Potent Protective Immune Responses to Senecavirus Induced by Virus-Like Particle Vaccine in Pigs
Abstract
1. Introduction
2. Results
2.1. Infectious Routes and Doses of SVA CH-HB-2017 Strain
2.2. Expression and Purification of SVA Capsid Proteins
2.3. Characterization and Quantification of SVA VLPs
2.4. Immunogenic Evaluation of SVA VLPs Vaccine in Pigs
2.5. SVA VLPs Vaccine-Induced INF-γ Response
2.6. Protection against SVA Challenge
3. Discussion
4. Materials and Methods
4.1. Viruses and Cells
4.2. Preparation and Characterization of SVA VLPs
4.3. Vaccine Preparation
4.4. Ethics Statement
4.5. Infection and Immunization of Pigs
4.6. Detection of Specific Antibodies, IgG1 and IgG2a
4.7. Neutralization Assay
4.8. Detection of Cytokines Interferon-γ
4.9. qRT-PCR
4.10. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Guo, B.; Piñeyro, P.E.; Rademacher, C.J.; Zheng, Y.; Li, G.; Yuan, J.; Hoang, H.; Gauger, P.C.; Madson, D.M.; Schwartz, K.J. Novel Senecavirus A in Swine with Vesicular Disease, United States, July 2015. Emerg. Infect. Dis. 2016, 22, 1325. [Google Scholar] [CrossRef] [PubMed]
- Pasma, T.; Davidson, S.; Shaw, S.L. Idiopathic vesicular disease in swine in Manitoba. Can. Vet. J. 2008, 49, 84–85. [Google Scholar]
- Zhang, J.; Pineyro, P.; Chen, Q.; Zheng, Y.; Li, G.; Rademacher, C.; Derscheid, R.; Guo, B.; Yoon, K.J.; Madson, D.; et al. Full-Length Genome Sequences of Senecavirus A from Recent Idiopathic Vesicular Disease Outbreaks in U.S. Swine. Genome Announc 2015, 3, e01270-15. [Google Scholar] [CrossRef]
- Leme, R.A.; Zotti, E.; Alcantara, B.K.; Oliveira, M.V.; Freitas, L.A.; Alfieri, A.F.; Alfieri, A.A. Senecavirus A: An Emerging Vesicular Infection in Brazilian Pig Herds. Transbound. Emerg. Dis. 2015, 62, 603–611. [Google Scholar] [CrossRef]
- Wu, Q.; Zhao, X.; Chen, Y.; He, X.; Zhang, G.; Ma, J. Complete Genome Sequence of Seneca Valley Virus CH-01-2015 Identified in China. Genome Announc. 2016, 4, e01509-15. [Google Scholar] [CrossRef]
- Saeng-Chuto, K.; Rodtian, P.; Temeeyasen, G.; Wegner, M.; Nilubol, D. The first detection of Senecavirus A in pigs in Thailand, 2016. Transbound. Emerg. Dis. 2018, 65, 285–288. [Google Scholar] [CrossRef] [PubMed]
- Sun, D.; Vannucci, F.; Knutson, T.P.; Corzo, C.; Marthaler, D.G. Emergence and whole-genome sequence of Senecavirus A in Colombia. Transbound. Emerg. Dis. 2017, 64, 1346–1349. [Google Scholar] [CrossRef] [PubMed]
- Arzt, J.; Bertram, M.R.; Vu, L.T.; Pauszek, S.J.; Hartwig, E.J.; Smoliga, G.R.; Palinski, R.; Stenfeldt, C.; Fish, I.H.; Hoang, B.H.; et al. First Detection and Genome Sequence of Senecavirus A in Vietnam. Microbiol. Resour. Announc. 2019, 8, e01247-18. [Google Scholar] [CrossRef]
- Segales, J.; Barcellos, D.; Alfieri, A.; Burrough, E.; Marthaler, D. Senecavirus A: An Emerging Pathogen Causing Vesicular Disease and Mortality in Pigs? Vet. Pathol. 2017, 54, 11–21. [Google Scholar] [CrossRef]
- Qian, S.; Fan, W.; Liu, T.; Wu, M.; Zhang, H.; Cui, X.; Zhou, Y.; Hu, J.; Wei, S.; Chen, H.; et al. Seneca Valley Virus Suppresses Host Type I Interferon Production by Targeting Adaptor Proteins MAVS, TRIF, and TANK for Cleavage. J. Virol. 2017, 91. [Google Scholar] [CrossRef]
- Strugnell, R.; Zepp, F.; Cunningham, A.; Tantawichien, T. Vaccine antigens. Perspect. Vaccinol. 2011, 1, 61–88. [Google Scholar] [CrossRef]
- Yang, F.; Zhu, Z.; Cao, W.; Liu, H.; Zhang, K.; Tian, H.; Liu, X.; Zheng, H. Immunogenicity and protective efficacy of an inactivated cell culture-derived Seneca Valley virus vaccine in pigs. Vaccine 2018, 36, 841–846. [Google Scholar] [CrossRef]
- Fuenmayor, J.; Godia, F.; Cervera, L. Production of virus-like particles for vaccines. New Biotechnol. 2017, 39, 174–180. [Google Scholar] [CrossRef]
- Mohsen, M.O.; Zha, L.; Cabral-Miranda, G.; Bachmann, M.F. Major findings and recent advances in virus-like particle (VLP)-based vaccines. Semin. Immunol. 2017, 34, 123–132. [Google Scholar] [CrossRef] [PubMed]
- Lua, L.H.; Connors, N.K.; Sainsbury, F.; Chuan, Y.P.; Wibowo, N.; Middelberg, A.P. Bioengineering virus-like particles as vaccines. Biotechnol. Bioeng. 2014, 111, 425–440. [Google Scholar] [CrossRef] [PubMed]
- Paavonen, J.; Jenkins, D.; Bosch, F.X.; Naud, P.; Salmeron, J.; Wheeler, C.M.; Chow, S.N.; Apter, D.L.; Kitchener, H.C.; Castellsague, X.; et al. Efficacy of a prophylactic adjuvanted bivalent L1 virus-like-particle vaccine against infection with human papillomavirus types 16 and 18 in young women: An interim analysis of a phase III double-blind, randomised controlled trial. Lancet 2007, 369, 2161–2170. [Google Scholar] [CrossRef]
- Liu, H.; Ma, Y.; Wang, H.; Liu, Q. Quantitative evaluation of the effect of the hepatitis B vaccine based on the HBsAg- and anti-HBs-positive rates in the Chinese population over the last 33 years. Vaccine 2012, 30, 3483–3487. [Google Scholar] [CrossRef] [PubMed]
- Michel, M.L.; Tiollais, P. Hepatitis B vaccines: Protective efficacy and therapeutic potential. Pathol. Biol. (Paris) 2010, 58, 288–295. [Google Scholar] [CrossRef]
- Noad, R.; Roy, P. Virus-like particles as immunogens. Trends. Microbiol. 2003, 11, 438–444. [Google Scholar] [CrossRef]
- Yin, S.; Sun, S.; Yang, S.; Shang, Y.; Cai, X.; Liu, X. Self-assembly of virus-like particles of porcine circovirus type 2 capsid protein expressed from Escherichia coli. Virol. J. 2010, 7, 166. [Google Scholar] [CrossRef]
- Guo, H.C.; Sun, S.Q.; Jin, Y.; Yang, S.L.; Wei, Y.Q.; Sun, D.H.; Yin, S.H.; Ma, J.W.; Liu, Z.X.; Guo, J.H.; et al. Foot-and-mouth disease virus-like particles produced by a SUMO fusion protein system in Escherichia coli induce potent protective immune responses in guinea pigs, swine and cattle. Vet. Res. 2013, 44, 48. [Google Scholar] [CrossRef] [PubMed]
- Guo, H.; Zhu, J.; Tan, Y.; Li, C.; Chen, Z.; Sun, S.; Liu, G. Self-assembly of virus-like particles of rabbit hemorrhagic disease virus capsid protein expressed in Escherichia coli and their immunogenicity in rabbits. Antivir. Res. 2016, 131, 85–91. [Google Scholar] [CrossRef] [PubMed]
- Xiao, Y.; Chen, H.Y.; Wang, Y.; Yin, B.; Lv, C.; Mo, X.; Yan, H.; Xuan, Y.; Huang, Y.; Pang, W.; et al. Large-scale production of foot-and-mouth disease virus (serotype Asia1) VLP vaccine in Escherichia coli and protection potency evaluation in cattle. BMC Biotechnol. 2016, 16, 56. [Google Scholar] [CrossRef] [PubMed]
- Liu, F.; Ge, S.; Li, L.; Wu, X.; Liu, Z.; Wang, Z. Virus-like particles: Potential veterinary vaccine immunogens. Res. Vet. Sci. 2012, 93, 553–559. [Google Scholar] [CrossRef]
- Strauss, M.; Jayawardena, N.; Sun, E.; Easingwood, R.; Burga, L.N.; Bostina, M. Cryo-EM Structure of Seneca Valley Virus Procapsid. J. Virol. 2017, 92, e01927-17. [Google Scholar] [CrossRef]
- Venkataraman, S.; Reddy, S.P.; Loo, J.; Idamakanti, N.; Hallenbeck, P.L.; Reddy, V.S. Crystallization and preliminary X-ray diffraction studies of Seneca Valley virus-001, a new member of the Picornaviridae family. Acta Crystallogr. Sect. F Struct. Biol. Cryst. Commun. 2008, 64, 293–296. [Google Scholar] [CrossRef]
- Hales, L.M.; Knowles, N.J.; Reddy, P.S.; Xu, L.; Hay, C.; Hallenbeck, P.L. Complete genome sequence analysis of Seneca Valley virus-001, a novel oncolytic picornavirus. J. Gen. Virol. 2008, 89, 1265–1275. [Google Scholar] [CrossRef]
- Maggioli, M.F.; Lawson, S.; de Lima, M.; Joshi, L.R.; Faccin, T.C.; Bauermann, F.V.; Diel, D.G. Adaptive Immune Responses following Senecavirus A Infection in Pigs. J. Virol. 2018, 92, e01717-17. [Google Scholar] [CrossRef]
- Sadr, V.; Saffar, B.; Emamzadeh, R. Functional expression and purification of recombinant Hepcidin25 production in Escherichia coli using SUMO fusion technology. Gene 2017, 610, 112–117. [Google Scholar] [CrossRef]
- Heinrich, J.; Drewniok, C.; Neugebauer, E.; Kellner, H.; Wiegert, T. The YoaW signal peptide directs efficient secretion of different heterologous proteins fused to a StrepII-SUMO tag in Bacillus subtilis. Microb. Cell Fact. 2019, 18, 31. [Google Scholar] [CrossRef]
- Butt, T.R.; Edavettal, S.C.; Hall, J.P.; Mattern, M.R. SUMO fusion technology for difficult-to-express proteins. Protein Expr. Purif. 2005, 43, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Marblestone, J.G.; Edavettal, S.C.; Lim, Y.; Lim, P.; Zuo, X.; Butt, T.R. Comparison of SUMO fusion technology with traditional gene fusion systems: Enhanced expression and solubility with SUMO. Protein. Sci. 2006, 15, 182–189. [Google Scholar] [CrossRef] [PubMed]
- Maggioli, M.F.; Fernandes, M.H.V.; Joshi, L.R.; Sharma, B.; Tweet, M.M.; Noll, J.C.G.; Bauermann, F.V.; Diel, D.G. Persistent infection and transmission of Senecavirus A from carrier sows to contact piglets. J. Virol. 2019, 93, e00819-19. [Google Scholar] [CrossRef] [PubMed]
- Joshi, L.R.; Fernandes, M.H.; Clement, T.; Lawson, S.; Pillatzki, A.; Resende, T.P.; Vannucci, F.A.; Kutish, G.F.; Nelson, E.A.; Diel, D.G. Pathogenesis of Senecavirus A infection in finishing pigs. J. Gen. Virol. 2016, 97, 3267–3279. [Google Scholar] [CrossRef]
- Xiaoran, G.; Shichong, H.; Xiaoli, Z.; Shiqi, S.; Xiaojun, M.; Huichen, G. Isolation, Identification and Biological Characteristics of the Chinese Strain of Senecavirus A. Chin. J. Virol. 2018, 34, 555–562. [Google Scholar]
- Oliveira, A.N.; Andrade, M.C.; Silva, M.V.; Moura, W.C.; Cortez Contreiras, E. Immune response in cattle vaccinated against rabies. Mem Inst. Oswaldo. Cruz. 2000, 95, 83–88. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Scodeller, E.A.; Lebendiker, M.A.; Dubra, M.S.; Crespo, O.A.; Basarab, O.; La Torre, J.L.; Vasquez, C. Inactivation of foot-and-mouth disease virus vaccine strains by activation of virus-associated endonuclease. J. Gen. Virol. 1984, 65, 1567–1573. [Google Scholar] [CrossRef] [PubMed]
- Mu, S.; Abdullah, S.W.; Zhang, Y.; Han, S.; Guo, H.; Li, M.; Dong, H.; Xu, J.; Teng, Z.; Wen, X.; et al. Development of a novel SYBR green I-based quantitative RT-PCR assay for Senecavirus A detection in clinical samples of pigs. Mol. Cell Probes 2020, 53, 101643. [Google Scholar] [CrossRef] [PubMed]
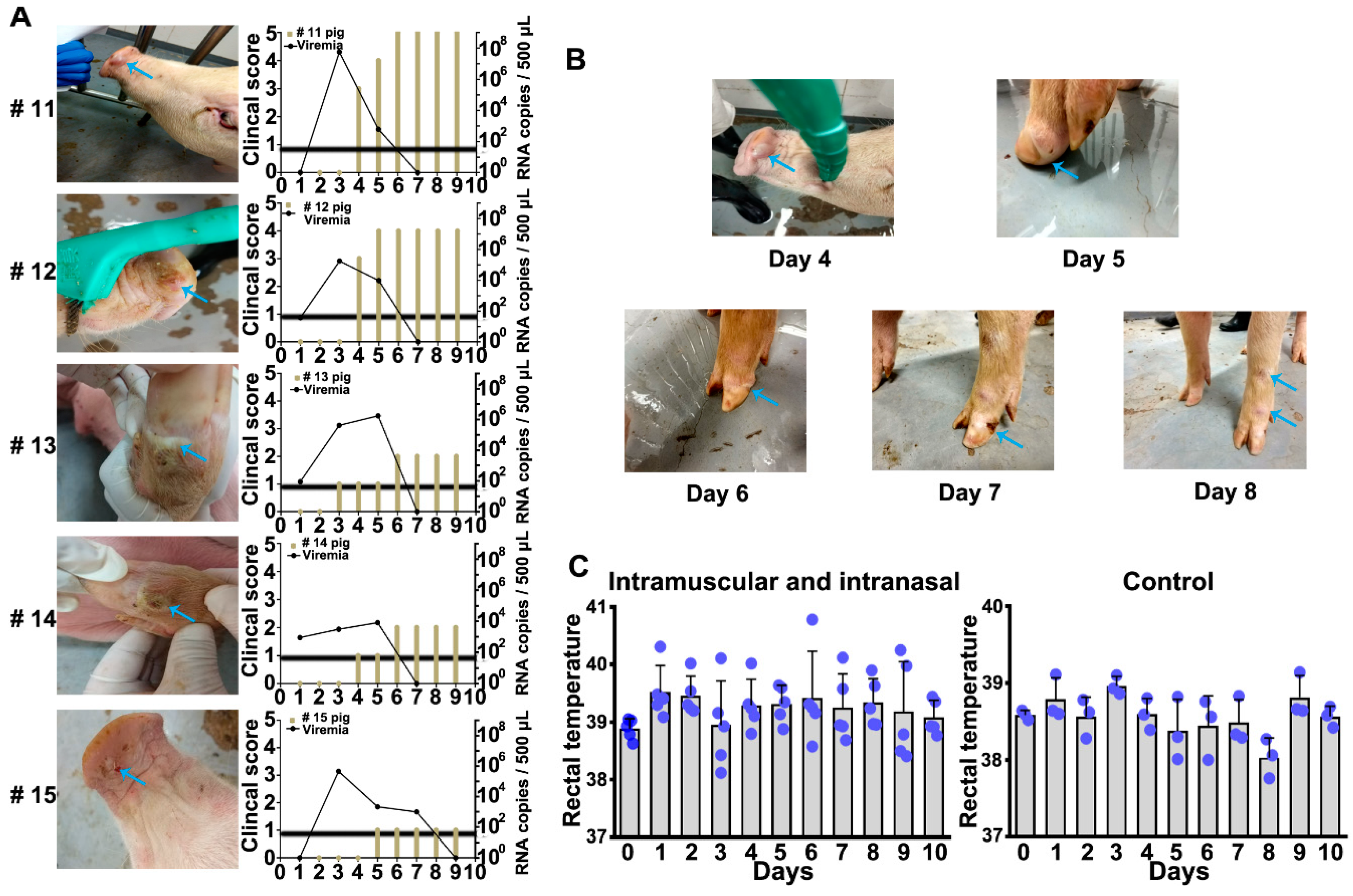

| Inoculation Method (Group) | Pig Number | Inoculation Dose (7 × 10−7.8 PFU/mL) | Infected (+/−) | Score | Incidence Rate (%) |
|---|---|---|---|---|---|
| Intramuscular andintranasal | 11 | 3 a mL+3 b mL | + | 5 | 100 |
| 12 | 3 a mL+3 b mL | + | 4 | ||
| 13 | 3 a mL+3 b mL | + | 2 | ||
| 14 | 3 a mL+3 b mL | + | 1 | ||
| 15 | 3 a mL+3 b mL | + | 2 | ||
| Intramuscular andintranasal | 16 | 1.5 a mL+1.5 b mL | + | 3 | 60 |
| 17 | 1.5 a mL+1.5 b mL | + | 2 | ||
| 18 | 1.5 a mL+1.5 b mL | + | 3 | ||
| 19 | 1.5 a mL+1.5 b mL | − | 0 | ||
| 20 | 1.5 a mL+1.5 b mL | − | 0 | ||
| Intramuscular | 21 | 6 mL | + | 2 | 80 |
| 22 | 6 mL | + | 3 | ||
| 23 | 6 mL | + | 2 | ||
| 24 | 6 mL | + | 1 | ||
| 25 | 6 mL | − | 0 | ||
| Ear vein | 31 | 3 mL | + | 2 | 80 |
| 32 | 3 mL | + | 1 | ||
| 33 | 3 mL | + | 1 | ||
| 34 | 3 mL | + | 1 | ||
| 35 | 3 mL | − | 0 | ||
| Control | 50 | None | − | 0 | 0 |
| 51 | None | − | 0 | ||
| 52 | None | − | 0 |
| Group | Pig Number | Infected (+/−) | Score | Viremia | Level of Protection |
|---|---|---|---|---|---|
| Inactivated vaccine | 1 | − | 0 | No | 100% |
| 2 | − | 0 | No | ||
| 3 | − | 0 | No | ||
| 4 | − | 0 | No | ||
| 5 | − | 0 | No | ||
| VLPs vaccine | 6 | − | 0 | No | 100% |
| 7 | − | 0 | No | ||
| 8 | − | 0 | No | ||
| 9 | − | 0 | No | ||
| 10 | − | 0 | No | ||
| Control | 11 | + | 3 | Yes | 0 |
| 12 | + | 2 | Yes | ||
| 13 | + | 2 | Yes | ||
| 14 | + | 2 | Yes | ||
| 15 | + | 2 | Yes |
| Clinical Symptoms | Clinical Score | Total Score |
|---|---|---|
| Left forefoot blisters | 1 | 5 |
| Right forefoot blisters | 1 | |
| Left hindfoot blisters | 1 | |
| Right hindfoot blisters | 1 | |
| Mouth and nose blisters | 1 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mu, S.; Sun, S.; Dong, H.; Bai, M.; Zhang, Y.; Teng, Z.; Ren, M.; Yin, S.; Guo, H. Potent Protective Immune Responses to Senecavirus Induced by Virus-Like Particle Vaccine in Pigs. Vaccines 2020, 8, 532. https://doi.org/10.3390/vaccines8030532
Mu S, Sun S, Dong H, Bai M, Zhang Y, Teng Z, Ren M, Yin S, Guo H. Potent Protective Immune Responses to Senecavirus Induced by Virus-Like Particle Vaccine in Pigs. Vaccines. 2020; 8(3):532. https://doi.org/10.3390/vaccines8030532
Chicago/Turabian StyleMu, Suyu, Shiqi Sun, Hu Dong, Manyuan Bai, Yun Zhang, Zhidong Teng, Mei Ren, Shuanghui Yin, and Huichen Guo. 2020. "Potent Protective Immune Responses to Senecavirus Induced by Virus-Like Particle Vaccine in Pigs" Vaccines 8, no. 3: 532. https://doi.org/10.3390/vaccines8030532
APA StyleMu, S., Sun, S., Dong, H., Bai, M., Zhang, Y., Teng, Z., Ren, M., Yin, S., & Guo, H. (2020). Potent Protective Immune Responses to Senecavirus Induced by Virus-Like Particle Vaccine in Pigs. Vaccines, 8(3), 532. https://doi.org/10.3390/vaccines8030532







