Promising Expectations for Pneumococcal Vaccination during COVID-19
Abstract
:1. Overview of COVID-19
1.1. COVID-19 Outbreak
1.2. COVID-19 Threat Factors
2. Overview of Streptococcus pneumoniae
3. Overlapping Risk Factors for Infection by Two Respiratory Pathogens
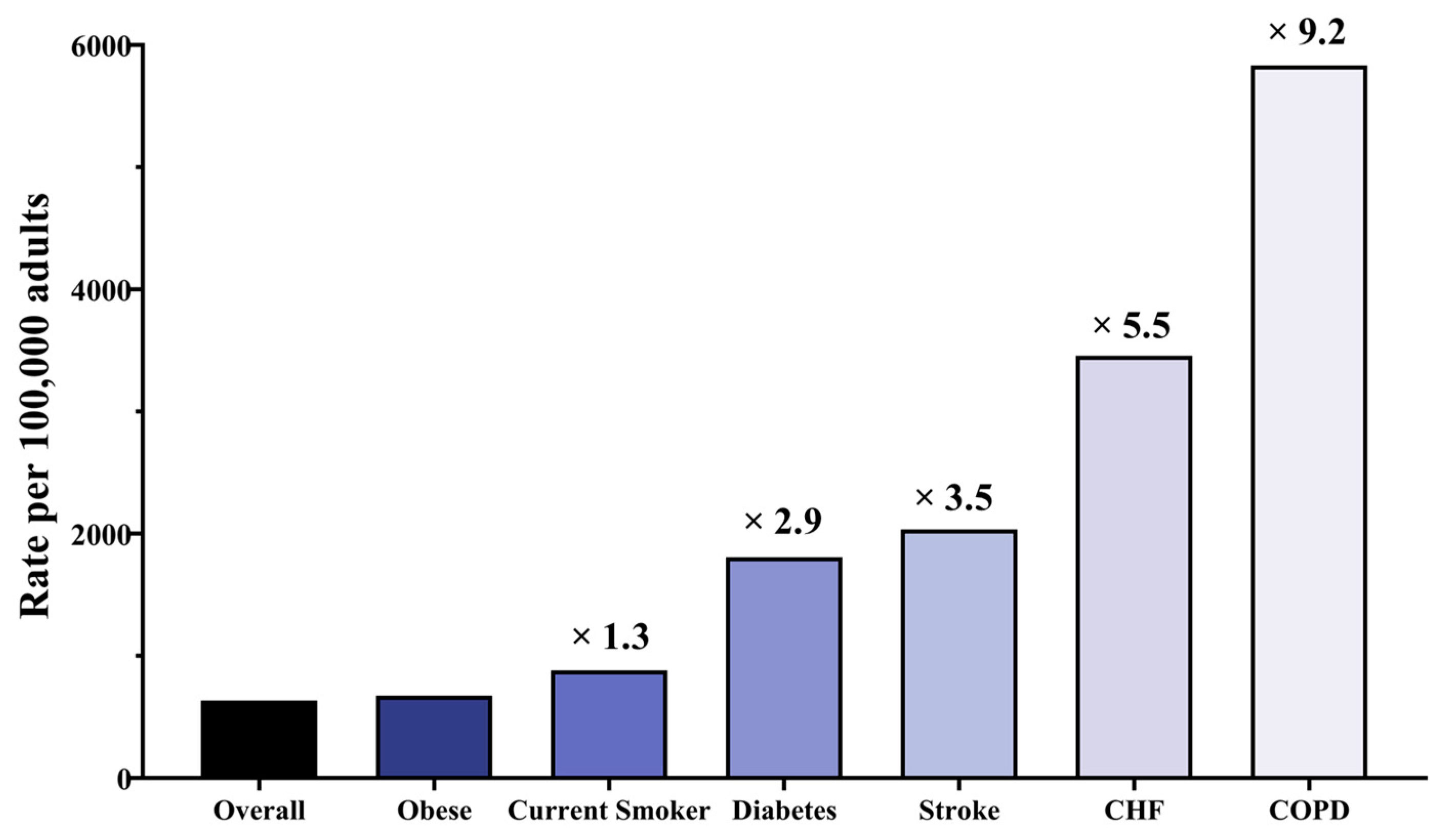
3.1. Age
3.2. Chronic Obstructive Pulmonary Disease (COPD)
3.3. Asthma
3.4. Cardiovascular Disease (CVD)
3.5. Chronic Heart Disease
3.6. Chronic Kidney Disease (CKD)
3.7. Chronic Liver Disease (CLD)
3.8. Diabetes
3.9. Cancer
3.10. Smoking
4. Combined Infection with SARS-CoV-2 and S. pneumoniae
5. Recommendations for Two Vaccine-Preventable Diseases (VPDs)
5.1. Impact of Pneumococcal Vaccines in COVID-19
5.2. Recommendations of Pneumococcal Vaccine for Reducing the Risk of COVID-19
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Williamson, E.J.; Walker, A.J.; Bhaskaran, K.; Bacon, S.; Bates, C.; Morton, C.E.; Curtis, H.J.; Mehrkar, A.; Evans, D.; Inglesby, P. Factors associated with COVID-19-related death using OpenSAFELY. Nature 2020, 584, 430–436. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. WHO Coronavirus (COVID-19) Dashboard. Available online: https://covid19.who.int/ (accessed on 15 December 2021).
- Maggi, S. Vaccination and healthy aging. Expert Rev. Vaccines 2010, 9, 3–6. [Google Scholar] [CrossRef] [PubMed]
- Sanyaolu, A.; Okorie, C.; Marinkovic, A.; Patidar, R.; Younis, K.; Desai, P.; Hosein, Z.; Padda, I.; Mangat, J.; Altaf, M. Comorbidity and its Impact on Patients with COVID-19. SN Compr. Clin. Med. 2020, 2, 1069–1076. [Google Scholar] [CrossRef] [PubMed]
- Kiani, F.Z.; Ahmadi, A. Prevalence of different comorbidities in chronic obstructive pulmonary disease among Shahrekord PERSIAN cohort study in southwest Iran. Sci. Rep. 2021, 11, 1548. [Google Scholar] [CrossRef]
- Zhou, F.; Yu, T.; Du, R.; Fan, G.; Liu, Y.; Liu, Z.; Xiang, J.; Wang, Y.; Song, B.; Gu, X.; et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: A retrospective cohort study. Lancet 2020, 395, 1054–1062. [Google Scholar] [CrossRef]
- Maddaloni, E.; Buzzetti, R. COVID-19 and diabetes mellitus: Unveiling the interaction of two pandemics. Diabetes Metab. Res. Rev. 2020, 36, e33213321. [Google Scholar] [CrossRef]
- Xu, L.; Mao, Y.; Chen, G. Risk factors for 2019 novel coronavirus disease (COVID-19) patients progressing to critical illness: A systematic review and meta-analysis. Aging 2020, 12, 12410. [Google Scholar] [CrossRef]
- Jordan, R.E.; Adab, P.; Cheng, K. COVID-19: Risk factors for severe disease and death. BMJ 2020, 368, m1198. [Google Scholar] [CrossRef] [Green Version]
- Fang, X.; Li, S.; Yu, H.; Wang, P.; Zhang, Y.; Chen, Z.; Li, Y.; Cheng, L.; Li, W.; Jia, H. Epidemiological, comorbidity factors with severity and prognosis of COVID-19: A systematic review and meta-analysis. Aging 2020, 12, 12493. [Google Scholar] [CrossRef] [PubMed]
- Sultana, J.; Mazzaglia, G.; Luxi, N.; Cancellieri, A.; Capuano, A.; Ferrajolo, C.; de Waure, C.; Ferlazzo, G.; Trifirò, G. Potential effects of vaccinations on the prevention of COVID-19: Rationale, clinical evidence, risks, and public health considerations. Expert Rev. Vaccines 2020, 19, 919–936. [Google Scholar] [CrossRef]
- Joseph, C.; Togawa, Y.; Shindo, N. Bacterial and viral infections associated with influenza. Influenza Other Respir. Viruses 2013, 7, 105–113. [Google Scholar] [CrossRef] [Green Version]
- Morris, D.E.; Cleary, D.W.; Clarke, S.C. Secondary bacterial infections associated with influenza pandemics. Front. Microbiol. 2017, 8, 1041. [Google Scholar] [CrossRef] [Green Version]
- Wang, L.; Fu, J.; Liang, Z.; Chen, J. Prevalence and serotype distribution of nasopharyngeal carriage of Streptococcus pneumoniae in China: A meta-analysis. BMC Infect. Dis. 2017, 17, 765. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Aykac, K.; Ozsurekci, Y.; Cura Yayla, B.C.; Evren, K.; Lacinel Gurlevik, S.; Oygar, P.D.; Yucel, M.; Karakoc, A.E.; Alp, A.; Cengiz, A.B. Pneumococcal carriage in children with COVID-19. Hum. Vaccines Immunother. 2021, 17, 1628–1634. [Google Scholar] [CrossRef]
- Rodriguez-Nava, G.; Yanez-Bello, M.A.; Trelles-Garcia, D.P.; Chung, C.W.; Egoryan, G.; Friedman, H.J. A retrospective study of coinfection of SARS-CoV-2 and Streptococcus pneumoniae in 11 hospitalized patients with severe COVID-19 pneumonia at a single center. Med. Sci. Monit. Int. Med. J. Exp. Clin. Res. 2020, 26, e928754-1. [Google Scholar] [CrossRef] [PubMed]
- Lozano, R.; Naghavi, M.; Foreman, K.; Lim, S.; Shibuya, K.; Aboyans, V.; Abraham, J.; Adair, T.; Aggarwal, R.; Ahn, S.Y. Global and regional mortality from 235 causes of death for 20 age groups in 1990 and 2010: A systematic analysis for the Global Burden of Disease Study 2010. Lancet 2012, 380, 2095–2128. [Google Scholar] [CrossRef]
- Mathers, C. The Global Burden of Disease: 2004 Update; World Health Organization: Geneva, Switzerland, 2008. [Google Scholar]
- Ramirez, J.A.; Wiemken, T.L.; Peyrani, P.; Arnold, F.W.; Kelley, R.; Mattingly, W.A.; Nakamatsu, R.; Pena, S.; Guinn, B.E.; Furmanek, S.P. Adults hospitalized with pneumonia in the United States: Incidence, epidemiology, and mortality. Clin. Infect. Dis. 2017, 65, 1806–1812. [Google Scholar] [CrossRef] [Green Version]
- McLaughlin, J.M.; McGinnis, J.J.; Tan, L.; Mercatante, A.; Fortuna, J. Estimated human and economic burden of four major adult vaccine-preventable diseases in the United States, 2013. J. Prim. Prev. 2015, 36, 259–273. [Google Scholar] [CrossRef] [Green Version]
- Sakamoto, A.; Chanyasanha, C.; Sujirarat, D.; Matsumoto, N.; Nakazato, M. Factors associated with pneumococcal vaccination in elderly people: A cross-sectional study among elderly club members in Miyakonojo City, Japan. BMC Public Health 2018, 18, 1172. [Google Scholar] [CrossRef] [PubMed]
- CDC. Assessing Risk Factors for Severe COVID-19 Illness. Available online: https://www.cdc.gov/coronavirus/2019-ncov/covid-data/investigations-discovery/assessing-risk-factors.html (accessed on 30 November 2021).
- World Health Organization. COVID-19 Clinical Management: Living Guidance, 25 January 2021; World Health Organization: Geneva, Switzerland, 2021. [Google Scholar]
- CDC. People with Certain Medical Conditions. Available online: https://www.cdc.gov/coronavirus/2019-ncov/need-extra-precautions/people-with-medical-conditions.html (accessed on 13 May 2021).
- CDC. Evidence Used to Update the List of Underlying Medical Conditions That Increase a Person’s Risk of Severe Illness from COVID-19; Centers for Disease Control and Prevention: Atlanta, GA, USA, 2021.
- World Health Organization. Clinical Management of Severe Acute Respiratory Infection (SARI) When COVID-19 Disease is Suspected: Interim Guidance, 13 March 2020; World Health Organization: Geneva, Switzerland, 2020. [Google Scholar]
- CDC. Pneumococcal Disease: Risk Factors and Transmission. Available online: https://www.cdc.gov/pneumococcal/about/risk-transmission.html (accessed on 1 September 2021).
- Shea, K.M.; Edelsberg, J.; Weycker, D.; Farkouh, R.A.; Strutton, D.R.; Pelton, S.I. Rates of pneumococcal disease in adults with chronic medical conditions. Open Forum Infect. Dis. 2014, 1, ofu024. [Google Scholar] [CrossRef]
- Australian Government, Department of Health, People at Higher Risk of Coronavirus (COVID-19). Available online: https://www.health.gov.au/news/health-alerts/novel-coronavirus-2019-ncov-health-alert/ongoing-support-during-coronavirus-covid-19/what-you-need-to-know-about-coronavirus-covid-19/people-at-higher-risk-of-coronavirus-covid-19 (accessed on 7 July 2021).
- González-Romo, F.; Picazo, J.J.; García Rojas, A.; Labrador Horrillo, M.; Barrios, V.; Magro, M.C.; Gregorio, P.G.; de la Cámara, R.; Rodríguez, A.; Barberán, J. Consenso sobre la vacunación anti-neumocócica en el adulto por riesgo de edad y patología de base. Actualización 2017. Rev. Esp. Quimioter. 2017, 30, 142–168. [Google Scholar] [PubMed]
- Government of Canada, Coronavirus Disease (COVID-19): Prevention and Risks. Available online: https://www.canada.ca/en/public-health/services/diseases/2019-novel-coronavirus-infection/prevention-risks.html (accessed on 16 August 2021).
- Zhang, D.; Petigara, T.; Yang, X. Clinical and economic burden of pneumococcal disease in US adults aged 19–64 years with chronic or immunocompromising diseases: An observational database study. BMC Infect. Dis. 2018, 18, 436. [Google Scholar] [CrossRef]
- Haut Conseil de la Santé Publique. Infections Invasives à Pneumocoque: Recommandations Vaccinales Pour les Personnes à Risque. 2013. Available online: https://www.hcsp.fr/Explore.cgi/AvisRapportsDomaine?clefr=355 (accessed on 1 September 2021).
- Imai, K.; Petigara, T.; Kohn, M.A.; Nakashima, K.; Aoshima, M.; Shito, A.; Kanazu, S. Risk of pneumococcal diseases in adults with underlying medical conditions: A retrospective, cohort study using two Japanese healthcare databases. BMJ Open 2018, 8, e018553. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lee, M.S.; Oh, J.Y.; Kang, C.-I.; Kim, E.S.; Park, S.; Rhee, C.K.; Jung, J.Y.; Jo, K.-W.; Heo, E.Y.; Park, D.-A.; et al. Guideline for Antibiotic Use in Adults with Community-acquired Pneumonia. Infect. Chemother. 2018, 50, 160–198. [Google Scholar] [CrossRef]
- Rivero-Calle, I.; Cebey-López, M.; Pardo-Seco, J.; Yuste, J.; Redondo, E.; Vargas, D.A.; Mascarós, E.; Díaz-Maroto, J.L.; Linares-Rufo, M.; Jimeno, I. Lifestyle and comorbid conditions as risk factors for community-acquired pneumonia in outpatient adults (NEUMO-ES-RISK project). BMJ Open Respir. Res. 2019, 6, e000359. [Google Scholar] [CrossRef] [Green Version]
- Vinogradova, Y.; Hippisley-Cox, J.; Coupland, C. Identification of new risk factors for pneumonia: Population-based case-control study. Br. J. Gen. Pract. 2009, 59, e329–e338. [Google Scholar] [CrossRef]
- Public Health England. Pneumococcal: The Green Book, Chapter 25; Public Health England Government: London, UK, 2020.
- O’Brien, D.G.K.L. Harrison’s Principles of Internal Medicine; McGraw-Hill Education: New York, NY, USA, 2018. [Google Scholar]
- Fine, M.J.; Smith, M.A.; Carson, C.A.; Mutha, S.S.; Sankey, S.S.; Weissfeld, L.A.; Kapoor, W.N. Prognosis and outcomes of patients with community-acquired pneumonia. A meta-analysis. JAMA 1996, 275, 134–141. [Google Scholar] [CrossRef]
- Örtqvist, Å.; Hedlund, J.; Kalin, M. Streptococcus pneumoniae: Epidemiology, risk factors, and clinical features. In Seminars in Respiratory and Critical Care Medicine; Thieme Medical Publishers, Inc.: New York, NY, USA, 2005; pp. 563–574. [Google Scholar]
- Public Health England. Disparities in the Risk and Outcomes of COVID-19; Public Health England: London, UK, 2020.
- CDC. Menu of State Long-Term Care Facility Pneumococcal Vaccination Laws. Available online: https://www.cdc.gov/phlp/publications/topic/menus/ltcpneumo/index.html (accessed on 28 February 2021).
- World Health Organization. Infection Prevention and Control Guidance for Long-Term Care Facilities in the Context of COVID-19: Interim Guidance, 21 March 2020; World Health Organization: Geneva, Switzerland, 2020. [Google Scholar]
- Qureshi, H.; Sharafkhaneh, A.; Hanania, N.A. Chronic obstructive pulmonary disease exacerbations: Latest evidence and clinical implications. Ther. Adv. Chronic Dis. 2014, 5, 212–227. [Google Scholar] [CrossRef] [PubMed]
- Guan, W.-J.; Liang, W.-H.; Zhao, Y.; Liang, H.-R.; Chen, Z.-S.; Li, Y.-M.; Liu, X.-Q.; Chen, R.-C.; Tang, C.-L.; Wang, T. Comorbidity and its impact on 1590 patients with COVID-19 in China: A nationwide analysis. Eur. Respir. J. 2020, 55, 2000547. [Google Scholar] [CrossRef] [Green Version]
- Yoon, H.K.; Park, Y.-B.; Rhee, C.K.; Lee, J.H.; Oh, Y.-M.; Committee of the Korean COPD Guideline. Summary of the Chronic Obstructive Pulmonary Disease Clinical Practice Guideline Revised in 2014 by the Korean Academy of Tuberculosis and Respiratory Disease. Tuberc. Respir. Dis. 2017, 80, 230–240. [Google Scholar] [CrossRef]
- Froes, F.; Roche, N.; Blasi, F. Pneumococcal vaccination and chronic respiratory diseases. Int. J. Chronic Obs. Pulmon. Dis. 2017, 12, 3457–3468. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Singh, D.; Agusti, A.; Anzueto, A.; Barnes, P.; Bourbeau, J.; Celli, B.; Criner, G.; Frith, P.; Halpin, D.; Han, M.; et al. Global Strategy for the Diagnosis, Management, and Prevention of Chronic Obstructive Lung Disease: The GOLD science committee report 2019. Eur. Respir. J. 2019, 18, 1900164. [Google Scholar] [CrossRef] [PubMed]
- Gillissen, A.; Paparoupa, M. Inflammation and infections in asthma. Clin. Respir. J. 2015, 9, 257–269. [Google Scholar] [CrossRef]
- Zaidi, S.R.; Blakey, J.D. Why are people with asthma susceptible to pneumonia? A review of factors related to upper airway bacteria. Respirology 2019, 24, 423–430. [Google Scholar] [CrossRef] [PubMed]
- Talbot, T.R.; Hartert, T.V.; Mitchel, E.; Halasa, N.B.; Arbogast, P.G.; Poehling, K.A.; Schaffner, W.; Craig, A.S.; Griffin, M.R. Asthma as a risk factor for invasive pneumococcal disease. N. Engl. J. Med. 2005, 352, 2082–2090. [Google Scholar] [CrossRef]
- Wyrwich, K.W.; Yu, H.; Sato, R.; Strutton, D.; Powers, J.H. Community-acquired pneumonia: Symptoms and burden of illness at diagnosis among US adults aged 50 years and older. Patient-Patient-Cent. Outcomes Res. 2013, 6, 125–134. [Google Scholar] [CrossRef]
- Izquierdo, J.L.; Almonacid, C.; González, Y.; Del Rio-Bermúdez, C.; Ancochea, J.; Cárdenas, R.; Soriano, J.B. The Impact of COVID-19 on Patients with Asthma. Eur. Respir. J. 2020, 57, 2003142. [Google Scholar] [CrossRef]
- The National Heart, Lung and Blood Institute. Morbidity and Mortality: 2012 Chartbook on Cardiovascular, Lung and Blood Diseases; US Department of Health and Human Services; Public Health Service, National Institutes of Health: Bethesda, MD, USA, 2012.
- Wong, N.D. Epidemiological studies of CHD and the evolution of preventive cardiology. Nat. Rev. Cardiol. 2014, 11, 276. [Google Scholar] [CrossRef]
- Warren-Gash, C.; Smeeth, L.; Hayward, A.C. Influenza as a trigger for acute myocardial infarction or death from cardiovascular disease: A systematic review. Lancet Infect. Dis. 2009, 9, 601–610. [Google Scholar] [CrossRef]
- Bova, I.; Bornstein, N.; Korczyn, A. Acute infection as a risk factor for ischemic stroke. Stroke 1996, 27, 2204–2206. [Google Scholar] [CrossRef]
- Yende, S.; D’Angelo, G.; Kellum, J.A.; Weissfeld, L.; Fine, J.; Welch, R.D.; Kong, L.; Carter, M.; Angus, D.C. Inflammatory markers at hospital discharge predict subsequent mortality after pneumonia and sepsis. Am. J. Respir. Crit. Care Med. 2008, 177, 1242–1247. [Google Scholar] [CrossRef] [Green Version]
- Corrales-Medina, V.F.; Alvarez, K.N.; Weissfeld, L.A.; Angus, D.C.; Chirinos, J.A.; Chang, C.C.; Newman, A.; Loehr, L.; Folsom, A.R.; Elkind, M.S.; et al. Association between hospitalization for pneumonia and subsequent risk of cardiovascular disease. JAMA 2015, 313, 264–274. [Google Scholar] [CrossRef]
- Story, A.N. ACC Clinical Bulletin Focuses on Cardiac Implications of Coronavirus (COVID-19). Available online: https://www.acc.org/latest-in-cardiology/articles/2020/02/13/12/42/acc-clinical-bulletin-focuses-on-cardiac-implications-of-coronavirus-2019-ncov (accessed on 13 February 2021).
- Friedberg, C.K. Diseases of the Heart. Acad. Med. 1950, 25, 159. [Google Scholar]
- Bennett, J.E.; Dolin, R.; Blaser, M.J. Mandell, Douglas, and Bennett’s Principles and Practice of Infectious Diseases: 2-Volume Set; Elsevier Health Sciences: London, UK, 2014; Volume 2. [Google Scholar]
- Mor, A.; Thomsen, R.W.; Ulrichsen, S.P.; Sørensen, H.T. Chronic heart failure and risk of hospitalization with pneumonia: A population-based study. Eur. J. Intern. Med. 2013, 24, 349–353. [Google Scholar] [CrossRef] [PubMed]
- The National Advisory Committee on Immunization. Pneumococcal Vaccine. Canadian Immunization Guide; Public Health Agency of Canada (PHAC): Ottawa, ON, USA, 2016. [Google Scholar]
- Vaziri, N.; Pahl, M.; Crum, A.; Norris, K. Effect of uremia on structure and function of immune system. J. Ren. Nutr. 2012, 22, 149–156. [Google Scholar] [CrossRef] [Green Version]
- Kato, S.; Chmielewski, M.; Honda, H.; Pecoits-Filho, R.; Matsuo, S.; Yuzawa, Y.; Tranaeus, A.; Stenvinkel, P.; Lindholm, B. Aspects of immune dysfunction in end-stage renal disease. Clin. J. Am. Soc. Nephrol. 2008, 3, 1526–1533. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Immunization of Immunocompromised Persons: Canadian Immunization Guide. Available online: https://www.canada.ca/en/public-health/services/publications/healthy-living/canadian-immunization-guide-part-3-vaccination-specific-populations/page-8-immunization-immunocompromised-persons.html (accessed on 23 November 2021).
- Gansevoort, R.T.; Hilbrands, L.B. CKD is a key risk factor for COVID-19 mortality. Nat. Rev. Nephrol. 2020, 16, 705–706. [Google Scholar] [CrossRef] [PubMed]
- Guan, W.-J.; Ni, Z.-Y.; Hu, Y.; Liang, W.-H.; Ou, C.-Q.; He, J.-X.; Liu, L.; Shan, H.; Lei, C.-L.; Hui, D.S.C.; et al. Clinical characteristics of coronavirus disease 2019 in China. N. Engl. J. Med. 2020, 382, 1708–1720. [Google Scholar] [CrossRef] [PubMed]
- Mao, R.; Qiu, Y.; He, J.-S.; Tan, J.-Y.; Li, X.-H.; Liang, J.; Shen, J.; Zhu, L.-R.; Chen, Y.; Iacucci, M.; et al. Manifestations and prognosis of gastrointestinal and liver involvement in patients with COVID-19: A systematic review and meta-analysis. Lancet Gastroenterol. Hepatol. 2020, 5, 667–678. [Google Scholar] [CrossRef]
- Immunization of Persons with Chronic Diseases: Canadian Immunization Guide. Available online: https://www.canada.ca/en/public-health/services/publications/healthy-living/canadian-immunization-guide-part-3-vaccination-specific-populations/page-7-immunization-persons-with-chronic-diseases.html (accessed on 23 November 2021).
- CDC. Vaccination of Adults with Liver Disease. Available online: https://www.cdc.gov/vaccines/adults/rec-vac/health-conditions/liver-disease.html (accessed on 2 May 2021).
- Geerlings, S.E.; Hoepelman, A.I. Immune dysfunction in patients with diabetes mellitus (DM). FEMS Immunol. Med. Microbiol. 1999, 26, 259–265. [Google Scholar] [CrossRef] [PubMed]
- Muller, L.M.; Gorter, K.J.; Hak, E.; Goudzwaard, W.L.; Schellevis, F.G.; Hoepelman, A.I.; Rutten, G.E. Increased risk of common infections in patients with type 1 and type 2 diabetes mellitus. Clin. Infect. Dis. 2005, 41, 281–288. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Peleg, A.Y.; Weerarathna, T.; McCarthy, J.S.; Davis, T.M. Common infections in diabetes: Pathogenesis, management and relationship to glycaemic control. Diabetes Metab. Res. Rev. 2007, 23, 3–13. [Google Scholar] [CrossRef] [PubMed]
- Vardakas, K.Z.; Siempos, I.I.; Falagas, M.E. Diabetes mellitus as a risk factor for nosocomial pneumonia and associated mortality. Diabet. Med. 2007, 24, 1168–1171. [Google Scholar] [CrossRef]
- Casqueiro, J.; Casqueiro, J.; Alves, C. Infections in patients with diabetes mellitus: A review of pathogenesis. Indian J. Endocrinol. Metab. 2012, 16, 27–36. [Google Scholar] [CrossRef]
- Joshi, N.; Caputo, G.M.; Weitekamp, M.R.; Karchmer, A.W. Infections in patients with diabetes mellitus. N. Engl. J. Med. 1999, 341, 1906–1912. [Google Scholar] [CrossRef] [PubMed]
- Kunutsor, S.K.; Mäkikallio, T.H.; Seidu, S.; de Araújo, C.G.S.; Dey, R.S.; Blom, A.W.; Laukkanen, J.A. Physical activity and risk of venous thromboembolism: Systematic review and meta-analysis of prospective cohort studies. Eur. J. Epidemiol. 2020, 35, 431–442. [Google Scholar] [CrossRef] [Green Version]
- Lim, S.; Bae, J.H.; Kwon, H.-S.; Nauck, M.A. COVID-19 and diabetes mellitus: From pathophysiology to clinical management. Nat. Rev. Endocrinol. 2021, 17, 11–30. [Google Scholar] [CrossRef]
- Control CFD. Prevention. Influenza and pneumococcal vaccination coverage among persons aged> or = 65 years and persons aged 18–64 years with diabetes or asthma–United States, 2003. MMWR Morb. Mortal. Wkly. Rep. 2004, 53, 1007–1012. [Google Scholar]
- Semenov, Y.R.; Starmer, H.M.; Gourin, C.G. The effect of pneumonia on short-term outcomes and cost of care after head and neck cancer surgery. Laryngoscope 2012, 122, 1994–2004. [Google Scholar] [CrossRef]
- Lipsky, B.A.; Boyko, E.J.; Inui, T.S.; Koepsell, T.D. Risk factors for acquiring pneumococcal infections. Arch. Intern. Med. 1986, 146, 2179–2185. [Google Scholar] [CrossRef] [PubMed]
- Pai, S. Vaccinations for Cancer Patients: What to Know. Available online: https://www.mdanderson.org/cancerwise/vaccinations-for-cancer-patients--what-to-know.h00-159149190.html (accessed on 16 October 2021).
- Docherty, A.; Harrison, E.; Green, C.; Hardwick, H.; Pius, R.; Norman, L.; Holden, K.; Read, J.; Dondelinger, F.; Carson, G.; et al. Features of 16,749 hospitalised UK patients with COVID-19 using the ISARIC WHO Clinical Characterisation Protocol. medRxiv 2020, 1, 20076042. [Google Scholar] [CrossRef]
- Raman, A.S.; Swinburne, A.J.; Fedullo, A.J. Pneumococcal adherence to the buccal epithelial cells of cigarette smokers. Chest 1983, 83, 23–27. [Google Scholar] [CrossRef] [Green Version]
- Jacups, S.P.; Cheng, A. The epidemiology of community acquired bacteremic pneumonia, due to Streptococcus pneumoniae, in the top end of the Northern Territory, Australia—Over 22 years. Vaccine 2011, 29, 5386–5392. [Google Scholar] [CrossRef]
- Hamer, M.; Kivimäki, M.; Gale, C.R.; Batty, G.D. Lifestyle risk factors, inflammatory mechanisms, and COVID-19 hospitalization: A community-based cohort study of 387,109 adults in UK. Brain Behav. Immun. 2020, 87, 184–187. [Google Scholar] [CrossRef] [PubMed]
- Vardavas, C.I.; Nikitara, K. COVID-19 and smoking: A systematic review of the evidence. Tob. Induc. Dis. 2020, 18, 20. [Google Scholar] [CrossRef]
- Grau, I.; Ardanuy, C.; Calatayud, L.; Schulze, M.H.; Liñares, J.; Pallares, R. Smoking and alcohol abuse are the most preventable risk factors for invasive pneumonia and other pneumococcal infections. Int. J. Infect. Dis. 2014, 25, 59–64. [Google Scholar] [CrossRef] [Green Version]
- Smith, A.M.; McCullers, J.A. Secondary bacterial infections in influenza virus infection pathogenesis. Influenza Pathog. Control-Vol. I 2014, 385, 327–356. [Google Scholar]
- Lai, C.-C.; Wang, C.-Y.; Hsueh, P.-R. Co-infections among patients with COVID-19: The need for combination therapy with non-anti-SARS-CoV-2 agents? J. Microbiol. Immunol. Infect. 2020, 53, 505–512. [Google Scholar] [CrossRef]
- Bengoechea, J.A.; Bamford, C.G. SARS-CoV-2, bacterial co-infections, and AMR: The deadly trio in COVID-19? EMBO Mol. Med. 2020, 12, e12560. [Google Scholar] [CrossRef] [PubMed]
- Guo, L.; Wei, D.; Zhang, X.; Wu, Y.; Li, Q.; Zhou, M.; Qu, J. Clinical Features Predicting Mortality Risk in Patients With Viral Pneumonia: The MuLBSTA Score. Front. Microbiol. 2019, 10, 2752. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cox, M.J.; Loman, N.; Bogaert, D.; O’Grady, J. Co-infections: Potentially lethal and unexplored in COVID-19. Lancet Microbe 2020, 1, e11. [Google Scholar] [CrossRef]
- Lansbury, L.; Lim, B.; Baskaran, V.; Lim, W.S. Co-infections in people with COVID-19: A systematic review and meta-analysis. J. Infect. 2020, 81, 266–275. [Google Scholar] [CrossRef] [PubMed]
- Nag, V.L.; Kaur, N. Superinfections in COVID-19 Patients: Role of Antimicrobials. Dubai Med. J. 2021, 4, 117–126. [Google Scholar] [CrossRef]
- Garcia-Vidal, C.; Sanjuan, G.; Moreno-García, E.; Puerta-Alcalde, P.; Garcia-Pouton, N.; Chumbita, M.; Fernandez-Pittol, M.; Pitart, C.; Inciarte, A.; Bodro, M.; et al. Incidence of co-infections and superinfections in hospitalized patients with COVID-19: A retrospective cohort study. Clin. Microbiol. Infect. 2021, 27, 83–88. [Google Scholar] [CrossRef]
- Rawson, T.M.; Wilson, R.C.; Holmes, A. Understanding the role of bacterial and fungal infection in COVID-19. Clin. Microbiol. Infect. 2021, 27, 9. [Google Scholar] [CrossRef]
- Pittet, L.; Posfay-Barbe, K. Pneumococcal vaccines for children: A global public health priority. Clin. Microbiol. Infect. 2012, 18, 25–36. [Google Scholar] [CrossRef] [Green Version]
- Vos, Q.; Lees, A.; Wu, Z.-Q.; Snapper, C.; Mond, J. B-cell activation by T-cell-independent type 2 antigens as an integral part of the humoral immune response to pathogenic microorganisms. Immunol. Rev. 2000, 176, 154–170. [Google Scholar]
- Defrance, T.; Taillardet, M.; Genestier, L. T cell-independent B cell memory. Curr. Opin. Immunol. 2011, 23, 330–336. [Google Scholar] [CrossRef]
- Papadatou, I.; Spoulou, V. Pneumococcal vaccination in high-risk individuals: Are we doing it right? Clin. Vaccine Immunol. 2016, 23, 388–395. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Avci, F.Y.; Kasper, D.L. How bacterial carbohydrates influence the adaptive immune system. Annu. Rev. Immunol. 2009, 28, 107–130. [Google Scholar] [CrossRef] [Green Version]
- Bonten, M.J.; Huijts, S.M.; Bolkenbaas, M.; Webber, C.; Patterson, S.; Gault, S.; van Werkhoven, C.H.; van Deursen, A.M.; Sanders, E.A.; Verheij, T.J. Polysaccharide conjugate vaccine against pneumococcal pneumonia in adults. N. Engl. J. Med. 2015, 372, 1114–1125. [Google Scholar] [CrossRef] [Green Version]
- CDC. Vaccines and Preventable Disease. Available online: https://www.cdc.gov/vaccines/vpd/pneumo/index.html (accessed on 21 November 2021).
- The National Institute for Communicable Diseases. Pneumococcal Conjugate Vaccine Use in the Light of the COVID-19 Pandemic. Available online: https://www.nicd.ac.za/diseases-a-z-index/disease-index-covid-19/advice-for-the-public/pneumococcal-conjugate-vaccine-use-in-the-light-of-the-covid-19-pandemic/ (accessed on 30 April 2021).
- Harvard Medical School. Preventing the Spread of the Coronavirus. Available online: https://www.health.harvard.edu/diseases-and-conditions/preventing-the-spread-of-the-coronavirus (accessed on 23 November 2021).
- World Health Organization. Coronavirus Disease (COVID-19) Advice for the Public: Mythbusters. Available online: https://www.who.int/emergencies/diseases/novel-coronavirus-2019/advice-for-public/myth-busters?gclid=Cj0KCQjw8IaGBhCHARIsAGIRRYorv3VefRlfgb7l5za4EEeus4hEII_CQX-UuZVr4mH1y8DN-cXIvnMaAkcpEALw_wcB#vaccines (accessed on 5 May 2021).
- Pawlowski, C.; Puranik, A.; Bandi, H.; Venkatakrishnan, A.J.; Agarwal, V.; Kennedy, R.; O’Horo, J.C.; Gores, G.J.; Williams, A.W.; Halamka, J.; et al. Exploratory analysis of immunization records highlights decreased SARS-CoV-2 rates in individuals with recent non-COVID-19 vaccinations. Sci. Rep. 2021, 11, 4741. [Google Scholar] [CrossRef] [PubMed]
- Root-Bernstein, R. Until a Coronavirus Vaccine is Ready, Pneumonia Vaccines may Reduce Deaths from COVID-19. Available online: https://www.discovermagazine.com/health/until-a-coronavirus-vaccine-is-ready-pneumonia-vaccines-may-reduce-deaths (accessed on 15 October 2021).
- Lewnard, J.A.; Bruxvoort, K.J.; Fischer, H.; Hong, V.X.; Grant, L.R.; Jódar, L.; Gessner, B.D.; Tartof, S.Y. Prevention of COVID-19 among older adults receiving pneumococcal conjugate vaccine suggests interactions between Streptococcus pneumoniae and SARS-CoV-2 in the respiratory tract. J. Infect. Dis. 2021, 3, jiab128. [Google Scholar] [CrossRef]
- Madhi, S.A.; Klugman, K.P.; Group, T.V.T. A role for Streptococcus pneumoniae in virus-associated pneumonia. Nat. Med. 2004, 10, 811–813. [Google Scholar] [CrossRef]
- Huijts, S.; Coenjaerts, F.; Bolkenbaas, M.; van Werkhoven, C.; Grobbee, D.; Bonten, M.; Team, C.S. The impact of 13-valent pneumococcal conjugate vaccination on virus-associated community-acquired pneumonia in elderly: Exploratory analysis of the CAPiTA trial. Clin. Microbiol. Infect. 2018, 24, 764–770. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nunes, M.C.; Cutland, C.L.; Klugman, K.P.; Madhi, S.A. Pneumococcal Conjugate Vaccine Protection against Coronavirus-Associated Pneumonia Hospitalization in Children Living with and without HIV. mBio 2021, 12, e02347-20. [Google Scholar] [CrossRef]
- Palmu, A.A.; Jokinen, J.; Borys, D.; Nieminen, H.; Ruokokoski, E.; Siira, L.; Puumalainen, T.; Lommel, P.; Hezareh, M.; Moreira, M. Effectiveness of the ten-valent pneumococcal Haemophilus influenzae protein D conjugate vaccine (PHiD-CV10) against invasive pneumococcal disease: A cluster randomised trial. Lancet 2013, 381, 214–222. [Google Scholar] [CrossRef] [Green Version]
- Root-Bernstein, R. Pneumococcal and Influenza Vaccination Rates and Pneumococcal Invasive Disease Rates Set Geographical and Ethnic Population Susceptibility to Serious COVID-19 Cases and Deaths. Vaccines 2021, 9, 474. [Google Scholar] [CrossRef] [PubMed]
- Thindwa, D.; Quesada, M.G.; Liu, Y.; Bennett, J.; Cohen, C.; Knoll, M.D.; von Gottberg, A.; Hayford, K.; Flasche, S. Use of seasonal influenza and pneumococcal polysaccharide vaccines in older adults to reduce COVID-19 mortality. Vaccine 2020, 38, 5398. [Google Scholar] [CrossRef]
- Matanock, A.; Lee, G.; Gierke, R.; Kobayashi, M.; Leidner, A.; Pilishvili, T. Use of 13-valent pneumococcal conjugate vaccine and 23-valent pneumococcal polysaccharide vaccine among adults aged ≥ 65 years: Updated recommendations of the Advisory Committee on Immunization Practices. Morb. Mortal. Wkly. Rep. 2019, 68, 1069. [Google Scholar] [CrossRef] [Green Version]
- Bonnave, C.; Mertens, D.; Peetermans, W.; Cobbaert, K.; Ghesquiere, B.; Deschodt, M.; Flamaing, J. Adult vaccination for pneumococcal disease: A comparison of the national guidelines in Europe. Eur. J. Clin. Microbiol. Infect. Dis. 2019, 38, 785–791. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Al-Ani, A.H.; Prentice, R.E.; Rentsch, C.A.; Johnson, D.; Ardalan, Z.; Heerasing, N.; Garg, M.; Campbell, S.; Sasadeusz, J.; Macrae, F.A. prevention, diagnosis and management of COVID-19 in the IBD patient. Aliment. Pharmacol. Ther. 2020, 52, 54–72. [Google Scholar] [CrossRef] [PubMed]
- Zanettini, C.; Omar, M.; Dinalankara, W.; Imada, E.L.; Colantuoni, E.; Parmigiani, G.; Marchionni, L. Influenza Vaccination and COVID-19 Mortality in the USA: An Ecological Study. Vaccines 2021, 9, 427. [Google Scholar] [CrossRef]
- Fink, G.; Orlova-Fink, N.; Schindler, T.; Grisi, S.; Ferrer, A.P.; Daubenberger, C.; Brentani, A. Inactivated trivalent influenza vaccination is associated with lower mortality among COVID-19 in Brazil. BMJ Evid. Based Med. 2021, 26, 192–199. [Google Scholar] [CrossRef]
- Marín-Hernández, D.; Schwartz, R.E.; Nixon, D.F. Epidemiological evidence for association between higher influenza vaccine uptake in the elderly and lower COVID-19 deaths in Italy. J. Med. Virol. 2021, 93, 64–65. [Google Scholar] [CrossRef]
- Elston, J.W.; Cartwright, C.; Ndumbi, P.; Wright, J. The health impact of the 2014–15 Ebola outbreak. Public Health 2017, 143, 60–70. [Google Scholar] [CrossRef]
- World Health Organization. Guidance on Routine Immunization Services during COVID-19 Pandemic in the WHO European Region; World Health Organization: Geneva, Switzerland; Regional Office for Europe: København, Denmark, 2020. [Google Scholar]
- Grech, V.; Borg, M. Influenza vaccination in the COVID-19 era. Early Hum. Dev. 2020, 148, 105116. [Google Scholar] [CrossRef] [PubMed]
- Gostin, L.O.; Salmon, D.A. The dual epidemics of COVID-19 and influenza: Vaccine acceptance, coverage, and mandates. JAMA 2020, 324, 335–336. [Google Scholar] [CrossRef] [PubMed]
- Portolés-Pérez, J.; Marques-Vidas, M.; Picazo, J.J.; González-Romo, F.; García-Rojas, A.; Pérez-Trallero, E.; Gil-Gregorio, P.; de la Cámara, R.; Morató, M.L.; Rodríguez, A.; et al. Recommendations for vaccination against pneumococcus in kidney patients in Spain. Nefrologia 2014, 34, 545–551. [Google Scholar] [CrossRef] [PubMed]
- COSMO Healthcare. Immunization: Pneumococcal Polysaccharide Vaccine. Available online: https://www.health.gov.on.ca/en/public/publications/immune/pnem.aspx (accessed on 28 October 2021).
- EACS & BHIVA. Statement on Risk of COVID-19 for People Living with HIV (EACS & BHIVA); EACS: Brussels, Belgium, 2020. [Google Scholar]
| Disease | COVID-19 | Pneumococcal | ||||
|---|---|---|---|---|---|---|
| Ref Risk Factor | CDC [28] | UK [1] | Australia [29] | CDC [30] | CANADA [31] | Research Articles |
| Lung Disease | 1 | 1 | 1 | 1 | 1 | 12 [19,28,30,32,33,34,35,36,37,38,39,40] |
| Heart Disease | 1 | 1 | 1 | 1 | 1 | 11 [28,30,32,33,34,35,36,37,38,40] |
| Kidney Disease | 1 | 1 | 1 | 1 | 1 | 8 [28,30,33,34,36,37,38,39] |
| Liver Disease | 1 | 1 | 1 | 1 | 1 | 9 [28,30,32,33,34,35,37,38,39] |
| Diabetes | 1 | 1 | 1 | 1 | 1 | 12 [19,28,30,32,33,34,35,36,37,38,39,40] |
| Cancer | 1 | 1 | 1 | 1 | 5 [32,33,34,37,38] | |
| Neurologic disorders | 1 | 1 | 1 | 1 [40] | ||
| Sickle cell disease | 1 | 1 | 1 | 2 [37,38] | ||
| HIV infection | 1 | 1 | 1 | 8 [28,30,32,33,36,37,38,39] | ||
| Stroke | 1 | 1 | 4 [19,30,36,37] | |||
| Rheumatoid arthritis | 1 | 1 [37] | ||||
| Smoking | 1 | 1 | 1 | 6 [19,28,30,35,36,40] | ||
| Medical Indication Group | Specific Underlying Medical Condition | PCV13 for Persons Aged ≥ 19 Years | PPSV23 * for Persons Aged 19–64 Years | PCV13 for Persons Aged ≥ 65 Years | PPSV23 for Persons Aged ≥ 65 Years |
|---|---|---|---|---|---|
| None | None of the below | No recommendation | No recommendation | Based on shared clinical decision-making † | 1 dose; if PCV13 has been administered, then administer PPSV23 ≥ 1 year after PCV13 |
| Immunocompetent persons | Alcoholism | No recommendation | 1 dose | Based on shared clinical decision-making † | 1 dose; if PCV13 has been administered, then administer PPSV23 ≥ 1 year after PCV13 and ≥ 5 years after any PPSV23 at age < 65 years |
| Chronic heart disease § | |||||
| Chronic liver disease | |||||
| Chronic lung disease ¶ | |||||
| Cigarette smoking | |||||
| Diabetes mellitus | |||||
| Cochlear implant | 1 dose | 1 dose ≥ 8 weeks after PCV13 | 1 dose if no previous PCV13 vaccination | 1 dose ≥ 8 weeks after PCV13 and ≥ 5 years after any PPSV23 at < 65 years | |
| CSF leak | |||||
| Immunocompromised persons | Congenital or acquired asplenia | 1 dose | 2 doses, 1st dose ≥ 8 weeks after PCV13 and 2nd dose ≥ 5 years after first PPSV23 dose | 1 dose if no previous PCV13 vaccination | dose ≥ 8 weeks after PCV13 and ≥ 5 years after any PPSV23 at < 65 years |
| Sickle cell disease/other hemoglobinopathies | |||||
| Chronic renal failure | |||||
| Congenital or acquired immunodeficiencies ** | |||||
| Generalized malignancy | |||||
| HIV infection | |||||
| Hodgkin disease | |||||
| Iatrogenic immunosuppression †† | |||||
| Leukemia | |||||
| Lymphoma | |||||
| Multiple myeloma | |||||
| Nephrotic syndrome | |||||
| Solid-organ transplant |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Im, H.; Ser, J.; Sim, U.; Cho, H. Promising Expectations for Pneumococcal Vaccination during COVID-19. Vaccines 2021, 9, 1507. https://doi.org/10.3390/vaccines9121507
Im H, Ser J, Sim U, Cho H. Promising Expectations for Pneumococcal Vaccination during COVID-19. Vaccines. 2021; 9(12):1507. https://doi.org/10.3390/vaccines9121507
Chicago/Turabian StyleIm, Hyobin, Jinhui Ser, Uk Sim, and Hoonsung Cho. 2021. "Promising Expectations for Pneumococcal Vaccination during COVID-19" Vaccines 9, no. 12: 1507. https://doi.org/10.3390/vaccines9121507
APA StyleIm, H., Ser, J., Sim, U., & Cho, H. (2021). Promising Expectations for Pneumococcal Vaccination during COVID-19. Vaccines, 9(12), 1507. https://doi.org/10.3390/vaccines9121507







