The Immunogenic and Immunoprotective Activities of Recombinant Chimeric T. gondii Proteins Containing AMA1 Antigen Fragments
Abstract
1. Introduction
2. Materials and Methods
2.1. Mice
2.2. Ethical Statement
2.3. Parasites
2.4. Production of Recombinant Chimeric Proteins
2.5. Mouse Immunization and Challenge
2.6. Brain Cyst Enumeration
2.7. Determination of the Specific IgG response and Isotype Profile after Immunization
2.8. In Vitro Splenocyte Proliferation
2.9. Determination of Cytokine Production by Stimulated Splenocytes
2.10. Statistical Analysis
3. Results
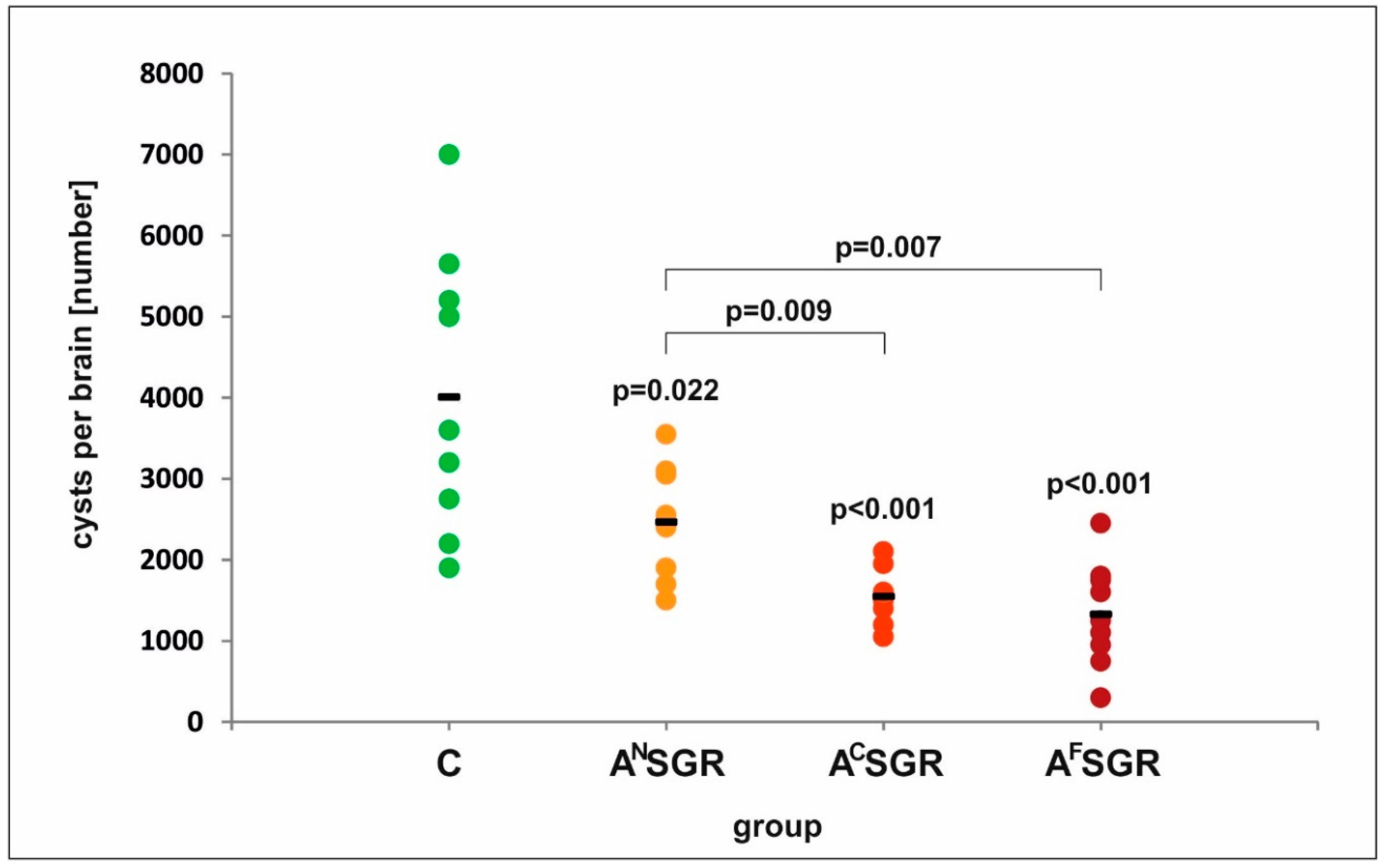
3.1. Protection against Cyst Formation in Mice
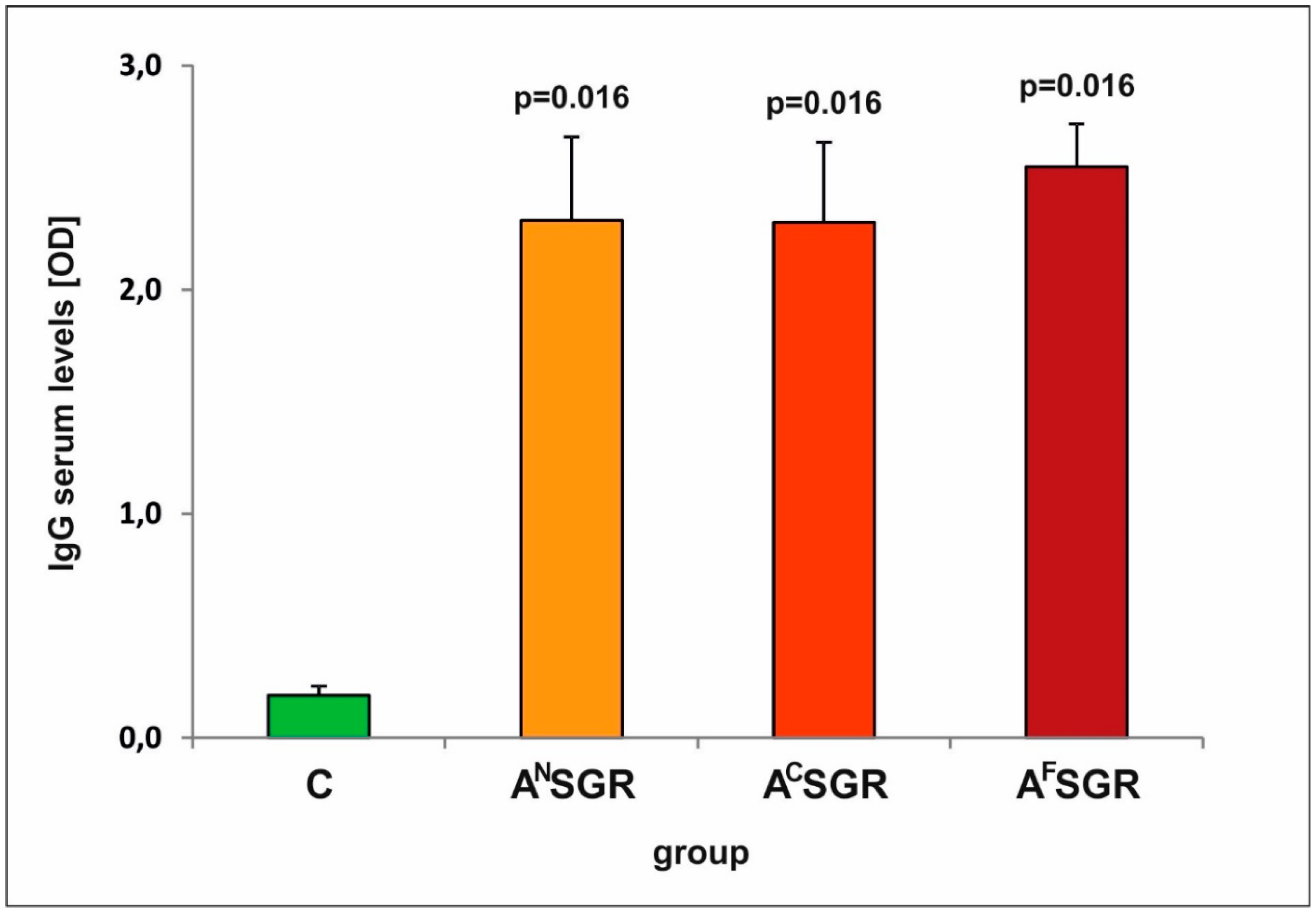
3.2. Analysis of the IgG production and Isotype Profile
3.3. Cellular Response Induced by Vaccination
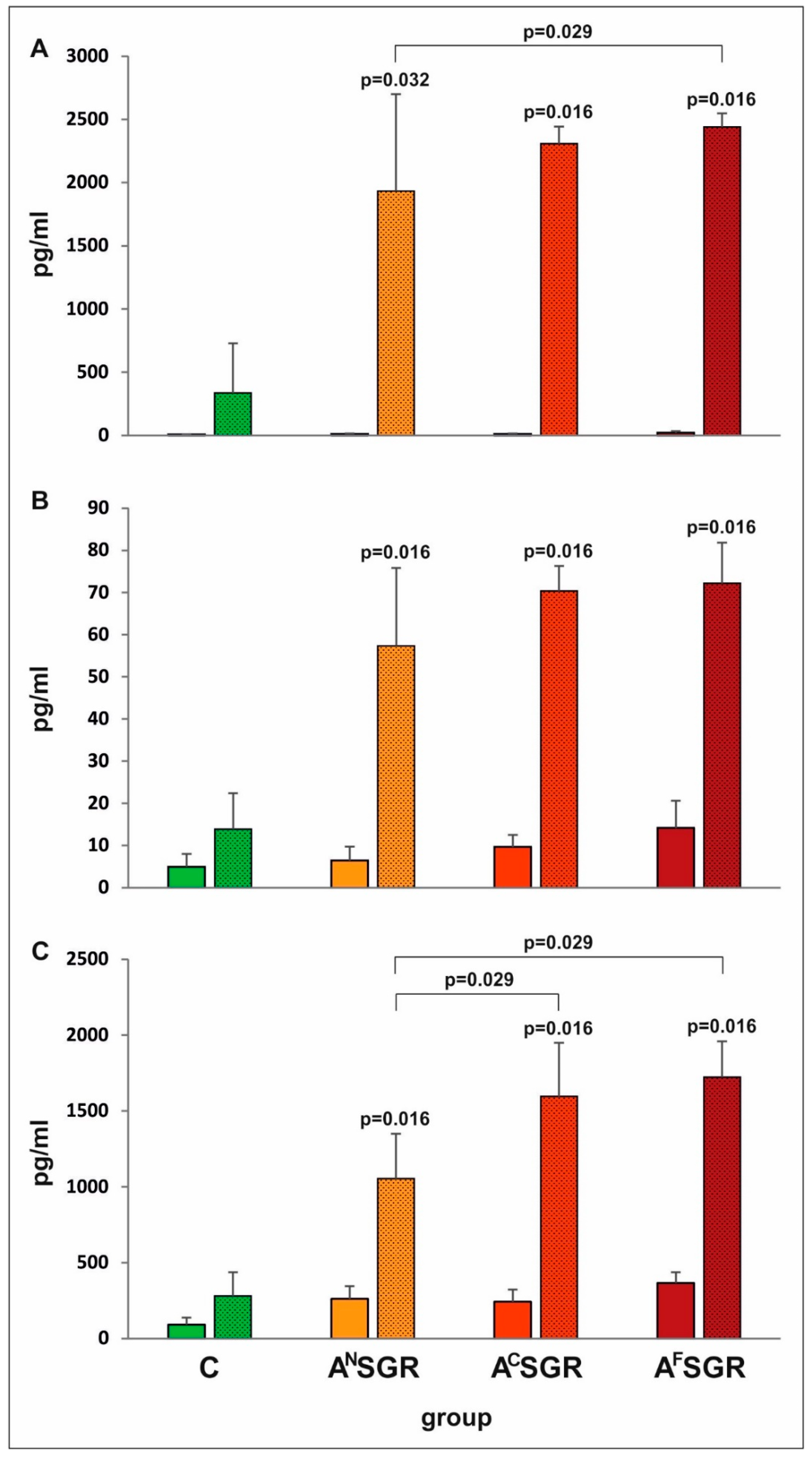
3.4. Determination of IL−2, IFN-γ and IL−10 In Vitro Synthesis
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Halonen, S.K.; Weiss, L.M. Toxoplasmosis. Handb. Clin. Neurol. 2013, 114, 125–145. [Google Scholar]
- Davies, A.P.; Chalmers, R.M. Cryptosporidiosis. BMJ 2009, 339, b4168. [Google Scholar] [CrossRef]
- Foreyt, W.J. Coccidiosis and cryptosporidiosis in sheep and goats. Vet. Clin. N. Am. Food Anim. Pract. 1990, 6, 655–670. [Google Scholar] [CrossRef]
- Anderson, M.; Barr, B.; Rowe, J.; Conrad, P. Neosporosis in dairy cattle. Jpn. J. Vet. Res. 2012, 60, S51–S54. [Google Scholar]
- Schnittger, L.; Rodriguez, A.E.; Florin-Christensen, M.; Morrison, D.A. Babesia: A world emerging. Infect. Genet. Evol. 2012, 12, 1788–1809. [Google Scholar] [CrossRef]
- Rich, S.M.; Leendertz, F.H.; Xu, G.; LeBreton, M.; Djoko, C.F.; Aminake, M.N.; Takang, E.E.; Diffo, J.L.; Pike, B.L.; Rosenthal, B.M.; et al. The origin of malignant malaria. Proc. Natl. Acad. Sci. USA 2009, 106, 14902–14907. [Google Scholar] [CrossRef]
- Buxton, D.; Innes, E.A. A commercial vaccine for ovine toxoplasmosis. Parasitology 1995, 110, S11–S16. [Google Scholar] [CrossRef]
- Li, Y.; Zhou, H. Moving towards improved vaccines for Toxoplasma gondii. Expert Opin. Biol. Ther. 2018, 18, 273–280. [Google Scholar] [CrossRef]
- Antczak, M.; Dzitko, K.; Długońska, H. Human toxoplasmosis-searching for novel chemotherapeutics. Biomed. Pharmacother. 2016, 82, 677–684. [Google Scholar] [CrossRef]
- Alday, P.H.; Doggett, J.S. Drugs in development for toxoplasmosis: Advances, challenges, and current status. Drug Des. Devel. Ther. 2017, 11, 273–293. [Google Scholar] [CrossRef]
- Garcia, J.L. Vaccination concepts against Toxoplasma gondii. Expert Rev. Vaccines 2009, 8, 215–225. [Google Scholar] [CrossRef]
- Zhang, N.Z.; Chen, J.; Wang, M.; Petersen, E.; Zhu, X.Q. Vaccines against Toxoplasma gondii: New developments and perspectives. Expert Rev. Vaccines 2013, 12, 1287–1299. [Google Scholar] [CrossRef]
- Morrison, W.I.; Tomley, F. Development of vaccines for parasitic diseases of animals: Challenges and opportunities. Parasite Immunol. 2016, 38, 707–708. [Google Scholar] [CrossRef]
- Pereira, K.S.; Franco, R.M.; Leal, D.A. Transmission of toxoplasmosis (Toxoplasma gondii) by foods. Adv. Food Nutr. Res. 2010, 60, 1–19. [Google Scholar]
- Jones, J.L.; Dubey, J.P. Foodborne toxoplasmosis. Clin. Infect. Dis. 2012, 55, 845–851. [Google Scholar] [CrossRef]
- Serranti, D.; Buonsenso, D.; Valentini, P. Congenital toxoplasmosis treatment. Eur. Rev. Med. Pharmacol. Sci. 2011, 15, 193–198. [Google Scholar]
- Khurana, S.; Batra, N. Toxoplasmosis in organ transplant recipients: Evaluation, implication, and prevention. Trop. Parasitol. 2016, 6, 123–128. [Google Scholar] [CrossRef]
- Tu, V.; Yakubu, R.; Weiss, L.M. Observations on bradyzoite biology. Microbes Infect. 2018, 20, 466–476. [Google Scholar] [CrossRef]
- Dubey, J.P. History of the discovery of the life cycle of Toxoplasma gondii. Int. J. Parasitol. 2009, 39, 877–882. [Google Scholar] [CrossRef]
- Dubey, J.P. The history of Toxoplasma gondii—The first 100 years. J. Eukaryot. Microbiol. 2008, 55, 467–475. [Google Scholar] [CrossRef]
- Sibley, L.D. Toxoplasma gondii: Perfecting an intracellular life style. Traffic 2003, 4, 581–586. [Google Scholar] [CrossRef]
- Peng, H.J.; Chen, X.G.; Lindsay, D.S. A review: Competence, compromise, and concomitance-reaction of the host cell to Toxoplasma gondii infection and development. J. Parasitol. 2011, 97, 620–628. [Google Scholar] [CrossRef]
- Gavinet, M.F.; Robert, F.; Firtion, G.; Delouvrier, E.; Hennequin, C.; Maurin, J.R.; Tourte-Schaefer, C.; Dupouy-Camet, J. Congenital toxoplasmosis due to maternal reinfection during pregnancy. J. Clin. Microbiol. 1997, 35, 1276–1277. [Google Scholar] [CrossRef]
- Lindsay, D.S.; Dubey, J.P. Toxoplasma gondii: The changing paradigm of congenital toxoplasmosis. Parasitology 2011, 138, 1829–1831. [Google Scholar] [CrossRef]
- Montoya, J.G.; Remington, J.S. Management of Toxoplasma gondii infection during pregnancy. Clin. Infect. Dis. 2008, 47, 554–566. [Google Scholar] [CrossRef]
- McCabe, R.E.; Brooks, R.G.; Dorfman, R.F.; Remington, J.S. Clinical spectrum in 107 cases of toxoplasmic lymphadenopathy. Rev. Infect. Dis. 1987, 9, 754–774. [Google Scholar] [CrossRef]
- Flegr, J.; Prandota, J.; Sovičková, M.; Israili, Z.H. Toxoplasmosis—A global threat. Correlation of latent toxoplasmosis with specific disease burden in a set of 88 countries. PLoS ONE 2014, 9, e90203. [Google Scholar]
- Montoya, J.G.; Liesenfeld, O. Toxoplasmosis. Lancet 2004, 363, 1965–1976. [Google Scholar] [CrossRef]
- Konstantinovic, N.; Guegan, H.; Stäjner, T.; Belaz, S.; Robert-Gangneux, F. Treatment of toxoplasmosis: Current options and future perspectives. Food Waterborne Parasitol. 2019, 15, e00036. [Google Scholar] [CrossRef]
- Dunay, I.R.; Gajurel, K.; Dhakal, R.; Liesenfeld, O.; Montoya, J.G. Treatment of toxoplasmosis: Historical perspective, animal models, and current clinical practice. Clin. Microbiol. Rev. 2018, 31, e00057-17. [Google Scholar] [CrossRef]
- Paneth, A.; Węglińska, L.; Bekier, A.; Stefaniszyn, E.; Wujec, M.; Trotsko, N.; Dzitko, K. Systematic identification of thiosemicarbazides for inhibition of Toxoplasma gondii growth in vitro. Molecules 2019, 24, 614. [Google Scholar] [CrossRef]
- Dzitko, K.; Paneth, A.; Plech, T.; Pawełczyk, J.; Węglińska, L.; Paneth, P. Triazole-based compound as a candidate to develop novel medicines to treat toxoplasmosis. Antimicrob. Agents Chemother. 2014, 58, 7583–7585. [Google Scholar] [CrossRef] [PubMed]
- Oz, H.S. Toxoplasmosis complications and novel therapeutic synergism combination of diclazuril plus atovaquone. Front. Microbiol. 2014, 5, 484. [Google Scholar] [CrossRef]
- Innes, E.A.; Vermeulen, A.N. Vaccination as a control strategy against the coccidial parasites Eimeria, Toxoplasma and Neospora. Parasitology 2006, 133, S145–S168. [Google Scholar] [CrossRef]
- Gatkowska, J.; Dzitko, K.; Ferra, B.T.; Holec-Gąsior, L.; Kawka, M.; Dziadek, B. The impact of the antigenic composition of chimeric proteins on their immunoprotective activity against chronic toxoplasmosis in mice. Vaccines 2019, 7, 154. [Google Scholar] [CrossRef] [PubMed]
- Mital, J.; Meissner, M.; Soldati, D.; Ward, G.E. Conditional expression of Toxoplasma gondii apical membrane antigen-1 (TgAMA1) demonstrates that TgAMA1 plays a critical role in host cell invasion. Mol. Biol. Cell. 2005, 16, 4341–4349. [Google Scholar] [CrossRef]
- Bargieri, D.Y.; Andenmatten, N.; Lagal, V.; Thiberge, S.; Whitelaw, J.A.; Tardieux, I.; Meissner, M.; Ménardet, R. Apical membrane antigen1 mediates apicomplexan parasite attachment but is dispensable for host cell invasion. Nat. Commun. 2013, 4, 2552. [Google Scholar] [CrossRef]
- Krishnamurthy, S.; Deng, B.; Del Rio, R.; Buchholz, K.R.; Treeck, M.; Urban, S.; Boothroyd, J.; Lam, Y.-W.; Ward, G.E. Not a simple tether: Binding of Toxoplasma gondii AMA1 to RON2 during Invasion protects AMA1 from rhomboid-mediated cleavage and leads to dephosphorylation of its cytosolic tail. mBio 2016, 7, e00754–e00816. [Google Scholar] [CrossRef]
- Dautu, G.; Munyaka, B.; Carmen, G.; Zhang, G.; Omata, Y.; Xuenan, X.; Igarashi, M. Toxoplasma gondii: DNA vaccination with genes encoding antigens MIC2, M2AP, AMA1 and BAG1 and evaluation of their immunogenic potential. Exp. Parasitol. 2007, 116, 273–282. [Google Scholar] [CrossRef]
- Roozbehani, M.; Falak, R.; Mohammadi, M.; Hemphill, A.; Razmjou, E.; Meamar, A.R.; Masoori, L.; Khoshmirsafa, M.; Moradi, M.; Gharavi, M.J. Characterization of a multi-epitope peptide with selective MHC-binding capabilities encapsulated in PLGA nanoparticles as a novel vaccine candidate against Toxoplasma gondii infection. Vaccine 2018, 36, 6124–6132. [Google Scholar] [CrossRef]
- Kim, M.-J.; Lee, S.-H.; Kang, H.-J.; Chu, K.-B.; Park, H.; Jin, H.; Moon, E.-K.; Kim, S.S.; Quan, F.-S. Virus-like particle vaccine displaying Toxoplasma gondii apical membrane antigen 1 induces protection against T. gondii ME49 infection in mice. Microb. Pathog. 2020, 142, 104090. [Google Scholar] [CrossRef]
- Ferra, B.T.; Holec-Gąsior, L.; Gatkowska, J.; Dziadek, B.; Dzitko, K.; Grąźlewska, W.; Lautenbach, D. The first study on the usefulness of recombinant tetravalent chimeric proteins containing fragments of SAG2, GRA1, ROP1 and AMA1 antigens in the detection of specific anti-Toxoplasma gondii antibodies in mouse and human sera. PLoS ONE 2019, 14, e0217866. [Google Scholar] [CrossRef]
- Dziadek, B.; Gatkowska, J.; Brzostek, A.; Dziadek, J.; Dzitko, K.; Grzybowski, M.; Dlugonska, H. Evaluation of three recombinant multi-antigenic vaccines composed of surface and secretory antigens of Toxoplasma gondii in murine models of experimental toxoplasmosis. Vaccine 2011, 29, 821–830. [Google Scholar] [CrossRef]
- Foroutan, M.; Ghaffarifar, F.; Sharifi, Z.; Dalimi, A.; Jorjani, O. Rhoptry antigens as Toxoplasma gondii vaccine target. Clin. Exp. Vaccine Res. 2019, 8, 4–26. [Google Scholar] [CrossRef]
- Wang, J.-L.; Zhang, N.-Z.; Li, T.-T.; He, J.-J.; Elsheikha, H.M.; Zhu, X.-Q. Advances in the development of anti-Toxoplasma gondii vaccines: Challenges, opportunities, and perspectives. Trends Parasitol. 2019, 35, 239–253. [Google Scholar] [CrossRef]
- Allahyari, M.; Mohabati, R.; Amiri, S.; Esmaeili Rastaghi, A.R.; Babaie, J.; Mahdavi, M.; Vatanara, A.; Golkar, M. Synergistic efect of rSAG1 and rGRA2 antigens formulated in PLGA microspheres in eliciting immune protection against Toxoplasama gondii. Exp. Parasitol. 2016, 170, 236–246. [Google Scholar] [CrossRef]
- Lagal, V.; Dinis, M.; Cannella, D.; Bargieri, D.; Gonzalez, V.; Andenmatten, N.; Meissner, M.; Tardieux, I. AMA1-deficient Toxoplasma gondii parasites transiently colonize mice and trigger an innate immune response that leads to long-lasting protective immunity. Infect. Immun. 2015, 83, 2475–2486. [Google Scholar] [CrossRef]
- Remarque, E.J.; Faber, B.W.; Kocken, C.H.; Thomas, A.W. Apical membrane antigen 1: A malaria vaccine candidate in review. Trends Parasitol. 2008, 24, 74–84. [Google Scholar] [CrossRef]
- Sagara, I.; Dicko, A.; Ellis, R.D.; Fay, M.P.; Diawara, S.I.; Assadou, M.H.; Sissoko, M.S.; Kone, M.; Diallo, A.I.; Saye, R.; et al. A randomized controlled phase 2 trial of the blood stage AMA1-C1/Alhydrogel malaria vaccine in children in Mali. Vaccine 2009, 27, 3090–3098. [Google Scholar] [CrossRef]
- Ferra, B.; Holec-Gąsior, L.; Gatkowska, J.; Dziadek, B.; Dzitko, K. Toxoplasma gondii recombinant antigen AMA1: Diagnostic utility of protein fragments for the detection of IgG and IgM antibodies. Pathogens 2020, 9, 43. [Google Scholar] [CrossRef]
- Vetrivel, U.; Muralikumar, S.; Mahalakshmi, B.; Lily Therese, K.; Madhavan, H.N.; Alameen, M.; Thirumudi, I. Multilevel precision-based rational design of chemical inhibitors targeting the hydrophobic cleft of Toxoplasma gondii apical membrane antigen 1 (AMA1). Genom. Inform. 2016, 14, 53–61. [Google Scholar] [CrossRef]
- Crawford, J.; Tonkin, M.L.; Grujic, O.; Boulanger, M.J. Structural characterization of apical membrane antigen 1 (AMA1) from Toxoplasma gondii. J. Biol. Chem. 2010, 285, 15644–15652. [Google Scholar] [CrossRef]
- Carruthers, V.; Boothroyd, J.C. Pulling together: An integrated model of Toxoplasma cell invasion. Curr. Opin. Microbiol. 2007, 10, 83–89. [Google Scholar] [CrossRef]
- Liu, Q.; Li, F.-C.; Zhou, C.-X.; Zhu, X.-Q. Research advances in interactions related to Toxoplasma gondii microneme proteins. Exp. Parasitol. 2017, 176, 89–98. [Google Scholar] [CrossRef]
- Rosenberg, C.; De Craeye, S.; Jongert, E.; Gargano, N.; Beghetto, E.; Del Porto, P.; Vorup-Jensen, T.; Petersen, E. Induction of partial protection against infection with Toxoplasma gondii genotype II by DNA vaccination with recombinant chimeric tachyzoite antigens. Vaccine 2009, 27, 2489–2498. [Google Scholar] [CrossRef]
- Scheiblhofer, S.; Laimer, J.; Machado, Y.; Weiss, R.; Thalhamer, J. Influence of protein fold stability on immunogenicity and its implications for vaccine design. Expert Rev. Vaccines 2017, 16, 479–489. [Google Scholar] [CrossRef]
| Mouse/Group | Antibody Titer | ||
|---|---|---|---|
| IgG1 | IgG2a | IgG1/IgG2a | |
| ANSGR | |||
| mouse 1 | >204,800 | 51,200 | >4/1 |
| mouse 2 | 204,800 | 25,600 | 8/1 |
| mouse 3 | 204,800 | 6400 | 32/1 |
| mouse 4 | 102,400 | 6400 | 16/1 |
| ACSGR | |||
| mouse 1 | >204,800 | 12,800 | >16/1 |
| mouse 2 | 204,800 | 6400 | 32/1 |
| mouse 3 | >204,800 | 6400 | >32/1 |
| mouse 4 | 204,800 | 6400 | 32/1 |
| AFSGR | |||
| mouse 1 | >204,800 | 25,600 | >8/1 |
| mouse 2 | >204,800 | 102,400 | >2/1 |
| mouse 3 | 204,800 | 25,600 | 8/1 |
| mouse 4 | 204,800 | 12,800 | 16/1 |
| Antigen/Group | Mean OD570 ± SD a | |||
|---|---|---|---|---|
| Control | ANSGR | ACSGR | AFSGR | |
| Medium | 0.254 ± 0.048 | 0.249 ± 0.021 | 0.215 ± 0.043 | 0.263 ± 0.058 |
| TLA | 0.505 ± 0.048 | 0.505 ± 0.038 | 0.542 ± 0.039 | 0.550 ± 0.044 a |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gatkowska, J.; Dzitko, K.; Ferra, B.T.; Holec-Gąsior, L.; Kawka, M.; Dziadek, B. The Immunogenic and Immunoprotective Activities of Recombinant Chimeric T. gondii Proteins Containing AMA1 Antigen Fragments. Vaccines 2020, 8, 724. https://doi.org/10.3390/vaccines8040724
Gatkowska J, Dzitko K, Ferra BT, Holec-Gąsior L, Kawka M, Dziadek B. The Immunogenic and Immunoprotective Activities of Recombinant Chimeric T. gondii Proteins Containing AMA1 Antigen Fragments. Vaccines. 2020; 8(4):724. https://doi.org/10.3390/vaccines8040724
Chicago/Turabian StyleGatkowska, Justyna, Katarzyna Dzitko, Bartłomiej Tomasz Ferra, Lucyna Holec-Gąsior, Malwina Kawka, and Bożena Dziadek. 2020. "The Immunogenic and Immunoprotective Activities of Recombinant Chimeric T. gondii Proteins Containing AMA1 Antigen Fragments" Vaccines 8, no. 4: 724. https://doi.org/10.3390/vaccines8040724
APA StyleGatkowska, J., Dzitko, K., Ferra, B. T., Holec-Gąsior, L., Kawka, M., & Dziadek, B. (2020). The Immunogenic and Immunoprotective Activities of Recombinant Chimeric T. gondii Proteins Containing AMA1 Antigen Fragments. Vaccines, 8(4), 724. https://doi.org/10.3390/vaccines8040724






