A Newcastle Disease Virus (NDV) Expressing a Membrane-Anchored Spike as a Cost-Effective Inactivated SARS-CoV-2 Vaccine
Abstract
1. Introduction
2. Materials and Methods
2.1. Ethics Statement
2.2. Plasmids
2.3. Cells and Viruses
2.4. Rescue of NDV LaSota Expressing the Spike of SARS-CoV-2
2.5. Preparation of Concentrated Virus
2.6. Evaluation of the Stability of the S-F in the Allantoic Fluid
2.7. Western Blot
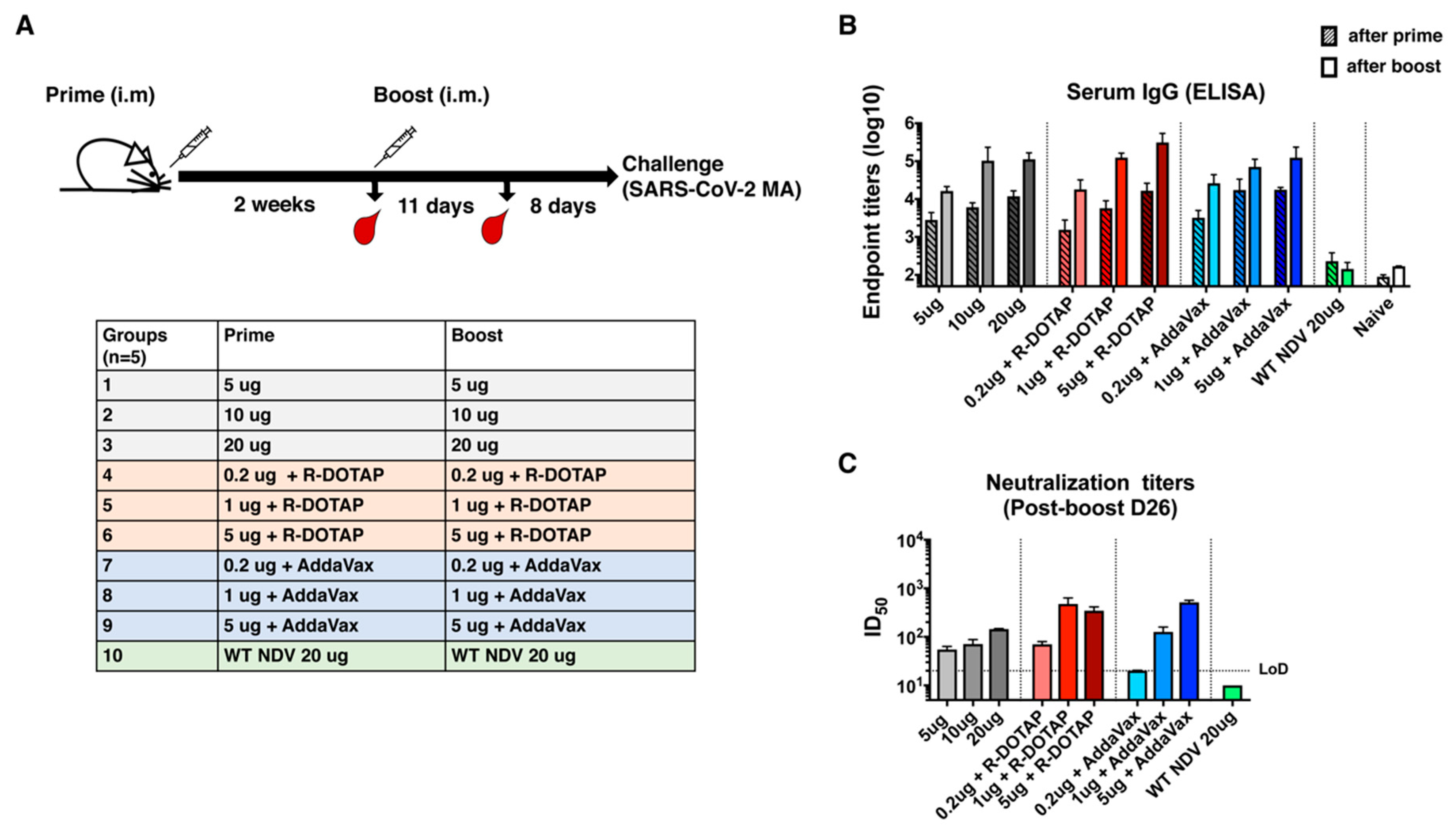
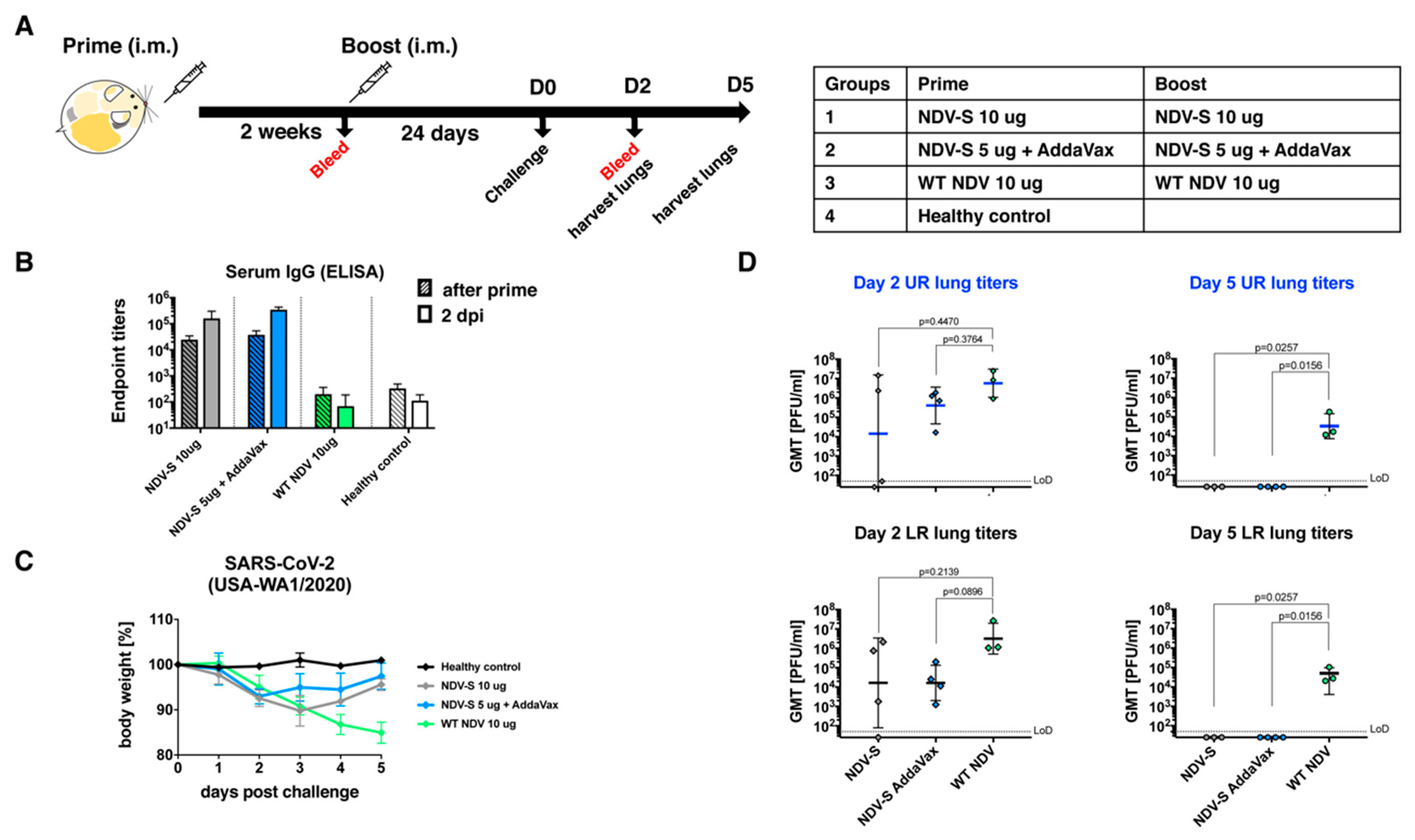
2.8. Immunization and Challenge Study in BALB/c Mice
2.9. Immunization and Challenge Study in Golden Syrian Hamsters
2.10. Lung Titers
2.11. Enzyme Linked Immunosorbent Assays (ELISAs)
2.12. Micro-Neutralization Assay
2.13. Statistics
3. Results
3.1. The Spike Protein Expressed on NDV Virions Is Stable in Allantoic Fluid
3.2. Inactivated NDV-S Vaccine Induced High Titers of Binding and Neutralizing Antibodies in Mice
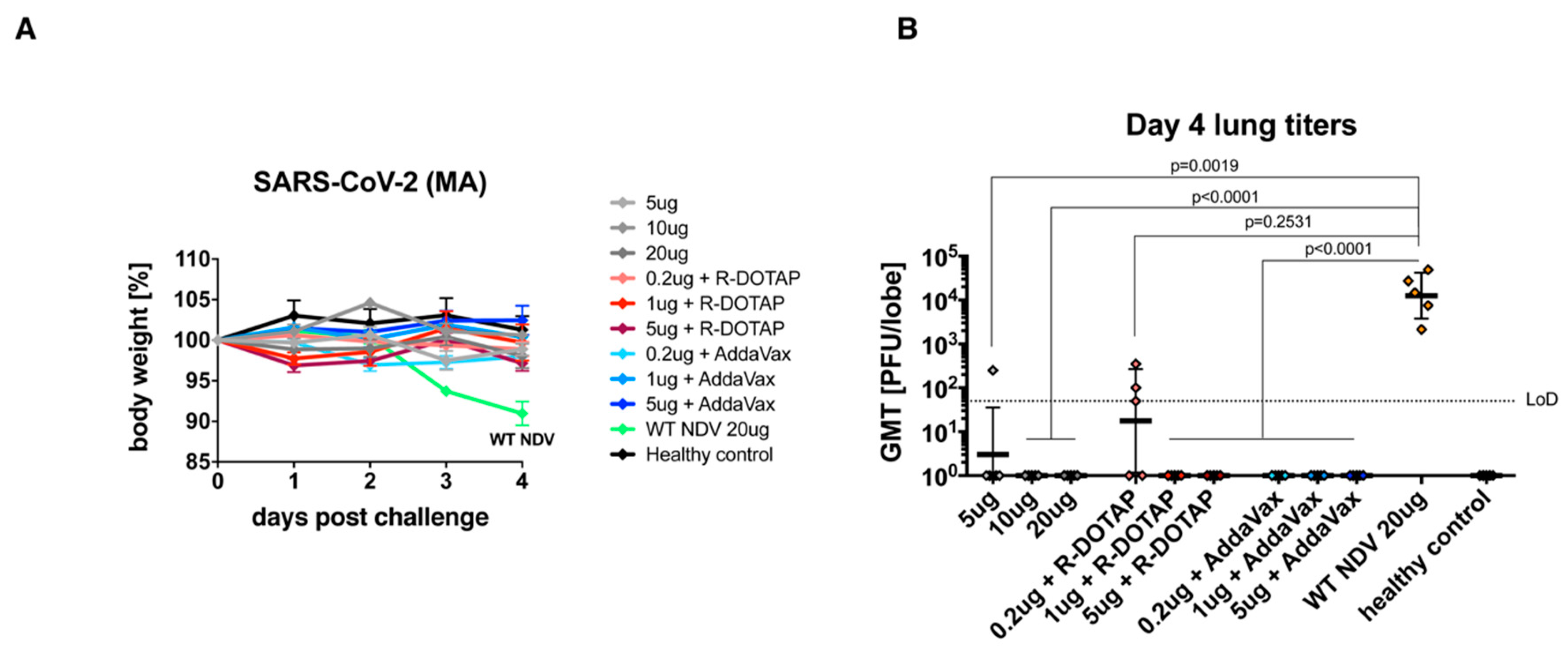
3.3. The Inactivated NDV-S Vaccine Protects Mice from Infection by a Mouse-Adapted SARS-CoV-2 Virus
3.4. The Inactivated NDV-S Vaccine Confers Protection against SARS-CoV-2 Infection in a Hamster Model
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Jackson, L.A.; Anderson, E.J.; Rouphael, N.G.; Roberts, P.C.; Makhene, M.; Coler, R.N.; McCullough, M.P.; Chappell, J.D.; Denison, M.R.; Stevens, L.J.; et al. An mRNA Vaccine against SARS-CoV-2—Preliminary Report. N. Engl. J. Med. 2020, 383, 1920–1931. [Google Scholar] [CrossRef] [PubMed]
- Corbett, K.S.; Edwards, D.K.; Leist, S.R.; Abiona, O.M.; Boyoglu-Barnum, S.; Gillespie, R.A.; Himansu, S.; Schafer, A.; Ziwawo, C.T.; DiPiazza, A.T.; et al. SARS-CoV-2 mRNA vaccine design enabled by prototype pathogen preparedness. Nature 2020, 586, 567–571. [Google Scholar] [CrossRef] [PubMed]
- Gao, Q.; Bao, L.; Mao, H.; Wang, L.; Xu, K.; Yang, M.; Li, Y.; Zhu, L.; Wang, N.; Lv, Z.; et al. Rapid development of an inactivated vaccine candidate for SARS-CoV-2. Science 2020, 369, 77–81. [Google Scholar] [CrossRef] [PubMed]
- Zhu, F.C.; Li, Y.H.; Guan, X.H.; Hou, L.H.; Wang, W.J.; Li, J.X.; Wu, S.P.; Wang, B.S.; Wang, Z.; Wang, L.; et al. Safety, tolerability, and immunogenicity of a recombinant adenovirus type-5 vectored COVID-19 vaccine: A dose-escalation, open-label, non-randomised, first-in-human trial. Lancet 2020, 395, 1845–1854. [Google Scholar] [CrossRef]
- DiNapoli, J.M.; Kotelkin, A.; Yang, L.; Elankumaran, S.; Murphy, B.R.; Samal, S.K.; Collins, P.L.; Bukreyev, A. Newcastle disease virus, a host range-restricted virus, as a vaccine vector for intranasal immunization against emerging pathogens. Proc. Natl. Acad. Sci. USA 2007, 104, 9788–9793. [Google Scholar] [CrossRef] [PubMed]
- Liu, R.Q.; Ge, J.Y.; Wang, J.L.; Shao, Y.; Zhang, H.L.; Wang, J.L.; Wen, Z.Y.; Bu, Z.G. Newcastle disease virus-based MERS-CoV candidate vaccine elicits high-level and lasting neutralizing antibodies in Bactrian camels. J. Integr. Agric. 2017, 16, 2264–2273. [Google Scholar] [CrossRef]
- Li, K.; Li, Z.; Wohlford-Lenane, C.; Meyerholz, D.K.; Channappanavar, R.; An, D.; Perlman, S.; McCray, P.B., Jr.; He, B. Single-Dose, Intranasal Immunization with Recombinant Parainfluenza Virus 5 Expressing Middle East Respiratory Syndrome Coronavirus (MERS-CoV) Spike Protein Protects Mice from Fatal MERS-CoV Infection. mBio 2020, 11, e00554-20. [Google Scholar] [CrossRef]
- Liniger, M.; Zuniga, A.; Tamin, A.; Azzouz-Morin, T.N.; Knuchel, M.; Marty, R.R.; Wiegand, M.; Weibel, S.; Kelvin, D.; Rota, P.A.; et al. Induction of neutralising antibodies and cellular immune responses against SARS coronavirus by recombinant measles viruses. Vaccine 2008, 26, 2164–2174. [Google Scholar] [CrossRef]
- Koch, T.; Dahlke, C.; Fathi, A.; Kupke, A.; Krahling, V.; Okba, N.M.A.; Halwe, S.; Rohde, C.; Eickmann, M.; Volz, A.; et al. Safety and immunogenicity of a modified vaccinia virus Ankara vector vaccine candidate for Middle East respiratory syndrome: An open-label, phase 1 trial. Lancet Infect. Dis. 2020, 20, 827–838. [Google Scholar] [CrossRef]
- Sun, W.; Leist, S.R.; McCroskery, S.; Liu, Y.; Slamanig, S.; Oliva, J.; Amanat, F.; Schaefer, A.; Dinnon, K.; Garcia-Sastre, A.; et al. Newcastle disease virus (NDV) expressing the spike protein of SARS-CoV-2 as vaccine candidate. EBioMedicine (Accepted) 2020. [Google Scholar] [CrossRef]
- Vasievich, E.A.; Ramishetti, S.; Zhang, Y.; Huang, L. Trp2 peptide vaccine adjuvanted with (R)-DOTAP inhibits tumor growth in an advanced melanoma model. Mol. Pharm. 2012, 9, 261–268. [Google Scholar] [CrossRef] [PubMed]
- Smalley Rumfield, C.; Pellom, S.T.; Morillon Ii, Y.M.; Schlom, J.; Jochems, C. Immunomodulation to enhance the efficacy of an HPV therapeutic vaccine. J. Immunother. Cancer 2020, 8, e000612. [Google Scholar] [CrossRef] [PubMed]
- Riehl, M.; Harms, M.; Hanefeld, A.; Baleeiro, R.B.; Walden, P.; Mader, K. Combining R-DOTAP and a particulate antigen delivery platform to trigger dendritic cell activation: Formulation development and in-vitro interaction studies. Int. J. Pharm 2017, 532, 37–46. [Google Scholar] [CrossRef]
- Gandhapudi, S.K.; Ward, M.; Bush, J.P.C.; Bedu-Addo, F.; Conn, G.; Woodward, J.G. Antigen Priming with Enantiospecific Cationic Lipid Nanoparticles Induces Potent Antitumor CTL Responses through Novel Induction of a Type I IFN Response. J. Immunol. 2019, 202, 3524–3536. [Google Scholar] [CrossRef] [PubMed]
- Langley, J.M.; Wang, L.; Aggarwal, N.; Bueso, A.; Chandrasekaran, V.; Cousin, L.; Halperin, S.A.; Li, P.; Liu, A.; McNeil, S.; et al. Immunogenicity and Reactogenicity of an Inactivated Quadrivalent Influenza Vaccine Administered Intramuscularly to Children 6 to 35 Months of Age in 2012–2013: A Randomized, Double-Blind, Controlled, Multicenter, Multicountry, Clinical Trial. J. Pediatr. Infect. Dis Soc. 2015, 4, 242–251. [Google Scholar] [CrossRef] [PubMed]
- Madan, A.; Ferguson, M.; Rheault, P.; Seiden, D.; Toma, A.; Friel, D.; Soni, J.; Li, P.; Innis, B.L.; Schuind, A. Immunogenicity and safety of an AS03-adjuvanted H7N1 vaccine in adults 65years of age and older: A phase II, observer-blind, randomized, controlled trial. Vaccine 2017, 35, 1865–1872. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Chandrasekaran, V.; Domachowske, J.B.; Li, P.; Innis, B.L.; Jain, V.K. Immunogenicity and Safety of an Inactivated Quadrivalent Influenza Vaccine in US Children 6-35 Months of Age During 2013–2014: Results From A Phase II Randomized Trial. J. Pediatr. Infect. Dis. Soc. 2016, 5, 170–179. [Google Scholar] [CrossRef] [PubMed]
- Arifin, M.A.; Mel, M.; Abdul Karim, M.I.; Ideris, A. Production of Newcastle disease virus by Vero cells grown on cytodex 1 microcarriers in a 2-litre stirred tank bioreactor. J. Biomed. Biotechnol. 2010, 2010, 586363. [Google Scholar] [CrossRef]
- Amanat, F.; Stadlbauer, D.; Strohmeier, S.; Nguyen, T.H.O.; Chromikova, V.; McMahon, M.; Jiang, K.; Arunkumar, G.A.; Jurczyszak, D.; Polanco, J.; et al. A serological assay to detect SARS-CoV-2 seroconversion in humans. Nat. Med. 2020, 26, 1033–1036. [Google Scholar] [CrossRef]
- Vijayakumar, G.; Zamarin, D. Design and Production of Newcastle Disease Virus for Intratumoral Immunomodulation. Methods Mol. Biol. 2020, 2058, 133–154. [Google Scholar]
- Dinnon, K.H., 3rd; Leist, S.R.; Schafer, A.; Edwards, C.E.; Martinez, D.R.; Montgomery, S.A.; West, A.; Yount, B.L., Jr.; Hou, Y.J.; Adams, L.E.; et al. A mouse-adapted model of SARS-CoV-2 to test COVID-19 countermeasures. Nature 2020, 586, 560–566. [Google Scholar] [CrossRef] [PubMed]
- Amanat, F.; White, K.M.; Miorin, L.; Strohmeier, S.; McMahon, M.; Meade, P.; Liu, W.C.; Albrecht, R.A.; Simon, V.; Martinez-Sobrido, L.; et al. An In Vitro Microneutralization Assay for SARS-CoV-2 Serology and Drug Screening. Curr. Protoc. Microbiol. 2020, 58, e108. [Google Scholar] [CrossRef]
- Ania Wajnberg, F.A.; Firpo, A.; Altman, D.; Bailey, M.; Mansour, M.; McMahon, M.; Meade, P.; Mendu, D.R.; Muellers, K.; Stadlbauer, D.; et al. SARS-CoV-2 infection induces robust, neutralizing antibody responses that are stable for at least three months. medRxiv 2020. [Google Scholar] [CrossRef]
- Imai, M.; Iwatsuki-Horimoto, K.; Hatta, M.; Loeber, S.; Halfmann, P.J.; Nakajima, N.; Watanabe, T.; Ujie, M.; Takahashi, K.; Ito, M.; et al. Syrian hamsters as a small animal model for SARS-CoV-2 infection and countermeasure development. Proc. Natl. Acad. Sci. USA 2020, 117, 16587–16595. [Google Scholar] [CrossRef] [PubMed]
- Chan, J.F.; Zhang, A.J.; Yuan, S.; Poon, V.K.; Chan, C.C.; Lee, A.C.; Chan, W.M.; Fan, Z.; Tsoi, H.W.; Wen, L.; et al. Simulation of the clinical and pathological manifestations of Coronavirus Disease 2019 (COVID-19) in golden Syrian hamster model: Implications for disease pathogenesis and transmissibility. Clin. Infect. Dis. 2020. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sun, W.; McCroskery, S.; Liu, W.-C.; Leist, S.R.; Liu, Y.; Albrecht, R.A.; Slamanig, S.; Oliva, J.; Amanat, F.; Schäfer, A.; et al. A Newcastle Disease Virus (NDV) Expressing a Membrane-Anchored Spike as a Cost-Effective Inactivated SARS-CoV-2 Vaccine. Vaccines 2020, 8, 771. https://doi.org/10.3390/vaccines8040771
Sun W, McCroskery S, Liu W-C, Leist SR, Liu Y, Albrecht RA, Slamanig S, Oliva J, Amanat F, Schäfer A, et al. A Newcastle Disease Virus (NDV) Expressing a Membrane-Anchored Spike as a Cost-Effective Inactivated SARS-CoV-2 Vaccine. Vaccines. 2020; 8(4):771. https://doi.org/10.3390/vaccines8040771
Chicago/Turabian StyleSun, Weina, Stephen McCroskery, Wen-Chun Liu, Sarah R. Leist, Yonghong Liu, Randy A. Albrecht, Stefan Slamanig, Justine Oliva, Fatima Amanat, Alexandra Schäfer, and et al. 2020. "A Newcastle Disease Virus (NDV) Expressing a Membrane-Anchored Spike as a Cost-Effective Inactivated SARS-CoV-2 Vaccine" Vaccines 8, no. 4: 771. https://doi.org/10.3390/vaccines8040771
APA StyleSun, W., McCroskery, S., Liu, W.-C., Leist, S. R., Liu, Y., Albrecht, R. A., Slamanig, S., Oliva, J., Amanat, F., Schäfer, A., Dinnon, K. H., III, Innis, B. L., García-Sastre, A., Krammer, F., Baric, R. S., & Palese, P. (2020). A Newcastle Disease Virus (NDV) Expressing a Membrane-Anchored Spike as a Cost-Effective Inactivated SARS-CoV-2 Vaccine. Vaccines, 8(4), 771. https://doi.org/10.3390/vaccines8040771






