Serological Response to SARS-CoV-2 Messenger RNA Vaccine: Real-World Evidence from Italian Adult Population
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Design and Population
2.2. Laboratory Methods
2.3. Statistical Analysis
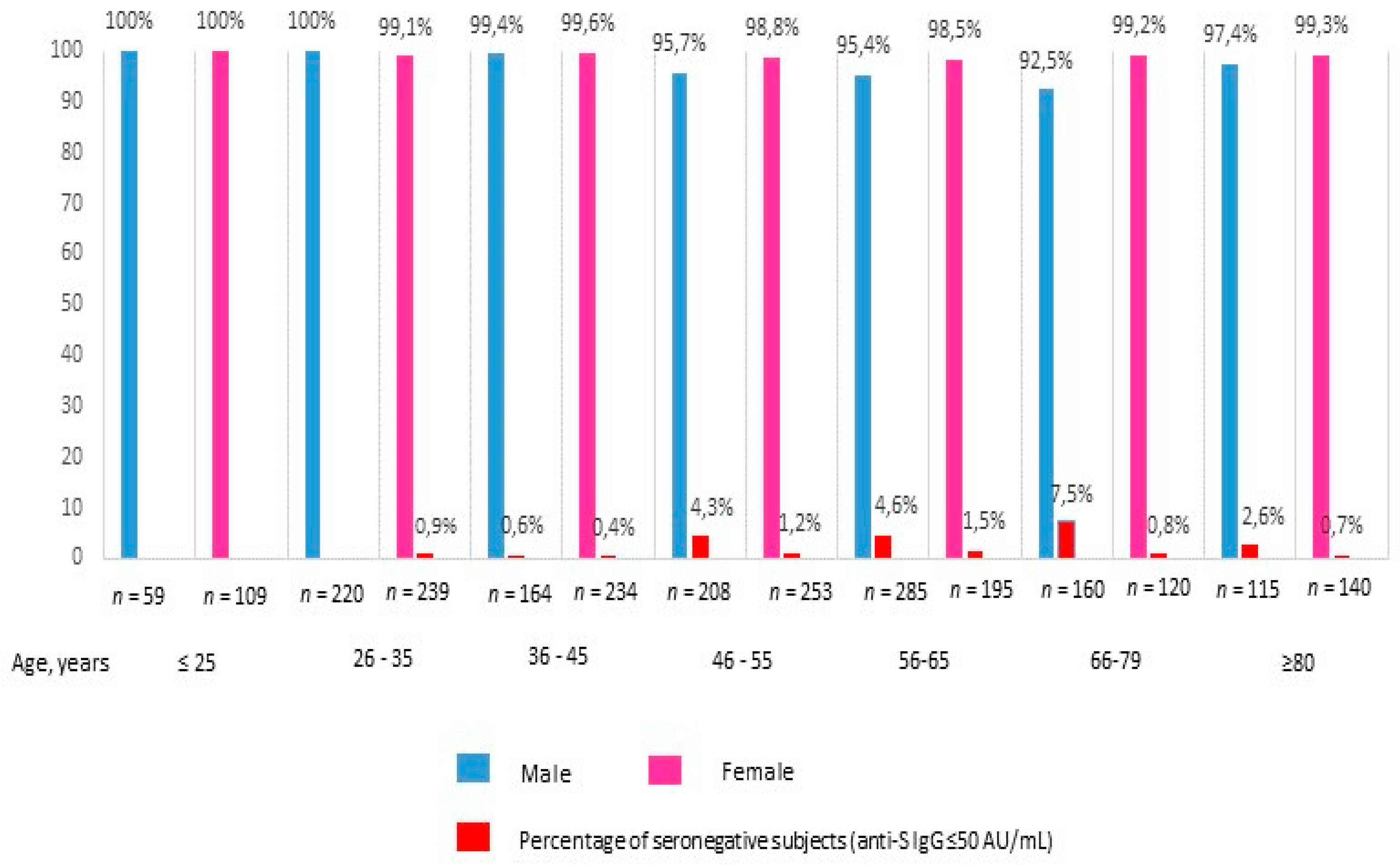
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Gorbalenya, A.E.; Baker, S.C.; Baric, R.S.; de Groot, R.J.; Drosten, C.; Gulyaeva, A.A.; Haagmans, B.L.; Lauber, C.; Leontovich, A.M.; Neuman, B.W.; et al. The species severe acute respiratory syndrome-related coronavirus: Classifying 2019-Ncov and naming it SARS-CoV-2. Nat. Microbiol. 2020, 5, 536–544. [Google Scholar] [CrossRef] [Green Version]
- Yang, X.; Yu, Y.; Xu, J.; Shu, H.; Xia, J.; Liu, H.; Wu, Y.; Zhang, L.; Yu, Z.; Fang, M.; et al. Clinical course and outcomes of critically ill patients with SARS-CoV-2 pneumonia in Wuhan, China: A single-centered, retrospective, observational study. Lancet Respir. Med. 2020, 8, 475–481. [Google Scholar] [CrossRef] [Green Version]
- Zhu, N.; Zhang, D.; Wang, W.; Li, X.; Yang, B.; Song, J.; Zhao, X.; Huang, B.; Shi, W.; Lu, R.; et al. A Novel Coronavirus from Patients with Pneumonia in China. N. Engl. J. Med. 2020, 382, 727–733. [Google Scholar] [CrossRef] [PubMed]
- Yang, P.; Wang, X. COVID-19: A new challenge for human beings. Cell. Mol. Immunol. 2020, 17, 555–557. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- World Health Organization (WHO). Weekly Epidemiological Update on COVID-19—3 August. Available online: https://www.who.int/publications/m/item/weekly-epidemiological-update-on-covid-19---3-august-2021 (accessed on 10 August 2021).
- Salzberger, B.; Buder, F.; Lampl, B.; Ehrenstein, B.; Hitzenbichler, F.; Holzmann, T.; Schmidt, B.; Hanses, F. Epidemiology of SARS-CoV-2. Infection 2021, 49, 233–239. [Google Scholar] [CrossRef]
- Horowitz, R.I.; Freeman, P.R. Three novel prevention, diagnostic, and treatment options for COVID-19 urgently necessitating controlled randomized trials. Med. Hypotheses 2020, 143, 109851. [Google Scholar] [CrossRef] [PubMed]
- Johansson, M.A.; Quandelacy, T.M.; Kada, S.; Prasad, P.V.; Steele, M.; Brooks, J.T.; Slayton, R.B.; Biggerstaff, M.; Butler, J.C. SARS-CoV-2 Transmission from people without COVID-19 symptoms. JAMA Netw. Open 2021, 4, e2035057. [Google Scholar] [CrossRef]
- Oran, A.D.P.; Topol, E.J. The proportion of SARS-CoV-2 infections that are asymptomatic: A Systematic Review. Ann. Intern. Med. 2021, 174, 655–662. [Google Scholar] [CrossRef]
- Sanders, J.M.; Monogue, M.L.; Jodlowski, T.Z.; Cutrell, J.B. Pharmacologic treatments for coronavirus disease 2019 (COVID-19): A Review. JAMA 2020, 32, 1824–1836. [Google Scholar] [CrossRef]
- Gallelli, L.; Zhang, L.; Wang, T.; Fu, F. Severe acute lung injury related to COVID-19 infection: A Review and the possible role for escin. J. Clin. Pharmacol. 2020, 60, 815–825. [Google Scholar] [CrossRef]
- Adam, L.; Rosenbaum, P.; Bonduelle, O.; Combadière, B. Strategies for immunomonitoring after vaccination and during infection. Vaccines 2021, 9, 365. [Google Scholar] [CrossRef] [PubMed]
- Long, Q.-X.; Liu, B.-Z.; Deng, H.-J.; Wu, G.-C.; Deng, K.; Chen, Y.-K.; Liao, P.; Qiu, J.-F.; Lin, Y.; Cai, X.-F.; et al. Antibody responses to SARS-CoV-2 in patients with COVID-19. Nat. Med. 2020, 26, 845–848. [Google Scholar] [CrossRef] [PubMed]
- Eyre, D.W.; Lumley, S.F.; Wei, J.; Cox, S.; James, T.; Justice, A.; Jesuthasan, G.; O’Donnell, D.; Howarth, A.; Hatch, S.B.; et al. Quantitative SARS-CoV-2 anti-spike responses to Pfizer–BioNTech and Oxford–AstraZeneca vaccines by previous infection status. Clin. Microbiol. Infect. 2021, 27, 1516.e7–1516.e14. [Google Scholar] [CrossRef]
- Ebinger, J.E.; Fert-Bober, J.; Printsev, I.; Wu, M.; Sun, N.; Prostko, J.C.; Frias, E.C.; Stewart, J.L.; Van Eyk, J.E.; Braun, J.G.; et al. Antibody responses to the BNT162b2 mRNA vaccine in individuals previously infected with SARS-CoV-2. Nat. Med. 2021, 27, 981–984. [Google Scholar] [CrossRef]
- Italian Ministry of Health. Official Strategic Plan 12 December 2021 Official Gazette no. 72 of 24 March 2021. Available online: https://www.gazzettaufficiale.it/eli/gu/2021/03/24/72/sg/pdf (accessed on 10 August 2021).
- Hanlon, P.; Nicholl, B.I.; Jani, B.D.; Lee, D.; McQueenie, R.; Mair, F.S. Frailty and pre-frailty in middle-aged and older adults and its association with multimorbidity and mortality: A prospective analysis of 493 737 UK Biobank participants. Lancet Public Health 2018, 3, e323–e332. [Google Scholar] [CrossRef]
- Fraley, E.; LeMaster, C.; Geanes, E.; Banerjee, D.; Khanal, S.; Grundberg, E.; Selvarangan, R.; Bradley, T. Humoral immune responses during SARS-CoV-2 mRNA vaccine administration in seropositive and seronegative individuals. BMC Med. 2021, 19, 169. [Google Scholar] [CrossRef]
- Wang, Z.; Schmidt, F.; Weisblum, Y.; Muecksch, F.; Barnes, C.O.; Finkin, S.; Schaefer-Babajew, D.; Cipolla, M.; Gaebler, C.; Lieberman, J.A.; et al. mRNA vaccine-elicited antibodies to SARS-CoV-2 and circulating variants. Nat. Cell Biol. 2021, 592, 616–622. [Google Scholar] [CrossRef] [PubMed]
- Pellini, R.; Venuti, A.; Pimpinelli, F.; Abril, E.; Blandino, G.; Campo, F.; Conti, L.; De Virgilio, A.; De Marco, F.; Di Domenico, E.G.; et al. Initial observations on age, gender, BMI and hypertension in antibody responses to SARS-CoV-2 BNT162b2 vaccine. EClinicalMedicine 2021, 36, 100928. [Google Scholar] [CrossRef]
- Amodio, E.; Capra, G.; Casuccio, A.; Grazia, S.; Genovese, D.; Pizzo, S.; Calamusa, G.; Ferraro, D.; Giammanco, G.; Vitale, F.; et al. Antibodies responses to SARS-CoV-2 in a large cohort of vaccinated subjects and seropositive patients. Vaccines 2021, 9, 714. [Google Scholar] [CrossRef]
- Abu Jabal, K.; Ben-Amram, H.; Beiruti, K.; Batheesh, Y.; Sussan, C.; Zarka, S.; Edelstein, M. Impact of age, ethnicity, sex and prior infection status on immunogenicity following a single dose of the BNT162b2 mRNA COVID-19 vaccine: Real-world evidence from healthcare workers, Israel, December 2020 to January 2021. Eurosurveillance 2021, 26, 2100096. [Google Scholar] [CrossRef]
- Wei, J.; Stoesser, N.; Matthews, P.C.; Ayoubkhani, D.; Studley, R.; Bell, I.; Bell, J.I.; Newton, J.N.; Farrar, J.; Diamond, I.; et al. Antibody responses to SARS-CoV-2 vaccines in 45,965 adults from the general population of the United Kingdom. Nat. Microbiol. 2021, 6, 1140–1149. [Google Scholar] [CrossRef] [PubMed]
- Attias, P.; Sakhi, H.; Rieu, P.; Soorkia, A.; Assayag, D.; Bouhroum, S.; Nizard, P.; El Karoui, K. Antibody response to the BNT162b2 vaccine in maintenance hemodialysis patients. Kidney Int. 2021, 99, 1490–1492. [Google Scholar] [CrossRef] [PubMed]
- Addeo, A.; Shah, P.K.; Bordry, N.; Hudson, R.D.; Albracht, B.; Di Marco, M.; Kaklamani, V.; Dietrich, P.-Y.; Taylor, B.S.; Simand, P.-F.; et al. Immunogenicity of SARS-CoV-2 messenger RNA vaccines in patients with cancer. Cancer Cell 2021, 39, 1091–1098.e2. [Google Scholar] [CrossRef]
- Pimpinelli, F.; Marchesi, F.; Piaggio, G.; Giannarelli, D.; Papa, E.; Falcucci, P.; Pontone, M.; Di Martino, S.; Laquintana, V.; La Malfa, A.; et al. Fifth-week immunogenicity and safety of anti-SARS-CoV-2 BNT162b2 vaccine in patients with multiple myeloma and myeloproliferative malignancies on active treatment: Preliminary data from a single institution. J. Hematol. Oncol. 2021, 14, 1–12. [Google Scholar] [CrossRef]
- Monin, L.; Laing, A.G.; Muñoz-Ruiz, M.; McKenzie, D.R.; del Molino del Barrio, I.; Alaguthurai, T.; Domingo-Vila, C.; Hayday, T.S.; Graham, C.; Seow, J.; et al. Safety and immunogenicity of one versus two doses of the COVID-19 vaccine BNT162b2 for patients with cancer: Interim analysis of a prospective observational study. Lancet Oncol. 2021, 22, 765–778. [Google Scholar] [CrossRef]
- Fischinger, S.; Boudreau, C.; Butler, A.L.; Streeck, H.; Alter, G. Sex differences in vaccine-induced humoral immunity. Semin. Immunopathol. 2019, 41, 239–249. [Google Scholar] [CrossRef] [Green Version]
- Klein, S.L.; Flanagan, K.L. Sex differences in immune responses. Nat. Rev. Immunol. 2016, 16, 626–638. [Google Scholar] [CrossRef]
- Klein, S.L.; Pekosz, A. Sex-based biology and the rational design of influenza vaccination strategies. J. Infect. Dis. 2014, 209 (Suppl. 3), S114–S119. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pileggi, C.; Papadopoli, R.; Bianco, A.; Pavia, M. Hepatitis B vaccine and the need for a booster dose after primary vaccination. Vaccine 2017, 35, 6302–6307. [Google Scholar] [CrossRef]
- Morgan, R.; Klein, S.L. The intersection of sex and gender in the treatment of in-fluenza. Curr. Opin. Virol. 2019, 35, 35–41. [Google Scholar] [CrossRef] [PubMed]
- Vassallo, A.; Shajahan, S.; Harris, K.; Hallam, L.; Hockham, C.; Womersley, K.; Woodward, M.; Sheel, M. Sex and gender in COVID-19 vaccine research: Substantial evidence gaps remain. Front. Glob. Women’s Health 2021, 2, 761511. [Google Scholar] [CrossRef] [PubMed]
- Polack, F.P.; Thomas, S.J.; Kitchin, N.; Absalon, J.; Gurtman, A.; Lockhart, S.; Perez, J.L.; Pérez Marc, G.; Moreira, E.D.; Zerbini, C.; et al. Safety and efficacy of the BNT162b2 mRNA COVID-19 vaccine. N. Engl. J. Med. 2020, 383, 2603–2615. [Google Scholar] [CrossRef]
- Tan, C.W.; Chia, W.N.; Qin, X.; Liu, P.; Chen, M.I.-C.; Tiu, C.; Hu, Z.; Chen, V.C.-W.; Young, B.E.; Sia, W.R.; et al. A SARS-CoV-2 surrogate virus neutralization test based on antibody-mediated blockage of ACE2–spike protein–protein interaction. Nat. Biotechnol. 2020, 38, 1073–1078. [Google Scholar] [CrossRef] [PubMed]
- Duysburgh, E.; Mortgat, L.; Barbezange, C.; Dierick, K.; Fischer, N.; Heyndrickx, L.; Hutse, V.; Thomas, I.; Van Gucht, S.; Vuylsteke, B.; et al. Persistence of IgG response to SARS-CoV-2. Lancet Infect. Dis. 2021, 21, 163–164. [Google Scholar] [CrossRef]
- Papadopoli, R.; De Sarro, C.; Torti, C.; Pileggi, C.; Pavia, M. Is there any opportunity to provide an HBV vaccine booster dose before anti-Hbs titer vanishes? Vaccines 2020, 8, 227. [Google Scholar] [CrossRef] [PubMed]
- Pinto, L.A.; Dillner, J.; Beddows, S.; Unger, E.R. Immunogenicity of HPV prophylactic vaccines: Serology assays and their use in HPV vaccine evaluation and development. Vaccine 2018, 36 Pt A, 4792–4799. [Google Scholar] [CrossRef]
- Napolitano, F.; Di Giuseppe, G.; Montemurro, M.; Molinari, A.; Donnarumma, G.; Arnese, A.; Pavia, M.; Angelillo, I. Seroprevalence of SARS-CoV-2 antibodies in adults and healthcare workers in southern Italy. Int. J. Environ. Res. Public Health 2021, 18, 4761. [Google Scholar] [CrossRef]
- Alter, M.J.; Arduino, M.J.; Lyerla, H.C.; Miller, E.R.; Tokars, J.I. Recommendations for preventing transmission of infections among chronic hemodialysis patients. MMWR Recomm. Rep. 2001, 50, RR-5. [Google Scholar]
- Bosaeed, M.; Kumar, D. Seasonal influenza vaccine in immunocompromised persons. Hum. Vaccines Immunother. 2018, 14, 1311–1322. [Google Scholar] [CrossRef] [PubMed]
| Characteristic | Total | Anti-S IgG (≤50 AU/mL) | Anti-S IgG (51–4159 AU/mL) | Anti-S IgG (≥4160 AU/mL) | ||||
|---|---|---|---|---|---|---|---|---|
| N (2591) | % | N (50) | 1.9% | N (602) | 23.2% | N (1939) | 74.8% | |
| Gender | ||||||||
| Male Female | 1208 1383 | 46.6 53.4 | 38 12 | 3.1 0.9 | 340 262 | 28.2 18.9 | 830 1109 | 68.7 80.2 |
| χ2 = 52.1, 2 df, p < 0.001 | ||||||||
| Age at testing, years | ||||||||
| Mean ± SD | 50.3 ± 18.5 | 61.2 ± 13.1 | 62.3 ± 17.9 | 46.3 ± 17.1 | ||||
| F = 211.08, 2 df, p < 0.001 | ||||||||
| Risk category | ||||||||
| Healthy subjects Frail subjects Over 80 years | 1910 427 25 | 73.7 16.5 9.8 | 2 44 4 | 0.1 10.3 1.6 | 288 178 136 | 15.1 41.7 53.5 | 1620 205 114 | 84.8 48 44.9 |
| Fisher exact p < 0.001 | ||||||||
| Anti-S IgG at T1(1641) * | ||||||||
| Seronegative (≤50 AU/mL) Seropositive (>50 AU/mL) | 162 1479 | 9.9 90.1 | 25 2 | 15.4 0.1 | 110 241 | 67.9 16.3 | 27 1236 | 16.7 83.6 |
| Fisher exact p < 0.001 | ||||||||
| Likelihood = −656.76; χ2 = 652.3 (10 df); p < 0.0001; No. of Observation = 1641 | ||||
|---|---|---|---|---|
| Outcome: Anti-SARS-CoV-2 Titer Measured after Full Course of Vaccination | ||||
| ≤50 AU/mL | 51–4159 AU/mL | |||
| RRR (95% CI) | p Value | RRR (95% CI) | p Value | |
| Gender Female Male as reference | 0.95 (0.32–2.81) | 0.931 | 0.81 (0.61–1.07) | 0.144 |
| Age at enrollment, continuous | 0.96 (0.92–1.00) | 0.064 | 1.01 (0.99–1.02) | 0.098 |
| Risk category | ||||
| Frail subjects | 4.21 (0.70–25.1) | 0.115 | 1.07 (0.69–1.65) | 0.744 |
| Over 80 | 1.03 (0.08–13.7) | 0.981 | 1.18 (0.64–2.16) | 0.594 |
| Healthy subjects as reference | ||||
| Anti-S IgG at T1 Seropositive (>50 AU/mL) Seronegative (≤50 AU/mL) as reference | 0.94 (0.90–0.97) | <0.001 | 0.99 (0.98–0.99) | <0.001 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Papadopoli, R.; De Sarro, C.; Palleria, C.; Gallelli, L.; Pileggi, C.; De Sarro, G. Serological Response to SARS-CoV-2 Messenger RNA Vaccine: Real-World Evidence from Italian Adult Population. Vaccines 2021, 9, 1494. https://doi.org/10.3390/vaccines9121494
Papadopoli R, De Sarro C, Palleria C, Gallelli L, Pileggi C, De Sarro G. Serological Response to SARS-CoV-2 Messenger RNA Vaccine: Real-World Evidence from Italian Adult Population. Vaccines. 2021; 9(12):1494. https://doi.org/10.3390/vaccines9121494
Chicago/Turabian StylePapadopoli, Rosa, Caterina De Sarro, Caterina Palleria, Luca Gallelli, Claudia Pileggi, and Giovambattista De Sarro. 2021. "Serological Response to SARS-CoV-2 Messenger RNA Vaccine: Real-World Evidence from Italian Adult Population" Vaccines 9, no. 12: 1494. https://doi.org/10.3390/vaccines9121494
APA StylePapadopoli, R., De Sarro, C., Palleria, C., Gallelli, L., Pileggi, C., & De Sarro, G. (2021). Serological Response to SARS-CoV-2 Messenger RNA Vaccine: Real-World Evidence from Italian Adult Population. Vaccines, 9(12), 1494. https://doi.org/10.3390/vaccines9121494







