Different Long-Term Duration of Seroprotection against Neisseria meningitidis in Adolescents and Middle-Aged Adults after a Single Meningococcal ACWY Conjugate Vaccination in The Netherlands
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design and Participants
2.2. Serological Analysis
2.3. Mathematical Model
2.4. Statistical Analysis
3. Results
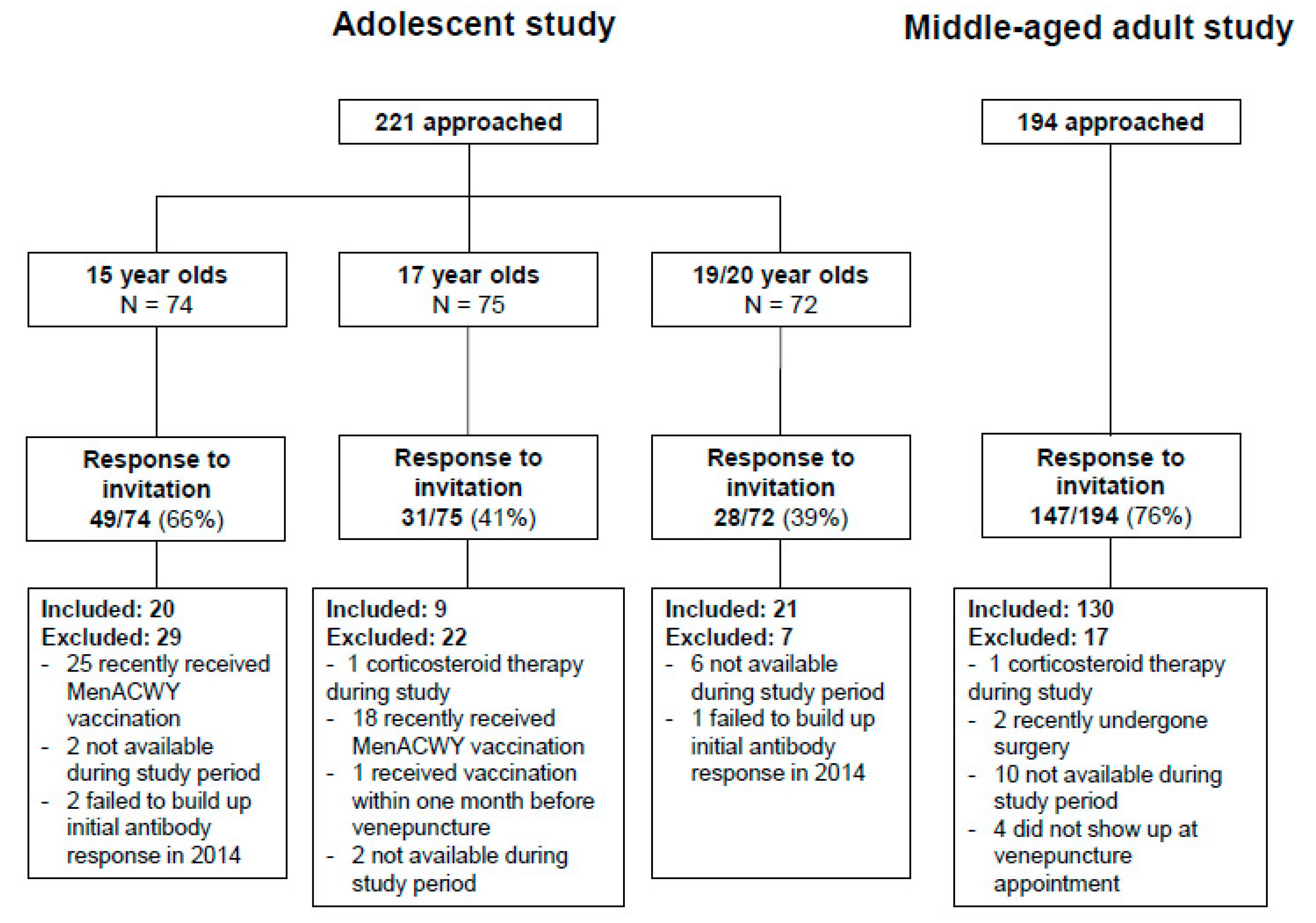
3.1. Study Participants
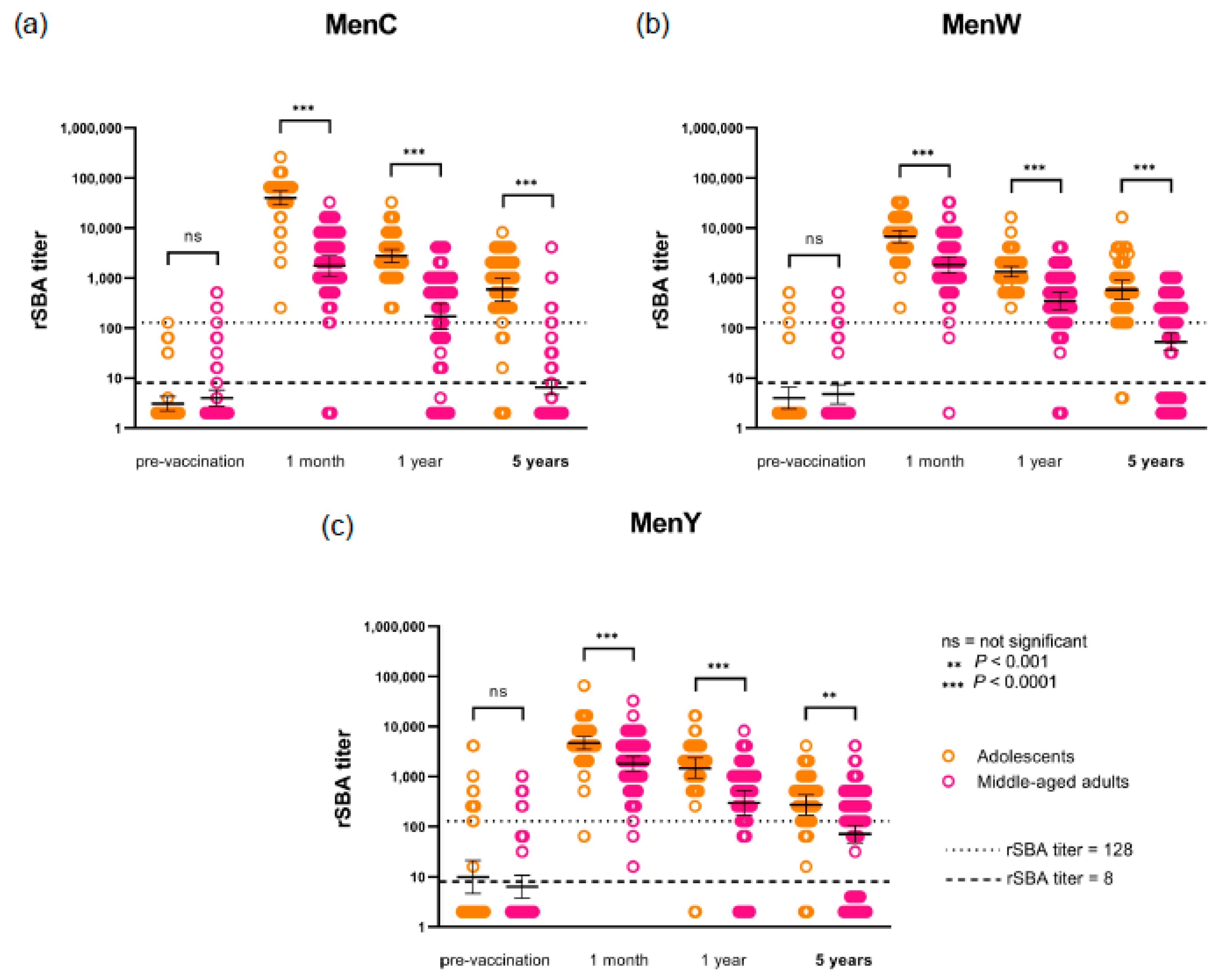
3.2. Persistence of Antibodies after MenACWY-TT Vaccination
3.3. Waning of Functional Antibodies and Duration of Protection
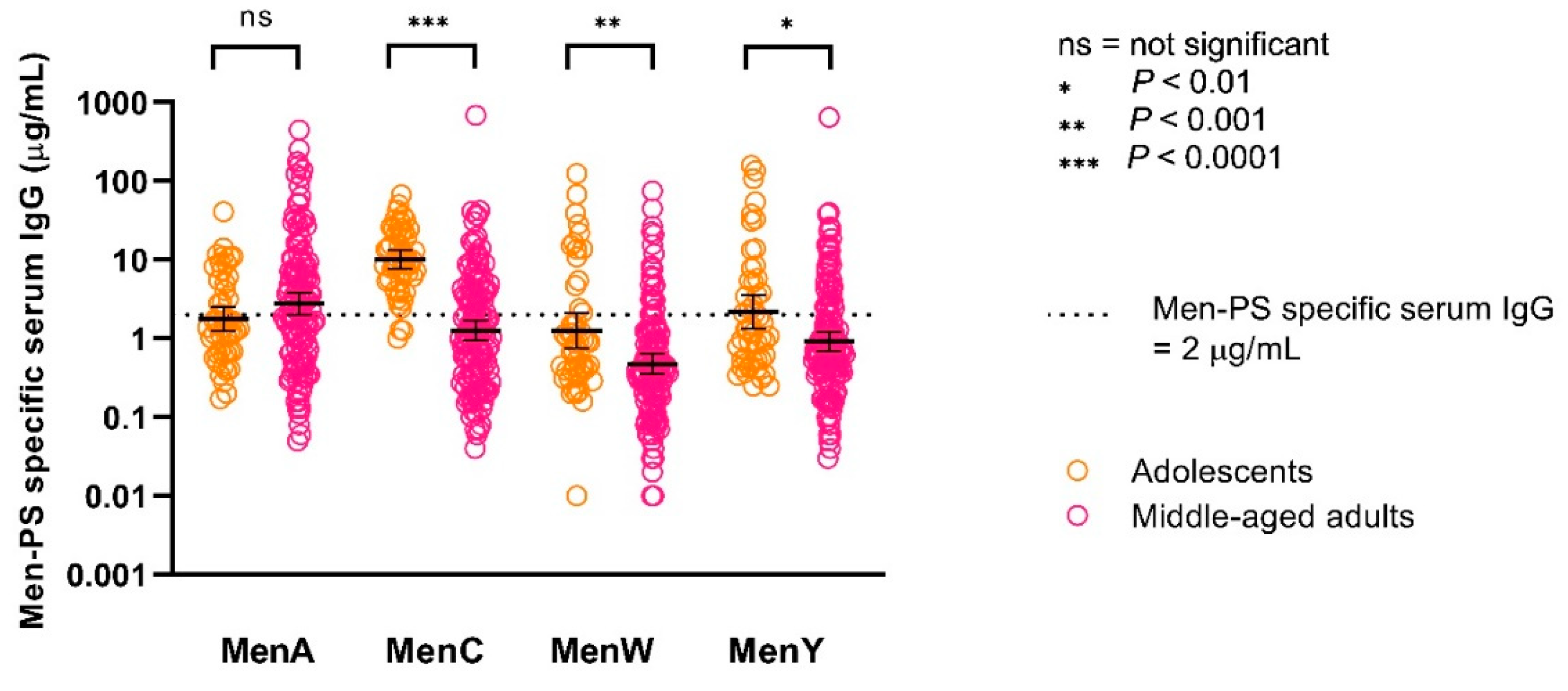
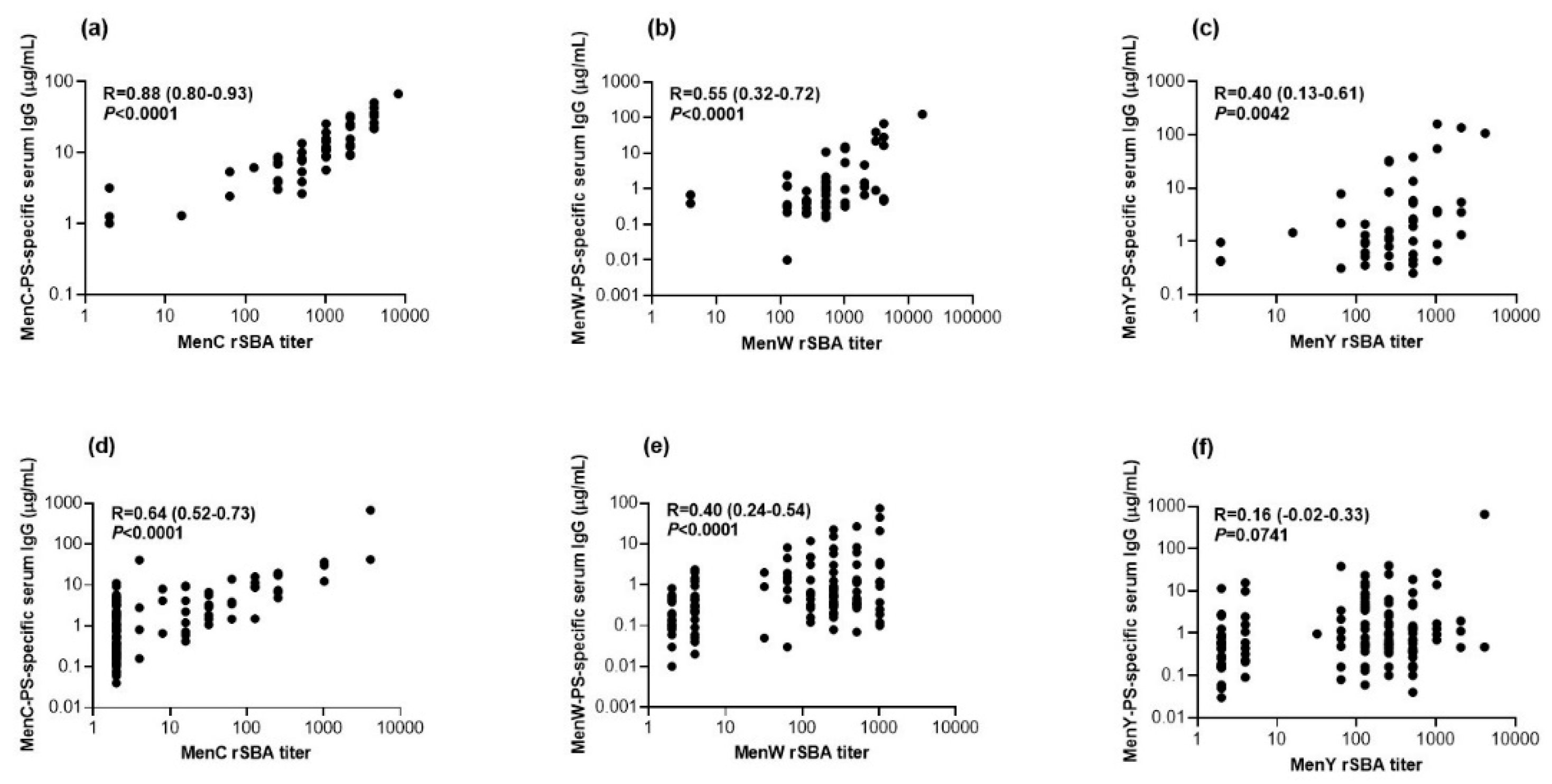
3.4. Correlation between Functional Antibodies and Serum IgG Concentrations
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Stephens, D.S.; Greenwood, B.; Brandtzaeg, P. Epidemic meningitis, meningococcaemia, and Neisseria meningitidis. Lancet 2007, 369, 2196–2210. [Google Scholar] [CrossRef]
- Brandtzaeg, P. Pathogenesis and Pathophysiology of Invasive Meningococcal. Handb. Meningococcal Dis. 2006, 427. [Google Scholar]
- Rosenstein, N.E.; Perkins, B.A.; Stephens, D.S.; Popovic, T.; Hughes, J.M. Meningococcal Disease. N. Eng. J. Med. 2001, 344, 1378–1388. [Google Scholar] [CrossRef]
- Edmond, K.; Clark, A.; Korczak, V.S.; Sanderson, C.; Griffiths, U.K.; Rudan, I. Global and regional risk of disabling sequelae from bacterial meningitis: A systematic review and meta-analysis. Lancet. Infect. Dis. 2010, 10, 317–328. [Google Scholar] [CrossRef]
- Perrett, K.P.; Jin, C.; Clutterbuck, E.; John, T.M.; Winter, A.P.; Kibwana, E.; Yu, L.M.; Curtis, N.; Pollard, A.J. B cell memory to a serogroup C meningococcal conjugate vaccine in childhood and response to booster: Little association with serum IgG antibody. J. Immunol. 2012, 189, 2673–2681. [Google Scholar] [CrossRef]
- Pollard, A.J.; Perrett, K.P.; Beverley, P.C. Maintaining protection against invasive bacteria with protein-polysaccharide conjugate vaccines. Nat. Rev. Immunol. 2009, 9, 213–220. [Google Scholar] [CrossRef]
- Lewis, L.; Ram, S. Meningococcal disease and the complement system. Virulence 2013, 5. [Google Scholar] [CrossRef]
- McIntosh, E.D.; Broker, M.; Wassil, J.; Welsch, J.A.; Borrow, R. Serum bactericidal antibody assays—The role of complement in infection and immunity. Vaccine 2015, 33, 4414–4421. [Google Scholar] [CrossRef]
- Erlich, K.S.; Congeni, B.L. Importance of circulating antibodies in protection against meningococcal disease. Hum. Vaccines Immunother. 2012, 8, 1029–1035. [Google Scholar] [CrossRef]
- Pichichero, M.E. Booster vaccinations: Can immunologic memory outpace disease pathogenesis? Pediatrics 2009, 124, 1633–1641. [Google Scholar] [CrossRef]
- De Greeff, S.C.; de Melker, H.E.; Spanjaard, L.; Schouls, L.M.; van Derende, A. Protection from routine vaccination at the age of 14 months with meningococcal serogroup C conjugate vaccine in The Netherlands. Pediatric Infect. Dis. J. 2006, 25, 79–80. [Google Scholar] [CrossRef]
- De Voer, R.M.; Mollema, L.; Schepp, R.M.; de Greeff, S.C.; van Gageldonk, P.G.; de Melker, H.E.; Sanders, E.A.; Berbers, G.A.; van der Klis, F.R. Immunity against Neisseria meningitidis serogroup C in the Dutch population before and after introduction of the meningococcal C conjugate vaccine. PLoS ONE 2010, 5, e12144. [Google Scholar] [CrossRef]
- Stoof, S.P.; van der Klis, F.R.; van Rooijen, D.M.; Knol, M.J.; Sanders, E.A.; Berbers, G.A. Timing of an adolescent booster after single primary meningococcal serogroup C conjugate immunization at young age; an intervention study among Dutch teenagers. PLoS ONE 2014, 9, e100651. [Google Scholar] [CrossRef]
- Van Ravenhorst, M.B.; van der Klis, F.R.M.; van Rooijen, D.M.; Sanders, E.A.M.; Berbers, G.A.M. Adolescent meningococcal serogroup A, W and Y immune responses following immunization with quadrivalent meningococcal A, C, W and Y conjugate vaccine: Optimal age for vaccination. Vaccine 2017, 35, 4753–4760. [Google Scholar] [CrossRef]
- Christensen, H.; May, M.; Bowen, L.; Hickman, M.; Trotter, C.L. Meningococcal carriage by age: A systematic review and meta-analysis. Lancet Infect. Dis. 2010, 10, 853–861. [Google Scholar] [CrossRef]
- Trotter, C.L.; Maiden, M.C. Meningococcal vaccines and herd immunity: Lessons learned from serogroup C conjugate vaccination programs. Expert Rev. Vaccines 2009, 8, 851–861. [Google Scholar] [CrossRef]
- De Melker, H.E.; Schurink-van ‘t Klooster, T.M. The National Immunisation Programme in The Netherlands: Surveillance and Developments in 2018–2019; National Institute for Public Health and the Environment, Ministry of Health, Welfare and Sport: Bilthoven, The Netherlands, 2019. [Google Scholar]
- Van der Heiden, M.; Boots, A.M.H.; Bonacic Marinovic, A.A.; de Rond, L.G.H.; van Maurik, M.; Tcherniaeva, I.; Berbers, G.A.M.; Buisman, A.M. Novel Intervention in the Aging Population: A Primary Meningococcal Vaccine Inducing Protective IgM Responses in Middle-Aged Adults. Front. Immunol. 2017, 8. [Google Scholar] [CrossRef]
- Ostergaard, L.; Lebacq, E.; Poolman, J.; Maechler, G.; Boutriau, D. Immunogenicity, reactogenicity and persistence of meningococcal A, C, W-135 and Y-tetanus toxoid candidate conjugate (MenACWY-TT) vaccine formulations in adolescents aged 15–25 years. Vaccine 2009, 27, 161–168. [Google Scholar] [CrossRef]
- Al-Mazrou, Y.; Khalil, M.; Findlow, H.; Chadha, H.; Bosch Castells, V.; Johnson, D.R.; Borrow, R. Immunogenicity and safety of a meningococcal quadrivalent conjugate vaccine in Saudi Arabian adolescents previously vaccinated with one dose of bivalent and quadrivalent meningococcal polysaccharide vaccines: A phase III, controlled, randomized, and modified blind-observer study. Clin. Vaccine Immunol. CVI 2012, 19, 999–1004. [Google Scholar] [CrossRef]
- Baxter, R.; Baine, Y.; Ensor, K.; Bianco, V.; Friedland, L.R.; Miller, J.M. Immunogenicity and safety of an investigational quadrivalent meningococcal ACWY tetanus toxoid conjugate vaccine in healthy adolescents and young adults 10 to 25 years of age. Pediatric Infect. Dis. J. 2011, 30, e41–e48. [Google Scholar] [CrossRef]
- Bermal, N.; Huang, L.M.; Dubey, A.P.; Jain, H.; Bavdekar, A.; Lin, T.Y.; Bianco, V.; Baine, Y.; Miller, J.M. Safety and immunogenicity of a tetravalent meningococcal serogroups A, C, W-135 and Y conjugate vaccine in adolescents and adults. Hum. Vaccines 2011, 7, 239–247. [Google Scholar] [CrossRef]
- Borja-Tabora, C.; Montalban, C.; Memish, Z.A.; Van der Wielen, M.; Bianco, V.; Boutriau, D.; Miller, J. Immune response, antibody persistence, and safety of a single dose of the quadrivalent meningococcal serogroups A, C, W-135, and Y tetanus toxoid conjugate vaccine in adolescents and adults: Results of an open, randomised, controlled study. BMC Infect. Dis. 2013, 13, 116. [Google Scholar] [CrossRef]
- Dbaibo, G.; El-Ayoubi, N.; Ghanem, S.; Hajar, F.; Bianco, V.; Miller, J.M.; Mesaros, N. Immunogenicity and safety of a quadrivalent meningococcal serogroups A, C, W-135 and Y tetanus toxoid conjugate vaccine (MenACWY-TT) administered to adults aged 56 Years and older: Results of an open-label, randomized, controlled trial. Drugs Aging 2013, 30, 309–319. [Google Scholar] [CrossRef]
- van der Heiden, M.; van Ravenhorst, M.B.; Bogaard, M.; Boots, A.M.H.; Berbers, G.A.M.; Buisman, A.M. Lower antibody functionality in middle-aged adults compared to adolescents after primary meningococcal vaccination: Role of IgM. Exp. Gerontol. 2018, 105, 101–108. [Google Scholar] [CrossRef]
- Borja-Tabora, C.F.; Montalban, C.; Memish, Z.A.; Boutriau, D.; Kolhe, D.; Miller, J.M.; Van der Wielen, M. Long-term immunogenicity and safety after a single dose of the quadrivalent meningococcal serogroups A, C, W, and Y tetanus toxoid conjugate vaccine in adolescents and adults: 5-year follow-up of an open, randomized trial. BMC Infect. Dis. 2015, 15, 409. [Google Scholar] [CrossRef]
- Borja-Tabora, C.F.C.; Peyrani, P.; Webber, C.; Van der Wielen, M.; Cheuvart, B.; De Schrevel, N.; Bianco, V.; Aris, E.; Cutler, M.; Li, P.; et al. A phase 2b/3b MenACWY-TT study of long-term antibody persistence after primary vaccination and immunogenicity and safety of a booster dose in individuals aged 11 through 55 years. BMC Infect. Dis. 2020, 20, 426. [Google Scholar] [CrossRef]
- Baxter, R.; Keshavan, P.; Welsch, J.A.; Han, L.; Smolenov, I. Persistence of the immune response after MenACWY-CRM vaccination and response to a booster dose, in adolescents, children and infants. Hum. Vaccines Immunother. 2016. [Google Scholar] [CrossRef]
- Van Ravenhorst, M.B.; van der Klis, F.R.M.; van Rooijen, D.M.; Knol, M.J.; Stoof, S.P.; Sanders, E.A.M.; Berbers, G.A.M. Meningococcal serogroup C immunogenicity, antibody persistence and memory B-cells induced by the monovalent meningococcal serogroup C versus quadrivalent meningococcal serogroup ACWY conjugate booster vaccine: A randomized controlled trial. Vaccine 2017, 35, 4745–4752. [Google Scholar] [CrossRef]
- Maslanka, S.E.; Gheesling, L.L.; Libutti, D.E.; Donaldson, K.B.; Harakeh, H.S.; Dykes, J.K.; Arhin, F.F.; Devi, S.J.; Frasch, C.E.; Huang, J.C.; et al. Standardization and a multilaboratory comparison of Neisseria meningitidis serogroup A and C serum bactericidal assays. The Multilaboratory Study Group. Clin. Diagn. Lab. Immunol. 1997, 4, 156–167. [Google Scholar] [CrossRef]
- Borrow, R.; Andrews, N.; Goldblatt, D.; Miller, E. Serological basis for use of meningococcal serogroup C conjugate vaccines in the United Kingdom: Reevaluation of correlates of protection. Infect. Immun. 2001, 69, 1568–1573. [Google Scholar] [CrossRef]
- Borrow, R.; Balmer, P.; Miller, E. Meningococcal surrogates of protection--serum bactericidal antibody activity. Vaccine 2005, 23, 2222–2227. [Google Scholar] [CrossRef] [PubMed]
- Andrews, N.; Borrow, R.; Miller, E. Validation of serological correlate of protection for meningococcal C conjugate vaccine by using efficacy estimates from postlicensure surveillance in England. Clin. Diagn. Lab. Immunol. 2003, 10, 780–786. [Google Scholar] [CrossRef]
- De Voer, R.M.; Schepp, R.M.; Versteegh, F.G.; van der Klis, F.R.; Berbers, G.A. Simultaneous detection of Haemophilus influenzae type b polysaccharide-specific antibodies and Neisseria meningitidis serogroup A, C, Y, and W-135 polysaccharide-specific antibodies in a fluorescent-bead-based multiplex immunoassay. Clin. Vaccine Immunol. CVI 2009, 16, 433–436. [Google Scholar] [CrossRef]
- De Voer, R.M.; van der Klis, F.R.; Engels, C.W.; Rijkers, G.T.; Sanders, E.A.; Berbers, G.A. Development of a fluorescent-bead-based multiplex immunoassay to determine immunoglobulin G subclass responses to Neisseria meningitidis serogroup A and C polysaccharides. Clin. Vaccine Immunol. CVI 2008, 15, 1188–1193. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Lal, G.; Balmer, P.; Joseph, H.; Dawson, M.; Borrow, R. Development and evaluation of a tetraplex flow cytometric assay for quantitation of serum antibodies to Neisseria meningitidis serogroups A, C, Y, and W-135. Clin. Diagn. Lab. Immunol. 2004, 11, 272–279. [Google Scholar] [CrossRef] [PubMed]
- Ceyhan, M.; Yildirim, I.; Balmer, P.; Riley, C.; Laher, G.; Andrews, N.; Borrow, R.; Kurt, N.; Turgut, M.; Aydogan, A.; et al. Age-specific seroprevalence of serogroup C meningococcal serum bactericidal antibody activity and serogroup A, C, W135 and Y-specific IgG concentrations in the Turkish population during 2005. Vaccine 2007, 25, 7233–7237. [Google Scholar] [CrossRef]
- Peltola, H.; Mäkelä, H.; Käyhty, H.; Jousimies, H.; Herva, E.; Hällström, K.; Sivonen, A.; Renkonen, O.V.; Pettay, O.; Karanko, V.; et al. Clinical efficacy of meningococcus group A capsular polysaccharide vaccine in children three months to five years of age. N. Eng. J. Med. 1977, 297, 686–691. [Google Scholar] [CrossRef]
- King, W.J.; MacDonald, N.E.; Wells, G.; Huang, J.; Allen, U.; Chan, F.; Ferris, W.; Diaz-Mitoma, F.; Ashton, F. Total and functional antibody response to a quadrivalent meningococcal polysaccharide vaccine among children. J. Pediatrics 1996, 128, 196–202. [Google Scholar] [CrossRef]
- Elias, J.; Findlow, J.; Borrow, R.; Tremmel, A.; Frosch, M.; Vogel, U. Persistence of antibodies in laboratory staff immunized with quadrivalent meningococcal polysaccharide vaccine. J. Occup. Med. Toxicol. 2013, 8, 4. [Google Scholar] [CrossRef]
- Findlow, H.; Tapia, M.D.; Sow, S.O.; Haidara, F.C.; Coulibaly, F.; Keita, A.M.; Diallo, F.; Doumbia, M.; Traore, A.; Schluterman, N.; et al. Kinetics of maternally-derived serogroup A, C, Y and W-specific meningococcal immunoglobulin G in Malian women and infants. Vaccine 2019, 37, 2477–2481. [Google Scholar] [CrossRef]
- van Ravenhorst, M.B.; Marinovic, A.B.; van der Klis, F.R.; van Rooijen, D.M.; van Maurik, M.; Stoof, S.P.; Sanders, E.A.; Berbers, G.A. Long-term persistence of protective antibodies in Dutch adolescents following a meningococcal serogroup C tetanus booster vaccination. Vaccine 2016. [Google Scholar] [CrossRef] [PubMed]
- De Graaf, W.F.; Kretzschmar, M.E.; Teunis, P.F.; Diekmann, O. A two-phase within-host model for immune response and its application to serological profiles of pertussis. Epidemics 2014, 9, 1–7. [Google Scholar] [CrossRef]
- Berbers, G.A.; van de Wetering, M.S.; van Gageldonk, P.G.; Schellekens, J.F.; Versteegh, F.G.; Teunis, P.F. A novel method for evaluating natural and vaccine induced serological responses to Bordetella pertussis antigens. Vaccine 2013, 31, 3732–3738. [Google Scholar] [CrossRef] [PubMed]
- Teunis, P.F.M.; van Eijkeren, J.C.H.; de Graaf, W.F.; Bonačić Marinović, A.A.; Kretzschmar, M.E.E. Linking the seroresponse to infection to within-host heterogeneity in antibody production. Epidemics 2016, 16, 33–39. [Google Scholar] [CrossRef]
- Plummer, M. JAGS: A program for analysis of Bayesian graphial models using Gibbs sampling. In Proceedings of the 3rd International Workshop on Distributed Statistical Computing (DSC 2003), Vienna, Austria, 20–22 March 2003; Technische Universität Wien: Vienna, Austria, 2003; pp. 1–10. [Google Scholar]
- Newcombe, R.G. Improved confidence intervals for the difference between binomial proportions based on paired data. Stat. Med. 1998, 17, 2635–2650. [Google Scholar] [CrossRef]
- McVernon, J.; Nolan, T.; Richmond, P.; Reynolds, G.; Nissen, M.; Lambert, S.B.; Marshall, H.; Papa, T.; Rehm, C. A randomized trial to assess safety and immunogenicity of alternative formulations of a quadrivalent meningococcal (A, C, Y, and W-135) tetanus protein conjugate vaccine in toddlers. Pediatric Infect. Dis. J. 2012, 31, e15–e23. [Google Scholar] [CrossRef] [PubMed]
- Findlow, H.; Borrow, R. Immunogenicity and safety of a meningococcal serogroup A, C, Y and W glycoconjugate vaccine, ACWY-TT. Adv. Ther. 2013, 30, 431–458. [Google Scholar] [CrossRef]
- Østergaard, L.; Van der Wielen, M.; Bianco, V.; Miller, J.M. Persistence of antibodies for 42 months following vaccination of adolescents with a meningococcal serogroups A, C, W-135, and Y tetanus toxoid conjugate vaccine (MenACWY-TT). Int. J. Infect. Dis. 2013, 17, e173–e176. [Google Scholar] [CrossRef]
- Baxter, R.; Baine, Y.; Kolhe, D.; Baccarini, C.I.; Miller, J.M.; Van der Wielen, M. Five-year Antibody Persistence and Booster Response to a Single Dose of Meningococcal A, C, W and Y Tetanus Toxoid Conjugate Vaccine in Adolescents and Young Adults: An Open, Randomized Trial. Pediatric Infect. Dis. J. 2015, 34, 1236–1243. [Google Scholar] [CrossRef]
- Klein, N.P.; Baine, Y.; Kolhe, D.; Baccarini, C.I.; Miller, J.M.; Van der Wielen, M. Five-year Antibody Persistence and Booster Response After 1 or 2 Doses of Meningococcal A, C, W and Y Tetanus Toxoid Conjugate Vaccine in Healthy Children. Pediatric Infect. Dis. J. 2016, 35, 662–672. [Google Scholar] [CrossRef]
- Ilyina, N.; Kharit, S.; Namazova-Baranova, L.; Asatryan, A.; Benashvili, M.; Tkhostova, E.; Bhusal, C.; Arora, A.K. Safety and immunogenicity of meningococcal ACWY CRM197-conjugate vaccine in children, adolescents and adults in Russia. Hum. Vaccines Immunother. 2014, 10, 2471–2481. [Google Scholar] [CrossRef]
- Lalwani, S.; Agarkhedkar, S.; Gogtay, N.; Palkar, S.; Agarkhedkar, S.; Thatte, U.; Vakil, H.; Jonnalagedda, R.; Pedotti, P.; Hoyle, M.; et al. Safety and immunogenicity of an investigational meningococcal ACWY conjugate vaccine (MenACWY-CRM) in healthy Indian subjects aged 2 to 75 years. Int. J. Infect. Dis. 2015, 38, 36–42. [Google Scholar] [CrossRef][Green Version]
- Reisinger, K.S.; Baxter, R.; Block, S.L.; Shah, J.; Bedell, L.; Dull, P.M. Quadrivalent meningococcal vaccination of adults: Phase III comparison of an investigational conjugate vaccine, MenACWY-CRM, with the licensed vaccine, Menactra. Clin. Vaccine Immunol. CVI 2009, 16, 1810–1815. [Google Scholar] [CrossRef] [PubMed]
- Ferlito, C.; Biselli, R.; Cattaruzza, M.S.; Teloni, R.; Mariotti, S.; Tomao, E.; Salerno, G.; Peragallo, M.S.; Lulli, P.; Caporuscio, S.; et al. Immunogenicity of meningococcal polysaccharide ACWY vaccine in primary immunized or revaccinated adults. Clin. Exp. Immunol. 2018, 194, 361–370. [Google Scholar] [CrossRef] [PubMed]
- Siegrist, C.A.; Aspinall, R. B-cell responses to vaccination at the extremes of age. Nat. Rev. Immunol. 2009, 9, 185–194. [Google Scholar] [CrossRef]
- Esteves-Jaramillo, A.; Koehler, T.; Jeanfreau, R.; Neveu, D.; Jordanov, E.; Singh Dhingra, M. Immunogenicity and safety of a quadrivalent meningococcal tetanus toxoid-conjugate vaccine (MenACYW-TT) in ≥56-year-olds: A Phase III randomized study. Vaccine 2020, 38, 4405–4411. [Google Scholar] [CrossRef]
- Weinberger, B.; Herndler-Brandstetter, D.; Schwanninger, A.; Weiskopf, D.; Grubeck-Loebenstein, B. Biology of immune responses to vaccines in elderly persons. Clin. Infect. Dis. Off. Publ. Infect. Dis. Soc. Am. 2008, 46, 1078–1084. [Google Scholar] [CrossRef]
- Stoof, S.P.; Rodenburg, G.D.; Knol, M.J.; Rumke, L.W.; Bovenkerk, S.; Berbers, G.A.; Spanjaard, L.; van der Ende, A.; Sanders, E.A. Disease Burden of Invasive Meningococcal Disease in the Netherlands Between June 1999 and June 2011: A Subjective Role for Serogroup and Clonal Complex. Clin. Infect. Dis. Off. Publ. Infect. Dis. Soc. Am. 2015, 61, 1281–1292. [Google Scholar] [CrossRef] [PubMed]
- Bijlsma, M.W.; Brouwer, M.C.; Spanjaard, L.; van de Beek, D.; van der Ende, A. A decade of herd protection after introduction of meningococcal serogroup C conjugate vaccination. Clin. Infect. Dis. Off. Publ. Infect. Dis. Soc. Am. 2014, 59, 1216–1221. [Google Scholar] [CrossRef]
- Nimenrix—Assessment Report—Procedure, No. EMEA/H/C/002226. Available online: https://www.ema.europa.eu/en/documents/assessment-report/nimenrix-epar-public-assessment-report_en.pdf (accessed on 23 September 2020).
- Bröker, M.; Berti, F.; Costantino, P. Factors contributing to the immunogenicity of meningococcal conjugate vaccines. Hum. Vaccines Immunother. 2016, 12, 1808–1824. [Google Scholar] [CrossRef]
- Bijlsma, M.W.; Bekker, V.; Brouwer, M.C.; Spanjaard, L.; van de Beek, D.; van der Ende, A. Epidemiology of invasive meningococcal disease in the Netherlands, 1960–2012: An analysis of national surveillance data. Lancet. Infect. Dis. 2014, 14, 805–812. [Google Scholar] [CrossRef]
- Knol, M.J.; de Melker, H.E.; Berbers, G.A.M.; van Ravenhorst, M.B.; Ruijs, W.L.M.; van Vliet, J.A.; Kemmeren, J.M.; Suijkerbuijk, A.; van Lier, E.A.; Sanders, E.A.M.; et al. Meningococcal Disease in The Netherlands. Background Information for the Health Council; National Institute for Public Health and the Environment: Bilthoven, The Netherlands, 2017. [Google Scholar]

| Antibody | Age Group | p-Value | ||
|---|---|---|---|---|
| Adolescents (n = 50) | Middle-Aged Adults (n = 130) | |||
| MenA | GMC MenA-PS-specific IgG µg/mL (95% CI) | 1.8 (1.2–2.5) | 2.8 (2.0–3.9) | 0.1532 |
| MenC | GMT (95% CI) | 588 (341–1014) | 6.5 (4.6–9.0) | <0.0001 |
| % rSBA-titer ≥8 (95% CI) | 94% (82–98) | 32% (24–40) | <0.0001 | |
| % rSBA-titer ≥128 (95% CI) | 88% (75–95) | 13% (8–20) | <0.0001 | |
| GMC MenC-PS-specific IgG µg/mL (95% CI) | 10.1 (7.7–13.2) | 1.3 (0.9–1.7) | <0.0001 | |
| MenW | GMT (95% CI) | 578 (372–898) | 52.5 (35.4–78.3) | <0.0001 |
| % rSBA-titer ≥8 (95% CI) | 96% (85–99) | 65% (56–73) | <0.0001 | |
| % rSBA-titer ≥128 (95% CI) | 96% (85–99) | 56% (47–65) | <0.0001 | |
| GMC MenW-PS-specific IgG µg/mL (95% CI) | 1.3 (0.8–2.1) | 0.5 (0.4–0.6) | 0.0008 | |
| MenY | GMT (95% CI) | 270 (170–430) | 70.5 (47.2–105) | 0.0002 |
| % rSBA-titer ≥8 (95% CI) | 94% (82–98) | 71% (62–79) | 0.0006 | |
| % rSBA-titer ≥128 (95% CI) | 86% (73–94) | 64% (55–72) | 0.0036 | |
| GMC MenY-PS-specific IgG µg/mL (95% CI) | 2.2 (1.3–3.6) | 0.9 (0.7–1.2) | 0.0045 | |
| Antibody | Adolescents | Middle-Aged Adults | |
|---|---|---|---|
| MenC | Fold-change 1 month vs. 1 year | 14.3 | 10.2 |
| Fold-change 1 year vs. 5 years | 4.7 | 26.6 | |
| Mean annual decay rate 1–5 years | 1.18 | 6.65 | |
| Minimal duration of protection | 4.6 years | 0.2 years | |
| Median duration of protection | 32.4 years | 3.7 years | |
| MenW | Fold-change 1 month vs. 1 year | 5.1 | 5.3 |
| Fold-change 1 year vs. 5 years | 2.3 | 6.6 | |
| Mean annual decay rate 1–5 years | 0.58 | 1.65 | |
| Minimal duration of protection | 7.0 years | 1.7 years | |
| Median duration of protection | 97.7 years | 13.9 years | |
| MenY | Fold-change 1 month vs. 1 year | 3.2 | 6.1 |
| Fold-change 1 year vs. 5 years | 5.4 | 4.2 | |
| Mean annual decay rate 1–5 years | 1.35 | 1.05 | |
| Minimal duration of protection | 3.7 years | 1.4 years | |
| Median duration of protection | 33.4 years | 20.8 years |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ohm, M.; van Rooijen, D.M.; Bonačić Marinović, A.A.; van Ravenhorst, M.B.; van der Heiden, M.; Buisman, A.-M.; Sanders, E.A.M.; Berbers, G.A.M. Different Long-Term Duration of Seroprotection against Neisseria meningitidis in Adolescents and Middle-Aged Adults after a Single Meningococcal ACWY Conjugate Vaccination in The Netherlands. Vaccines 2020, 8, 624. https://doi.org/10.3390/vaccines8040624
Ohm M, van Rooijen DM, Bonačić Marinović AA, van Ravenhorst MB, van der Heiden M, Buisman A-M, Sanders EAM, Berbers GAM. Different Long-Term Duration of Seroprotection against Neisseria meningitidis in Adolescents and Middle-Aged Adults after a Single Meningococcal ACWY Conjugate Vaccination in The Netherlands. Vaccines. 2020; 8(4):624. https://doi.org/10.3390/vaccines8040624
Chicago/Turabian StyleOhm, Milou, Debbie M. van Rooijen, Axel A. Bonačić Marinović, Mariëtte B. van Ravenhorst, Marieke van der Heiden, Anne-Marie Buisman, Elisabeth A.M. Sanders, and Guy A.M. Berbers. 2020. "Different Long-Term Duration of Seroprotection against Neisseria meningitidis in Adolescents and Middle-Aged Adults after a Single Meningococcal ACWY Conjugate Vaccination in The Netherlands" Vaccines 8, no. 4: 624. https://doi.org/10.3390/vaccines8040624
APA StyleOhm, M., van Rooijen, D. M., Bonačić Marinović, A. A., van Ravenhorst, M. B., van der Heiden, M., Buisman, A.-M., Sanders, E. A. M., & Berbers, G. A. M. (2020). Different Long-Term Duration of Seroprotection against Neisseria meningitidis in Adolescents and Middle-Aged Adults after a Single Meningococcal ACWY Conjugate Vaccination in The Netherlands. Vaccines, 8(4), 624. https://doi.org/10.3390/vaccines8040624





