Google Scholar] [CrossRef]
- Wagner, D.K.; Muelenaer, P.; Henderson, F.W.; Snyder, M.H.; Reimer, C.B.; Walsh, E.E.; Anderson, L.J.; Nelson, D.L.; Murphy, B.R. Serum immunoglobulin G antibody subclass response to respiratory syncytial virus F and G glycoproteins after first, second, and third infections. J. Clin. Microbiol. 1989, 27, 589–592. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, A.; McCarthy, K.R.; Kuraoka, M.; Schmidt, A.G.; Adachi, Y.; Onodera, T.; Tonouchi, K.; Caradonna, T.M.; Bajic, G.; Song, S.; et al. Antibodies to a Conserved Influenza Head Interface Epitope Protect by an IgG Subtype-Dependent Mechanism. Cell 2019, 177, 1124–1135.e1116. [Google Scholar] [CrossRef] [PubMed]
- Greenberg, S.B.; Criswell, B.S.; Six, H.R.; Couch, R.B. Lymphocyte cytotoxicity to influenza virus-infected cells: Response to vaccination and virus infection. Infect. Immun. 1978, 20, 640–645. [Google Scholar] [CrossRef] [PubMed]
- Greenberg, S.B.; Six, H.R.; Drake, S.; Couch, R.B. Cell cytotoxicity due to specific influenza antibody production in vitro after recent influenza antigen stimulation. Proc. Natl. Acad. Sci. USA 1979, 76, 4622–4626. [Google Scholar] [CrossRef] [PubMed]
- Meguro, H.; Kervina, M.; Wright, P.F. Antibody-Dependent Cell-Mediated Cytotoxicity against Cells Infected with Respir-atory Syncytial Virus: Characterization of in Vitro and In Vivo Properties. J. Immunol. 1979, 122, 2521. [Google Scholar] [PubMed]
- Scott, R.; de Landazuri, M.O.; Gardner, P.S.; Owen, J.J. Human antibody-dependent cell-mediated cytotoxicity against target cells infected with respiratory syncytial virus. Clin. Exp. Immunol. 1977, 28, 19–26. [Google Scholar]
- Vella, S.; Rocchi, G.; Resta, S.; Marcelli, M.; De Felici, A. Antibody reactive in antibody-dependent cell-mediated cytotoxicity following influenza virus vaccination. J. Med. Virol. 1980, 6, 203–211. [Google Scholar] [CrossRef]
- Von Holle, T.A.; Moody, M.A. Influenza and Antibody-Dependent Cellular Cytotoxicity. Front. Immunol. 2019, 10, 1457. [Google Scholar] [CrossRef]
- Collins, A.M. IgG subclass co-expression brings harmony to the quartet model of murine IgG function. Immunol. Cell Biol. 2016, 94, 949–954. [Google Scholar] [CrossRef]
- Khanna, M.; Jackson, R.J.; Alcantara, S.; Amarasena, T.H.; Li, Z.; Kelleher, A.D.; Kent, S.J.; Ranasinghe, C. Mucosal and systemic SIV-specific cytotoxic CD4+ T cell hierarchy in protection following intranasal/intramuscular recombinant pox-viral vaccination of pigtail macaques. Sci. Rep. 2019, 9, 5661. [Google Scholar] [CrossRef]
- Panagioti, E.; Klenerman, P.; Lee, L.N.; van der Burg, S.H.; Arens, R. Features of Effective T Cell-Inducing Vaccines against Chronic Viral Infections. Front. Immunol. 2018, 9, 276. [Google Scholar] [CrossRef] [PubMed]
- Stark, G.R.; Kerr, I.M.; Williams, B.R.; Silverman, R.H.; Schreiber, R.D. How cells respond to interferons. Annu. Rev. Biochem. 1998, 67, 227–264. [Google Scholar] [CrossRef] [PubMed]
- Hem, S.L.; Harm, H. Aluminum-Containing Adjuvants: Properties, Formulation, and Use. In Vaccine Adjuvants and Delivery Systems; Wiley: Hoboken, NJ, USA, 2007; pp. 81–114. [Google Scholar] [CrossRef]
- Al-Shakhshir, R.H.; Regnier, F.E.; White, J.L.; Hem, S.L. Contribution of electrostatic and hydrophobic interactions to the adsorption of proteins by aluminium-containing adjuvants. Vaccine 1995, 13, 41–44. [Google Scholar] [CrossRef]
- Jully, V.; Mathot, F.; Moniotte, N.; Préat, V.; Lemoine, D. Mechanisms of Antigen Adsorption Onto an Aluminum-Hydroxide Adjuvant Evaluated by High-Throughput Screening. J. Pharm. Sci. 2016, 105, 1829–1836. [Google Scholar] [CrossRef]
- Chang, C.B.; Knobler, C.M.; Gelbart, W.M.; Mason, T.G. Curvature dependence of viral protein structures on encapsidated nanoemulsion droplets. ACS Nano. 2008, 2, 281–286. [Google Scholar] [CrossRef]
- Chesko, J.; Kazzaz, J.; Ugozzoli, M.; O’Hagan, D.T.; Singh, M. An investigation of the factors controlling the adsorption of protein antigens to anionic PLG microparticles. J. Pharm. Sci. 2005, 94, 2510–2519. [Google Scholar] [CrossRef]
- Fox, C.B.; Kramer, R.M.; Barnes, V.L.; Dowling, Q.M.; Vedvick, T.S. Working together: Interactions between vaccine antigens and adjuvants. Ther. Adv. Vaccines 2013, 1, 7–20. [Google Scholar] [CrossRef]
- Watson, D.S.; Endsley, A.N.; Huang, L. Design considerations for liposomal vaccines: Influence of formulation parameters on antibody and cell-mediated immune responses to liposome associated antigens. Vaccine 2012, 30, 2256–2272. [Google Scholar] [CrossRef]
- Watson, D.S.; Platt, V.M.; Cao, L.; Venditto, V.J.; Szoka, F.C., Jr. Antibody response to polyhistidine-tagged peptide and protein antigens attached to liposomes via lipid-linked nitrilotriacetic acid in mice. Clin. Vaccine Immunol. 2011, 18, 289–297. [Google Scholar] [CrossRef]
- Hansen, B.; Malyala, P.; Singh, M.; Sun, Y.; Srivastava, I.; Hogenesch, H.; Hem, S.L. Effect of the strength of adsorption of HIV 1 SF162dV2gp140 to aluminum-containing adjuvants on the immune response. J. Pharm. Sci. 2011, 100, 3245–3250. [Google Scholar] [CrossRef]
- Ozorowski, G.; Cupo, A.; Golabek, M.; LoPiccolo, M.; Ketas, T.A.; Cavallary, M.; Cottrell, C.A.; Klasse, P.J.; Ward, A.B.; Moore, J.P. Effects of Adjuvants on HIV-1 Envelope Glycoprotein SOSIP Trimers In Vitro. J. Virol. 2018, 92, e00381-18. [Google Scholar] [CrossRef] [PubMed]
- Funchal, G.A.; Jaeger, N.; Czepielewski, R.S.; Machado, M.S.; Muraro, S.P.; Stein, R.T.; Bonorino, C.B.C.; Porto, B.N. Respiratory syncytial virus fusion protein promotes TLR-4-dependent neutrophil extracellular trap formation by human neutrophils. PLoS ONE 2015, 10, e0124082. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.-C.; Lin, S.-C.; Yu, Y.-L.; Chu, C.-L.; Wu, S.-C. Dendritic Cell Activation by Recombinant Hemagglutinin Proteins of H1N1 and H5N1 Influenza A Viruses. J. Virol. 2010, 84, 12011. [Google Scholar] [CrossRef] [PubMed]
- Modhiran, N.; Watterson, D.; Muller, D.A.; Panetta, A.K.; Sester, D.P.; Liu, L.; Hume, D.A.; Stacey, K.J.; Young, P.R. Dengue virus NS1 protein activates cells via Toll-like receptor 4 and disrupts endothelial cell monolayer integrity. Sci. Transl. Med. 2015, 7, 304ra142. [Google Scholar] [CrossRef] [PubMed]
- Rallabhandi, P.; Phillips, R.L.; Boukhvalova, M.S.; Pletneva, L.M.; Shirey, K.A.; Gioannini, T.L.; Weiss, J.P.; Chow, J.C.; Hawkins, L.D.; Vogel, S.N.; et al. Respiratory Syncytial Virus Fusion Protein-Induced Toll-Like Receptor 4 (TLR4) Signaling Is Inhibited by the TLR4 Antagonists Rhodobacter sphaeroides Lipopolysaccharide and Eritoran (E5564) and Requires Direct Interaction with MD-2. mBio 2012, 3, e00218-12. [Google Scholar] [CrossRef] [PubMed]
- Bieback, K.; Lien, E.; Klagge, I.M.; Avota, E.; Schneider-Schaulies, J.; Duprex, W.P.; Wagner, H.; Kirschning, C.J.; Ter Meulen, V.; Schneider-Schaulies, S. Hemagglutinin protein of wild-type measles virus activates toll-like receptor 2 signaling. J. Virol. 2002, 76, 8729–8736. [Google Scholar] [CrossRef]
- Hiatt, A.; Bohorova, N.; Bohorov, O.; Goodman, C.; Kim, D.; Pauly, M.H.; Velasco, J.; Whaley, K.J.; Piedra, P.A.; Gilbert, B.E.; et al. Glycan variants of a respiratory syncytial virus antibody with enhanced effector function and in vivo efficacy. Proc. Natl. Acad. Sci. USA 2014, 111, 5992. [Google Scholar] [CrossRef]
- Lu, Y.; Welsh, J.P.; Swartz, J.R. Production and stabilization of the trimeric influenza hemagglutinin stem domain for potentially broadly protective influenza vaccines. Proc. Natl. Acad. Sci. USA 2014, 111, 125. [Google Scholar] [CrossRef]
- Valkenburg, S.A.; Mallajosyula, V.V.A.; Li, O.T.W.; Chin, A.W.H.; Carnell, G.; Temperton, N.; Varadarajan, R.; Poon, L.L.M. Stalking influenza by vaccination with pre-fusion headless HA mini-stem. Sci. Rep. 2016, 6, 22666. [Google Scholar] [CrossRef]
- DiLillo, D.J.; Palese, P.; Wilson, P.C.; Ravetch, J.V. Broadly neutralizing anti-influenza antibodies require Fc receptor engagement for in vivo protection. J. Clin. Investig. 2016, 126, 605–610. [Google Scholar] [CrossRef]
- Sedova, E.S.; Scherbinin, D.N.; Lysenko, A.A.; Alekseeva, S.V.; Artemova, E.A.; Shmarov, M.M. Non-neutralizing Antibodies Directed at Conservative Influenza Antigens. Acta Nat. 2019, 11, 22–32. [Google Scholar] [CrossRef] [PubMed]
- Zhang, N.; Channappanavar, R.; Ma, C.; Wang, L.; Tang, J.; Garron, T.; Tao, X.; Tasneem, S.; Lu, L.; Tseng, C.-T.K.; et al. Identification of an ideal adjuvant for receptor-binding domain-based subunit vaccines against Middle East respiratory syndrome coronavirus. Cell Mol. Immunol. 2016, 13, 180–190. [Google Scholar] [CrossRef] [PubMed]
- Park, S.A.; Song, E.S.; Cho, Y.J.; Ahn, B.Y.; Ha, S.-H.; Seong, B.L.; Lee, K.H.; Lee, N.G. Immune Responses of Mice to Influenza Subunit Vaccine in Combination with CIA07 as an Adjuvant. Microbiol. Immunol. 2007, 51, 1099–1107. [Google Scholar] [CrossRef] [PubMed]
- Williams, M.A.; Holmes, B.J.; Sun, J.C.; Bevan, M.J. Developing and maintaining protective CD8+ memory T cells. Immunol. Rev. 2006, 211, 146–153. [Google Scholar] [CrossRef] [PubMed]
- Seder, R.A.; Darrah, P.A.; Roederer, M. T-cell quality in memory and protection: Implications for vaccine design. Nat. Rev. Immunol. 2008, 8, 247–258. [Google Scholar] [CrossRef]
- Zimmerli, S.C.; Harari, A.; Cellerai, C.; Vallelian, F.; Bart, P.-A.; Pantaleo, G. HIV-1-specific IFN-γ/IL-2-secreting CD8 T cells support CD4-independent proliferation of HIV-1-specific CD8 T cells. Proc. Natl. Acad. Sci. USA 2005, 102, 7239. [Google Scholar] [CrossRef]
- Lichterfeld, M.; Yu, X.G.; Waring, M.T.; Mui, S.K.; Johnston, M.N.; Cohen, D.; Addo, M.M.; Zaunders, J.; Alter, G.; Pae, E.; et al. HIV-1–specific cytotoxicity is preferentially mediated by a subset of CD8+ T cells producing both interferon-γ and tumor necrosis factor–α. Blood 2004, 104, 487–494. [Google Scholar] [CrossRef]
- Teijaro, J.R.; Turner, D.; Pham, Q.; Wherry, E.J.; Lefrançois, L.; Farber, D.L. Cutting Edge: Tissue-Retentive Lung Memory CD4 T Cells Mediate Optimal Protection to Respiratory Virus Infection. J. Immunol. 2011, 187, 5510. [Google Scholar] [CrossRef]
- Van de Sandt, C.E.; Kreijtz, J.H.C.M.; de Mutsert, G.; Geelhoed-Mieras, M.M.; Hillaire, M.L.B.; Vogelzang-van Trierum, S.E.; Osterhaus, A.D.M.E.; Fouchier, R.A.M.; Rimmelzwaan, G.F. Human Cytotoxic T Lymphocytes Directed to Seasonal Influenza A Viruses Cross-React with the Newly Emerging H7N9 Virus. J. Virol. 2014, 88, 1684. [Google Scholar] [CrossRef]
- Zhou, X.; Hopkins, J.W.; Wang, C.; Brahmakshatriya, V.; Swain, S.L.; Kuchel, G.A.; Haynes, L.; McElhaney, J.E. IL-2 and IL-6 cooperate to enhance the generation of influenza-specific CD8 T cells responding to live influenza virus in aged mice and humans. Oncotarget 2016, 7, 39171–39183. [Google Scholar] [CrossRef][Green Version]
- Strutt, T.M.; Dhume, K.; Finn, C.M.; Hwang, J.H.; Castonguay, C.; Swain, S.L.; McKinstry, K.K. IL-15 supports the generation of protective lung-resident memory CD4 T cells. Mucosal Immunol. 2018, 11, 668–680. [Google Scholar] [CrossRef] [PubMed]
- Strutt, T.M.; McKinstry, K.K.; Kuang, Y.; Bradley, L.M.; Swain, S.L. Memory CD4 T-cell–mediated protection depends on secondary effectors that are distinct from and superior to primary effectors. Proc. Natl. Acad. Sci. USA 2012, 109, E2551. [Google Scholar] [CrossRef] [PubMed]
- Quiñones-Parra, S.; Grant, E.; Loh, L.; Nguyen, T.H.O.; Campbell, K.-A.; Tong, S.Y.C.; Miller, A.; Doherty, P.C.; Vijaykrishna, D.; Rossjohn, J.; et al. Preexisting CD8 T-cell immunity to the H7N9 influenza A virus varies across ethnicities. Proc. Natl. Acad. Sci. USA 2014, 111, 1049. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Wan, Y.; Qiu, C.; Quiñones-Parra, S.; Zhu, Z.; Loh, L.; Tian, D.; Ren, Y.; Hu, Y.; Zhang, X.; et al. Recovery from severe H7N9 disease is associated with diverse response mechanisms dominated by CD8+ T cells. Nat. Commun. 2015, 6, 6833. [Google Scholar] [CrossRef]
- Hemann, E.A.; Kang, S.-M.; Legge, K.L. Protective CD8 T Cell–Mediated Immunity against Influenza A Virus Infection following Influenza Virus–like Particle Vaccination. J. Immunol. 2013, 191, 2486. [Google Scholar] [CrossRef]
- Li, Z.; Khanna, M.; Grimley, S.L.; Ellenberg, P.; Gonelli, C.A.; Lee, W.S.; Amarasena, T.H.; Kelleher, A.D.; Purcell, D.F.J.; Kent, S.J.; et al. Mucosal IL-4R antagonist HIV vaccination with SOSIP-gp140 booster can induce high-quality cytotoxic CD4+/CD8+ T cells and humoral responses in macaques. Sci. Rep. 2020, 10, 22077. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Isaacs, A.; Li, Z.; Cheung, S.T.M.; Wijesundara, D.K.; McMillan, C.L.D.; Modhiran, N.; Young, P.R.; Ranasinghe, C.; Watterson, D.; Chappell, K.J. Adjuvant Selection for Influenza and RSV Prefusion Subunit Vaccines. Vaccines 2021, 9, 71. https://doi.org/10.3390/vaccines9020071
Isaacs A, Li Z, Cheung STM, Wijesundara DK, McMillan CLD, Modhiran N, Young PR, Ranasinghe C, Watterson D, Chappell KJ. Adjuvant Selection for Influenza and RSV Prefusion Subunit Vaccines. Vaccines. 2021; 9(2):71. https://doi.org/10.3390/vaccines9020071
Chicago/Turabian StyleIsaacs, Ariel, Zheyi Li, Stacey T. M. Cheung, Danushka K. Wijesundara, Christopher L. D. McMillan, Naphak Modhiran, Paul R. Young, Charani Ranasinghe, Daniel Watterson, and Keith J. Chappell. 2021. "Adjuvant Selection for Influenza and RSV Prefusion Subunit Vaccines" Vaccines 9, no. 2: 71. https://doi.org/10.3390/vaccines9020071
APA StyleIsaacs, A., Li, Z., Cheung, S. T. M., Wijesundara, D. K., McMillan, C. L. D., Modhiran, N., Young, P. R., Ranasinghe, C., Watterson, D., & Chappell, K. J. (2021). Adjuvant Selection for Influenza and RSV Prefusion Subunit Vaccines. Vaccines, 9(2), 71. https://doi.org/10.3390/vaccines9020071






