First Report of Two Cases of Löfgren’s Syndrome after SARS-CoV-2 Vaccination-Coincidence or Causality?
Abstract
:1. Introduction
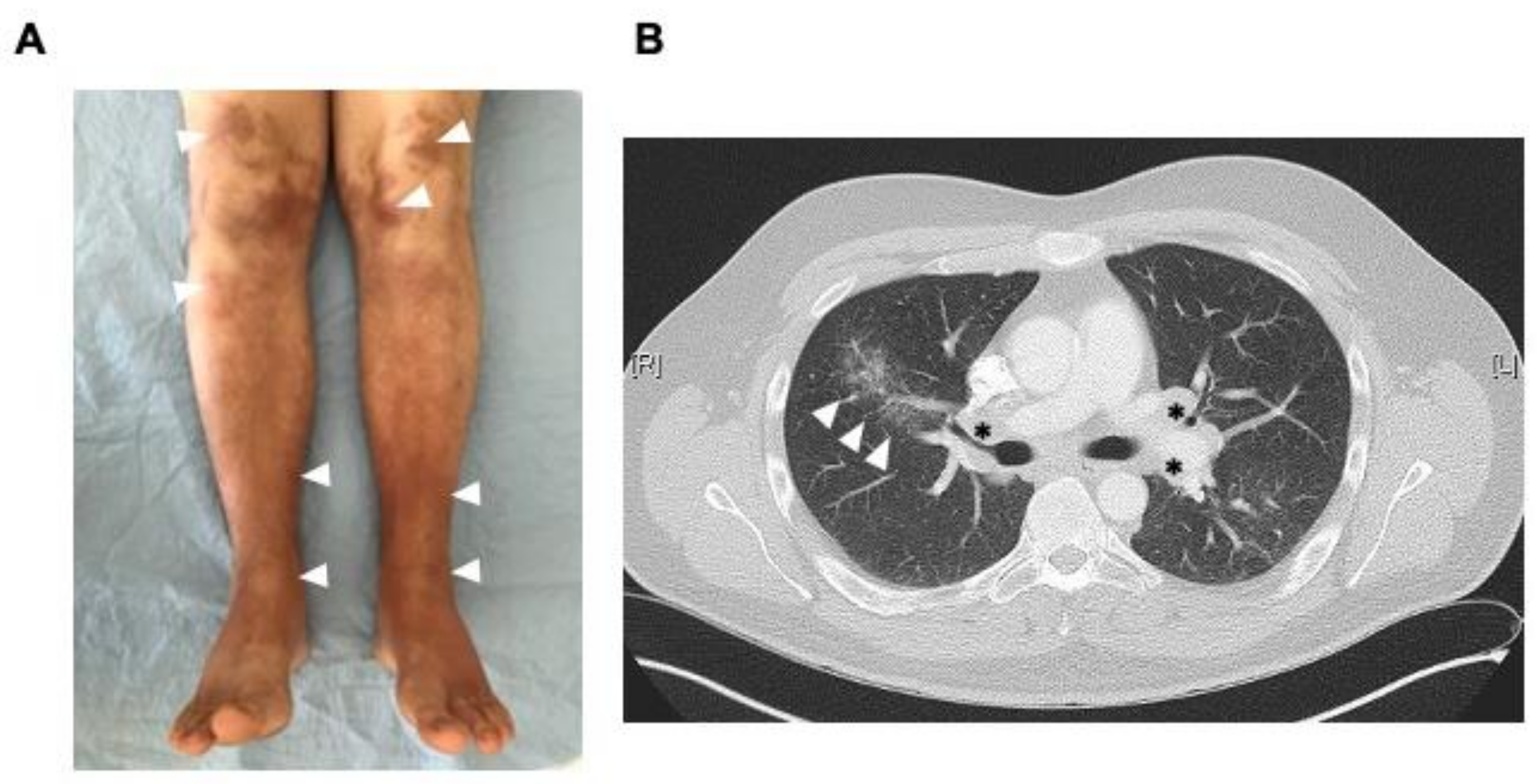
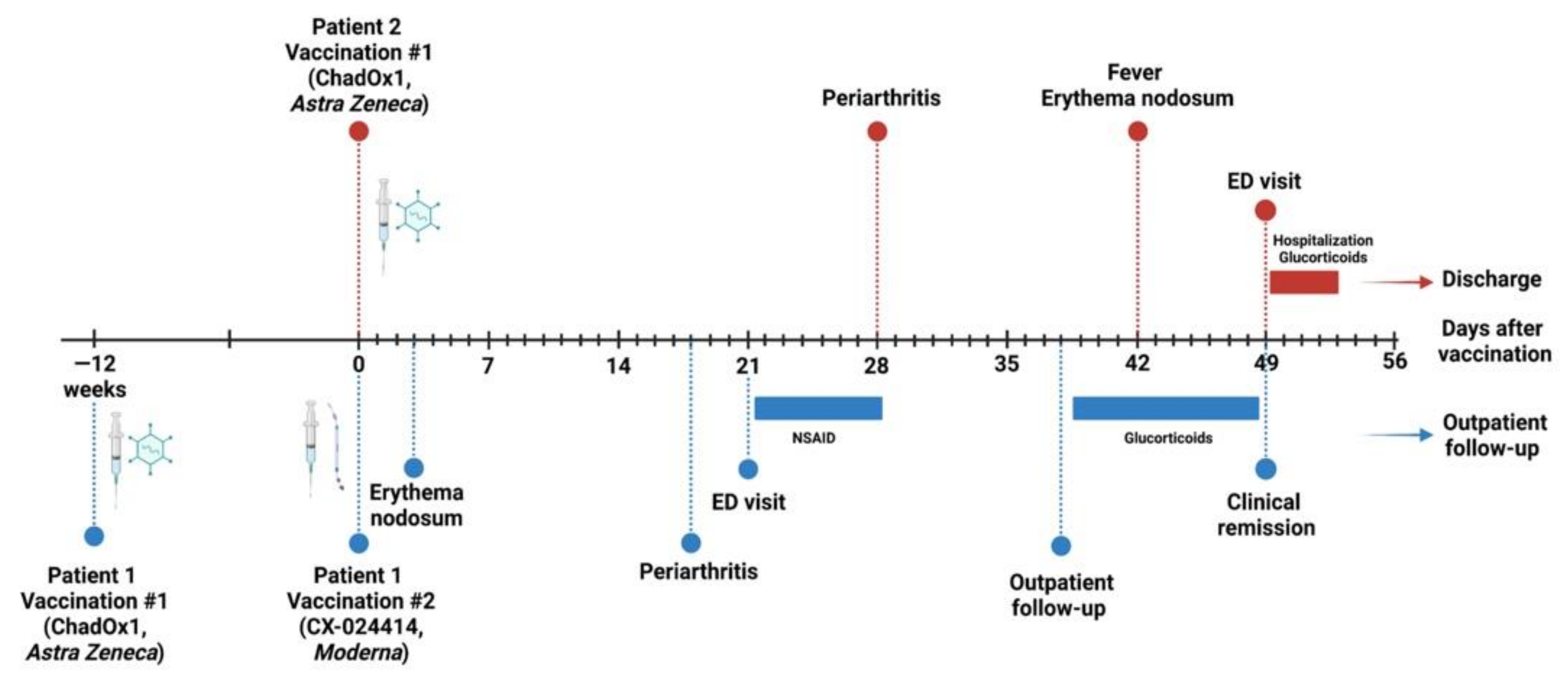
2. Case Reports
3. Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Patel, S.P.; Patel, G.S.; Suthar, J.V. Inside the Story about the Research and Development of COVID-19 Vaccines. Clin. Exp. Vaccine Res. 2021, 10, 154–170. [Google Scholar] [CrossRef] [PubMed]
- Bogdanov, G.; Bogdanov, I.; Kazandjieva, J.; Tsankov, N. Cutaneous Adverse Effects of the Available COVID-19 Vaccines. Clin. Dermatol. 2021, 39, 523–531. [Google Scholar] [CrossRef]
- Pavord, S.; Scully, M.; Hunt, B.J.; Lester, W.; Bagot, C.; Craven, B.; Rampotas, A.; Ambler, G.; Makris, M. Clinical Features of Vaccine-Induced Immune Thrombocytopenia and Thrombosis. N. Engl. J. Med. 2021, 385, 1680–1689. [Google Scholar] [CrossRef] [PubMed]
- Holzmann-Littig, C.; Braunisch, M.C.; Kranke, P.; Popp, M.; Seeber, C.; Fichtner, F.; Littig, B.; Carbajo-Lozoya, J.; Allwang, C.; Frank, T.; et al. COVID-19 Vaccination Acceptance and Hesitancy among Healthcare Workers in Germany. Vaccines 2021, 9, 777. [Google Scholar] [CrossRef]
- Andrade, G. Predictive demographic factors of Covid-19 vaccine hesitancy in Venezuela: A cross-sectional study. Vacunas 2021. [Google Scholar] [CrossRef] [PubMed]
- Mondal, P.; Sinharoy, A.; Su, L. Sociodemographic Predictors of COVID-19 Vaccine Acceptance: A Nationwide US-Based Survey Study. Public Health 2021, 198, 252–259. [Google Scholar] [CrossRef] [PubMed]
- Rosenblum, H.G.; Hadler, S.C.; Moulia, D.; Shimabukuro, T.T.; Su, J.R.; Tepper, N.K.; Ess, K.C.; Woo, E.J.; Mba-Jonas, A.; Alimchandani, M.; et al. Use of COVID-19 Vaccines After Reports of Adverse Events Among Adult Recipients of Janssen (Johnson & Johnson) and MRNA COVID-19 Vaccines (Pfizer-BioNTech and Moderna): Update from the Advisory Committee on Immunization Practices—United States, July 2021. MMWR Morb. Mortal. Wkly. Rep. 2021, 70, 1094–1099. [Google Scholar] [CrossRef] [PubMed]
- Valeyre, D.; Prasse, A.; Nunes, H.; Uzunhan, Y.; Brillet, P.-Y.; Müller-Quernheim, J. Sarcoidosis. Lancet 2014, 383, 1155–1167. [Google Scholar] [CrossRef]
- Crouser, E.D.; Maier, L.A.; Wilson, K.C.; Bonham, C.A.; Morgenthau, A.S.; Patterson, K.C.; Abston, E.; Bernstein, R.C.; Blankstein, R.; Chen, E.S.; et al. Diagnosis and Detection of Sarcoidosis. An Official American Thoracic Society Clinical Practice Guideline. Am. J. Respir. Crit. Care Med. 2020, 201, e26–e51. [Google Scholar] [CrossRef]
- Visser, H.; Vos, K.; Zanelli, E.; Verduyn, W.; Schreuder, G.M.T.; Speyer, I.; Breedveld, F.C.; Hazes, J.M.W. Sarcoid Arthritis: Clinical Characteristics, Diagnostic Aspects, and Risk Factors. Ann. Rheum. Dis. 2002, 61, 499–504. [Google Scholar] [CrossRef] [Green Version]
- Grunewald, J.; Eklund, A. Löfgren’s Syndrome: Human Leukocyte Antigen Strongly Influences the Disease Course. Am. J. Respir. Crit. Care Med. 2009, 179, 307–312. [Google Scholar] [CrossRef] [PubMed]
- Tejera Segura, B.; Holgado, S.; Mateo, L.; Pego-Reigosa, J.M.; Carnicero Iglesias, M.; Olivé, A. Löfgren syndrome: A study of 80 cases. Med. Clin. (Barc.) 2014, 143, 166–169. [Google Scholar] [CrossRef]
- Boekel, L.; Kummer, L.Y.; van Dam, K.P.J.; Hooijberg, F.; van Kempen, Z.; Vogelzang, E.H.; Wieske, L.; Eftimov, F.; van Vollenhoven, R.; Kuijpers, T.W.; et al. Adverse Events after First COVID-19 Vaccination in Patients with Autoimmune Diseases. Lancet Rheumatol. 2021, 3, E542–E545. [Google Scholar] [CrossRef]
- Watad, A.; De Marco, G.; Mahajna, H.; Druyan, A.; Eltity, M.; Hijazi, N.; Haddad, A.; Elias, M.; Zisman, D.; Naffaa, M.E.; et al. Immune-Mediated Disease Flares or New-Onset Disease in 27 Subjects Following MRNA/DNA SARS-CoV-2 Vaccination. Vaccines 2021, 9, 435. [Google Scholar] [CrossRef] [PubMed]
- Barbhaiya, M.; Levine, J.M.; Bykerk, V.P.; Jannat-Khah, D.; Mandl, L.A. Systemic Rheumatic Disease Flares after SARS-CoV-2 Vaccination among Rheumatology Outpatients in New York City. Ann. Rheum. Dis. 2021, 80, 1352–1354. [Google Scholar] [CrossRef] [PubMed]
- Keshavarz, P.; Yazdanpanah, F.; Rafiee, F.; Mizandari, M. Lymphadenopathy Following COVID-19 Vaccination: Imaging Findings Review. Acad. Radiol. 2021. [Google Scholar] [CrossRef] [PubMed]
- Coronavirus—Todesfälle in Deutschland Nach Alter. Available online: https://de.statista.com/statistik/daten/studie/1104173/umfrage/todesfaelle-aufgrund-des-coronavirus-in-deutschland-nach-geschlecht/ (accessed on 26 October 2021).
- RKI—Impfen—COVID-19 Und Impfen: Antworten Auf Häufig Gestellte Fragen (FAQ). Available online: https://www.rki.de/SharedDocs/FAQ/COVID-Impfen/gesamt.html (accessed on 26 October 2021).
- Bauckneht, M.; Aloè, T.; Tagliabue, E.; Cittadini, G.; Guadagno, A.; Morbelli, S.; Barisione, E. Beyond Covid-19 Vaccination-Associated Pitfalls on [(18)F]Fluorodeoxyglucose (FDG) PET: A Case of a Concomitant Sarcoidosis. Eur. J. Nucl. Med. Mol. Imaging 2021, 48, 2661–2662. [Google Scholar] [CrossRef]
- Psaltis, N.M.; Gardner, R.G.; Denton, W.J. Systemic Sarcoidosis and Red Dye Granulomatous Tattoo Inflammation after Influenza Vaccination: A Case Report and Review of Literature. Ocul. Immunol. Inflamm. 2014, 22, 314–321. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Heydari-Kamjani, M.; Vante, I.; Uppal, P.; Demory Beckler, M.; Kesselman, M.M. Uveitis Sarcoidosis Presumably Initiated After Administration of Shingrix Vaccine. Cureus 2019, 11, e4920. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Arakawa, A.; Kambe, N.; Nishikomori, R.; Tanabe, A.; Ueda, M.; Nishigori, C.; Miyachi, Y.; Kanazawa, N. NOD2 Mutation-Associated Case with Blau Syndrome Triggered by BCG Vaccination. Children 2021, 8, 117. [Google Scholar] [CrossRef] [PubMed]
- Pfisterer, R.; Wespi, H.; Herzog, H. Observations on cases of Boeck’s disease after BCG vaccination. Helv. Med. Acta 1954, 21, 439–444. [Google Scholar] [PubMed]
- Fried, K.H.; Genz, H. Sarcoidosis (Besnier-Boeck-Schaumann) disease after BCG vaccination. Tuberkulosearzt 1958, 12, 558–569. [Google Scholar] [PubMed]
- Ellman, P.; Andrews, L.G.B.C.G. Sarcoidosis. Br. Med. J. 1959, 1, 1433–1435. [Google Scholar] [CrossRef] [PubMed]
- Press, P.; Wacker, T. Lymph node-pulmonary images of sarcoid appearance in BCG-vaccinated subjects. Poumon. Coeur. 1962, 18, 937–949. [Google Scholar] [PubMed]
- Fried, K.H. BCG and Sarcoidosis in Berlin West. Acta Med. Scand. Suppl. 1964, 425, 260–265. [Google Scholar] [CrossRef] [PubMed]
- Sutherland, I.; Mitchell, D.N.; Hart, P.D. Incidence of intrathoracic sarcoidosis among young adults participating in a trial of tuberculosis vaccines. Br. Med. J. 1965, 2, 497–503. [Google Scholar] [CrossRef] [Green Version]
- Renard, J.; Puvinel, B.; Bertrand, Y. Apropos of a case of Besnier-Boeck-Schaumann disease in a subject vaccinated with BCG. Poumon. Coeur. 1962, 18, 951–956. [Google Scholar] [PubMed]
- Greally, J.F.; Manning, D.; McNicholl, B. Sarcoidosis Following B.C.G. Vaccination in a Lymphopaenic Boy. Sarcoidosis 1989, 6, 156–157. [Google Scholar]
- Osborne, G.E.N.; Mallon, E.; Mayou, S.C. Juvenile Sarcoidosis after BCG Vaccination. J. Am. Acad. Dermatol. 2003, 48, S99–S102. [Google Scholar] [CrossRef]
- Chen, E.S.; Moller, D.R. Etiologic Role of Infectious Agents. Semin. Respir. Crit. Care Med. 2014, 35, 285–295. [Google Scholar] [CrossRef] [PubMed]
- Svendsen, C.B.; Milman, N.; Rasmussen, E.M.; Thomsen, V.Ø.; Andersen, C.B.; Krogfelt, K.A. The Continuing Search for Mycobacterium Tuberculosis Involvement in Sarcoidosis: A Study on Archival Biopsy Specimens. Clin. Respir. J. 2011, 5, 99–104. [Google Scholar] [CrossRef] [PubMed]
- Oswald-Richter, K.A.; Beachboard, D.C.; Seeley, E.H.; Abraham, S.; Shepherd, B.E.; Jenkins, C.A.; Culver, D.A.; Caprioli, R.M.; Drake, W.P. Dual Analysis for Mycobacteria and Propionibacteria in Sarcoidosis BAL. J. Clin. Immunol. 2012, 32, 1129–1140. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Oswald-Richter, K.A.; Beachboard, D.C.; Zhan, X.; Gaskill, C.F.; Abraham, S.; Jenkins, C.; Culver, D.A.; Drake, W. Multiple Mycobacterial Antigens Are Targets of the Adaptive Immune Response in Pulmonary Sarcoidosis. Respir. Res. 2010, 11, 161. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Manansala, M.; Chopra, A.; Baughman, R.P.; Novak, R.; Lower, E.E.; Culver, D.A.; Korsten, P.; Drake, W.P.; Judson, M.A.; Sweiss, N. COVID-19 and Sarcoidosis, Readiness for Vaccination: Challenges and Opportunities. Front. Med. 2021, 8, 672028. [Google Scholar] [CrossRef] [PubMed]
- Sweiss, N.J.; Korsten, P.; Syed, H.J.; Syed, A.; Baughman, R.P.; Yee, A.M.F.; Culver, D.A.; Sosenko, T.; Azuma, A.; Bonella, F.; et al. When the Game Changes: Guidance to Adjust Sarcoidosis Management During the Coronavirus Disease 2019 Pandemic. Chest 2020, 158, 892–895. [Google Scholar] [CrossRef] [PubMed]
| Type of Vaccine | Timing of Onset | Population | Clinical Manifestations | Laboratory Manifestations | Outcome | Reference |
|---|---|---|---|---|---|---|
| SARS-CoV-2 (BNT162b2, Pfizer/BioNTech) | One day | Adult | Axillary and mediastinal lymphadenopathy | Not reported | Not reported | [19] |
| Influenza | Two months | Adult | Granulomatous uveitis, Tattoo sarcoidosis, pulmonary lymphadenopathy | ACE elevation | Near-complete remission with a need for topical treatment | [20] |
| Varicella-zoster (Shingrix) | Four days | Adult | Granulomatous uveitis Bilateral hilar and mediastinal adenopathy | ACE elevation Hypercalcemia Positive RF | Remission with local GC | [21] |
| BCG | 1–84 months | Adult and pediatric | NOD2-associated Blau syndrome with skin, bone. Lung findings and mediastinal lymphadenopathy, EN | ESR elevation in some cases | Spontaneous resolution in most cases | [22,23,24,25,26,27,28,29,30,31] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rademacher, J.-G.; Tampe, B.; Korsten, P. First Report of Two Cases of Löfgren’s Syndrome after SARS-CoV-2 Vaccination-Coincidence or Causality? Vaccines 2021, 9, 1313. https://doi.org/10.3390/vaccines9111313
Rademacher J-G, Tampe B, Korsten P. First Report of Two Cases of Löfgren’s Syndrome after SARS-CoV-2 Vaccination-Coincidence or Causality? Vaccines. 2021; 9(11):1313. https://doi.org/10.3390/vaccines9111313
Chicago/Turabian StyleRademacher, Jan-Gerd, Björn Tampe, and Peter Korsten. 2021. "First Report of Two Cases of Löfgren’s Syndrome after SARS-CoV-2 Vaccination-Coincidence or Causality?" Vaccines 9, no. 11: 1313. https://doi.org/10.3390/vaccines9111313
APA StyleRademacher, J.-G., Tampe, B., & Korsten, P. (2021). First Report of Two Cases of Löfgren’s Syndrome after SARS-CoV-2 Vaccination-Coincidence or Causality? Vaccines, 9(11), 1313. https://doi.org/10.3390/vaccines9111313







