Evaluation of Chimpanzee Adenovirus and MVA Expressing TRAP and CSP from Plasmodium cynomolgi to Prevent Malaria Relapse in Nonhuman Primates
Abstract
1. Introduction
2. Materials and Methods
2.1. Antigen Design in ChAdOx1 and MVA Vaccines Expressing TRAP and the CSP from Plasmodium cynomolgi
2.2. Design, Production and Purification of PvCSP and PvTRAP Proteins in HEK293T Cells
2.3. Ethics Statement and Immunization in Mice
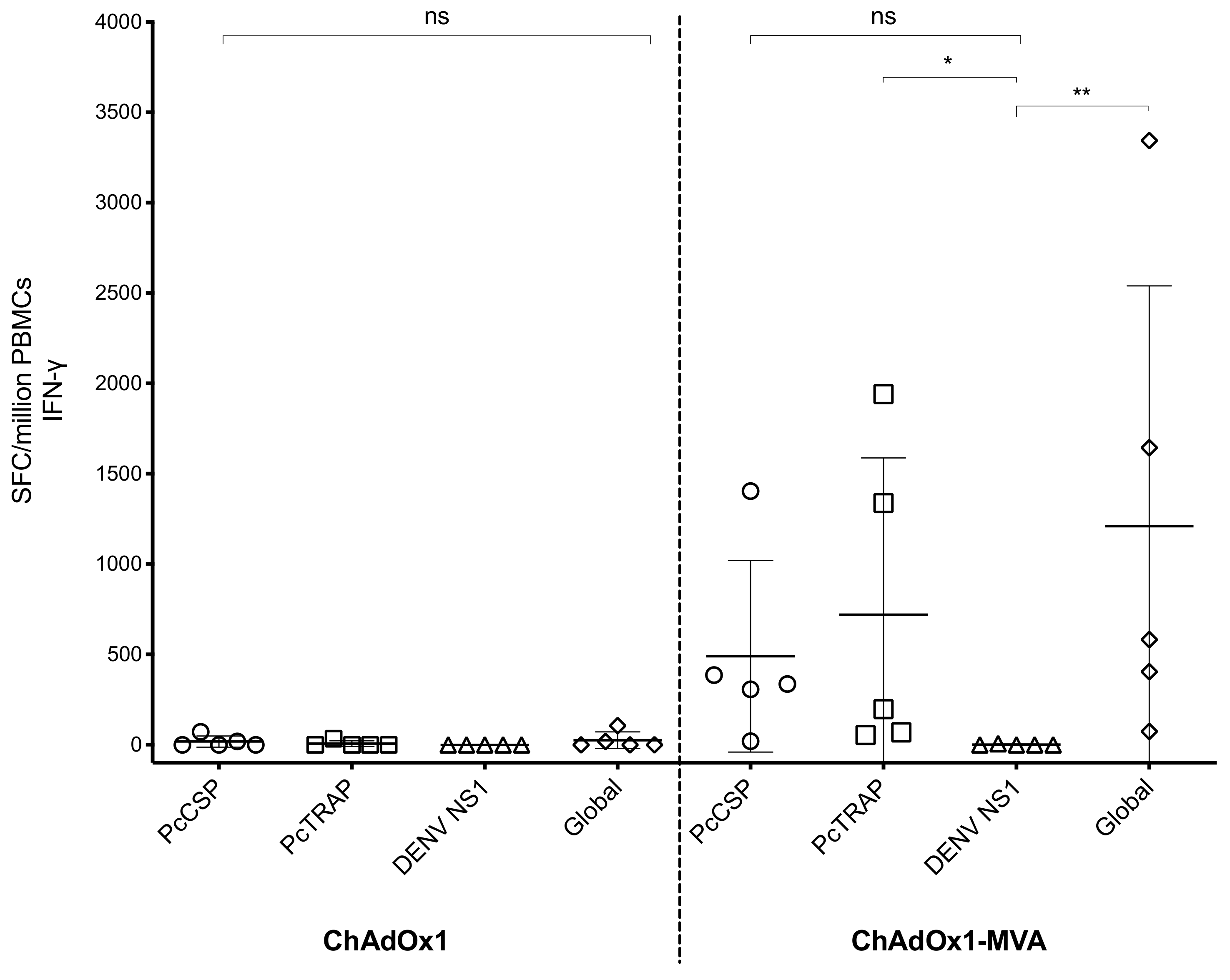
2.4. Ex-Vivo IFNγ ELISpot Assay
2.5. Enzyme-Linked Immunosorbent Assay
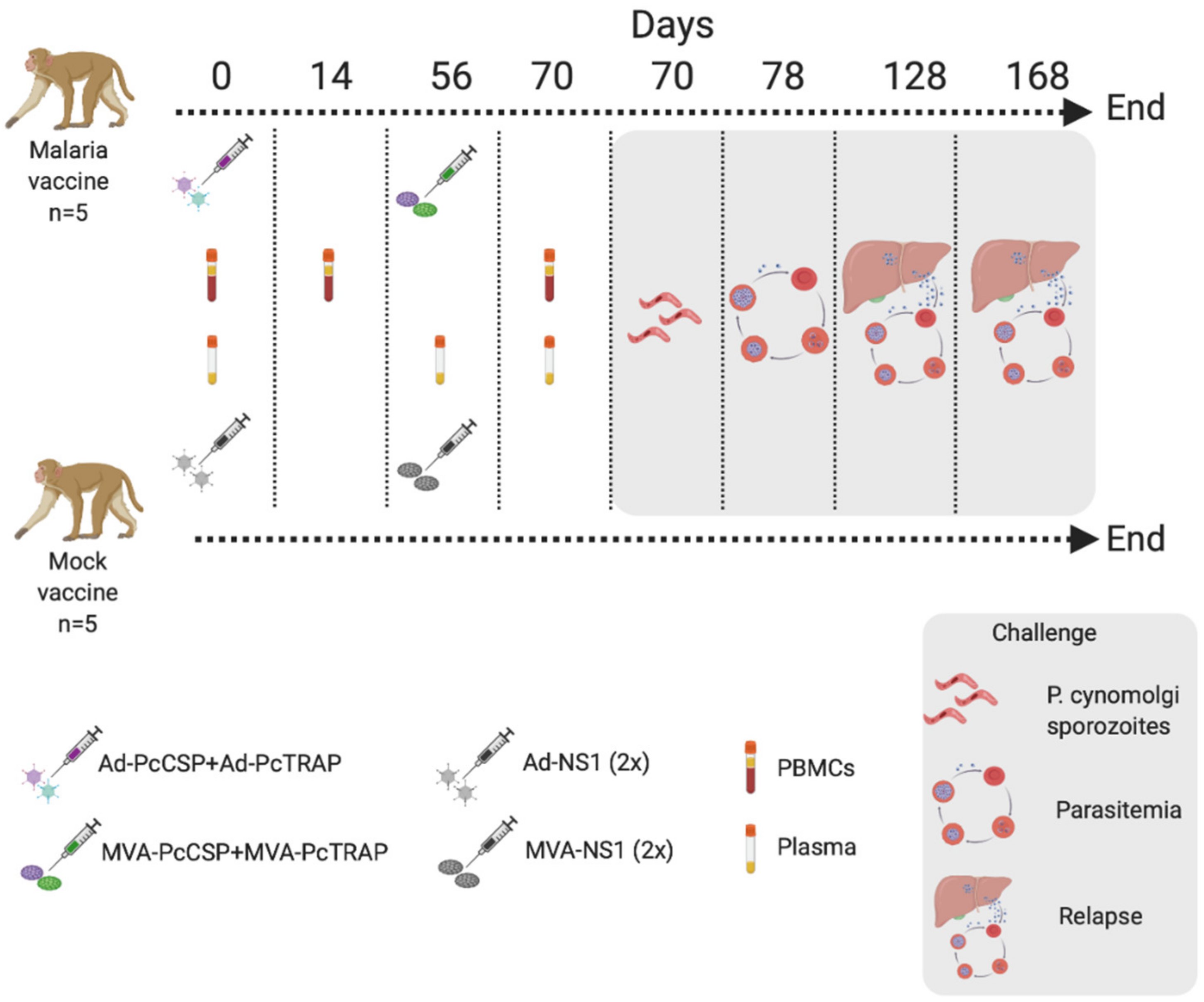
2.6. Immunogenicity Responses and Challenge in Rhesus Macaque
3. Results
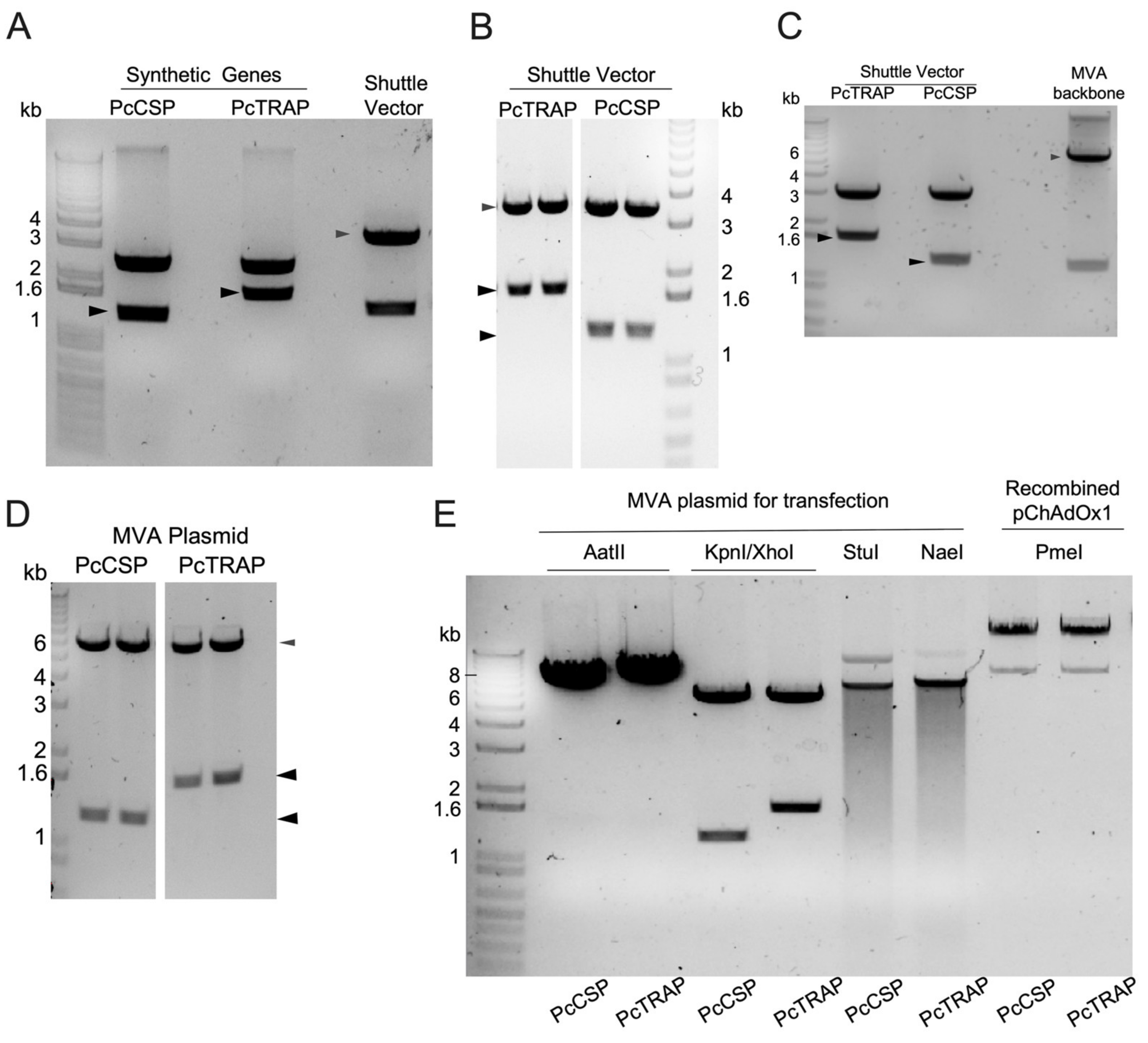
3.1. Transgene Design in ChAdOx1 and MVA Vaccines Expressing TRAP and the CSP
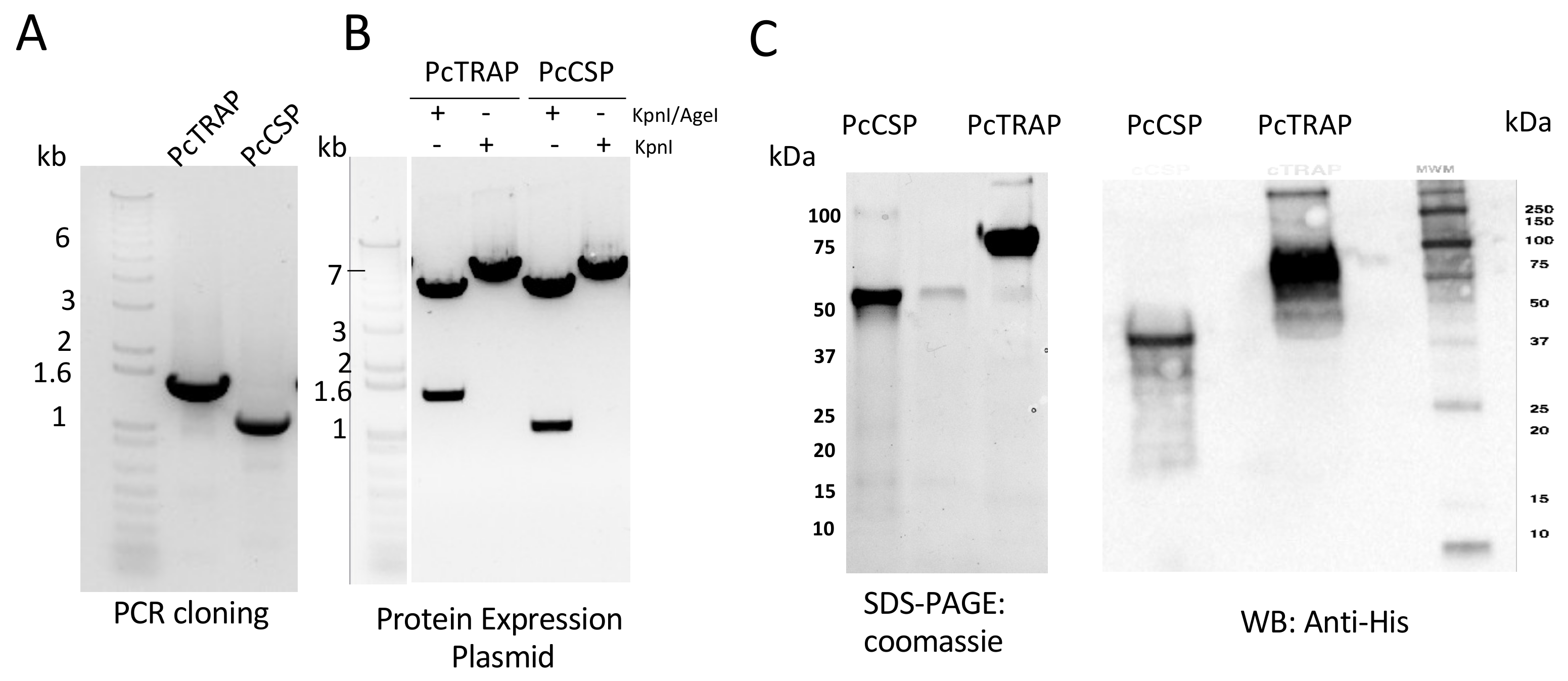
3.2. Design and Production of PcTRAP and the PcCSP Proteins
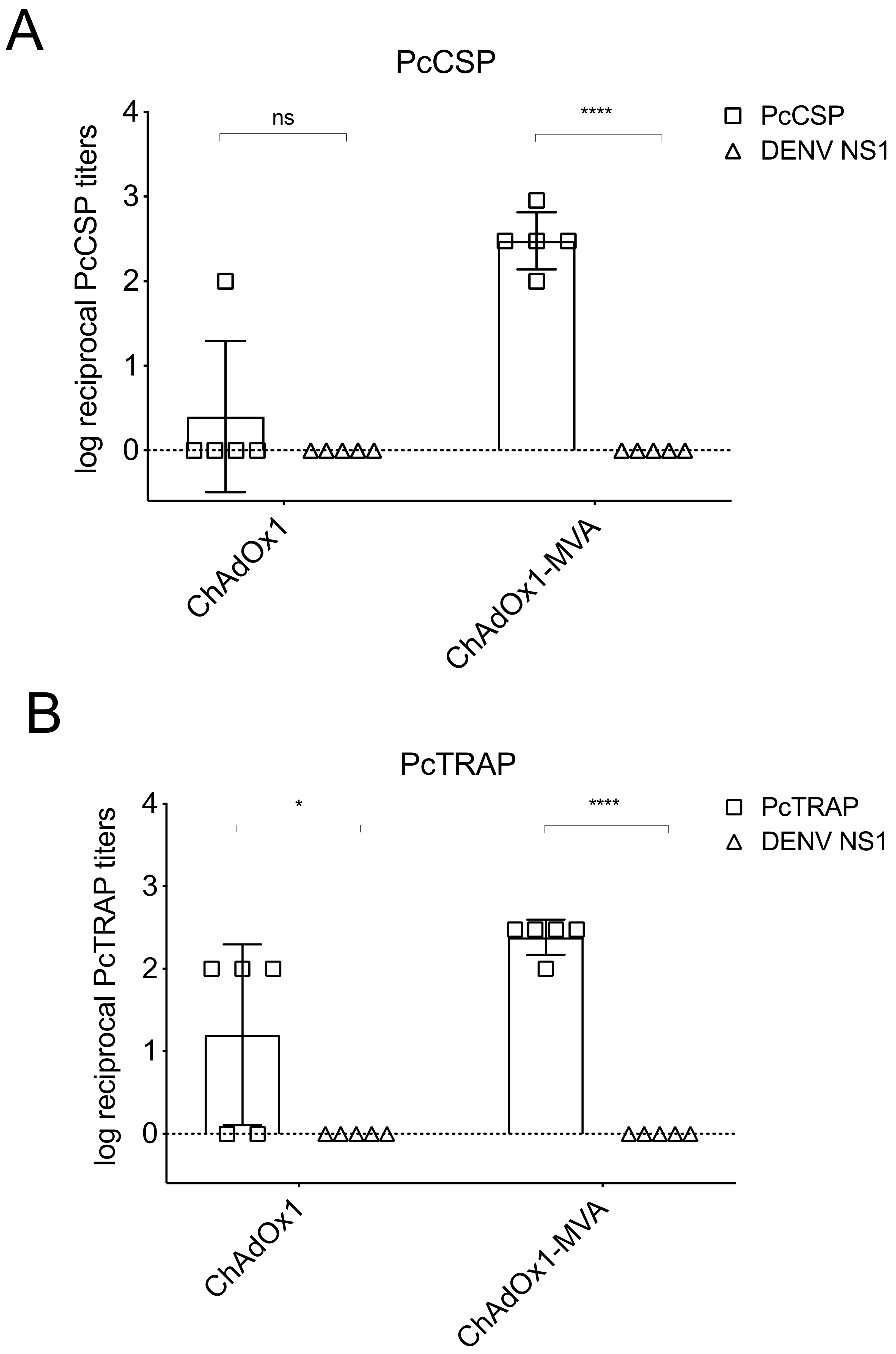
3.3. Pre-Clinical Immune Responses after Vaccination in Mice
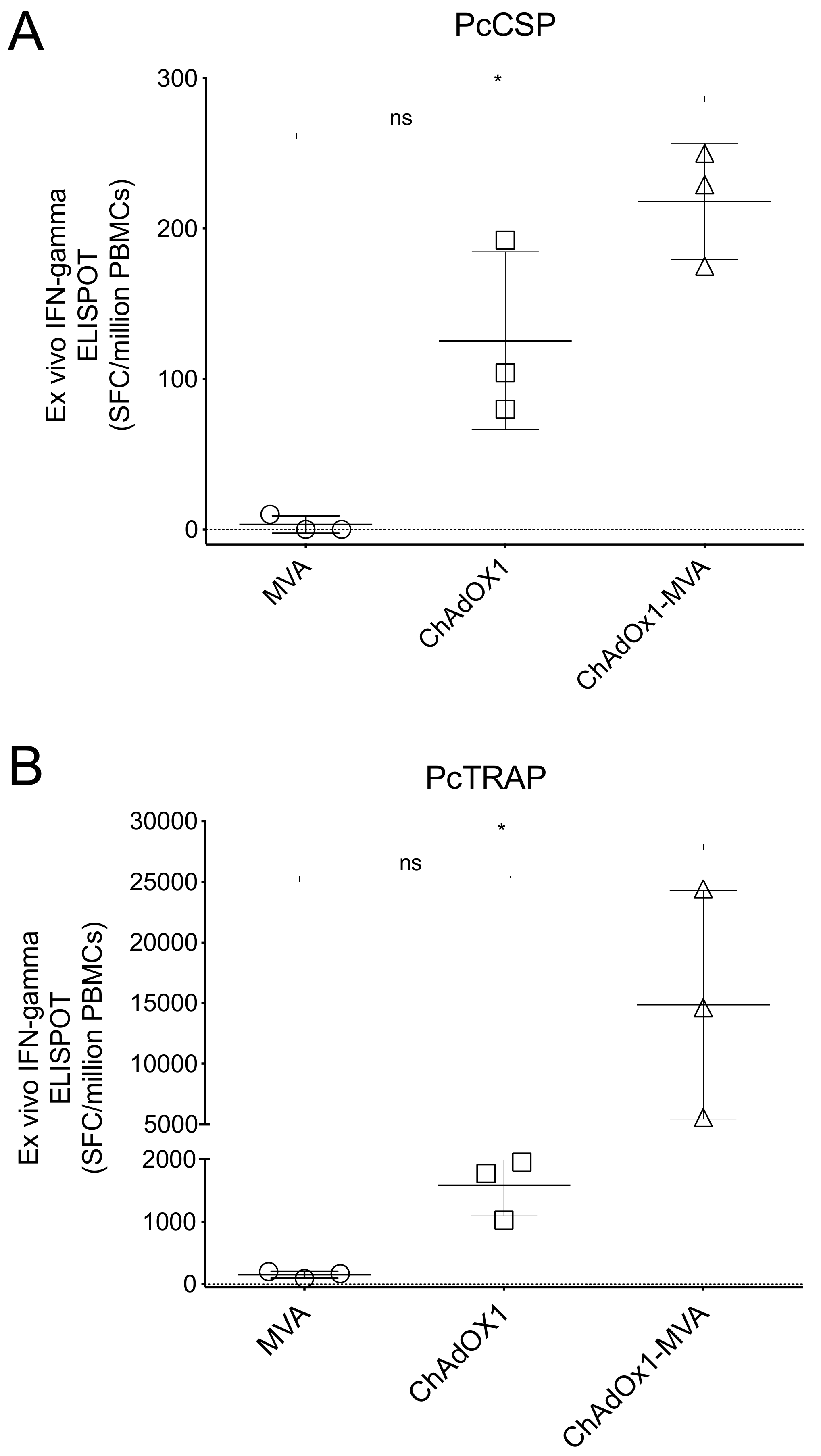
3.4. Humoral and Cellular Immune Responses after Vaccination in Rhesus Macaque Prior to Challenge
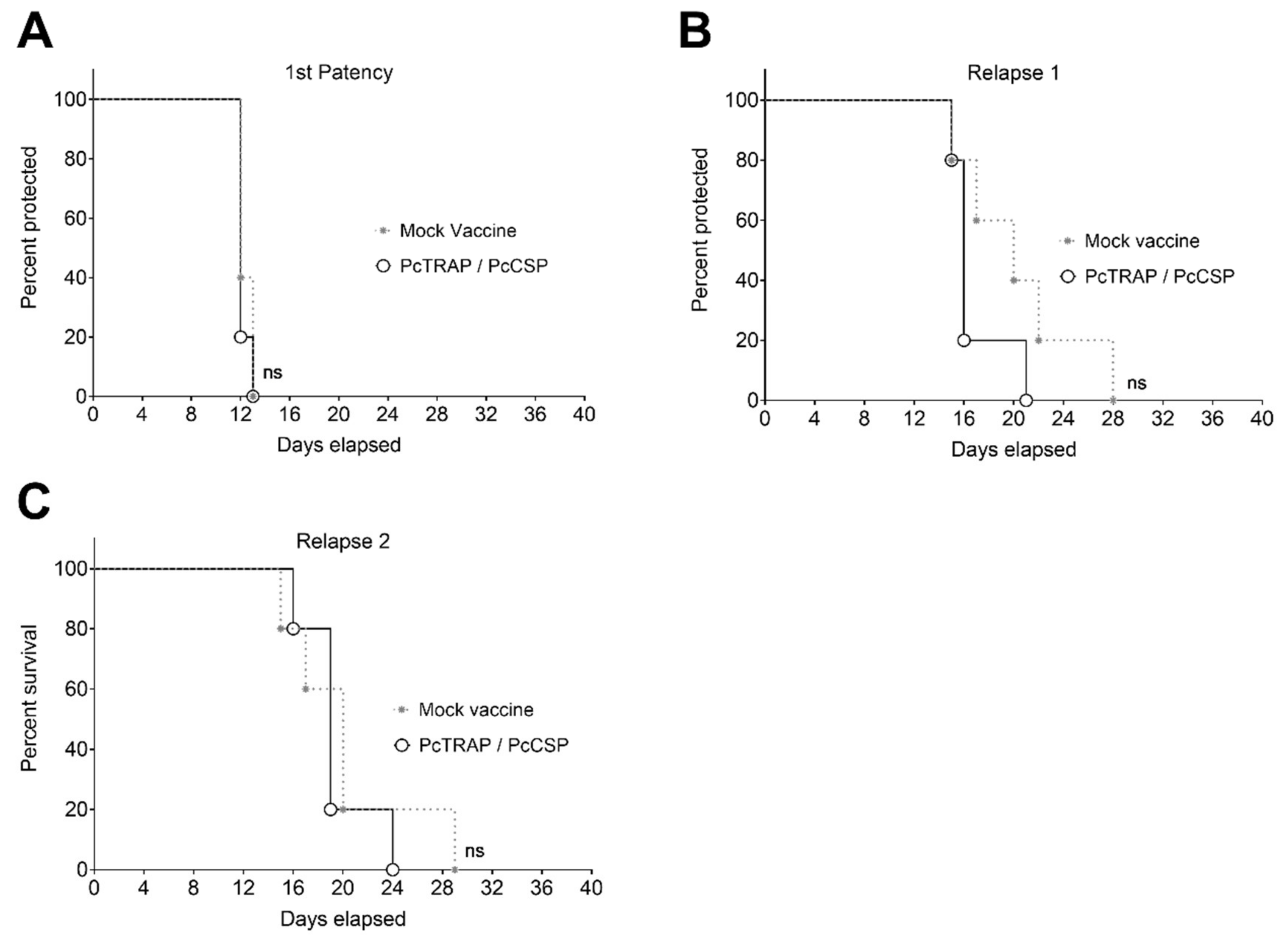
3.5. Challenge of NHPs with P. cynomolgi sporozoites
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Guerra, C.A.; Howes, R.E.; Patil, A.P.; Gething, P.W.; Van Boeckel, T.P.; Temperley, W.H.; Kabaria, C.W.; Tatem, A.J.; Manh, B.H.; Elyazar, I.R.F.; et al. The international limits and population at risk of Plasmodium vivax transmission in 2009. PLoS Negl. Trop. Dis. 2010, 4, e774. [Google Scholar] [CrossRef] [PubMed]
- Price, R.N.; Tjitra, E.; Guerra, C.A.; Yeung, S.; White, N.J.; Anstey, N.M. Vivax malaria: Neglected and not benign. Am. J. Trop. Med. Hyg. 2007, 77, 79–87. [Google Scholar] [CrossRef] [PubMed]
- Markus, M.B. Malaria: Origin of the term “hypnozoite”. J. Hist. Biol. 2011, 44, 781–786. [Google Scholar] [CrossRef] [PubMed]
- White, N.J. Determinants of relapse periodicity in Plasmodium vivax malaria. Malar. J. 2011, 10, 297. [Google Scholar] [CrossRef]
- Luxemburger, C.; van Vugt, M.; Jonathan, S.; McGready, R.; Looareesuwan, S.; White, N.J.; Nosten, F. Treatment of vivax malaria on the western border of Thailand. Trans. R. Soc. Trop. Med. Hyg. 1999, 93, 433–438. [Google Scholar] [CrossRef]
- Robinson, L.J.; Wampfler, R.; Betuela, I.; Karl, S.; White, M.T.; Li Wai Suen, C.S.N.; Hofmann, N.E.; Kinboro, B.; Waltmann, A.; Brewster, J.; et al. Strategies for understanding and reducing the Plasmodium vivax and Plasmodium ovale hypnozoite reservoir in Papua New Guinean children: A randomised placebo-controlled trial and mathematical model. PLoS Med. 2015, 12, e1001891. [Google Scholar] [CrossRef]
- White, M.; Amino, R.; Mueller, I. Theoretical Implications of a Pre-Erythrocytic Plasmodium vivax Vaccine for Preventing Relapses. Trends Parasitol. 2017, 33, 260–263. [Google Scholar] [CrossRef]
- Rishikesh, K.; Saravu, K. Primaquine treatment and relapse in Plasmodium vivax malaria. Pathog. Glob. Health 2016, 110, 1–8. [Google Scholar] [CrossRef]
- Schmidt, L.H. Appraisals of compounds of diverse chemical classes for capacities to cure infections with sporozoites of Plasmodium cynomolgi. Am. J. Trop. Med. Hyg. 1983, 32, 231–257. [Google Scholar] [CrossRef]
- Reyes-Sandoval, A.; Wyllie, D.H.; Bauza, K.; Milicic, A.; Forbes, E.K.; Rollier, C.S.; Hill, A.V.S. CD8+ T effector memory cells protect against liver-stage malaria. J. Immunol. 2011, 187, 1347–1357. [Google Scholar] [CrossRef]
- Vaughan, A.M.; Kappe, S.H.I. Malaria Parasite Liver Infection and Exoerythrocytic Biology. Cold Spring Harb. Perspect. Med. 2017, 7. [Google Scholar] [CrossRef] [PubMed]
- Bauza, K.; Malinauskas, T.; Pfander, C.; Anar, B.; Jones, E.Y.; Billker, O.; Hill, A.V.S.; Reyes-Sandoval, A. Efficacy of a Plasmodium vivax Malaria Vaccine Using ChAd63 and Modified Vaccinia Ankara Expressing Thrombospondin-Related Anonymous Protein as Assessed with Transgenic Plasmodium berghei Parasites. Infect. Immun. 2014, 82, 1277–1286. [Google Scholar] [CrossRef] [PubMed]
- Bauza, K.; Atcheson, E.; Malinauskas, T.; Blagborough, A.M.; Reyes-Sandoval, A. Tailoring a Combination Preerythrocytic Malaria Vaccine. Infect. Immun. 2016, 84, 622–634. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.-Y.; Song, W.-T.; Li, Y.; Chen, W.-J.; Yang, D.; Zhong, G.-C.; Zhou, H.-Z.; Ren, C.-Y.; Yu, H.-T.; Ling, H. Improved expression of secretory and trimeric proteins in mammalian cells via the introduction of a new trimer motif and a mutant of the tPA signal sequence. Appl. Microbiol. Biotechnol. 2011, 91, 731–740. [Google Scholar] [CrossRef]
- Kou, Y.; Xu, Y.; Zhao, Z.; Liu, J.; Wu, Y.; You, Q.; Wang, L.; Gao, F.; Cai, L.; Jiang, C. Tissue plasminogen activator (tPA) signal sequence enhances immunogenicity of MVA-based vaccine against tuberculosis. Immunol. Lett. 2017, 190, 51–57. [Google Scholar] [CrossRef] [PubMed]
- Hancock, G.; Blight, J.; Lopez-Camacho, C.; Kopycinski, J.; Pocock, M.; Byrne, W.; Price, M.J.; Kemlo, P.; Evans, R.I.; Bloss, A.; et al. A multi-genotype therapeutic human papillomavirus vaccine elicits potent T cell responses to conserved regions of early proteins. Sci. Rep. 2019, 9, 18713. [Google Scholar] [CrossRef]
- Aricescu, A.R.; Lu, W.; Jones, E.Y. A time- and cost-efficient system for high-level protein production in mammalian cells. Acta Cryst. D Biol. Cryst. 2006, 62, 1243–1250. [Google Scholar] [CrossRef]
- López-Camacho, C.; Kim, Y.C.; Blight, J.; Lazaro Moreli, M.; Montoya-Diaz, E.; Huiskonen, J.T.; Kümmerer, B.M.; Reyes-Sandoval, A. Assessment of Immunogenicity and Neutralisation Efficacy of Viral-Vectored Vaccines Against Chikungunya Virus. Viruses 2019, 11. [Google Scholar] [CrossRef]
- López-Camacho, C.; Kim, Y.C.; Abbink, P.; Larocca, R.A.; Huiskonen, J.T.; Barouch, D.H.; Reyes-Sandoval, A. Assessment of Immunogenicity and Efficacy of a Zika Vaccine Using Modified Vaccinia Ankara Virus as Carriers. Pathogens 2019, 8, 216. [Google Scholar] [CrossRef]
- Kim, Y.C.; Lopez-Camacho, C.; Nettleship, J.E.; Rahman, N.; Hill, M.L.; Silva-Reyes, L.; Ortiz-Martinez, G.; Figueroa-Aguilar, G.; Mar, M.A.; Vivanco-Cid, H.; et al. Optimization of Zika virus envelope protein production for ELISA and correlation of antibody titers with virus neutralization in Mexican patients from an arbovirus endemic region. Virol. J. 2018, 15, 193. [Google Scholar] [CrossRef]
- Salman, A.M.; Montoya-Díaz, E.; West, H.; Lall, A.; Atcheson, E.; Lopez-Camacho, C.; Ramesar, J.; Bauza, K.; Collins, K.A.; Brod, F.; et al. Rational development of a protective P. vivax vaccine evaluated with transgenic rodent parasite challenge models. Sci. Rep. 2017, 7, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Atcheson, E.; Bauza, K.; Salman, A.M.; Alves, E.; Blight, J.; Viveros-Sandoval, M.E.; Janse, C.J.; Khan, S.M.; Hill, A.V.S.; Reyes-Sandoval, A. Tailoring a Plasmodium vivax Vaccine To Enhance Efficacy through a Combination of a CSP Virus-Like Particle and TRAP Viral Vectors. Infect. Immun. 2018, 86. [Google Scholar] [CrossRef]
- Reyes-Sandoval, A.; Sridhar, S.; Berthoud, T.; Moore, A.C.; Harty, J.T.; Gilbert, S.C.; Gao, G.; Ertl, H.C.J.; Wilson, J.C.; Hill, A.V.S. Single-dose immunogenicity and protective efficacy of simian adenoviral vectors against Plasmodium berghei. Eur. J. Immunol. 2008, 38, 732–741. [Google Scholar] [CrossRef] [PubMed]
- Rampling, T.; Ewer, K.J.; Bowyer, G.; Edwards, N.J.; Wright, D.; Sridhar, S.; Payne, R.; Powlson, J.; Bliss, C.; Venkatraman, N.; et al. Safety and efficacy of novel malaria vaccine regimens of RTS,S/AS01B alone, or with concomitant ChAd63-MVA-vectored vaccines expressing ME-TRAP. NPJ Vaccines 2018, 3, 1–9. [Google Scholar] [CrossRef]
- Dunachie, S.; Hill, A.V.S.; Fletcher, H.A. Profiling the host response to malaria vaccination and malaria challenge. Vaccine 2015, 33, 5316–5320. [Google Scholar] [CrossRef] [PubMed][Green Version]
- De Camargo, T.M.; de Freitas, E.O.; Gimenez, A.M.; Lima, L.C.; de Almeida Caramico, K.; Françoso, K.S.; Bruna-Romero, O.; Andolina, C.; Nosten, F.; Rénia, L.; et al. Prime-boost vaccination with recombinant protein and adenovirus-vector expressing Plasmodium vivax circumsporozoite protein (CSP) partially protects mice against Pb/Pv sporozoite challenge. Sci. Rep. 2018, 8, 1118. [Google Scholar] [CrossRef] [PubMed]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, Y.C.; Dema, B.; Rodriguez-Garcia, R.; López-Camacho, C.; Leoratti, F.M.S.; Lall, A.; Remarque, E.J.; Kocken, C.H.M.; Reyes-Sandoval, A. Evaluation of Chimpanzee Adenovirus and MVA Expressing TRAP and CSP from Plasmodium cynomolgi to Prevent Malaria Relapse in Nonhuman Primates. Vaccines 2020, 8, 363. https://doi.org/10.3390/vaccines8030363
Kim YC, Dema B, Rodriguez-Garcia R, López-Camacho C, Leoratti FMS, Lall A, Remarque EJ, Kocken CHM, Reyes-Sandoval A. Evaluation of Chimpanzee Adenovirus and MVA Expressing TRAP and CSP from Plasmodium cynomolgi to Prevent Malaria Relapse in Nonhuman Primates. Vaccines. 2020; 8(3):363. https://doi.org/10.3390/vaccines8030363
Chicago/Turabian StyleKim, Young Chan, Barbara Dema, Roberto Rodriguez-Garcia, César López-Camacho, Fabiana M. S. Leoratti, Amar Lall, Edmond J. Remarque, Clemens H. M. Kocken, and Arturo Reyes-Sandoval. 2020. "Evaluation of Chimpanzee Adenovirus and MVA Expressing TRAP and CSP from Plasmodium cynomolgi to Prevent Malaria Relapse in Nonhuman Primates" Vaccines 8, no. 3: 363. https://doi.org/10.3390/vaccines8030363
APA StyleKim, Y. C., Dema, B., Rodriguez-Garcia, R., López-Camacho, C., Leoratti, F. M. S., Lall, A., Remarque, E. J., Kocken, C. H. M., & Reyes-Sandoval, A. (2020). Evaluation of Chimpanzee Adenovirus and MVA Expressing TRAP and CSP from Plasmodium cynomolgi to Prevent Malaria Relapse in Nonhuman Primates. Vaccines, 8(3), 363. https://doi.org/10.3390/vaccines8030363




