The Role of Birds of Prey in West Nile Virus Epidemiology
Abstract
1. Introduction
2. Infection and Immunity
2.1. Infection in Birds of Prey
2.2. Immunity against WNV in Birds of Prey
3. WNV Transmission
3.1. Mosquito-Borne Transmission
3.2. Contact Transmission
3.3. Oral Transmission
4. Clinical Features and Gross Pathology
4.1. Non-Specific Clinical Signs
4.2. Neurologic and Ocular Clinical Signs
4.3. Gross Pathological Lesions
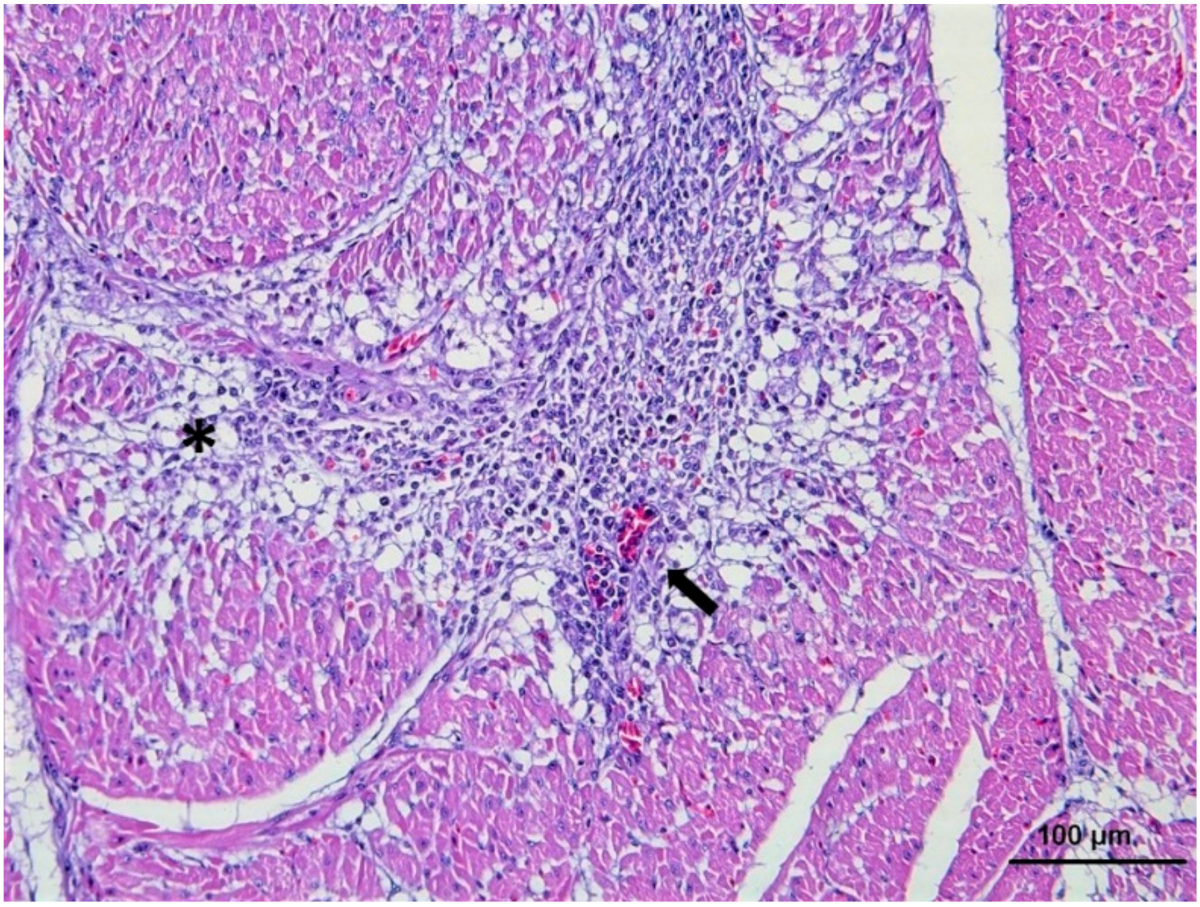
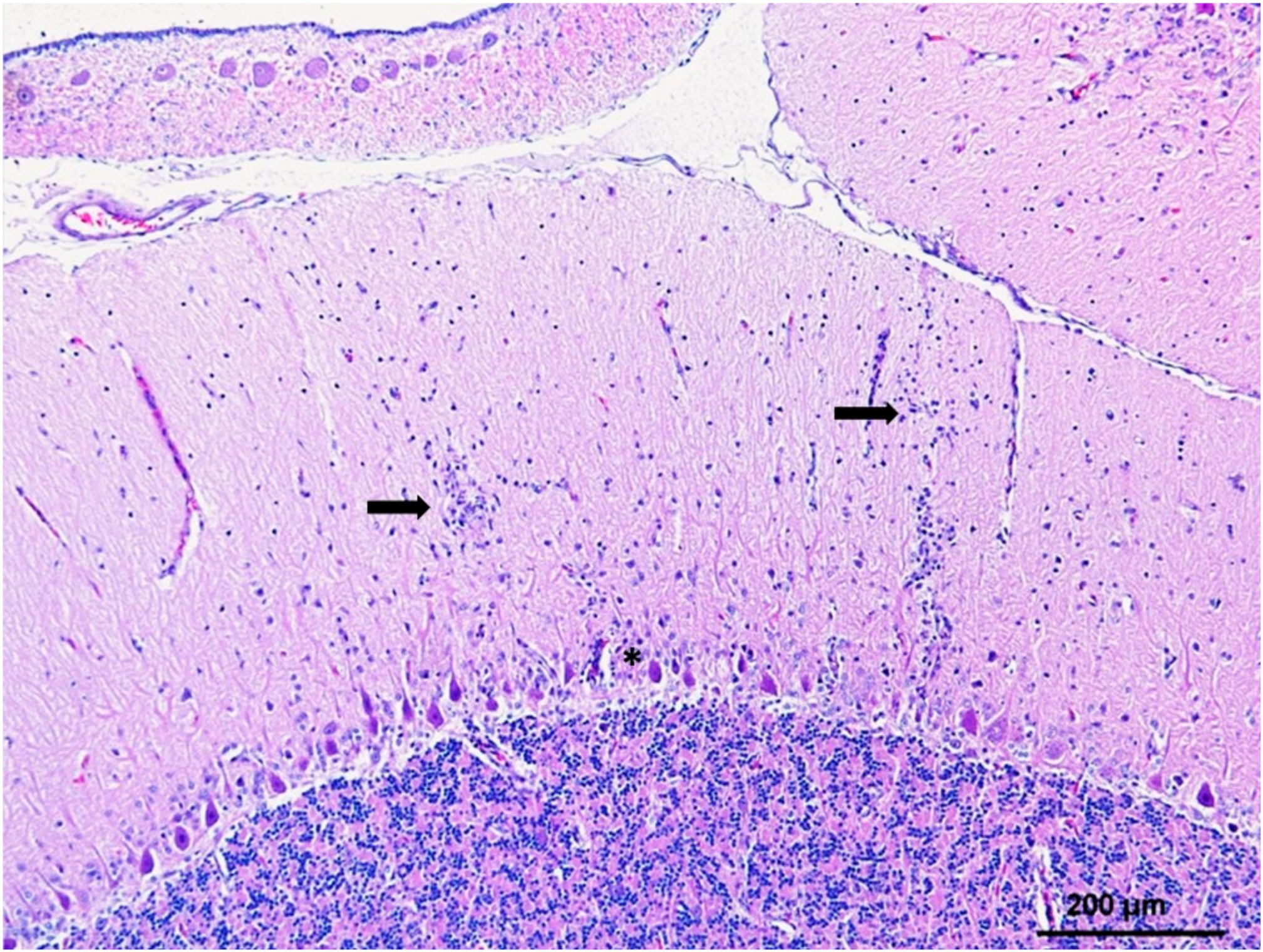
5. Histopathology and WNV Antigen Distribution
5.1. Heart, Skeletal Muscle and Kidney
5.2. CNS and Eye
5.3. Spleen, Liver and Lymphoid Organs
5.4. Lungs and Other Organs
6. Experimental WNV Infection
7. WNV Epidemiology
7.1. North America
7.2. Europe
8. Diagnosis and Surveillance
9. Vaccination against West Nile Virus in Avian Species
10. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Rizzoli, A.; Jimenez-Clavero, M.A.; Barzon, L.; Cordioli, P.; Figuerola, J.; Koraka, P.; Martina, B.; Moreno, A.; Nowotny, N.; Pardigon, N.; et al. The challenge of West Nile virus in Europe: Knowledge gaps and research priorities. Eurosurveillance 2015, 20, 21135. [Google Scholar] [CrossRef] [PubMed]
- Mackenzie, J.S.; Williams, D.T. The zoonotic flaviviruses of southern, south-eastern and eastern Asia, and Australasia: The potential for emergent viruses. Zoonoses Public Health 2009, 56, 338–356. [Google Scholar] [CrossRef] [PubMed]
- Perez-Ramirez, E.; Llorente, F.; Del Amo, J.; Fall, G.; Sall, A.A.; Lubisi, A.; Lecollinet, S.; Vazquez, A.; Jimenez-Clavero, M.A. Pathogenicity evaluation of twelve West Nile virus strains belonging to four lineages from five continents in a mouse model: Discrimination between three pathogenicity categories. J. Gen. Virol. 2017, 98, 662–670. [Google Scholar] [CrossRef]
- Ulbert, S. West Nile virus vaccines—Current situation and future directions. Hum. Vaccines Immunother. 2019, 15, 2337–2342. [Google Scholar] [CrossRef]
- Smithburn, K.C.; Hughes, T.P.; Burke, A.W.; Paul, J.H. A Neurotropic Virus Isolated from the Blood of a Native of Uganda. Am. J. Trop. Med. Hyg. 1940, 20, 471–472. [Google Scholar] [CrossRef]
- Fall, G.; Di Paola, N.; Faye, M.; Dia, M.; Freire, C.C.M.; Loucoubar, C.; Zanotto, P.M.A.; Faye, O.; Sall, A.A. Biological and phylogenetic characteristics of West African lineages of West Nile virus. PLoS Negl. Trop. Dis. 2017, 11, e0006078. [Google Scholar] [CrossRef]
- Murgue, B.; Murri, S.; Triki, H.; Deubel, V.; Zeller, H.G. West Nile in the Mediterranean basin: 1950–2000. Ann. N. Y. Acad. Sci. 2001, 951, 117–126. [Google Scholar] [CrossRef]
- May, F.J.; Davis, C.T.; Tesh, R.B.; Barrett, A.D. Phylogeography of West Nile virus: From the cradle of evolution in Africa to Eurasia, Australia, and the Americas. J. Virol. 2011, 85, 2964–2974. [Google Scholar] [CrossRef]
- Lanciotti, R.S.; Roehrig, J.T.; Deubel, V.; Smith, J.; Parker, M.; Steele, K.; Crise, B.; Volpe, K.E.; Crabtree, M.B.; Scherret, J.H.; et al. Origin of the West Nile virus responsible for an outbreak of encephalitis in the northeastern United States. Science 1999, 286, 2333–2337. [Google Scholar] [CrossRef]
- Hernandez-Triana, L.M.; Jeffries, C.L.; Mansfield, K.L.; Carnell, G.; Fooks, A.R.; Johnson, N. Emergence of west nile virus lineage 2 in europe: A review on the introduction and spread of a mosquito-borne disease. Front. Public Health 2014, 2, 271. [Google Scholar] [CrossRef]
- Burki, T. Increase of West Nile virus cases in Europe for 2018. Lancet 2018, 392, 1000. [Google Scholar] [CrossRef]
- ECDC West Nile virus in Europe in 2020—Human Cases. Available online: https://www.ecdc.europa.eu/en/publications-data/west-nile-virus-europe-2020-human-cases-updated-3-september-2020 (accessed on 8 September 2020).
- CDC West Nile Virus Disease Cases by State 2019, USA. Available online: https://www.cdc.gov/westnile/statsmaps/preliminarymapsdata2019/disease-cases-state-2019.html (accessed on 8 September 2020).
- ECDC Epidemiological Update: West Nile Virus Transmission Season in Europe. 2019. Available online: https://www.ecdc.europa.eu/en/west-nile-fever/surveillance-and-disease-data/disease-data-ecdc (accessed on 21 May 2020).
- Gossner, C.M.; Marrama, L.; Carson, M.; Allerberger, F.; Calistri, P.; Dilaveris, D.; Lecollinet, S.; Morgan, D.; Nowotny, N.; Paty, M.C.; et al. West Nile virus surveillance in Europe: Moving towards an integrated animal-human-vector approach. EuroSurveillance 2017, 22, 22. [Google Scholar] [CrossRef] [PubMed]
- Johnson, N.; Fernandez de Marco, M.; Giovannini, A.; Ippoliti, C.; Danzetta, M.L.; Svartz, G.; Erster, O.; Groschup, M.H.; Ziegler, U.; Mirazimi, A.; et al. Emerging Mosquito-Borne Threats and the Response from European and Eastern Mediterranean Countries. Int. J. Environ. Res. Public Health 2018, 15, 2775. [Google Scholar] [CrossRef] [PubMed]
- Ana, A.; Perez Andres, M.; Julia, P.; Pedro, P.; Arno, W.; Kimberly, V.W.; Julio, A.; Michelle, W. Syndromic surveillance for West Nile virus using raptors in rehabilitation. BMC Vet. Res. 2017, 13, 368. [Google Scholar] [CrossRef]
- Nemeth, N.M.; Kratz, G.E.; Bates, R.; Scherpelz, J.A.; Bowen, R.A.; Komar, N. Clinical evaluation and outcomes of naturally acquired West Nile virus infection in raptors. J. Zoo Wildl. Med. 2009, 40, 51–63. [Google Scholar] [CrossRef] [PubMed]
- Jimenez-Clavero, M.A.; Sotelo, E.; Fernandez-Pinero, J.; Llorente, F.; Blanco, J.M.; Rodriguez-Ramos, J.; Perez-Ramirez, E.; Hofle, U. West Nile virus in golden eagles, Spain, 2007. Emerg. Infect. Dis. 2008, 14, 1489–1491. [Google Scholar] [CrossRef]
- Ip, H.S.; Van Wettere, A.J.; McFarlane, L.; Shearn-Bochsler, V.; Dickson, S.L.; Baker, J.; Hatch, G.; Cavender, K.; Long, R.; Bodenstein, B. West nile virus transmission in winter: The 2013 great salt lake bald eagle and eared grebes mortality event. PLoS Curr. 2014, 6. [Google Scholar] [CrossRef]
- Banet-Noach, C.; Simanov, L.; Malkinson, M. Direct (non-vector) transmission of West Nile virus in geese. Avian Pathol. 2003, 32, 489–494. [Google Scholar] [CrossRef] [PubMed]
- Austin, R.J.; Whiting, T.L.; Anderson, R.A.; Drebot, M.A. An outbreak of West Nile virus-associated disease in domestic geese (Anser anser domesticus) upon initial introduction to a geographic region, with evidence of bird to bird transmission. Can. Vet. J. 2004, 45, 117–123. [Google Scholar] [PubMed]
- Glavits, R.; Ferenczi, E.; Ivanics, E.; Bakonyi, T.; Mato, T.; Zarka, P.; Palya, V. Co-occurrence of West Nile Fever and circovirus infection in a goose flock in Hungary. Avian Pathol. 2005, 34, 408–414. [Google Scholar] [CrossRef]
- Kritzik, K.L.; Kratz, G.; Panella, N.A.; Burkhalter, K.; Clark, R.J.; Biggerstaff, B.J.; Komar, N. Determining Raptor Species and Tissue Sensitivity for Improved West Nile Virus Surveillance. J. Wildl. Dis. 2018, 54, 528–533. [Google Scholar] [CrossRef] [PubMed]
- Hubalek, Z.; Tomesek, M.; Kosina, M.; Sikutova, S.; Strakova, P.; Rudolf, I. West Nile virus outbreak in captive and wild raptors, Czech Republic, 2018. Zoonoses Public Health 2019, 66, 978–981. [Google Scholar] [CrossRef] [PubMed]
- Zana, B.; Erdelyi, K.; Nagy, A.; Mezei, E.; Nagy, O.; Takacs, M.; Bakonyi, T.; Forgach, P.; Korbacska-Kutasi, O.; Feher, O.; et al. Multi-Approach Investigation Regarding the West Nile Virus Situation in Hungary, 2018. Viruses 2020, 12, 123. [Google Scholar] [CrossRef] [PubMed]
- Kolodziejek, J.; Jungbauer, C.; Aberle, S.W.; Allerberger, F.; Bago, Z.; Camp, J.V.; Dimmel, K.; de Heus, P.; Kolodziejek, M.; Schiefer, P.; et al. Integrated analysis of human-animal-vector surveillance: West Nile virus infections in Austria, 2015–2016. Emerg. Microbes Infect. 2018, 7, 25. [Google Scholar] [CrossRef] [PubMed]
- Feyer, S.; Bartenschlager, F.; Bertram, C.A.; Ziegler, U.; Fast, C.; Klopfleisch, R.; Muller, K. Clinical, pathological and virological aspects of fatal West Nile virus infections in ten free-ranging goshawks (Accipiter gentilis) in Germany. Transbound. Emerg. Dis. 2020. [Google Scholar] [CrossRef]
- Komar, N.; Langevin, S.; Hinten, S.; Nemeth, N.; Edwards, E.; Hettler, D.; Davis, B.; Bowen, R.; Bunning, M. Experimental infection of North American birds with the New York 1999 strain of West Nile virus. Emerg. Infect. Dis. 2003, 9, 311–322. [Google Scholar] [CrossRef]
- Gancz, A.Y.; Barker, I.K.; Lindsay, R.; Dibernardo, A.; McKeever, K.; Hunter, B. West Nile virus outbreak in North American owls, Ontario, 2002. Emerg. Infect. Dis. 2004, 10, 2135–2142. [Google Scholar] [CrossRef]
- Nemeth, N.; Gould, D.; Bowen, R.; Komar, N. Natural and experimental West Nile virus infection in five raptor species. J. Wildl. Dis. 2006, 42, 1–13. [Google Scholar] [CrossRef]
- Busquets, N.; Bertran, K.; Costa, T.P.; Rivas, R.; de la Fuente, J.G.; Villalba, R.; Solanes, D.; Bensaid, A.; Majo, N.; Pages, N. Experimental West Nile virus infection in Gyr-Saker hybrid falcons. Vector Borne Zoonotic Dis. 2012, 12, 482–489. [Google Scholar] [CrossRef]
- Ziegler, U.; Angenvoort, J.; Fischer, D.; Fast, C.; Eiden, M.; Rodriguez, A.V.; Revilla-Fernandez, S.; Nowotny, N.; de la Fuente, J.G.; Lierz, M.; et al. Pathogenesis of West Nile virus lineage 1 and 2 in experimentally infected large falcons. Vet. Microbiol. 2013, 161, 263–273. [Google Scholar] [CrossRef]
- Smith, K.A.; Campbell, G.D.; Pearl, D.L.; Jardine, C.M.; Salgado-Bierman, F.; Nemeth, N.M. A Retrospective Summary of Raptor Mortality in Ontario, Canada (1991–2014), Including the Effects of West Nile Virus. J. Wildl. Dis. 2018, 54, 261–271. [Google Scholar] [CrossRef] [PubMed]
- Ferraguti, M.; De La Puente, J.M.; Soriguer, R.; Llorente, F.; Jiménez-Clavero, M.Á.; Figuerola, J. West Nile virus-neutralizing antibodies in wild birds from southern Spain. Epidemiol. Infect. 2016, 144, 1907–1911. [Google Scholar] [CrossRef] [PubMed]
- Nemeth, N.M.; Oesterle, P.T.; Bowen, R.A. Humoral immunity to West Nile virus is long-lasting and protective in the house sparrow (Passer domesticus). Am. J. Trop. Med. Hyg. 2009, 80, 864–869. [Google Scholar] [CrossRef] [PubMed]
- Nemeth, N.M.; Kratz, G.E.; Bates, R.; Scherpelz, J.A.; Bowen, R.A.; Komar, N. Naturally induced humoral immunity to West Nile virus infection in raptors. Ecohealth 2008, 5, 298–304. [Google Scholar] [CrossRef]
- Gangoso, L.; Grande, J.M.; Llorente, F.; Jimenez-Clavero, M.A.; Perez, J.M.; Figuerola, J. Prevalence of neutralizing antibodies to West Nile virus in Eleonora’s Falcons in the Canary Islands. J. Wildl. Dis. 2010, 46, 1321–1324. [Google Scholar] [CrossRef]
- Michel, F.; Sieg, M.; Fischer, D.; Keller, M.; Eiden, M.; Reuschel, M.; Schmidt, V.; Schwehn, R.; Rinder, M.; Urbaniak, S.; et al. Evidence for West Nile Virus and Usutu Virus Infections in Wild and Resident Birds in Germany, 2017 and 2018. Viruses 2019, 11, 674. [Google Scholar] [CrossRef]
- Dube, M.C.; Bird, D.M.; Dibernardo, A.; Lindsay, L.R.; Charest, H. Prevalence of West Nile virus in wild American Kestrels (Falco sparverius) of southern Quebec, Canada. J. Wildl. Dis. 2010, 46, 603–607. [Google Scholar] [CrossRef]
- Quaglia, A.I.; Diaz, L.A.; Argibay, H.; Contigiani, M.S.; Saggese, M.D. West Nile and st. Louis encephalitis viruses antibodies surveillance in captive and free-ranging birds of prey from Argentina. Ecohealth 2014, 11, 603–609. [Google Scholar] [CrossRef]
- Hahn, D.C.; Nemeth, N.M.; Edwards, E.; Bright, P.R.; Komar, N. Passive West Nile virus antibody transfer from maternal Eastern screech-owls (Megascops asio) to progeny. Avian Dis. 2006, 50, 454–455. [Google Scholar] [CrossRef]
- Nemeth, N.M.; Oesterle, P.T.; Bowen, R.A. Passive immunity to West Nile virus provides limited protection in a common passerine species. Am. J. Trop. Med. Hyg. 2008, 79, 283–290. [Google Scholar] [CrossRef]
- Soltesz, Z.; Erdelyi, K.; Bakonyi, T.; Barna, M.; Szentpali-Gavaller, K.; Solt, S.; Horvath, E.; Palatitz, P.; Kotyman, L.; Dan, A.; et al. West Nile virus host-vector-pathogen interactions in a colonial raptor. Parasites Vectors 2017, 10, 449. [Google Scholar] [CrossRef] [PubMed]
- Saito, E.K.; Sileo, L.; Green, D.E.; Meteyer, C.U.; McLaughlin, G.S.; Converse, K.A.; Docherty, D.E. Raptor mortality due to West Nile virus in the United States, 2002. J. Wildl. Dis. 2007, 43, 206–213. [Google Scholar] [CrossRef] [PubMed]
- Bakonyi, T.; Ferenczi, E.; Erdelyi, K.; Kutasi, O.; Csorgo, T.; Seidel, B.; Weissenbock, H.; Brugger, K.; Ban, E.; Nowotny, N. Explosive spread of a neuroinvasive lineage 2 West Nile virus in Central Europe, 2008/2009. Vet. Microbiol. 2013, 165, 61–70. [Google Scholar] [CrossRef] [PubMed]
- Perez-Ramirez, E.; Llorente, F.; Jimenez-Clavero, M.A. Experimental infections of wild birds with West Nile virus. Viruses 2014, 6, 752–781. [Google Scholar] [CrossRef]
- Turell, M.J.; O’Guinn, M.; Oliver, J. Potential for New York mosquitoes to transmit West Nile virus. Am. J. Trop. Med. Hyg. 2000, 62, 413–414. [Google Scholar] [CrossRef]
- Nemeth, N.M.; Hahn, D.C.; Gould, D.H.; Bowen, R.A. Experimental West Nile virus infection in Eastern Screech Owls (Megascops asio). Avian Dis. 2006, 50, 252–258. [Google Scholar] [CrossRef]
- Holicki, C.M.; Ziegler, U.; Raileanu, C.; Kampen, H.; Werner, D.; Schulz, J.; Silaghi, C.; Groschup, M.H.; Vasic, A. West Nile Virus Lineage 2 Vector Competence of Indigenous Culex and Aedes Mosquitoes from Germany at Temperate Climate Conditions. Viruses 2020, 12, 561. [Google Scholar] [CrossRef]
- Lwande, O.W.; Lutomiah, J.; Obanda, V.; Gakuya, F.; Mutisya, J.; Mulwa, F.; Michuki, G.; Chepkorir, E.; Fischer, A.; Venter, M.; et al. Isolation of tick and mosquito-borne arboviruses from ticks sampled from livestock and wild animal hosts in Ijara District, Kenya. Vector Borne Zoonotic Dis. 2013, 13, 637–642. [Google Scholar] [CrossRef]
- Victoriano Llopis, I.; Tomassone, L.; Grego, E.; Serrano, E.; Mosca, A.; Vaschetti, G.; Andrade, D.; Rossi, L. Evaluating the feeding preferences of West Nile virus mosquito vectors using bird-baited traps. Parasites Vectors 2016, 9, 479. [Google Scholar] [CrossRef]
- Burkett-Cadena, N.D.; Bingham, A.M.; Porterfield, C.; Unnasch, T.R. Innate preference or opportunism: Mosquitoes feeding on birds of prey at the Southeastern Raptor Center. J. Vector Ecol. 2014, 39, 21–31. [Google Scholar] [CrossRef]
- Figuerola, J.; Jimenez-Clavero, M.A.; Lopez, G.; Rubio, C.; Soriguer, R.; Gomez-Tejedor, C.; Tenorio, A. Size matters: West Nile Virus neutralizing antibodies in resident and migratory birds in Spain. Vet. Microbiol. 2008, 132, 39–46. [Google Scholar] [CrossRef] [PubMed]
- Collins, C.T. Diurnal body temperature cycle in the northern Hawk-Owl (Surnia ulula). J. Raptor Res. 1991, 25, 92–93. [Google Scholar]
- Hinton, M.G.; Reisen, W.K.; Wheeler, S.S.; Townsend, A.K. West Nile Virus Activity in a Winter Roost of American Crows (Corvus brachyrhynchos): Is Bird-To-Bird Transmission Important in Persistence and Amplification? J. Med. Entomol. 2015, 52, 683–692. [Google Scholar] [CrossRef] [PubMed]
- Foss, L.; Padgett, K.; Reisen, W.K.; Kjemtrup, A.; Ogawa, J.; Kramer, V. West Nile Virus-Related Trends in Avian Mortality in California, USA, 2003–2012. J. Wildl. Dis. 2015, 51, 576–588. [Google Scholar] [CrossRef]
- Steinman, A.; Banet-Noach, C.; Tal, S.; Levi, O.; Simanov, L.; Perk, S.; Malkinson, M.; Shpigel, N. West Nile virus infection in crocodiles. Emerg. Infect. Dis. 2003, 9, 887–889. [Google Scholar] [CrossRef]
- Nemeth, N.M.; Beckett, S.; Edwards, E.; Klenk, K.; Komar, N. Avian mortality surveillance for West Nile virus in Colorado. Am. J. Trop. Med. Hyg. 2007, 76, 431–437. [Google Scholar] [CrossRef]
- Rimoldi, G.; Mete, A.; Adaska, J.M.; Anderson, M.L.; Symmes, K.P.; Diab, S. West Nile Virus Infection in Sheep. Vet. Pathol. 2017, 54, 155–158. [Google Scholar] [CrossRef]
- Garmendia, A.E.; Van Kruiningen, H.J.; French, R.A.; Anderson, J.F.; Andreadis, T.G.; Kumar, A.; West, A.B. Recovery and identification of West Nile virus from a hawk in winter. J. Clin. Microbiol. 2000, 38, 3110–3111. [Google Scholar] [CrossRef]
- Garcia-Salgado, G.; Rebollo, S.; Perez-Camacho, L.; Martinez-Hesterkamp, S.; Navarro, A.; Fernandez-Pereira, J.M. Evaluation of trail-cameras for analyzing the diet of nesting raptors using the Northern Goshawk as a model. PLoS ONE 2015, 10, e0127585. [Google Scholar] [CrossRef]
- Busquets, N.; Laranjo-Gonzalez, M.; Soler, M.; Nicolas, O.; Rivas, R.; Talavera, S.; Villalba, R.; San Miguel, E.; Torner, N.; Aranda, C.; et al. Detection of West Nile virus lineage 2 in North-Eastern Spain (Catalonia). Transbound. Emerg. Dis. 2019, 66, 617–621. [Google Scholar] [CrossRef]
- Napp, S.; Montalvo, T.; Pinol-Baena, C.; Gomez-Martin, M.B.; Nicolas-Francisco, O.; Soler, M.; Busquets, N. Usefulness of Eurasian Magpies (Pica pica) for West Nile virus Surveillance in Non-Endemic and Endemic Situations. Viruses 2019, 11, 716. [Google Scholar] [CrossRef] [PubMed]
- Dahlin, C.R.; Hughes, D.F.; Meshaka, W.E., Jr.; Coleman, C.; Henning, J.D. Wild snakes harbor West Nile virus. One Health 2016, 2, 136–138. [Google Scholar] [CrossRef] [PubMed]
- Steele, K.E.; Linn, M.J.; Schoepp, R.J.; Komar, N.; Geisbert, T.W.; Manduca, R.M.; Calle, P.P.; Raphael, B.L.; Clippinger, T.L.; Larsen, T.; et al. Pathology of fatal West Nile virus infections in native and exotic birds during the 1999 outbreak in New York City, New York. Vet. Pathol. 2000, 37, 208–224. [Google Scholar] [CrossRef]
- Lopes, H.; Redig, P.; Glaser, A.; Armien, A.; Wunschmann, A. Clinical findings, lesions, and viral antigen distribution in great gray owls (Strix nebulosa) and barred owls (Strix varia) with spontaneous West Nile virus infection. Avian Dis. 2007, 51, 140–145. [Google Scholar] [CrossRef]
- Joyner, P.H.; Kelly, S.; Shreve, A.A.; Snead, S.E.; Sleeman, J.M.; Pettit, D.A. West Nile virus in raptors from Virginia during 2003: Clinical, diagnostic, and epidemiologic findings. J. Wildl. Dis. 2006, 42, 335–344. [Google Scholar] [CrossRef][Green Version]
- Hofle, U.; Blanco, J.M.; Crespo, E.; Naranjo, V.; Jimenez-Clavero, M.A.; Sanchez, A.; de la Fuente, J.; Gortazar, C. West Nile virus in the endangered Spanish imperial eagle. Vet. Microbiol. 2008, 129, 171–178. [Google Scholar] [CrossRef] [PubMed]
- Erdelyi, K.; Ursu, K.; Ferenczi, E.; Szeredi, L.; Ratz, F.; Skare, J.; Bakonyi, T. Clinical and pathologic features of lineage 2 West Nile virus infections in birds of prey in Hungary. Vector Borne Zoonotic Dis. 2007, 7, 181–188. [Google Scholar] [CrossRef] [PubMed]
- Ziegler, U.; Luhken, R.; Keller, M.; Cadar, D.; van der Grinten, E.; Michel, F.; Albrecht, K.; Eiden, M.; Rinder, M.; Lachmann, L.; et al. West Nile virus epizootic in Germany, 2018. Antivir. Res. 2019, 162, 39–43. [Google Scholar] [CrossRef]
- Fitzgerald, S.D.; Patterson, J.S.; Kiupel, M.; Simmons, H.A.; Grimes, S.D.; Sarver, C.F.; Fulton, R.M.; Steficek, B.A.; Cooley, T.M.; Massey, J.P.; et al. Clinical and pathologic features of West Nile virus infection in native North American owls (Family strigidae). Avian Dis. 2003, 47, 602–610. [Google Scholar] [CrossRef]
- Ellis, A.E.; Mead, D.G.; Allison, A.B.; Stallknecht, D.E.; Howerth, E.W. Pathology and epidemiology of natural West Nile viral infection of raptors in Georgia. J. Wildl. Dis. 2007, 43, 214–223. [Google Scholar] [CrossRef]
- Gancz, A.Y.; Smith, D.A.; Barker, I.K.; Lindsay, R.; Hunter, B. Pathology and tissue distribution of West Nile virus in North American owls (family: Strigidae). Avian Pathol. 2006, 35, 17–29. [Google Scholar] [CrossRef] [PubMed]
- Wunschmann, A.; Timurkaan, N.; Armien, A.G.; Bueno Padilla, I.; Glaser, A.; Redig, P.T. Clinical, pathological, and immunohistochemical findings in bald eagles (Haliaeetus leucocephalus) and golden eagles (Aquila chrysaetos) naturally infected with West Nile virus. J. Vet. Diagn. Investig. 2014, 26, 599–609. [Google Scholar] [CrossRef] [PubMed]
- Harris, M.C.; Sleeman, J.M. Morbidity and mortality of bald eagles (Haliaeetus leucocephalus) and peregrine falcons (Falco peregrinus) admitted to the Wildlife Center of Virginia, 1993–2003. J. Zoo Wildl. Med. 2007, 38, 62–66. [Google Scholar] [CrossRef] [PubMed]
- Wunschmann, A.; Shivers, J.; Bender, J.; Carroll, L.; Fuller, S.; Saggese, M.; van Wettere, A.; Redig, P. Pathologic findings in red-tailed hawks (Buteo jamaicensis) and Cooper’s hawks (Accipiter cooper) naturally infected with West Nile virus. Avian Dis. 2004, 48, 570–580. [Google Scholar] [CrossRef]
- Pauli, A.M.; Cruz-Martinez, L.A.; Ponder, J.B.; Redig, P.T.; Glaser, A.L.; Klauss, G.; Schoster, J.V.; Wunschmann, A. Ophthalmologic and oculopathologic findings in red-tailed hawks and Cooper’s hawks with naturally acquired West Nile virus infection. J. Am. Vet. Med. Assoc. 2007, 231, 1240–1248. [Google Scholar] [CrossRef] [PubMed]
- Wodak, E.; Richter, S.; Bago, Z.; Revilla-Fernandez, S.; Weissenbock, H.; Nowotny, N.; Winter, P. Detection and molecular analysis of West Nile virus infections in birds of prey in the eastern part of Austria in 2008 and 2009. Vet. Microbiol. 2011, 149, 358–366. [Google Scholar] [CrossRef]
- Wunschmann, A.; Shivers, J.; Bender, J.; Carroll, L.; Fuller, S.; Saggese, M.; van Wettere, A.; Redig, P. Pathologic and immunohistochemical findings in goshawks (Accipiter gentilis) and great horned owls (Bubo virginianus) naturally infected with West Nile virus. Avian Dis. 2005, 49, 252–259. [Google Scholar] [CrossRef] [PubMed]
- Hubalek, Z.; Kosina, M.; Rudolf, I.; Mendel, J.; Strakova, P.; Tomesek, M. Mortality of Goshawks (Accipiter gentilis) Due to West Nile Virus Lineage 2. Vector Borne Zoonotic Dis. 2018, 18, 624–627. [Google Scholar] [CrossRef]
- Savini, G.; Puggioni, G.; Di Gennaro, A.; Di Francesco, G.; Rocchigiani, A.M.; Polci, A.; Marini, V.; Pinoni, C.; Rolesu, S.; Marruchella, G.; et al. West Nile virus lineage 2 in Sardinian wild birds in 2012: A further threat to public health. Epidemiol. Infect. 2013, 141, 2313–2316. [Google Scholar] [CrossRef]
- Wunschmann, A.; Armien, A.G.; Khatri, M.; Martinez, L.C.; Willette, M.; Glaser, A.; Alvarez, J.; Redig, P. Ocular Lesions in Red-Tailed Hawks ( Buteo jamaicensis) With Naturally Acquired West Nile Disease. Vet. Pathol. 2017, 54, 277–287. [Google Scholar] [CrossRef]
- Wojick, K.; Carpenter, J.W.; Johnson-Neitman, J.L.; Debey, B. What is your diagnosis? West Nile virus infection in a hawk. J. Avian Med. Surg. 2008, 22, 268–273. [Google Scholar] [PubMed]
- Angenvoort, J.; Fischer, D.; Fast, C.; Ziegler, U.; Eiden, M.; de la Fuente, J.G.; Lierz, M.; Groschup, M.H. Limited efficacy of West Nile virus vaccines in large falcons (Falco spp.). Vet. Res. 2014, 45, 41. [Google Scholar] [CrossRef] [PubMed]
- Lim, S.M.; van den Ham, H.J.; Oduber, M.; Martina, E.; Zaaraoui-Boutahar, F.; Roose, J.M.; van, I.W.F.J.; Osterhaus, A.; Andeweg, A.C.; Koraka, P.; et al. Transcriptomic Analyses Reveal Differential Gene Expression of Immune and Cell Death Pathways in the Brains of Mice Infected with West Nile Virus and Chikungunya Virus. Front. Microbiol. 2017, 8, 1556. [Google Scholar] [CrossRef] [PubMed]
- Guerrero-Sanchez, S.; Cuevas-Romero, S.; Nemeth, N.M.; Trujillo-Olivera, M.T.; Worwa, G.; Dupuis, A.; Brault, A.C.; Kramer, L.D.; Komar, N.; Estrada-Franco, J.G. West Nile virus infection of birds, Mexico. Emerg. Infect. Dis. 2011, 17, 2245–2252. [Google Scholar] [CrossRef] [PubMed]
- Gamino, V.; Hofle, U. Pathology and tissue tropism of natural West Nile virus infection in birds: A review. Vet. Res. 2013, 44, 39. [Google Scholar] [CrossRef]
- Work, T.H.; Hurlbut, H.S.; Taylor, R.M. Indigenous wild birds of the Nile Delta as potential West Nile virus circulating reservoirs. Am. J. Trop. Med. Hyg. 1955, 4, 872–888. [Google Scholar] [CrossRef]
- Partners in Flight (PIF) Population Estimates Database. Available online: http://pif.birdconservancy.org/PopEstimates (accessed on 15 April 2020).
- McDonough, K. Review of Rhe Web Site all about Birds, by CORnell Lab of Ornitology; CORnell Lab of Ornitology: Ithaca, NY, USA, 2013; p. 26. [Google Scholar]
- Hamer, G.L.; Kitron, U.D.; Goldberg, T.L.; Brawn, J.D.; Loss, S.R.; Ruiz, M.O.; Hayes, D.B.; Walker, E.D. Host selection by Culex pipiens mosquitoes and West Nile virus amplification. Am. J. Trop. Med. Hyg. 2009, 80, 268–278. [Google Scholar] [CrossRef]
- Edman, J.D.; Webber, L.A.; Schmid, A.A. Effect of host defenses on the feeding pattern of Culex nigripalpus when offered a choice of blood sources. J. Parasitol. 1974, 60, 874–883. [Google Scholar] [CrossRef]
- International, B. European Birds of Conservation Concrn: Populations, Trends and National Responsibilities. Available online: https://www.birdlife.org/sites/default/files/attachments/European%20Birds%20of%20Conservation%20Concern_Low.pdf (accessed on 15 April 2020).
- International, B. IUCN Red List for Birds. Available online: http://www.birdlife.org (accessed on 1 July 2020).
- The IUCN Red List of Threatened Species. Available online: https://www.iucnredlist.org (accessed on 25 April 2020).
- Bakonyi, T.; Ivanics, E.; Erdelyi, K.; Ursu, K.; Ferenczi, E.; Weissenbock, H.; Nowotny, N. Lineage 1 and 2 strains of encephalitic West Nile virus, central Europe. Emerg. Infect. Dis. 2006, 12, 618–623. [Google Scholar] [CrossRef]
- Csank, T.; Drzewniokova, P.; Korytar, L.; Major, P.; Gyuranecz, M.; Pistl, J.; Bakonyi, T. A Serosurvey of Flavivirus Infection in Horses and Birds in Slovakia. Vector Borne Zoonotic Dis. 2018, 18, 206–213. [Google Scholar] [CrossRef]
- Petrovic, T.; Blazquez, A.B.; Lupulovic, D.; Lazic, G.; Escribano-Romero, E.; Fabijan, D.; Kapetanov, M.; Lazic, S.; Saiz, J. Monitoring West Nile virus (WNV) infection in wild birds in Serbia during 2012: First isolation and characterisation of WNV strains from Serbia. EuroSurveillance 2013, 18, 20622. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Bocanegra, I.; Busquets, N.; Napp, S.; Alba, A.; Zorrilla, I.; Villalba, R.; Arenas, A. Serosurvey of West Nile virus and other flaviviruses of the Japanese encephalitis antigenic complex in birds from Andalusia, southern Spain. Vector Borne Zoonotic Dis. 2011, 11, 1107–1113. [Google Scholar] [CrossRef] [PubMed]
- Jurado-Tarifa, E.; Napp, S.; Lecollinet, S.; Arenas, A.; Beck, C.; Cerda-Cuellar, M.; Fernandez-Morente, M.; Garcia-Bocanegra, I. Monitoring of West Nile virus, Usutu virus and Meaban virus in waterfowl used as decoys and wild raptors in southern Spain. Comp. Immunol. Microbiol. Infect. Dis. 2016, 49, 58–64. [Google Scholar] [CrossRef]
- Alba, A.; Allepuz, A.; Napp, S.; Soler, M.; Selga, I.; Aranda, C.; Casal, J.; Pages, N.; Hayes, E.B.; Busquets, N. Ecological surveillance for West Nile in Catalonia (Spain), learning from a five-year period of follow-up. Zoonoses Public Health 2014, 61, 181–191. [Google Scholar] [CrossRef] [PubMed]
- Di Sabatino, D.; Bruno, R.; Sauro, F.; Danzetta, M.L.; Cito, F.; Iannetti, S.; Narcisi, V.; De Massis, F.; Calistri, P. Epidemiology of West Nile disease in Europe and in the Mediterranean Basin from 2009 to 2013. Biomed. Res. Int. 2014, 2014, 907852. [Google Scholar] [CrossRef]
- Balanca, G.; Gaidet, N.; Savini, G.; Vollot, B.; Foucart, A.; Reiter, P.; Boutonnier, A.; Lelli, R.; Monicat, F. Low West Nile virus circulation in wild birds in an area of recurring outbreaks in Southern France. Vector Borne Zoonotic Dis. 2009, 9, 737–741. [Google Scholar] [CrossRef]
- OIE. West Nile Fever. In Terrestral Manual; OIE: Paris, France, 2018. [Google Scholar]
- Nemeth, N.; Kratz, G.; Edwards, E.; Scherpelz, J.; Bowen, R.; Komar, N. Surveillance for West Nile virus in clinic-admitted raptors, Colorado. Emerg. Infect. Dis. 2007, 13, 305–307. [Google Scholar] [CrossRef]
- Komar, N.; Lanciotti, R.; Bowen, R.; Langevin, S.; Bunning, M. Detection of West Nile virus in oral and cloacal swabs collected from bird carcasses. Emerg. Infect. Dis. 2002, 8, 741–742. [Google Scholar] [CrossRef]
- Foss, L.; Reisen, W.K.; Fang, Y.; Kramer, V.; Padgett, K. Evaluation of Nucleic Acid Preservation Cards for West Nile Virus Testing in Dead Birds. PLoS ONE 2016, 11, e0157555. [Google Scholar] [CrossRef]
- Lecollinet, S.; Pronost, S.; Coulpier, M.; Beck, C.; Gonzalez, G.; Leblond, A.; Tritz, P. Viral Equine Encephalitis, a Growing Threat to the Horse Population in Europe? Viruses 2019, 12, 23. [Google Scholar] [CrossRef]
- Jimenez de Oya, N.; Escribano-Romero, E.; Blazquez, A.B.; Martin-Acebes, M.A.; Saiz, J.C. Current Progress of Avian Vaccines Against West Nile Virus. Vaccines 2019, 7, 126. [Google Scholar] [CrossRef] [PubMed]
- Samina, I.; Khinich, Y.; Simanov, M.; Malkinson, M. An inactivated West Nile virus vaccine for domestic geese-efficacy study and a summary of 4 years of field application. Vaccine 2005, 23, 4955–4958. [Google Scholar] [CrossRef] [PubMed]
- Jarvi, S.I.; Hu, D.; Misajon, K.; Coller, B.A.; Wong, T.; Lieberman, M.M. Vaccination of captive nene (Branta sandvicensis) against West Nile virus using a protein-based vaccine (WN-80E). J. Wildl. Dis. 2013, 49, 152–156. [Google Scholar] [CrossRef] [PubMed]
- Fischer, D.; Angenvoort, J.; Ziegler, U.; Fast, C.; Maier, K.; Chabierski, S.; Eiden, M.; Ulbert, S.; Groschup, M.H.; Lierz, M. DNA vaccines encoding the envelope protein of West Nile virus lineages 1 or 2 administered intramuscularly, via electroporation and with recombinant virus protein induce partial protection in large falcons (Falco spp.). Vet. Res. 2015, 46, 87. [Google Scholar] [CrossRef] [PubMed]
- USGS. Is there a West Nile Virus Vaccine Available for Birds? Available online: https://www.usgs.gov/faqs/there-a-west-nile-virus-vaccine-available-birds?qt-news_science_products=3#qt-news_science_products (accessed on 16 September 2020).
- Lustig, S.; Olshevsky, U.; Ben-Nathan, D.; Lachmi, B.E.; Malkinson, M.; Kobiler, D.; Halevy, M. A live attenuated West Nile virus strain as a potential veterinary vaccine. Viral. Immunol. 2000, 13, 401–410. [Google Scholar] [CrossRef] [PubMed]
- Johnson, S. Avian titer development against West nile virus after extralabel use of an equine vaccine. J. Zoo Wildl. Med. 2005, 36, 257–264. [Google Scholar] [CrossRef] [PubMed]
- Fassbinder-Orth, C.A.; Hofmeister, E.K.; Weeks-Levy, C.; Karasov, W.H. Oral and parenteral immunization of chickens (Gallus gallus) against West Nile virus with recombinant envelope protein. Avian Dis. 2009, 53, 502–509. [Google Scholar] [CrossRef]
- Chvala, S.; Kolodziejek, J.; Nowotny, N.; Weissenbock, H. Pathology and viral distribution in fatal Usutu virus infections of birds from the 2001 and 2002 outbreaks in Austria. J. Comp. Pathol. 2004, 131, 176–185. [Google Scholar] [CrossRef]
- Toyne, E.P. Breeding season diet of the Goshawk Accipiter gentilis in Wales. Ibis 1998, 140, 569–579. [Google Scholar] [CrossRef]
- Rutz, C. Breeding season diet of Northern Goshawks Accipiter gentilis in the city of Hamburg, Germany. Corax 2004, 19, 311–322. [Google Scholar]
- Rutz, C. Assessing the breeding season diet of goshawks Accipiter gentilis: Biases of plucking analysis quantified by means of continuous radio-monitoring. J. Zool. 2006, 259, 209–217. [Google Scholar] [CrossRef]
- Stout, W.E.; Temple, S.A.; Papp, J.M. Landscape Correlates of Reproductive Success for an Urban-Suburban Red-Tailed Hawk Population. J. Wildl. Manag. 2006, 70, 989–997. [Google Scholar] [CrossRef]
- Coleman, J.L.; Bird, D.M.; Jacobs, E.A. Habitat Use and Productivity of Sharp-Shinned Hawks Nesting in an Urban Area. Wilson Bull. 2002, 114, 467–473. [Google Scholar] [CrossRef]
- Swetnam, D.; Widen, S.G.; Wood, T.G.; Reyna, M.; Wilkerson, L.; Debboun, M.; Symonds, D.A.; Mead, D.G.; Beaty, B.J.; Guzman, H.; et al. Terrestrial Bird Migration and West Nile Virus Circulation, United States. Emerg. Infect. Dis. 2018, 24, 2184–2194. [Google Scholar] [CrossRef] [PubMed]
| Raptor Species | Clinical Signs | Gross Lesions d | Histological Lesions | IHC | References | |||
|---|---|---|---|---|---|---|---|---|
| Neurologic a | Ocular b | Non-Specific c | CNS | Others | ||||
| Bald eagle (Haliaeetus leucocephalus) | HT, TR, AC, HLR, MD, IN | NY, BL | EM, FA, RE, DP | Brain, eyes, heart, | Brain, spinal cord | Heart, liver, kidney | Brain, heart, spleen, kidney, endocrine, lung, eye, endothelium | [17,18,45,66,74,75,76] |
| Cooper’s hawk (Accipiter cooperii) | HT, TR, AC, MD, HLR, DY, SE, DE | NY, BL | EM, FA | Cachexia, spleen | Brain, spinal cord | Eye, heart, pancreas, pectoral muscle, liver, kidney | Kidney, heart, brain, spinal cord, eye, pancreas, lung, spleen, GIT | [17,18,45,73,77,78] |
| Northern goshawk (Accipiter gentilis) | TR, IN, UN, SE, AC, HLR, HT | BL | DH, EM | Liver, spleen, heart, brain | Brain, spinal cord | Eye, heart, liver, pectoral muscle, thyroid, pancreas, spleen, eye | Brain, spinal cord, spleen, liver, eye, heart, kidney, lung, muscle | [17,28,63,70,71,73,79,80,81,82] |
| Red-tailed hawk (Buteo jamaicensis) | AC, HT, TR, HLR, CO, SE, DY | APu, NY | EM, DH, RE, HY, GI, FA, | Heart, malacia | Brain, spinal cord | Heart, liver, kidney, pancreas, pectoral muscle, arteries | Brain, eye, Kidney, heart, brain, spinal cord, GIT, liver, lung, spleen, pectoral muscle | [17,18,31,45,73,77,78,83] |
| Sharped-shinned hawk (Accipiter striatus) | AC, HT, AP, DY | APu | Abs | ND | Brain | Heart | Heart, liver, lung, kidney, spleen, GIT, brain, pectoral muscle | [18,45,73] |
| Peregrine falcon (Falco peregrinus) | Abs | Abs | Abs | ND | Brain | ND | ND | [18,45,76] |
| American kestrel (Falco sparverius) | AC, MD, HT, DY, | APu | EM, DH. FA, | Heart | Brain | Arteries, heart, liver, kidney | ND | [18,31] |
| Great horned owl (Bubo virginianus) | HT, TR, AC, HLR, MD, SE, CO, IN, DY | APu, NY | RE, EM, FA, GI, HY, DP | PA, BH, SA, GIT, cachexia, heart, malacia | Brain, spinal cord | Heart, liver, kidney, spleen, arteries, eye, pancreas | Brain, heart, kidney, liver, spleen, GIT, endocrine | [17,18,31,45,72,73,74,80] |
| Eastern screech owl (Megascops asio) | Abs/ SE | ND | FA, RE, EM, DP | PA, heart | Brain | Heart, pectoral muscle, kidney, phlebitis | Heart, lung, kidney, spleen, GIT | [18,49,73] |
| Great grey owl (Strix nebulosa) | Abs | ND | Abs/FA, EM | Liver, spleen, heart, BH, eye | Brain, spinal cord, sciatic nerve | Liver, spleen, kidney, lung, heart, GIT | brain, heart, kidney, liver, lungs, spleen, GIT, endothelium, spinal cord, endocrine, gonad, sciatic nerve, eye, pectoral muscle, thymus | [67,71,74] |
| Barred owl (Strix varia) | TR, IN, HT, SE | Abs | EM, RE, DP | Cachexia, Absent | Brain | Heart, liver, spleen, kidney, eye | Heart, kidney, spleen, lung, brain, liver, GIT | [45,67,72,73] |
| Swainson’s hawk (Buteo swainsoni) | Abs | ND | EM, FA, GI, HY, DH, DP | ND | ND | ND | ND | [18] |
| Barn owl (Tyto alba) | Abs/ AC, HLR, MD | ND | EM, DH, FA, DP | ND | ND | Liver | ND | [18,31] |
| Ferruginous hawk (Buteo regalis) | HT, TR, MD, AC, HLR, MD | NY, APu | EM, CP, DV, FA, GI, HY, DP | ND | Brain | Heart, pectoral muscle | Heart | [18,84] |
| Golden eagle (Aquila chrysaetos) | AC, MD, HLR, TR, CO, SE, DYS | APu, NY | DH, RE, EM, FA, GI, DP | Absent | Brain | Liver, eye, heart, kidney | Brain, eye, heart, kidney | [18,19,25,31,75] |
| Gyr falcon (Falco rusticolus) and hybrid falcons | HT, AC, SE | ND | CP, FA, EM, DH, EC, RE | Spleen, heart | Brain | Heart, arteries | brain, heart, liver, kidney, pancreas, peripheral nerves, skeletal muscle | [25,33,79,85] |
| Sparrow hawk (Accipiter Nisus) | IN, TR, SE, AC, HT | BL | EM, DP | ND | ND | ND | ND | [25] |
| Harris’s hawk (Parabuteo unicinctus) | IN, TR, SE, AC, HT | BL | EM, DP | ND | ND | ND | ND | [25] |
| Snowy owl (Bubo scandiacus) | IN, HT, TR, SE | ND | RE, DP | Spleen | Brain, spinal cord, sciatic nerve | Heart, liver, kidney, spleen, GIT, pectoral muscle | Brain, heart, liver, kidney, intestine, pancreas, lung, gonad, spinal cord, GIT, spleen, skin, trachea, endocrine, pectoral muscle, thymus | [30,66,72,74] |
| Short-eared owl (Asio flammeus) | UN | ND | ND | ND | ND | heart | Heart, liver, kidney, lung | [72] |
| Boreal owl (Aegolius funereus) | ND | ND | ND | UN | Brain | Liver, spleen, kidney, heart, pectoral muscle | Heart, liver, lung, kidney, spleen, GIT, eye, pectoral muscle, thymus, gonad, endocrine, trachea, pancreas, spinal cord | [30,73,74] |
| Northern-hawk owl (Surnia ulula) | ND | ND | ND | UN | Brain, spinal cord, sciatic nerve | Liver, spleen, kidney, heart, pectoral muscle | Brain, spinal cord, liver, kidney, heart, pancreas, skin, trachea, GIT, endocrine, gonad, eye, thymus | [30,74] |
| Saw-whet owl (Aegolius acadicus) | ND | ND | ND | UN | Brain, spinal cord, sciatic nerve | Liver, spleen, kidney, heart, pectoral muscle | Brain, liver, spleen, kidney, lung, heart, pancreas, GIT, skin, endocrine, gonad, sciatic nerve, thymus | [30,74] |
| Bearded vulture (Gypaetus barbatus) | Abs/UN | ND | Abs | ND | ND | ND | ND | [63] |
| Spanish imperial eagle (Aquila adalberti) | ND | ND | FA | ND | Brain | Liver, intestine, heart | Brain, GIT, liver, kidney, lung | [69] |
| Bonelli’s eagle (Aquila fasciata) | IN | BL | DP | ND | ND | ND | ND | [19] |
| Prairie falcon (Falco mexicanus) | Abs | Abs | FA | ND | Brain | ND | ND | [18,45] |
| Red shouldered hawk (Buteo lineatus) | ND | ND | ND | ND | ND | ND | Heart, brain, GIT | [73] |
| Long-eared owl (Asio otus) Northern harrier (Circus cyaneus) Rough-ledged hawk (Buteo lagopus) | Abs | ND | EM, FA, GI, DP | ND | ND | ND | ND | [18] |
| Order | Family | Species | Tested Strain | Competent Host a | Mortality | Oral Infection b | Infection by Direct Contact c | Viral Shedding | Viral Detection in Organs | Viral Detection in Feathers | Reference | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Oral | Cloacal | |||||||||||
| Falconiformes | Falconidae | Falco sparverius | NY99 (L1) | YES | NO | YES(only in 1/8) | NO | YES | YES | YES (14 dpi) | NA | [29,31] |
| Falco rusticolus | NY99 (L1) | YES | 33% | NA | NA | YES | YES | YES (14 dpi) | NA | [33] | ||
| AUS09 (L2) | YES | 33% | NA | NA | YES | YES | YES (21 dpi) | NA | ||||
| Falco tinunculus | Ar-248 (L1) | NO | NO | NA | NA | NA | NA | NA | NA | [89] | ||
| F. rusticolus x F. cherrug | NY99 (L1) | NO | NO | NA | NA | YES | YES | YES (14 dpi) | YES | [32] | ||
| Accipitriformes | Accipitridae | Buteo jamaicensis | NY99 (L1) | YES | NO | YES | NO | YES | YES | YES (up to 27 dpi) | NA | [31] |
| Strigiformes | Strigidae | Bubo virginianus | NY99 (L1) | YES | NO | YES | NA | YES | YES | NO or at very low levels (14 dpi) | NA | [29,31] |
| Megascops asio | NY99 (L1) | YES | 40% | YES (only in 1/5) | NO | YES | YES | YES (14 dpi) | YES | [49] | ||
| Tytonidae | Tyto alba | NY99 (L1) | NO | NO | NA | NA | YES | NO | NO (15 dpi) | NA | [31] | |
| Order | Family | Species | Scientific Name | Captive vs. Wild | Positive/Tested a | Detection b | Location | Year | Reference |
|---|---|---|---|---|---|---|---|---|---|
| Accipitriformes | Accipitridae | Cooper’s hawk | Accipiter cooperii | Wild | 10/103 | IHC/virus isolation | US (Georgia) | 2001–2004 | [73] |
| Wild | 3/3 | ELISA + SNT | US (Colorado) | 2002–2005 | [18] | ||||
| Captive/Wild | 3/100 | RT-PCR | Canada (Ontario) | 2001–2014 | [34] | ||||
| Wild | 10/11 | IHC/RT-PCR | US (Minnesota) | 2001–2014 | [77] | ||||
| Northern goshawk | Accipiter gentilis | Wild | 1/1 | RT-PCR/virus isolation | US (Various) | 2002 | [45] | ||
| Captive/Wild | 3/23 | RT-PCR | Canada (Ontario) | 2001–2014 | [34] | ||||
| 9 Captive/3 Wild | 12/12 | IHC/ RT-PCR | US (Minnesota) | 2002–2003 | [80] | ||||
| Sharp-shinned hawk | Accipiter striatus | Wild | 8/40 | IHC/virus isolation | US (Georgia) | 2001–2004 | [73] | ||
| Wild | 1/1 | RT-PCR | US (Virginia) | 2003 | [68] | ||||
| Wild | 2/5 | ELISA + SNT | US (Colorado) | 2002–2005 | [18] | ||||
| Wild | 2/5 | RT-PCR/virus isolation | US (Various) | 2002 | [45] | ||||
| Captive/Wild | 10/91 | PCR | Canada (Ontario) | 2001–2014 | [34] | ||||
| Golden eagle | Aquila chrysaetos | Wild | 4/12 | ELISA + SNT | US (Colorado) | 2002–2005 | [18] | ||
| Captive/Wild | 3 | IHC/RT-PCR | US (Minnesota) | 2004–2013 | [75] | ||||
| Red-tailed hawk | Buteo jamaicensis | Wild | 10/56 | IHC/virus isolation | US (Georgia) | 2001–2004 | [73] | ||
| Wild | 1 | Virus isolation | US (New York) | 2000 | [61] | ||||
| Wild | 15/20 | RT-PCR | US (Virginia) | 2003 | [68] | ||||
| Wild | 18/33 | ELISA + SNT | US (Colorado) | 2002–2005 | [18] | ||||
| Wild | 10/20 | RT-PCR/virus isolation | US (Various) | 2002 | [45] | ||||
| Captive/Wild | 21/300 | RT-PCR | Canada (Ontario) | 2001–2014 | [34] | ||||
| Wild | 11/13 | IHC/RT-PCR | US (Minnesota) | 2002–2003 | [77] | ||||
| Roughhawk-legged | Buteo lagopus | Captive/Wild | 1/20 | RT-PCR | Canada (Ontario) | 2001–2014 | [34] | ||
| Red-shouldered hawk | Buteo lineatus | Wild | 2/43 | IHC/virus isolation | US (Georgia) | 2001–2004 | [73] | ||
| Broad wing hawk | Buteo platypterus | Wild | 2/2 | RT-PCR | US (Virginia) | 2003 | [68] | ||
| Ferruginous hawk | Buteo regalis | Wild | 2/2 | ELISA + SNT | US (Colorado) | 2002–2005 | [18] | ||
| Swainson’s hawk | Buteo swainsoni | Wild | 32/56 | ELISA + SNT | US (Colorado) | 2002–2005 | [18] | ||
| Bald eagle | Haliaeetus leucocephalus | Captive/Wild | 8/95 | RT-PCR | US (Virginia) | Up to 2003 | [76] | ||
| Wild | 9/9 | Virus isolation | US (Utah) | 2013 | [20] | ||||
| Wild | 2/2 | RT-PCR | US (Virginia) | 2003 | [68] | ||||
| Wild | 2/3 | RT-PCR/virus isolation | US (Various) | 2002 | [45] | ||||
| Captive/Wild | 4/80 | RT-PCR | Canada (Ontario) | 2001–2014 | [34] | ||||
| Captive/Wild | 15 | IHC/RT-PCR | US (Minnesota) | 2004–2013 | [75] | ||||
| Mississippi kite | Ictinia mississippiensis | Wild | 1/1 | ELISA + SNT | US (Colorado) | 2002–2005 | [18] | ||
| Pandionidae | Osprey | Pandion haliaetus | Wild | 1/5 | IHC/virus isolation | US (Georgia) | 2001–2004 | [73] | |
| Cathartiformes | Cathartidae | Black vulture | Coragyps atratus | Wild | 1/1 | RT-PCR | US (Virginia) | 2003 | [68] |
| Falconiformes | Falconidae | Merlin | Falco columbarius | Captive/Wild | 5/50 | RT-PCR | Canada (Ontario) | 2001–2014 | [34] |
| Prairie falcon | Falco mexicanus | Wild | 1/1 | RT-PCR/virus isolation | US (Various) | 2002 | [45] | ||
| Peregrine falcon | Falco peregrinus | Captive/Wild | 3/16 | RT-PCR | US (Virginia) | Up to 2004 | [76] | ||
| Wild | 1/2 | RT-PCR | US (Virginia) | 2003 | [68] | ||||
| Wild | 1/2 | ELISA + SNT | US (Colorado) | 2002–2005 | [18] | ||||
| American kestrel | Falco sparverius | Wild | 1/2 | RT-PCR | US (Virginia) | 2003 | [68] | ||
| Wild | 24/68 | ELISA + SNT | US (Colorado) | 2002–2005 | [18] | ||||
| Captive/Wild | 5/71 | RT-PCR | Canada (Ontario) | 2001–2021 | [34] | ||||
| Strigiformes | Strigidae | Northern saw-whet owl | Aegolius acadicus | Captive | 12/12 | RT-PCR | Canada (Ontario) | 2002 | [30] |
| Boreal owl | Aegolius funereus | Captive | 10/11 | RT-PCR | Canada (Ontario) | 2002 | [30] | ||
| Short-eared owl | Asio flammeus | Captive/Wild | 1 | IHC/ RT-PCR | US (Michigan/Ohio) | 2002 | [72] | ||
| Captive | 2/2 | RT-PCR | Canada (Ontario) | 2002 | [30] | ||||
| Long-eared owl | Asio otus | Captive | 3/3 | RT-PCR | Canada (Ontario) | 2002 | [30] | ||
| Snowy owl | Bubo scandiacus | Captive/Wild | 7 | IHC/ RT-PCR | US (Michigan/Ohio) | 2002 | [72] | ||
| Captive | 11/11 | RT-PCR | Canada (Ontario) | 2002 | [30] | ||||
| Great horned owl | Bubo virginianus | Wild | 1/18 | IHC/virus isolation | US (Georgia) | 2001–2004 | [73] | ||
| Captive/Wild | 4 | IHC/ RT-PCR | US (Michigan/Ohio) | 2002 | [72] | ||||
| Captive | 1/1 | RT-PCR | Canada (Ontario) | 2002 | [30] | ||||
| Wild | 16/22 | RT-PCR | US (Virginia) | 2003 | [68] | ||||
| Wild | 33/56 | ELISA + SNT | US (Colorado) | 2002–2005 | [18] | ||||
| Wild | 9/16 | RT-PCR/virus isolation | US (Various) | 2002 | [45] | ||||
| Captive/Wild | 18/225 | RT-PCR | Canada (Ontario) | 2001–2014 | [34] | ||||
| 1 Captive/24 Wild | 12/25 | IHC/RT-PCR | US (Minnesota) | 2002–2003 | [80] | ||||
| Northern pygmy owl | Glaucidium californicum | Captive | 1/1 | RT-PCR | Canada (Ontario) | 2002 | [30] | ||
| Eastern Screech Owl | Megascops asio | Wild | 4/42 | IHC/virus isolation | US (Georgia) | 2001–2004 | [73] | ||
| Wild | 4/6 | ELISA + SNT | US (Colorado) | 2002–2005 | [18] | ||||
| Captive/Wild | 1/100 | RT-PCR | Canada (Ontario) | 2001–2014 | [34] | ||||
| Flammulated owl | Otus flammeolus | Captive | 1/1 | RT-PCR | Canada (Ontario) | 2002 | [30] | ||
| Tawny owl | Strix aluco | Captive | 1/1 | RT-PCR | Canada (Ontario) | 2002 | [30] | ||
| Great gray owl | Strix nebulosa | Captive | 21/23 | RT-PCR | Canada (Ontario) | 2002 | [30] | ||
| Wild | 6 | Antigen detection | US (Minnesota) | 2003–2005 | [67] | ||||
| Captive/Wild | 4/27 | RT-PCR | Canada (Ontario) | 2001–2014 | [34] | ||||
| Spotted owl | Strix occidentalis | Captive | 1/1 | RT-PCR | Canada (Ontario) | 2002 | [30] | ||
| Barred owl | Strix varia | Wild | 4/27 | IHC/virus isolation | US (Georgia) | 2001–2004 | [73] | ||
| Captive/Wild | 1 | IHC/RT-PCR | US (Michigan/Ohio) | 2002 | [72] | ||||
| Wild | 2 | Antigen detection | US (Minnesota) | 2003–2005 | [67] | ||||
| Captive/Wild | 1/50 | RT-PCR | Canada (Ontario) | 2001–2014 | [34] | ||||
| Northern hawk owl | Surnia ulula | Captive | 17/17 | RT-PCR | Canada (Ontario) | 2002 | [30] | ||
| Tytonidae | Barn owl | Tyto alba | Wild | 1/3 | RT-PCR | US (Virginia) | 2003 | [68] |
| Order | Family | Species | Scientific Name | Captive vs. Wild | Positive/Tested a | Detection b | L c | Country | Year | Reference |
|---|---|---|---|---|---|---|---|---|---|---|
| Accipitriformes | Accipitridae | Northern goshawk | Accipiter gentilis | Wild | 4 | RT-PCR | 2 | Hungary | 2004–2005 | [97] |
| Wild | 35/57 | RT-PCR | 2 | Hungary/ Austria | 2008–2009 | [46] | ||||
| 2 Captive + 1 Wild | 3 | 2 RT-PCR 1ELISA | 2 | Spain | 2017 | [63] | ||||
| Captive + Wild | 3 | ELISA + SNT | Slovakia | 2012–2014 | [98] | |||||
| Captive + Wild | 5 | RT-PCR | 2 | Hungary | 2004 | [70] | ||||
| Captive | 2 | RT-PCR | 2 | Czech Republic | 2017 | [81] | ||||
| 12 Captive + 5 Wild | 17 | RT-PCR | 2 | Czech Republic | 2018 | [25] | ||||
| Captive * + Wild | 1 | ELISA + SNT | Germany | 2018 | [39] | |||||
| Captive + Wild | 3/3 | RT-PCR | 2 | Serbia | 2012 | [99] | ||||
| Wild | 1 | RT-PCR | 2 | Italy | 2012 | [82] | ||||
| Wild | 5/5 | RT-PCR | 2 | Austria | 2008 | [79] | ||||
| 1 Captive + 1 Wild | 2 | RT-PCR | 2 | Germany | 2018 | [71] | ||||
| Wild | 10 | RT-PCR + ELISA + IHC | Germany | 2019 | [28] | |||||
| Sparrowhawk | Accipiter nisus | Wild | 2 | RT-PCR | 2 | Hungary | 2005 | [97] | ||
| Wild | 1/12 | RT-PCR | 2 | Hungary | 2009 | [46] | ||||
| Eurasian sparrowhawk | Accipiter nisus | Captive | 1 | RT-PCR | 2 | Hungary | 2004 | [70] | ||
| Wild | 2 | RT-PCR | 2 | Czech Republic | 2018 | [81] | ||||
| Spanish imperial eagle | Aquila adalberti | Wild | 1 | ELISA + SNT | Spain | 2009 | [100] | |||
| 4 Captive + 4 Wild | 8 | RT-PCR | Spain | 2001–2005 | [69] | |||||
| Golden eagle | Aquila chrysaetos | Captive + Wild | 1 | ELISA + SNT | Slovakia | 2012-2014 | [98] | |||
| Captive | 1 | RT-PCR | 2 | Czech Republic | 2018 | [81] | ||||
| 1 Captive + 1 Wild | 2 | RT-PCR | 1 | Spain | 2007 | [19] | ||||
| Bonelli’s eagle | Aquila fasciata | Captive | 1 | RT-PCR | 1 | Spain | 2007 | [19] | ||
| Booted eagle | Aquila pennata | Captive | 1/5 | ELISA + SNT | Spain | 2011–2014 | [101] | |||
| Common buzzard | Buteo buteo | Captive + Wild | 1 | ELISA + SNT | Slovakia | 2012–2014 | [98] | |||
| Buteo buteo | Captive * + Wild | 7 | ELISA + SNT | Germany | 2017–2018 | [39] | ||||
| Short-toed snake eagle | Circaetus gallicus | Wild | 5/9 | ELISA + SNT | Spain | 2010–2011 | [102] | |||
| Circaetus gallicus | Captive | 3/5 | ELISA + SNT | Spain | 2011–2014 | [101] | ||||
| Western marsh-harrier | Circus aeruginosus | Captive + Wild | 1 | ELISA + SNT | Slovakia | 2012–2014 | [98] | |||
| Captive * + Wild | 2 | ELISA + SNT | Germany | 2018 | [39] | |||||
| Hen Harrier | Circus cyaneus | Wild | 1/1 | ELISA + SNT | Spain | 2010–2014 | [102] | |||
| Montagu’s harrier | Circus pygargus | Captive | 1/2 | ELISA + SNT | Spain | 2011–2014 | [101] | |||
| Bearded vulture | Gypaetus barbatus | Wild | 1/1 | RT-PCR | 2 | Austria | 2008 | [46] | ||
| Captive | 13 | ELISA + SNT | Spain | 2017 | [63] | |||||
| White-tailed Eagle | Haliaeetus albicilla | Captive * + Wild | 1 | ELISA + SNT | Germany | 2018 | [39] | |||
| Captive + Wild | 1/8 | ELISA + SNT | Serbia | 2012 | [99] | |||||
| Black Kite | Milvus migrans | Wild | 1/4 | ELISA + SNT | Spain | 2010–2012 | [102] | |||
| Captive * + Wild | 1 | ELISA + SNT | Germany | 2018 | [39] | |||||
| Red Kite | Milvus milvus | Wild | 1/7 | ELISA + SNT | Spain | 2010–2013 | [102] | |||
| Harris hawk | Parabuteo unicinctus | Captive | 2/2 | RT-PCR | 2 | Hungary | 2008 | [46] | ||
| Captive | 1 | RT-PCR | 2 | Czech Republic | 2018 | [81] | ||||
| Pandionidae | Osprey | Pandion haliaetus | Captive + Wild | 1 | ELISA + SNT | Slovakia | 2012–2014 | [98] | ||
| Falconiformes | Falconidae | Peregrine falcon | Falco peregrinus | Wild | 1/1 | RT-PCR | 2 | Austria | 2009 | [46] |
| Gyrfalcon | Falco rusticolus | Wild | 3/4 | RT-PCR | 2 | Hungary | 2008 | [46] | ||
| Wild | 1/1 | RT-PCR | 2 | Austria | 2008 | [79] | ||||
| European Kestrel | Falco tinnunculus | Captive * + Wild | 3 | ELISA + SNT | Germany | 2017–2018 | [39] | |||
| Strigiformes | Strigidae | Long-eared owl | Asio otus | Captive | 2/10 | ELISA + SNT | Spain | 2011–2014 | [101] | |
| Little owl | Athene noctua | Wild | 1 | RT-PCR | 1 | Italy | 2011 | [103] | ||
| Eurasian eagle-owl | Bubo bubo | Captive + Wild | 1 | ELISA + SNT | Slovakia | 2012–2014 | [98] | |||
| Captive | 1/24 | ELISA + SNT | Spain | 2011–2014 | [101] | |||||
| Snowy owl | Nyctea scandiaca | Wild | 1/1 | RT-PCR | 2 | Austria | 2009 | [46] | ||
| Scops owl | Otus scops | Wild | 2/6 | ELISA + SNT | France | 2005–2006 | [104] | |||
| European scops owl | Otus scops | Captive | 1/5 | ELISA + SNT | Spain | 2011–2014 | [101] | |||
| Great grey owl | Strix nebulosa | Captive | 2 | RT-PCR | 2 | Germany | 2018 | [71] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vidaña, B.; Busquets, N.; Napp, S.; Pérez-Ramírez, E.; Jiménez-Clavero, M.Á.; Johnson, N. The Role of Birds of Prey in West Nile Virus Epidemiology. Vaccines 2020, 8, 550. https://doi.org/10.3390/vaccines8030550
Vidaña B, Busquets N, Napp S, Pérez-Ramírez E, Jiménez-Clavero MÁ, Johnson N. The Role of Birds of Prey in West Nile Virus Epidemiology. Vaccines. 2020; 8(3):550. https://doi.org/10.3390/vaccines8030550
Chicago/Turabian StyleVidaña, Beatriz, Núria Busquets, Sebastian Napp, Elisa Pérez-Ramírez, Miguel Ángel Jiménez-Clavero, and Nicholas Johnson. 2020. "The Role of Birds of Prey in West Nile Virus Epidemiology" Vaccines 8, no. 3: 550. https://doi.org/10.3390/vaccines8030550
APA StyleVidaña, B., Busquets, N., Napp, S., Pérez-Ramírez, E., Jiménez-Clavero, M. Á., & Johnson, N. (2020). The Role of Birds of Prey in West Nile Virus Epidemiology. Vaccines, 8(3), 550. https://doi.org/10.3390/vaccines8030550







