Safety and Efficacy of the East Coast Fever Muguga Cocktail Vaccine: A Systematic Review
Abstract
1. Introduction
2. Materials and Methods
2.1. Protocol
2.2. Review Research Questions
- What is the safety and efficacy of the Muguga cocktail vaccine?
- What is the safety and efficacy of oxytetracycline?
2.3. Eligibility Criteria
2.4. Scoping Review
2.5. Database Sources
2.6. Search Strategy
2.7. Study Selection
2.8. Data Management
2.9. Data Extraction
2.10. Data Analysis
3. Results
3.1. Efficacy
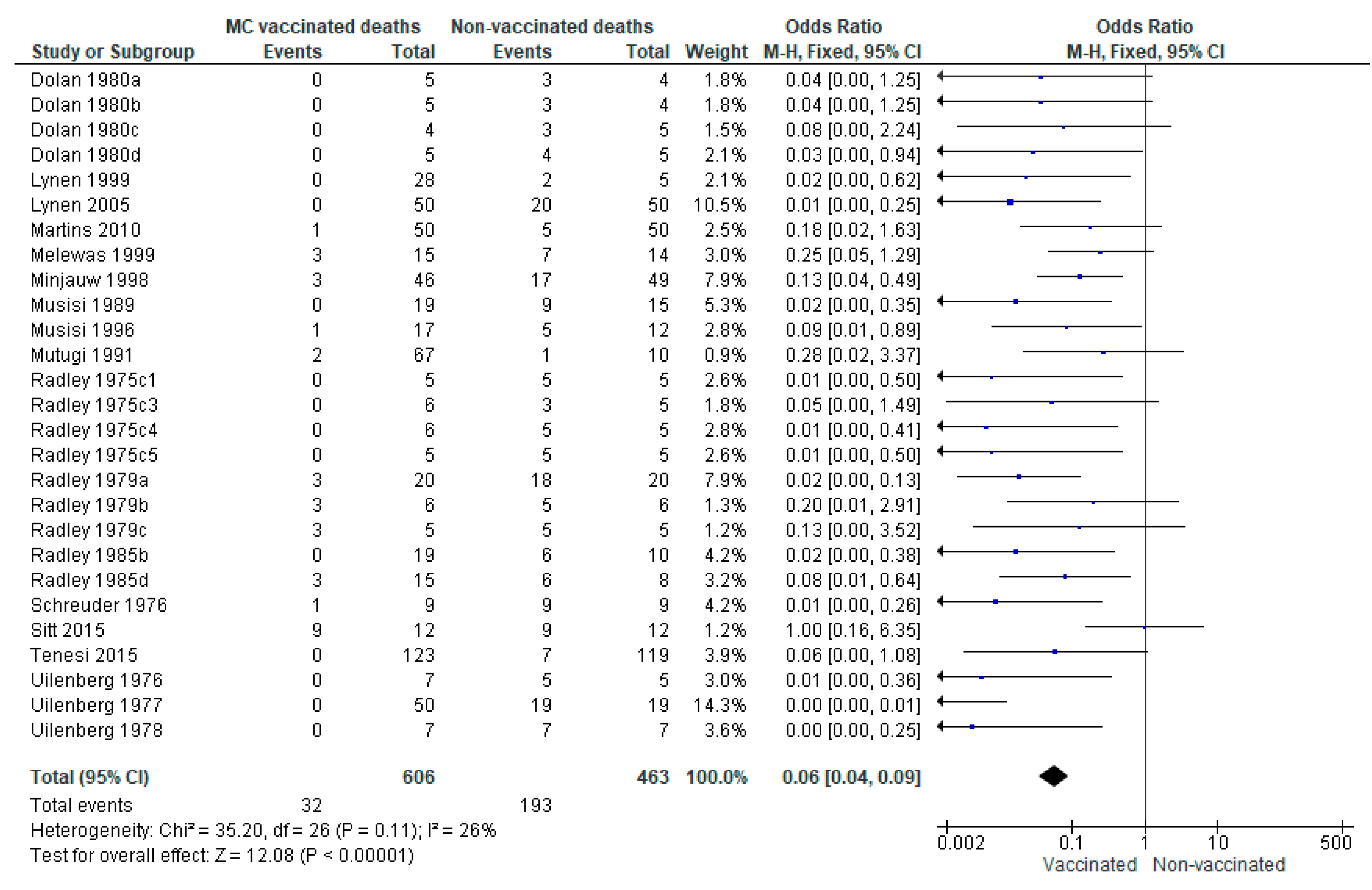
3.1.1. Protection against ECF Mortality in Immunisation Trials
Risk of Bias of Included Mortality Studies
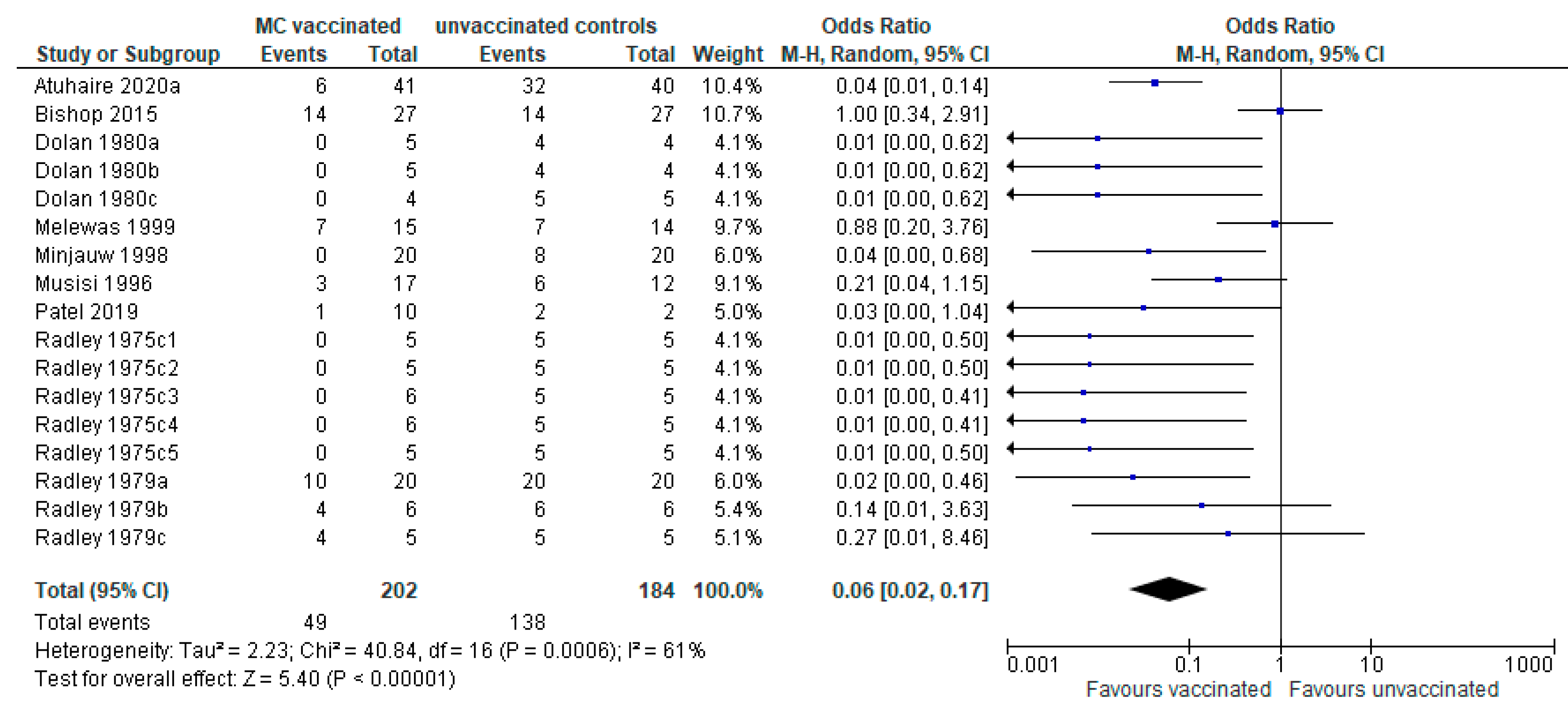
3.1.2. Efficacy in Response to Experimental Challenge
Risk of Bias of Severe Reaction Studies
3.1.3. Efficacy Based on Seroconversion as a Measure of Protection
3.1.4. Efficacy as a Percentage (Quantitative)
3.1.5. Onset of Immunity
3.1.6. Duration of Immunity
3.1.7. Efficacy as a Statement (Qualitative)
3.1.8. Efficacy near Buffalo
3.1.9. Efficacy and Storage Temperature
3.2. Safety
3.2.1. Safety as a Statement (Qualitative)
3.2.2. Shed and Transmission
3.3. OTC Safety and Efficacy
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References and Notes
- Ocaido, M.; Muwazi, R.T.; Opuda, J.A.; Anim, T.; Opuda, J.A. Economic impact of ticks and tick-borne diseases on cattle production systems around Lake Mburo National Park in South Western Uganda. Trop. Anim. Health Prod. 2009, 41, 731–739. [Google Scholar] [CrossRef]
- Onono, J.O.; Wieland, B.; Rushton, J. Constraints to cattle production in a semiarid pastoral system in Kenya. Trop. Anim. Health Prod. 2013, 45, 1415–1422. [Google Scholar] [CrossRef] [PubMed]
- Perry, B.D. The control of East Coast fever of cattle by live parasite vaccination: A science-to-impact narrative. One Health 2016, 2, 103–114. [Google Scholar] [CrossRef]
- Theiler, A. The Immunisation of Cattle against East Coast Fever. Onderstepoort J. Vet. Res. Second Rep. Dir. Vet. Res. 1912, 2, 266–314. [Google Scholar] [CrossRef]
- Nene, V.; Kiara, H.; Lacasta, A.; Pelle, R.; Svitek, N.; Steinaa, L. The biology of Theileria parva and control of East Coast fever—Current status and future trends. Ticks Tick Borne Dis. 2016, 7, 549–564. [Google Scholar] [CrossRef] [PubMed]
- GALVmed East Coast Fever. Available online: https://www.galvmed.org/livestock-and-diseases/livestock-diseases/east-coast-fever/ (accessed on 28 July 2021).
- Dolan, T.T. Dogmas and misunderstandings in East Coast fever. Trop. Med. Int. Health 1999, 4, A3–A11. [Google Scholar] [CrossRef]
- McHardy, N. Multinational research on the use of buparvaquone (Butalex) for the control of theileriosis. In Proceedings of the International Symposium on Mycoplasmosis and Theileriosis, Pendik, Turkey, 11–13 October 1989; pp. 114–126. [Google Scholar]
- Morrison, W.I.; McKeever, D.J. Current status of vaccine development against Theileria parasites. Parasitology 2006, 133, S169–S187. [Google Scholar] [CrossRef]
- Radley, D.E.; Brown, C.G.D.; Burridge, M.J.; Cunningham, M.P.; Kirimi, I.M.; Purnell, R.E.; Young, A.S. East coast fever: 1. Chemoprophylactic immunization of cattle against Theileria parva (Muguga) and five theilerial strains. Vet. Parasitol. 1975, 1, 35–41. [Google Scholar] [CrossRef]
- Radley, D.E.; Young, A.S.; Brown, C.G.D.; Burridge, M.J.; Cunningham, M.P.; Musisi, F.L.; Purnell, R.E. East Coast fever: 2. Cross-Immunity Trials with a Kenya Strain of Theileria Lawrencei. Vet. Parasitol. 1975, 1, 43–50. [Google Scholar] [CrossRef]
- Radley, D.E.; Brown, C.G.D.; Cunningham, M.P.; Kimber, C.D.; Musisi, F.L.; Payne, R.C.; Purnell, R.E.; Stagg, S.M.; Young, A.S. East coast fever: 3. Chemoprophylactic immunization of cattle using oxytetracycline and a combination of theilerial strains. Vet. Parasitol. 1975, 1, 51–60. [Google Scholar] [CrossRef]
- Di Giulio, G.; Lynen, G.; Morzaria, S.; Oura, C.; Bishop, R. Live immunization against East Coast fever-current status. Rev. Trends Parasitol. 2009, 25, 85–92. [Google Scholar] [CrossRef]
- De Castro, J.J. Sustainable tick and tickborne disease control in livestock improvement in developing countries. Vet. Parasitol. 1997, 71, 77–97. [Google Scholar] [CrossRef]
- De Castro, J.J. Reply to comments on the paper “Sustainable tick and tickborne disease control in livestock in developing countries”. Vet. Parasitol. 1998, 77, 213–215. [Google Scholar]
- Berkvens, D.L.; Geysen, D.M.; Brandt, J.R.A. Comments on the paper “Sustainable tick and tickborne disease control in livestock improvement in developing countries”. Vet. Parasitol. 1998, 77, 211–212. [Google Scholar] [CrossRef]
- McKeever, D.J. Live immunisation against Theileria parva: Containing or spreading the disease? Trends Parasitol. 2007, 23, 565–568. [Google Scholar] [CrossRef] [PubMed]
- Principles of Veterianary Vaccine Production. Available online: https://www.oie.int/fileadmin/Home/eng/Health_standards/tahm/1.01.08_VACCINE_PRODUCTION.pdf (accessed on 28 October 2021).
- Peters, A.R.; Toye, P.; Spooner, P.; di Giulio, G.; Lynen, G. Registration of the East Coast Fever Infection and Treatment Method vaccine (Muguga cocktail) in East Africa. Gates Open Res. 2020, 4, 100. [Google Scholar] [CrossRef]
- Cook, D.J.; Mulrow, C.D.; Haynes, R.B. Systematic reviews: Synthesis of best evidence for clinical decisions. Ann. Intern. Med. 1997, 126, 376–380. [Google Scholar] [CrossRef] [PubMed]
- Greenhalgh, T. How to read a paper: Papers that summarise other papers (systematic reviews and meta-analyses). Br. Med. J. 1997, 315, 672–675. [Google Scholar] [CrossRef] [PubMed]
- Vesterinen, H.M.; Sena, E.S.; Egan, K.J.; Hirst, T.C.; Churolov, L.; Currie, G.L.; Antonic, A.; Howells, D.W.; Macleod, M.R. Meta-analysis of data from animal studies: A practical guide. J. Neurosci. Methods 2014, 221, 92–102. [Google Scholar] [CrossRef]
- Sargeant, J.M.; O’Connor, A.M. Introduction to Systematic Reviews in Animal Agriculture and Veterinary Medicine. Zoonoses Public Health 2014, 61, 3–9. [Google Scholar] [CrossRef]
- Pound, P.; Ebrahim, S.; Sandercock, P.; Bracken, M.B.; Roberts, I. Where is the evidence that animal research benefits humans? Br. Med. J. 2004, 328, 514–517. [Google Scholar] [CrossRef]
- Van der Worp, H.B.; Howells, D.W.; Sena, E.S.; Porritt, M.J.; Rewell, S.; O’Collins, V.; Macleod, M.R. Can Animal Models of Disease Reliably Inform Human Studies? PLoS Med. 2010, 7, e1000245. [Google Scholar] [CrossRef]
- De Vries, R.B.M.; Hooijmans, C.R.; Langendam, M.W.; van Luijk, J.; Leenaars, M.; Ritskes-Hoitinga, M.; Wever, K.E. A protocol format for the preparation, registration and publication of systematic reviews of animal intervention studies. Evid.-Based Preclin. Med. 2015, 1, 1–9. [Google Scholar] [CrossRef]
- Green, S.; Higgins, J. Preparing a Cochrane Review. Available online: https://handbook-5-1.cochrane.org/chapter_2/2_preparing_a_cochrane_review.htm (accessed on 29 July 2021).
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. Br. Med. J. 2021, 372, 1–9. [Google Scholar] [CrossRef]
- Grindlay, D.J.C.; Brennan, M.L.; Dean, R.S. Searching the veterinary literature: A comparison of the coverage of veterinary journals by nine bibliographic databases. J. Vet. Med. Educ. 2012, 39, 404–412. [Google Scholar] [CrossRef]
- Towards the Digital Library: Findings of an Investigation to Establish the Current Status of University Libraries in Africa; International Network for the Availability of Scientific Publications (INASP); Available online: https://www.inasp.info/sites/default/files/2018-04/towards_the_digital_library_full.pdf (accessed on 20 October 2021).
- Tawfik, G.M.; Dila, K.A.S.; Mohamed, M.Y.F.; Tam, D.N.H.; Kien, N.D.; Ahmed, A.M.; Huy, N.T. A step by step guide for conducting a systematic review and meta-analysis with simulation data. Trop. Med. Health 2019, 47, 1–9. [Google Scholar] [CrossRef]
- Review Manager (RevMan), version 5.4.1; The Nordic Cochrane Centre, The Cochrane Collaboration: Copenhagen, Denmark, 2020.
- Thrusfield, M. Veterinary Epidemiology, 3rd ed.; Blackwell Science Ltd.: Oxford, UK, 2005. [Google Scholar]
- European Commission EudraLex-Volume-6-Notice to Applicants and Regulatory Guidelines for Medicinal Products for Veterinary Use. Available online: https://ec.europa.eu/health/documents/eudralex/vol-6_en (accessed on 4 August 2021).
- Hooijmans, C.R.; Rovers, M.M.; De Vries, R.B.; Leenaars, M.; Ritskes-Hoitinga, M.; Langendam, M.W. SYRCLE’s risk of bias tool for animal studies. BMC Med. Res. Methodol. 2014, 14, 1–9. [Google Scholar] [CrossRef]
- Higgins, J.P.; Altman, D.G.; Gotzsche, P.C.; Juni, P.; Moher, D.; Oxman, A.D.; Savovic, J.; Schulz, K.F.; Weeks, L.; Sterne, J.A.; et al. The Cochrane Collaboration’s tool for assessing risk of bias in randomised trials. Br. Med. J. 2011, 343, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Uilenberg, G.; Schreuder, B.E.C.; Mpangala, C.; Silayo, R.S.; Tondeur, W.; Tatchell, R.J.; Sanga, H.J.N. Immunization Against East Coast fever. In Tick-Borne Diseases and Their Vectors, Proceedings of an International Conference, Edinburgh, UK, 27 September–1 October 1976; Wilde, J.K.H., Ed.; Edinburgh University Press: Edinburgh, UK, 1978; pp. 307–314. [Google Scholar]
- Radley, D.E.; Young, A.S.; Grootenhuis, J.G.; Cunningham, M.P.; Dolan, T.T.; Morzaria, S.P. Further studies on the immunization of cattle against Theileria lawrencei by infection and chemoprophylaxis. Vet. Parasitol. 1979, 5, 117–128. [Google Scholar] [CrossRef]
- Dolan, T.T.; Radley, D.E.; Brown, C.G.D.; Cunningham, M.P.; Morzaria, S.P.; Young, A.S. East Coast fever: 4. Further Studies on the Protection of Cattle Immunized with a Combination of Theilerial Strains. Vet. Parasitol. 1980, 6, 325–332. [Google Scholar] [CrossRef]
- Radley, D.E. East Coast fever immunization—Field trials in Malawi. In Proceedings of the Immunization Against Theileriosis in Africa, Nairobi, Kenya, 1–5 October 1984; Irvin, A.D., Ed.; ILRAD: Nairobi, Kenya, 1985; pp. 58–61. [Google Scholar]
- Nambota, A. Immunization against theileriosis in the Southern Province of Zambia. In Theileriosis in Eastern, Central and Southern Africa, Proceedings of a Workshop on East Coast Fever Immunization, Lilongew, Malawi, 20–22 September 1988; Dolan, T.T., Ed.; The International Laboratory for Research on Animal Diseases: Nairobi, Kenya, 1989; pp. 87–89. [Google Scholar]
- Schreuder, B.E.C.; Silayo, R.S.; Uilenberg, G.; Mpangala, C.; Sanga, H.J.N. Studies on Theileriidae (Sporozoa) in Tanzania. VI. Second Field Trial on Immunization against Cattle Theileriosis. Tropenmed. Parasitol. 1976, 27, 26–34. [Google Scholar]
- Uilenberg, G.; Silayo, R.S.; Mpangala, C.; Tondeur, W.; Tatchell, R.J.; Sanga, H.J.N. Studies on Theileriidae (Sporozoa) in Tanzania. X. A Large-scale Field Trial on Immunization Against Cattle Theileriosis. Tropenmed. Parasitol. 1977, 28, 499–506. [Google Scholar] [PubMed]
- Sitt, T.; Poole, E.J.; Ndambuki, G.; Mwaura, S.; Njoroge, T.; Omondi, G.P.; Mutinda, M.; Mathenge, J.; Prettejohn, G.; Morrison, W.I.; et al. Exposure of vaccinated and naive cattle to natural challenge from buffalo-derived Theileria parva. Int. J. Parasitol. Parasites Wildl. 2015, 4, 244–251. [Google Scholar] [CrossRef]
- Anon. Classification of Theileria parva reactions in cattle. In Theileriosis in Eastern, Central and Southern Africa, Proceedings of a Workshop on East Coast Fever Immunization, Lilongew, Malawi, 20–22 September 1988; Dolan, T.T., Ed.; English Press Ltd.: Nairobi, Kenya, 1989; pp. 187–188. [Google Scholar]
- Atuhaire, D.K.; Muleya, W.; Mbao, V.; Bazarusanga, T.; Gafarasi, I.; Salt, J.; Namangala, B.; Musoke, A.J. Sequence diversity of cytotoxic T cell antigens and satellite marker analysis of Theileria parva informs the immunization against East Coast fever in Rwanda. Parasit. Vectors 2020, 13, 1–20. [Google Scholar] [CrossRef]
- Rowlands, G.J.; Musoke, A.J.; Morzaria, S.P.; Nagda, S.M.; Ballingall, K.T.; McKeever, D.J. A statistically derived index for classifying East Coast fever reactions in cattle challenged with Theileria parva under experimental conditions. Parasitology 2000, 120, 371–381. [Google Scholar] [CrossRef]
- Melewas, J.; Majaliwa, M.; Lynen, L. ECF immunisation in Tanzania. In Proceedings of the FAO/ILRI/OAU-IBAR Workshop “Live Vaccines for Theileria Parva: Deployment in Eastern, Central and Southern Africa”, Nairobi, Kenya, 10–12 March 1997; Morzaria, S., Williamson, S., Eds.; ILRI: Nairobi, Kenya, 1999; pp. 13–20. [Google Scholar]
- Bishop, R.; Hemmink, J.; Morrison, W.; Weir, W.; Toye, P.; Sitt, T.; Spooner, P.; Musoke, A.; Skilton, R.; Odongo, D. The African buffalo parasite Theileria. sp. (buffalo) can infect and immortalize cattle leukocytes and encodes divergent orthologues of Theileria parva antigen genes. Int. J. Parasitol. Parasites Wildl. 2015, 4, 333–342. [Google Scholar] [CrossRef] [PubMed]
- Anon. FAO-1 composite stabilate. Evaluation of efficacy at 1:80 and 1:100 dilutions and treatment with 30% oxytetracycline. 1998. [Google Scholar]
- Anon. FAO-1-2 composite stabilate. Evaluation of efficacy at 1:80 direct dilution and treatment with 30% oxytetracycline. 1999. [Google Scholar]
- Kiraithe, Z.N. Efficacy of the Muguga Cocktail East Coast Fever Vaccine at a Cattle-Buffalo Interface in Laikipia County. Master’s Thesis, University of Nairobi, Nairobi, Kenya, 2015. [Google Scholar]
- Kazungu, Y.E.M.; Mwega, E.; Neselle, M.O.; Sallu, R.; Kimera, S.I.; Gwakisa, P. Incremental effect of natural tick challenge on the infection and treatment method-induced immunity against T. parva in cattle under agro-pastoral systems in Northern Tanzania. Ticks Tick Borne Dis. 2015, 6, 587–591. [Google Scholar] [CrossRef] [PubMed]
- Martins, S.B.; Di Giulio, G.; Lynen, G.; Peters, A.; Rushton, J. Assessing the impact of East Coast Fever immunisation by the infection and treatment method in Tanzanian pastoralist systems. Prev. Vet. Med. 2010, 97, 175–182. [Google Scholar] [CrossRef]
- Mbassa, G.K.; Kweka, L.E.; Dulla, P.N. Immunization against East Coast Fever in field cattle with low infectivity Theileria parva stabilate—Preliminary assessment. Vet. Parasitol. 1998, 77, 41–48. [Google Scholar] [CrossRef]
- Patel, E.; Mwaura, S.; Di Giulio, G.; Cook, E.A.J.; Lynen, G.; Toye, P. Infection and treatment method (ITM) vaccine against East Coast fever: Reducing the number of doses per straw for use in smallholder dairy herds by thawing, diluting and refreezing already packaged vaccine. BMC Vet. Res. 2019, 15. [Google Scholar] [CrossRef]
- Turasha, G.K. Pilot Delivery of ECF Vaccine in Smallholder Dairy Production Systems in Kenya; Project Report; VetAID Kenya: Nairobi, Kenya, 2011. [Google Scholar]
- Tenesi, M.G. An Assessment of the Efficacy of Infection and Treatment Method against East Coast Fever and the Financial and Economic Benefits in Narok County of Kenya. Ph.D. Thesis, University of Nairobi, Nairobi, Kenya, 2015. [Google Scholar]
- Wesonga, F.; Mugambi, J.M.; Karanja-Lumumba, T. East Coast fever (ECF) Vaccine Efficacy Follow Up Studies Under OSRO/KEN/103/SWE Project in Selected Diary Areas and Laikipia. 2013. [Google Scholar]
- Dohoo, I.R.; Martin, S.W.; Stryhn, H. Veterinary Epidemiologic Research, 2nd ed.; Prince Edward Island: Charlottetown, PE, Canada, 2009. [Google Scholar]
- Lynen, G.; Yrjö-Koskinen, A.E.; Bakuname, C.; Di Giulio, G.; Mlinga, N.; Khama, I.; Hanks, J.; Taylor, N.M.; James, A.D.; McKeever, D.; et al. East Coast fever immunisation field trial in crossbred dairy cattle in Hanang and Handeni districts in northern Tanzania. Trop. Anim. Health Prod. 2012, 44, 567–572. [Google Scholar] [CrossRef] [PubMed]
- Magwisha, H.B.; Ruheta, M.R.; Mbwambo, H.A.; Ranga, E.E.; Makungu, L.S. The Potential of East Coast Fever (ECF) Vaccine on Improved and Indigenous Cattle in Tanzania. J. Commonw. Vet. Assoc. 2011, 27, 87–92. [Google Scholar]
- Nsubuga-Mutaka, R.C.; Otim, C.P.; Kakaire-Nyende, M.; Erima, S.; Ongyera, S. Immunization against east coast fever by infection and treatment method in Uganda. Uganda J. Agric. Sci. 2000, 5, 1–2. [Google Scholar]
- Nsubuga-Mutaka, R. ECF immunisation in Uganda. In Proceedings of the FAO/ILRI/OAU-IBAR Workshop “Live Vaccines for Theileria Parva: Deployment in Eastern, Central and Southern Africa”, Nairobi, Kenya, 10–12 March 1997; Morzaria, S.P., Williamson, S., Eds.; ILRI: Nairobi, Kenya, 1999; pp. 21–23. [Google Scholar]
- Anon Infectivity/viability test of FAO-1 stabilate after 2.5 years storage at CTTBD, Lilongwe, Malawi, July 1999. 1999.
- Oura, C.A.L.; Bishop, R.; Wampande, E.M.; Lubega, G.W.; Tait, A. The persistence of component Theileria parva stocks in cattle immunized with the “Muguga cocktail” live vaccine against East Coast fever in Uganda. Parasitology 2004, 129, 27–42. [Google Scholar] [CrossRef] [PubMed]
- Akoolo, L.; Pellé, R.; Saya, R.; Awino, E.; Nyanjui, J.; Taracha, E.L.N.; Kanyari, P.; Mwangi, D.M.; Graham, S.P. Evaluation of the recognition of Theileria parva vaccine candidate antigens by cytotoxic T lymphocytes from Zebu cattle. Vet. Immunol. Immunopathol. 2008, 121, 216–221. [Google Scholar] [CrossRef]
- Anon Applying ITM Immunisation in Tanzania using the FAO-1 Vaccine Batch (1998–2007). 2007.
- Homewood, K.; Trench, P.; Randall, S.; Lynen, G.; Bishop, B. Livestock health and socio-economic impacts of a veterinary intervention in Maasailand: Infection-and-treatment vaccine against East Coast fever. Agric. Syst. 2006, 89, 248–271. [Google Scholar] [CrossRef]
- Mutugi, J.; Njuguna, L.; Musisi, F.; Morzaria, S.P.; Spooner, P.R.; Duchateau, L. Determining a Safe and Protective Immunizing Dose for the FAO-1 Theileria Parva Composite Stabilate for Use in Field Vaccinations against East Coast Fever—FAO Technical Report AG:GCP/RAF/291/DEN & GCP/RAF/293/BEL; FAO: Rome, Italy, 1997. [Google Scholar]
- Patel, E.; Mwaura, S.; Kiara, H.; Morzaria, S.; Peters, A.; Toye, P. Production and dose determination of the Infection and Treatment Method (ITM) Muguga cocktail vaccine used to control East Coast fever in cattle. Ticks Tick Borne Dis. 2016, 7, 306–314. [Google Scholar] [CrossRef]
- Mutugi, J. Review of safety and effiacy of ECF-ITM Muguga cocktail.
- Steinaa, L.; Svitek, N.; Awino, E.; Saya, R.; Toye, P. Cytotoxic T lymphocytes from cattle sharing the same MHC class I haplotype and immunized with live Theileria parva sporozoites differ in antigenic specificity. BMC Res. Notes 2018, 11, 44. [Google Scholar] [CrossRef]
- Pelle, R.; Graham, S.P.; Njahira, M.N.; Osaso, J.; Saya, R.M.; Odongo, D.O.; Toye, P.G.; Spooner, P.R.; Musoke, A.J.; Mwangi, D.M.; et al. Two Theileria parva CD8 T cell antigen genes are more variable in buffalo than cattle parasites, but differ in pattern of sequence diversity. PLoS ONE 2011, 6, e19015. [Google Scholar] [CrossRef]
- Obara, I.; Ulrike, S.; Musoke, T.; Spooner, P.R.; Jabbar, A.; Odongo, D.; Kemp, S.; Silva, J.C.; Bishop, R.P. Molecular evolution of a central region containing B cell epitopes in the gene encoding the p67 sporozoite antigen within a field population of Theileria parva. Parasitol. Res. 2015, 114, 1729–1737. [Google Scholar] [CrossRef] [PubMed]
- Lynen, G.; di Giulio, G.; Homewood, K.; Reid, R.; Mwilawa, A. Deployment of a Live ECF Vaccine in Pastoral Areas: Lessons Learned from Tanzania. In The Role of Biotechnology in Animal Agriculture to Address Poverty in Africa: Opportunities and Challenges, Proceedings of the 4th All African Conference on Animal Agriculture and the 31st Annual Meeting of the Tanzania Society for Animal Production (TSAP), Arusha, Tanzania, 20–24 September 2005; All Africa Society for Animal Production (AASAP): Addis Ababa, Ethiopia, 2005; pp. 193–201. [Google Scholar]
- Turasha Technical Meeting on the Infection and Treatment Method of East Coast fever Immunization and the Way Forward in Kenya; Report to Meeting at Naivasha Country Club, Kenya, 7 March 2005.
- Atuhaire, D.K.; Lieberman, D.; Marcotty, T.; Musoke, A.J.; Madan, D. An alternative cold chain for storing and transporting East Coast fever vaccine. Vet. Parasitol. 2020, 288, 109304. [Google Scholar] [CrossRef] [PubMed]
- ILRI Titration of the FAO-1 stabilate. 1996.
- Mbyuzi, A.O.; Komba, E.V.G.; Magwisha, H.B.; Salum, M.R.; Kafiriti, E.M.; Malamla, L.J. Preliminary evidence of vertical transmission of Theileria parva sporozoites from ECF immunized cows to offspring in southern Tanzania. Res. Opin. Anim. Vet. Sci 2013, 3, 92–100. [Google Scholar]
- Amzati, G.S.; Djikeng, A.; Odongo, D.O.; Nimpaye, H.; Sibeko, K.P.; Muhigwa, J.-B.B.; Madder, M.; Kirschvink, N.; Marcotty, T. Genetic and antigenic variation of the bovine tick-borne pathogen Theileria parva in the Great Lakes region of Central Africa. Parasit. Vectors 2019, 12, 19. [Google Scholar] [CrossRef] [PubMed]
- Chatanga, E.; Hayashida, K.; Muleya, W.; Kusakisako, K.; Moustafa, M.A.M.; Salim, B.; Katakura, K.; Sugimoto, C.; Nonaka, N.; Nakao, R. Genetic Diversity and Sequence Polymorphism of Two Genes Encoding Theileria parva Antigens Recognized by CD8+ T Cells among Vaccinated and Unvaccinated Cattle in Malawi. Pathogens 2020, 9, 334. [Google Scholar] [CrossRef]
- De Deken, R.; Martin, V.; Saido, A.; Madder, M.; Brandt, J.; Geysen, D. An outbreak of East Coast Fever on the Comoros: A consequence of the import of immunised cattle from Tanzania? Vet. Parasitol. 2007, 143, 245–253. [Google Scholar] [CrossRef]
- Mwega, E.; Hasan, S.D.; Njahira, M.; Rukambile, E.; Skilton, R.; Gwakisa, P. Genetic and antigenic diversity of Theileria parva in cattle in Eastern and Southern zones of Tanzania. A study to support control of East Coast fever. Parasitology 2015, 142, 698–705. [Google Scholar] [CrossRef]
- Geysen, D.; Bishop, R.; Skilton, R.; Dolan, T.T.; Morzaria, S. Molecular epidemiology of Theileria parva in the field. Trop. Med. Int. Health 1999, 4, A21–A27. [Google Scholar] [CrossRef]
- Gwakisa, P.; Kindoro, F.; Mwega, E.; Kimera, S.; Obara, I.; Ahmed, J.; Clausen, P.; Bishop, R. Monitoring vaccinated cattle for induction and longevity of persistent tick-transmissible infection: Implications for wider deployment of live vaccination against East Coast fever in Tanzania. Transbound. Emerg. Dis. 2020, 67, 79–87. [Google Scholar] [CrossRef]
- Kerario, I.I.; Chenyambuga, S.W.; Mwega, E.D.; Rukambile, E.; Simulundu, E.; Simuunza, M.C. Diversity of two Theileria parva CD8+ antigens in cattle and buffalo-derived parasites in Tanzania. Ticks Tick Borne Dis. 2019, 10, 1003–1017. [Google Scholar] [CrossRef] [PubMed]
- Magulu, E.; Kindoro, F.; Mwega, E.; Kimera, S.; Shirima, G.; Gwakisa, P. Detection of carrier state and genetic diversity of Theileria parva in ECF-vaccinated and naturally exposed cattle in Tanzania. Vet. Parasitol. Reg. Stud. Rep. 2019, 17, 100312. [Google Scholar] [CrossRef]
- Nambota, A.M.; Lovelace, C.E.A.; Chitambo, H.; Kakuda, T.; Sugimoto, C.; Onuma, M. Characterization of Some Theileria parva Stocks from Zambia Using Monoclonal Antibodies. J. Vet. Med. Sci. 1997, 59, 1–4. [Google Scholar] [CrossRef]
- Oura, C.A.L.; Bishop, R.; Asiimwe, B.B.; Spooner, P.; Lubega, G.W.; Tait, A. Theileria parva live vaccination: Parasite transmission, persistence and heterologous challenge in the field. Parasitology 2007, 134, 1205–1213. [Google Scholar] [CrossRef]
- Rukambile, E.J.; Mbassa, G.K.; Luziga, C.D.; Mwaijibe, L.R.; Magwisha, H.B.; Mamiro, K.A.; Makondo, Z.E. Molecular epidemiology of Theileria parva in Eastern Zone of Tanzania. Tanzan. Vet. J. 2014, 29, 27–34. [Google Scholar]
- Lynen, G.; Melewas, J.; Majaliwa, K.; Bakuname, C.; di Giulio, G. The Use of a 30% Formulation of Oxytetracycline Long-Acting in East Coast fever Immunisation in Tanzania. In Animal Health and Production for Development, Proceedings of IX International Conference of Associations of Institutions of Tropical Veterinary Medicine, Harare, Zimbabwe, 14–18 September 1998; Mukaratirwa, S., Obwolo, M.J., Eds.; University of Utrecht Press: Utrecht, The Netherlands, 1999; Volume 1, pp. 367–375. [Google Scholar]
- Clarke, C.R.; Wang, Z.; Cudd, L.; Burrows, G.E.; Kirkpatrick, J.G.; Brown, M.D. Pharmacokinetics of two long-acting oxytetracycline products administered subcutaneously and intramuscularly. J. Vet. Pharmacol. Ther. 1999, 22, 65–67. [Google Scholar] [CrossRef] [PubMed]
- Di Giulio, G.; Lynen, G.; Ulicky, E.; Katende, J.; Morzaria, S. Use of Two Different Dose Rates of Oxytetracycline in East Coast fever Immunisation in Tanzania. In Proceedings of the 3rd International Conference on Tick and Tick-Borne Pathogens, Bratislava, Slovakia, 30 August–3 September 1999; Institute of Zoology: Bratislava, Slovakia, 2000; pp. 121–123. [Google Scholar]
- Rashid, A.; Rasheed, K.; Akhtar, M. Factors Influencing Vaccine Efficacy—A General Review. J. Anim. Plant Sci. 2009, 19, 22–25. [Google Scholar]
- Conrad, P.A.; Ole-Moiyoi, O.K.; Baldwin, C.L.; Dolan, T.T.; O’Callaghan, C.J.; Njamunggehr, R.E.G.; Grootenhuis, J.G.; Stagg, D.A.; Leitch, B.L.; Young, A.S. Characterization of buffalo-derived theilerial parasites with monoclonal antibodies and DNA probes. Parasitology 1989, 98, 179–188. [Google Scholar] [CrossRef] [PubMed]
- Oura, C.A.L.; Tait, A.; Asiimwe, B.; Lubega, G.W.; Weir, W. Theileria parva genetic diversity and haemoparasite prevalence in cattle and wildlife in and around Lake Mburo National Park in Uganda. Parasitol. Res. 2011, 108, 1365–1375. [Google Scholar] [CrossRef] [PubMed]
- Hemmink, J.D.; Sitt, T.; Pelle, R.; de Klerk-Lorist, L.M.; Shiels, B.; Toye, P.G.; Morrison, W.I.; Weir, W. Ancient diversity and geographical sub-structuring in African buffalo Theileria parva populations revealed through metagenetic analysis of antigen-encoding loci. Int. J. Parasitol. 2018, 48, 287–296. [Google Scholar] [CrossRef] [PubMed]
- Allan, F.K.; Jayaraman, S.; Paxton, E.; Sindoya, E.; Kibona, T.; Fyumagwa, R.; Mramba, F.; Torr, S.J.; Hemmink, J.D.; Toye, P.; et al. Antigenic Diversity in Theileria parva Populations From Sympatric Cattle and African Buffalo Analyzed Using Long Read Sequencing. Front. Genet. 2021, 12, 684127. [Google Scholar] [CrossRef]
- Head, J.R.; Vos, A.; Blanton, J.; Müller, T.; Chipman, R.; Pieracci, E.G.; Cleaton, J.; Wallace, R. Environmental distribution of certain modified live-virus vaccines with a high safety profile presents a low-risk, high-reward to control zoonotic diseases. Nat. Sci. Rep. 2019, 9, 1–12. [Google Scholar] [CrossRef]
- Boutron, I.; Page, M.J.; Higgins, J.P.; Altman, D.G.; Lundh, A.; Hróbjartsson, A. Considering bias and conflicts of interest among the included studies. In Cochrane Handbook for Systematic Reviews of Interventions, version 6.2; Higgins, J., Thomas, J., Chandler, J., Cumpston, M., Li, T., Page, M., Welch, V., Eds.; The Cochrane Collaboration: Cochrane, AB, Canada, 2021. [Google Scholar]
- Sterne, J.A.; Sutton, A.J.; Ioannidis, J.P.; Terrin, N.; Jones, D.R.; Lau, J.; Carpenter, J.; Rucker, G.; Harbord, R.M.; Schmid, C.H.; et al. Recommendations for examining and interpreting funnel plot asymmetry in meta-analyses of randomised controlled trials. Br. Med. J. 2011, 342, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Kilkenny, C.; Parsons, N.; Kadyszewski, E.; Festing, M.F.W.; Cuthill, I.C.; Fry, D.; Hutton, J.; Altman, D.G. Survey of the Quality of Experimental Design, Statistical Analysis and Reporting of Research Using Animals. PLoS ONE 2009, 4, e7824. [Google Scholar] [CrossRef] [PubMed]
- Lankinen, K.S.; Pastila, S.; Terhi, K.; Nohynek, H.; Mäkelä, P.H.; Olin, P. Vaccinovigilance in Europe—Need for timeliness, standardization and resources. Bull. World Health Organ. 2004, 82, 828–835. [Google Scholar] [PubMed]
- Clements, C.J.; Larsen, G.; Jodar, L. Technologies that make administration of vaccines safer. Vaccine 2004, 22, 2054–2058. [Google Scholar] [CrossRef]
- Kivaria, F.M.; Ruheta, M.R.; Mkonyi, P.A.; Malamsha, P.C. Epidemiological aspects and economic impact of bovine theileriosis (East Coast fever) and its control: A preliminary assessment with special reference to Kibaha district, Tanzania. Vet. J. 2007, 173, 384–390. [Google Scholar] [CrossRef]

| PICO Scheme | Question Elements |
|---|---|
| Population | Cattle |
| Intervention | Immunisation with the Muguga cocktail ITM vaccine |
| Comparison | Nonimmunised (control) cattle |
| Outcome | Efficacy of Muguga cocktail vaccination (demonstrated as reduction in mortality and/or reduction in severe reactors in immunised compared to nonimmunised controls) and safety of Muguga cocktail and OTC (demonstrated by lack or local or systemic reactions in animals or the environment (including nontarget animals)) |
| Search Term | Database | Number of Results |
|---|---|---|
| ECF ITM vaccine | Web of Science | 12 |
| PubMed | 12 | |
| CAB Direct | 13 | |
| African Journals Online | 1 | |
| Muguga Cocktail | Web of Science | 24 |
| PubMed | 26 | |
| CAB Direct | 27 | |
| African Journals Online | 4 | |
| ECF Muguga vaccine | Web of Science | 14 |
| PubMed | 16 | |
| CAB Direct | 26 | |
| African Journals Online | 5 | |
| ECF Muguga Cocktail | Web of Science | 11 |
| PubMed | 13 | |
| CAB Direct | 12 | |
| African Journals Online | 3 | |
| ECF ITM vaccine OR Muguga cocktail | Web of Science | 30 |
| PubMed | 33 | |
| CAB Direct | 36 | |
| African Journals Online | 0 | |
| Google Scholar | 296 |
| Domain | Criteria |
|---|---|
| Date range | All |
| Geographical scope | Global |
| Type | Peer-reviewed journal articles, research reports including published and unpublished field studies, theses, conference proceedings. Challenge trials, natural infections, and observational studies. |
| Specific details | Reported efficacy and safety of use of ECF ITM (Muguga cocktail) vaccine |
| Exclusions | Abstract or full text unavailable |
| Language | English only |
| Issue | Parameter |
|---|---|
| Efficacy | Efficacy (quantitative) demonstrated by mortality rates |
| Efficacy in response to challenge (quantitative) | |
| Efficacy based on Seroconversion as a measure of protection | |
| Efficacy as a percentage | |
| Onset of immunity | |
| Duration of immunity | |
| Efficacy (qualitative) | |
| Efficacy in proximity to buffalo | |
| Efficacy and storage temperature | |
| Safety | Safety (qualitative) |
| Shed and transmission | |
| Safety and efficacy of OTC | Safety and efficacy of OTC |
| Study | Description of Safety |
|---|---|
| Anon 2007 [68] | Reported that vaccine FAO-1 was accepted as safe in Tanzania. |
| Anon 1998 [50] | The study compared 1:80 and 1:100 dilutions of FAO-1 with OTC LA 30 mg/kg and showed that both dilutions were very safe for field conditions, with no reactors encountered. |
| Anon 1999 [51] | Negligible reactors to either vaccine batch FAO-1 or FAO-2 with a 1:80 dilution and OTC LA 30mg/kg and were both confirmed as being safe. |
| ILRI 1996 [79] | Four groups of two cattle were immunised with FAO-1 vaccine dilutions of 1:10, 1:20, 1:40, and 1:80, with concurrent 20 mg/kg OTC (Terramycin LA 20%), followed by homologous challenge of all immunised as well as two control animals. None of the vaccinated animals underwent severe reactions, whereas the controls both reacted severely. It was concluded that even doses of 1:10 were safe. |
| Mbyuzi et al., 2013 [80] | Observed mortality of calves born from immunised dams and reported the requirement for further safety evaluation and verification. |
| Mutugi et al., 1997 [70] | Reported a 1:80 dose of FAO-1 to be safe, in terms of survival and few reactions. |
| Mutugi [72] | A review of the safety of the FAO-1 vaccine stated that “the refinement of the vaccine used has given greater confidence that the ITM procedure is safe and does not compromise productivity”. The review reported that the majority of the 37,000 cattle immunised in Uganda (1998–2007) showed inapparent or only mild reactions, with 2.7% (1000)undergoing severe reactions and five deaths (0.01%) associated. The 1:80 FAO-1 dilution used in Kenya “provided a bigger safety margin while at the same time being efficacious (than the 1:60 or 1:100)”, with a “very high degree of safety margin observed in the field”. |
| Patel et al., 2016 [71] | A safety and dose determination study was conducted using a three-stage immunisation and challenge trial (immunizing with ILRI-0804 Muguga cocktail and challenging with FAO-1). Severe reactions were observed in all animals given undiluted vaccine and all given dilutions from 1:10 to 1:80, with the remaining animals undergoing mild or inapparent reactions. All control animals underwent severe reactions to challenge. It was concluded that 1 ml of a 1:100 vaccine dose was considered both safe and efficacious. |
| Tenesi 2015 [58] | Vaccinated cattle were followed for five months, and no reactors were observed, concluding that the Muguga cocktail vaccine was safe. |
| Turasha 2005 [77] | An immunisation trial in Kenya reported a total of 87 reactors in 4000 immunised cattle (2.2%) and 46 mortalities (1.1%). Calf mortality had previously been 20–40%, hence safety and efficacy of the vaccine in Kenyan field conditions were demonstrated. |
| Study | Year of Study | Country | Sample Size | Description of Possible Transmission of MC Vaccine Components |
|---|---|---|---|---|
| Amzati et al., 2019 [81] | 2015 | DRC and Burundi | 480 | Muguga vaccine component alleles present in all AEZs, may be associated with unrestricted movement of cattle in the region |
| Chatanga et al., 2020 [82] | 2018 | Malawi | 254 | Possible spread of vaccine components into unvaccinated cattle |
| De Deken et al., 2007 [83] | 2003–2004 | Comoros | 21 & 6 | Vaccinated can shed Muguga and Kiambu 5 |
| Mwega et al., 2015 [84] | 2012 | Tanzania | 39 | Some field samples genetically related to Muguga vaccine isolate |
| Geysen et al., 1999 [85] | 1996–1997 | Zambia | 12 (isolates) | Possible tick transmission of MC vaccine components |
| Gwakisa et al., 2020 [86] | Tanzania | 410 | Detection of vaccine component in co-grazing unvaccinated, possible vaccine transmission | |
| Kerario et al., 2019 [87] | 2014–2015 | Tanzania | 130 | Indication of genotype sharing as Muguga vaccine isolate clustering despite no vaccination |
| Magulu et al., 2019 [88] | Tanzania | 336 | Muguga vaccine-specific alleles detected in unvaccinated cattle—potentially transmitted by ticks from vaccinated to unvaccinated | |
| Mbyuzi et al., 2013 [80] | 2010–2011 | Tanzania | 768 | 84% ECF cases in calves born to immunised cattle—possibility of vertical transmission of Muguga cocktail vaccine parasites |
| Nambota et al., 1997 [89] | Zambia | 17 (stocks) | Possible introduction of Theileria stocks via MC use | |
| Oura et al., 2004 [66] | Observed persistence of the Kiambu 5 strain in cattle for up to two years in Uganda, in comparison to the Muguga and Serengeti strains which were largely eliminated by three months. There was no evidence of transmission of vaccine stocks to unvaccinated in-contact animals (over a one-year period) | |||
| Oura et al., 2007 [90] | 2000–2004 | Uganda | 36 | Presence of Kiambu 5 stock in unvaccinated cattle, indicating “foreign” vaccine stocks will be introduced to local cattle and tick populations |
| Rukambile et al., 2014 [91] | Tanzania | 102 | Isolation of Muguga stock in areas where no immunisation. Cannot exclude possibility of vaccine derived as extensive nomadism, thus sharing of stocks likely to occur |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Allan, F.K.; Peters, A.R. Safety and Efficacy of the East Coast Fever Muguga Cocktail Vaccine: A Systematic Review. Vaccines 2021, 9, 1318. https://doi.org/10.3390/vaccines9111318
Allan FK, Peters AR. Safety and Efficacy of the East Coast Fever Muguga Cocktail Vaccine: A Systematic Review. Vaccines. 2021; 9(11):1318. https://doi.org/10.3390/vaccines9111318
Chicago/Turabian StyleAllan, Fiona K., and Andrew R. Peters. 2021. "Safety and Efficacy of the East Coast Fever Muguga Cocktail Vaccine: A Systematic Review" Vaccines 9, no. 11: 1318. https://doi.org/10.3390/vaccines9111318
APA StyleAllan, F. K., & Peters, A. R. (2021). Safety and Efficacy of the East Coast Fever Muguga Cocktail Vaccine: A Systematic Review. Vaccines, 9(11), 1318. https://doi.org/10.3390/vaccines9111318






