Next-Generation Pertussis Vaccines Based on the Induction of Protective T Cells in the Respiratory Tract
Abstract
1. Introduction
2. Immune Responses That Control a Primary Infection with B. pertussis
3. Natural and Vaccine-Induced Immunity-Correlates of Protection
3.1. Antibody Responses
3.1.1. Serum IgG
3.1.2. Secretory IgA
3.2. CD4 Th Cell Subtypes
3.2.1. Th1 Cells Protect in the Lung
3.2.2. Th17 Cells Protect Against Nasal Colonization
3.3. TRM Cells Maintain Sustained Protective Immunity in the Respiratory Tract
4. Experimental Vaccines
4.1. Live Attenuated Pertussis Vaccine-BPZE1
4.2. OMV Vaccines
4.3. aP Vaccines with New Antigens
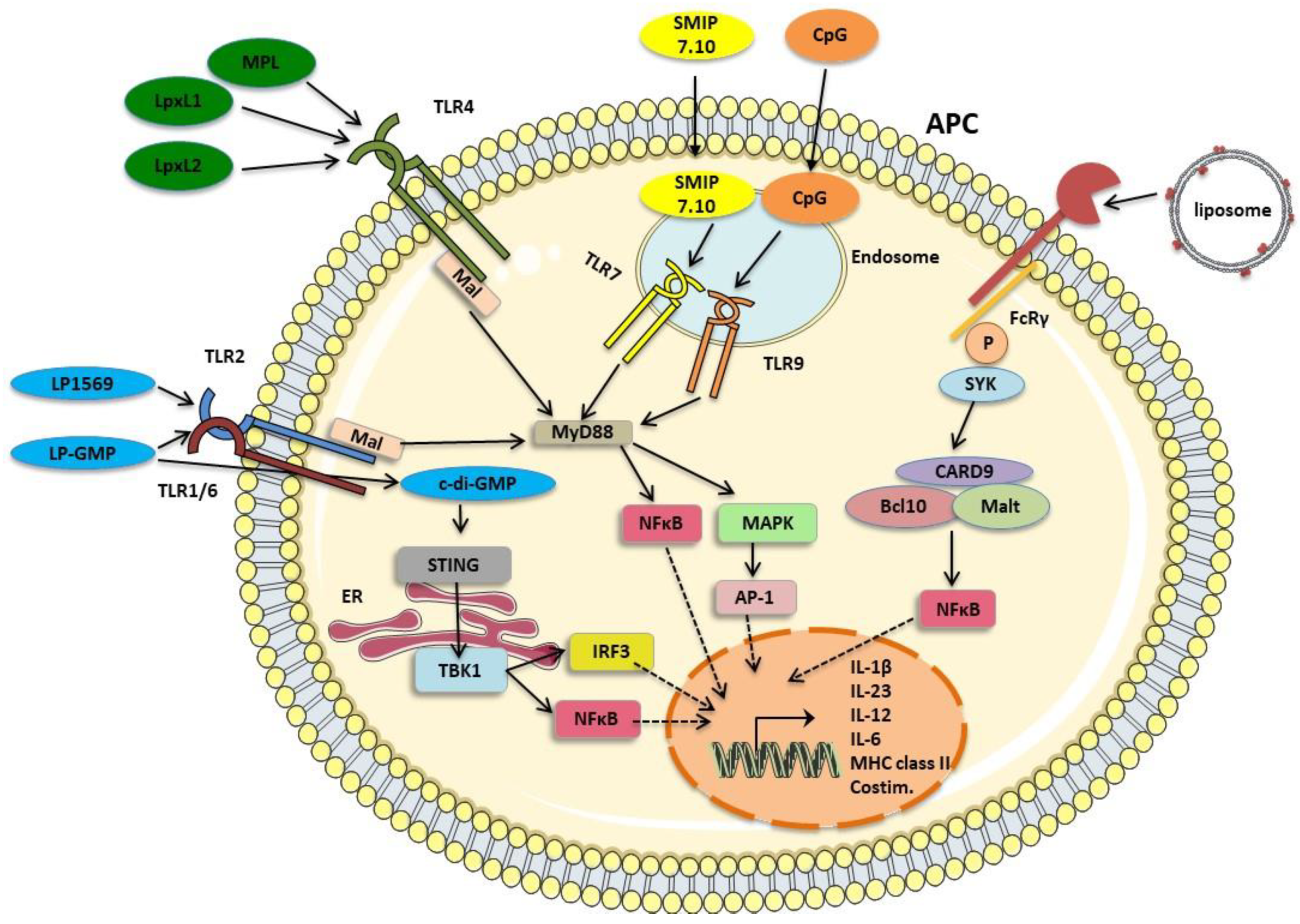
4.4. aP Vaccines with New Adjuvants
4.4.1. Alum
4.4.2. TLR4 Agonists
4.4.3. TLR 9 Agonists
4.4.4. TLR7 Agonists
4.4.5. TLR2 Agonists
4.4.6. STING and TLR Agonists
4.4.7. Particulate Antigen Delivery Systems, Live Vectors and Nucleic Acid Vaccines
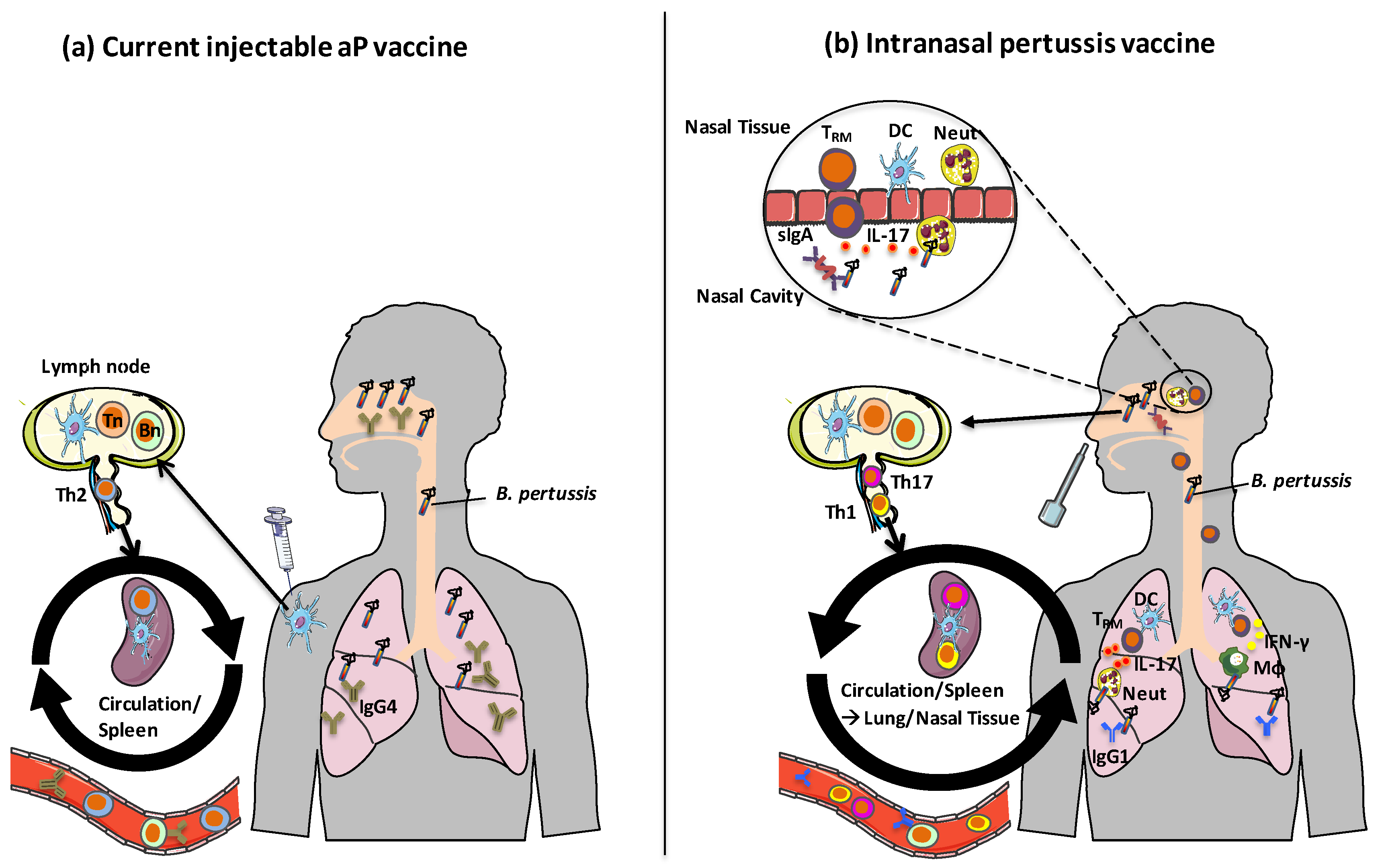
5. Route of Vaccine Administration—A Case for an Intranasal Pertussis Vaccine
6. Concluding Remarks
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Mattoo, S.; Cherry, J.D. Molecular pathogenesis, epidemiology, and clinical manifestations of respiratory infections due to Bordetella pertussis and other Bordetella subspecies. Clin. Microbiol. Rev. 2005, 18, 326–382. [Google Scholar] [CrossRef] [PubMed]
- Hewlett, E.L.; Burns, D.L.; Cotter, P.A.; Harvill, E.T.; Merkel, T.J.; Quinn, C.P.; Stibitz, E.S. Pertussis Pathogenesis—What We Know and What We Don’t Know. J. Infect. Dis. 2014, 209, 982–985. [Google Scholar] [CrossRef] [PubMed]
- Janda, W.M.; Santos, E.; Stevens, J.; Celig, D.; Terrile, L.; Schreckenberger, P.C. Unexpected isolation of Bordetella pertussis from a blood culture. J. Clin. Microbiol. 1994, 32, 2851–2853. [Google Scholar] [CrossRef] [PubMed]
- Cody, C.L.; Baraff, L.J.; Cherry, J.D.; Marcy, S.M.; Manclark, C.R. Nature and Rates of Adverse Reactions Associated with DTP and DT Immunizations in Infants and Children. Pediatrics 1981, 68, 650–660. [Google Scholar]
- Decker, M.D.; Edwards, K.M.; Steinhoff, M.C.; Rennels, M.B.; Pichichero, M.E.; Englund, J.A.; Anderson, E.L.; Deloria, M.A.; Reed, G.F. Comparison of 13 acellular pertussis vaccines: Adverse reactions. Pediatrics 1995, 96, 557–566. [Google Scholar]
- Edwards, K.M. Unraveling the challenges of pertussis. Proc. Natl. Acad. Sci. USA 2014, 111, 575–576. [Google Scholar] [CrossRef]
- Gorringe, A.R.; Vaughan, T.E. Bordetella pertussis fimbriae (Fim): Relevance for vaccines. Expert Rev. Vaccines 2014, 13, 1205–1214. [Google Scholar] [CrossRef]
- Edwards, K.M.; Meade, B.D.; Decker, M.D.; Reed, G.F.; Rennels, M.B.; Steinhoff, M.C.; Anderson, E.L.; Englund, J.A.; Pichichero, M.E.; Deloria, M.A. Comparison of 13 acellular pertussis vaccines: Overview and serologic response. Pediatrics 1995, 96, 548–557. [Google Scholar]
- World Health Organization. Pertussis vaccines: WHO position paper, August 2015—Recommendations. Vaccine 2016, 34, 1423–1425. [Google Scholar] [CrossRef]
- Broder, K.R.; Cortese, M.M.; Iskander, J.K.; Kretsinger, K.; Slade, B.A.; Brown, K.H.; Mijalski, C.M.; Tiwari, T.; Weston, E.J.; Cohn, A.C.; et al. Preventing tetanus, diphtheria, and pertussis among adolescents: Use of tetanus toxoid, reduced diphtheria toxoid and acellular pertussis vaccines recommendations of the Advisory Committee on Immunization Practices (ACIP). MMWR Recomm. Rep. 2006, 55, 1–34. [Google Scholar]
- Lambert, L.C. Pertussis Vaccine Trials in the 1990s. J. Infect. Dis. 2014, 209, S4–S9. [Google Scholar] [CrossRef] [PubMed]
- Olin, P.; Rasmussen, F.; Gustafsson, L.; Hallander, H.O.; Heijbel, H. Randomised controlled trial of two-component, three-component, and five-component acellular pertussis vaccines compared with whole-cell pertussis vaccine. Lancet 1997, 350, 1569–1577. [Google Scholar] [CrossRef]
- Gustafsson, L.; Hallander, H.O.; Olin, P.; Reizenstein, E.; Storsaeter, J. A Controlled Trial of a Two-Component Acellular, a Five-Component Acellular, and a Whole-Cell Pertussis Vaccine. N. Engl. J. Med. 1996, 334, 349–356. [Google Scholar] [CrossRef] [PubMed]
- Warfel, J.M.; Zimmerman, L.I.; Merkel, T.J. Acellular pertussis vaccines protect against disease but fail to prevent infection and transmission in a nonhuman primate model. Proc. Natl. Acad. Sci. USA 2014, 111, 787–792. [Google Scholar] [CrossRef]
- Wilk, M.M.; Borkner, L.; Misiak, A.; Curham, L.; Allen, A.C.; Mills, K.H.G. Immunization with whole cell but not acellular pertussis vaccines primes CD4 T RM cells that sustain protective immunity against nasal colonization with Bordetella pertussis. Emerg. Microbes Infect. 2019, 8, 169–185. [Google Scholar] [CrossRef]
- Ross, P.J.; Sutton, C.E.; Higgins, S.; Allen, A.C.; Walsh, K.; Misiak, A.; Lavelle, E.C.; McLoughlin, R.M.; Mills, K.H.G. Relative Contribution of Th1 and Th17 Cells in Adaptive Immunity to Bordetella pertussis: Towards the Rational Design of an Improved Acellular Pertussis Vaccine. PLoS Pathog. 2013, 9, e1003264. [Google Scholar] [CrossRef]
- Kapil, P.; Merkel, T.J. Pertussis vaccines and protective immunity. Curr. Opin. Immunol. 2019, 59, 72–78. [Google Scholar] [CrossRef]
- Arsenault, C.; Johri, M.; Nandi, A.; Mendoza Rodríguez, J.M.; Hansen, P.M.; Harper, S. Country-level predictors of vaccination coverage and inequalities in Gavi-supported countries. Vaccine 2017, 35, 2479–2488. [Google Scholar] [CrossRef]
- Sobanjo-ter Meulen, A.; Duclos, P.; McIntyre, P.; Lewis, K.D.C.; Van Damme, P.; O’Brien, K.L.; Klugman, K.P. Assessing the Evidence for Maternal Pertussis Immunization: A Report From the Bill & Melinda Gates Foundation Symposium on Pertussis Infant Disease Burden in Low- and Lower-Middle-Income Countries. Clin. Infect. Dis. 2016, 63, S123–S133. [Google Scholar]
- Diavatopoulos, D.A.; Mills, K.H.G.; Kester, K.E.; Kampmann, B.; Silerova, M.; Heininger, U.; van Dongen, J.J.M.; van der Most, R.G.; Huijnen, M.A.; Siena, E.; et al. PERISCOPE: Road towards effective control of pertussis. Lancet Infect. Dis. 2019, 19, e179–e186. [Google Scholar] [CrossRef]
- Klein, N.P.; Bartlett, J.; Fireman, B.; Rowhani-Rahbar, A.; Baxter, R. Comparative Effectiveness of Acellular Versus Whole-Cell Pertussis Vaccines in Teenagers. Pediatrics 2013, 131, e1716–e1722. [Google Scholar] [CrossRef]
- Sheridan, S.L.; Ware, R.S.; Grimwood, K.; Lambert, S.B. Number and order of whole cell pertussis vaccines in infancy and disease protection. JAMA 2012, 308, 454–456. [Google Scholar] [CrossRef] [PubMed]
- Klein, N.P.; Bartlett, J.; Rowhani-Rahbar, A.; Fireman, B.; Baxter, R. Waning Protection after Fifth Dose of Acellular Pertussis Vaccine in Children. N. Engl. J. Med. 2012, 367, 1012–1019. [Google Scholar] [CrossRef] [PubMed]
- Winter, K.; Harriman, K.; Zipprich, J.; Schechter, R.; Talarico, J.; Watt, J.; Chavez, G. California Pertussis Epidemic, 2010. J. Pediatr. 2012, 161, 1091–1096. [Google Scholar] [CrossRef]
- Klein, N.P.; Bartlett, J.; Fireman, B.; Baxter, R. Waning Tdap Effectiveness in Adolescents. Pediatrics 2016, 137, e20153326. [Google Scholar] [CrossRef]
- Martin, S.W.; Pawloski, L.; Williams, M.; Weening, K.; DeBolt, C.; Qin, X.; Reynolds, L.; Kenyon, C.; Giambrone, G.; Kudish, K.; et al. Pertactin-Negative Bordetella pertussis Strains: Evidence for a Possible Selective Advantage. Clin. Infect. Dis. 2014, 60, 223–227. [Google Scholar] [CrossRef] [PubMed]
- Octavia, S.; Maharjan, R.P.; Sintchenko, V.; Stevenson, G.; Reeves, P.R.; Gilbert, G.L.; Lan, R. Insight into Evolution of Bordetella pertussis from Comparative Genomic Analysis: Evidence of Vaccine-Driven Selection. Mol. Biol. Evol. 2010, 28, 707–715. [Google Scholar] [CrossRef]
- Safarchi, A.; Octavia, S.; Luu, L.D.W.; Tay, C.Y.; Sintchenko, V.; Wood, N.; Marshall, H.; McIntyre, P.; Lan, R. Pertactin negative Bordetella pertussis demonstrates higher fitness under vaccine selection pressure in a mixed infection model. Vaccine 2015, 33, 6277–6281. [Google Scholar] [CrossRef]
- Ryan, M.; Murphy, G.; Gothefors, L.; Nilsson, L.; Storsaeter, J.; Mills, K.H.G. Bordetella pertussis Respiratory Infection in Children Is Associated with Preferential Activation of Type 1 T Helper Cells. J. Infect. Dis. 1997, 175, 1246–1250. [Google Scholar] [CrossRef]
- Mahon, B.P.; Sheahan, B.J.; Griffin, F.; Murphy, G.; Mills, K.H. Atypical disease after Bordetella pertussis respiratory infection of mice with targeted disruptions of interferon-gamma receptor or immunoglobulin mu chain genes. J. Exp. Med. 1997, 186, 1843–1851. [Google Scholar] [CrossRef]
- Warfel, J.M.; Zimmerman, L.I.; Merkel, T.J. Comparison of Three Whole-Cell Pertussis Vaccines in the Baboon Model of Pertussis. Clin. Vaccine Immunol. 2015, 23, 47–54. [Google Scholar] [CrossRef] [PubMed]
- Wilk, M.M.; Misiak, A.; McManus, R.M.; Allen, A.C.; Lynch, M.A.; Mills, K.H.G. Lung CD4 Tissue-Resident Memory T Cells Mediate Adaptive Immunity Induced by Previous Infection of Mice with Bordetella pertussis. J. Immunol. 2017, 199, 233–243. [Google Scholar] [CrossRef] [PubMed]
- Allen, A.C.; Wilk, M.M.; Misiak, A.; Borkner, L.; Murphy, D.; Mills, K.H.G. Sustained protective immunity against Bordetella pertussis nasal colonization by intranasal immunization with a vaccine-adjuvant combination that induces IL-17-secreting TRM cells. Mucosal Immunol. 2018, 11, 1763–1776. [Google Scholar] [CrossRef] [PubMed]
- Solans, L.; Debrie, A.-S.; Borkner, L.; Aguiló, N.; Thiriard, A.; Coutte, L.; Uranga, S.; Trottein, F.; Martín, C.; Mills, K.H.G.; et al. IL-17-dependent SIgA-mediated protection against nasal Bordetella pertussis infection by live attenuated BPZE1 vaccine. Mucosal Immunol. 2018, 11, 1753–1762. [Google Scholar] [CrossRef]
- Althouse, B.M.; Scarpino, S.V. Asymptomatic transmission and the resurgence of Bordetella pertussis. BMC Med. 2015, 13, 146. [Google Scholar] [CrossRef]
- Choi, Y.H.; Campbell, H.; Amirthalingam, G.; van Hoek, A.J.; Miller, E. Investigating the pertussis resurgence in England and Wales, and options for future control. BMC Med. 2016, 14, 121. [Google Scholar] [CrossRef]
- Burdin, N.; Handy, L.K.; Plotkin, S.A. What is wrong with pertussis vaccine immunity? The problem of waning effectiveness of pertussis vaccines. Cold Spring Harb. Perspect. Biol. 2017, 9, a029454. [Google Scholar] [CrossRef]
- Brummelman, J.; Wilk, M.M.; Han, W.G.H.; van Els, C.A.C.M.; Mills, K.H.G. Roads to the development of improved pertussis vaccines paved by immunology. Pathog. Dis. 2015, 73, ftv067. [Google Scholar] [CrossRef]
- Coutte, L.; Alonso, S.; Reveneau, N.; Willery, E.; Quatannens, B.; Locht, C.; Jacob-Dubuisson, F. Role of adhesin release for mucosal colonization by a bacterial pathogen. J. Exp. Med. 2003, 197, 735–742. [Google Scholar] [CrossRef]
- Ishibashi, Y.; Nishikawa, A. Bordetella pertussis infection of human respiratory epithelial cells up-regulates intercellular adhesion molecule-1 expression: Role of filamentous hemagglutinin and pertussis toxin. Microb. Pathog. 2002, 33, 115–125. [Google Scholar] [CrossRef]
- Jahnsen, F.L.; Strickland, D.H.; Thomas, J.A.; Tobagus, I.T.; Napoli, S.; Zosky, G.R.; Turner, D.J.; Sly, P.D.; Stumbles, P.A.; Holt, P.G. Accelerated Antigen Sampling and Transport by Airway Mucosal Dendritic Cells following Inhalation of a Bacterial Stimulus. J. Immunol. 2006, 177, 5861–5867. [Google Scholar] [CrossRef] [PubMed]
- Carbonetti, N.H.; Artamonova, G.V.; Van Rooijen, N.; Ayala, V.I. Pertussis toxin targets airway macrophages to promote Bordetella pertussis infection of the respiratory tract. Infect. Immun. 2007, 75, 1713–1720. [Google Scholar] [CrossRef] [PubMed]
- Bernard, N.J.; Finlay, C.M.; Tannahill, G.M.; Cassidy, J.P.; O’Neill, L.A.; Mills, K.H. A critical role for the TLR signaling adapter Mal in alveolar macrophage-mediated protection against Bordetella pertussis. Mucosal Immunol. 2015, 8, 982–992. [Google Scholar] [CrossRef] [PubMed]
- Raeven, R.H.M.; van der Maas, L.; Tilstra, W.; Uittenbogaard, J.P.; Bindels, T.H.E.; Kuipers, B.; van der Ark, A.; Pennings, J.L.A.; van Riet, E.; Jiskoot, W.; et al. Immunoproteomic Profiling of Bordetella pertussis Outer Membrane Vesicle Vaccine Reveals Broad and Balanced Humoral Immunogenicity. J. Proteome Res. 2015, 14, 2929–2942. [Google Scholar] [CrossRef] [PubMed]
- Lambrecht, B.N.; Prins, J.B.; Hoogsteden, H.C. Lung dendritic cells and host immunity to infection. Eur. Respir. J. 2001, 18, 692–704. [Google Scholar]
- Higgins, S.C.; Lavelle, E.C.; McCann, C.; Keogh, B.; McNeela, E.; Byrne, P.; O’Gorman, B.; Jarnicki, A.; McGuirk, P.; Mills, K.H.G. Toll-Like Receptor 4-Mediated Innate IL-10 Activates Antigen-Specific Regulatory T Cells and Confers Resistance to Bordetella pertussis by Inhibiting Inflammatory Pathology. J. Immunol. 2003, 171, 3119–3127. [Google Scholar] [CrossRef]
- Dunne, P.J.; Moran, B.; Cummins, R.C.; Mills, K.H.G. CD11c + CD8α + Dendritic Cells Promote Protective Immunity to Respiratory Infection with Bordetella pertussis. J. Immunol. 2009, 183, 400–410. [Google Scholar] [CrossRef]
- Misiak, A.; Wilk, M.M.; Raverdeau, M.; Mills, K.H.G. IL-17–Producing Innate and Pathogen-Specific Tissue Resident Memory γδ T Cells Expand in the Lungs of Bordetella pertussis–Infected Mice. J. Immunol. 2016, 198, 363–374. [Google Scholar] [CrossRef]
- Andreasen, C.; Carbonetti, N.H. Role of neutrophils in response to Bordetella pertussis infection in mice. Infect. Immun. 2009, 77, 1182–1188. [Google Scholar] [CrossRef]
- Boehm, D.T.; Varney, M.E.; Wong, T.Y.; Nowak, E.S.; Sen-Kilic, E.; Hall, J.; Bradford, S.D.; DeRoos, K.; Bevere, J.; Epperly, M.; et al. Characterizing the innate and adaptive responses of immunized mice to Bordetella pertussis infection using in vivo imaging and transcriptomic analysis. bioRxiv 2019. [Google Scholar] [CrossRef]
- Byrne, P.; McGuirk, P.; Todryk, S.; Mills, K.H.G. Depletion of NK cells results in disseminating lethal infection with Bordetella pertussis associated with a reduction of antigen-specific Th1 and enhancement of Th2, but not Tr1 cells. Eur. J. Immunol. 2004, 34, 2579–2588. [Google Scholar] [CrossRef] [PubMed]
- Barbic, J.; Leef, M.F.; Burns, D.L.; Shahin, R.D. Role of gamma interferon in natural clearance of Bordetella pertussis infection. Infect. Immun. 1997, 65, 4904–4908. [Google Scholar] [CrossRef] [PubMed]
- MacMicking, J.D. Interferon-inducible effector mechanisms in cell-autonomous immunity. Nat. Rev. Immunol. 2012, 12, 367–382. [Google Scholar] [CrossRef] [PubMed]
- Higgs, R.; Higgins, S.C.; Ross, P.J.; Mills, K.H.G. Immunity to the respiratory pathogen Bordetella pertussis. Mucosal Immunol. 2012, 5, 485–500. [Google Scholar] [CrossRef]
- Mills, K.H.; Barnard, A.; Watkins, J.; Redhead, K. Cell-mediated immunity to Bordetella pertussis: Role of Th1 cells in bacterial clearance in a murine respiratory infection model. Infect. Immun. 1993, 61, 399–410. [Google Scholar] [CrossRef]
- Warfel, J.M.; Merkel, T.J. Bordetella pertussis infection induces a mucosal IL-17 response and long-lived Th17 and Th1 immune memory cells in nonhuman primates. Mucosal Immunol. 2013, 6, 787–796. [Google Scholar] [CrossRef]
- Vinuesa, C.G.; Linterman, M.A.; Yu, D.; MacLennan, I.C.M. Follicular Helper T Cells. Annu. Rev. Immunol. 2016, 34, 335–368. [Google Scholar] [CrossRef]
- Rowe, J.; Macaubas, C.; Monger, T.M.; Holt, B.J.; Harvey, J.; Poolman, J.T.; Sly, P.D.; Holt, P.G. Antigen-Specific Responses to Diphtheria-Tetanus-Acellular Pertussis Vaccine in Human Infants Are Initially Th2 Polarized. Infect. Immun. 2000, 68, 3873–3877. [Google Scholar] [CrossRef]
- Bancroft, T.; Dillon, M.B.C.; da Silva Antunes, R.; Paul, S.; Peters, B.; Crotty, S.; Lindestam Arlehamn, C.S.; Sette, A. Th1 versus Th2 T cell polarization by whole-cell and acellular childhood pertussis vaccines persists upon re-immunization in adolescence and adulthood. Cell. Immunol. 2016, 304–305, 35–43. [Google Scholar] [CrossRef]
- Greco, D.; Salmaso, S.; Mastrantonio, P.; Giuliano, M.; Tozzi, A.E.; Anemona, A.; Ciofi degli Atti, M.L.; Giammanco, A.; Panei, P.; Blackwelder, W.C.; et al. A Controlled Trial of Two Acellular Vaccines and One Whole-Cell Vaccine against Pertussis. N. Engl. J. Med. 1996, 334, 341–349. [Google Scholar] [CrossRef]
- Sealey, K.L.; Belcher, T.; Preston, A. Bordetella pertussis epidemiology and evolution in the light of pertussis resurgence. Infect. Genet. Evol. 2016, 40, 136–143. [Google Scholar] [CrossRef] [PubMed]
- Gambhir, M.; Clark, T.A.; Cauchemez, S.; Tartof, S.Y.; Swerdlow, D.L.; Ferguson, N.M. A change in vaccine efficacy and duration of protection explains recent rises in pertussis incidence in the United States. PLoS Comput. Biol. 2015, 11, e1004138. [Google Scholar] [CrossRef] [PubMed]
- Cherry, J.D.; Heininger, U.; Richards, D.M.; Storsaeter, J.; Gustafsson, L.; Ljungman, M.; Hallander, H.O. Antibody response patterns to Bordetella pertussis antigens in vaccinated (primed) and unvaccinated (unprimed) young children with pertussis. Clin. Vaccine Immunol. 2010, 17, 741–747. [Google Scholar] [CrossRef] [PubMed]
- Hendrikx, L.H.; Schure, R.-M.; Öztürk, K.; de Rond, L.G.H.; de Greeff, S.C.; Sanders, E.A.M.; Berbers, G.A.M.; Buisman, A.-M. Different IgG-subclass distributions after whole-cell and acellular pertussis infant primary vaccinations in healthy and pertussis infected children. Vaccine 2011, 29, 6874–6880. [Google Scholar] [CrossRef]
- Mills, K.H.G.; Ryan, M.; Ryan, E.; Mahon, B.P. A Murine Model in Which Protection Correlates with Pertussis Vaccine Efficacy in Children Reveals Complementary Roles for Humoral and Cell-Mediated Immunity in Protection againstBordetella pertussis. Infect. Immun. 1998, 66, 594–602. [Google Scholar] [CrossRef]
- Coffman, R.L.; Seymour, B.W.P.; Lebman, D.A.; Hiraki, D.D.; Christiansen, J.A.; Shrader, B.; Cherwinski, H.M.; Savelkoul, H.F.J.; Finkelman, F.D.; Bond, M.W.; et al. The Role of Helper T Cell Products in Mouse B Cell Differentiation and Isotype Regulation. Immunol. Rev. 1988, 102, 5–28. [Google Scholar] [CrossRef]
- Bossie, A.; Vitetta, E.S. IFN-γ enhances secretion of IgG2a from IgG2a-committed LPS-stimulated murine B cells: Implications for the role of IFN-γ in class switching. Cell. Immunol. 1991, 135, 95–104. [Google Scholar] [CrossRef]
- Zhang, Z.; Goldschmidt, T.; Salter, H. Possible allelic structure of IgG2a and IgG2c in mice. Mol. Immunol. 2012, 50, 169–171. [Google Scholar] [CrossRef]
- Moon, H.B.; Severinson, E.; Heusser, C.; Johansson, S.G.O.; Möller, G.; Persson, U. Regulation of IgG1 and IgE Synthesis by Interleukin 4 in Mouse B Cells. Scand. J. Immunol. 1989, 30, 355–361. [Google Scholar] [CrossRef]
- Hellwig, S.M.M.; van Oirschot, H.F.L.M.; Hazenbos, W.L.W.; van Spriel, A.B.; Mooi, F.R.; van de Winkel, J.G.J. Targeting to Fcγ Receptors, But Not CR3 (CD11b/CD18), Increases Clearance of Bordetella pertussis. J. Infect. Dis. 2001, 183, 871–879. [Google Scholar] [CrossRef]
- Vidarsson, G.; Dekkers, G.; Rispens, T. IgG Subclasses and Allotypes: From Structure to Effector Functions. Front. Immunol. 2014, 5, 520. [Google Scholar] [CrossRef] [PubMed]
- Lin, A.; Apostolovic, D.; Jahnmatz, M.; Liang, F.; Ols, S.; Tecleab, T.; Wu, C.; van Hage, M.; Solovay, K.; Rubin, K.; et al. Live attenuated pertussis vaccine BPZE1 induces a broad antibody response in humans. J. Clin. Investig. 2020, 130, 2332–2346. [Google Scholar] [CrossRef] [PubMed]
- Tuomanen, E.I.; Zapiain, L.A.; Galvan, P.; Hewlett, E.L. Characterization of antibody inhibiting adherence of Bordetella pertussis to human respiratory epithelial cells. J. Clin. Microbiol. 1984, 20, 167–170. [Google Scholar] [CrossRef] [PubMed]
- Hellwig, S.M.; van Spriel, A.B.; Schellekens, J.F.; Mooi, F.R.; van de Winkel, J.G. Immunoglobulin A-mediated protection against Bordetella pertussis infection. Infect. Immun. 2001, 69, 4846–4850. [Google Scholar] [CrossRef]
- Christensen, D.; Mortensen, R.; Rosenkrands, I.; Dietrich, J.; Andersen, P. Vaccine-induced Th17 cells are established as resident memory cells in the lung and promote local IgA responses. Mucosal Immunol. 2017, 10, 260–270. [Google Scholar] [CrossRef]
- Jaffar, Z.; Ferrini, M.E.; Herritt, L.A.; Roberts, K. Cutting Edge: Lung Mucosal Th17-Mediated Responses Induce Polymeric Ig Receptor Expression by the Airway Epithelium and Elevate Secretory IgA Levels. J. Immunol. 2009, 182, 4507–4511. [Google Scholar] [CrossRef]
- Redhead, K.; Watkins, J.; Barnard, A.; Mills, K.H. Effective immunization against Bordetella pertussis respiratory infection in mice is dependent on induction of cell-mediated immunity. Infect. Immun. 1993, 61, 3190–3198. [Google Scholar] [CrossRef]
- Nagamatsu, K.; Kuwae, A.; Konaka, T.; Nagai, S.; Yoshida, S.; Eguchi, M.; Watanabe, M.; Mimuro, H.; Koyasu, S.; Abe, A. Bordetella evades the host immune system by inducing IL-10 through a type III effector, BopN. J. Exp. Med. 2009, 206, 3073–3088. [Google Scholar] [CrossRef]
- Mosser, D.M.; Zhang, X. Interleukin-10: New perspectives on an old cytokine. Immunol. Rev. 2008, 226, 205–218. [Google Scholar] [CrossRef]
- Saraiva, M.; O’Garra, A.; Saraiva, M.; O’Garra, A. The regulation of IL-10 production by immune cells. Nat. Rev. Immunol. 2010, 10, 170–181. [Google Scholar] [CrossRef]
- McGuirk, P.; McCann, C.; Mills, K.H.G. Pathogen-specific T regulatory 1 cells induced in the respiratory tract by a bacterial molecule that stimulates interleukin 10 production by dendritic cells: A novel strategy for evasion of protective T helper type 1 responses by Bordetella pertussis. J. Exp. Med. 2002, 195, 221–231. [Google Scholar] [CrossRef] [PubMed]
- Dirix, V.; Verscheure, V.; Vermeulen, F.; De Schutter, I.; Goetghebuer, T.; Locht, C.; Mascart, F. Both CD4+ and CD8+ lymphocytes participate in the IFN-γ response to filamentous hemagglutinin from Bordetella pertussis in infants, children, and adults. Clin. Dev. Immunol. 2012, 2012, 795958. [Google Scholar] [CrossRef] [PubMed]
- Raeven, R.H.M.; Brummelman, J.; Pennings, J.L.A.; Nijst, O.E.M.; Kuipers, B.; Blok, L.E.R.; Helm, K.; van Riet, E.; Jiskoot, W.; van Els, C.A.C.M.; et al. Molecular signatures of the evolving immune response in mice following a Bordetella pertussis infection. PLoS ONE 2014, 9, e104548. [Google Scholar] [CrossRef]
- Argondizo-Correia, C.; Rodrigues, A.K.S.; de Brito, C.A. Neonatal Immunity to Bordetella pertussis Infection and Current Prevention Strategies. J. Immunol. Res. 2019, 2019, 7134168. [Google Scholar] [CrossRef]
- Webster, R.B.; Rodriguez, Y.; Klimecki, W.T.; Vercelli, D. The human IL-13 locus in neonatal CD4+ T cells is refractory to the acquisition of a repressive chromatin architecture. J. Biol. Chem. 2007, 282, 700–709. [Google Scholar] [CrossRef] [PubMed]
- Hebel, K.; Weinert, S.; Kuropka, B.; Knolle, J.; Kosak, B.; Jorch, G.; Arens, C.; Krause, E.; Braun-Dullaeus, R.C.; Brunner-Weinzierl, M.C. CD4+ T cells from human neonates and infants are poised spontaneously to run a nonclassical IL-4 program. J. Immunol. 2014, 192, 5160–5170. [Google Scholar] [CrossRef] [PubMed]
- Adkins, B.; Yoshimoto, M. Epigenetic Regulation of the Th2 Locus in Fetal and Neonatal T Cells. Adv. Neuroimmune Biol. 2014, 5, 69–73. [Google Scholar] [CrossRef]
- Mascart, F.; Verscheure, V.; Malfroot, A.; Hainaut, M.; Piérard, D.; Temerman, S.; Peltier, A.; Debrie, A.-S.; Levy, J.; Del Giudice, G.; et al. Bordetella pertussis Infection in 2-Month-Old Infants Promotes Type 1 T Cell Responses. J. Immunol. 2003, 170, 1504–1509. [Google Scholar] [CrossRef]
- Langrish, C.L.; Chen, Y.; Blumenschein, W.M.; Mattson, J.; Basham, B.; Sedgwick, J.D.; McClanahan, T.; Kastelein, R.A.; Cua, D.J. IL-23 drives a pathogenic T cell population that induces autoimmune inflammation. J. Exp. Med. 2005, 201, 233–240. [Google Scholar] [CrossRef]
- Mills, K.H.G. Induction, function and regulation of IL-17-producing T cells. Eur. J. Immunol. 2008, 38, 2636–2649. [Google Scholar] [CrossRef]
- Kolls, J.K.; Khader, S.A. The role of Th17 cytokines in primary mucosal immunity. Cytokine Growth Factor Rev. 2010, 21, 443–448. [Google Scholar] [CrossRef] [PubMed]
- Locht, C.; Papin, J.F.; Lecher, S.; Debrie, A.-S.; Thalen, M.; Solovay, K.; Rubin, K.; Mielcarek, N. Live Attenuated Pertussis Vaccine BPZE1 Protects Baboons against Bordetella pertussis Disease and Infection. J. Infect. Dis. 2017, 216, 117–124. [Google Scholar] [CrossRef] [PubMed]
- Higgins, S.C.; Jarnicki, A.G.; Lavelle, E.C.; Mills, K.H.G. TLR4 Mediates Vaccine-Induced Protective Cellular Immunity to Bordetella pertussis: Role of IL-17-Producing T Cells. J. Immunol. 2006, 177, 7980–7989. [Google Scholar] [CrossRef]
- Dunne, A.; Ross, P.J.; Pospisilova, E.; Masin, J.; Meaney, A.; Sutton, C.E.; Iwakura, Y.; Tschopp, J.; Sebo, P.; Mills, K.H.G. Inflammasome Activation by Adenylate Cyclase Toxin Directs Th17 Responses and Protection against Bordetella pertussis. J. Immunol. 2010, 185, 1711–1719. [Google Scholar] [CrossRef] [PubMed]
- da Silva Antunes, R.; Babor, M.; Carpenter, C.; Khalil, N.; Cortese, M.; Mentzer, A.J.; Seumois, G.; Petro, C.D.; Purcell, L.A.; Vijayanand, P.; et al. Th1/Th17 polarization persists following whole-cell pertussis vaccination despite repeated acellular boosters. J. Clin. Investig. 2018, 128, 3853–3865. [Google Scholar] [CrossRef] [PubMed]
- van der Lee, S.; Hendrikx, L.H.; Sanders, E.A.M.; Berbers, G.A.M.; Buisman, A.-M. Whole-Cell or Acellular Pertussis Primary Immunizations in Infancy Determines Adolescent Cellular Immune Profiles. Front. Immunol. 2018, 9, 51. [Google Scholar] [CrossRef] [PubMed]
- Sallusto, F.; Lenig, D.; Forster, R.; Lipp, M.; Lanzavecchia, A. Two subsets of memory T lymphocytes with distinct homing potentials and effector functions. Nature 1999, 401, 708–712. [Google Scholar] [CrossRef]
- Mueller, S.N.; Mackay, L.K. Tissue-resident memory T cells: Local specialists in immune defence. Nat. Rev. Immunol. 2016, 16, 79–89. [Google Scholar] [CrossRef]
- Gebhardt, T.; Wakim, L.M.; Eidsmo, L.; Reading, P.C.; Heath, W.R.; Carbone, F.R. Memory T cells in nonlymphoid tissue that provide enhanced local immunity during infection with herpes simplex virus. Nat. Immunol. 2009, 10, 524–530. [Google Scholar] [CrossRef]
- Masopust, D.; Choo, D.; Vezys, V.; Wherry, E.J.; Duraiswamy, J.; Akondy, R.; Wang, J.; Casey, K.A.; Barber, D.L.; Kawamura, K.S.; et al. Dynamic T cell migration program provides resident memory within intestinal epithelium. J. Exp. Med. 2010, 207, 553–564. [Google Scholar] [CrossRef]
- Kumar, B.V.; Ma, W.; Miron, M.; Granot, T.; Guyer, R.S.; Carpenter, D.J.; Senda, T.; Sun, X.; Ho, S.-H.; Lerner, H.; et al. Human Tissue-Resident Memory T Cells Are Defined by Core Transcriptional and Functional Signatures in Lymphoid and Mucosal Sites. Cell Rep. 2017, 20, 2921–2934. [Google Scholar] [CrossRef] [PubMed]
- Mackay, L.K.; Braun, A.; Macleod, B.L.; Collins, N.; Tebartz, C.; Bedoui, S.; Carbone, F.R.; Gebhardt, T. Cutting edge: CD69 interference with sphingosine-1-phosphate receptor function regulates peripheral T cell retention. J. Immunol. 2015, 194, 2059–2063. [Google Scholar] [CrossRef] [PubMed]
- Teijaro, J.R.; Turner, D.; Pham, Q.; Wherry, E.J.; Lefrançois, L.; Farber, D.L. Cutting Edge: Tissue-Retentive Lung Memory CD4 T Cells Mediate Optimal Protection to Respiratory Virus Infection. J. Immunol. 2011, 187, 5510–5514. [Google Scholar] [CrossRef] [PubMed]
- Pham, O.H.; McSorley, S.J. Divergent behavior of mucosal memory T cells. Mucosal Immunol. 2015, 8, 731–734. [Google Scholar] [CrossRef]
- Schenkel, J.M.; Fraser, K.A.; Beura, L.K.; Pauken, K.E.; Vezys, V.; Masopust, D. Resident memory CD8 T cells trigger protective innate and adaptive immune responses. Science 2014, 346, 98–101. [Google Scholar] [CrossRef]
- Schenkel, J.M.; Fraser, K.A.; Vezys, V.; Masopust, D. Sensing and alarm function of resident memory CD8+ T cells. Nat. Immunol. 2013, 14, 509–513. [Google Scholar] [CrossRef]
- Holmkvist, P.; Roepstorff, K.; Uronen-Hansson, H.; Sandén, C.; Gudjonsson, S.; Patschan, O.; Grip, O.; Marsal, J.; Schmidtchen, A.; Hornum, L.; et al. A major population of mucosal memory CD4+ T cells, coexpressing IL-18Rα and DR3, display innate lymphocyte functionality. Mucosal Immunol. 2015, 8, 545–558. [Google Scholar] [CrossRef]
- Masopust, D.; Jiang, J.; Shen, H.; Lefrançois, L. Direct Analysis of the Dynamics of the Intestinal Mucosa CD8 T Cell Response to Systemic Virus Infection. J. Immunol. 2001, 166, 2348–2356. [Google Scholar] [CrossRef]
- Khanna, K.M.; Bonneau, R.H.; Kinchington, P.R.; Hendricks, R.L. Herpes simplex virus-specific memory CD8+ T cells are selectively activated and retained in latently infected sensory ganglia. Immunity 2003, 18, 593–603. [Google Scholar] [CrossRef]
- Iijima, N.; Iwasaki, A. T cell memory. A local macrophage chemokine network sustains protective tissue-resident memory CD4 T cells. Science 2014, 346, 93–98. [Google Scholar] [CrossRef]
- Kinnear, E.; Lambert, L.; McDonald, J.U.; Cheeseman, H.M.; Caproni, L.J.; Tregoning, J.S. Airway T cells protect against RSV infection in the absence of antibody. Mucosal Immunol. 2018, 11, 249–256. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.; Zhao, J.; Mangalam, A.K.; Channappanavar, R.; Fett, C.; Meyerholz, D.K.; Agnihothram, S.; Baric, R.S.; David, C.S.; Perlman, S. Airway Memory CD4+ T Cells Mediate Protective Immunity against Emerging Respiratory Coronaviruses. Immunity 2016, 44, 1379–1391. [Google Scholar] [CrossRef] [PubMed]
- Zens, K.D.; Chen, J.K.; Farber, D.L. Vaccine-generated lung tissue-resident memory T cells provide heterosubtypic protection to influenza infection. JCI Insight 2016, 1, e85832. [Google Scholar] [CrossRef] [PubMed]
- McMaster, S.R.; Wilson, J.J.; Wang, H.; Kohlmeier, J.E. Airway-Resident Memory CD8 T Cells Provide Antigen-Specific Protection against Respiratory Virus Challenge through Rapid IFN-γ Production. J. Immunol. 2015, 195, 203–209. [Google Scholar] [CrossRef]
- Paik, D.H.; Farber, D.L. Influenza infection fortifies local lymph nodes to promote lung-resident heterosubtypic immunity. J. Exp. Med. 2020, 218, e20200218. [Google Scholar] [CrossRef]
- Smith, N.M.; Wasserman, G.A.; Coleman, F.T.; Hilliard, K.L.; Yamamoto, K.; Lipsitz, E.; Malley, R.; Dooms, H.; Jones, M.R.; Quinton, L.J.; et al. Regionally compartmentalized resident memory T cells mediate naturally acquired protection against pneumococcal pneumonia. Mucosal Immunol. 2018, 11, 220–235. [Google Scholar] [CrossRef]
- Ge, C.; Monk, I.R.; Pizzolla, A.; Wang, N.; Bedford, J.G.; Stinear, T.P.; Westall, G.P.; Wakim, L.M. Bystander Activation of Pulmonary Trm Cells Attenuates the Severity of Bacterial Pneumonia by Enhancing Neutrophil Recruitment. Cell Rep. 2019, 29, 4236–4244.e3. [Google Scholar] [CrossRef]
- Woodworth, J.S.; Cohen, S.B.; Moguche, A.O.; Plumlee, C.R.; Agger, E.M.; Urdahl, K.B.; Andersen, P. Subunit vaccine H56/CAF01 induces a population of circulating CD4 T cells that traffic into the Mycobacterium tuberculosis-infected lung. Mucosal Immunol. 2017, 10, 555–564. [Google Scholar] [CrossRef]
- Hansen, S.G.; Zak, D.E.; Xu, G.; Ford, J.C.; Marshall, E.E.; Malouli, D.; Gilbride, R.M.; Hughes, C.M.; Ventura, A.B.; Ainslie, E.; et al. Prevention of tuberculosis in rhesus macaques by a cytomegalovirus-based vaccine. Nat. Med. 2018, 24, 130–143. [Google Scholar] [CrossRef]
- Bull, N.C.; Kaveh, D.A.; Garcia-Pelayo, M.C.; Stylianou, E.; McShane, H.; Hogarth, P.J. Induction and maintenance of a phenotypically heterogeneous lung tissue-resident CD4+ T cell population following BCG immunisation. Vaccine 2018, 36, 5625–5635. [Google Scholar] [CrossRef]
- Mielcarek, N.; Debrie, A.-S.; Raze, D.; Bertout, J.; Rouanet, C.; Younes, A.B.; Creusy, C.; Engle, J.; Goldman, W.E.; Locht, C. Live attenuated B. pertussis as a single-dose nasal vaccine against whooping cough. PLoS Pathog. 2006, 2, e65. [Google Scholar] [CrossRef] [PubMed]
- Thorstensson, R.; Trollfors, B.; Al-Tawil, N.; Jahnmatz, M.; Bergstrom, J.; Ljungman, M.; Torner, A.; Wehlin, L.; Van Broekhoven, A.; Bosman, F.; et al. A phase I clinical study of a live attenuated Bordetella pertussis vaccine--BPZE1; a single centre, double-blind, placebo-controlled, dose-escalating study of BPZE1 given intranasally to healthy adult male volunteers. PLoS ONE 2014, 9, e83449. [Google Scholar] [CrossRef] [PubMed]
- Feunou, P.F.; Kammoun, H.; Debrie, A.-S.; Mielcarek, N.; Locht, C. Long-term immunity against pertussis induced by a single nasal administration of live attenuated B. pertussis BPZE1. Vaccine 2010, 28, 7047–7053. [Google Scholar] [CrossRef] [PubMed]
- Feunou Feunou, P.; Bertout, J.; Locht, C. T- and B-Cell-Mediated Protection Induced by Novel, Live Attenuated Pertussis Vaccine in Mice. Cross Protection against Parapertussis. PLoS ONE 2010, 5, e10178. [Google Scholar] [CrossRef]
- Skerry, C.M.; Mahon, B.P. A Live, Attenuated Bordetella pertussis Vaccine Provides Long-Term Protection against Virulent Challenge in a Murine Model. Clin. Vaccine Immunol. 2011, 18, 187–193. [Google Scholar] [CrossRef]
- Debrie, A.-S.; Mielcarek, N.; Lecher, S.; Roux, X.; Sirard, J.-C.; Locht, C. Early Protection against Pertussis Induced by Live Attenuated Bordetella pertussis BPZE1 Depends on TLR4. J. Immunol. 2019, 203, 3293–3300. [Google Scholar] [CrossRef]
- Fedele, G.; Bianco, M.; Debrie, A.-S.; Locht, C.; Ausiello, C.M. Attenuated Bordetella pertussis Vaccine Candidate BPZE1 Promotes Human Dendritic Cell CCL21-Induced Migration and Drives a Th1/Th17 Response. J. Immunol. 2011, 186, 5388–5396. [Google Scholar] [CrossRef]
- Schwechheimer, C.; Kuehn, M.J. Outer-membrane vesicles from Gram-negative bacteria: Biogenesis and functions. Nat. Rev. Microbiol. 2015, 13, 605–619. [Google Scholar] [CrossRef]
- Raeven, R.H.M.; Brummelman, J.; Pennings, J.L.A.; van der Maas, L.; Tilstra, W.; Helm, K.; van Riet, E.; Jiskoot, W.; van Els, C.A.C.M.; Han, W.G.H.; et al. Bordetella pertussis outer membrane vesicle vaccine confers equal efficacy in mice with milder inflammatory responses compared to a whole-cell vaccine. Sci. Rep. 2016, 6, 38240. [Google Scholar] [CrossRef]
- Roberts, R.; Moreno, G.; Bottero, D.; Gaillard, M.E.; Fingermann, M.; Graieb, A.; Rumbo, M.; Hozbor, D. Outer membrane vesicles as acellular vaccine against pertussis. Vaccine 2008, 26, 4639–4646. [Google Scholar] [CrossRef]
- Zurita, M.E.; Wilk, M.M.; Carriquiriborde, F.; Bartel, E.; Moreno, G.; Misiak, A.; Mills, K.H.G.; Hozbor, D. A Pertussis Outer Membrane Vesicle-Based Vaccine Induces Lung-Resident Memory CD4 T Cells and Protection Against Bordetella pertussis, Including Pertactin Deficient Strains. Front. Cell. Infect. Microbiol. 2019, 9, 125. [Google Scholar] [CrossRef]
- Bottero, D.; Gaillard, M.E.; Zurita, E.; Moreno, G.; Martinez, D.S.; Bartel, E.; Bravo, S.; Carriquiriborde, F.; Errea, A.; Castuma, C.; et al. Characterization of the immune response induced by pertussis OMVs-based vaccine. Vaccine 2016, 34, 3303–3309. [Google Scholar] [CrossRef] [PubMed]
- Gaillard, M.E.; Bottero, D.; Errea, A.; Ormazábal, M.; Zurita, M.E.; Moreno, G.; Rumbo, M.; Castuma, C.; Bartel, E.; Flores, D.; et al. Acellular pertussis vaccine based on outer membrane vesicles capable of conferring both long-lasting immunity and protection against different strain genotypes. Vaccine 2014, 32, 931–937. [Google Scholar] [CrossRef] [PubMed]
- Asensio, C.J.A.; Gaillard, M.E.; Moreno, G.; Bottero, D.; Zurita, E.; Rumbo, M.; van der Ley, P.; van der Ark, A.; Hozbor, D. Outer membrane vesicles obtained from Bordetella pertussis Tohama expressing the lipid A deacylase PagL as a novel acellular vaccine candidate. Vaccine 2011, 29, 1649–1656. [Google Scholar] [CrossRef] [PubMed]
- Armstrong, M.E.; Loscher, C.E.; Lynch, M.A.; Mills, K.H.G. IL-1β-dependent neurological effects of the whole cell pertussis vaccine: A role for IL-1-associated signalling components in vaccine reactogenicity. J. Neuroimmunol. 2003, 136, 25–33. [Google Scholar] [CrossRef]
- Raeven, R.H.M.; Brummelman, J.; Pennings, J.L.A.; van der Maas, L.; Helm, K.; Tilstra, W.; van der Ark, A.; Sloots, A.; van der Ley, P.; van Eden, W.; et al. Molecular and cellular signatures underlying superior immunity against Bordetella pertussis upon pulmonary vaccination. Mucosal Immunol. 2018, 11, 979–993. [Google Scholar] [CrossRef]
- Raeven, R.H.M.; Rockx-Brouwer, D.; Kanojia, G.; van der Maas, L.; Bindels, T.H.E.; ten Have, R.; van Riet, E.; Metz, B.; Kersten, G.F.A. Intranasal immunization with outer membrane vesicle pertussis vaccine confers broad protection through mucosal IgA and Th17 responses. Sci. Rep. 2020, 10, 7396. [Google Scholar] [CrossRef]
- Pawloski, L.C.; Queenan, A.M.; Cassiday, P.K.; Lynch, A.S.; Harrison, M.J.; Shang, W.; Williams, M.M.; Bowden, K.E.; Burgos-Rivera, B.; Qin, X.; et al. Prevalence and molecular characterization of pertactin-deficient Bordetella pertussis in the United States. Clin. Vaccine Immunol. 2014, 21, 119–125. [Google Scholar] [CrossRef]
- Mooi, F.R.; van Loo, I.H.M.; van Gent, M.; He, Q.; Bart, M.J.; Heuvelman, K.J.; de Greeff, S.C.; Diavatopoulos, D.; Teunis, P.; Nagelkerke, N.; et al. Bordetella pertussis strains with increased toxin production associated with pertussis resurgence. Emerg. Infect. Dis. 2009, 15, 1206–1213. [Google Scholar] [CrossRef]
- Williams, M.M.; Sen, K.; Weigand, M.R.; Skoff, T.H.; Cunningham, V.A.; Halse, T.A.; Tondella, M.L.; CDC Pertussis Working Group. Bordetella pertussis Strain Lacking Pertactin and Pertussis Toxin. Emerg. Infect. Dis. 2016, 22, 319–322. [Google Scholar] [CrossRef]
- Bouchez, V.; Brun, D.; Cantinelli, T.; Dore, G.; Njamkepo, E.; Guiso, N. First report and detailed characterization of B. pertussis isolates not expressing pertussis toxin or pertactin. Vaccine 2009, 27, 6034–6041. [Google Scholar] [CrossRef]
- Barkoff, A.-M.; Mertsola, J.; Pierard, D.; Dalby, T.; Hoegh, S.V.; Guillot, S.; Stefanelli, P.; van Gent, M.; Berbers, G.; Vestrheim, D.; et al. Pertactin-deficient Bordetella pertussis isolates: Evidence of increased circulation in Europe, 1998 to 2015. Euro Surveill. 2019, 24, 1700832. [Google Scholar] [CrossRef] [PubMed]
- Miyaji, Y.; Otsuka, N.; Toyoizumi-Ajisaka, H.; Shibayama, K.; Kamachi, K. Genetic Analysis of Bordetella pertussis Isolates from the 2008–2010 Pertussis Epidemic in Japan. PLoS ONE 2013, 8, e77165. [Google Scholar] [CrossRef] [PubMed]
- Dorji, D.; Mooi, F.; Yantorno, O.; Deora, R.; Graham, R.M.; Mukkur, T.K. Bordetella Pertussis virulence factors in the continuing evolution of whooping cough vaccines for improved performance. Med. Microbiol. Immunol. 2018, 207, 3–26. [Google Scholar] [CrossRef] [PubMed]
- Decker, K.B.; James, T.D.; Stibitz, S.; Hinton, D.M. The Bordetella pertussis model of exquisite gene control by the global transcription factor BvgA. Microbiology 2012, 158, 1665–1676. [Google Scholar] [CrossRef]
- Karimova, G.; Bellalou, J.; Ullmann, A. Phosphorylation-dependent binding of BvgA to the upstream region of the cyaA gene of Bordetella pertussis. Mol. Microbiol. 1996, 20, 489–496. [Google Scholar] [CrossRef]
- Kamanova, J.; Kofronova, O.; Masin, J.; Genth, H.; Vojtova, J.; Linhartova, I.; Benada, O.; Just, I.; Sebo, P. Adenylate Cyclase Toxin Subverts Phagocyte Function by RhoA Inhibition and Unproductive Ruffling. J. Immunol. 2008, 181, 5587–5597. [Google Scholar] [CrossRef]
- Arciniega, J.L.; Hewlett, E.L.; Johnson, F.D.; Deforest, A.; Wassilak, S.G.; Onorato, I.M.; Manclark, C.R.; Burns, D.L. Human serologic response to envelope-associated proteins and adenylate cyclase toxin of Bordetella pertussis. J. Infect. Dis. 1991, 163, 135–142. [Google Scholar] [CrossRef]
- Farfel, Z.; Könen, S.; Wiertz, E.; Klapmuts, R.; Addy, P.A.; Hanski, E. Antibodies to Bordetella pertussis adenylate cyclase are produced in man during pertussis infection and after vaccination. J. Med. Microbiol. 1990, 32, 173–177. [Google Scholar] [CrossRef]
- Arciniega, J.L.; Hewlett, E.L.; Edwards, K.M.; Burns, D.L. Antibodies to Bordetella pertussis adenylate cyclase toxin in neonatal and maternal sera. FEMS Immunol. Med. Microbiol. 1993, 6, 325–330. [Google Scholar] [CrossRef]
- Guiso, N.; Rocancourt, M.; Szatanik, M.; Alonso, J. Bordetella adenylate cyclase is a virulent associated factor and an immunoprotective antigen. Microb. Pathog. 1989, 7, 373–380. [Google Scholar] [CrossRef]
- Hormozi, K.; Parton, R.; Coote, J. Adjuvant and protective properties of native and recombinant Bordetella pertussis adenylate cyclase toxin preparations in mice. FEMS Immunol. Med. Microbiol. 1999, 23, 273–282. [Google Scholar] [CrossRef] [PubMed]
- Guiso, N.; Szatanik, M.; Rocancourt, M. Protective activity of Bordetella adenylate cyclase-hemolysin against bacterial colonization. Microb. Pathog. 1991, 11, 423–431. [Google Scholar] [CrossRef]
- Khelef, N.; Sakamoto, H.; Guiso, N. Both adenylate cyclase and hemolytic activities are required by Bordetella pertussis to initiate infection. Microb. Pathog. 1992, 12, 227–235. [Google Scholar] [CrossRef]
- Cheung, G.Y.C.; Xing, D.; Prior, S.; Corbel, M.J.; Parton, R.; Coote, J.G. Effect of different forms of adenylate cyclase toxin of Bordetella pertussis on protection afforded by an acellular pertussis vaccine in a murine model. Infect. Immun. 2006, 74, 6797–6805. [Google Scholar] [CrossRef]
- Gray, M.C.; Donato, G.M.; Jones, F.R.; Kim, T.; Hewlett, E.L. Newly secreted adenylate cyclase toxin is responsible for intoxication of target cells by Bordetella pertussis. Mol. Microbiol. 2004, 53, 1709–1719. [Google Scholar] [CrossRef]
- Hanski, E.; Farfel, Z. Bordetella pertussis invasive adenylate cyclase. Partial resolution and properties of its cellular penetration. J. Biol. Chem. 1985, 260, 5526–5532. [Google Scholar]
- Wang, X.; Gray, M.C.; Hewlett, E.L.; Maynard, J.A. The Bordetella adenylate cyclase repeat-in-toxin (RTX) domain is immunodominant and elicits neutralizing antibodies. J. Biol. Chem. 2015, 290, 3576–3591. [Google Scholar] [CrossRef]
- Boehm, D.T.; Hall, J.M.; Wong, T.Y.; DiVenere, A.M.; Sen-Kilic, E.; Bevere, J.R.; Bradford, S.D.; Blackwood, C.B.; Elkins, C.M.; DeRoos, K.A.; et al. Evaluation of Adenylate Cyclase Toxoid Antigen in Acellular Pertussis Vaccines by Using a Bordetella pertussis Challenge Model in Mice. Infect. Immun. 2018, 86, e00857-17. [Google Scholar] [CrossRef]
- Barnes, M.G.; Weiss, A.A. BrkA Protein of Bordetella pertussis Inhibits the Classical Pathway of Complement after C1 Deposition. Infect. Immun. 2001, 69, 3067–3072. [Google Scholar] [CrossRef]
- Marr, N.; Oliver, D.C.; Laurent, V.; Poolman, J.; Denoel, P.; Fernandez, R.C. Protective activity of the Bordetella pertussis BrkA autotransporter in the murine lung colonization model. Vaccine 2008, 26, 4306–4311. [Google Scholar] [CrossRef] [PubMed]
- Gasperini, G.; Biagini, M.; Arato, V.; Gianfaldoni, C.; Vadi, A.; Norais, N.; Bensi, G.; Delany, I.; Pizza, M.; Aricò, B.; et al. Outer Membrane Vesicles (OMV)-based and Proteomics-driven Antigen Selection Identifies Novel Factors Contributing to Bordetella pertussis Adhesion to Epithelial Cells. Mol. Cell. Proteom. 2018, 17, 205–215. [Google Scholar] [CrossRef] [PubMed]
- de Gouw, D.; de Jonge, M.I.; Hermans, P.W.M.; Wessels, H.J.C.T.; Zomer, A.; Berends, A.; Pratt, C.; Berbers, G.A.; Mooi, F.R.; Diavatopoulos, D.A. Proteomics-Identified Bvg-Activated Autotransporters Protect against Bordetella pertussis in a Mouse Model. PLoS ONE 2014, 9, e105011. [Google Scholar]
- Coffman, R.L.; Sher, A.; Seder, R.A. Vaccine Adjuvants: Putting Innate Immunity to Work. Immunity 2010, 33, 492–503. [Google Scholar] [CrossRef] [PubMed]
- Baraff, L.J.; Shields, W.D.; Beckwith, L.; Strome, G.; Marcy, S.M.; Cherry, J.D.; Manclark, C.R. Infants and children with convulsions and hypotonic-hyporesponsive episodes following diphtheria-tetanus-pertussis immunization: Follow-up evaluation. Pediatrics 1988, 81, 789–794. [Google Scholar] [PubMed]
- McKee, A.S.; MacLeod, M.K.L.; Kappler, J.W.; Marrack, P. Immune mechanisms of protection: Can adjuvants rise to the challenge? BMC Biol. 2010, 8, 37. [Google Scholar] [CrossRef]
- Villa, P.; Ghezzi, P. Animal Models of Endotoxic Shock. In Tumor Necrosis Factor; Humana Press: Totowa, NJ, USA, 2004; pp. 199–206. [Google Scholar]
- Glenny, A.T.; Pope, C.G.; Waddington, H.; Wallace, U. Immunological notes. XVII–XXIV. J. Pathol. Bacteriol. 1926, 29, 31–40. [Google Scholar] [CrossRef]
- Oleszycka, E.; McCluskey, S.; Sharp, F.A.; Muñoz-Wolf, N.; Hams, E.; Gorman, A.L.; Fallon, P.G.; Lavelle, E.C. The vaccine adjuvant alum promotes IL-10 production that suppresses Th1 responses. Eur. J. Immunol. 2018, 48, 705–715. [Google Scholar] [CrossRef]
- Glenny, A.T.; Buttle, G.A.H.; Stevens, M.F. Rate of disappearance of diphtheria toxoid injected into rabbits and guinea - pigs: Toxoid precipitated with alum. J. Pathol. Bacteriol. 1931, 34, 267–275. [Google Scholar] [CrossRef]
- Harrison, W.T. Some Observations on the Use of Alum Precipitated Diphtheria Toxoid. Am. J. Public Health Nation’s Health 1935, 25, 298–300. [Google Scholar] [CrossRef]
- Morefield, G.L.; Sokolovska, A.; Jiang, D.; HogenEsch, H.; Robinson, J.P.; Hem, S.L. Role of aluminum-containing adjuvants in antigen internalization by dendritic cells in vitro. Vaccine 2005, 23, 1588–1595. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Willingham, S.B.; Ting, J.P.-Y.; Re, F. Cutting Edge: Inflammasome Activation by Alum and Alum’s Adjuvant Effect Are Mediated by NLRP3. J. Immunol. 2008, 181, 17–21. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Nookala, S.; Re, F. Aluminum hydroxide adjuvants activate caspase-1 and induce IL-1beta and IL-18 release. J. Immunol. 2007, 178, 5271–5276. [Google Scholar] [CrossRef]
- Franchi, L.; Núñez, G. The Nlrp3 inflammasome is critical for aluminium hydroxide-mediated IL-1beta secretion but dispensable for adjuvant activity. Eur. J. Immunol. 2008, 38, 2085–2089. [Google Scholar] [CrossRef]
- Mori, A.; Oleszycka, E.; Sharp, F.A.; Coleman, M.; Ozasa, Y.; Singh, M.; O’Hagan, D.T.; Tajber, L.; Corrigan, O.I.; McNeela, E.A.; et al. The vaccine adjuvant alum inhibits IL-12 by promoting PI3 kinase signaling while chitosan does not inhibit IL-12 and enhances Th1 and Th17 responses. Eur. J. Immunol. 2012, 42, 2709–2719. [Google Scholar] [CrossRef] [PubMed]
- Plotkin, S.A. Correlates of protection induced by vaccination. Clin. Vaccine Immunol. 2010, 17, 1055–1065. [Google Scholar] [CrossRef]
- Banus, S.; Stenger, R.M.; Gremmer, E.R.; Dormans, J.A.M.A.; Mooi, F.R.; Kimman, T.G.; Vandebriel, R.J. The role of Toll-like receptor-4 in pertussis vaccine-induced immunity. BMC Immunol. 2008, 9, 21. [Google Scholar] [CrossRef]
- Moreno, G.; Errea, A.; Van Maele, L.; Roberts, R.; Leger, H.; Sirard, J.C.; Benecke, A.; Rumbo, M.; Hozbor, D. Toll-like receptor 4 orchestrates neutrophil recruitment into airways during the first hours of Bordetella pertussis infection. Microbes Infect. 2013, 15, 708–718. [Google Scholar] [CrossRef]
- Opal, S.M. Endotoxins and Other Sepsis Triggers. In Endotoxemia and Endotoxin Shock; KARGER: Basel, Switzerland, 2010; Volume 167, pp. 14–24. ISBN 0302-5144. [Google Scholar]
- Ireton, G.C.; Reed, S.G. Adjuvants containing natural and synthetic Toll-like receptor 4 ligands. Expert Rev. Vaccines 2013, 12, 793–807. [Google Scholar] [CrossRef]
- Garçon, N.; Morel, S.; Didierlaurent, A.; Descamps, D.; Wettendorff, M.; Van Mechelen, M. Development of an AS04-Adjuvanted HPV Vaccine with the Adjuvant System Approach. BioDrugs 2011, 25, 217–226. [Google Scholar] [CrossRef]
- Leroux-Roels, G.; Van Belle, P.; Vandepapeliere, P.; Horsmans, Y.; Janssens, M.; Carletti, I.; Garçon, N.; Wettendorff, M.; Van Mechelen, M. Vaccine Adjuvant Systems containing monophosphoryl lipid A and QS-21 induce strong humoral and cellular immune responses against hepatitis B surface antigen which persist for at least 4 years after vaccination. Vaccine 2015, 33, 1084–1091. [Google Scholar] [CrossRef] [PubMed]
- Geurtsen, J.; Banus, H.A.; Gremmer, E.R.; Ferguson, H.; de la Fonteyne-Blankestijn, L.J.J.; Vermeulen, J.P.; Dormans, J.A.M.A.; Tommassen, J.; van der Ley, P.; Mooi, F.R.; et al. Lipopolysaccharide Analogs Improve Efficacy of Acellular Pertussis Vaccine and Reduce Type I Hypersensitivity in Mice. Clin. Vaccine Immunol. 2007, 14, 821–829. [Google Scholar] [CrossRef] [PubMed]
- Brummelman, J.; Helm, K.; Hamstra, H.-J.; van der Ley, P.; Boog, C.J.P.; Han, W.G.H.; van Els, C.A.C.M. Modulation of the CD4+ T cell response after acellular pertussis vaccination in the presence of TLR4 ligation. Vaccine 2015, 33, 1483–1491. [Google Scholar] [CrossRef] [PubMed]
- Brummelman, J.; Raeven, R.H.M.; Helm, K.; Pennings, J.L.A.; Metz, B.; van Eden, W.; van Els, C.A.C.M.; Han, W.G.H. Transcriptome signature for dampened Th2 dominance in acellular pertussis vaccine-induced CD4+ T cell responses through TLR4 ligation. Sci. Rep. 2016, 6, 25064. [Google Scholar] [CrossRef] [PubMed]
- Vollmer, J.; Krieg, A.M. Immunotherapeutic applications of CpG oligodeoxynucleotide TLR9 agonists. Adv. Drug Deliv. Rev. 2009, 61, 195–204. [Google Scholar] [CrossRef]
- Weeratna, R.D.; Brazolot Millan, C.L.; McCluskie, M.J.; Davis, H.L. CpG ODN can re-direct the Th bias of established Th2 immune responses in adult and young mice. FEMS Immunol. Med. Microbiol. 2001, 32, 65–71. [Google Scholar] [CrossRef]
- Bode, C.; Zhao, G.; Steinhagen, F.; Kinjo, T.; Klinman, D.M. CpG DNA as a vaccine adjuvant. Expert Rev. Vaccines 2011, 10, 499–511. [Google Scholar] [CrossRef]
- Asokanathan, C.; Corbel, M.; Xing, D. A CpG-containing oligodeoxynucleotide adjuvant for acellular pertussis vaccine improves the protective response against Bordetella pertussis. Hum. Vaccines Immunother. 2013, 9, 325–331. [Google Scholar] [CrossRef]
- Sugai, T.; Mori, M.; Nakazawa, M.; Ichino, M.; Naruto, T.; Kobayashi, N.; Kobayashi, Y.; Minami, M.; Yokota, S. A CpG-containing oligodeoxynucleotide as an efficient adjuvant counterbalancing the Th1/Th2 immune response in diphtheria–tetanus–pertussis vaccine. Vaccine 2005, 23, 5450–5456. [Google Scholar] [CrossRef]
- Bakhshaei, P.; Kazemi, M.H.; Golara, M.; Abdolmaleki, S.; Khosravi-Eghbal, R.; Khoshnoodi, J.; Judaki, M.A.; Salimi, V.; Douraghi, M.; Jeddi-Tehrani, M.; et al. Investigation of the Cellular Immune Response to Recombinant Fragments of Filamentous Hemagglutinin and Pertactin of Bordetella pertussis in BALB/c Mice. J. Interf. Cytokine Res. 2018, 38, 161–170. [Google Scholar] [CrossRef]
- Buonsanti, C.; Balocchi, C.; Harfouche, C.; Corrente, F.; Galli Stampino, L.; Mancini, F.; Tontini, M.; Malyala, P.; Bufali, S.; Baudner, B.; et al. Novel adjuvant Alum-TLR7 significantly potentiates immune response to glycoconjugate vaccines. Sci. Rep. 2016, 6, 29063. [Google Scholar] [CrossRef] [PubMed]
- Mancini, F.; Monaci, E.; Lofano, G.; Torre, A.; Bacconi, M.; Tavarini, S.; Sammicheli, C.; Arcidiacono, L.; Galletti, B.; Laera, D.; et al. One Dose of Staphylococcus aureus 4C-Staph Vaccine Formulated with a Novel TLR7-Dependent Adjuvant Rapidly Protects Mice through Antibodies, Effector CD4+ T Cells, and IL-17A. PLoS ONE 2016, 11, e0147767. [Google Scholar] [CrossRef] [PubMed]
- Bagnoli, F.; Fontana, M.R.; Soldaini, E.; Mishra, R.P.N.; Fiaschi, L.; Cartocci, E.; Nardi-Dei, V.; Ruggiero, P.; Nosari, S.; De Falco, M.G.; et al. Vaccine composition formulated with a novel TLR7-dependent adjuvant induces high and broad protection against Staphylococcus aureus. Proc. Natl. Acad. Sci. USA 2015, 112, 3680–3685. [Google Scholar] [CrossRef] [PubMed]
- Wu, T.Y.-H.; Singh, M.; Miller, A.T.; De Gregorio, E.; Doro, F.; D’Oro, U.; Skibinski, D.A.G.; Mbow, M.L.; Bufali, S.; Herman, A.E.; et al. Rational design of small molecules as vaccine adjuvants. Sci. Transl. Med. 2014, 6, 263ra160. [Google Scholar] [CrossRef]
- Misiak, A.; Leuzzi, R.; Allen, A.C.; Galletti, B.; Baudner, B.C.; D’Oro, U.; O’Hagan, D.T.; Pizza, M.; Seubert, A.; Mills, K.H.G. Addition of a TLR7 agonist to an acellular pertussis vaccine enhances Th1 and Th17 responses and protective immunity in a mouse model. Vaccine 2017, 35, 5256–5263. [Google Scholar] [CrossRef]
- Dunne, A.; Mielke, L.A.; Allen, A.C.; Sutton, C.E.; Higgs, R.; Cunningham, C.C.; Higgins, S.C.; Mills, K.H.G. A novel TLR2 agonist from Bordetella pertussis is a potent adjuvant that promotes protective immunity with an acellular pertussis vaccine. Mucosal Immunol. 2015, 8, 607–617. [Google Scholar] [CrossRef]
- Xiao, T.S.; Fitzgerald, K.A. The cGAS-STING Pathway for DNA Sensing. Mol. Cell 2013, 51, 135–139. [Google Scholar] [CrossRef]
- Burdette, D.L.; Vance, R.E. STING and the innate immune response to nucleic acids in the cytosol. Nat. Immunol. 2013, 14, 19–26. [Google Scholar] [CrossRef]
- Burdette, D.L.; Monroe, K.M.; Sotelo-Troha, K.; Iwig, J.S.; Eckert, B.; Hyodo, M.; Hayakawa, Y.; Vance, R.E. STING is a direct innate immune sensor of cyclic di-GMP. Nature 2011, 478, 515–518. [Google Scholar] [CrossRef]
- Ishikawa, H.; Barber, G.N. STING is an endoplasmic reticulum adaptor that facilitates innate immune signalling. Nature 2008, 455, 674–678. [Google Scholar] [CrossRef]
- Tanaka, Y.; Chen, Z.J. STING Specifies IRF3 Phosphorylation by TBK1 in the Cytosolic DNA Signaling Pathway. Sci. Signal. 2012, 5, ra20. [Google Scholar] [CrossRef] [PubMed]
- Elahi, S.; Van Kessel, J.; Kiros, T.G.; Strom, S.; Hayakawa, Y.; Hyodo, M.; Babiuk, L.A.; Gerdts, V. c-di-GMP enhances protective innate immunity in a murine model of pertussis. PLoS ONE 2014, 9, e109778. [Google Scholar] [CrossRef]
- Gschwandtner, M.; Derler, R.; Midwood, K.S. More Than Just Attractive: How CCL2 Influences Myeloid Cell Behavior Beyond Chemotaxis. Front. Immunol. 2019, 10, 2759. [Google Scholar] [CrossRef] [PubMed]
- Allavena, P.; Bianchi, G.; Zhou, D.; Van Damme, J.; Jílek, P.; Sozzani, S.; Mantovani, A. Induction of natural killer cell migration by monocyte chemotactic protein−1, −2 and −3. Eur. J. Immunol. 1994, 24, 3233–3236. [Google Scholar] [CrossRef] [PubMed]
- Andreasen, C.; Carbonetti, N.H. Pertussis toxin inhibits early chemokine production to delay neutrophil recruitment in response to Bordetella pertussis respiratory tract infection in mice. Infect. Immun. 2008, 76, 5139–5148. [Google Scholar] [CrossRef] [PubMed]
- Temizoz, B.; Kuroda, E.; Ohata, K.; Jounai, N.; Ozasa, K.; Kobiyama, K.; Aoshi, T.; Ishii, K.J. TLR9 and STING agonists synergistically induce innate and adaptive type-II IFN. Eur. J. Immunol. 2015, 45, 1159–1169. [Google Scholar] [CrossRef]
- Conway, M.A.; Madrigal-Estebas, L.; McClean, S.; Brayden, D.J.; Mills, K.H.G. Protection against Bordetella pertussis infection following parenteral or oral immunization with antigens entrapped in biodegradable particles: Effect of formulation and route of immunization on induction of Th1 and Th2 cells. Vaccine 2001, 19, 1940–1950. [Google Scholar] [CrossRef]
- Lindenstrøm, T.; Moguche, A.; Damborg, M.; Agger, E.M.; Urdahl, K.; Andersen, P. T Cells Primed by Live Mycobacteria Versus a Tuberculosis Subunit Vaccine Exhibit Distinct Functional Properties. EBioMedicine 2018, 27, 27–39. [Google Scholar] [CrossRef]
- Krammer, F. SARS-CoV-2 vaccines in development. Nature 2020. [Google Scholar] [CrossRef]
- Wallis, J.; Shenton, D.P.; Carlisle, R.C. Novel approaches for the design, delivery and administration of vaccine technologies. Clin. Exp. Immunol. 2019, 196, 189–204. [Google Scholar] [CrossRef]
- Wilk, M.M.; Mills, K.H.G. CD4 TRM Cells Following Infection and Immunization: Implications for More Effective Vaccine Design. Front. Immunol. 2018, 9, 1860. [Google Scholar] [CrossRef]
- Miquel-Clopés, A.; Bentley, E.G.; Stewart, J.P.; Carding, S.R. Mucosal vaccines and technology. Clin. Exp. Immunol. 2019, 196, 205–214. [Google Scholar] [CrossRef] [PubMed]
- Perdomo, C.; Zedler, U.; Kühl, A.A.; Lozza, L.; Saikali, P.; Sander, L.E.; Vogelzang, A.; Kaufmann, S.H.E.; Kupz, A. Mucosal BCG Vaccination Induces Protective Lung-Resident Memory T Cell Populations against Tuberculosis. MBio 2016, 7, e01686-16. [Google Scholar] [CrossRef]
- Bull, N.C.; Stylianou, E.; Kaveh, D.A.; Pinpathomrat, N.; Pasricha, J.; Harrington-Kandt, R.; Garcia-Pelayo, M.C.; Hogarth, P.J.; McShane, H. Enhanced protection conferred by mucosal BCG vaccination associates with presence of antigen-specific lung tissue-resident PD-1+ KLRG1− CD4+ T cells. Mucosal Immunol. 2019, 12, 555–564. [Google Scholar] [CrossRef] [PubMed]
- Van Dis, E.; Sogi, K.M.; Rae, C.S.; Sivick, K.E.; Surh, N.H.; Leong, M.L.; Kanne, D.B.; Metchette, K.; Leong, J.J.; Bruml, J.R.; et al. STING-Activating Adjuvants Elicit a Th17 Immune Response and Protect against Mycobacterium tuberculosis Infection. Cell Rep. 2018, 23, 1435–1447. [Google Scholar] [CrossRef] [PubMed]
- Santosuosso, M.; Zhang, X.; McCormick, S.; Wang, J.; Hitt, M.; Xing, Z. Mechanisms of mucosal and parenteral tuberculosis vaccinations: Adenoviral-based mucosal immunization preferentially elicits sustained accumulation of immune protective CD4 and CD8 T cells within the airway lumen. J. Immunol. 2005, 174, 7986–7994. [Google Scholar] [CrossRef]
- Dietrich, J.; Andersen, C.; Rappuoli, R.; Doherty, T.M.; Jensen, C.G.; Andersen, P. Mucosal Administration of Ag85B-ESAT-6 Protects against Infection with Mycobacterium tuberculosis and Boosts Prior Bacillus Calmette-Guérin Immunity. J. Immunol. 2006, 177, 6353–6360. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Wang, J.; Zganiacz, A.; Xing, Z. Single Intranasal Mucosal Mycobacterium bovis BCG Vaccination Confers Improved Protection Compared to Subcutaneous Vaccination against Pulmonary Tuberculosis. Infect. Immun. 2004, 72, 238–246. [Google Scholar] [CrossRef]
- Aguilo, N.; Toledo, A.M.; Lopez-Roman, E.M.; Perez-Herran, E.; Gormley, E.; Rullas-Trincado, J.; Angulo-Barturen, I.; Martin, C. Pulmonary Mycobacterium bovis BCG Vaccination Confers Dose-Dependent Superior Protection Compared to That of Subcutaneous Vaccination. Clin. Vaccine Immunol. 2014, 21, 594–597. [Google Scholar] [CrossRef]
- Barclay, W.R.; Busey, W.M.; Dalgard, D.W.; Good, R.C.; Janicki, B.W.; Kasik, J.E.; Ribi, E.; Ulrich, C.E.; Wolinsky, E. Protection of monkeys against airborne tuberculosis by aerosol vaccination with bacillus Calmette-Guerin. Am. Rev. Respir. Dis. 1973, 107, 351–358. [Google Scholar]
- Storsaeter, J.; Hallander, H.O.; Gustafsson, L.; Olin, P. Levels of anti-pertussis antibodies related to protection after household exposure to Bordetella pertussis. Vaccine 1998, 16, 1907–1916. [Google Scholar] [CrossRef]
- Solans, L.; Locht, C. The Role of Mucosal Immunity in Pertussis. Front. Immunol. 2019, 9, 3068. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chasaide, C.N.; Mills, K.H.G. Next-Generation Pertussis Vaccines Based on the Induction of Protective T Cells in the Respiratory Tract. Vaccines 2020, 8, 621. https://doi.org/10.3390/vaccines8040621
Chasaide CN, Mills KHG. Next-Generation Pertussis Vaccines Based on the Induction of Protective T Cells in the Respiratory Tract. Vaccines. 2020; 8(4):621. https://doi.org/10.3390/vaccines8040621
Chicago/Turabian StyleChasaide, Caitlín Ní, and Kingston H.G. Mills. 2020. "Next-Generation Pertussis Vaccines Based on the Induction of Protective T Cells in the Respiratory Tract" Vaccines 8, no. 4: 621. https://doi.org/10.3390/vaccines8040621
APA StyleChasaide, C. N., & Mills, K. H. G. (2020). Next-Generation Pertussis Vaccines Based on the Induction of Protective T Cells in the Respiratory Tract. Vaccines, 8(4), 621. https://doi.org/10.3390/vaccines8040621




