Adenoviral Vector-Based Vaccine Platforms for Developing the Next Generation of Influenza Vaccines
Abstract
1. Introduction
2. Current Seasonal Influenza Vaccines and Their Potential Limitations
3. Adjuvant Impact of Adenoviral Vectors
4. Significance of Ad Vector Immunity for Vaccine Platform
5. Ad Vector-Based Influenza Vaccines in Preclinical Trials
5.1. Human Ad (HAd) Vectors in Preclinical Trials
5.2. Nonhuman Ad Vectors in Preclinical Trials
6. Ad Vector-Based Influenza Vaccines in Clinical Trials
6.1. Human Ad Vectors in Clinical Trials
6.2. Nonhuman Ad Vectors in Clinical Trials
7. Potential of Ad Vectors for Developing Universal Influenza Vaccines
8. Longevity of Adenoviral Vector-Based Influenza Immunity
9. Conclusions and Future Directions
Disclaimer
Author Contributions
Funding
Conflicts of Interest
References
- Human Adenovirus Working Group. Available online: http://hadvwg.gmu.edu/ (accessed on 25 August 2020).
- Harrach, B.; Benkö, M.; Both, G.W.; Brown, M.; Davison, A.J.; Echavarría, M.; Hess, M.; Jones, M.S.; Kajon, A.; Lehmkuhl, H.D.; et al. Adenoviridae—dsDNA Viruses. In Virus Taxonomy; Ninth Report of the International Committee on Taxonomy of Viruses; International Committee on Taxonomy of Viruses; Elsevier: Amsterdam, The Netherlands, 2011. [Google Scholar]
- MacLachlan, N.J.; Dubovi, E.J. Adenoviridae. In Fenner’s Veterinary Virology, 5th ed.; MacLachlan, N.J., Dubovi, E.J., Eds.; Academic Press: Cambridge, MA, USA, 2017; pp. 217–227. [Google Scholar]
- Liu, L. Fields Virology, 6th Edition. Clin. Infect. Dis. 2014, 59, 613. [Google Scholar] [CrossRef]
- Vectors Used in Gene Therapy Clinical Trials. Available online: http://www.abedia.com/wiley/vectors.php (accessed on 20 July 2020).
- Appledorn, D.M.; Patial, S.; McBride, A.; Godbehere, S.; Van Rooijen, N.; Parameswaran, N.; Amalfitano, A. Adenovirus vector-induced innate inflammatory mediators, MAPK signaling, as well as adaptive immune responses are dependent upon both TLR2 and TLR9 in vivo. J. Immunol. 2008, 181, 2134–2144. [Google Scholar] [CrossRef] [PubMed]
- Fejer, G.; Freudenberg, M.; Greber, U.F.; Gyory, I. Adenovirus-triggered innate signalling pathways. Eur. J. Microbiol. Immunol. 2011, 1, 279–288. [Google Scholar] [CrossRef] [PubMed]
- Zhu, J.; Huang, X.; Yang, Y. Innate Immune Response to Adenoviral Vectors is Mediated by both Toll-Like Receptor-Dependent and -Independent Pathways. J. Virol. 2007, 81, 3170–3180. [Google Scholar] [CrossRef]
- Kovesdi, I.; Hedley, S.J. Adenoviral Producer Cells. Viruses 2010, 2, 1681–1703. [Google Scholar] [CrossRef]
- Payne, S. Family Orthomyxoviridae. In Viruses; Chapter 23; Payne, S., Ed.; Academic Press: Cambridge, MA, USA, 2017; pp. 197–208. [Google Scholar]
- International Committee on Taxonomy of Viruses (ICTV). Orthomyxoviridae; Negative Sense RNA Viruses; Negative Sense RNA Viruses. 2011. Available online: https://talk.ictvonline.org/ictv-reports/ictv_9th_report/negative-sense-rna-viruses-2011/w/negrna_viruses/209/orthomyxoviridae (accessed on 20 July 2020).
- Global Influenza Strategy 2019–2030. Available online: https://www.who.int/influenza/Global_Influenza_Strategy_2019_2030_Summary_English.pdf? (accessed on 20 July 2020).
- CDC. Frequently Asked Influenza (Flu) Questions: 2020–2021 Season. Available online: https://www.cdc.gov/flu/season/faq-flu-season-2020-2021.htm (accessed on 20 July 2020).
- CDC. People 65 Years and Older & Influenza. Available online: https://www.cdc.gov/flu/highrisk/65over.htm (accessed on 20 July 2020).
- Tregoning, J.S.; Russell, R.F.; Kinnear, E. Adjuvanted influenza vaccines. Hum. Vaccines Immunother. 2018, 14, 550–564. [Google Scholar] [CrossRef]
- Tricco, A.C.; Chit, A.; Soobiah, C.; Hallett, D.; Meier, G.; Chen, M.H.; Tashkandi, M.F.; Bauch, C.T.; Loeb, M. Comparing influenza vaccine efficacy against mismatched and matched strains: A systematic review and meta-analysis. BMC Med. 2013, 11, 153. [Google Scholar] [CrossRef]
- Belongia, E.A.; Kieke, B.A.; Donahue, J.G.; Greenlee, R.T.; Balish, A.; Foust, A.; Lindstrom, S.; Shay, D.K.; Marshfield Influenza Study Group. Effectiveness of Inactivated Influenza Vaccines Varied Substantially with Antigenic Match from the 2004–2005 Season to the 2006–2007 Season. J. Infect. Dis. 2009, 199, 159–167. [Google Scholar] [CrossRef]
- Zost, S.; Parkhouse, K.; Gumina, M.; Kim, K.; Diaz, P.S.; Wilson, P.; Treanor, J.; Sant, A.; Cobey, S.; Hensley, S. Contemporary H3N2 influenza viruses have a glycosylation site that alters binding of antibodies elicited by egg-adapted vaccine strains. Proc. Natl. Acad. Sci. USA 2017, 114, 12578–12583. [Google Scholar] [CrossRef]
- Garçon, N.; Vaughn, D.W.; Didierlaurent, A.M. Development and evaluation of AS03, an Adjuvant System containing α-tocopherol and squalene in an oil-in-water emulsion. Expert Rev. Vaccines 2012, 11, 349–366. [Google Scholar] [CrossRef]
- Vesikari, T.; Pépin, S.; Kusters, I.; Hoffenbach, A.; Denis, M. Assessment of squalene adjuvanted and non-adjuvanted vaccines against pandemic H1N1 influenza in children 6 months to 17 years of age. Hum. Vaccines Immunother. 2012, 8, 1283–1292. [Google Scholar] [CrossRef]
- Petrovsky, N. Vaxine. Hum. Vaccines Immunother. 2016, 12, 2726–2728. [Google Scholar] [CrossRef][Green Version]
- Vemula, S.V.; Mittal, S.K. Production of adenovirus vectors and their use as a delivery system for influenza vaccines. Expert Opin. Biol. Ther. 2010, 10, 1469–1487. [Google Scholar] [CrossRef] [PubMed]
- Ahi, Y.S.; Bangari, D.S.; Mittal, S.K. Adenoviral vector immunity: Its implications and circumvention strategies. Curr. Gene Ther. 2011, 11, 307–320. [Google Scholar] [CrossRef]
- Sharma, A.; Tandon, M.; Ahi, Y.S.; Bangari, D.S.; Vemulapalli, R.; Mittal, S.K. Evaluation of cross-reactive cell-mediated immune responses among human, bovine and porcine adenoviruses. Gene Ther. 2010, 17, 634–642. [Google Scholar] [CrossRef] [PubMed]
- Muruve, D.A. The Innate Immune Response to Adenovirus Vectors. Hum. Gene Ther. 2004, 15, 1157–1166. [Google Scholar] [CrossRef] [PubMed]
- Morelli, A.E.; Larregina, A.T.; Ganster, R.W.; Zahorchak, A.F.; Plowey, J.M.; Takayama, T.; Logar, A.J.; Robbins, P.D.; Falo, L.D.; Thomson, A.W. Recombinant Adenovirus Induces Maturation of Dendritic Cells via an NF-κB-Dependent Pathway. J. Virol. 2000, 74, 9617–9628. [Google Scholar] [CrossRef]
- Muruve, D.A.; Barnes, M.J.; Stillman, I.E.; Libermann, T.A. Adenoviral Gene Therapy Leads to Rapid Induction of Multiple Chemokines and Acute Neutrophil-Dependent Hepatic Injury in Vivo. Hum. Gene Ther. 1999, 10, 965–976. [Google Scholar] [CrossRef]
- Gregory, S.M.; Nazir, S.A.; Metcalf, J.P. Implications of the innate immune response to adenovirus and adenoviral vectors. Future Virol. 2011, 6, 357–374. [Google Scholar] [CrossRef]
- Atasheva, S.; Shayakhmetov, D.M. Adenovirus sensing by the immune system. Curr. Opin. Virol. 2016, 21, 109–113. [Google Scholar] [CrossRef]
- Sharma, A.; Bangari, D.S.; Tandon, M.; HogenEsch, H.; Mittal, S.K. Evaluation of innate immunity and vector toxicity following inoculation of bovine, porcine or human adenoviral vectors in a mouse model. Virus Res. 2010, 153, 134–142. [Google Scholar] [CrossRef] [PubMed]
- Nociari, M.; Ocheretina, O.; Schoggins, J.W.; Falck-Pedersen, E. Sensing Infection by Adenovirus: Toll-Like Receptor-Independent Viral DNA Recognition Signals Activation of the Interferon Regulatory Factor 3 Master Regulator. J. Virol. 2007, 81, 4145–4157. [Google Scholar] [CrossRef] [PubMed]
- Liu, Q.; Muruve, D.A. Molecular basis of the inflammatory response to adenovirus vectors. Gene Ther. 2003, 10, 935–940. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Nunes, F.; Berencsi, K.; Furth, E.; Gönczöl, E.; Wilson, J. Cellular immunity to viral antigens limits E1-deleted adenoviruses for gene therapy. Proc. Natl. Acad. Sci. USA 1994, 91, 4407–4411. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Ertl, H.C.; Wilson, J.M. MHC class I-cestricted cytotoxic T lymphocytes to viral antigens destroy hepatocytes in mice infected with E1-deleted recombinant adenoviruses. Immunity 1994, 1, 433–442. [Google Scholar] [CrossRef]
- Yang, Y.; Jooss, K.U.; Su, Q.; Ertl, H.C.; Wilson, J.M. Immune responses to viral antigens versus transgene product in the elimination of recombinant adenovirus-infected hepatocytes in vivo. Gene Ther. 1996, 3, 137–144. [Google Scholar]
- Yang, Y.; Wilson, J.M. Clearance of adenovirus-infected hepatocytes by MHC class I-restricted CD4+ CTLs in vivo. J. Immunol. 1995, 155, 2564–2570. [Google Scholar]
- Yang, Y.; Li, Q.; Ertl, H.C.; Wilson, J.M. Cellular and humoral immune responses to viral antigens create barriers to lung-directed gene therapy with recombinant adenoviruses. J. Virol. 1995, 69, 2004–2015. [Google Scholar] [CrossRef]
- Merkley, S.D.; Chock, C.J.; Yang, X.O.; Harris, J.; Castillo, E.F. Modulating T Cell Responses via Autophagy: The Intrinsic Influence Controlling the Function of Both Antigen-Presenting Cells and T Cells. Front. Immunol. 2018, 9, 9. [Google Scholar] [CrossRef]
- Germic, N.; Frangez, Z.; Yousefi, S.; Simon, H.U. Regulation of the innate immune system by autophagy: Monocytes, macrophages, dendritic cells and antigen presentation. Cell Death Differ. 2019, 26, 715–727. [Google Scholar] [CrossRef]
- Saini, N.K.; Baena, A.; Ng, T.W.; Venkataswamy, M.M.; Kennedy, S.C.; Kunnath-Velayudhan, S.; Carreño, L.J.; Xu, J.; Chan, J.; Larsen, M.H.; et al. Suppression of autophagy and antigen presentation by Mycobacterium tuberculosis PE_PGRS47. Nat. Microbiol. 2016, 1, 16133. [Google Scholar] [CrossRef]
- Nwanegbo, E.; Vardas, E.; Gao, W.; Whittle, H.; Sun, H.; Rowe, D.; Robbins, P.D.; Gambotto, A. Prevalence of Neutralizing Antibodies to Adenoviral Serotypes 5 and 35 in the Adult Populations of The Gambia, South Africa, and the United States. Clin. Diagn. Lab. Immunol. 2004, 11, 351–357. [Google Scholar] [CrossRef] [PubMed]
- Yu, B.; Zhou, Y.; Wu, H.; Wang, Z.; Zhan, Y.; Feng, X.; Geng, R.; Wu, Y.; Kong, W.; Yu, X. Seroprevalence of neutralizing antibodies to human adenovirus type 5 in healthy adults in China. J. Med Virol. 2012, 84, 1408–1414. [Google Scholar] [CrossRef] [PubMed]
- Thorner, A.R.; Vogels, R.; Kaspers, J.; Weverling, G.J.; Holterman, L.; Lemckert, A.A.C.; Dilraj, A.; McNally, L.M.; Jeena, P.M.; Jepsen, S.; et al. Age Dependence of Adenovirus-Specific Neutralizing Antibody Titers in Individuals from Sub-Saharan Africa. J. Clin. Microbiol. 2006, 44, 3781–3783. [Google Scholar] [CrossRef] [PubMed]
- Barouch, D.H.; Kik, S.V.; Weverling, G.J.; Dilan, R.; King, S.L.; Maxfield, L.F.; Clark, S.; Ng’Ang’A, D.; Brandariz, K.L.; Abbink, P.; et al. International seroepidemiology of adenovirus serotypes 5, 26, 35, and 48 in pediatric and adult populations. Vaccine 2011, 29, 5203–5209. [Google Scholar] [CrossRef] [PubMed]
- Mast, T.C.; Kierstead, L.; Gupta, S.B.; Nikas, A.; Kallas, E.G.; Novitsky, V.; Mbewe, B.; Pitisuttithum, P.; Schechter, M.; Vardas, E.; et al. International epidemiology of human pre-existing adenovirus (Ad) type-5, type-6, type-26 and type-36 neutralizing antibodies: Correlates of high Ad5 titers and implications for potential HIV vaccine trials. Vaccine 2010, 28, 950–957. [Google Scholar] [CrossRef]
- Pandey, A.; Singh, N.; Vemula, S.V.; Couëtil, L.; Katz, J.M.; Donis, R.; Sambhara, S.; Mittal, S.K. Impact of Preexisting Adenovirus Vector Immunity on Immunogenicity and Protection Conferred with an Adenovirus-Based H5N1 Influenza Vaccine. PLoS ONE 2012, 7, e33428. [Google Scholar] [CrossRef]
- Bangari, D.S.; Mittal, S.K. Porcine adenoviral vectors evade preexisting humoral immunity to adenoviruses and efficiently infect both human and murine cells in culture. Virus Res. 2004, 105, 127–136. [Google Scholar] [CrossRef]
- Sumida, S.M.; Truitt, D.M.; Lemckert, A.A.C.; Vogels, R.; Custers, J.H.H.V.; Addo, M.; Lockman, S.; Peter, T.; Peyerl, F.W.; Kishko, M.G.; et al. Neutralizing Antibodies to Adenovirus Serotype 5 Vaccine Vectors are Directed Primarily against the Adenovirus Hexon Protein. J. Immunol. 2005, 174, 7179–7185. [Google Scholar] [CrossRef]
- Aldhamen, Y.A.; Seregin, S.S.; Amalfitano, A. Immune Recognition of Gene Transfer Vectors: Focus on Adenovirus as a Paradigm. Front. Immunol. 2011, 2, 40. [Google Scholar] [CrossRef]
- Zaiss, A.K.; Machado, H.B.; Herschman, H.R. The influence of innate and pre-existing immunity on adenovirus therapy. J. Cell. Biochem. 2009, 108, 778–790. [Google Scholar] [CrossRef] [PubMed]
- Thacker, E.E.; Timares, L.; Matthews, Q.L. Strategies to overcome host immunity to adenovirus vectors in vaccine development. Expert Rev. Vaccines 2009, 8, 761–777. [Google Scholar] [CrossRef] [PubMed]
- Sayedahmed, E.E.; Kumari, R.; Shukla, S.; Hassan, A.O.; Mohammed, S.I.; York, I.A.; Gangappa, S.; Sambhara, S.; Mittal, S.K. Longevity of adenovirus vector immunity in mice and its implications for vaccine efficacy. Vaccine 2018, 36, 6744–6751. [Google Scholar] [CrossRef] [PubMed]
- Roy, S.; Kobinger, G.P.; Lin, J.; Figueredo, J.; Calcedo, R.; Kobasa, D.; Wilson, J.M. Partial protection against H5N1 influenza in mice with a single dose of a chimpanzee adenovirus vector expressing nucleoprotein. Vaccine 2007, 25, 6845–6851. [Google Scholar] [CrossRef]
- Singh, N.; Pandey, A.; Jayashankar, L.; Mittal, S.K. Bovine adenoviral vector-based H5N1 influenza vaccine overcomes exceptionally high levels of pre-existing immunity against human adenovirus. Mol. Ther. 2008, 16, 965–971. [Google Scholar] [CrossRef]
- Vogels, R.; Zuijdgeest, D.; van Rijnsoever, R.; Hartkoorn, E.; Damen, I.; de Béthune, M.P.; Kostense, S.; Penders, G.; Helmus, N.; Koudstaal, W.; et al. Replication-deficient human adenovirus type 35 vectors for gene transfer and vaccination: Efficient human cell infection and bypass of preexisting adenovirus immunity. J. Virol. 2003, 77, 8263–8271. [Google Scholar] [CrossRef]
- Holterman, L.; Vogels, R.; van der Vlugt, R.; Sieuwerts, M.; Grimbergen, J.; Kaspers, J.; Geelen, E.; van der Helm, E.; Lemckert, A.; Gillissen, G.; et al. Novel replication-incompetent vector derived from adenovirus type 11 (Ad11) for vaccination and gene therapy: Low seroprevalence and non-cross-reactivity with Ad5. J. Virol. 2004, 78, 13207–13215. [Google Scholar] [CrossRef]
- Abbink, P.; Lemckert, A.A.; Ewald, B.A.; Lynch, D.M.; Denholtz, M.; Smits, S.; Holterman, L.; Damen, I.; Vogels, R.; Thorner, A.R.; et al. Comparative seroprevalence and immunogenicity of six rare serotype recombinant adenovirus vaccine vectors from subgroups B and D. J. Virol. 2007, 81, 4654–4663. [Google Scholar] [CrossRef]
- Kahl, C.A.; Bonnell, J.; Hiriyanna, S.; Fultz, M.; Nyberg-Hoffman, C.; Chen, P.; King, C.R.; Gall, J.G. Potent immune responses and in vitro pro-inflammatory cytokine suppression by a novel adenovirus vaccine vector based on rare human serotype 28. Vaccine 2010, 28, 5691–5702. [Google Scholar] [CrossRef]
- Lapuente, D.; Ruzsics, Z.; Thirion, C.; Tenbusch, M. Evaluation of adenovirus 19a as a novel vector for mucosal vaccination against influenza A viruses. Vaccine 2018, 36, 2712–2720. [Google Scholar] [CrossRef]
- Crosby, C.M.; Matchett, W.E.; Anguiano-Zarate, S.S.; Parks, C.A.; Weaver, E.A.; Pease, L.R.; Webby, R.J.; Barry, M.A. Replicating Single-Cycle Adenovirus Vectors Generate Amplified Influenza Vaccine Responses. J. Virol. 2017, 91. [Google Scholar] [CrossRef] [PubMed]
- Weaver, E.A.; Barry, M.A. Low seroprevalent species D adenovirus vectors as influenza vaccines. PLoS ONE 2013, 8, e73313. [Google Scholar] [CrossRef] [PubMed]
- Mittal, S.K.; Prevec, L.; Graham, F.L.; Babiuk, L.A. Development of a bovine adenovirus type 3-based expression vector. J. Gen. Virol. 1995, 76, 93–102. [Google Scholar] [CrossRef]
- Kremer, E.J.; Boutin, S.; Chillon, M.; Danos, O. Canine adenovirus vectors: An alternative for adenovirus-mediated gene transfer. J. Virol. 2000, 74, 505–512. [Google Scholar] [CrossRef]
- Mittal, S.K.; Ahi, Y.S.; Vemula, S.V. Xenogenic Adenoviral Vectors. In Adenoviral Vectors for Gene Therapy, 2nd ed.; Chapter 19; Curiel, D.T., Ed.; Academic Press: Cambridge, MA, USA, 2016; pp. 495–528. [Google Scholar]
- Sailaja, G.; HogenEsch, H.; North, A.; Hays, J.; Mittal, S.K. Encapsulation of recombinant adenovirus into alginate microspheres circumvents vector-specific immune response. Gene Ther. 2002, 9, 1722–1729. [Google Scholar] [CrossRef]
- Mittal, S.K.; Aggarwal, N.; Sailaja, G.; van Olphen, A.; HogenEsch, H.; North, A.; Hays, J.; Moffatt, S. Immunization with DNA, adenovirus or both in biodegradable alginate microspheres: Effect of route of inoculation on immune response. Vaccine 2000, 19, 253–263. [Google Scholar] [CrossRef]
- Gall, J.G.; Crystal, R.G.; Falck-Pedersen, E. Construction and characterization of hexon-chimeric adenoviruses: Specification of adenovirus serotype. J. Virol. 1998, 72, 10260–10264. [Google Scholar] [CrossRef]
- Tang, M.; Harp, J.A.; Wesley, R.D. Recombinant adenovirus encoding the HA gene from swine H3N2 influenza virus partially protects mice from challenge with heterologous virus: A/HK/1/68 (H3N2). Arch. Virol. 2002, 147, 2125–2141. [Google Scholar] [CrossRef]
- Gao, W.; Soloff, A.C.; Lu, X.; Montecalvo, A.; Nguyen, D.C.; Matsuoka, Y.; Robbins, P.D.; Swayne, D.E.; Donis, R.O.; Katz, J.M.; et al. Protection of mice and poultry from lethal H5N1 avian influenza virus through adenovirus-based immunization. J. Virol. 2006, 80, 1959–1964. [Google Scholar] [CrossRef] [PubMed]
- Hoelscher, M.A.; Garg, S.; Bangari, D.S.; Belser, J.A.; Lu, X.; Stephenson, I.; Bright, R.A.; Katz, J.M.; Mittal, S.K.; Sambhara, S. Development of adenoviral-vector-based pandemic influenza vaccine against antigenically distinct human H5N1 strains in mice. Lancet 2006, 367, 475–481. [Google Scholar] [CrossRef]
- Steitz, J.; Barlow, P.; Hossain, J.; Kim, E.; Okada, K.; Kenniston, T.; Rea, S.; Donis, R.; Gambotto, A. A candidate H1N1 pandemic influenza vaccine elicits protective immunity in mice. PLoS ONE 2010, 5. [Google Scholar] [CrossRef] [PubMed]
- Hoelscher, M.; Jayashankar, L.; Garg, S.; Veguilla, V.; lu, X.; Singh, N.; Katz, J.; Mittal, S.; Sambhara, S. New Pre-pandemic Influenza Vaccines: An Egg-and Adjuvant-independent Human Adenoviral Vector Strategy Induces Long-lasting Protective Immune Responses in Mice. Clin. Pharmacol. Ther. 2007, 82, 665–671. [Google Scholar] [CrossRef]
- Hoelscher, M.A.; Singh, N.; Garg, S.; Jayashankar, L.; Veguilla, V.; Pandey, A.; Matsuoka, Y.; Katz, J.M.; Donis, R.; Mittal, S.K.; et al. A broadly protective vaccine against globally dispersed clade 1 and clade 2 H5N1 influenza viruses. J. Infect. Dis. 2008, 197, 1185–1188. [Google Scholar] [CrossRef] [PubMed]
- Holman, D.H.; Wang, D.; Raja, N.U.; Luo, M.; Moore, K.M.; Woraratanadharm, J.; Mytle, N.; Dong, J.Y. Multi-antigen vaccines based on complex adenovirus vectors induce protective immune responses against H5N1 avian influenza viruses. Vaccine 2008, 26, 2627–2639. [Google Scholar] [CrossRef] [PubMed]
- Epstein, S.L.; Kong, W.P.; Misplon, J.A.; Lo, C.Y.; Tumpey, T.M.; Xu, L.; Nabel, G.J. Protection against multiple influenza A subtypes by vaccination with highly conserved nucleoprotein. Vaccine 2005, 23, 5404–5410. [Google Scholar] [CrossRef]
- Tompkins, S.M.; Zhao, Z.S.; Lo, C.Y.; Misplon, J.A.; Liu, T.; Ye, Z.; Hogan, R.J.; Wu, Z.; Benton, K.A.; Tumpey, T.M.; et al. Matrix protein 2 vaccination and protection against influenza viruses, including subtype H5N1. Emerg. Infect. Dis. 2007, 13, 426–435. [Google Scholar] [CrossRef]
- Leung, H.C.; Chan, C.C.; Poon, V.K.; Zhao, H.J.; Cheung, C.Y.; Ng, F.; Huang, J.D.; Zheng, B.J. An H5N1-based matrix protein 2 ectodomain tetrameric peptide vaccine provides cross-protection against lethal infection with H7N9 influenza virus. Emerg. Microbes Infect. 2015, 4, e22. [Google Scholar] [CrossRef]
- Scallan, C.D.; Lindbloom, J.D.; Tucker, S.N. Oral Modeling of an Adenovirus-Based Quadrivalent Influenza Vaccine in Ferrets and Mice. Infect. Dis. Ther. 2016, 5, 165–183. [Google Scholar] [CrossRef]
- Cao, W.; Liepkalns, J.S.; Hassan, A.O.; Kamal, R.P.; Hofstetter, A.R.; Amoah, S.; Kim, J.H.; Reber, A.J.; Stevens, J.; Katz, J.M.; et al. A highly immunogenic vaccine against A/H7N9 influenza virus. Vaccine 2016, 34, 744–749. [Google Scholar] [CrossRef][Green Version]
- Sayedahmed, E.E.; Hassan, A.O.; Kumari, R.; Cao, W.; Gangappa, S.; York, I.; Sambhara, S.; Mittal, S.K. A bovine adenoviral vector-based H5N1 influenza -vaccine provides enhanced immunogenicity and protection at a significantly low dose. Mol. Ther. Methods Clin. Dev. 2018, 10, 210–222. [Google Scholar] [CrossRef]
- Rao, S.; Kong, W.; Wei, C.; Van Hoeven, N.; Gorres, J.; Nason, M.; Andersen, H.; Tumpey, T.; Nabel, G. Comparative efficacy of hemagglutinin, nucleoprotein, and matrix 2 protein gene-based vaccination against H5N1 influenza in mouse and ferret. PLoS ONE 2010, 5, e9812. [Google Scholar] [CrossRef] [PubMed]
- Price, G.E.; Soboleski, M.R.; Lo, C.Y.; Misplon, J.A.; Pappas, C.; Houser, K.V.; Tumpey, T.M.; Epstein, S.L. Vaccination focusing immunity on conserved antigens protects mice and ferrets against virulent H1N1 and H5N1 influenza A viruses. Vaccine 2009, 27, 6512–6521. [Google Scholar] [CrossRef] [PubMed]
- Ciaran, D.S.; Debora, W.T.; Jonathan, D.L.; James, S.T.; Sean, N.T. An adenovirus-based vaccine with a double-stranded RNA adjuvant protects mice and ferrets against H5N1 avian influenza in oral delivery models. Clin Vaccine Immunol 2013, 20, 85–94. [Google Scholar] [CrossRef]
- Wesley, R.D.; Tang, M.; Lager, K.M. Protection of weaned pigs by vaccination with human adenovirus 5 recombinant viruses expressing the hemagglutinin and the nucleoprotein of H3N2 swine influenza virus. Vaccine 2004, 22, 3427–3434. [Google Scholar] [CrossRef] [PubMed]
- Wesley, R.; Lager, K. Evaluation of a recombinant human adenovirus-5 vaccine administered via needle-free device and intramuscular injection for vaccination of pigs against swine influenza virus. Am. J. Vet. Res. 2005, 66, 1943–1947. [Google Scholar] [CrossRef]
- Braucher, D.; Henningson, J.; Loving, C.; Vincent, A.; Kim, E.; Steitz, J.; Gambotto, A.; Kehrli, M. Intranasal vaccination with replication-defective adenovirus type 5 encoding influenza virus hemagglutinin elicits protective immunity to homologous challenge and partial protection to heterologous challenge in pigs. Clin. Vaccine Immunol. 2012, 19, 1722–1729. [Google Scholar] [CrossRef]
- Alexander, J.; Ward, S.; Mendy, J.; Manayani, D.; Farness, P.; Avanzini, J.; Guenther, B.; Garduno, F.; Jow, L.; Snarsky, V.; et al. Pre-clinical evaluation of a replication-competent recombinant adenovirus serotype 4 vaccine expressing influenza H5 hemagglutinin. PLoS ONE 2012, 7, e31177. [Google Scholar] [CrossRef]
- Vitelli, A.; Quirion, M.R.; Lo, C.Y.; Misplon, J.A.; Grabowska, A.K.; Pierantoni, A.; Ammendola, V.; Price, G.E.; Soboleski, M.R.; Cortese, R.; et al. Vaccination to conserved influenza antigens in mice using a novel simian adenovirus vector, PanAd3, derived from the bonobo Pan paniscus. PLoS ONE 2013, 8, e55435. [Google Scholar] [CrossRef]
- Cheng, T.; Wang, X.; Song, Y.; Tang, X.; Zhang, C.; Zhang, H.; Jin, X.; Zhou, D. Chimpanzee adenovirus vector-based avian influenza vaccine completely protects mice against lethal challenge of H5N1. Vaccine 2016, 34, 4875–4883. [Google Scholar] [CrossRef]
- Wang, X.; Fu, W.; Yuan, S.; Yang, X.; Song, Y.; Liu, L.; Chi, Y.; Cheng, T.; Xing, M.; Zhang, Y.; et al. Both haemagglutinin-specific antibody and T cell responses induced by a chimpanzee adenoviral vaccine confer protection against influenza H7N9 viral challenge. Sci. Rep. 2017, 7, 1854. [Google Scholar] [CrossRef]
- Li, X.; Bangari, D.S.; Sharma, A.; Mittal, S.K. Bovine adenovirus serotype 3 utilizes sialic acid as a cellular receptor for virus entry. Virology 2009, 392, 162–168. [Google Scholar] [CrossRef] [PubMed]
- Roelvink, P.; Lizonova, A.; Lee, J.; Li, Y.; Bergelson, J.; Finberg, R.; Brough, D.; Kovesdi, I.; Wickham, T. The coxsackievirus-adenovirus receptor protein can function as a cellular attachment protein for adenovirus serotypes from subgroups A, C, D, E, and F. J. Virol. 1998, 72, 7909–7915. [Google Scholar] [CrossRef]
- Nestić, D.; Uil, T.; Ma, J.; Roy, S.; Vellinga, J.; Baker, A.; Custers, J.; Majhen, D. αvβ3 integrin is required for efficient infection of epithelial cells with human adenovirus type 26. J. Virol. 2018, 93, e01474-18. [Google Scholar] [CrossRef] [PubMed]
- Cohen, C.; Xiang, Z.; Gao, G.; Ertl, H.; Wilson, J.; Bergelson, J. Chimpanzee adenovirus CV-68 adapted as a gene delivery vector interacts with the coxsackievirus and adenovirus receptor. J. Gen. Virol. 2002, 83, 151–155. [Google Scholar] [CrossRef]
- Patel, A.; Tikoo, S.; Kobinger, G. A porcine adenovirus with low human seroprevalence is a promising alternative vaccine vector to human adenovirus 5 in an H5N1 virus disease model. PLoS ONE 2010, 5, e15301. [Google Scholar] [CrossRef]
- Peters, W.; Brandl, J.; Lindbloom, J.; Martinez, C.; Scallan, C.; Trager, G.; Tingley, D.; Kabongo, M.; Tucker, S. Oral administration of an adenovirus vector encoding both an avian influenza A hemagglutinin and a TLR3 ligand induces antigen specific granzyme B and IFN-γ T cell responses in humans. Vaccine 2013, 31, 1752–1758. [Google Scholar] [CrossRef] [PubMed]
- Liebowitz, D.; Lindbloom, J.D.; Brandl, J.R.; Garg, S.J.; Tucker, S.N. High titre neutralising antibodies to influenza after oral tablet immunisation: A phase 1, randomised, placebo-controlled trial. Lancet Infect. Dis. 2015, 15, 1041–1048. [Google Scholar] [CrossRef]
- ClinicalTrials.gov. Safety and Immunogenicity Study of Adenovirus-Vectored, Intranasal Pandemic Influenza Vaccine; Full Text View. Available online: https://clinicaltrials.gov/ct2/show/NCT00755703 (accessed on 20 July 2020).
- Liebowitz, D.; Gottlieb, K.; Kolhatkar, N.S.; Garg, S.J.; Asher, J.M.; Nazareno, J.; Kim, K.; McIlwain, D.R.; Tucker, S.N. Efficacy, immunogenicity, and safety of an oral influenza vaccine: A placebo-controlled and active-controlled phase 2 human challenge study. Lancet Infect. Dis. 2020, 20, 435–444. [Google Scholar] [CrossRef]
- Kim, L.; Martinez, C.J.; Hodgson, K.A.; Trager, G.R.; Brandl, J.R.; Sandefer, E.P.; Doll, W.J.; Liebowitz, D.; Tucker, S.N. Systemic and mucosal immune responses following oral adenoviral delivery of influenza vaccine to the human intestine by radio controlled capsule. Sci. Rep. 2016, 6. [Google Scholar] [CrossRef]
- Kolhatkar, N.; Gottlieb, K.; Kasparek, K.; Hodgson, K.; Tucker, S.; Liebowitz, D. 1947. Influenza Vaccination via Oral Tablet is Protective and Induces a Unique Mucosal Immune Response. Open Forum Infect. Dis. 2018, 5, S561–S562. [Google Scholar] [CrossRef]
- ClinicalTrials.gov. Single-Ascending-Dose Study of the Safety and Immunogenicity of NasoVAX—Study Results. Available online: https://clinicaltrials.gov/ct2/show/results/NCT03232567 (accessed on 20 July 2020).
- Gurwith, M.; Lock, M.; Taylor, E.M.; Ishioka, G.; Alexander, J.; Mayall, T.; Ervin, J.E.; Greenberg, R.N.; Strout, C.; Treanor, J.J.; et al. Safety and immunogenicity of an oral, replicating adenovirus serotype 4 vector vaccine for H5N1 influenza: A randomised, double-blind, placebo-controlled, phase 1 study. Lancet Infect. Dis. 2013, 13, 238–250. [Google Scholar] [CrossRef]
- Khurana, S.; Coyle, E.M.; Manischewitz, J.; King, L.R.; Ishioka, G.; Alexander, J.; Smith, J.; Gurwith, M.; Golding, H. Oral priming with replicating adenovirus serotype 4 followed by subunit H5N1 vaccine boost promotes antibody affinity maturation and expands H5N1 cross-clade neutralization. PLoS ONE 2015, 10, e0115476. [Google Scholar] [CrossRef]
- Antrobus, R.D.; Berthoud, T.K.; Mullarkey, C.E.; Hoschler, K.; Coughlan, L.; Zambon, M.; Hill, A.V.; Gilbert, S.C. Coadministration of seasonal influenza vaccine and MVA-NP+M1 simultaneously achieves potent humoral and cell-mediated responses. Mol. Ther. 2014, 22, 233–238. [Google Scholar] [CrossRef] [PubMed]
- Dicks, M.D.; Spencer, A.J.; Edwards, N.J.; Wadell, G.; Bojang, K.; Gilbert, S.C.; Hill, A.V.; Cottingham, M.G. A novel chimpanzee adenovirus vector with low human seroprevalence: Improved systems for vector derivation and comparative immunogenicity. PLoS ONE 2012, 7, e40385. [Google Scholar] [CrossRef] [PubMed]
- Antrobus, R.D.; Coughlan, L.; Berthoud, T.K.; Dicks, M.D.; Hill, A.V.; Lambe, T.; Gilbert, S.C. Clinical assessment of a novel recombinant simian adenovirus ChAdOx1 as a vectored vaccine expressing conserved Influenza A antigens. Mol. Ther. 2014, 22, 668–674. [Google Scholar] [CrossRef]
- Coughlan, L.; Sridhar, S.; Payne, R.; Edmans, M.; Milicic, A.; Venkatraman, N.; Lugonja, B.; Clifton, L.; Qi, C.; Folegatti, P.M.; et al. Heterologous Two-Dose Vaccination with Simian Adenovirus and Poxvirus Vectors Elicits Long-Lasting Cellular Immunity to Influenza Virus A in Healthy Adults. EBioMedicine 2018, 29, 146–154. [Google Scholar] [CrossRef]
- Nakajima, K. The mechanism of antigenic shift and drift of human influenza virus. Nihon Rinsho Jpn. J. Clin. Med. 2003, 61, 1897–1903. [Google Scholar]
- Gregory, V.; Bennett, M.; Orkhan, M.H.; Al Hajjar, S.; Varsano, N.; Mendelson, E.; Zambon, M.; Ellis, J.; Hay, A.; Lin, Y.P. Emergence of influenza A H1N2 reassortant viruses in the human population during 2001. Virology 2002, 300, 1–7. [Google Scholar] [CrossRef]
- Al Faress, S.; Cartet, G.; Ferraris, O.; Norder, H.; Valette, M.; Lina, B. Divergent genetic evolution of hemagglutinin in influenza A H1N1 and A H1N2 subtypes isolated in the south-France since the winter of 2001–2002. J. Clin. Virol. 2005, 33, 230–236. [Google Scholar] [CrossRef]
- Lin, Y.P.; Gregory, V.; Bennett, M.; Hay, A. Recent changes among human influenza viruses. Virus Res. 2004, 103, 47–52. [Google Scholar] [CrossRef]
- Lewnard, J.; Cobey, S. Immune History and Influenza Vaccine Effectiveness. Vaccines 2018, 6, 28. [Google Scholar] [CrossRef] [PubMed]
- Hassan, A.O.; Amen, O.; Sayedahmed, E.E.; Vemula, S.V.; Amoah, S.; York, I.; Gangappa, S.; Sambhara, S.; Mittal, S.K. Adenovirus vector-based multi-epitope vaccine provides partial protection against H5, H7, and H9 avian influenza viruses. PLoS ONE 2017, 12, e0186244. [Google Scholar] [CrossRef] [PubMed]
- Vemula, S.V.; Ahi, Y.S.; Swaim, A.M.; Katz, J.M.; Donis, R.; Sambhara, S.; Mittal, S.K. Broadly protective adenovirus-based multivalent vaccines against highly pathogenic avian influenza viruses for pandemic preparedness. PLoS ONE 2013, 8, e62496. [Google Scholar] [CrossRef] [PubMed]
- Price, G.E.; Lo, C.Y.; Misplon, J.A.; Epstein, S.L. Mucosal immunization with a candidate universal influenza vaccine reduces virus transmission in a mouse model. J. Virol. 2014. [Google Scholar] [CrossRef]
- Fan, X.; Hashem, A.M.; Chen, Z.; Li, C.; Doyle, T.; Zhang, Y.; Yi, Y.; Farnsworth, A.; Xu, K.; Li, Z.; et al. Targeting the HA2 subunit of influenza A virus hemagglutinin via CD40L provides universal protection against diverse subtypes. Mucosal Immunol. 2015, 8, 211–220. [Google Scholar] [CrossRef]
- Price, G.E.; Lo, C.Y.; Misplon, J.A.; Epstein, S.L. Reduction of influenza virus transmission from mice immunized against conserved viral antigens is influenced by route of immunization and choice of vaccine antigen. Vaccine 2018, 36, 4910–4918. [Google Scholar] [CrossRef]
- Lapuente, D.; Bonsmann, M.S.G.; Maaske, A.; Stab, V.; Heinecke, V.; Watzstedt, K.; Heß, R.; Westendorf, A.M.; Bayer, W.; Ehrhardt, C.; et al. IL-1β as mucosal vaccine adjuvant: The specific induction of tissue-resident memory T cells improves the heterosubtypic immunity against influenza A viruses. Mucosal Immunol. 2018, 11, 1265–1278. [Google Scholar] [CrossRef] [PubMed]
- Tutykhina, I.; Esmagambetov, I.; Bagaev, A.; Pichugin, A.; Lysenko, A.; Shcherbinin, D.; Sedova, E.; Logunov, D.; Shmarov, M.; Ataullakhanov, R.; et al. Vaccination potential of B and T epitope-enriched NP and M2 against Influenza A viruses from different clades and hosts. PLoS ONE 2018, 13, e0191574. [Google Scholar] [CrossRef]
- Lingel, A.; Bullard, B.L.; Weaver, E.A. Efficacy of an adenoviral vectored multivalent centralized influenza Vaccine. Sci. Rep. 2017, 7, 14912. [Google Scholar] [CrossRef] [PubMed]
- Kim, E.H.; Han, G.Y.; Nguyen, H. An adenovirus-vectored influenza vaccine induces durable cross-protective hemagglutinin stalk antibody responses in mice. Viruses 2017, 9, 234. [Google Scholar] [CrossRef] [PubMed]
- Tang, X.; Yang, Y.; Xia, X.; Zhang, C.; Yang, X.; Song, Y.; Dai, X.; Wang, M.; Zhou, D. Recombinant Adenoviruses Displaying Matrix 2 Ectodomain Epitopes on Their Fiber Proteins as Universal Influenza Vaccines. J. Virol. 2017, 91. [Google Scholar] [CrossRef] [PubMed]
- Uddback, I.E.; Pedersen, L.M.; Pedersen, S.R.; Steffensen, M.A.; Holst, P.J.; Thomsen, A.R.; Christensen, J.P. Combined local and systemic immunization is essential for durable T-cell mediated heterosubtypic immunity against influenza A virus. Sci. Rep. 2016, 6, 20137. [Google Scholar] [CrossRef]
- Kim, E.H.; Park, H.J.; Han, G.Y.; Song, M.K.; Pereboev, A.; Hong, J.S.; Chang, J.; Byun, Y.H.; Seong, B.L.; Nguyen, H.H. Intranasal adenovirus-vectored vaccine for induction of long-lasting humoral immunity-mediated broad protection against influenza in mice. J. Virol. 2014, 88, 9693–9703. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.H.; Kim, J.Y.; Choi, Y.; Nguyen, H.H.; Song, M.K.; Chang, J. Mucosal vaccination with recombinant adenovirus encoding nucleoprotein provides potent protection against influenza virus infection. PLoS ONE 2013, 8, e75460. [Google Scholar] [CrossRef]
- Hashem, A.; Jaentschke, B.; Gravel, C.; Tocchi, M.; Doyle, T.; Rosu-Myles, M.; He, R.; Li, X. Subcutaneous immunization with recombinant adenovirus expressing influenza A nucleoprotein protects mice against lethal viral challenge. Hum. Vaccin. Immunother. 2012, 8, 425–430. [Google Scholar] [CrossRef] [PubMed]
- Zhou, D.; Wu, T.L.; Lasaro, M.O.; Latimer, B.P.; Parzych, E.M.; Bian, A.; Li, Y.; Li, H.; Erikson, J.; Xiang, Z.; et al. A universal influenza A vaccine based on adenovirus expressing matrix-2 ectodomain and nucleoprotein protects mice from lethal challenge. Mol. Ther. 2010, 18, 2182–2189. [Google Scholar] [CrossRef]
- Krammer, F. The human antibody response to influenza A virus infection and vaccination. Nat. Rev. Immunol. 2019, 19, 383–397. [Google Scholar] [CrossRef]
- Sun, H.; Xiao, Y.; Liu, J.; Wang, D.; Li, F.; Wang, C.; Li, C.; Zhu, J.; Song, J.; Sun, H.; et al. Prevalent Eurasian avian-like H1N1 swine influenza virus with 2009 pandemic viral genes facilitating human infection. Proc. Natl. Acad. Sci. USA 2020. [Google Scholar] [CrossRef]
- Zhu, F.C.; Wurie, A.H.; Hou, L.H.; Liang, Q.; Li, Y.H.; Russell, J.B.; Wu, S.P.; Li, J.X.; Hu, Y.M.; Guo, Q.; et al. Safety and immunogenicity of a recombinant adenovirus type-5 vector-based Ebola vaccine in healthy adults in Sierra Leone: A single-centre, randomised, double-blind, placebo-controlled, phase 2 trial. Lancet 2017, 389, 621–628. [Google Scholar] [CrossRef]
- De Santis, O.; Audran, R.; Pothin, E.; Warpelin-Decrausaz, L.; Vallotton, L.; Wuerzner, G.; Cochet, C.; Estoppey, D.; Steiner-Monard, V.; Lonchampt, S.; et al. Safety and immunogenicity of a chimpanzee adenovirus-vectored Ebola vaccine in healthy adults: A randomised, double-blind, placebo-controlled, dose-finding, phase 1/2a study. Lancet Infect. Dis. 2016, 16, 311–320. [Google Scholar] [CrossRef]
- Zhu, F.C.; Guan, X.H.; Li, Y.H.; Huang, J.Y.; Jiang, T.; Hou, L.H.; Li, J.X.; Yang, B.F.; Wang, L.; Wang, W.J.; et al. Immunogenicity and safety of a recombinant adenovirus type-5-vectored COVID-19 vaccine in healthy adults aged 18 years or older: A randomised, double-blind, placebo-controlled, phase 2 trial. Lancet 2020, 396, 479–488. [Google Scholar] [CrossRef]
- Folegatti, P.M.; Ewer, K.J.; Aley, P.K.; Angus, B.; Becker, S.; Belij-Rammerstorfer, S.; Bellamy, D.; Bibi, S.; Bittaye, M.; Clutterbuck, E.A.; et al. Safety and immunogenicity of the ChAdOx1 nCoV-19 vaccine against SARS-CoV-2: A preliminary report of a phase 1/2, single-blind, randomised controlled trial. Lancet 2020, 396, 467–478. [Google Scholar] [CrossRef]
- Ledgerwood, J.E.; DeZure, A.D.; Stanley, D.A.; Coates, E.E.; Novik, L.; Enama, M.E.; Berkowitz, N.M.; Hu, Z.; Joshi, G.; Ploquin, A.; et al. Chimpanzee Adenovirus Vector Ebola Vaccine. N. Engl. J. Med. 2017, 376, 928–938. [Google Scholar] [CrossRef] [PubMed]
- Dolzhikova, I.V.; Tokarskaya, E.A.; Dzharullaeva, A.S.; Tukhvatulin, A.I.; Shcheblyakov, D.V.; Voronina, O.L.; Syromyatnikova, S.I.; Borisevich, S.V.; Pantyukhov, V.B.; Babira, V.F.; et al. Virus-Vectored Ebola Vaccines. Acta Naturae 2017, 9, 4–11. [Google Scholar] [CrossRef] [PubMed]
- Weaver, E.A.; Nehete, P.N.; Buchl, S.S.; Senac, J.S.; Palmer, D.; Ng, P.; Sastry, K.J.; Barry, M.A. Comparison of replication-competent, first generation, and helper-dependent adenoviral vaccines. PLoS ONE 2009, 4, e5059. [Google Scholar] [CrossRef]
- Weaver, E.A.; Rubrum, A.M.; Webby, R.J.; Barry, M.A. Protection against divergent influenza H1N1 virus by a centralized influenza hemagglutinin. PLoS ONE 2011, 6, e18314. [Google Scholar] [CrossRef]
- Vemula, S.V.; Amen, O.; Katz, J.M.; Donis, R.; Sambhara, S.; Mittal, S.K. Beta-defensin 2 enhances immunogenicity and protection of an adenovirus-based H5N1 influenza vaccine at an early time. Virus Res. 2013, 178, 398–403. [Google Scholar] [CrossRef]
- Vemula, S.V.; Pandey, A.; Singh, N.; Katz, J.M.; Donis, R.; Sambhara, S.; Mittal, S.K. Adenoviral vector expressing murine β-defensin 2 enhances immunogenicity of an adenoviral vector based H5N1 influenza vaccine in aged mice. Virus Res. 2013, 177, 55–61. [Google Scholar] [CrossRef][Green Version]
- Yang, X.; Wang, X.; Song, Y.; Zhou, P.; Li, D.; Zhang, C.; Jin, X.; Huang, Z.; Zhou, D. Chimpanzee adenoviral vector prime-boost regimen elicits potent immune responses against Ebola virus in mice and rhesus macaques. Emerg. Microbes Infect. 2019, 8, 1086–1097. [Google Scholar] [CrossRef]
- Reyes-Sandoval, A.; Berthoud, T.; Alder, N.; Siani, L.; Gilbert, S.C.; Nicosia, A.; Colloca, S.; Cortese, R.; Hill, A.V. Prime-boost immunization with adenoviral and modified vaccinia virus Ankara vectors enhances the durability and polyfunctionality of protective malaria CD8+ T-cell responses. Infect. Immun. 2010, 78, 145–153. [Google Scholar] [CrossRef]
- Rollier, C.S.; Hill, A.V.S.; Reyes-Sandoval, A. Influence of adenovirus and MVA vaccines on the breadth and hierarchy of T cell responses. Vaccine 2016, 34, 4470–4474. [Google Scholar] [CrossRef]
- Vierboom, M.P.M.; Chenine, A.L.; Darrah, P.A.; Vervenne, R.A.W.; Boot, C.; Hofman, S.O.; Sombroek, C.C.; Dijkman, K.; Khayum, M.A.; Stammes, M.A.; et al. Evaluation of heterologous prime-boost vaccination strategies using chimpanzee adenovirus and modified vaccinia virus for TB subunit vaccination in rhesus macaques. NPJ Vaccines 2020, 5, 39. [Google Scholar] [CrossRef] [PubMed]
- Lin, S.C.; Liu, W.C.; Lin, Y.F.; Huang, Y.H.; Liu, J.H.; Wu, S.C. Heterologous prime-boost immunization regimens using adenovirus vector and virus-like particles induce broadly neutralizing antibodies against H5N1 avian influenza viruses. Biotechnol. J. 2013, 8, 1315–1322. [Google Scholar] [CrossRef] [PubMed]
- Lambe, T.; Carey, J.B.; Li, Y.; Spencer, A.J.; van Laarhoven, A.; Mullarkey, C.E.; Vrdoljak, A.; Moore, A.C.; Gilbert, S.C. Immunity against heterosubtypic influenza virus induced by adenovirus and MVA expressing nucleoprotein and matrix protein-1. Sci. Rep. 2013, 3, 1443. [Google Scholar] [CrossRef] [PubMed]
- Boyd, A.C.; Ruiz-Hernandez, R.; Peroval, M.Y.; Carson, C.; Balkissoon, D.; Staines, K.; Turner, A.V.; Hill, A.V.; Gilbert, S.C.; Butter, C. Towards a universal vaccine for avian influenza: Protective efficacy of modified Vaccinia virus Ankara and Adenovirus vaccines expressing conserved influenza antigens in chickens challenged with low pathogenic avian influenza virus. Vaccine 2013, 31, 670–675. [Google Scholar] [CrossRef]
- Lo, C.; Wu, Z.; Misplon, J.; Price, G.; Pappas, C.; Kong, W.; Tumpey, T.; Epstein, S. Comparison of vaccines for induction of heterosubtypic immunity to influenza A virus: Cold-adapted vaccine versus DNA prime-adenovirus boost strategies. Vaccine 2008, 26, 2062–2072. [Google Scholar] [CrossRef] [PubMed]
- Sedegah, M.; Hollingdale, M.R.; Farooq, F.; Ganeshan, H.; Belmonte, M.; Kim, Y.; Peters, B.; Sette, A.; Huang, J.; McGrath, S.; et al. Sterile immunity to malaria after DNA prime/adenovirus boost immunization is associated with effector memory CD8+T cells targeting AMA1 class I epitopes. PLoS ONE 2014, 9, e106241. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Yang, D.; Xu, B.; Liang, W.; Sui, J.; Chen, Y.; Yang, H.; Chen, H.; Wei, P.; Qiao, C. Immune efficacy of an adenoviral vector-based swine influenza vaccine against antigenically distinct H1N1 strains in mice. Antivir. Res. 2017, 147, 29–36. [Google Scholar] [CrossRef] [PubMed]
- García, M.; Misplon, J.A.; Price, G.E.; Lo, C.Y.; Epstein, S.L. Age Dependence of Immunity Induced by a Candidate Universal Influenza Vaccine in Mice. PLoS ONE 2016, 11, e0153195. [Google Scholar] [CrossRef] [PubMed]
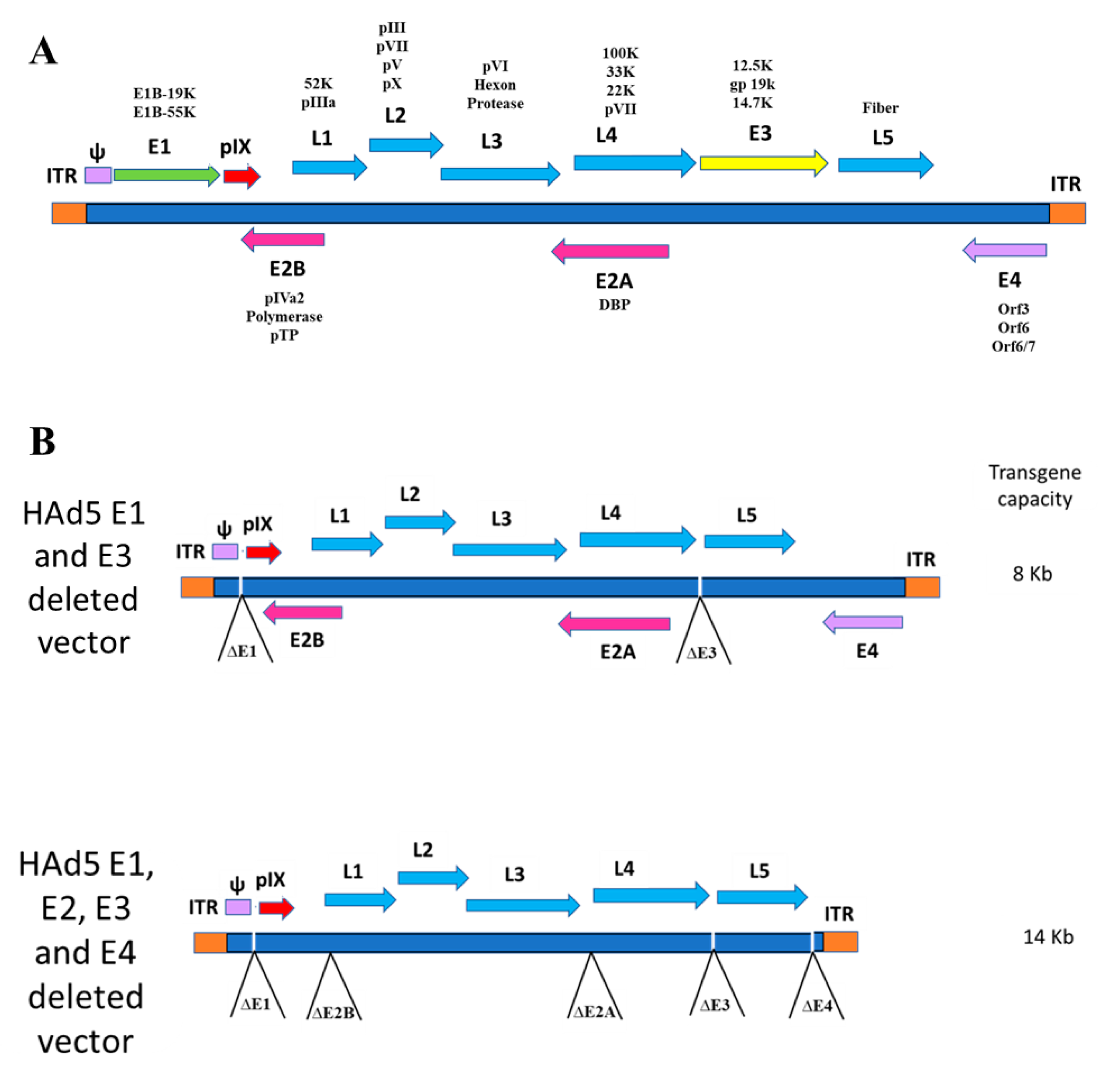
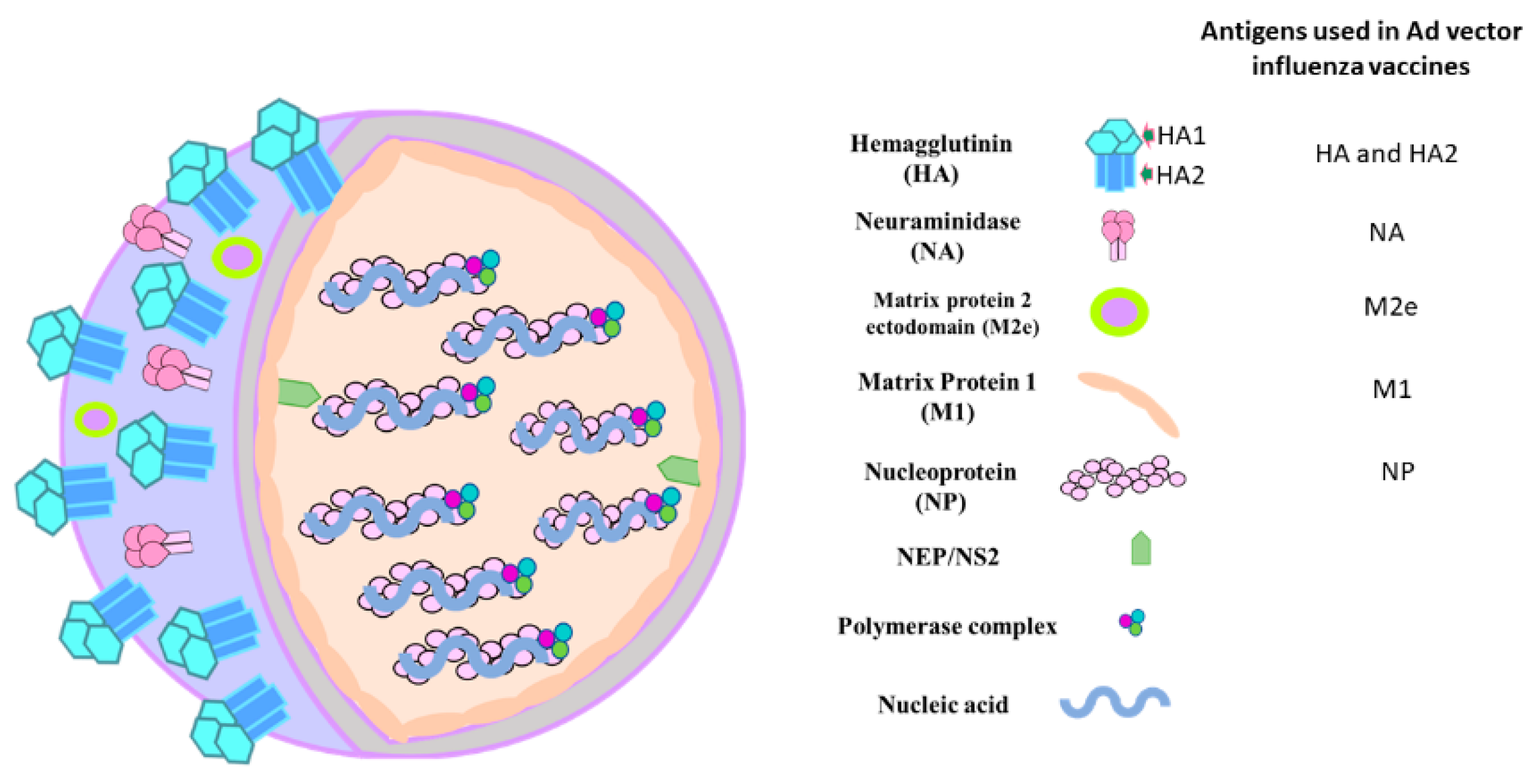
| Vector | Gene Insertion Site | Immunogen | Dose | Route of Inoculation | Immune RESPONSEEVALUATED | Host | Challenge Virus | Protection Level | References |
|---|---|---|---|---|---|---|---|---|---|
| HAd5 | E1 | HA [A/Swine/Iowa/1999(H3N2)] | 5× 108 TCID50 | IM | Humoral | Mice | [A/HK/1/1968(H3N2)] | Partial | [69] |
| HAd5 | E1 | HA [A/Hong Kong/156/1997(H5N1)] | 1 × 108 PFU | IM | Humoral, CMI | Mice | A/HK/483/1997 (H5N1) A/VN/1203/200 (H5N1) A/HK/213/2003 (H5N1) | Complete | [71] |
| HAd5 | E1 | HA [VN/1203/2004(H5N1)] HA [HK/156/1997(H5N1)] | 5 × 1010 VP | IM, SC, IN | Humoral, CMI | Mice, Chicken | A/VN/1203/2004(H5N1) | Complete | [70] |
| HAd5 | E1 | HA [A/Ca/4/2009 (H1N1)] | 5 × 1010 VP | IM | Humoral, CMI | Mice | A/Ohio/7/2009(H1N1) | Complete | [72] |
| HAd5 | E1 | HA [A/HK/156/1997 (H5N1)] | Multiple | IM | Humoral, CMI | Mice | A/HK/483/1997(H5N1) | Complete | [73] |
| HAd5 | E1 | HA and NP [VN/1203/2004 (H5N1)] | 1 × 108 PFU | IM | Humoral, CMI | Mice | PR8 reassortant A/Indo/05/2005(H5N1) A/VN/1203/2004(H5N1) | Complete | [74] |
| HAd5 | E1 | NP [A/PR/8/1934(H1N1)] NP [B/Ann Arbor/1/1986) | 1 × 1010 VP | IM | Humoral, CMI | Mice | A/PR/8/1934(H1N1) A/HK/483/1997(H5N1) A/HK/156/1997(H5N1) | Partial | [76] |
| HAd5 | E1 | M2 consensus sequence | 1 × 1010 VP | IM | Humoral, CMI | Mice | A/PR/8/1934(H1N1) A/Thailand/SP-83/2004 (H5N1) | Complete | [77] |
| HAd5 | E1 | M2 consensus sequence NP [A/PR/8/193 (H1N1)] NP [B/Ann Arbor/1/1986) | 1 × 1010 VP | IM | CMI | Mice | A/PR/8/1934(H1N1)] | Complete | [150] |
| A/VN/1203/2004(H5N1) | Partial | ||||||||
| HAd5 | E1, E3, E4 | HA, NP, and M2 A/Thailand/1/KAN-1/2004 | 1 × 1010 VP | IM | Humoral | Mice Ferrets | A/VN/1203/2004(H5N1) | Complete | [82] |
| HAd5 | E1 | HA and NP A/Swine/Iowa/1999(H3N2) | 2× 1010 TCID50 | IM | Humoral | Pig | A/Swine/Iowa/1999(H3N2) | Complete | [85] |
| AdC7 | E1 | NP[A/PR/8/1934 (H1N1)] | 1 × 1011 VP | IM | CMI | Mice | A/VM/1203/2004(H5N1) A/HK/483/1997(H5N1) | Partial | [53] |
| BAd3 | E1 | HA [A/HK/156/1997 (H5N1)] | 1 × 108 PFU | IM | Humoral, CMI | Mice | [A/Hong Kong/483/1997(H5N1)] | Complete | [54] |
| BAd3 | E1 | HA [A/HK/156/1997 (H5N1)] | Multiple | IM, IN | Humoral, CMI | Mice | A/Vietnam/1203/2004(H5N1)-PR8/CDC-RG | Complete | [81] |
| Vector | Vector Deletions | Insert | Phase | Route | Clinical Trial Number | Sponsor |
|---|---|---|---|---|---|---|
| Human adenovirus type 4 (HAd4) | Partial E3 deletion | H5HA | I | Oral | NCT01006798 | PaxVax |
| I | Intranasal | NCT01806909 | PaxVax | |||
| I | Oral, tonsillar | NCT01443936 | PaxVax | |||
| Human adenovirus type 5 (HAd5) | E1 and E3 | H1HA+ dsRNA | II |
Oral + H1N1 challenge | NCT02918006 | VaxArt |
| I | Oral | NCT01688297 | VaxArt | |||
| I | Oral | NCT03121339 | VaxArt | |||
| I | Ileum radio-controlled capsule | NCT01761123 | VaxArt | |||
| H5HA+ dsRNA | I | Oral | NCT01335347 | VaxArt | ||
| H1HA | IIa | Intranasal | NCT03232567 | Altimmune | ||
| H5HA | I | Intranasal | NCT00755703 | Vaxin/Altimmune | ||
| Chimpanzee adenovirus (ChAd) + Modified Vaccinia Ankara (MVA) | E1 and E3 | NP+M1 of H3N2 | I | IM heterologous prime-boost | NCT01623518 | Jenner Institute |
| I | IM heterologous prime-boost | NCT01818362 | Jenner Institute |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sayedahmed, E.E.; Elkashif, A.; Alhashimi, M.; Sambhara, S.; Mittal, S.K. Adenoviral Vector-Based Vaccine Platforms for Developing the Next Generation of Influenza Vaccines. Vaccines 2020, 8, 574. https://doi.org/10.3390/vaccines8040574
Sayedahmed EE, Elkashif A, Alhashimi M, Sambhara S, Mittal SK. Adenoviral Vector-Based Vaccine Platforms for Developing the Next Generation of Influenza Vaccines. Vaccines. 2020; 8(4):574. https://doi.org/10.3390/vaccines8040574
Chicago/Turabian StyleSayedahmed, Ekramy E., Ahmed Elkashif, Marwa Alhashimi, Suryaprakash Sambhara, and Suresh K. Mittal. 2020. "Adenoviral Vector-Based Vaccine Platforms for Developing the Next Generation of Influenza Vaccines" Vaccines 8, no. 4: 574. https://doi.org/10.3390/vaccines8040574
APA StyleSayedahmed, E. E., Elkashif, A., Alhashimi, M., Sambhara, S., & Mittal, S. K. (2020). Adenoviral Vector-Based Vaccine Platforms for Developing the Next Generation of Influenza Vaccines. Vaccines, 8(4), 574. https://doi.org/10.3390/vaccines8040574







