A Review on Current COVID-19 Vaccines and Evaluation of Particulate Vaccine Delivery Systems
Abstract
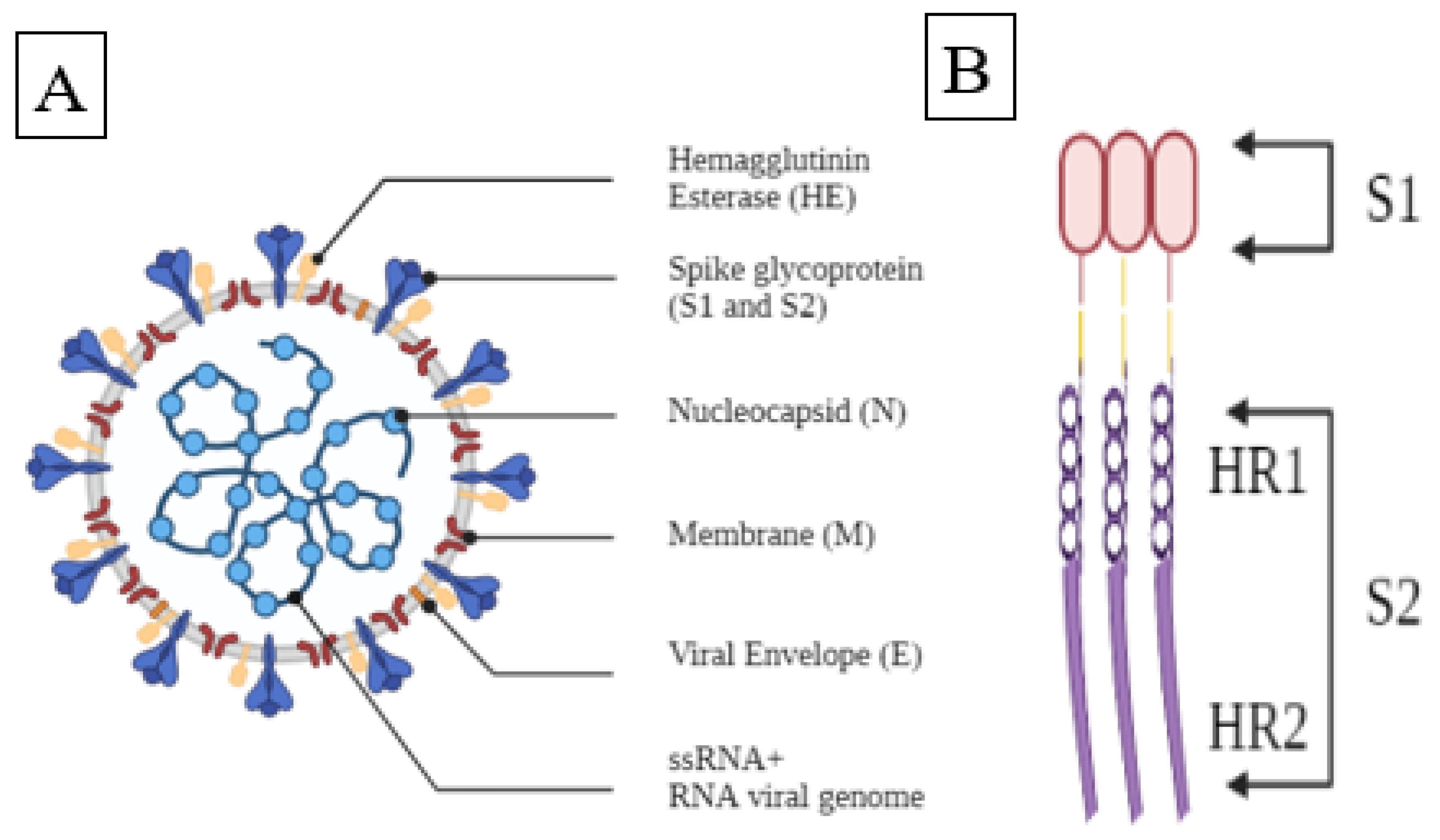
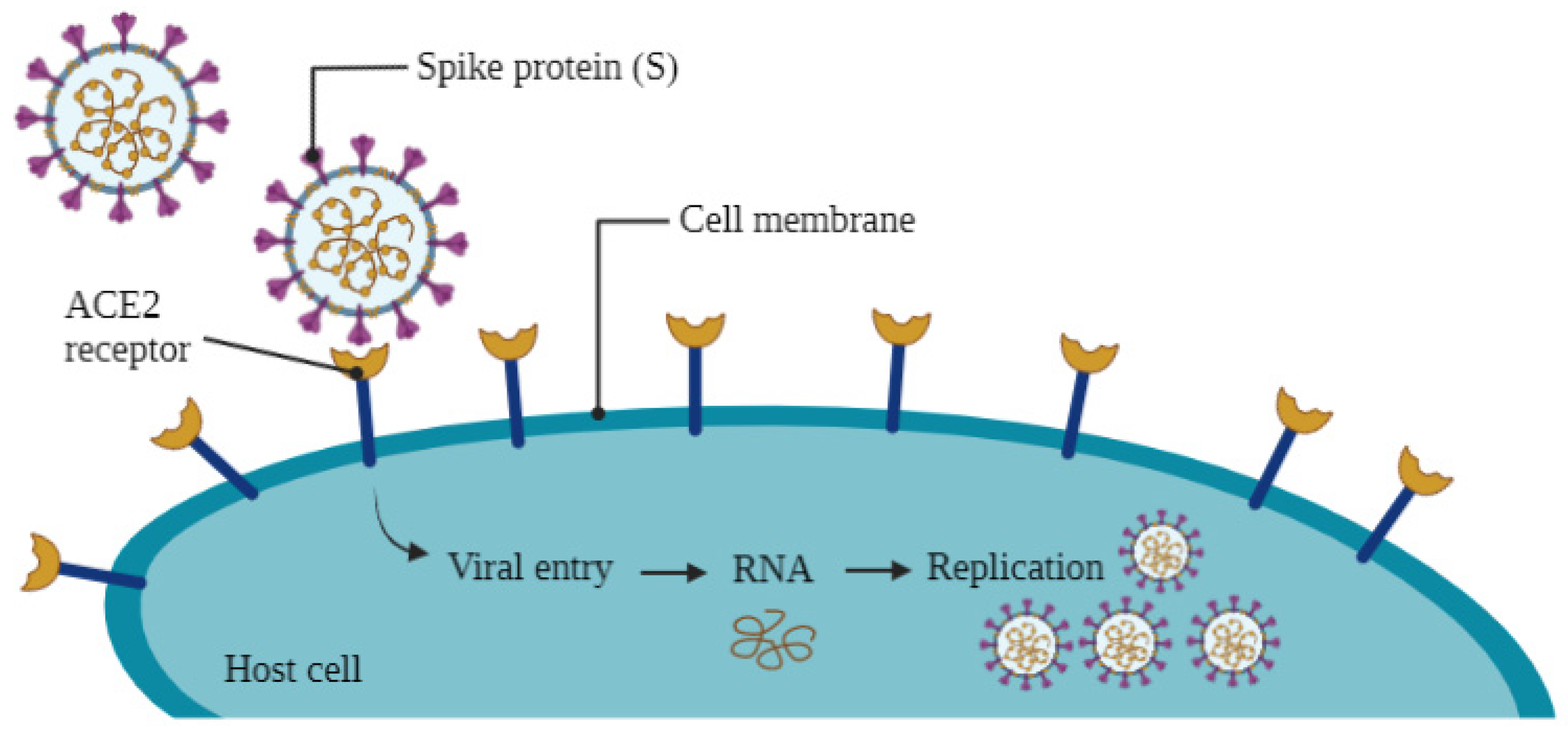
1. Introduction
2. Current COVID-19 Vaccine Platforms
2.1. mRNA Based COVID Vaccines
2.2. Recombinant Protein Based COVID Vaccines
2.3. Viral Vector Based COVID Vaccines
2.4. Plasmid DNA-Based COVID Vaccines
3. Limitations of Current COVID-19 Vaccines
3.1. Side Effects
3.2. Refusal of Vaccines
3.3. Blood Clotting
3.4. Needle Fear
3.5. Route of Administration
4. Potential of Micro- or Nano Particulate COVID Vaccines
5. Possible Particulate COVID Vaccine Delivery System for Oral, Sublingual and Buccal Administrations
5.1. Oral Administration
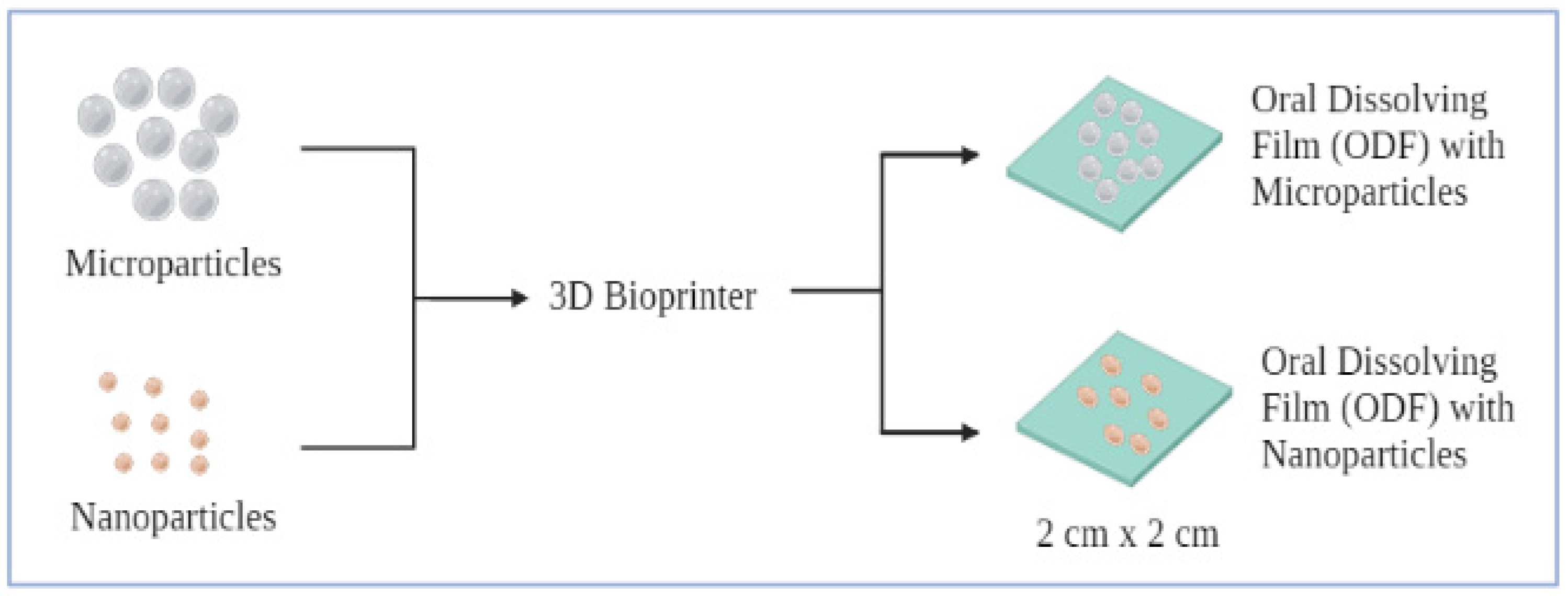
5.2. Film-Based Particulate COVID Vaccine for Sublingual and Buccal Administration
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kahn, J.S.; McIntosh, K. Discussion. Pediatr. Infect. Dis. J. 2005, 24, 223–227. [Google Scholar] [CrossRef]
- McIntosh, K.; Dees, J.H.; Becker, W.B.; Kapikian, A.Z.; Chanock, R.M. Recovery in Tracheal Organ Cultures of Novel Viruses from Patients with Respiratory Disease. Proc. Natl. Acad. Sci. USA 1967, 57, 933. [Google Scholar] [CrossRef] [PubMed]
- Tyrrel, D.A.J.; Lm Eida, J.D.A.; Akstelskaya, L.Z.; Easterday, B.C.; Bingham, R.W. Coronaviridae1. Intervirology 1975, 5, 76. [Google Scholar] [CrossRef] [PubMed]
- First Travel-Related Case of 2019 Novel Coronavirus Detected in United States|CDC Online Newsroom|CDC. Available online: https://www.cdc.gov/media/releases/2020/p0121-novel-coronavirus-travel-case.html (accessed on 19 October 2020).
- Second Travel-Related Case of 2019 Novel Coronavirus Detected in United States|CDC Online Newsroom|CDC. Available online: https://www.cdc.gov/media/releases/2020/p0124-s-travel-coronavirus.html (accessed on 19 October 2020).
- New-Type Coronavirus Causes Pneumonia in Wuhan: Expert-Xinhua|English.news.cn. Available online: http://www.xinhuanet.com/english/2020-01/09/c_138690570.htm (accessed on 19 October 2020).
- Coronavirus|Human Coronavirus Types|CDC. Available online: https://www.cdc.gov/coronavirus/types.html (accessed on 19 October 2020).
- Coronavirus (COVID-19) Frequently Asked Questions | CDC. Available online: https://www.cdc.gov/coronavirus/2019-ncov/faq.html (accessed on 19 October 2020).
- Kuba, K.; Imai, Y.; Rao, S.; Gao, H.; Guo, F.; Guan, B.; Huan, Y.; Yang, P.; Zhang, Y.; Deng, W.; et al. A crucial role of angiotensin converting enzyme 2 (ACE2) in SARS coronavirus-induced lung injury. Nat. Med. Vol. 2005, 11, 875–879. [Google Scholar] [CrossRef] [PubMed]
- Jeffers, S.A.; Tusell, S.M.; Gillim-Ross, L.; Hemmila, E.M.; Achenbach, J.E.; Babcock, G.J.; Thomas, W.D.; Thackray, L.B.; Young, M.D.; Mason, R.J.; et al. Cd209l (L-Sign) Is a Receptor for Severe Acute Respiratory Syndrome Coronavirus. Proc. Natl. Acad. Sci. USA 2004, 101, 15748–15753. [Google Scholar] [CrossRef] [PubMed]
- Belouzard, S.; Millet, J.K.; Licitra, B.N.; Whittaker, G.R. Mechanisms of Coronavirus Cell Entry Mediated by the Viral Spike Protein. Viruses 2012, 4, 1011–1033. [Google Scholar] [CrossRef]
- Callaway, E. The Race for coronavirus vaccines. Nature 2020, 580, 576–577. [Google Scholar] [CrossRef]
- Knight, S.C.; Stagg, A.J. Antigen-presenting cell types. Curr. Opin. Immunol. 1993, 5, 374–382. [Google Scholar] [CrossRef]
- Levi, R.; Azzolini, E.; Pozzi, C.; Ubaldi, L.; Lagioia, M.; Mantovani, A.; Rescigno, M. One dose of SARS-CoV-2 vaccine exponentially increases antibodies in individuals who have recovered from symptomatic COVID-19. J. Clin. Investig. 2021, 131, e149154. [Google Scholar] [CrossRef]
- Ledford, H. COVID vaccines and blood clots: Five key questions. Nature 2021, 592, 495–496. [Google Scholar] [CrossRef]
- Greinacher, A.; Thiele, T.; Warkentin, T.E.; Weisser, K.; Kyrle, P.A.; Eichinger, S. Thrombotic Thrombocytopenia after ChAdOx1 nCov-19 Vaccination. N. Engl. J. Med. 2021, 384, 2092–2101. [Google Scholar] [CrossRef]
- Uddin, M.N.; Kouzi, S.A.; Hussain, M.D. Strategies for Developing Oral Vaccines for Human Papillomavirus (HPV) Induced Cancer Using Nanoparticle Mediated Delivery System. J. Pharm. Pharm. Sci. 2015, 18, 220. [Google Scholar] [CrossRef]
- Copland, M.J.; Baird, M.A.; Rades, T.; McKenzie, J.L.; Becker, B.; Reck, F.; Tyler, P.C.; Davies, N.M. Liposomal delivery of antigen to human dendritic cells. Vaccine 2003, 21, 883–890. [Google Scholar] [CrossRef]
- Schijns, V.E. Immunological concepts of vaccine adjuvant activity. Curr. Opin. Immunol. 2000, 12, 456–463. [Google Scholar] [CrossRef]
- Kreuter, J. Nanoparticles and microparticles for drug and vaccine delivery. J. Anat. 1996, 189 Pt 3, 503–505. [Google Scholar]
- Parums, D.V. Editorial: SARS-CoV-2 mRNA Vaccines and the Possible Mechanism of Vaccine-Induced Immune Thrombotic Thrombocytopenia (VITT). Med. Sci. Monit. 2021, 27, e932899. [Google Scholar] [CrossRef]
- Verga, D. mRNA and the Future of Vaccine Manufacturing | PATH. Available online: https://www.path.org/articles/mrna-and-future-vaccine-manufacturing/ (accessed on 23 August 2020).
- Florindo, H.F.; Kleiner, R.; Vaskovich-Koubi, D.; Acúrcio, R.C.; Carreira, B.; Yeini, E.; Tiram, G.; Liubomirski, Y.; Satchi-Fainaro, R. Immune-mediated approaches against COVID-19. Nat. Nanotechnol. 2020, 15, 630–645. [Google Scholar] [CrossRef] [PubMed]
- Hou, X.; Zaks, T.; Langer, R.; Dong, Y. Lipid nanoparticles for mRNA delivery. Nat. Rev. Mater. 2021. [Google Scholar] [CrossRef] [PubMed]
- He, Y.; Zhou, Y.; Liu, S.; Kou, Z.; Li, W.; Farzan, M.; Jiang, S. Receptor-binding domain of SARS-CoV spike protein induces highly potent neutralizing antibodies: Implication for developing subunit vaccine. Biochem. Biophys. Res. Commun. 2004, 324, 773–781. [Google Scholar] [CrossRef] [PubMed]
- Du, L.; Zhao, G.; He, Y.; Guo, Y.; Zheng, B.-J.; Jiang, S.; Zhou, Y. Receptor-binding domain of SARS-CoV spike protein induces long-term protective immunity in an animal model. Vaccine 2007, 25, 2832–2838. [Google Scholar] [CrossRef] [PubMed]
- Fang, M.; Cheng, H.; Dai, Z.; Bu, Z.; Sigal, L.J. Immunization with a single extracellular enveloped virus protein produced in bacteria provides partial protection from a lethal orthopoxvirus infection in a natural host. Virology 2006, 345, 231–243. [Google Scholar] [CrossRef]
- Galmiche, M.C.; Goenaga, J.; Wittek, R.; Rindisbacher, L. Neutralizing and Protective Antibodies Directed against Vaccinia Virus Envelope Antigens. Virology 1999, 254, 71–80. [Google Scholar] [CrossRef]
- Novavax to Present COVID-19 Vaccine Candidate Progress in World Vaccine Congress Webinar Series | Novavax Inc.—IR Site. Available online: https://ir.novavax.com/news-releases/news-release-details/novavax-present-covid-19-vaccine-candidate-progress-world (accessed on 11 May 2021).
- Thi, T.T.H.; Suys, E.J.A.; Lee, J.S.; Nguyen, D.H.; Park, K.D.; Truong, N.P. Lipid-Based Nanoparticles in the Clinic and Clinical Trials: From Cancer Nanomedicine to COVID-19 Vaccines. Vaccines 2021, 9, 359. [Google Scholar] [CrossRef]
- Du, L.; Zhao, G.; Lin, Y.; Sui, H.; Chan, C.; Ma, S.; He, Y.; Jiang, S.; Wu, C.; Yuen, K.-Y.; et al. Intranasal Vaccination of Recombinant Adeno-Associated Virus Encoding Receptor-Binding Domain of Severe Acute Respiratory Syndrome Coronavirus (SARS-CoV) Spike Protein Induces Strong Mucosal Immune Responses and Provides Long-Term Protection against SARS-CoV infection. J. Immunol. 2008, 180, 948–956. [Google Scholar] [CrossRef]
- Voysey, M.; Clemens, S.A.C.; Madhi, S.A.; Weckx, L.Y.; Folegatti, P.M.; Aley, P.K.; Angus, B.; Baillie, V.L.; Barnabas, S.L.; Bhorat, Q.E.; et al. Safety and efficacy of the ChAdOx1 nCoV-19 vaccine (AZD1222) against SARS-CoV-2: An interim analysis of four randomised controlled trials in Brazil, South Africa, and the UK. Lancet 2021, 397, 99–111. [Google Scholar] [CrossRef]
- Lewis, D. China’s coronavirus vaccine shows military’s growing role in medical research. Nature 2020, 585, 494–495. [Google Scholar] [CrossRef]
- Cohen, J. Russia’s approval of a COVID-19 vaccine is less than meets the press release. Science 2020, 10. [Google Scholar] [CrossRef]
- Wang, J.; Peng, Y.; Xu, H.; Cui, Z.; Williams, R.O. The COVID-19 Vaccine Race: Challenges and Opportunities in Vaccine Formulation. AAPS PharmSciTech 2020, 21, 1–12. Available online: http://link.springer.com/10.1208/s12249-020-01744-7 (accessed on 17 September 2021). [CrossRef] [PubMed]
- Uddin, M.; Henry, B.; Carter, K.D.; Roni, M.A.; Kouzi, S.A. A Novel Formulation Strategy to Deliver Combined DNA and VLP Based HPV Vaccine. J. Pharm. Pharm. Sci. 2019, 22, 536–547. [Google Scholar] [CrossRef]
- Blumenthal, K.G.; Freeman, E.E.; Saff, R.R.; Robinson, L.B.; Wolfson, A.R.; Foreman, R.K.; Hashimoto, D.; Banerji, A.; Li, L.; Anvari, S.; et al. Delayed Large Local Reactions to mRNA-1273 Vaccine against SARS-CoV-2. N. Engl. J. Med. 2021, 384, 1273–1277. [Google Scholar] [CrossRef] [PubMed]
- Pfizer vs. Moderna Vaccines: Does One Have More Side Effects?—NBC Chicago. Available online: https://www.nbcchicago.com/news/local/pfizer-vs-moderna-vaccines-does-one-have-more-side-effects-than-the-other/2499425/ (accessed on 13 May 2021).
- Solís Arce, J.S.; Warren, S.S.; Meriggi, N.F.; Scacco, A.; McMurry, N.; Voors, M.; Syunyaev, G.; Malik, A.A.; Aboutajdine, S.; Adeojo, O.; et al. COVID-19 vaccine acceptance and hesitancy in low- and middle-income countries. Nat. Med. 2021, 27, 1385–1394. [Google Scholar] [CrossRef]
- Yigit, M.; Ozkaya-Parlakay, A.; Senel, E. Evaluation of COVID-19 Vaccine Refusal in Parents. Pediatr. Infect. Dis. J. 2021, 40, e134–e136. [Google Scholar] [CrossRef]
- Joint CDC and FDA Statement on Johnson & Johnson COVID-19 Vaccine | CDC Online Newsroom | CDC. Available online: https://www.cdc.gov/media/releases/2021/s0413-JJ-vaccine.html (accessed on 13 May 2021).
- Johnson & Johnson Delays Its COVID-19 Vaccine Rollout in EUROPE—The New York Times. Available online: https://www.nytimes.com/2021/04/13/world/johnson-covid-vaccine-europe.html (accessed on 8 July 2021).
- McLenon, J.; Rogers, M.A.M. The fear of needles: A systematic review and meta-analysis. J. Adv. Nurs. 2019, 75, 30–42. [Google Scholar] [CrossRef]
- Belyakov, I.M.; Ahlers, J.D. What Role Does the Route of Immunization Play in the Generation of Protective Immunity against Mucosal Pathogens? J. Immunol. 2009, 183, 6883–6892. [Google Scholar] [CrossRef]
- Mohanan, D.; Slütter, B.; Henriksen-Lacey, M.; Jiskoot, W.; Bouwstra, J.A.; Perrie, Y.; Kündig, T.M.; Gander, B.; Johansen, P. Administration routes affect the quality of immune responses: A cross-sectional evaluation of particulate antigen-delivery systems. J. Control. Release 2010, 147, 342–349. [Google Scholar] [CrossRef]
- Gala, R.P.; Popescu, C.; Knipp, G.T.; McCain, R.R.; Ubale, R.V.; Addo, R.; Bhowmik, T.; Kulczar, C.D.; D’Souza, M.J. Physicochemical and Preclinical Evaluation of a Novel Buccal Measles Vaccine. AAPS PharmSciTech 2017, 18, 283–292. [Google Scholar] [CrossRef]
- Anderson, E.J.; Rouphael, N.G.; Widge, A.T.; Jackson, L.A.; Roberts, P.C.; Makhene, M.; Chappell, J.D.; Denison, M.R.; Stevens, L.J.; Pruijssers, A.J.; et al. Safety and Immunogenicity of SARS-CoV-2 mRNA-1273 Vaccine in Older Adults. N. Engl. J. Med. 2020, 383, 2427–2438. [Google Scholar] [CrossRef] [PubMed]
- Walter, E.; Dreher, D.; Kok, M.; Thiele, L.; Kiama, S.G.; Gehr, P.; Merkle, H.P. Hydrophilic poly(dl-lactide-co-glycolide) microspheres for the delivery of DNA to human-derived macrophages and dendritic cells. J. Control. Release 2001, 76, 149–168. [Google Scholar] [CrossRef]
- Slütter, B.; Soema, P.C.; Ding, Z.; Verheul, R.; Hennink, W.; Jiskoot, W. Conjugation of ovalbumin to trimethyl chitosan improves immunogenicity of the antigen. J. Control. Release 2010, 143, 207–214. [Google Scholar] [CrossRef] [PubMed]
- O’Hagan, D. Microparticles and polymers for the mucosal delivery of vaccines. Adv. Drug Deliv. Rev. 1998, 34, 305–320. [Google Scholar] [CrossRef]
- Panyam, J.; Labhasetwar, V. Biodegradable nanoparticles for drug and gene delivery to cells and tissue. Adv. Drug Deliv. Rev. 2003, 55, 329–347. [Google Scholar] [CrossRef]
- Tobío, M.; Gref, R.; Sánchez, A.; Langer, R.; Alonso, M.J. Stealth PLA-PEG nanoparticles as protein carriers for nasal administration. Pharm. Res. 1998, 15, 270–275. [Google Scholar] [CrossRef]
- Parkin, J.; Cohen, B. An overview of the immune system. Lancet 2001, 357, 1777–1789. [Google Scholar] [CrossRef]
- Gamvrellis, A.; Gloster, S.; Jefferies, M.; Mottram, P.L.; Smooker, P.; Plebanski, M.; Scheerlinck, J.-P.Y. Characterisation of local immune responses induced by a novel nano-particle based carrier-adjuvant in sheep. Vet. Immunol. Immunopathol. 2013, 155, 21–29. [Google Scholar] [CrossRef] [PubMed]
- Thiele, L.; Merkle, H.P.; Walter, E. Phagocytosis and phagosomal fate of surface-modified microparticles in dendritic cells and macrophages. Pharm. Res. 2003, 20, 221–228. [Google Scholar] [CrossRef]
- Elamanchili, P.; Diwan, M.; Cao, M.; Samuel, J. Characterization of poly (d,l-lactic-co-glycolic acid) based nanoparticulate system for enhanced delivery of antigens to dendritic cells. Vaccine 2004, 22, 2406–2412. [Google Scholar] [CrossRef] [PubMed]
- Hamdy, S.; Haddadi, A.; Hung, R.W.; Lavasanifar, A. Targeting dendritic cells with nano-particulate PLGA cancer vaccine formulations. Adv. Drug. Deliv. Rev. 2011, 63, 943–955. [Google Scholar] [CrossRef]
- Elamanchili, P.; Lutsiak, C.M.E.; Hamdy, S.; Diwan, M.; Samuel, J. “Pathogen-Mimicking” Nanoparticles for Vaccine Delivery to Dendritic Cells. J. Immunother. 2007, 30, 378–395. [Google Scholar] [CrossRef]
- Nettey, H.; Haswani, D.; Oettinger, C.W.; D’Souza, M.J. Formulation and testing of vancomycin loaded albumin microspheres prepared by spray-drying. J. Microencapsul. 2006, 23, 632–642. [Google Scholar] [CrossRef]
- Joshi, D.; Chbib, C.; Uddin, M.N.; D’Souza, M.J. Evaluation of Microparticulate (S)-4,5-Dihydroxy-2,3-pentanedione (DPD) as a Potential Vaccine Adjuvant. AAPS J. 2021, 23, 84. [Google Scholar] [CrossRef]
- Combadière, B.; Mahé, B. Particle-based vaccines for transcutaneous vaccination. Comp. Immunol. Microbiol. Infect. Dis. 2008, 31, 293–315. [Google Scholar] [CrossRef]
- O’Grady, M.; Bruner, P.J. Polio Vaccine. StatPearls. 2021. Available online: http://www.ncbi.nlm.nih.gov/pubmed/30252295 (accessed on 19 September 2021).
- Poliomyelitis. Available online: https://www.who.int/teams/health-product-policy-and-standards/standards-and-specifications/vaccines-quality/poliomyelitis (accessed on 27 August 2021).
- Yeh, M.T.; Bujaki, E.; Dolan, P.T.; Smith, M.; Wahid, R.; Konz, J.; Weiner, A.J.; Bandyopadhyay, A.S.; Van Damme, P.; De Coster, I.; et al. Engineering the Live-Attenuated Polio Vaccine to Prevent Reversion to Virulence. Cell Host Microbe 2020, 27, 736–751.e8. [Google Scholar] [CrossRef]
- Cesta, M.F. Normal Structure, Function, and Histology of Mucosa-Associated Lymphoid Tissue. Toxicol. Pathol. 2006, 34, 599–608. [Google Scholar] [CrossRef]
- Goldsby, R.; Kindt, T.; Osborne, B.; Kuby, J. Immunology, 5th ed.; W. H. Freeman: New York, NY, USA, 2002; ISBN 0716749475. [Google Scholar]
- Pabst, R. The anatomical basis for the immune function of the gut. Anat. Embryol. 1987, 176, 135–144. [Google Scholar] [CrossRef]
- Thönes, N.; Müller, M. Oral immunization with different assembly forms of the HPV 16 major capsid protein L1 induces neutralizing antibodies and cytotoxic T-lymphocytes. Virology 2007, 369, 375–388. [Google Scholar] [CrossRef] [PubMed]
- Chang, S.; Warner, J.; Liang, L.; Fairman, J. A novel vaccine adjuvant for recombinant flu antigens. Biologicals 2009, 37, 141–147. [Google Scholar] [CrossRef] [PubMed]
- Smith, J.H.; Papania, M.; Knaus, D.; Brooks, P.; Haas, D.L.; Mair, R.; Barry, J.; Tompkins, S.M.; Tripp, R.A. Nebulized live-attenuated influenza vaccine provides protection in ferrets at a reduced dose. Vaccine 2012, 30, 3026–3033. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Panraksa, P.; Udomsom, S.; Rachtanapun, P.; Chittasupho, C.; Ruksiriwanich, W.; Jantrawut, P. Hydroxypropyl Methylcellulose E15: A Hydrophilic Polymer for Fabrication of Orodispersible Film Using Syringe Extrusion 3D Printer. Polymers 2020, 12, 2666. [Google Scholar] [CrossRef] [PubMed]
- Uddin, M.N.; Allon, A.; Roni, M.A.; Kouzi, S. Overview and Future Potential of Fast Dissolving Buccal Films as Drug Delivery System for Vaccines. J. Pharm. Pharm. Sci. 2019, 22, 388–406. [Google Scholar] [CrossRef] [PubMed]
- Anselmo, A.C.; Gokarn, Y.; Mitragotri, S. Non-invasive delivery strategies for biologics. Nat. Rev. Drug Discov. 2019, 18, 19–40. [Google Scholar] [CrossRef]
- Sinjari, B.; D’Ardes, D.; Santilli, M.; Rexhepi, I.; D’Addazio, G.; Di Carlo, P.; Chiacchiaretta, P.; Caputi, S.; Cipollone, F. SARS-CoV-2 and Oral Manifestation: An Observational, Human Study. J. Clin. Med. 2020, 9, 3218. [Google Scholar] [CrossRef] [PubMed]
- Huang, N.; Pérez, P.; Kato, T.; Mikami, Y.; Okuda, K.; Gilmore, R.C.; Conde, C.D.; Gasmi, B.; Stein, S.; Beach, M.; et al. SARS-CoV-2 infection of the oral cavity and saliva. Nat. Med. 2021, 27, 892–903. [Google Scholar] [CrossRef] [PubMed]
- Brandtzaeg, P. Function of mucosa-associated lymphoid tissue in antibody formation. Immunol. Investig. 2010, 39, 303–355. [Google Scholar] [CrossRef] [PubMed]
- Amorij, J.-P.; Kersten, G.F.A.; Saluja, V.; Tonnis, W.F.; Hinrichs, W.L.J.; Slütter, B.; Bal, S.M.; Bouwstra, J.A.; Huckriede, A.; Jiskoot, W. Towards tailored vaccine delivery: Needs, challenges and perspectives. J. Control. Release 2012, 161, 363–376. [Google Scholar] [CrossRef] [PubMed]
| Vaccine Candidates | Classification of Vaccine | Clinical Phase | Lead Development Company/Collaboration |
|---|---|---|---|
| GRAd-COV2 | Adenovirus-based vaccine | Phase 1 | ReiThera; Leukocare; Univercells; Lazzaro Spallanzani National Institute for Infection |
| ChAd-SARS-CoV-2-S | Adenovirus-based vaccine | Preclinical | Washington University School of Medicine in St. Louis |
| LinealDNA | DNA Vaccine | Preclinical | Takis Biotech |
| AG0301-COVID19 | DNA vaccine | Phase 1/2 | AnGes, Inc. |
| GX-19 | DNA vaccine | Phase 1/2 | Genexine |
| INO-4800 | DNA vaccine (plasmid) | Phase 1/2 | Inovio Pharmaceuticals; Center for Pharmaceutical Research, Kansas City. Mo.; University of Pennsylvania, Philadelphia |
| ZyCoV-D | DNA vaccine (plasmid) | Phase 2 | Zydus Cadila |
| AAVCOVID | Gene-based vaccine | Preclinical | Massachusetts General Hospital; University of Pennsylvania |
| No name given | gp96-based vaccine | Preclinical | Heat Biologics; University of Miami Miller School of Medicine |
| No name given | Ii-Key peptide COVID-19 vaccine | Preclinical | Generex Biotechnology |
| No name given | Inactivated vaccine | Phase 1/2 | Research Institute for Biological Safety Problems, Rep of Kazakhstan |
| No name given | Inactivated vaccine | Phase 3 | Wuhan Institute of Biological Products; China National Pharmaceutical Group (Sinopharm); Henan Provincial Center for Disease Control and Prevention |
| Covaxin | Inactivated vaccine | Phase 2 | Bharat Biotech; National Institute of Virology |
| No name given | Inactivated vaccine | Phase 1/2 | Chinese Academy of Medical Sciences, Institute of Medical Biology; West China Second University Hospital, Yunnan Center for Disease Control and Prevention |
| No name given | Inactivated vaccine | Preclinical | Shenzhen Kangtai Biological Products |
| BBIBP-CorV | Inactivated vaccine | Phase 3 | Beijing Institute of Biological Products; China National Pharmaceutical Group (Sinopharm); Henan Provincial Center for Disease Control and Prevention |
| CoronaVac | Inactivated vaccine (formalin with alum adjuvant) | Phase 3 | Sinovac; Sinovac Research and Development Co., Ltd. |
| AdCOVID | Intranasal vaccine | Preclinical | Altimmune; University of Alabama at Birmingham |
| T-COVIDTM | Intranasal vaccine | Preclinical | Altimmune |
| Bacillus Calmette-Guerin (BCG) vaccine | Live-attenuated vaccine | Phase 2/3 | University of Melbourne and Murdoch Children’s Research Institute; Radboud University Medical Center; Faustman Lab at Massachusetts General Hospital |
| V591 | Replicating viral vector | Phase 1 | University of Pittsburgh’s Center for Vaccine Research; Themis Biosciences; Institut Pasteur |
| bacTRL-Spike | Monovalent oral vaccine (bifidobacteria) | Preclinical | Symvivo |
| COVAX-19 | Protein subunit Monovalent spike protein vaccine | Phase 1 | Vaxine Pty Ltd.; Royal Adelaide Hospital |
| No name announced | mRNA lipid nanoparticle | Early research | CanSino Biologics, Precision NanoSystems |
| mRNA-1273 | LNP-encapsulated mRNA vaccine | Phase 3 | Moderna; Kaiser Permanente Washington Health Research Institute |
| BNT162 | 3 LNP-mRNAs-based vaccine | Phase 3 | Pfizer, BioNTech |
| CVnCoV | mRNA-based vaccine | Phase 1 | CureVac |
| No name given | mRNA-based vaccine | Preclinical | Chulalongkorn University’s Center of Excellence in Vaccine Research and Development |
| UB-612 | Multitope peptide-based vaccine | Phase 1 | COVAXX; United Biomedical Inc. (UBI) |
| JNJ-78436735 | Non-replicating viral vector | Phase 3 | Johnson & Johnson |
| Sputnik V COVID-Vac; Гам-КОВИД-Вак20 | Non-replicating viral vector Adenovirus-based vaccine | Phase 3 | Gamaleya Research Institute, Acellena Contract Drug Research and Development |
| EpiVacCorona | Peptide vaccine | Phase 1/2 | Federal Budgetary Research Institution State Research Center of Virology and Biotechnology |
| No name given | Plant-based adjuvant vaccine | Phase 1 | Medicago; GSK; Dynavax |
| No name given | Protein subunit vaccine S protein20 | Phase 1/2 | Sanofi; GlaxoSmithKline |
| No name given | Protein subunit RBD-based vaccine | Phase 1/2 | Kentucky Bioprocessing, Inc. |
| AdimrSC-2f | Protein subunit vaccine | Phase 1 | Adimmune |
| No name given | Protein subunit vaccine | Phase 1 | CSL; The University of Queensland; Seqirus |
| SCB-2019 | Protein subunit vaccine | Phase 1 | GlaxoSmithKline, Sanofi, Clover Biopharmaceuticals, Dynavax and Xiamen Innovax; Linear Clinical Research (Australia) |
| No name given | Protein subunit vaccine | Preclinical | University of Saskatchewan Vaccine and Infectious Disease Organization-International Vaccine Centre |
| PittCoVacc | Recombinant protein subunit vaccine | Preclinical | UPMC/University of Pittsburgh School of Medicine |
| RBD-Dimer (relief) | Protein subunit Adjuvanted recombinant protein | Phase 2 | Anhui Zhifei Longcom Biopharmaceutical, Institute of Microbiology of the Chinese Academy of Sciences |
| No name given | Recombinant vaccine | Preclinical | Sanofi, Translate Bio |
| Ad5-nCoV | Recombinant vaccine (adenovirus type 5 vector) | Phase 3 | CanSino Biologics; Tongji Hospital |
| VXA-CoV2-1 | Recombinant vaccine (adenovirus vector) | Phase 1 | Vaxart |
| V590 | Recombinant vaccine (stomatitis virus) | Phase 1 | Merck; IAVI |
| DelNS1-2019-nCoV-RBD-OPT1 | Replicating viral vector | Phase 1 | Xiamen University, Beijing Wantai Biological Pharmacy; Jiangsu Provincial Centre for Disease Control and Prevention |
| No name announced | Replicating viral vector | Preclinical | Federal Budgetary Research Institution (FBRI); State research center of virology and biotechnology “VECTOR” |
| AZD1222 ChAdOx1-S20 | Non-Replicating viral vector vaccine | Phase 3 | The University of Oxford; AstraZeneca; IQVIA; Serum Institute of India |
| HDT-301 | RNA vaccine | Preclinical | University of Washington; National Institutes of Health Rocky Mountain Laboratories; HDT Bio Corp. |
| LNP-CoVsaRNA | Self-amplifying RNA vaccine | Phase 1/2 | Imperial College London |
| HaloVax | Self-assembling vaccine | Preclinical | Voltron Therapeutics, Inc.; Hoth Therapeutics, Inc.; MGH Vaccine and Immunotherapy Center |
| LUNAR-COV19 [36] | Self-replicating RNA vaccine | Phase 1/2 | Arcturus Therapeutics and Duke-NUS Medical School |
| CDX-005 [10,12] | Weakened | Phase 1 | Codagenix, Serum Institute of India |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shah, S.M.; Alsaab, H.O.; Rawas-Qalaji, M.M.; Uddin, M.N. A Review on Current COVID-19 Vaccines and Evaluation of Particulate Vaccine Delivery Systems. Vaccines 2021, 9, 1086. https://doi.org/10.3390/vaccines9101086
Shah SM, Alsaab HO, Rawas-Qalaji MM, Uddin MN. A Review on Current COVID-19 Vaccines and Evaluation of Particulate Vaccine Delivery Systems. Vaccines. 2021; 9(10):1086. https://doi.org/10.3390/vaccines9101086
Chicago/Turabian StyleShah, Sarthak M., Hashem O. Alsaab, Mutasem M. Rawas-Qalaji, and Mohammad N. Uddin. 2021. "A Review on Current COVID-19 Vaccines and Evaluation of Particulate Vaccine Delivery Systems" Vaccines 9, no. 10: 1086. https://doi.org/10.3390/vaccines9101086
APA StyleShah, S. M., Alsaab, H. O., Rawas-Qalaji, M. M., & Uddin, M. N. (2021). A Review on Current COVID-19 Vaccines and Evaluation of Particulate Vaccine Delivery Systems. Vaccines, 9(10), 1086. https://doi.org/10.3390/vaccines9101086








