Occurrence of Antibodies against SARS-CoV-2 in the Domestic Cat Population of Germany
Abstract
1. Introduction
2. Materials and Methods
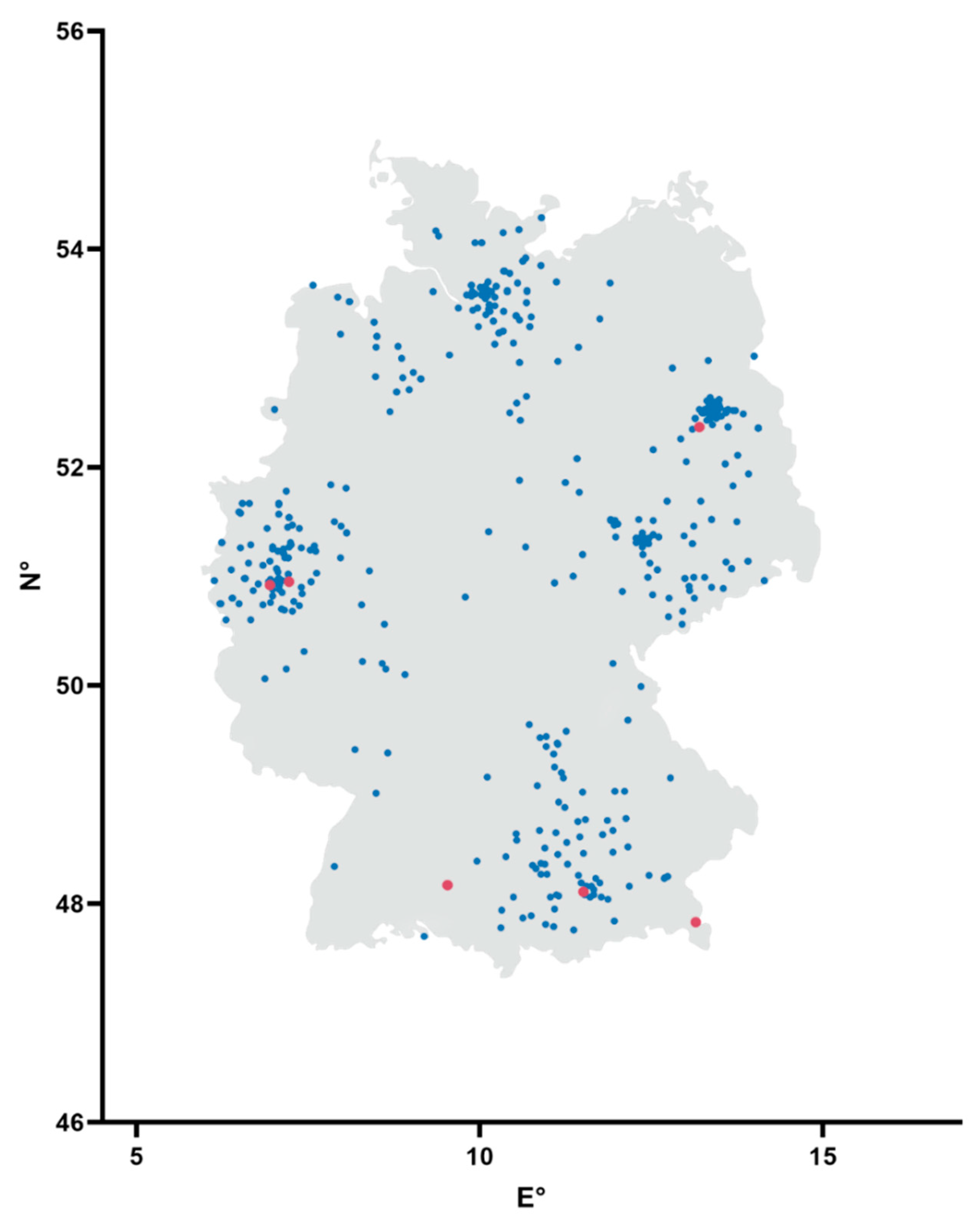
2.1. Serum Samples
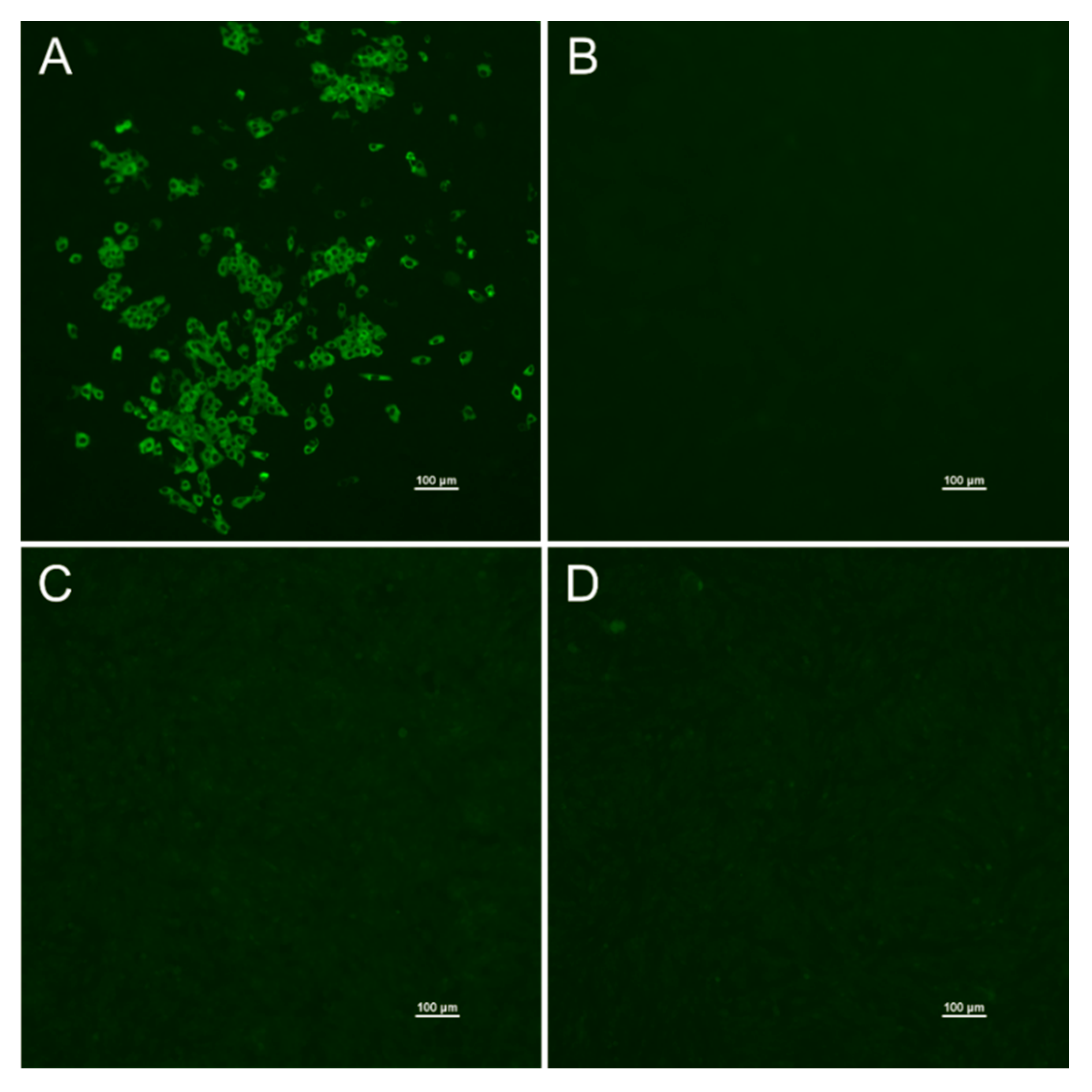
2.2. Antibody Detection
3. Results
3.1. Detection of Antibodies against SARS-CoV-2
3.2. Detection of Antibodies against FCoV by VNT
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Ethical Statement
References
- Krajcarz, M.; Krajcarz, M.; Baca, M.; Baumann, C.; Van Neer, W.; Popovic, D.; Sudoł, M.; Wach, B.; Wilczyński, J.; Wojenka, M.; et al. Ancestors of domestic cats in Neolithic Central Europe: Isotopic evidence of a synanthropic diet. Proc. Natl. Acad. Sci. USA 2020, 117, 201918884. [Google Scholar] [CrossRef] [PubMed]
- Overgaauw, P.A.M.; Vinke, C.M.; Hagen, M.A.E.V.; Lipman, L.J.A. A One Health Perspective on the Human-Companion Animal Relationship with Emphasis on Zoonotic Aspects. Int. J. Environ. Res. Public Health 2020, 17, 3789. [Google Scholar] [CrossRef] [PubMed]
- Chalkowski, K.; Wilson, A.E.; Lepczyk, C.A.; Zohdy, S. Who let the cats out? A global meta-analysis on risk of parasitic infection in indoor versus outdoor domestic cats (Felis catus). Biol. Lett. 2019, 15, 20180840. [Google Scholar] [CrossRef] [PubMed]
- Chomel, B.B. Emerging and Re-Emerging Zoonoses of Dogs and Cats. Animals 2014, 4, 434–445. [Google Scholar] [CrossRef]
- Wu, F.; Zhao, S.; Yu, B.; Chen, Y.M.; Wang, W.; Song, Z.G.; Hu, Y.; Tao, Z.W.; Tian, J.H.; Pei, Y.Y.; et al. A new coronavirus associated with human respiratory disease in China. Nature 2020, 579, 265–269. [Google Scholar] [CrossRef]
- Shi, J.; Wen, Z.; Zhong, G.; Yang, H.; Wang, C.; Huang, B.; Liu, R.; He, X.; Shuai, L.; Sun, Z.; et al. Susceptibility of ferrets, cats, dogs, and other domesticated animals to SARS-coronavirus 2. Science 2020, 368, 1016–1020. [Google Scholar] [CrossRef]
- McNamara, T.; Richt, J.A.; Glickman, L. A Critical Needs Assessment for Research in Companion Animals and Livestock Following the Pandemic of COVID-19 in Humans. Vector Borne Zoonotic Dis. 2020, 20, 393–405. [Google Scholar] [CrossRef]
- Martina, B.E.; Haagmans, B.L.; Kuiken, T.; Fouchier, R.A.; Rimmelzwaan, G.F.; Van Amerongen, G.; Peiris, J.S.; Lim, W.; Osterhaus, A.D. Virology: SARS virus infection of cats and ferrets. Nature 2003, 425, 915. [Google Scholar] [CrossRef]
- Halfmann, P.J.; Hatta, M.; Chiba, S.; Maemura, T.; Fan, S.; Takeda, M.; Kinoshita, N.; Hattori, S.I.; Sakai-Tagawa, Y.; Iwatsuki-Horimoto, K.; et al. Transmission of SARS-CoV-2 in Domestic Cats. N. Engl. J. Med. 2020, 383, 592–594. [Google Scholar] [CrossRef]
- Gaudreault, N.N.; Trujillo, J.D.; Carossino, M.; Meekins, D.A.; Morozov, I.; Madden, D.W.; Indran, S.V.; Bold, D.; Balaraman, V.; Kwon, T.; et al. SARS-CoV-2 infection, disease and transmission in domestic cats. Emerg. Microbes Infect. 2020, 9, 2322–2332. [Google Scholar] [CrossRef]
- Bosco-Lauth, A.M.; Hartwig, A.E.; Porter, S.M.; Gordy, P.W.; Nehring, M.; Byas, A.D.; VandeWoude, S.; Ragan, I.K.; Maison, R.M.; Bowen, R.A. Experimental infection of domestic dogs and cats with SARS-CoV-2: Pathogenesis, transmission, and response to reexposure in cats. Proc. Natl. Acad. Sci. USA 2020, 117, 26382–26388. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Zhang, H.; Gao, J.; Huang, K.; Yang, Y.; Hui, X.; He, X.; Li, C.; Gong, W.; Zhang, Y.; et al. A serological survey of SARS-CoV-2 in cat in Wuhan. Emerg. Microbes Infect. 2020, 9, 2013–2019. [Google Scholar] [CrossRef]
- Fritz, M.; Rosolen, B.; Krafft, E.; Becquart, P.; Elguero, E.; Vratskikh, O.; Denolly, S.; Boson, B.; Vanhomwegen, J.; Gouilh, M.A.; et al. High prevalence of SARS-CoV-2 antibodies in pets from COVID-19+ households. One Health 2020, 11, 100192. [Google Scholar] [CrossRef] [PubMed]
- Sailleau, C.; Dumarest, M.; Vanhomwegen, J.; Delaplace, M.; Caro, V.; Kwasiborski, A.; Hourdel, V.; Chevaillier, P.; Barbarino, A.; Comtet, L.; et al. First detection and genome sequencing of SARS-CoV-2 in an infected cat in France. Transbound. Emerg. Dis. 2020. [Google Scholar] [CrossRef] [PubMed]
- Newman, A.; Smith, D.; Ghai, R.R.; Wallace, R.M.; Torchetti, M.K.; Loiacono, C.; Murrell, L.S.; Carpenter, A.; Moroff, S.; Rooney, J.A.; et al. First Reported Cases of SARS-CoV-2 Infection in Companion Animals—New York, March–April 2020. MMWR Morb. Mortal Wkly. Rep. 2020, 69, 710–713. [Google Scholar] [CrossRef] [PubMed]
- Garigliany, M.; Van Laere, A.S.; Clercx, C.; Giet, D.; Escriou, N.; Huon, C.; van der Werf, S.; Eloit, M.; Desmecht, D. SARS-CoV-2 Natural Transmission from Human to Cat, Belgium, March 2020. Emerg. Infect. Dis. 2020, 26. [Google Scholar] [CrossRef]
- Hosie, M.J.; Epifano, I.; Herder, V.; Orton, R.J.; Stevenson, A.; Johnson, N.; MacDonald, E.; Dunbar, D.; McDonald, M.; Howie, F.; et al. Respiratory disease in cats associated with human-to-cat transmission of SARS-CoV-2 in the UK. bioRxiv 2020. [Google Scholar] [CrossRef]
- Temmam, S.; Barbarino, A.; Maso, D.; Behillil, S.; Enouf, V.; Huon, C.; Jaraud, A.; Chevallier, L.; Backovic, M.; Pérot, P.; et al. Absence of SARS-CoV-2 infection in cats and dogs in close contact with a cluster of COVID-19 patients in a veterinary campus. One Health 2020, 10, 100164. [Google Scholar] [CrossRef]
- Zhao, S.; Li, W.; Schuurman, N.; van Kuppeveld, F.; Bosch, B.J.; Egberink, H. Serological Screening for Coronavirus Infections in Cats. Viruses 2019, 11, 743. [Google Scholar] [CrossRef]
- Wernike, K.; Aebischer, A.; Michelitsch, A.; Hoffmann, D.; Freuling, C.; Balkema-Buschmann, A.; Graaf, A.; Müller, T.; Osterrieder, N.; Rissmann, M.; et al. Multi-species ELISA for the detection of antibodies against SARS-CoV-2 in animals. Transbound. Emerg. Dis. 2020. [Google Scholar] [CrossRef]
- Schlottau, K.; Rissmann, M.; Graaf, A.; Schon, J.; Sehl, J.; Wylezich, C.; Hoper, D.; Mettenleiter, T.C.; Balkema-Buschmann, A.; Harder, T.; et al. SARS-CoV-2 in fruit bats, ferrets, pigs, and chickens: An experimental transmission study. Lancet Microbe 2020. [Google Scholar] [CrossRef]
- WHO. Laboratory Testing for Coronavirus Disease 2019 (COVID-19) in Suspected Human Cases: Interim Guidance, 2 March 2020; World Health Organization: Geneva, Switzerland, 2019. [Google Scholar]
- Robert Koch-Institut. Coronavirus Disease 2019 (COVID-19) Daily Situation Report of the Robert Koch Institute—30/09/2020—UPDATED STATUS FOR GERMANY; Robert Koch-Institut: Berlin, Germany, 2020. [Google Scholar]
- Flaxman, S.; Mishra, S.; Gandy, A.; Unwin, H.J.T.; Mellan, T.A.; Coupland, H.; Whittaker, C.; Zhu, H.; Berah, T.; Eaton, J.W.; et al. Estimating the effects of non-pharmaceutical interventions on COVID-19 in Europe. Nature 2020, 584, 257–261. [Google Scholar] [CrossRef] [PubMed]
- Fischer, B.; Knabbe, C.; Vollmer, T. SARS-CoV-2 IgG seroprevalence in blood donors located in three different federal states, Germany, March to June 2020. Euro Surveill. 2020, 25, 2001285. [Google Scholar] [CrossRef] [PubMed]
- Patterson, E.I.; Elia, G.; Grassi, A.; Giordano, A.; Desario, C.; Medardo, M.; Smith, S.L.; Anderson, E.R.; Prince, T.; Patterson, G.T.; et al. Evidence of exposure to SARS-CoV-2 in cats and dogs from households in Italy. bioRxiv 2020. [Google Scholar] [CrossRef]
- Klein-Richers, U.; Hartmann, K.; Hofmann-Lehmann, R.; Unterer, S.; Bergmann, M.; Rieger, A.; Leutenegger, C.; Pantchev, N.; Balzer, J.; Felten, S. Prevalence of Feline Coronavirus Shedding in German Catteries and Associated Risk Factors. Viruses 2020, 12, 1000. [Google Scholar] [CrossRef] [PubMed]
- Leroy, E.M.; Ar Gouilh, M.; Brugère-Picoux, J. The risk of SARS-CoV-2 transmission to pets and other wild and domestic animals strongly mandates a one-health strategy to control the COVID-19 pandemic. One Health 2020, 10, 100133. [Google Scholar] [CrossRef]
- SARS-Coronavirus (CoV)-2 and Cats. Available online: http://www.abcdcatsvets.org/sars-coronavirus-2-and-cats/ (accessed on 7 December 2020).
| Federal State | Month 2020 | ∑ | |||||
|---|---|---|---|---|---|---|---|
| April | May | June | July | August | September | ||
| Baden-Wuerttemberg | 0/0 | 0/0 | 1/2 | 0/3 | 0/0 | 0/0 | 1/5 |
| Bavaria | 0/0 | 0/39 | 1/18 | 1/32 | 0/0 | 0/9 | 2/98 |
| Berlin | 0/0 | 0/40 | 0/1 | 0/56 | 0/30 | 0/5 | 0/132 |
| Brandenburg | 0/0 | 0/15 | 0/0 | 1/39 | 0/13 | 0/1 | 1/68 |
| Bremen | 0/0 | 0/1 | 0/1 | 0/0 | 0/2 | 0/0 | 0/4 |
| Hamburg | 0/0 | 0/16 | 0/18 | 0/10 | 0/36 | 0/23 | 0/103 |
| Hesse | 0/0 | 0/0 | 0/2 | 0/8 | 0/2 | 0/0 | 0/12 |
| Lower Saxony | 0/0 | 0/13 | 0/9 | 0/1 | 0/7 | 0/14 | 0/44 |
| Mecklenburg-Western Pomerania | 0/1 | 0/2 | 0/0 | 0/2 | 0/0 | 0/2 | 0/7 |
| North Rhine-Westphalia | 0/0 | 0/16 | 1/30 | 1/74 | 0/58 | 0/2 | 2/180 |
| Rhineland-Palatinate | 0/0 | 0/0 | 0/1 | 0/1 | 0/1 | 0/0 | 0/3 |
| Saxony | 0/19 | 0/38 | 0/36 | 0/46 | 0/39 | 0/4 | 0/182 |
| Saxony-Anhalt | 0/2 | 0/5 | 0/4 | 0/8 | 0/9 | 0/1 | 0/29 |
| Schleswig-Holstein | 0/0 | 0/14 | 0/5 | 0/4 | 0/15 | 0/10 | 0/48 |
| Thuringia | 0/0 | 0/0 | 0/1 | 0/2 | 0/2 | 0/0 | 0/5 |
| ∑ | 0/22 | 0/199 | 3/128 | 3/286 | 0/214 | 0/71 | 6/920 |
| Collection Date | RBD ELISA | iIFT SARS-CoV-2 | VNT SARS-CoV-2 | VNT FCoV |
|---|---|---|---|---|
| Absorbance/Result | ND50 | ND50 | ||
| 16 June2020 | 1.292/positive | 1/4096 | neg. 1 | neg. 1 |
| 7 July 2020 | 0.850/positive | 1/512 | 1/101.6 | neg. 1 |
| 3 June 2020 | 0.650/positive | 1/1024 | neg. 1 | neg. 1 |
| 15 July 2020 | 0.369/positive | 1/512 | neg. 1 | neg. 1 |
| 25 July 2020 | 0.334/positive | 1/1024 | 1/20.16 | neg. 1 |
| 16 June 2020 | 0.320/positive | 1/2048 | neg. 1 | neg. 1 |
| 10 September 2020 | 0.242/borderline | neg. 1 | neg. 1 | neg. 1 |
| 9 September 2020 | 0.239/borderline | 1/512 | neg. 1 | neg. 1 |
| 9 September 2020 | 0.225/borderline | neg. 1 | neg. 1 | neg. 1 |
| 8 July 2020 | 0.207/borderline | 1/256 | neg. 1 | neg. 1 |
| 30 April 2020 | 0.201/borderline | neg. 1 | neg. 1 | neg. 1 |
| Federal State | Month 2020 | ∑ | |||||
|---|---|---|---|---|---|---|---|
| April | May | June | July | August | September | ||
| Baden-Wuerttemberg | 0/0 | 0/0 | 0/2 | 0/3 | 0/0 | 0/0 | 0/5 |
| Bavaria | 0/0 | 4/39 | 1/18 | 1/32 | 0/0 | 0/9 | 6/98 |
| Berlin | 0/0 | 2/40 | 0/1 | 0/56 | 0/30 | 0/5 | 2/132 |
| Brandenburg | 0/0 | 0/15 | 0/0 | 0/39 | 1/13 | 0/1 | 1/68 |
| Bremen | 0/0 | 0/1 | 0/1 | 0/0 | 0/2 | 0/0 | 0/4 |
| Hamburg | 0/0 | 0/16 | 0/18 | 0/10 | 1/36 | 0/23 | 1/103 |
| Hesse | 0/0 | 0/0 | 0/2 | 1/8 | 0/2 | 0/0 | 1/12 |
| Lower Saxony | 0/0 | 4/13 | 0/9 | 0/1 | 0/7 | 0/14 | 4/44 |
| Mecklenburg-Western Pomerania | 0/1 | 0/2 | 0/0 | 0/2 | 0/0 | 1/2 | 1/7 |
| North Rhine-Westphalia | 0/0 | 1/16 | 0/30 | 1/74 | 0/58 | 0/2 | 2/180 |
| Rhineland-Palatinate | 0/0 | 0/0 | 0/1 | 0/1 | 0/1 | 0/0 | 0/3 |
| Saxony | 0/19 | 1/38 | 1/36 | 2/46 | 1/39 | 0/4 | 5/182 |
| Saxony-Anhalt | 0/2 | 0/5 | 0/4 | 0/8 | 0/9 | 0/1 | 0/29 |
| Schleswig-Holstein | 0/0 | 0/14 | 0/5 | 1/4 | 0/15 | 1/10 | 2/48 |
| Thuringia | 0/0 | 0/0 | 0/1 | 0/2 | 0/2 | 0/0 | 0/5 |
| ∑ | 0/22 | 12/199 | 2/128 | 6/286 | 3/214 | 2/71 | 25/920 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Michelitsch, A.; Hoffmann, D.; Wernike, K.; Beer, M. Occurrence of Antibodies against SARS-CoV-2 in the Domestic Cat Population of Germany. Vaccines 2020, 8, 772. https://doi.org/10.3390/vaccines8040772
Michelitsch A, Hoffmann D, Wernike K, Beer M. Occurrence of Antibodies against SARS-CoV-2 in the Domestic Cat Population of Germany. Vaccines. 2020; 8(4):772. https://doi.org/10.3390/vaccines8040772
Chicago/Turabian StyleMichelitsch, Anna, Donata Hoffmann, Kerstin Wernike, and Martin Beer. 2020. "Occurrence of Antibodies against SARS-CoV-2 in the Domestic Cat Population of Germany" Vaccines 8, no. 4: 772. https://doi.org/10.3390/vaccines8040772
APA StyleMichelitsch, A., Hoffmann, D., Wernike, K., & Beer, M. (2020). Occurrence of Antibodies against SARS-CoV-2 in the Domestic Cat Population of Germany. Vaccines, 8(4), 772. https://doi.org/10.3390/vaccines8040772





