Maternal Autogenous Inactivated Virus Vaccination Boosts Immunity to PRRSV in Piglets
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design, Animals, and Sample Processing
2.2. Virus Propagation, Titration, and Evaluation
2.3. Viremia
2.4. Neutralizing Antibody Validation and Determination of Positive Samples
2.5. Necropsy Procedures, Scoring, and Tissue Cell Iisolations
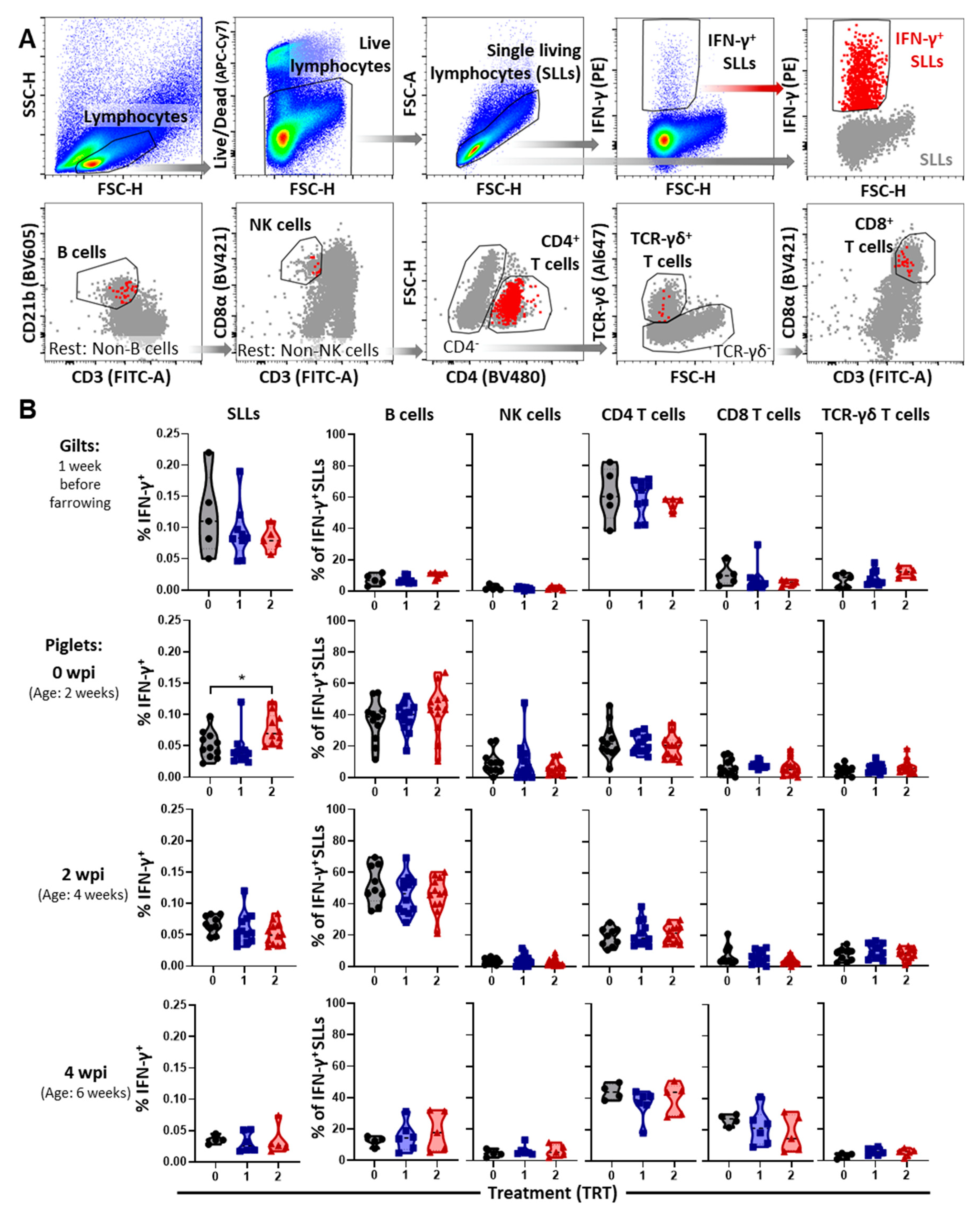
2.6. Peripheral Blood Isolation, Viral Stimulation, and IFN-γ FCM Staining/Analysis
2.7. Statistical Analysis
3. Results
3.1. Study Design and Vaccine Efficacy—Clinical Signs, Viremia, Weight Gain, and Lung Pathology
3.2. Induction of Maternal Neutralizing Antibodies and Their Transfer to Piglets
3.3. The Systemic Cell-Mediated Immune Response to PRRSV
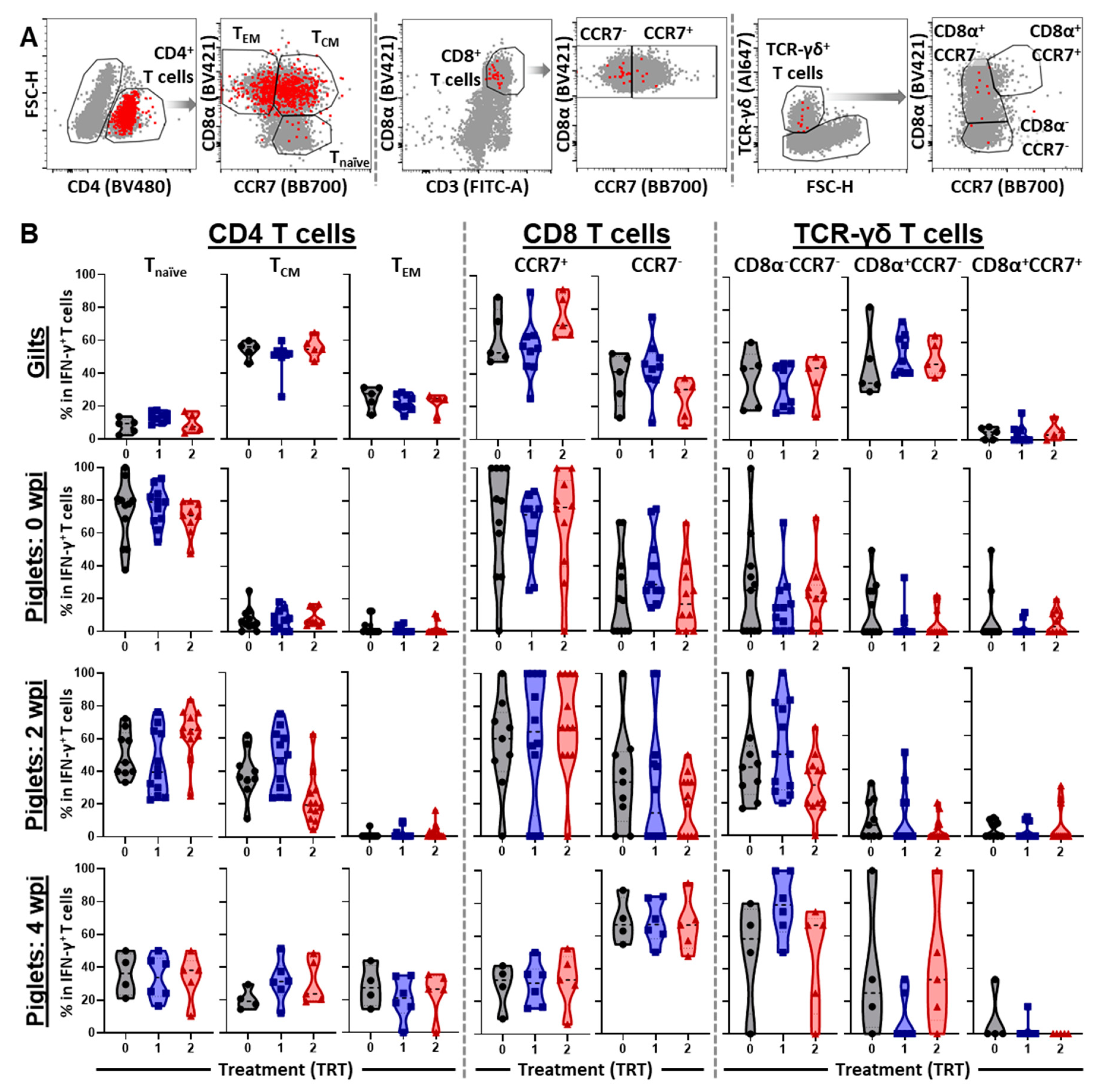
3.4. The Differentiation and Homing of IFN-γ Producing T-Cell Subsets in Blood
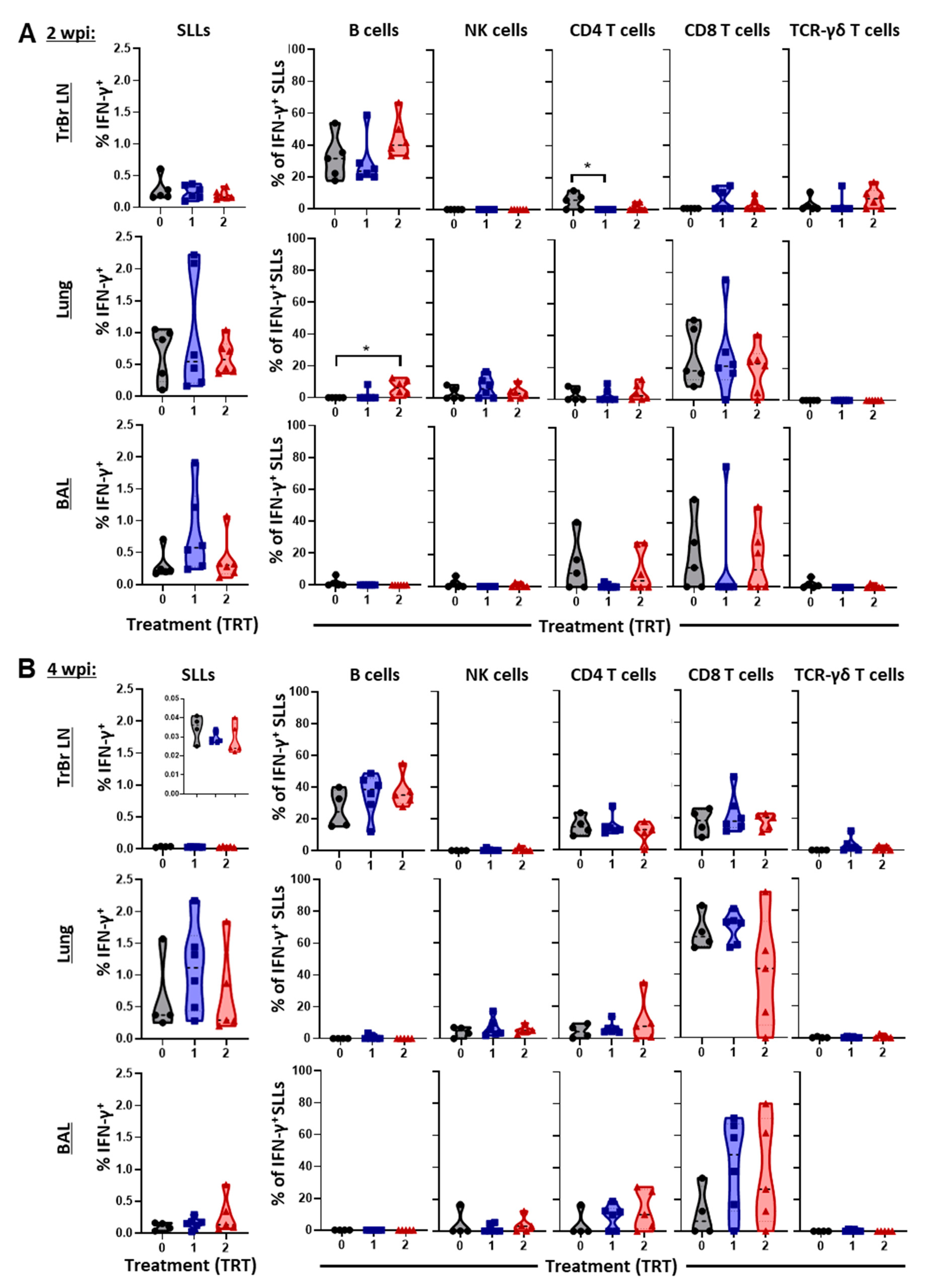
3.5. The Local Cell-Mediated Immune Response to PRRSV—BAL, Lung, and Tracheobronchial Lymph Nodes
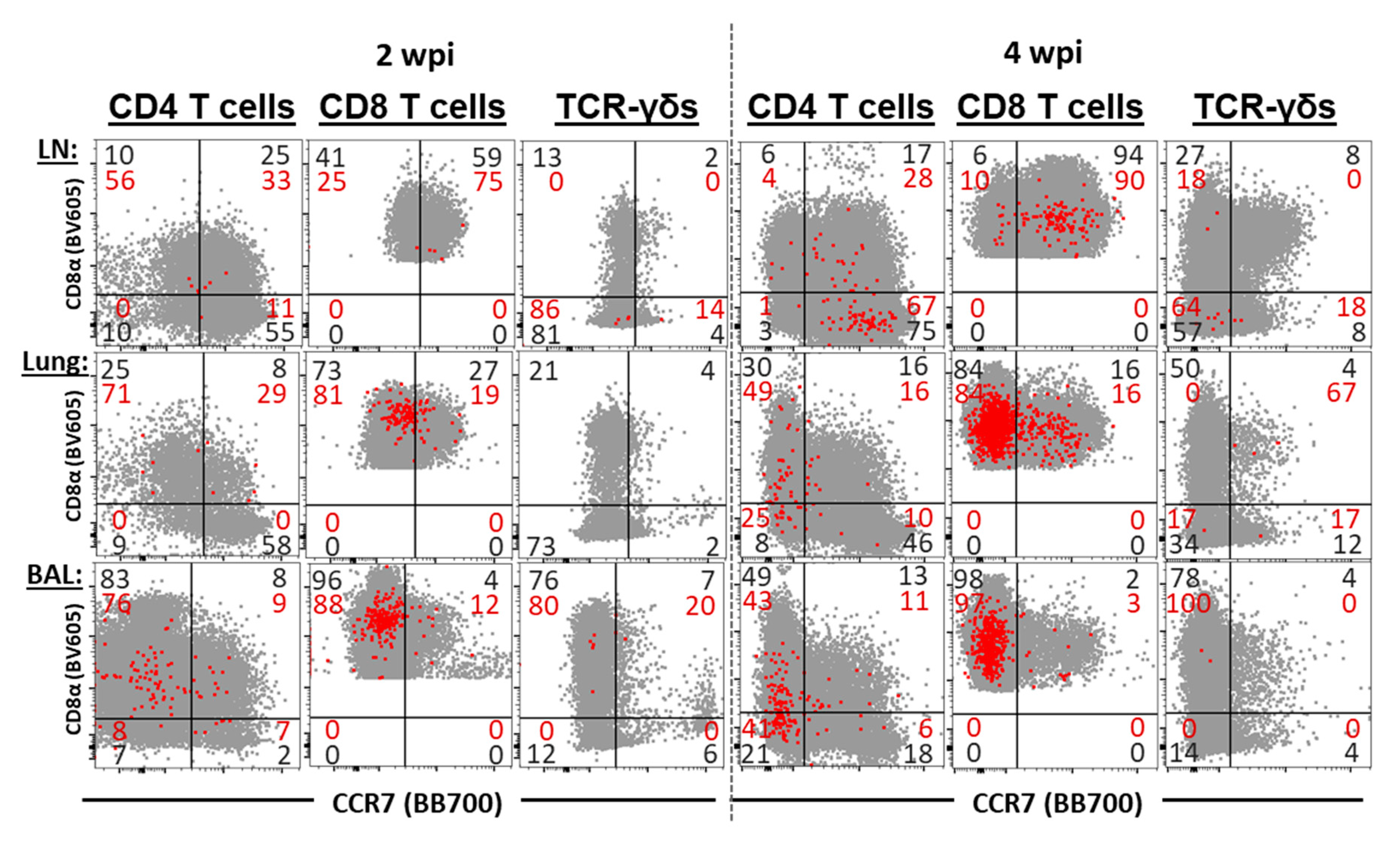
3.6. The Differentiation and Homing of IFN-γ Producing T-Cell Subsets in BAL, Lung, and Tracheobronchial Lymph Nodes
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Merriam-Webster Inc. The Merriam-Webster Dictionary; Merriam-Webster, Incorporated: Springfield, MA, USA, 2019. [Google Scholar]
- Butler, J.E.; Wertz, N.; Sinkora, M. Antibody Repertoire Development in Swine. Annu. Rev. Anim. Biosci. 2017, 5, 255–279. [Google Scholar] [CrossRef] [PubMed]
- Hurley, W.L.; Theil, P.K. Perspectives on Immunoglobulins in Colostrum and Milk. Nutrients 2011, 3, 442–474. [Google Scholar] [CrossRef] [PubMed]
- Jennewein, M.F.; Abu-Raya, B.; Jiang, Y.; Alter, G.; Marchant, A. Transfer of maternal immunity and programming of the newborn immune system. Semin. Immunopathol. 2017, 39, 605–613. [Google Scholar] [CrossRef] [PubMed]
- Poonsuk, K.; Zimmerman, J. Historical and contemporary aspects of maternal immunity in swine. Anim. Health Res. Rev. 2017, 19, 31–45. [Google Scholar] [CrossRef]
- Declerck, I.; Dewulf, J.; Sarrazin, S.; Maes, D. Long-term effects of colostrum intake in piglet mortality and performance1. J. Anim. Sci. 2016, 94, 1633–1643. [Google Scholar] [CrossRef]
- Ogawa, S.; Tsukahara, T.; Imaoka, T.; Nakanishi, N.; Ushida, K.; Inoue, R. The effect of colostrum ingestion during the first 24 hours of life on early postnatal development of piglet immune systems. Anim. Sci. J. 2016, 87, 1511–1515. [Google Scholar] [CrossRef]
- Bandrick, M.; Ariza-Nieto, C.; Baidoo, S.K.; Molitor, T.W. Colostral antibody-mediated and cell-mediated immunity contributes to innate and antigen-specific immunity in piglets. Dev. Comp. Immunol. 2014, 43, 114–120. [Google Scholar] [CrossRef]
- Gerner, W.; Käser, T.; Saalmüller, A. Porcine T lymphocytes and NK cells—An update. Dev. Comp. Immunol. 2009, 33, 310–320. [Google Scholar] [CrossRef]
- Saalmüller, A.; Reddehase, M.J.; Bühring, H.-J.; Jonjić, S.; Koszinowski, U.H. Simultaneous expression of CD4 and CD8 antigens by a substantial proportion of resting porcine T lymphocytes. Eur. J. Immunol. 1987, 17, 1297–1301. [Google Scholar] [CrossRef]
- Saalmuller, A.; Werner, T.; Fachinger, V. T-helper cells from naive to committed. Vet. Immunol. Immunopathol. 2002, 87, 137–145. [Google Scholar] [CrossRef]
- Reutner, K.; Leitner, J.; Müllebner, A.; Ladinig, A.; Essler, S.E.; Duvigneau, J.C.; Ritzmann, M.; Steinberger, P.; Saalmuller, A.; Gerner, W. CD27 expression discriminates porcine T helper cells with functionally distinct properties. Vet. Res. 2013, 44, 18. [Google Scholar] [CrossRef]
- Kick, A.R.; Amaral, A.F.; Cortes, L.M.; Fogle, J.E.; Crisci, E.; Almond, G.W.; Käser, T. The T-Cell Response to Type 2 Porcine Reproductive and Respiratory Syndrome Virus (PRRSV). Viruses 2019, 11, 796. [Google Scholar] [CrossRef]
- Holtkamp, D.J.; Kliebenstein, J.B.; Neumann, E.J.; Zimmerman, J.J.; Rotto, H.; Yoder, T.K.; Wang, C.; Yeske, P.; Mowrer, C.; Haley, C. Assessment of the economic impact of porcine reproductive and respiratory syndrome virus on United States pork producers. J. Swine Health Prod. 2013, 21, 72–84. [Google Scholar]
- VanderWaal, K.; Deen, J. Global trends in infectious diseases of swine. Proc. Natl. Acad. Sci. USA 2018, 115, 11495–11500. [Google Scholar] [CrossRef]
- Montaner-Tarbes, S.; Del Portillo, H.A.; Montoya, M.; Fraile, L. Key Gaps in the Knowledge of the Porcine Respiratory Reproductive Syndrome Virus (PRRSV). Front. Vet. Sci. 2019, 6, 38. [Google Scholar] [CrossRef]
- Rahe, M.C.; Murtaugh, M.P. Effector mechanisms of humoral immunity to porcine reproductive and respiratory syndrome virus. Vet. Immunol. Immunopathol. 2017, 186, 15–18. [Google Scholar] [CrossRef]
- Loving, C.L.; Osorio, F.A.; Murtaugh, M.P.; Zuckermann, F.A. Innate and adaptive immunity against Porcine Reproductive and Respiratory Syndrome Virus. Vet. Immunol. Immunopathol. 2015, 167, 1–14. [Google Scholar] [CrossRef]
- Lunney, J.K.; Fang, Y.; Ladinig, A.; Chen, N.; Li, Y.; Rowland, R.R.R.; Renukaradhya, G.J. Porcine Reproductive and Respiratory Syndrome Virus (PRRSV): Pathogenesis and Interaction with the Immune System. Annu. Rev. Anim. Biosci. 2016, 4, 129–154. [Google Scholar] [CrossRef]
- Balka, G.; Dreckmann, K.; Papp, G.; Kraft, C. Vaccination of piglets at 2 and 3 weeks of age with Ingelvac PRRSFLEX® EU provides protection against heterologous field challenge in the face of homologous maternally derived antibodies. Porc. Health Manag. 2016, 2, 24. [Google Scholar] [CrossRef]
- Kang, S.; Brown, H.M.; Hwang, S. Direct Antiviral Mechanisms of Interferon-Gamma. Immune. Netw. 2018, 18, e33. [Google Scholar] [CrossRef]
- Kraft, C.; Hennies, R.; Dreckmann, K.; Noguera, M.; Rathkjen, P.H.; Gassel, M.; Gereke, M. Evaluation of PRRSv specific, maternally derived and induced immune response in Ingelvac PRRSFLEX EU vaccinated piglets in the presence of maternally transferred immunity. PLoS ONE 2019, 14, e0223060. [Google Scholar] [CrossRef] [PubMed]
- Wu, W.-H.; Fang, Y.; Farwell, R.; Steffen-Bien, M.; Rowland, R.R.; Christopher-Hennings, J.; Nelson, E.A. A 10-kDa Structural Protein of Porcine Reproductive and Respiratory Syndrome Virus Encoded by ORF2b. Virology 2001, 287, 183–191. [Google Scholar] [CrossRef] [PubMed]
- Halbur, P.G.; Paul, P.S.; Frey, M.L.; Landgraf, J.; Eernisse, K.; Meng, X.-J.; Lum, M.A.; Andrews, J.J.; Rathje, J.A. Comparison of the Pathogenicity of Two US Porcine Reproductive and Respiratory Syndrome Virus Isolates with that of the Lelystad Virus. Vet. Pathol. 1995, 32, 648–660. [Google Scholar] [CrossRef] [PubMed]
- Bandrick, M.; Pieters, M.; Pijoan, C.; Molitor, T.W. Passive Transfer of Maternal Mycoplasma hyopneumoniae-Specific Cellular Immunity to Piglets. Clin. Vaccine Immunol. 2008, 15, 540–543. [Google Scholar] [CrossRef]
- Houben, S.; Van Reeth, K.; Pensaert, M.B. Pattern of Infection with the Porcine Reproductive and Respiratory Syndrome Virus on Swine Farms in Belgium. J. Vet. Med. Ser. B 1995, 42, 209–215. [Google Scholar] [CrossRef]
- Geldhof, M.F.; Van Breedam, W.; De Jong, E.; Rodríguez, A.L.; Karniychuk, U.; Vanhee, M.; Van Doorsselaere, J.; Maes, D.; Nauwynck, H.J. Antibody response and maternal immunity upon boosting PRRSV-immune sows with experimental farm-specific and commercial PRRSV vaccines. Vet. Microbiol. 2013, 167, 260–271. [Google Scholar] [CrossRef]
- Drigo, M.; Giacomini, E.; Lazzaro, M.; Pasotto, D.; Bilato, D.; Ruggeri, J.; Boniotti, M.B.; Alborali, G.L.; Amadori, M. Comparative evaluation of immune responses of swine in PRRS-stable and unstable herds. Vet. Immunol. Immunopathol. 2018, 200, 32–39. [Google Scholar] [CrossRef]
- Dvorak, C.M.T.; Payne, B.J.; Seate, J.L.; Murtaugh, M.P. Effect of Maternal Antibody Transfer on Antibody Dynamics and Control of Porcine Circovirus Type 2 Infection in Offspring. Viral Immunol. 2018, 31, 40–46. [Google Scholar] [CrossRef]
- Osorio, F.A.; Galeota, J.; Nelson, E.; Brodersen, B.; Doster, A.; Wills, R.; Zuckermann, F.; Laegreid, W.W. Passive Transfer of Virus-Specific Antibodies Confers Protection against Reproductive Failure Induced by a Virulent Strain of Porcine Reproductive and Respiratory Syndrome Virus and Establishes Sterilizing Immunity. Virology 2002, 302, 9–20. [Google Scholar] [CrossRef]
- Lopez, O.J.; Oliveira, M.F.; Garcia, E.A.; Kwon, B.J.; Doster, A.; Osorio, F.A. Protection against Porcine Reproductive and Respiratory Syndrome Virus (PRRSV) Infection through Passive Transfer of PRRSV-Neutralizing Antibodies Is Dose Dependent. Clin. Vaccine Immunol. 2007, 14, 269–275. [Google Scholar] [CrossRef]
- Correas, I.; Osorio, F.A.; Steffen, D.; Pattnaik, A.K.; Vu, H.L.X. Cross reactivity of immune responses to porcine reproductive and respiratory syndrome virus infection. Vaccine 2017, 35, 782–788. [Google Scholar] [CrossRef] [PubMed]
- Balasch, M.; Fort, M.; Taylor, L.P.; Díaz, I.; Mateu, E.; Calvert, J.G. Immune response development after vaccination of 1-day-old naive pigs with a Porcine Reproductive and Respiratory Syndrome 1-based modified live virus vaccine. Porc. Health Manag. 2019, 5, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Jeong, J.; Choi, K.; Kang, I.; Park, C.; Chae, C. Evaluation of a 20 year old porcine reproductive and respiratory syndrome (PRRS) modified live vaccine (Ingelvac((R)) PRRS MLV) against two recent type 2 PRRS virus isolates in South Korea. Vet. Microbiol. 2016, 192, 102–109. [Google Scholar] [CrossRef] [PubMed]
- Park, C.; Baek, J.H.; Cho, S.H.; Jeong, J.; Chae, C.; You, S.-H.; Cha, S.-H. Field porcine reproductive and respiratory syndrome viruses (PRRSV) attenuated by codon pair deoptimization (CPD) in NSP1 protected pigs from heterologous challenge. Virology 2020, 540, 172–183. [Google Scholar] [CrossRef]
- Meier, W.A.; Galeota, J.; Osorio, F.A.; Husmann, R.J.; Schnitzlein, W.M.; Zuckermann, F.A. Gradual development of the interferon-gamma response of swine to porcine reproductive and respiratory syndrome virus infection or vaccination. Virology 2003, 309, 18–31. [Google Scholar] [CrossRef]
- Yang, S.; Oh, T.; Cho, H.; Chae, C. A comparison of commercial modified-live PRRSV-1 and PRRSV-2 vaccines against a dual heterologous PRRSV-1 and PRRSV-2 challenge in late term pregnancy gilts. Comp. Immunol. Microbiol. Infect. Dis. 2020, 69, 101423. [Google Scholar] [CrossRef]
- Yang, S.; Oh, T.; Kang, I.; Park, S.-J.; Chae, C. Efficacy of concurrent vaccination with modified-live PRRSV-1 and PRRSV-2 vaccines against heterologous dual PRRSV-1 and PRRSV-2 challenge in late term pregnancy gilts. Vet. Microbiol. 2019, 239, 108497. [Google Scholar] [CrossRef]
- Jeong, J.; Kim, S.; Park, K.H.; Kang, I.; Park, S.-J.; Park, C.; Chae, C. Evaluation of the effect of a porcine reproductive and respiratory syndrome (PRRS) modified-live virus vaccine on sow reproductive performance in endemic PRRS farms. Vet. Microbiol. 2017, 208, 47–52. [Google Scholar] [CrossRef]
- Han, K.; Seo, H.W.; Park, C.; Chae, C. Vaccination of sows against type 2 Porcine Reproductive and Respiratory Syndrome Virus (PRRSV) before artificial insemination protects against type 2 PRRSV challenge but does not protect against type 1 PRRSV challenge in late gestation. Vet. Res. 2014, 45, 12. [Google Scholar] [CrossRef]
- Harris, D.P.; Goodrich, S.; Gerth, A.J.; Peng, S.L.; Lund, F.E. Regulation of IFN-gamma production by B effector 1 cells: Essential roles for T-bet and the IFN-gamma receptor. J. Immunol. 2005, 174, 6781–6790. [Google Scholar] [CrossRef]
- Bao, Y.; Liu, X.; Han, C.; Xu, S.; Xie, B.; Zhang, Q.; Gu, Y.; Hou, J.; Qian, L.; Qian, C.; et al. Identification of IFN-gamma-producing innate B cells. Cell Res. 2014, 24, 161–176. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Carreño, M.P.; López-Fuertes, L.; Revilla, C.; Ezquerra, A.; Alonso, F.; Domínguez, J. Phenotypic characterization of porcine IFN-gamma-producing lymphocytes by flow cytometry. J. Immunol. Methods 2002, 259, 171–179. [Google Scholar] [CrossRef]
- Murphy, K.; Travers, P.; Walport, M.; Janeway, C. Janeway’s Immunobiology, 7th ed.; Garland Science: New York, NY, USA, 2008. [Google Scholar]
- Sallusto, F.; Lenig, D.; Förster, R.; Lipp, M.; Lanzavecchia, A. Two subsets of memory T lymphocytes with distinct homing potentials and effector functions. Nature 1999, 401, 708–712. [Google Scholar] [CrossRef] [PubMed]
- Hamann, D.; Baars, P.A.; Rep, M.H.; Hooibrink, B.; Kerkhof-Garde, S.R.; Klein, M.R.; Van Lier, R.A. Phenotypic and Functional Separation of Memory and Effector Human CD8+ T Cells. J. Exp. Med. 1997, 186, 1407–1418. [Google Scholar] [CrossRef] [PubMed]
- Sallusto, F.; Geginat, J.; Lanzavecchia, A. CentralMemory andEffectorMemoryT CellSubsets: Function, Generation, and Maintenance. Annu. Rev. Immunol. 2004, 22, 745–763. [Google Scholar] [CrossRef] [PubMed]
- Tian, Y.; Babor, M.; Lane, J.; Schulten, V.; Patil, V.S.; Seumois, G.; Rosales, S.L.; Fu, Z.; Picarda, G.; Burel, J.; et al. Unique phenotypes and clonal expansions of human CD4 effector memory T cells re-expressing CD45RA. Nat. Commun. 2017, 8, 1–13. [Google Scholar] [CrossRef]
- Mair, K.H.; Koinig, H.; Gerner, W.; Höhne, A.; Bretthauer, J.; Kroll, J.J.; Roof, M.B.; Saalmüller, A.; Stadler, K.; Libanova, R. Carbopol improves the early cellular immune responses induced by the modified-life vaccine Ingelvac PRRS(R) MLV. Vet. Microbiol. 2015, 176, 352–357. [Google Scholar] [CrossRef]
- Park, C.; Seo, H.W.; Han, K.; Kang, I.; Chae, C. Evaluation of the efficacy of a new modified live porcine reproductive and respiratory syndrome virus (PRRSV) vaccine (Fostera PRRS) against heterologous PRRSV challenge. Vet. Microbiol. 2014, 172, 432–442. [Google Scholar] [CrossRef]



| Antigen | Clone | Isotype | Fluorochrome | Labeling Strategy | Primary Ab Source | 2nd Ab Source |
|---|---|---|---|---|---|---|
| CD3 | PPT3 | IgG1 | FITC | Directly conjugated | Southern Biotech | - |
| CD4 | 74-12-4 | IgG2b | Brilliant Violet 480 | Secondary antibody | BEI Resources | Jackson Immunoresearch |
| CD8α | 76-2-11 | IgG2a | Brilliant Violet 421 | Secondary antibody | BEI Resources | Jackson Immunoresearch |
| CD21α | BB6-11C9.6 | IgG1 | Brilliant Violet 605 | Biotin-streptavidin | Novus Bio | Biolegend |
| TCR-γδ | PGBL22A | IgG1 | Alexa Fluor 647 | Directly conjugated | Kingfisher | Invitrogen |
| CCR7 | 3D12 | rIgG2a | Brilliant Blue 700 | Directly conjugated | BD Biosciences | - |
| IFN-γ | P2G10 | IgG1 | PE | Directly conjugated | BD Biosciences | - |
| Live/Dead | - | - | Near Infra-red | - | Invitrogen | - |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kick, A.R.; Wolfe, Z.C.; Amaral, A.F.; Cortes, L.M.; Almond, G.W.; Crisci, E.; Gauger, P.C.; Pittman, J.; Käser, T. Maternal Autogenous Inactivated Virus Vaccination Boosts Immunity to PRRSV in Piglets. Vaccines 2021, 9, 106. https://doi.org/10.3390/vaccines9020106
Kick AR, Wolfe ZC, Amaral AF, Cortes LM, Almond GW, Crisci E, Gauger PC, Pittman J, Käser T. Maternal Autogenous Inactivated Virus Vaccination Boosts Immunity to PRRSV in Piglets. Vaccines. 2021; 9(2):106. https://doi.org/10.3390/vaccines9020106
Chicago/Turabian StyleKick, Andrew R., Zoe C. Wolfe, Amanda F. Amaral, Lizette M. Cortes, Glen W. Almond, Elisa Crisci, Phillip C. Gauger, Jeremy Pittman, and Tobias Käser. 2021. "Maternal Autogenous Inactivated Virus Vaccination Boosts Immunity to PRRSV in Piglets" Vaccines 9, no. 2: 106. https://doi.org/10.3390/vaccines9020106
APA StyleKick, A. R., Wolfe, Z. C., Amaral, A. F., Cortes, L. M., Almond, G. W., Crisci, E., Gauger, P. C., Pittman, J., & Käser, T. (2021). Maternal Autogenous Inactivated Virus Vaccination Boosts Immunity to PRRSV in Piglets. Vaccines, 9(2), 106. https://doi.org/10.3390/vaccines9020106










