Duration of Cellular and Humoral Responses after Quadrivalent Human Papillomavirus Vaccination in Healthy Female Adults with or without Prior Type 16 and/or 18 Exposure
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design and Vaccine
2.2. Cellular Immunological Assays
2.3. Multiplexed Enzyme-Linked Immunosorbent Assay (M4ELISA)
2.4. Pseudovirion-Based Neutralization Assay Using Secreted Alkaline Phosphatase (PBNA)
2.5. HPV Detection in Cervical Swab Specimens
2.6. Statistical Analysis
3. Results
3.1. Study Populations
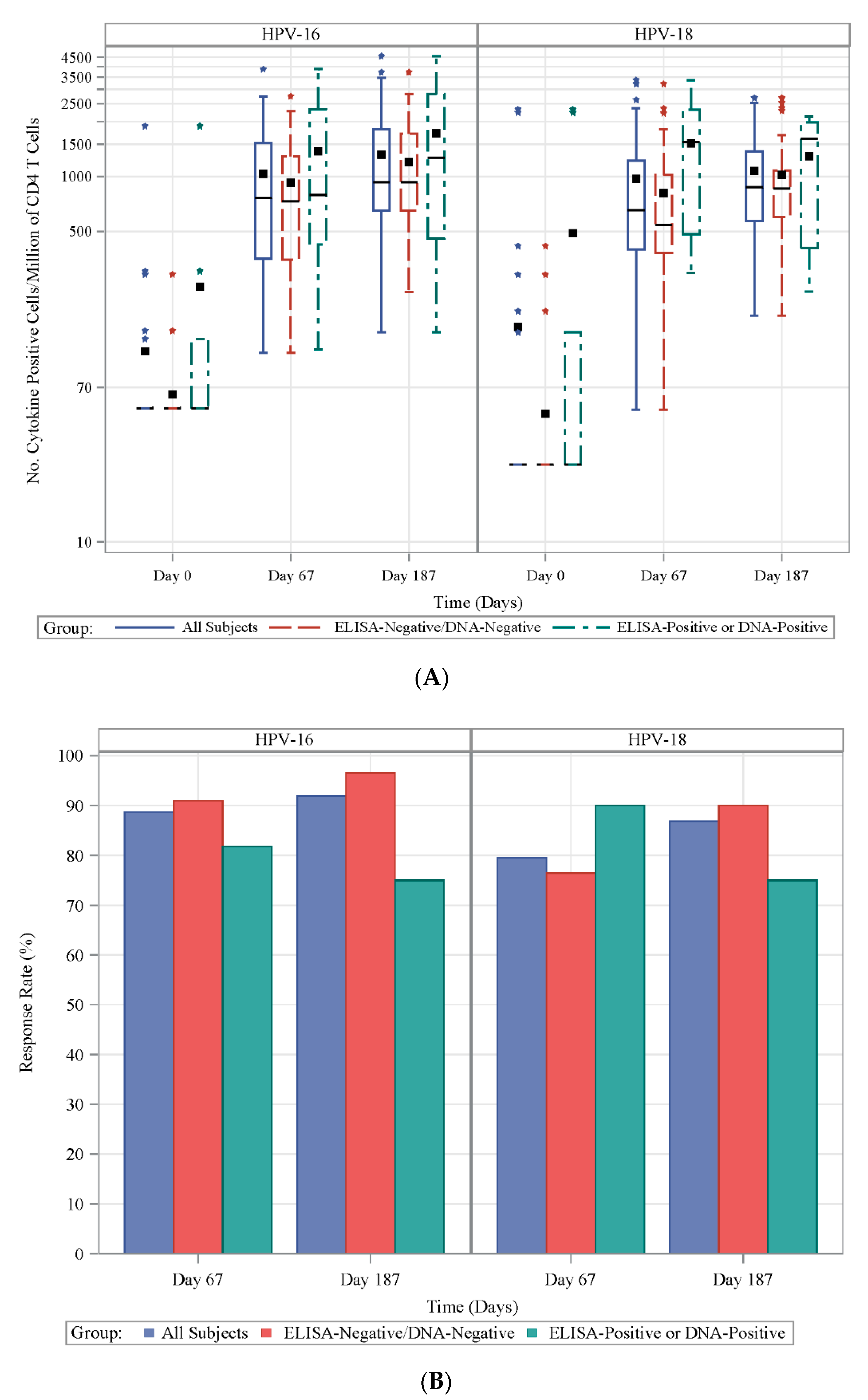
3.2. Frequencies of HPV-Specific CD4+ T Cells
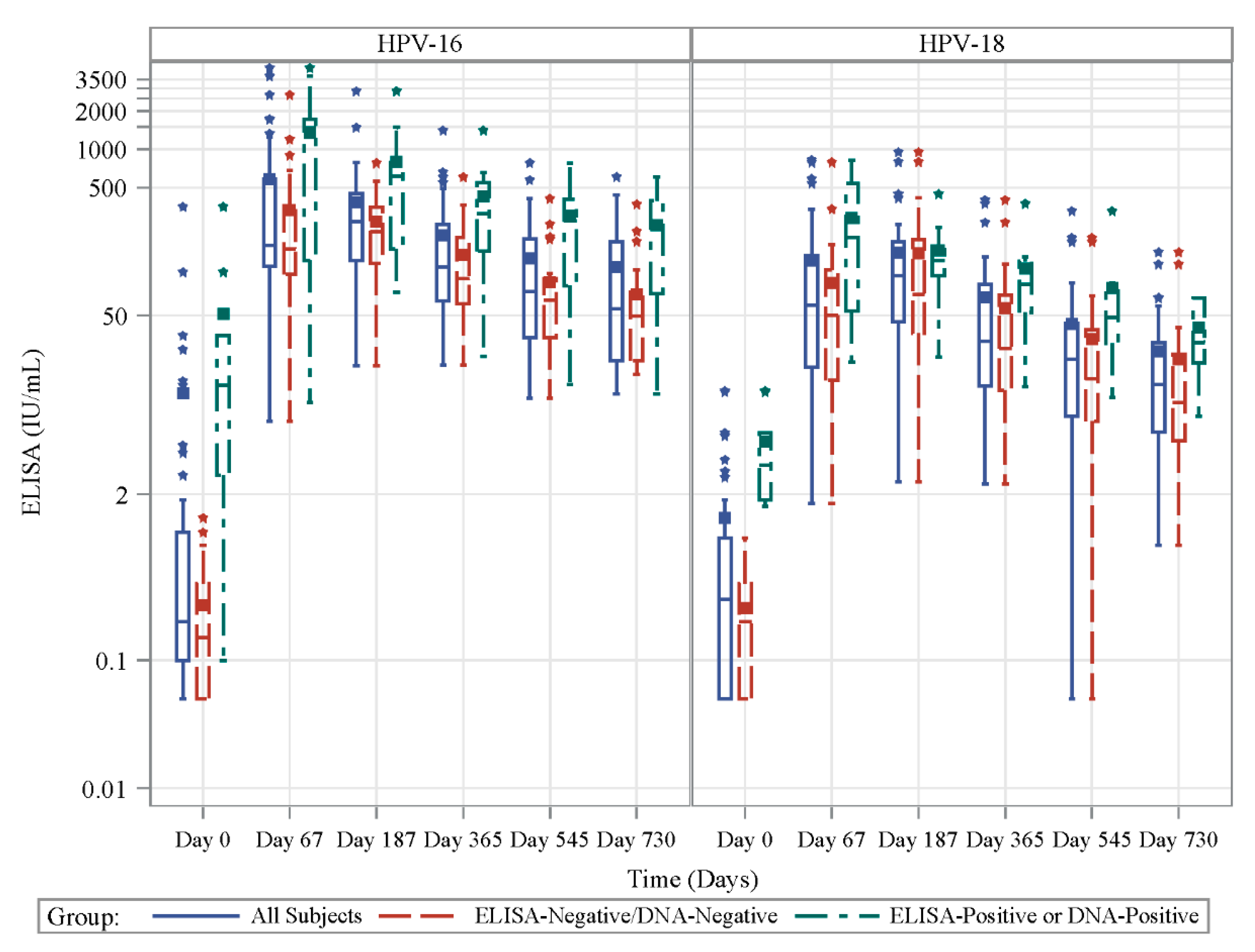
3.3. Antibody Response to Vaccination in Prospective Cohort
3.4. T Lymphocyte Proliferative Capacity
3.5. Correlations between CD4+ T Cell and Ab Responses
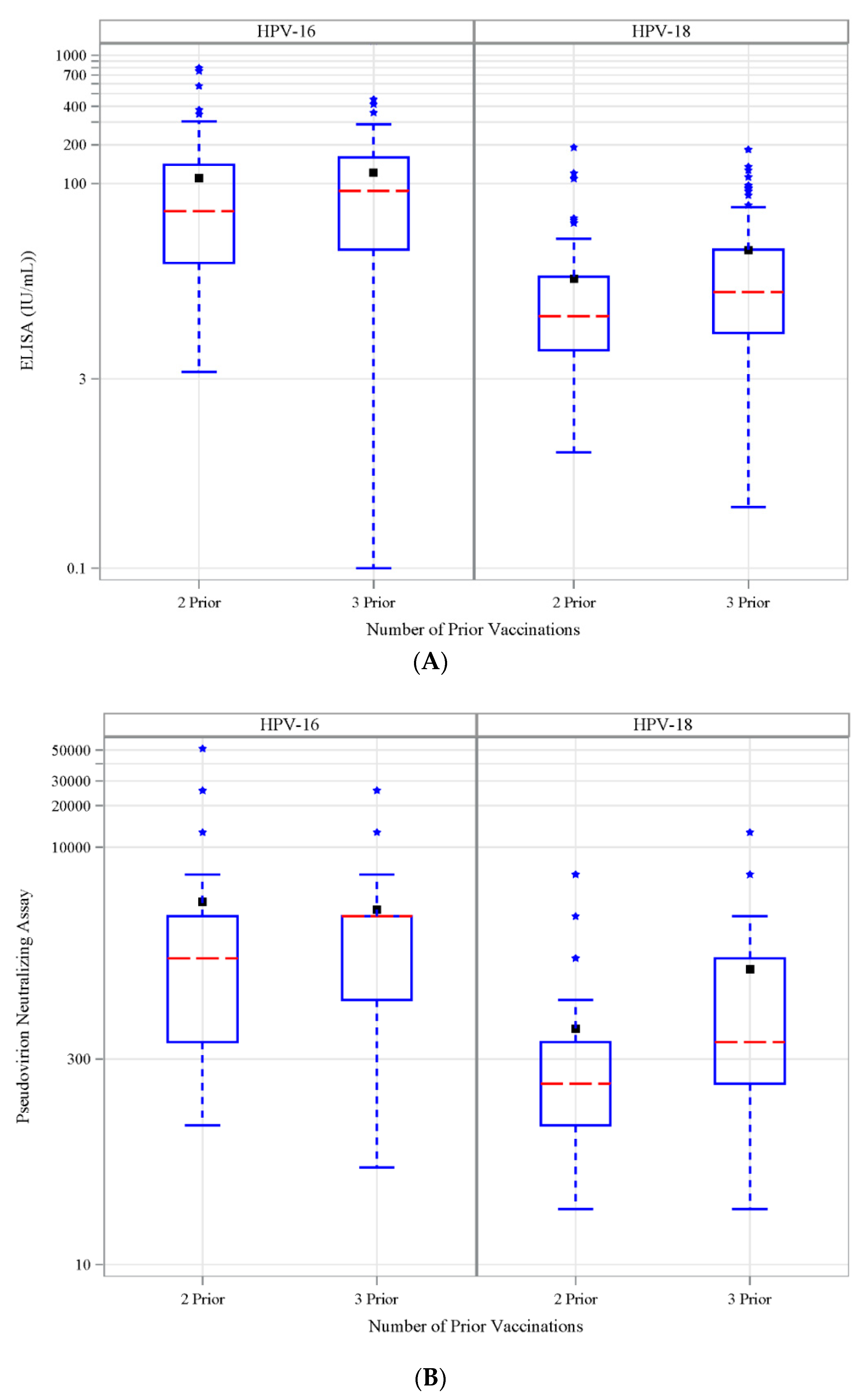
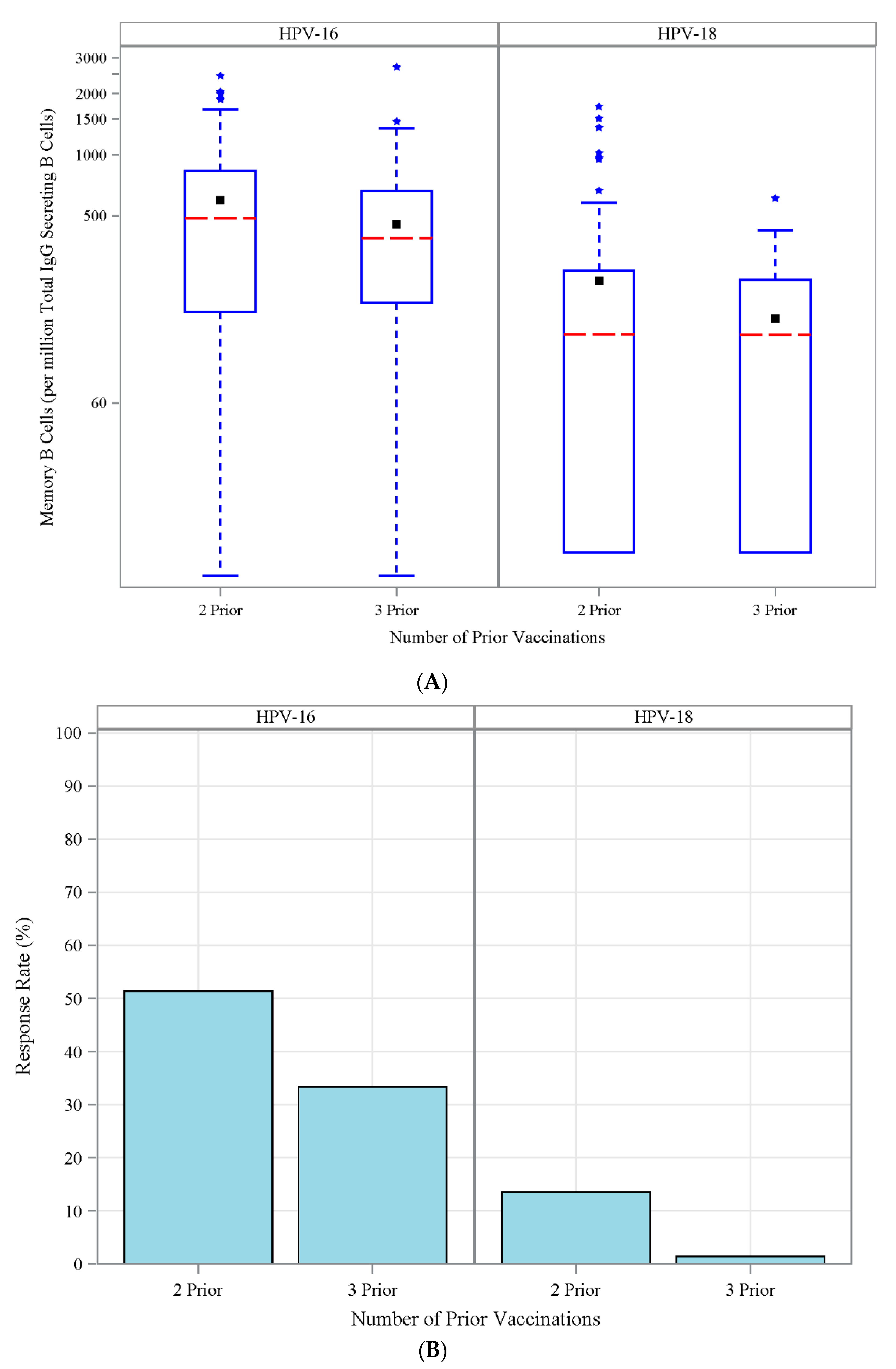
3.6. Antibody Response to Vaccination-Retrospective Cohort
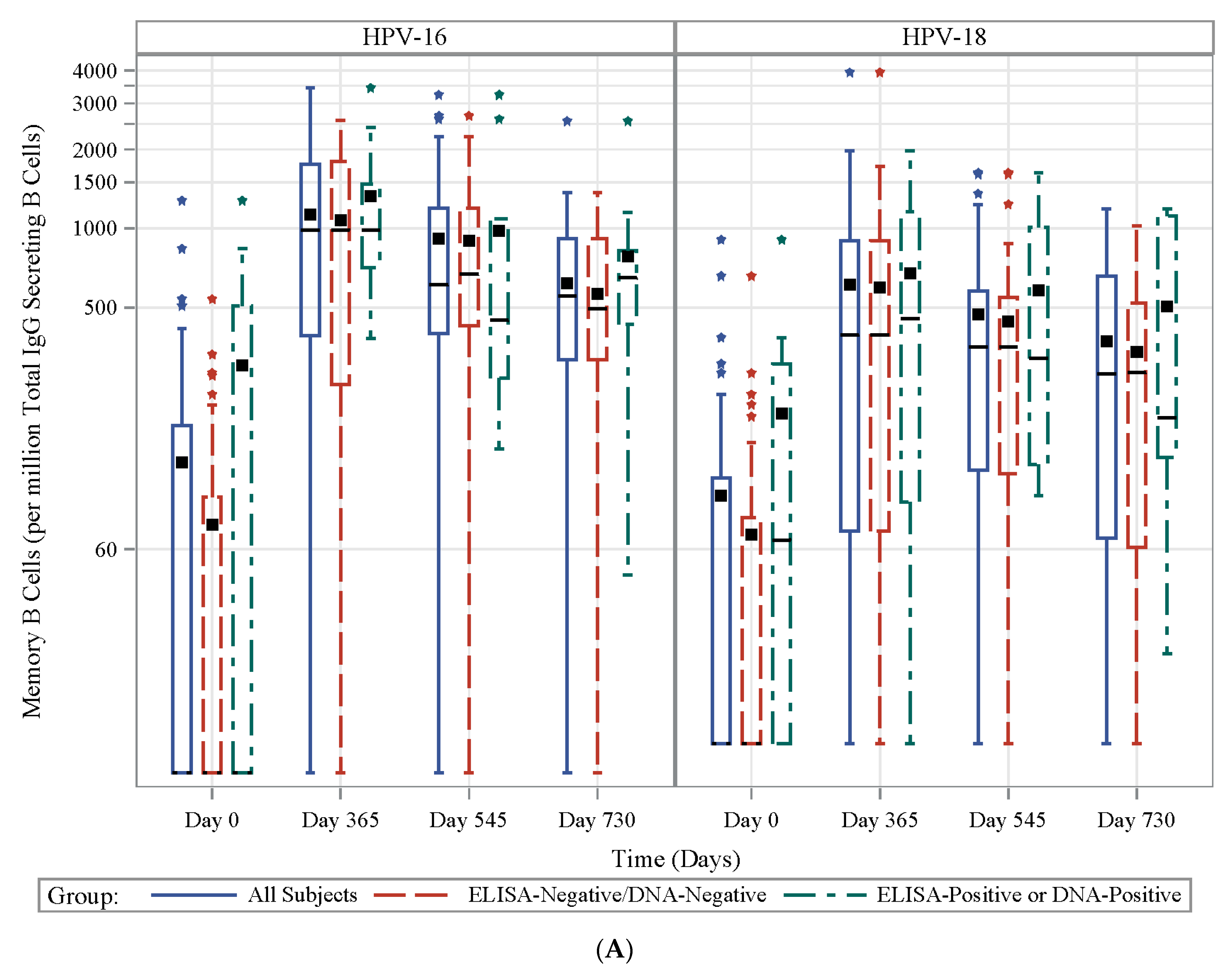
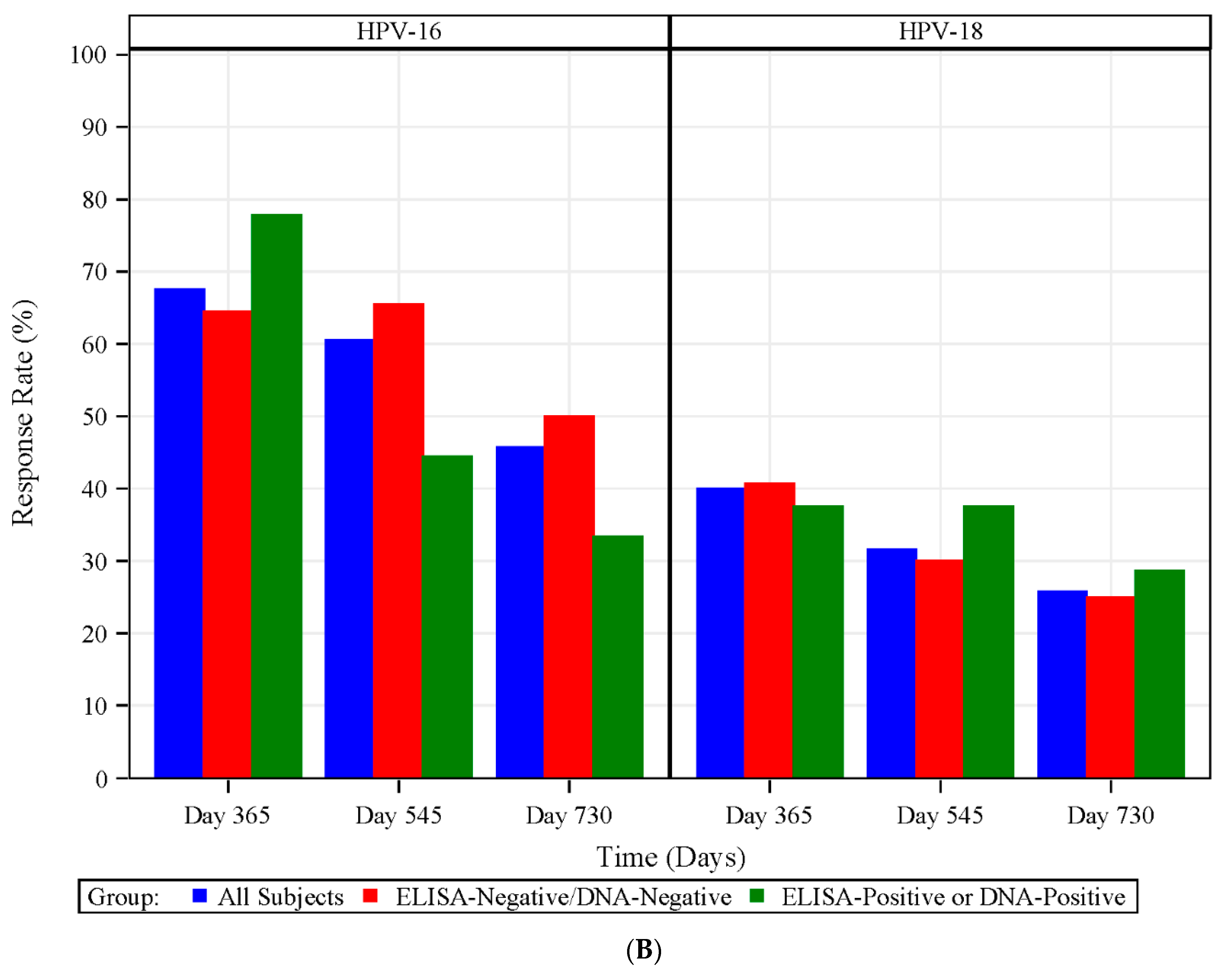
3.7. HPV Type-Specific MBCs
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A
Appendix A.1. Multiplexed Enzyme-Linked Immunosorbent Assay
Appendix A.2. Intracellular Cytokine Assay
Appendix A.3. Memory B-Cell (MBC) ELISpot Assay
Appendix A.4. Carboxyfluorescein Succinimidyl Ester (CFSE) Assay
Appendix A.5. Statistical Analysis
References
- Dillner, J.; Kjaer, S.K.; Wheeler, C.M.; Sigurdsson, K.; Iversen, O.E.; Hernandez-Avila, M.; Perez, G.; Brown, D.R.; Koutsky, L.A.; Future I/II Study Group; et al. Four year efficacy of prophylactic human papillomavirus quadrivalent vaccine against low grade cervical, vulvar, and vaginal intraepithelial neoplasia and anogenital warts: Randomised controlled trial. BMJ 2010, 341, c3493. [Google Scholar] [CrossRef] [PubMed]
- Paovonen, J.; Naud, P.; Salmeron, J.; Wheeler, C.M.; Chow, S.N.; Apter, D.; Kitchener, H.; Castellsague, X.; Teixeira, J.C.; Skinner, S.R.; et al. Efficacy of human papillomavirus (HPV)-16/18 AS04-adjuvanted vaccine against cervical infection and precancer caused by oncogenic HPV types (PATRICIA): Final analysis of a double-blind, randomised study in young women. Lancet 2009, 374, 301–314. [Google Scholar] [CrossRef]
- Naud, P.S.; Roteli-Martins, C.M.; De Carvalho, N.S.; Teixeira, J.C.; de Borba, P.C.; Sanchez, N.; Zahaf, T.; Catteau, G.; Geeraerts, B.; Descamps, D. Sustained efficacy, immunogenicity, and safety of the HPV-16/18 AS04-adjuvanted vaccine Final analysis of a long-term follow-up study up to 9.4 years post-vaccination. Hum. Vaccines Immunother. 2014, 10, 2147–2162. [Google Scholar] [CrossRef] [PubMed]
- Ferris, D.; Samakoses, R.; Block, S.L.; Lazcano-Ponce, E.; Restrepo, J.A.; Reisinger, K.S.; Mehlsen, J.; Chatterjee, A.; Iversen, O.E.; Sings, H.L.; et al. Long-term study of a quadrivalent human papillomavirus vaccine. Pediatrics 2014, 134, E657–E665. [Google Scholar] [CrossRef]
- Kjaer, S.K.; Nygard, M.; Dillner, J.; Marshall, J.B.; Radley, D.; Li, M.; Munk, C.; Hansen, B.T.; Sigurdardottir, L.G.; Hortlund, M.; et al. A 12-year follow-up on the long-term effectiveness of the quadrivalent human papillomavirus vaccine in 4 Nordic countries. Clin. Infect. Dis. 2018, 66, 339–345. [Google Scholar] [CrossRef]
- Nygard, M.; Saah, A.; Munk, C.; Tryggvadottir, L.; Enerly, E.; Hortlund, M.; Sigurdardottir, L.G.; Vuocolo, S.; Kjaer, S.K.; Dillner, J. Evaluation of the long-term anti-human papillomavirus 6 (HPV6), 11, 16, and 18 immune responses generated by the quadrivalent HPV Vaccine. Clin. Vaccine Immunol. 2015, 22, 943–948. [Google Scholar] [CrossRef]
- WHO. Summary of the SAGE April 2014 Meeting. Available online: https://www.who.int/immunization/sage/meetings/2014/april/report_summary_april_2014/en/ (accessed on 4 September 2018).
- Meites, E.; Kempe, A.; Markowitz, L.E. Use of a 2-dose schedule for human papillomavirus vaccination—Updated recommendations of the Advisory Committee on Immunization Practices. MMWR Morb. Mortal. Wkly. Rep. 2016, 65, 1405–1408. [Google Scholar] [CrossRef]
- Spinner, C.; Ding, L.; Bernstein, D.I.; Brown, D.R.; Franco, E.L.; Covert, C.; Kahn, J.A. Human papillomavirus vaccine effectiveness and herd protection in young women. Pediatrics 2019, 143, e20181902. [Google Scholar] [CrossRef]
- Drolet, M.; Bénard, É.; Pérez, N.; Brisson, M.; Ali, H.; Boily, M.-C.; Baldo, V.; Brassard, P.; Brotherton, J.M.L.; Callander, D.; et al. Population-level impact and herd effects following the introduction of human papillomavirus vaccination programmes: Updated systematic review and meta-analysis. Lancet 2019, 394, 497–509. [Google Scholar] [CrossRef]
- Einstein, M.H.; Baron, M.; Levin, M.J.; Chatterjee, A.; Edwards, R.P.; Zepp, F.; Carletti, I.; Dessy, F.J.; Trofa, A.F.; Schuind, A.; et al. Comparison of the immunogenicity and safety of Cervarix and Gardasil human papillomavirus (HPV) cervical cancer vaccines in healthy women aged 18–45 years. Hum. Vaccines 2009, 5, 705–719. [Google Scholar] [CrossRef]
- Olsson, S.E.; Kjaer, S.K.; Sigurdsson, K.; Iversen, O.E.; Hernandez-Avila, M.; Wheeler, C.M.; Perez, G.; Brown, D.R.; Koutsky, L.A.; Tay, E.H.; et al. Evaluation of quadrivalent HPV 6/11/16/18 vaccine efficacy against cervical and anogenital disease in subjects with serological evidence of prior vaccine type HPV infection. Hum. Vaccines 2009, 5, 696–704. [Google Scholar] [CrossRef]
- Panicker, G.; Rajbhandari, I.; Gurbaxani, B.M.; Querec, T.D.; Unger, E.R. Development and evaluation of multiplexed immunoassay for detection of antibodies to HPV vaccine types. J. Immunol. Methods 2015, 417, 107–114. [Google Scholar] [CrossRef]
- Grabowska, K.; Wang, X.; Jacobsson, A.; Dillner, J. Evaluation of cost-precision rations of different strategies for ELISA measurement of serum antibody levels. J. Immunol. Methods 2002, 271, 1–15. [Google Scholar] [CrossRef]
- Buck, C.B.; Thompson, C.D. Production of papillomavirus-based gene transfer vectors. Curr. Protoc. Cell Biol. 2007, 37, 26.1.1–26.1.19. [Google Scholar] [CrossRef] [PubMed]
- Pastrana, D.V.; Buck, C.B.; Pang, Y.-Y.S.; Thompson, C.D.; Castle, P.E.; FitzGerald, P.C.; Krüger Kjaer, S.; Lowy, D.R.; Schiller, J.T. Reactivity of human sera in a sensitive, high-throughput pseudovirus-based papillomavirus neutralization assay for HPV16 and HPV18. Virology 2004, 321, 205–216. [Google Scholar] [CrossRef]
- World Health Organization (WHO). Human Papillomavirus Laboratory Manual; World Health Organization: Geneva, Switzerland, 2010. [Google Scholar]
- Steinau, M.; Reddy, D.; Sumbry, A.; Reznik, D.; Gunthel, C.J.; del Rio, C.; Lennox, J.L.; Unger, E.R.; Nguyen, M.L.T. Oral sampling and human papillomavirus genotyping in HIV-infected patients: Oral sampling and HPV typing. J. Oral Pathol. Med. 2012, 41, 288–291. [Google Scholar] [CrossRef]
- Ferris, D.G.; Samakoses, R.; Block, S.L.; Lazcano-Ponce, E.; Restrepo, J.A.; Mehlsen, J.; Chatterjee, A.; Iversen, O.E.; Joshi, A.; Chu, J.L.; et al. 4-Valent human papillomavirus (4vHPV) vaccine in preadolescents and adolescents after 10 years. Pediatrics 2017, 140, e20163947. [Google Scholar] [CrossRef] [PubMed]
- Villa, L.L.; Ault, K.A.; Giuliano, A.R.; Costa, R.L.; Petta, C.A.; Andrade, R.P.; Brown, D.R.; Ferenczy, A.; Harper, D.M.; Koutsky, L.A.; et al. Immunologic responses following administration of a vaccine targeting human papillomavirus Types 6, 11, 16, and 18. Vaccine 2006, 24, 5571–5583. [Google Scholar] [CrossRef]
- Toh, Z.Q.; Russell, F.M.; Reyburn, R.; Fong, J.; Tuivaga, E.; Ratu, T.; Nguyen, C.D.; Devi, R.; Kama, M.; Matanitobua, S.; et al. Sustained antibody responses 6 years following 1, 2, or 3 doses of quadrivalent human papillomavirus (HPV) vaccine in adolescent Fijian girls, and subsequent responses to a single dose of bivalent HPV vaccine: A prospective cohort study. Clin. Infect. Dis. 2017, 64, 852–859. [Google Scholar] [CrossRef]
- Olsson, S.-E.; Villa, L.L.; Costa, R.L.R.; Petta, C.A.; Andrade, R.P.; Malm, C.; Iversen, O.-E.; Høye, J.; Steinwall, M.; Riis-Johannessen, G.; et al. Induction of immune memory following administration of a prophylactic quadrivalent human papillomavirus (HPV) types 6/11/16/18 L1 virus-like particle (VLP) vaccine. Vaccine 2007, 25, 4931–4939. [Google Scholar] [CrossRef]
- Scherer, E.M.; Smith, R.A.; Gallego, D.F.; Carter, J.J.; Wipf, G.C.; Hoyos, M.; Stern, M.; Thurston, T.; Trinklein, N.D.; Wald, A.; et al. A single human papillomavirus vaccine dose improves B cell memory in previously infected subjects. EBioMedicine 2016, 10, 55–64. [Google Scholar] [CrossRef]
- Kim, D.K.; Hunter, P. Advisory committee on immunization practices recommended immunization schedule for adults aged 19 years or older—United States, 2019. MMWR Morb. Mortal. Wkly. Rep. 2019, 68, 115–118. [Google Scholar] [CrossRef]
- Einstein, M.H.; Levin, M.J.; Chatterjee, A.; Chakhtoura, N.; Takacs, P.; Catteau, G.; Dessy, F.J.; Moris, P.; Lin, L.; Struyf, F.; et al. Comparative humoral and cellular immunogenicity and safety of human papillomavirus (HPV)-16/18 AS04-adjuvanted vaccine and HPV-6/11/16/18 vaccine in healthy women aged 18–45 years: Follow-up through month 48 in a phase III randomized study. Hum. Vaccines Immunother. 2014, 10, 3455–3465. [Google Scholar] [CrossRef]
- Herrin, D.M.; Coates, E.E.; Costner, P.J.; Kemp, T.J.; Nason, M.C.; Saharia, K.K.; Pan, Y.; Sarwar, U.N.; Holman, L.; Yamshchikov, G.; et al. Comparison of adaptive and innate immune responses induced by licensed vaccines for human papillomavirus. Hum. Vaccines Immunother. 2014, 10, 3446–3454. [Google Scholar] [CrossRef]
- Matsui, K.; Adelsberger, J.W.; Kemp, T.J.; Baseler, M.W.; Ledgerwood, J.E.; Pinto, L.A. Circulating CXCR5(+)CD4(+) T follicular-like helper cell and memory B cell responses to human papillomavirus vaccines. PLoS ONE 2015, 10, e0137195. [Google Scholar] [CrossRef]
- Pinto, L.A.; Edwards, J.; Castle, P.E.; Harro, C.D.; Lowy, D.R.; Schiller, J.T.; Wallace, D.; Kopp, W.; Adelsberger, J.W.; Baseler, M.W.; et al. Cellular immune responses to human papillomavirus (HPV)-16 L1 in healthy volunteers immunized with recombinant HPV-16 L1 Virus-Like Particles. J. Infect. Dis. 2003, 188, 327–338. [Google Scholar] [CrossRef]
- Brotherton, J.M.; Budd, A.; Rompotis, C.; Bartlett, N.; Malloy, M.J.; Andersen, R.L.; Coulter, K.A.; Couvee, P.W.; Steel, N.; Ward, G.H.; et al. Is one dose of human papillomavirus vaccine as effective as three? A national cohort analysis. Papillomavirus Res. 2019, 8, 100177. [Google Scholar] [CrossRef]
- Lai, L.; Davey, R.; Beck, A.; Xu, Y.; Suffredini, A.F.; Palmore, T.; Kabbani, S.; Rogers, S.; Kobinger, G.; Alimonti, J.; et al. Emergency postexposure vaccination with vesicular stomatitis virus-vectored Ebola vaccine after needlestick. JAMA 2015, 313, 1249. [Google Scholar] [CrossRef]
- Crotty, S.; Felgner, P.; Davies, H.; Glidewell, J.; Villarreal, L.; Ahmed, R. Cutting edge: Long-term B cell memory in humans after smallpox vaccination. J. Immunol. 2003, 171, 4969–4973. [Google Scholar] [CrossRef]
- Lyons, A.B. Analysing cell division in vivo and in vitro using flow cytometric measurement of CFSE dye dilution. J. Immunol. Methods 2000, 243, 147–154. [Google Scholar] [CrossRef]
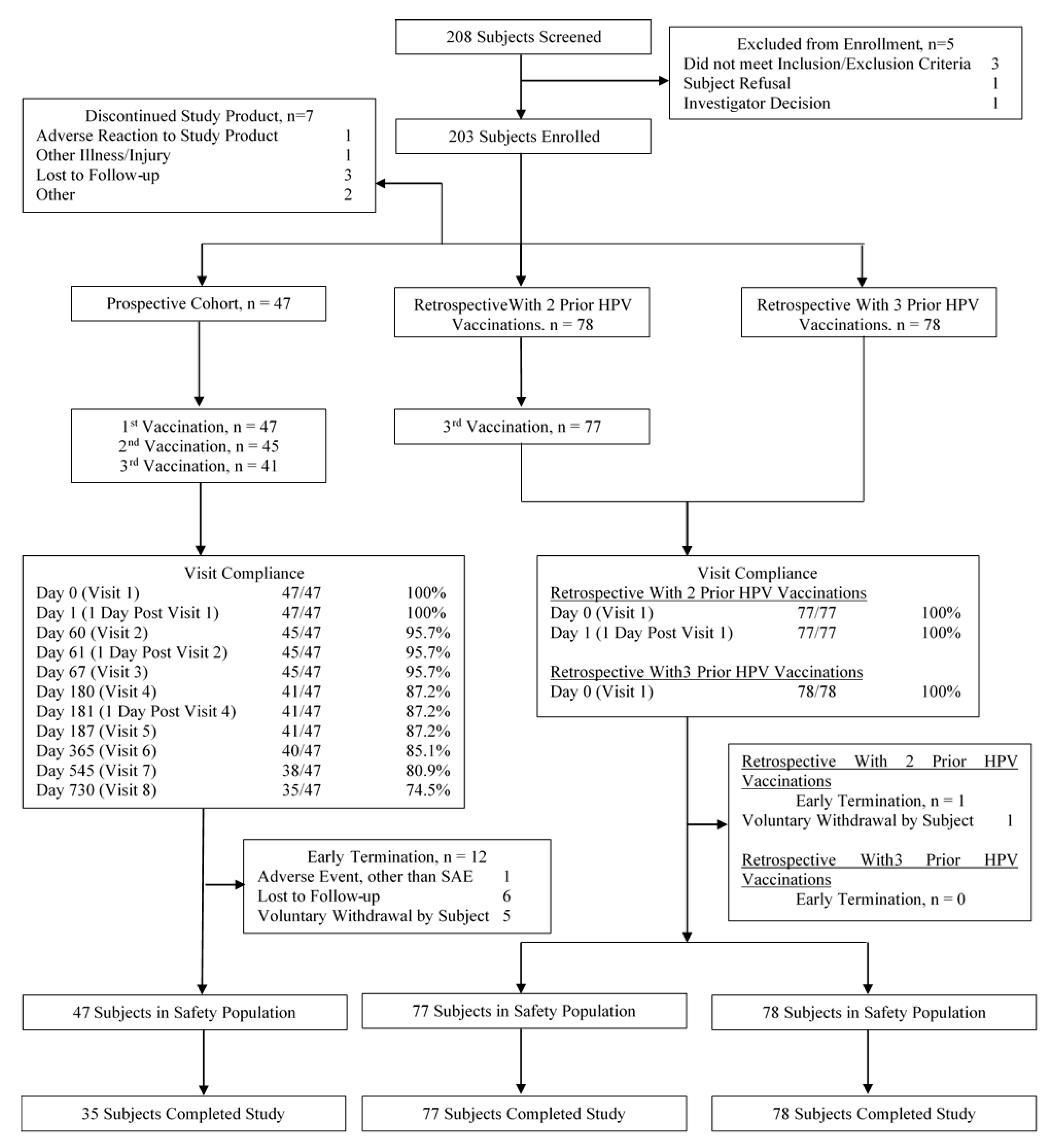
| Characteristic Measured | Prospective (n = 47) | Retrospective with 2 Prior HPV Vaccinations (n = 78) | Retrospective with 3 Prior HPV Vaccinations (n = 78) | All Cohorts (n = 203) |
|---|---|---|---|---|
| Gender, n (% 1) | - | - | - | - |
| Female | 47 (100) | 78 (100) | 78 (100) | 203 (100) |
| Ethnicity, n (% 1) | - | - | - | - |
| Non-Hispanic or Non-Latino | 47 (100) | 72 (92) | 75 (96) | 194 (96) |
| Hispanic or Latino | - | 6 (8) | 3 (4) | 9 (4) |
| Race-n (% 1) | - | - | - | - |
| American Indian/Alaskan Native | 1 (2) | - | - | 1 (0) |
| Asian | 7 (15) | 17 (22) | 3 (4) | 27 (13) |
| Black or African American | 14 (30) | 16 (21) | 8 (10) | 38 (19) |
| White | 24 (51) | 44 (56) | 64 (82) | 132 (65) |
| Multi-Racial | - | - | 3 (4) | 3 (1) |
| Other/Unknown | 1 (2) | 1 (1) | - | 2 (1) |
| Age (years) | - | - | - | - |
| Mean (SD) | 24 (2) | 25 (3) | 25 (3) | 25 (3) |
| Median | 24 | 25 | 24 | 25 |
| Min, Max | 18, 26 | 19, 30 | 18, 30 | 18, 30 |
| Baseline HPV exposure | - | - | ||
| ELISA-negative and DNA-negative | - | |||
| HPV-16 | 36 (77) | |||
| HPV-18 | 37 (79) | |||
| ELISA-Positive or DNA-Positive | - | |||
| HPV-16 | 11 (23) | |||
| HPV-18 | 10 (21) | |||
| Timepoint | HPV-16 | HPV-18 | ||||
|---|---|---|---|---|---|---|
| Days Postvaccination | ELISA− and PCR− | ELISA+ Or PCR+ | p-Value | ELISA− and PCR− | ELISA+ or PCR+ | p-Value |
| Day 0 | 0 | 10 | <0.0001 | 0 | 4 | <0.0001 |
| Day 67 | 165 | 498 | 0.0330 | 39 | 165 | 0.0060 |
| Day 187 | 208 | 442 | 0.0350 | 74 | 121 | 0.3080 |
| Day 365 | 111 | 243 | 0.0290 | 28 | 79 | 0.0340 |
| Day 545 | 65 | 173 | 0.0120 | 14 | 51 | 0.0230 |
| Day 730 | 51 | 143 | 0.0120 | 11 | 32 | 0.0370 |
| Timepoint | Antibody Titer by Study Time Point | ||||||
|---|---|---|---|---|---|---|---|
| Type | Cytokine Study Time Point | Statistics | Day 67 (Dose 2 + 7 Days) | Day 187 (Dose 3 + 7 Days) | Day 365 | Day 545 | Day 730 |
| HPV-16 | Day 67 (Day 7 postdose 2) | Number of Subjects | 44 | 40 | 39 | 37 | 34 |
| Correlation Statistic 1 | 0.366 | 0.568 | 0.529 | 0.470 | 0.479 | ||
| p-value 2 | 0.0146 | 0.0001 | 0.0005 | 0.0033 | 0.0041 | ||
| Day 187 (Day 7 postdose 3) | Number of Subjects | 37 | 37 | 36 | 34 | 32 | |
| Correlation Statistic 1 | 0.378 | 0.525 | 0.530 | 0.379 | 0.400 | ||
| p-value 2 | 0.0211 | 0.0008 | 0.0009 | 0.0269 | 0.0232 | ||
| HPV-18 | Day 67 (Day 7 postdose 2) | Number of Subjects | 44 | 40 | 39 | 37 | 34 |
| Correlation Statistic 1 | 0.416 | 0.334 | 0.415 | 0.376 | 0.275 | ||
| p-value 2 | 0.0050 | 0.0352 | 0.0087 | 0.0217 | 0.1150 | ||
| Day 187 (Day 7 postdose 3) | Number of Subjects | 38 | 38 | 37 | 35 | 33 | |
| Correlation Statistic 1 | 0.400 | 0.383 | 0.311 | 0.287 | 0.210 | ||
| p-value 2 | 0.0128 | 0.0175 | 0.0607 | 0.0946 | 0.2395 | ||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lai, L.; Ault, K.; Rouphael, N.; Beck, A.; Domjahn, B.; Xu, Y.; Anderson, E.J.; Cheng, A.; Nakamura, A.; Hoagland, R.J.; et al. Duration of Cellular and Humoral Responses after Quadrivalent Human Papillomavirus Vaccination in Healthy Female Adults with or without Prior Type 16 and/or 18 Exposure. Vaccines 2020, 8, 348. https://doi.org/10.3390/vaccines8030348
Lai L, Ault K, Rouphael N, Beck A, Domjahn B, Xu Y, Anderson EJ, Cheng A, Nakamura A, Hoagland RJ, et al. Duration of Cellular and Humoral Responses after Quadrivalent Human Papillomavirus Vaccination in Healthy Female Adults with or without Prior Type 16 and/or 18 Exposure. Vaccines. 2020; 8(3):348. https://doi.org/10.3390/vaccines8030348
Chicago/Turabian StyleLai, Lilin, Kevin Ault, Nadine Rouphael, Allison Beck, Briyana Domjahn, Yongxian Xu, Evan J. Anderson, Andrew Cheng, Aya Nakamura, Rebecca J. Hoagland, and et al. 2020. "Duration of Cellular and Humoral Responses after Quadrivalent Human Papillomavirus Vaccination in Healthy Female Adults with or without Prior Type 16 and/or 18 Exposure" Vaccines 8, no. 3: 348. https://doi.org/10.3390/vaccines8030348
APA StyleLai, L., Ault, K., Rouphael, N., Beck, A., Domjahn, B., Xu, Y., Anderson, E. J., Cheng, A., Nakamura, A., Hoagland, R. J., Kelley, C., Edupuganti, S., Mask, K., Nesin, M., Unger, E. R., Panicker, G., David, H., & Mulligan, M. J. (2020). Duration of Cellular and Humoral Responses after Quadrivalent Human Papillomavirus Vaccination in Healthy Female Adults with or without Prior Type 16 and/or 18 Exposure. Vaccines, 8(3), 348. https://doi.org/10.3390/vaccines8030348





